Zero-Valent Iron Nanoparticles for Soil and Groundwater Remediation
Abstract
1. Introduction
2. Zero-Valent Iron
3. Zero-Valent Iron Nanoparticles and Nanoremediation
3.1. Bimetallic Iron-Based Nanoparticles
Pilot and Full-Scale Test for Bimetallic Iron-Based Nanoparticles
3.2. Emulsified Zero-Valent Iron
Pilot and Full-Scale Test for Emulsified Zero-Valent Iron
3.3. Polymer Coated NZVI Particles
3.3.1. NZVI Coated with Synthetic Polymers
3.3.2. NZVI Coated with Natural Polymers
3.3.3. Pilot and Full-Scale Test for Polymer Coated NZVI Particles
4. Limitations and Risks Derived from NZVI Nanoremediation
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviation
| ·OH | hydroxyl radical |
| 124TCB | 1,2,4-trichlorobenzene |
| 2,3,7,8-TCDD | 2,3,7,8-tetraclorodibenzo-p-dioxine |
| BDE-209 | decabromodiphenyl ether |
| BDE-47 | 2,2′,4,4′-tetrabromodiphenyl ether |
| BMP | bimetallic iron-based nanoparticle |
| cis-DCE | cis-dichloroethylene |
| CMC | carboxymethylcellulose |
| DDT | Dichlorodiphenyltrichloroethan |
| DNAPL | dense non-aqueous liquid phase |
| EZVI | emulsified iron nanoparticles |
| GG | guar gum |
| MCB | monochlorobenzene |
| n/a | Not available |
| NZVI | Nanoscale zero-valent iron |
| OMA | Olefin maleic acid |
| PAA | Poly(acrylic acid) |
| PAH | polycyclic aromatic hydrocarbons |
| PAM | polyacrylamide |
| PAS | polyaspartate |
| PCB | polychlorinated biphenyl |
| PCE | Tetrachloroethylene or Perchloroethylene |
| PEG | polyethylen glycol |
| PMMA | poly(methacrylic acid) |
| PRB | permeable reactive barrier |
| PSS | polystyrene sulfonate |
| PTHF | polytetrahydrofuran |
| PV3A | polyvinyl alcohol-co-vinyl acetate-co-itaconic acid |
| PVP | polyvinylpyrrolidone |
| TC | tetracycline |
| TCA | trichloroethane |
| TCC | Triclocarban |
| TCE | trichloroethylene |
| TNT | 2,4,6-trinitrotoluene |
| trans-DCE | trans-dichloroethylene |
| VC | vinyl chloride |
| VOC | Volatile organic compound |
| VOCl | Volatile organic chlorides |
| XG | xanthan gum |
| ZVI | Zero-valent iron |
References
- Mareddy, A.R. Technology in EIA. In Environmental Impact Assessment; Elsevier: Amsterdam, The Netherlands, 2017; pp. 421–490. [Google Scholar]
- Lombi, E.; Hamon, R.E. Remediation of polluted soils. In Encyclopedia of Soils in the Environment; Elsevier: Amsterdam, The Netherlands, 2005; pp. 379–385. [Google Scholar]
- USEPA US EPA Engineering Issue In Situ and Ex Situ Biodegradation Technologies for Remediation of Contaminated Sites. US Res. Off. Transf. Support Div. Technol. Grosse Douglas 2006, 1–22.
- Gomes, H.I.; Dias-Ferreira, C.; Ribeiro, A.B. Overview of in situ and ex situ remediation technologies for PCB-contaminated soils and sediments and obstacles for full-scale application. Sci. Total Environ. 2013, 445–446, 237–260. [Google Scholar] [CrossRef] [PubMed]
- Paula, R.J.; Paula, A.J. Treatment of Soil Pollutants With Nanomaterials. In Nanomaterials Applications for Environmental Matrices; Elsevier: Amsterdam, The Netherlands, 2019; pp. 355–368. [Google Scholar]
- Horváthová, H.; Lászlová, K.; Dercová, K. Bioremediation vs. Nanoremediation: Degradation of Polychlorinated Biphenyls (PCBS) Using Integrated Remediation Approaches. Water Air Soil Pollut. 2019, 230, 204. [Google Scholar] [CrossRef]
- Lu, F.; Astruc, D. Nanocatalysts and other nanomaterials for water remediation from organic pollutants. Coord. Chem. Rev. 2020, 408, 213180. [Google Scholar] [CrossRef]
- Gudeppu, M.; Varier, K.M.; Chinnasamy, A.; Thangarajan, S.; Balasubramanian, J.; Li, Y.; Gajendran, B. Nanobiotechnology Approach for the Remediation of Environmental Hazards Generated from Industrial Waste. In Emerging Nanostructured Materials for Energy and Environmetal Sience; Springer Nature: Cham, Switzerland, 2019; pp. 531–561. [Google Scholar]
- Cai, Z.; Zhao, X.; Duan, J.; Zhao, D.; Dang, Z.; Lin, Z. Remediation of soil and groundwater contaminated with organic chemicals using stabilized nanoparticles: Lessons from the past two decades. Front. Environ. Sci. Eng. 2020, 14, 84. [Google Scholar] [CrossRef]
- Reinikainen, J.; Sorvari, J.; Tikkanen, S. Finnish policy approach and measures for the promotion of sustainability in contaminated land management. J. Environ. Manag. 2016, 184, 108–119. [Google Scholar] [CrossRef]
- Mulligan, C.N.; Yong, R.N.; Gibbs, B.F. An evaluation of technologies for the heavy metal remediation of dredged sediments. J. Hazard. Mater. 2001, 85, 145–163. [Google Scholar] [CrossRef]
- Eugris. 2019. Available online: http://www.eugris.info/ (accessed on 26 October 2019).
