Characteristics of Microbial Factors of Healthcare-Associated Infections Including Multidrug-Resistant Pathogens and Antibiotic Consumption at the University Intensive Care Unit in Poland in the Years 2011–2018
Abstract
1. Introduction
2. Material and Methods
2.1. Data Collection
2.2. Microbiological Diagnosis of Infections
2.3. Data Analysis
2.4. Ethical Approval
3. Findings
3.1. The Most Prevalent Microorganisms Responsible for HAIs
3.2. Changes in the Prevalence of “Alert Pathogens” between 2011 and 2018
3.3. ”Alert Pathogens” Antibiogram
3.4. Selected Consumption of Antibiotics among ICU Patients
4. Discussion
5. Conclusions
- The most frequently isolated group of microorganisms in patients with HAIs, regardless of the clinical form of the infection, was Gram-negative bacilli (71.4%).
- The total number of strains responsible for VAP was the highest, being slightly higher (54.1%) than for UTI and BSI together.
- The variation among etiological factors of different clinical forms of HAIs was proven. Among the pathogens that are responsible for VAP, Gram-negative MDR A. baumannii and K. pneumoniae ESBL(+) were dominant. In the case of UTI, A. baumannii MDR and Candida spp. were most common, whereas in CLA-BSI, MRCNS and K. pneumoniae were most frequently isolated.
- Alert pathogens were found in about half of patients with HAIs, whereas MDR GNB were found in one-third and two-thirds of patients in 2011 and 2018, respectively.
- The analysis demonstrated an almost twofold increase in the isolation of “alert pathogens” from HAIs during the observed period
- A 10-fold increase in resistance of A. baumannii and a 100% sensitivity of K. pneumoniae to carbapenems was found.
- An increase in the consumption of antibiotics belonging to the group of carbapenems as well as a slight decrease in the consumption of aminoglycosides and the fourfold decrease in the consumption of fluoroquinolones were proven.
- The data regarding the microbiological profile of the unit and percentage of resistance allow for a more effective selection of the optimal therapy and the development of the antibiotic policy to avoid further increase in resistance among microorganisms from the hospital environment.
- The strengthening of microbiological diagnostics and monitoring of antibiotic consumption facilitates the establishment of the antibiotic policy and the development of procedures to protect against the spread of resistance mechanisms transmitted by microorganisms and, consequently, the increase in infections caused by alert pathogens.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Vincent, J.-L.; Bihari, D.J.; Suter, P.M.; Bruining, H.A.; White, J.; Nicolas-Chanoin, M.H.; Wolff, M.; Spencer, R.C.; Hemmer, M. The prevalence of nosocomial infection in intensive care units in Europe. Results of the European Prevalence of Infection in Intensive Care (EPIC) Study. EPIC International Advisory Committee. JAMA 1995, 274, 639–644. [Google Scholar] [CrossRef]
- Vincent, J.-L.; Rello, J.; Marshall, J.; Silva, E.; Anzueto, A.; Martin-Loeches, I.; Moreno, R.; Lipman, J.; Gomersall, C.D.; Sakr, Y.; et al. International study of the prevalence and outcomes of infection in intensive care units. JAMA 2009, 302, 2323. [Google Scholar] [CrossRef]