- Sweeny, K.H.; Fischer, J.R. Reductive degradation of halogenated pesticides. U.S. Patent US3640821A, 8 February 1972. [Google Scholar]
- Gillham, R.W.; O’Hannesin, S.F. Enhanced Degradation of Halogenated Aliphatics by Zero-Valent Iron. Ground Water 1994, 32, 958–967. [Google Scholar] [CrossRef]
- O’Hannesin, S.F.; Gillham, R.W. Long-Term Performance of an In Situ “Iron Wall” for Remediation of VOCs. Ground Water 1998, 36, 164–170. [Google Scholar] [CrossRef]
- Tratnyek, P. Permeable Reactive Barriers of Iron and Other Zero- Valent Metals. In Chemical degradation methods for wastes and pollutants: Environmental and industrial applications; Marcel Dekker: New York, NY, USA, 2003. [Google Scholar]
- Orth, W.S.; Gillham, R.W. Dechlorination of Trichloroethene in Aqueous Solution Using Fe 0. Environ. Sci. Technol. 1996, 30, 66–71. [Google Scholar] [CrossRef]
- Arnold, W.A.; Roberts, A.L. Pathways and Kinetics of Chlorinated Ethylene and Chlorinated Acetylene Reaction with Fe(0) Particles. Environ. Sci. Technol. 2000, 34, 1794–1805. [Google Scholar] [CrossRef]
- Hwang, Y.-H.; Kim, D.-G.; Shin, H.-S. Mechanism study of nitrate reduction by nano zero valent iron. J. Hazard. Mater. 2011, 185, 1513–1521. [Google Scholar] [CrossRef] [PubMed]
- Cantrell, K.J.; Kaplan, D.I.; Wietsma, T.W. Zero-valent iron for the in situ remediation of selected metals in groundwater. J. Hazard. Mater. 1995, 42, 201–212. [Google Scholar] [CrossRef]
- Gould, J.P. The kinetics of hexavalent chromium reduction by metallic iron. Water Res. 1982, 16, 871–877. [Google Scholar] [CrossRef]
- Bandstra, J.Z.; Miehr, R.; Johnson, R.L.; Tratnyek, P.G. Reduction of 2,4,6-Trinitrotoluene by Iron Metal: Kinetic Controls on Product Distributions in Batch Experiments. Environ. Sci. Technol. 2005, 39, 230–238. [Google Scholar] [CrossRef]
- Matheson, L.J.; Tratnyek, P.G. Reductive Dehalogenation of Chlorinated Methanes by Iron Metal. Environ. Sci. Technol. 1994, 28, 2045–2053. [Google Scholar] [CrossRef]
- Agrawal, A.; Tratnyek, P.G. Reduction of Nitro Aromatic Compounds by Zero-Valent Iron Metal. Environ. Sci. Technol. 1996, 30, 153–160. [Google Scholar] [CrossRef]
- Johnson, T.L.; Scherer, M.M.; Tratnyek, P.G. Kinetics of Halogenated Organic Compound Degradation by Iron Metal. Environ. Sci. Technol. 1996, 30, 2634–2640. [Google Scholar] [CrossRef]
- Alonso, F.; Beletskaya, I.P.; Yus, M. Metal-Mediated Reductive Hydrodehalogenation of Organic Halides. Chem. Rev. 2002, 102, 4009–4092. [Google Scholar] [CrossRef]
- Alowitz, M.J.; Scherer, M.M. Kinetics of Nitrate, Nitrite, and Cr(VI) Reduction by Iron Metal. Environ. Sci. Technol. 2002, 36, 299–306. [Google Scholar] [CrossRef]
- Wang, C.-B.; Zhang, W. Synthesizing Nanoscale Iron Particles for Rapid and Complete Dechlorination of TCE and PCBs. Environ. Sci. Technol. 1997, 31, 2154–2156. [Google Scholar] [CrossRef]
- Zhao, X.; Liu, W.; Cai, Z.; Han, B.; Qian, T.; Zhao, D. An overview of preparation and applications of stabilized zero-valent iron nanoparticles for soil and groundwater remediation. Water Res. 2016, 100, 245–266. [Google Scholar] [CrossRef] [PubMed]
- O’Carroll, D.; Sleep, B.; Krol, M.; Boparai, H.; Kocur, C. Nanoscale zero valent iron and bimetallic particles for contaminated site remediation. Adv. Water Resour. 2013, 51, 104–122. [Google Scholar] [CrossRef]
- Deng, B.; Hu, S. Reductive dechlorination of chlorinated solvents on zerovalent iron surfaces. Physicochem. Groundw. Remediat. 2001, 139–159. [Google Scholar]
- U.S. Epa Nanotechnology for Site Remediation Fact Sheet. U.S. Environ. Prot. Agency 2008, 1–17.
- Cundy, A.B.; Hopkinson, L.; Whitby, R.L.D. Use of iron-based technologies in contaminated land and groundwater remediation: A review. Sci. Total Environ. 2008, 400, 42–51. [Google Scholar] [CrossRef]
- Macé, C.; Desrocher, S.; Gheorghiu, F.; Kane, A.; Pupeza, M.; Cernik, M.; Kvapil, P.; Venkatakrishnan, R.; Zhang, W. Nanotechnology and groundwater remediation: A step forward in technology understanding. Remediat. J. 2006, 16, 23–33. [Google Scholar] [CrossRef]
- Keenan, C.R.; Sedlak, D.L. Factors Affecting the Yield of Oxidants from the Reaction of Nanoparticulate Zero-Valent Iron and Oxygen. Environ. Sci. Technol. 2008, 42, 1262–1267. [Google Scholar] [CrossRef]
- Araújo, R.; Castro, A.C.M.; Fiúza, A. The Use of Nanoparticles in Soil and Water Remediation Processes. Mater. Today Proc. 2015, 2, 315–320. [Google Scholar] [CrossRef]
- Trujillo-Reyes, J.; Peralta-Videa, J.R.; Gardea-Torresdey, J.L. Supported and unsupported nanomaterials for water and soil remediation: Are they a useful solution for worldwide pollution? J. Hazard. Mater. 2014, 280, 487–503. [Google Scholar] [CrossRef]
- Mitra, S.; Sarkar, A.; Sen, S. Removal of chromium from industrial effluents using nanotechnology: A review. Nanotechnol. Environ. Eng. 2017, 2, 11. [Google Scholar] [CrossRef]
- Patil, P.M.; Bohara, R.A. Nanoparticles impact in biomedical waste management. Waste Manag. Res. 2020, 0734242X2093676. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Wang, Y.; Zhao, D.; Pan, G. Immobilization of arsenic in soils by stabilized nanoscale zero-valent iron, iron sulfide (FeS), and magnetite (Fe3O4) particles. Chinese Sci. Bull. 2010, 55, 365–372. [Google Scholar] [CrossRef]
- Gil-Díaz, M.; Diez-Pascual, S.; González, A.; Alonso, J.; Rodríguez-Valdés, E.; Gallego, J.R.; Lobo, M.C. A nanoremediation strategy for the recovery of an As-polluted soil. Chemosphere 2016, 149, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Baragaño, D.; Alonso, J.; Gallego, J.R.; Lobo, M.C.; Gil-Díaz, M. Magnetite nanoparticles for the remediation of soils co-contaminated with As and PAHs. Chem. Eng. J. 2020, 399, 125809. [Google Scholar] [CrossRef]
- Mar Gil-Díaz, M.; Pérez-Sanz, A.; Ángeles Vicente, M.; Carmen Lobo, M. Immobilisation of Pb and Zn in Soils Using Stabilised Zero-valent Iron Nanoparticles: Effects on Soil Properties. CLEAN Soil Air Water 2014, 42, 1776–1784. [Google Scholar] [CrossRef]
- Wang, Y.; Fang, Z.; Kang, Y.; Tsang, E.P. Immobilization and phytotoxicity of chromium in contaminated soil remediated by CMC-stabilized nZVI. J. Hazard. Mater. 2014, 275, 230–237. [Google Scholar] [CrossRef]
- Zhu, F.; Li, L.; Ma, S.; Shang, Z. Effect factors, kinetics and thermodynamics of remediation in the chromium contaminated soils by nanoscale zero valent Fe/Cu bimetallic particles. Chem. Eng. J. 2016, 302, 663–669. [Google Scholar] [CrossRef]
- Singh, R.; Misra, V.; Singh, R.P. Removal of Cr(VI) by Nanoscale Zero-valent Iron (nZVI) From Soil Contaminated with Tannery Wastes. Bull. Environ. Contam. Toxicol. 2012, 88, 210–214. [Google Scholar] [CrossRef]
- Su, H.; Fang, Z.; Tsang, P.E.; Zheng, L.; Cheng, W.; Fang, J.; Zhao, D. Remediation of hexavalent chromium contaminated soil by biochar-supported zero-valent iron nanoparticles. J. Hazard. Mater. 2016, 318, 533–540. [Google Scholar] [CrossRef]
- Chen, C.-F.; Binh, N.T.; Chen, C.-W.; Dong, C.-D. Removal of polycyclic aromatic hydrocarbons from sediments using sodium persulfate activated by temperature and nanoscale zero-valent iron. J. Air Waste Manag. Assoc. 2015, 65, 375–383. [Google Scholar] [CrossRef] [PubMed]
- Dorjee, P.; Amarasiriwardena, D.; Xing, B. Antimony adsorption by zero-valent iron nanoparticles (nZVI): Ion chromatography–inductively coupled plasma mass spectrometry (IC–ICP-MS) study. Microchem. J. 2014, 116, 15–23. [Google Scholar] [CrossRef]