- Dudeck, M.A.; Weiner, L.M.; Allen-Bridson, K.; Malpiedi, P.J.; Peterson, K.D.; Pollock, D.A.; Sievert, D.M.; Edwards, J.R. National Healthcare Safety Network (NHSN) report, data summary for 2012, Device-associated module. Am. J. Infect. Control 2013, 41, 1148–1166. [Google Scholar] [CrossRef]
- Kübler, A.; Duszynska, W.; Rosenthal, V.D.; Fleischer, M.; Kaiser, T.; Szewczyk, E.; Barteczko-Grajek, B. Device-associated infection rates and extra length of stay in an intensive care unit of a university hospital in Wroclaw, Poland: International Nosocomial Infection Control Consortium’s (INICC) findings. J. Crit. Care 2012, 27, 105.e5–105.e10. [Google Scholar] [CrossRef] [PubMed]
- Vincent, J.-L.; Sakr, Y.; Singer, M.; Martin-Loeches, I.; Machado, F.R.; Marshall, J.C.; Finfer, S.; Pelosi, P.; Brazzi, L.; Aditianingsih, D.; et al. Prevalence and Outcomes of Infection Among Patients in Intensive Care Units in 2017. JAMA 2020, 24, 1478–1487. [Google Scholar] [CrossRef]
- Kubler, A.; Adamik, B.; Durek, G.; Mayzner-Zawadzka, E.; Gaszynski, W.; Karpel, E.; Duszynska, W. Results of the severe sepsis registry in intensive care units I Poland from 2003–2009. Anestezjol. Intensywna Ter. 2015, 47, 7–13. [Google Scholar] [CrossRef]
- Boucher, H.W.; Talbot, G.H.; Bradley, J.S.; Edwards, J.E.; Gilbert, D.; Rice, L.B.; Scheld, M.; Spellberg, B.; Bartlett, J. Bad bugs, no drugs: No ESKAPE! An update from the Infectious Diseases Society of America. Clin. Infect. Dis. 2009, 48. [Google Scholar] [CrossRef] [PubMed]
- Brusselaers, N.; Vogelaers, D.; Blot, S. The rising problem of antimicrobial resistance in the intensive care unit. Ann. Intensiv. Care. 2011, 1, 47. [Google Scholar] [CrossRef]
- Lob, S.H.; Hoban, D.J.; Sahm, D.F.; Badal, R.E. Regional differences and trends in antimicrobial susceptibility of Acinetobacter baumannii. Int. J. Antimicrob. Agents. 2016, 47, 317–323. [Google Scholar] [CrossRef]
- Duszynska, W.; Litwin, A.; Rojek, S.; Szczęsny, A.; Ciasullo, A.; Goździk, W. Analysis of Acinetobacter baumannii hospital infections in patients treated at the intensive care unit of the University Hospital, Wroclaw, Poland: A 6-year, single-center, retrospective study. Infect. Drug Resist. 2018, 11, 629–635. [Google Scholar] [CrossRef]
- Weiner, L.M.; Webb, A.K.; Limbago, B.; Dudeck, M.A.; Patel, J.; Kallen, A.J.; Edwards, J.R.; Sievert, D.M. Antimicrobial-resistant pathogens associated with healthcare-associated infections: Summary of data reported to the national healthcare safety network at the Centers for Disease Control and Prevention, 2011–2014. Infect. Control Hosp. Epidemiol. 2016, 37, 1288–1301. [Google Scholar] [CrossRef] [PubMed]
- Despotovic, A.; Milosevic, B.; Milosevic, I.; Mitrovic, N.; Cirkovic, A.; Jovanovic, S.; Stevanovic, G. Hospital-acquired infections in the adult intensive care unit-Epidemiology, antimicrobial resistance patterns, and risk factors for acquisition and mortality. Am. J. Infect. Control 2020, 48, 1211–1215. [Google Scholar] [CrossRef] [PubMed]
- Talaat, M.; El-Shokry, M.; El-Kholy, J.; Ismail, G.; Kotb, S.; Hafez, S.; Attia, E.; Lessa, F.C. National surveillance of health care–associated infections in Egypt: Developing a sustainable program in a resource-limited country. Am. J. Infect. Control 2016, 44, 1296–1301. [Google Scholar] [CrossRef] [PubMed]
- Mitharwal, S.M.; Yaddanapudi, S.; Bhardwaj, N.; Gautam, V.; Biswal, M.; Yaddanapudi, L. Intensive care unit-acquired infections in a tertiary care hospital: An epidemiologic survey and influence on patient outcomes. Am. J. Infect. Control 2016, 44, e113–e117. [Google Scholar] [CrossRef] [PubMed]