- Zhang, Z.; Liu, J.; Cao, X.; Luo, X.; Hua, R.; Liu, Y.; Yu, X.; He, L.; Liu, Y. Comparison of U(VI) adsorption onto nanoscale zero-valent iron and red soil in the presence of U(VI)–CO 3 /Ca–U(VI)–CO 3 complexes. J. Hazard. Mater. 2015, 300, 633–642. [Google Scholar] [CrossRef]
- Li, X.Q.; Elliott, D.W.; Zhang, W.X. Zero-Valent Iron Nanoparticles for Abatement of Environmental Pollutants: Materials and Engineering Aspects. Crit. Rev. Solid State Mater. Sci. 2006, 31, 111–122. [Google Scholar] [CrossRef]
- Zhang, W. Nanoscale Iron Particles for Environmental Remediation: An Overview. J. Nanoparticle Res. 2003, 5, 323–332. [Google Scholar] [CrossRef]
- Pardo, F.; Santos, A.; Romero, A. Fate of iron and polycyclic aromatic hydrocarbons during the remediation of a contaminated soil using iron-activated persulfate: A column study. Sci. Total Environ. 2016, 566–567, 480–488. [Google Scholar] [CrossRef]
- Chang, M.-C.; Shu, H.-Y.; Hsieh, W.-P.; Wang, M.-C. Remediation of Soil Contaminated with Pyrene Using Ground Nanoscale Zero-Valent Iron. J. Air Waste Manag. Assoc. 2007, 57, 221–227. [Google Scholar] [CrossRef]
- Peluffo, M.; Pardo, F.; Santos, A.; Romero, A. Use of different kinds of persulfate activation with iron for the remediation of a PAH-contaminated soil. Sci. Total Environ. 2016, 563–564, 649–656. [Google Scholar] [CrossRef]
- Chowdhury, A.I.A.; Krol, M.M.; Kocur, C.M.; Boparai, H.K.; Weber, K.P.; Sleep, B.E.; O’Carroll, D.M. nZVI injection into variably saturated soils: Field and modeling study. J. Contam. Hydrol. 2015, 183, 16–28. [Google Scholar] [CrossRef]
- Kustov, L.M.; Finashina, E.D.; Shuvalova, E.V.; Tkachenko, O.P.; Kirichenko, O.A. Pd–Fe nanoparticles stabilized by chitosan derivatives for perchloroethene dechlorination. Environ. Int. 2011, 37, 1044–1052. [Google Scholar] [CrossRef]
- Machado, S.; Pacheco, J.G.; Nouws, H.P.A.; Albergaria, J.T.; Delerue-Matos, C. Characterization of green zero-valent iron nanoparticles produced with tree leaf extracts. Sci. Total Environ. 2015, 533, 76–81. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.-C.; Lei, M.; Chen, T.-B.; Li, X.-Y.; Liang, Q.; Ma, C. Application of zerovalent iron (Fe0) to enhance degradation of HCHs and DDX in soil from a former organochlorine pesticides manufacturing plant. Chemosphere 2010, 79, 727–732. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Shi, N.; Wang, H.; Pan, X.; Fang, H.; Yu, Y. Nanoscale zerovalent iron-mediated degradation of DDT in soil. Environ. Sci. Pollut. Res. 2016, 23, 6253–6263. [Google Scholar] [CrossRef]
- Jiamjitrpanich, W.; Parkpian, P.; Polprasert, C.; Laurent, F.; Kosanlavit, R. The tolerance efficiency of Panicum maximum and Helianthus annuus in TNT-contaminated soil and nZVI-contaminated soil. J. Environ. Sci. Heal. Part A 2012, 47, 1506–1513. [Google Scholar] [CrossRef] [PubMed]
- Binh, N.D.; Imsapsangworn, C.; Kim Oanh, N.T.; Parkpian, P.; Karstensen, K.; Giao, P.H.; DeLaune, R.D. Sequential anaerobic–aerobic biodegradation of 2,3,7,8-TCDD contaminated soil in the presence of CMC-coated nZVI and surfactant. Environ. Technol. 2016, 37, 388–398. [Google Scholar] [CrossRef]
- Gomes, H.I.; Ottosen, L.M.; Ribeiro, A.B.; Dias-Ferreira, C. Treatment of a suspension of PCB contaminated soil using iron nanoparticles and electric current. J. Environ. Manag. 2015, 151, 550–555. [Google Scholar] [CrossRef]
- San Román, I.; Alonso, M.L.; Bartolomé, L.; Galdames, A.; Goiti, E.; Ocejo, M.; Moragues, M.; Alonso, R.M.; Vilas, J.L. Relevance study of bare and coated zero valent iron nanoparticles for lindane degradation from its by-product monitorization. Chemosphere 2013, 93, 1324–1332. [Google Scholar] [CrossRef]
- San Román, I.; Galdames, A.; Alonso, M.L.; Bartolomé, L.; Vilas, J.L.; Alonso, R.M. Effect of coating on the environmental applications of zero valent iron nanoparticles: The lindane case. Sci. Total Environ. 2016, 565, 795–803. [Google Scholar] [CrossRef]
- Schrick, B.; Hydutsky, B.W.; Blough, J.L.; Mallouk, T.E. Delivery Vehicles for Zerovalent Metal Nanoparticles in Soil and Groundwater. Chem. Mater. 2004, 16, 2187–2193. [Google Scholar] [CrossRef]
- Hennebel, T.; Simoen, H.; De Windt, W.; Verloo, M.; Boon, N.; Verstraete, W. Biocatalytic dechlorination of trichloroethylene with bio-palladium in a pilot-scale membrane reactor. Biotechnol. Bioeng. 2009, 102, 995–1002. [Google Scholar] [CrossRef]
- Lefevre, E.; Bossa, N.; Wiesner, M.R.; Gunsch, C.K. A review of the environmental implications of in situ remediation by nanoscale zero valent iron (nZVI): Behavior, transport and impacts on microbial communities. Sci. Total Environ. 2016, 565, 889–901. [Google Scholar] [CrossRef] [PubMed]
- Otto, M.; Floyd, M.; Bajpai, S. Nanotechnology for site remediation. Remediat. J. 2008, 19, 99–108. [Google Scholar] [CrossRef]
- Karn, B.; Kuiken, T.; Otto, M. Nanotechnology and in Situ Remediation: A Review of the Benefits and Potential Risks. Environ. Health Perspect. 2009, 117, 1813–1831. [Google Scholar] [CrossRef] [PubMed]
- Lowry, G.V.; Casman, E.A. Nanomaterial Transport, Transformation, and Fate in the Environment. In Nanomaterials: Risks and Benefits; Springer: Dordrecht, The Netherlands, 2009; pp. 125–137. [Google Scholar]
- Tratnyek, P.G.; Johnson, R.L. Nanotechnologies for environmental cleanup. Nano Today 2006, 1, 44–48. [Google Scholar] [CrossRef]
- Saleh, N.; Kim, H.-J.; Phenrat, T.; Matyjaszewski, K.; Tilton, R.D.; Lowry, G.V. Ionic Strength and Composition Affect the Mobility of Surface-Modified Fe 0 Nanoparticles in Water-Saturated Sand Columns. Environ. Sci. Technol. 2008, 42, 3349–3355. [Google Scholar] [CrossRef]
- Comba, S.; Di Molfetta, A.; Sethi, R. A Comparison Between Field Applications of Nano-, Micro-, and Millimetric Zero-Valent Iron for the Remediation of Contaminated Aquifers. Water. Air. Soil Pollut. 2011, 215, 595–607. [Google Scholar] [CrossRef]
- Zhu, B.-W.; Lim, T.-T. Catalytic Reduction of Chlorobenzenes with Pd/Fe Nanoparticles: Reactive Sites, Catalyst Stability, Particle Aging, and Regeneration. Environ. Sci. Technol. 2007, 41, 7523–7529. [Google Scholar] [CrossRef]
- Choi, H.; Al-Abed, S.R.; Agarwal, S.; Dionysiou, D.D. Synthesis of Reactive Nano-Fe/Pd Bimetallic System-Impregnated Activated Carbon for the Simultaneous Adsorption and Dechlorination of PCBs. Chem. Mater. 2008, 20, 3649–3655. [Google Scholar] [CrossRef]
- Zhuang, Y.; Ahn, S.; Seyfferth, A.L.; Masue-Slowey, Y.; Fendorf, S.; Luthy, R.G. Dehalogenation of Polybrominated Diphenyl Ethers and Polychlorinated Biphenyl by Bimetallic, Impregnated, and Nanoscale Zerovalent Iron. Environ. Sci. Technol. 2011, 45, 4896–4903. [Google Scholar] [CrossRef]
- Lien, H.-L.; Zhang, W. Hydrodechlorination of Chlorinated Ethanes by Nanoscale Pd/Fe Bimetallic Particles. J. Environ. Eng. 2005, 131, 4–10. [Google Scholar] [CrossRef]
- Schrick, B.; Blough, J.L.; Jones, A.D.; Mallouk, T.E. Hydrodechlorination of Trichloroethylene to Hydrocarbons Using Bimetallic Nickel−Iron Nanoparticles. Chem. Mater. 2002, 14, 5140–5147. [Google Scholar] [CrossRef]
- Wang, R.; Lu, G.; Lin, H.; Huang, K.; Tang, T.; Xue, X.; Yang, X.; Yin, H.; Dang, Z. Relative roles of H-atom transfer and electron transfer in the debromination of polybrominated diphenyl ethers by palladized nanoscale zerovalent iron. Environ. Pollut. 2017, 222, 331–337. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Fang, Z.; Cheng, W.; Tsang, P.E.; Zhao, D. Remediation of polybrominated diphenyl ethers in soil using Ni/Fe bimetallic nanoparticles: Influencing factors, kinetics and mechanism. Sci. Total Environ. 2014, 485–486, 363–370. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Jin, X.; Owens, G.; Chen, Z. Simultaneous removal of mixed contaminants triclosan and copper by green synthesized bimetallic iron/nickel nanoparticles. Sci. Total Environ. 2019, 695, 133878. [Google Scholar] [CrossRef] [PubMed]