- Ardoino, I.; Zangirolami, F.; Iemmi, D.; Lanzoni, M.; Cargnelutti, M.; Biganzoli, E.M.; Castaldi, S. Risk factors and epidemiology of Acinetobacter baumannii infections in a university hospital in Northern Italy: A case-control study. Am. J. Infect. Control 2016, 44, 1600–1605. [Google Scholar] [CrossRef] [PubMed]
- Maraki, S.; Mantadakis, E.; Mavromanolaki, V.E.; Kofteridis, D.P.; Samonis, G. A 5-year Surveillance study on antimicrobial resistance of Acinetobacter baumannii clinical isolates from a tertiary Greek hospital. Infect. Chemother. 2016, 48, 190–198. [Google Scholar] [CrossRef]
- Kritsotakis, E.; Kontopidou, F.; Astrinaki, E.; Roumbelaki, M.; Ioannidou, E.; Gikas, A. Prevalence, incidence burden, and clinical impact of healthcare-associated infections and antimicrobial resistance: A national prevalent cohort study in acute care hospitals in Greece. Infect. Drug Resist. 2017, 10, 317–328. [Google Scholar] [CrossRef]
- Tomaszewski, D.; Rybicki, Z.; Duszyńska, W. The Polish Prevalence of Infection in Intensive Care (PPIC): A one-day point prevalence multicenter study. Adv. Clin. Exp. Med. 2019, 28, 907–912. [Google Scholar] [CrossRef]
- European Centre for Disease Prevention and Control. Surveillance of Antimicrobial Resistance in Europe. Annual Report of the European Antimicrobial Resistance Surveillance Network; EARS-Net 2016. Available online: http://ecdc.europea.eu/sites/portal/files/documents/AMR-surveillance-Europe-2016.pdf (accessed on 15 November 2017). [CrossRef]
- Centers for Disease Control and Prevention. Core Elements of Hospital Antibiotic Stewardship Programs. 2016. Available online: http://www.cdc.gov/getsmart/healthcare/implementation/core-elements.html (accessed on 31 August 2020).
- Campion, M.; Scully, G. Antibiotic Use in the intensive care unit: Optimization and de-escalation. J. Intensiv. Care Med. 2018, 33, 647–655. [Google Scholar] [CrossRef]
- Horan, T.C.; Andrus, M.; Dudeck, M.A. CDC/NHSN surveillance definition of health care–associated infection and criteria for specific types of infections in the acute care setting. Am. J. Infect. Control 2008, 36, 309–332. [Google Scholar] [CrossRef]
- EUCAST. Breakpoint Tables for Interpretation of MICs and Zone Diameters. Version 6.0 2016. Available online: http://www.eucast.org/clinical_breakpoints/ (accessed on 1 January 2016).
- The Council of European Communities. Programme of Community action in the field of public health (2003 to 2008). Off. J. Eur. Union. 2002, 9, 1–12. [Google Scholar]
- Rosenthal, V.D.; Al-Abdely, H.M.; El-Kholy, A.A.; Alkhawaja, S.A.A.; Leblebicioglu, H.; Mehta, Y.; Rai, V.; Hung, N.V.; Kanj, S.S.; Salama, M.F.; et al. International Nosocomial Infection Control Consortium report, data summary of 50 countries for 2010–2015: Device-associated module. Am. J. Infect. Control 2016, 44, 1495–1504. [Google Scholar] [CrossRef] [PubMed]
- European Centre for Disease Prevention and Control. Healthcare-Associated Infections Acquired in Intensive Care Units. In ECDC Annual Epidemiological Report for 2016; ECDC: Stockholm, Sweden, 2018; Available online: https://www.ecdc.europa.eu/en/publications-data/healthcare-associated-infections-intensive-care-units-annual-epidemiological-0 (accessed on 7 January 2017).