- Ou, J.-H.; Sheu, Y.-T.; Tsang, D.C.W.; Sun, Y.-J.; Kao, C.-M. Application of iron/aluminum bimetallic nanoparticle system for chromium-contaminated groundwater remediation. Chemosphere 2020, 256, 127158. [Google Scholar] [CrossRef] [PubMed]
- Foster, S.L.; Estoque, K.; Voecks, M.; Rentz, N.; Greenlee, L.F. Removal of Synthetic Azo Dye Using Bimetallic Nickel-Iron Nanoparticles. J. Nanomater. 2019, 2019, 1–12. [Google Scholar] [CrossRef]
- Niksefat, M.; Ayati, B. Reduction and reuse of Fe-Ni bimetallic nanoparticles oxide and evaluating its ability in acid red 14 removal. Environ. Prog. Sustain. Energy 2016, 35, 1646–1656. [Google Scholar] [CrossRef]
- Sarvari, H.; Goharshadi, E.K.; Samiee, S.; Ashraf, N. Removal of methyl orange from aqueous solutions by ferromagnetic Fe/Ni nanoparticles. Phys. Chem. Res. 2018, 6, 433–446. [Google Scholar] [CrossRef]
- Gautam, R.K.; Rawat, V.; Banerjee, S.; Sanroman, M.A.; Soni, S.; Singh, S.K.; Chattopadhyaya, M.C. Synthesis of bimetallic Fe–Zn nanoparticles and its application towards adsorptive removal of carcinogenic dye malachite green and Congo red in water. J. Mol. Liq. 2015, 212, 227–236. [Google Scholar] [CrossRef]
- Luo, F.; Yang, D.; Chen, Z.; Megharaj, M.; Naidu, R. One-step green synthesis of bimetallic Fe/Pd nanoparticles used to degrade Orange II. J. Hazard. Mater. 2016, 303, 145–153. [Google Scholar] [CrossRef]
- Dong, H.; Jiang, Z.; Zhang, C.; Deng, J.; Hou, K.; Cheng, Y.; Zhang, L.; Zeng, G. Removal of tetracycline by Fe/Ni bimetallic nanoparticles in aqueous solution. J. Colloid Interface Sci. 2018, 513, 117–125. [Google Scholar] [CrossRef] [PubMed]
- Lien, H.-L.; Zhang, W.-X. Nanoscale Pd/Fe bimetallic particles: Catalytic effects of palladium on hydrodechlorination. Appl. Catal. B Environ. 2007, 77, 110–116. [Google Scholar] [CrossRef]
- He, F.; Zhao, D. Preparation and characterization of a new class of starch-stabilized bimetallic nanoparticles for degradation of chlorinated hydrocarbons in water. Environ. Sci. Technol. 2005, 39, 3314–3320. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.X.; Elliott, D.W. Applications of iron nanoparticles for groundwater remediation. Remediation 2006, 16, 7–21. [Google Scholar] [CrossRef]
- Lien, H.-L.; Zhang, W. Transformation of Chlorinated Methanes by Nanoscale Iron Particles. J. Environ. Eng. 1999, 125, 1042–1047. [Google Scholar] [CrossRef]
- Lien, H.-L.; Zhang, W. Nanoscale iron particles for complete reduction of chlorinated ethenes. Colloids Surfaces A Physicochem. Eng. Asp. 2001, 191, 97–105. [Google Scholar] [CrossRef]
- Lin, Y.; Chen, Z.; Megharaj, M.; Naidu, R. Degradation of scarlet 4BS in aqueous solution using bimetallic Fe/Ni nanoparticles. J. Colloid Interface Sci. 2012, 381, 30–35. [Google Scholar] [CrossRef]
- Bokare, A.D.; Chikate, R.C.; Rode, C.V.; Paknikar, K.M. Iron-nickel bimetallic nanoparticles for reductive degradation of azo dye Orange G in aqueous solution. Appl. Catal. B Environ. 2008, 79, 270–278. [Google Scholar] [CrossRef]
- Luo, S.; Yang, S.; Sun, C.; Gu, J.-D. Improved debromination of polybrominated diphenyl ethers by bimetallic iron–silver nanoparticles coupled with microwave energy. Sci. Total Environ. 2012, 429, 300–308. [Google Scholar] [CrossRef]
- Gavaskar, A.; Tatar, L.; Condit, W. Cost and performance report nanoscale zero-valent iron technologies for source remediation; Naval Facilities Engineering Service Center: Port Hueneme, CA, USA, 2005. [Google Scholar]
- Elliott, D.W.; Zhang, W.X. Field assessment of nanoscale bimetallic particles for groundwater treatment. Environ. Sci. Technol. 2001, 35, 4922–4926. [Google Scholar] [CrossRef]
- He, F.; Zhao, D. Manipulating the Size and Dispersibility of Zerovalent Iron Nanoparticles by Use of Carboxymethyl Cellulose Stabilizers. Environ. Sci. Technol. 2007, 41, 6216–6221. [Google Scholar] [CrossRef] [PubMed]
- Henn, K.W.; Waddill, D.W. Utilization of nanoscale zero-valent iron for source remediation—A case study. Remediat. J. 2006, 16, 57–77. [Google Scholar] [CrossRef]
- Wei, Y.T.; Wu, S.C.; Chou, C.M.; Che, C.H.; Tsai, S.M.; Lien, H.L. Influence of nanoscale zero-valent iron on geochemical properties of groundwater and vinyl chloride degradation: A field case study. Water Res. 2010, 44, 131–140. [Google Scholar] [CrossRef] [PubMed]
- Reinhart, D.R.; Clausen, C.; Geiger, C.L.; Quinn, J.; Brooks, K. Zero-valent metal emulsion for reductive dehalogenation of DNAPLs. U.S. Patent US7037946B1, 31 October 2003. [Google Scholar]
- Singh, R.; Misra, V. Stabilization of Zero-Valent Iron Nanoparticles: Role of Polymers and Surfactants. In Handbook of Nanoparticles; Springer International Publishing: Cham, Switwerland, 2016; pp. 985–1007. ISBN 9783319153384. [Google Scholar]
- O’Hara, S.; Krug, T.; Quinn, J.; Clausen, C.; Geiger, C. Field and laboratory evaluation of the treatment of DNAPL source zones using emulsified zero-valent iron. Remediation 2006, 16, 35–56. [Google Scholar] [CrossRef]
- Li, J.; Wen, C.; Dong, J.; Liu, D.; Zhang, W.; Qin, C.; Jiang, H.; Hong, M. Study on degradation of nitrobenzene in groundwater using emulsified nano-zero-valent iron. J. Nanoparticle Res. 2015, 17. [Google Scholar] [CrossRef]
- Quinn, J.; Geiger, C.; Clausen, C.; Brooks, K.; Coon, C.; O’Hara, S.; Krug, T.; Major, D.; Yoon, W.S.; Gavaskar, A.; et al. Field demonstration of DNAPL dehalogenation using emulsified zero-valent iron. Environ. Sci. Technol. 2005, 39, 1309–1318. [Google Scholar] [CrossRef]
- McDowall, L. Degradation of toxic chemicals by zero-valent metal nanoparticles—A literature review. Def. Sci. Technol. Organ. 2005, DSTO-GD-04, 1–40. [Google Scholar]
- Krug, T.A.; Puls, R.W.; Ruiz, N.E.; Quinn, J.W.; Watling, M.T.; Su, C.; O’Hara, S.K.; Puls, R.W.; Krug, T.A.; Watling, M.T.; et al. A two and half-year-performance evaluation of a field test on treatment of source zone tetrachloroethene and its chlorinated daughter products using emulsified zero valent iron nanoparticles. Water Res. 2012, 46, 5071–5084. [Google Scholar] [CrossRef]
- NAVFAC; Consultants, G. Emulsified Zero-Valent Iron (EZVI) Treatment of Chlorinated Solvent. Available online: https://clu-in.org/download/techfocus/reduction/patrick-air-force-base.pdf (accessed on 20 March 2019).
- Su, C.; Puls, R.W.; Krug, T.A.; Watling, M.T.; O’Hara, S.K.; Quinn, J.W.; Ruiz, N.E. Travel distance and transformation of injected emulsified zerovalent iron nanoparticles in the subsurface during two and half years. Water Res. 2013, 47, 4095–4106. [Google Scholar] [CrossRef]
- Adusei-Gyamfi, J.; Acha, V. Carriers for nano zerovalent iron (nZVI): Synthesis, application and efficiency. RSC Adv. 2016, 6, 91025–91044. [Google Scholar] [CrossRef]
- Shi, Z.; Fan, D.; Johnson, R.L.; Tratnyek, P.G.; Nurmi, J.T.; Wu, Y.; Williams, K.H. Methods for characterizing the fate and effects of nano zerovalent iron during groundwater remediation. J. Contam. Hydrol. 2015, 181, 17–35. [Google Scholar] [CrossRef]
- Comba, S.; Dalmazzo, D.; Santagata, E.; Sethi, R. Rheological characterization of xanthan suspensions of nanoscale iron for injection in porous media. J. Hazard. Mater. 2011, 185, 598–605. [Google Scholar] [CrossRef]
- Zhong, L.; Oostrom, M.; Truex, M.J.; Vermeul, V.R.; Szecsody, J.E. Rheological behavior of xanthan gum solution related to shear thinning fluid delivery for subsurface remediation. J. Hazard. Mater. 2013, 244–245, 160–170. [Google Scholar] [CrossRef]
- Xue, D.; Sethi, R. Viscoelastic gels of guar and xanthan gum mixtures provide long-term stabilization of iron micro- and nanoparticles. J. Nanoparticle Res. 2012, 14, 1239. [Google Scholar] [CrossRef]