- Wałaszek, M.; Różańska, A.; Wałaszek, M.; Wójkowska-Mach, J.; Polish Society of Hospital Infections Team; The Polish Society of Hospital Infections Team; Domańska, J. Epidemiology of Ventilator-Associated Pneumonia, microbiological diagnostics and the length of antimicrobial treatment in the Polish Intensive Care Units in the years 2013–2015. BMC Infect. Dis. 2018, 18, 308. [Google Scholar] [CrossRef]
- Kołpa, M.; Wałaszek, M.; Gniadek, A.; Wolak, Z.; Dobros, W. Incidence, microbiological profile and risk factors of healthcare-associated infections in intensive care units: A 10 year observation in a provincial hospital in Southern Poland. Int. J. Environ. Res. Public Health 2018, 15, 112. [Google Scholar] [CrossRef] [PubMed]
- European Centre for Disease Prevention and Control. Healthcare-Associated Infections Acquired in Intensive Care Units. In ECDC Annual Epidemiological Report for 2017; ECDC: Stockholm, Sweden, 2019; Available online: https://www.ecdc.europa.eu/en/publications-data/healthcare-associated-infections-intensive-care-units-annual-epidemiological-1 (accessed on 10 October 2019).
- European Centre for Disease Prevention and Control. Surveillance of Antimicrobial Resistance in Europe. Annual Report of the European Antimicrobial Resistance Surveillance Network (EARS-Net). ECDC (online) 2017. Available online: https://ecdc.europa.eu/sites/porta/files/documents/EARS-Net-report-2017-update-jan-2019.pdf (accessed on 15 November 2018).
- Rosenthal, V.D.; Bat-Erdene, I.; Gupta, D.; Belkebir, S.; Rajhans, P.; Zand, F.; Myatra, S.N.; Afeef, M.; Tanzi, V.L.; Muralidharan, S.; et al. International Nosocomial Infection Control Consortium (INICC) report, data summary of 45 countries for 2012–2017: Device-associated module. Am. J. Infect. Control 2020, 48, 423–432. [Google Scholar] [CrossRef] [PubMed]
- Dahal, M.; Schwan, W.R. Management of methicillin-resistant Staphylococcus aureus mediated ventilator-associated pneumonia. Curr Trends Microbiol. 2018, 12, 95–107. [Google Scholar]
- Trejnowska, E.; Deptuła, A.; Tarczyńska-Słomian, M.; Knapik, P.; Jankowski, M.; Misiewska-Kaczur, A.; Tamowicz, B.; Smiechowicz, J.; Antończyk, R.; Armatowicz, P.; et al. Surveillance of Antibiotic Prescribing in Intensive Care Units in Poland. Can. J. Infect. Dis. Med Microbiol. 2018, 2018, 5670238. [Google Scholar] [CrossRef]
- Council of the European Union. Council Recommendation of 9 June 2009 on patient safety, including the prevention and control of healthcare associated infections. Off. J. Eur. Union. 2009, 1, 1–6. [Google Scholar]
| Pathogen | VAP | UTI | CLA-BSI | Sum |
|---|---|---|---|---|
| n (%) | n (%) | n (%) | n (%) | |
| Acinetobacter baumannii MDR | 192 (41.9) | 60 (21.9) | 11 (9.6) | 263 (31.08) |
| Pseudomonas aeruginosa | 41 (8.9) | 19 (6.9) | 4 (3.5) | 64 (7.6) |
| Pseudomonas aeruginosa MDR | 22 (4.8) | 10 (3.6) | 3 (2.6) | 35 (4.1) |
| Stenotrophomonas maltophilia | 15 (3.3) | 1 (0.4) | 2 (0.7) | 18 (2.1) |
| Klebsiella pneumoniae ESBLs | 53 (11.6) | 28 (10.2) | 15 (13.2) | 96 (11.3) |
| Enterobacter cloacae ESBLs | 6 (1.3) | 3 (1.09) | 3 (1.09) | 12 (1.4) |
| Enterobacter aerogenes ESBLs | - | - | 1 (0.4) | 1 (0.11) |
| Escherichia coli ESBLs | 2 (0.44) | 7 (2.5) | - | 9 (1.06) |
| Citrobacter freundii ESBLs | 1 (0.2) | - | - | 1 (0.11) |
| Serratia ficaria ESBLs | - | 1 (0.4) | - | 1 (0.11) |
| Proteus mirabilis ESBLs | - | 3 (1.09) | - | 3 (0.35) |
| Proteus vulgaris ESBLs | - | 1 (0.4) | - | 1 (0.11) |
| Enterobacter cloacae AMP C | 2 (0.44) | - | - | 2 (0.2) |
| Proteus mirabilis AMP C | 1 (0.2) | 1 (0.4) | - | 2 (0.2) |
| Proteus vulgaris | 1 (0.4) | - | 1 (0.11) | |
| Klebsiella pneumoniae | 20 (4.4) | 6 (2.2) | 4 (3.5) | 30 (3.5) |
| Klebsiella oxytoca | 1 (0.2) | 1 (0.4) | 1 (0.4) | 3 (0.35) |
| Escherichia coli | 10 (2.2) | 11 (4.0) | 1 (0.4) | 22 (2.6) |
| Enterobacter cloacae | 5 (1.09) | 3 (1.09) | 1 (0.4) | 9 (1.06) |
| Enterobacter aerogenes | - | - | 1 (0.4) | 1 (0.11) |
| Morganella morganii | 2 (0.44) | - | - | 2 (0.2) |
| Serratia marcescens | 5 (1.09) | - | 4 (3.5) | 9 (1.06) |
| Proteus vulgaris | 1 (0.2) | 1 (0.4) | - | 2 (0.2) |
| Proteus mirabilis | 1 (0.2) | 12 (4.4) | - | 13 (1.5) |
| Proteus pennerii | 1 (0.2) | - | - | 1 (0.11) |
| Enterobacter aerogenes | 3 (0.6) | - | - | 3 (0.35) |
| Candida spp. | 10 (2.2) | 36 (13.1) | 13 (11.4) | 59 (7.0) |
| MSCNS | 2 (0.44) | 1 (0.4) | 1 (0.4) | 4 (0.5) |
| MRCNS | - | 1 (0.4) | 34 (29.8) | 35 (4.1) |
| MRSA | 18 (3.9) | - | 1 (0.4) | 19 (2.2) |
| MSSA | 29 (6.3) | - | 1 (0.4) | 30 (3.5) |
| Enterococcus faecalis | 2 (0.44) | 13 (4.7) | 5 (4.4) | 20 (2.4) |
| Enterococcus faecium | 9 (2.0) | 7 (2.5) | - | 16 (1.9) |
| Enterococcus faecium VRE | 1 (0.2) | 5 (1.8) | - | 6 (0.7) |
| Enterococcus faecium HLAR VRE | - | 11 (4.0) | - | 11 (1.3) |
| Enterococcus faecalis HLAR | - | 9 (3.3) | 2 (0.7) | 11 (1.3) |
| Enterococcus faecium HLAR | - | 22 (8.0) | 5 (4.4) | 27 (3.2) |
| Streptococcus pneumoniae | 3 (0.6) | - | - | 3 (0.35) |
| Granulicatella spp. | - | - | 1 (0.4) | 1 (0.11) |
| Sum (%) | 458 (54.1) | 274 (32.4) | 114 (13.5) | 846 |
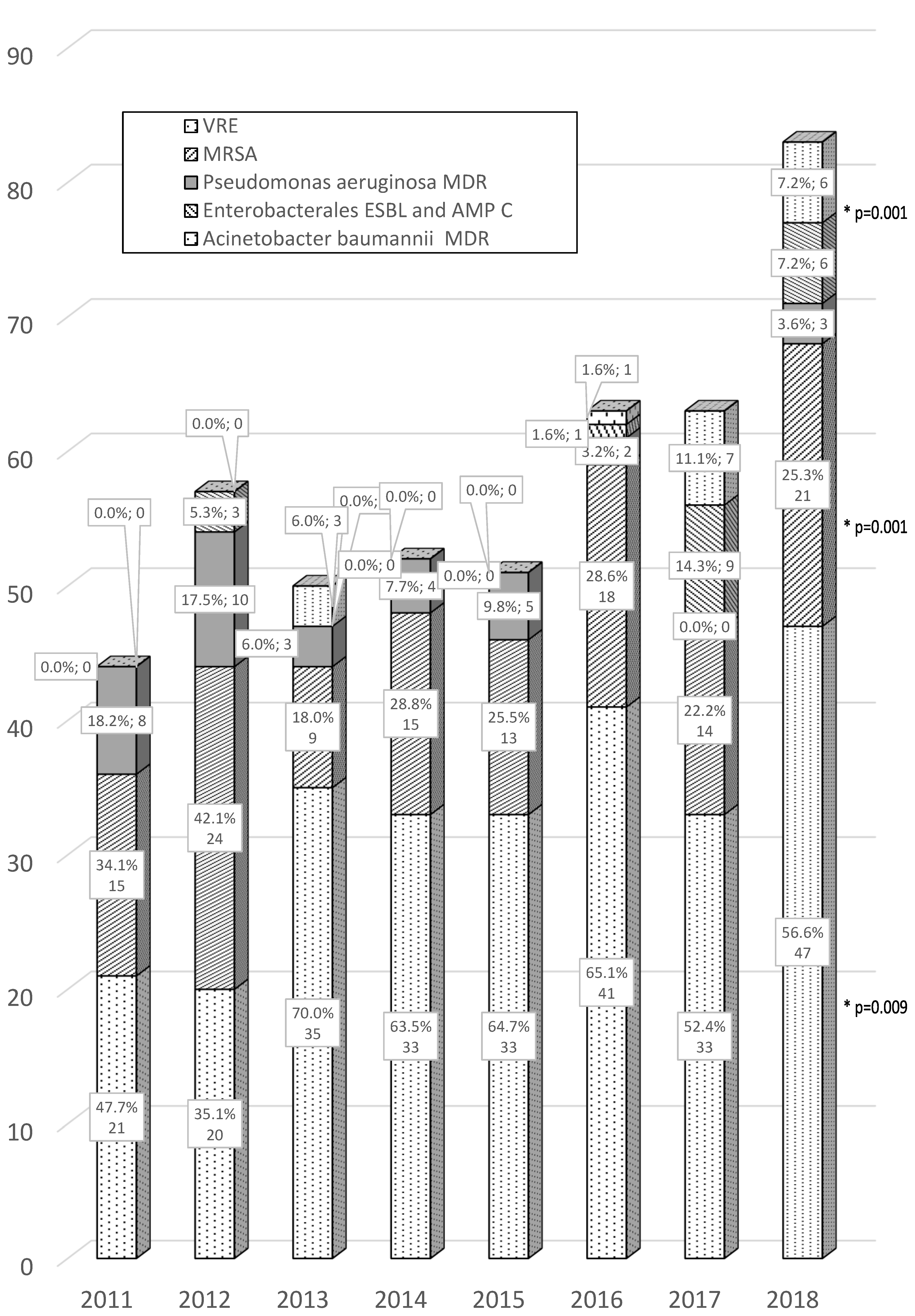
| 2011 n = 127 | 2012 n = 115 | 2013 n = 80 | 2014 n = 81 | 2015 n = 86 | 2016 n = 100 | 2017 n = 121 | 2018 n = 136 | Sum | p | |
|---|---|---|---|---|---|---|---|---|---|---|
| Acinetobacter baumannii MDR, n (%) | 21 (16.53) | 20 (17.39) | 35 (43.75) | 33 (40.74) | 33 (38.37) | 41 (41) | 33 (27.27) | 47 (34.56) | 263 | 0.0009 |
| Enterobacterales ESBL/AMPC | 15 (11.8) | 24 (20.87) | 9 (11.25) | 15 (18.52) | 13 (15.12) | 18 (18) | 14 (11.57) | 21 (15.44) | 129 | 0.3920 |
| Pseudomonas aeruginosa MDR | 8 (6.3) | 10 (8.69) | 3 (3.75) | 4 (4.94) | 5 (5.81) | 2 (2) | 0 (0) | 3 (2.21) | 35 | 0.1774 |
| MRSA | 0 (0) | 3 (2.61) | 0 (0) | 0 (0) | 0 (0) | 1 (1) | 9 (7.34) | 6 (4.4) | 19 | <0.0001 |
| VRE | 0 (0) | 0 (0) | 3 (3.75) | 0 (0) | 0 (0) | 1 (1) | 7 (7.34) | 6 (4.4) | 17 | <0.0001 |
| Sum of “alert pathogens” | 44 | 57 | 50 | 52 | 51 | 63 | 63 | 83 | 463 |
| 2011 | 2012 | 2013 | 2014 | 2015 | 2016 | 2017 | 2018 | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| MEM | CIP | AN | MEM | CIP | AN | MEM | CIP | AN | MEM | CIP | AN | MEM | CIP | AN | MEM | CIP | AN | MEM | CIP | AN | MEM | CIP | AN | |
| Acinetobacter baumannii MDR n/(%”R”) | 4/(19) | 19/(90) | 13 (62) | 11/(55) | 19/(95) | 13/(65) | 33/(94) | 34/(98) | 31/(89) | 33/(100) | 33/(100) | 28/(88) | 32/(97) | 33/(100) | 28/(85) | 38/(93) | 39/(95) | 36/(88) | 31/(94) | 31/(94) | 31/(94) | 47 (100) | 47/(100) | 43/(92) |
| Pseudomonas aeruginosa MDR n/(%”R”) | 9/(45) | 11/(55) | 8/(40) | 8/(44) | 11/(61) | 5/(28) | 4/(57) | 2/(19) | 1/(14) | 6/(46) | 9/(69) | 3/(23) | 4/(33) | 5/(42) | 2/(17) | 3/(60) | 3/(60) | 1/(20) | 3/(21) | 0/(0) | 0/(0) | 4/(40) | 4/(40) | 3/(30) |
| Klebsiella pneumoniae ESBLs n/(%”R”) | 0/(0) | 9/(93) | 7/(74) | 0/(0) | 12/(92) | 6/(40) | 0/(0) | 9/(100) | 3/(33) | 0/(0) | 15/(100) | 7/(50) | 0/(0) | 12/(92) | 5/(46) | 0/(0) | 11/(78) | 4/(25) | 0/(0) | 8/(86) | 3/(29) | 0/(0) | 14/(95) | 5/(34) |
| DDD/1000 Patient-Days | |||
|---|---|---|---|
| 2011 | 2014 | 2018 | |
| Amikacin | 33.50 | 44.23 | 30.70 |
| Ciprofloxacin | 119.03 | 67.47 | 27.71 |
| Carbapenems | 197.74 | 228.97 | 236.88 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Litwin, A.; Fedorowicz, O.; Duszynska, W. Characteristics of Microbial Factors of Healthcare-Associated Infections Including Multidrug-Resistant Pathogens and Antibiotic Consumption at the University Intensive Care Unit in Poland in the Years 2011–2018. Int. J. Environ. Res. Public Health 2020, 17, 6943. https://doi.org/10.3390/ijerph17196943
Litwin A, Fedorowicz O, Duszynska W. Characteristics of Microbial Factors of Healthcare-Associated Infections Including Multidrug-Resistant Pathogens and Antibiotic Consumption at the University Intensive Care Unit in Poland in the Years 2011–2018. International Journal of Environmental Research and Public Health. 2020; 17(19):6943. https://doi.org/10.3390/ijerph17196943
Chicago/Turabian StyleLitwin, Agnieszka, Olga Fedorowicz, and Wieslawa Duszynska. 2020. "Characteristics of Microbial Factors of Healthcare-Associated Infections Including Multidrug-Resistant Pathogens and Antibiotic Consumption at the University Intensive Care Unit in Poland in the Years 2011–2018" International Journal of Environmental Research and Public Health 17, no. 19: 6943. https://doi.org/10.3390/ijerph17196943
APA StyleLitwin, A., Fedorowicz, O., & Duszynska, W. (2020). Characteristics of Microbial Factors of Healthcare-Associated Infections Including Multidrug-Resistant Pathogens and Antibiotic Consumption at the University Intensive Care Unit in Poland in the Years 2011–2018. International Journal of Environmental Research and Public Health, 17(19), 6943. https://doi.org/10.3390/ijerph17196943