- Crane, R.A.A.; Scott, T.B.B. Nanoscale zero-valent iron: Future prospects for an emerging water treatment technology. J. Hazard. Mater. 2012, 211–212, 112–125. [Google Scholar] [CrossRef]
- Tosco, T.; Petrangeli Papini, M.; Cruz Viggi, C.; Sethi, R. Nanoscale zerovalent iron particles for groundwater remediation: A review. J. Clean. Prod. 2014, 77, 10–21. [Google Scholar] [CrossRef]
- Sun, Y.P.; Li, X.Q.; Zhang, W.X.; Wang, H.P. A method for the preparation of stable dispersion of zero-valent iron nanoparticles. Colloids Surfaces A Physicochem. Eng. Asp. 2007, 308, 60–66. [Google Scholar] [CrossRef]
- Chen, H.; Luo, H.; Lan, Y.; Dong, T.; Hu, B.; Wang, Y. Removal of tetracycline from aqueous solutions using polyvinylpyrrolidone (PVP-K30) modified nanoscale zero valent iron. J. Hazard. Mater. 2011. [Google Scholar] [CrossRef]
- Tian, H.; Liang, Y.; Yang, D.; Sun, Y. Characteristics of PVP–stabilised NZVI and application to dechlorination of soil–sorbed TCE with ionic surfactant. Chemosphere 2020, 239, 124807. [Google Scholar] [CrossRef]
- Sakulchaicharoen, N.; O’Carroll, D.M.; Herrera, J.E. Enhanced stability and dechlorination activity of pre-synthesis stabilized nanoscale FePd particles. J. Contam. Hydrol. 2010, 118, 117–127. [Google Scholar] [CrossRef]
- Cirtiu, C.M.; Raychoudhury, T.; Ghoshal, S.; Moores, A. Systematic comparison of the size, surface characteristics and colloidal stability of zero valent iron nanoparticles pre- and post-grafted with common polymers. Colloids Surfaces A Physicochem. Eng. Asp. 2011, 390, 95–104. [Google Scholar] [CrossRef]
- Sirk, K.M.; Saleh, N.B.; Phenrat, T.; Kim, H.-J.; Dufour, B.; Ok, J.; Golas, P.L.; Matyjaszewski, K.; Lowry, G.V.; Tilton, R.D. Effect of Adsorbed Polyelectrolytes on Nanoscale Zero Valent Iron Particle Attachment to Soil Surface Models. Environ. Sci. Technol. 2009, 43, 3803–3808. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-J.; Phenrat, T.; Tilton, R.D.; Lowry, G.V. Fe 0 Nanoparticles Remain Mobile in Porous Media after Aging Due to Slow Desorption of Polymeric Surface Modifiers. Environ. Sci. Technol. 2009, 43, 3824–3830. [Google Scholar] [CrossRef] [PubMed]
- Saleh, N.; Phenrat, T.; Sirk, K.; Dufour, B.; Ok, J.; Sarbu, T.; Matyjaszewski, K.; Tilton, R.D.; Lowry, G.V. Adsorbed Triblock Copolymers Deliver Reactive Iron Nanoparticles to the Oil/Water Interface. Nano Lett. 2005, 5, 2489–2494. [Google Scholar] [CrossRef]
- Phenrat, T.; Cihan, A.; Kim, H.J.; Mital, M.; Illangasekare, T.; Lowry, G.V. Transport and deposition of polymer-modified Fe0 nanoparticles in 2-D heterogeneous porous media: Effects of particle concentration, Fe 0 content, and coatings. Environ. Sci. Technol. 2010, 44, 9086–9093. [Google Scholar] [CrossRef]
- He, F.; Zhao, D.; Paul, C. Field assessment of carboxymethyl cellulose stabilized iron nanoparticles for in situ destruction of chlorinated solvents in source zones. Water Res. 2010, 44, 2360–2370. [Google Scholar] [CrossRef]
- Hou, S.; Wu, B.; Peng, D.; Wang, Z.; Wang, Y.; Xu, H. Remediation performance and mechanism of hexavalent chromium in alkaline soil using multi-layer loaded nano-zero-valent iron. Environ. Pollut. 2019, 252, 553–561. [Google Scholar] [CrossRef]
- Comba, S.; Sethi, R. Stabilization of highly concentrated suspensions of iron nanoparticles using shear-thinning gels of xanthan gum. Water Res. 2009, 43, 3717–3726. [Google Scholar] [CrossRef]
- Hydutsky, B.W.; Mack, E.J.; Beckerman, B.B.; Skluzacek, J.M.; Mallouk, T.E. Optimization of Nano- and Microiron Transport through Sand Columns Using Polyelectrolyte Mixtures. Environ. Sci. Technol. 2007, 41, 6418–6424. [Google Scholar] [CrossRef]
- Phenrat, T.; Long, T.C.; Lowry, G.V.; Veronesi, B. Partial Oxidation (“Aging”) and Surface Modification Decrease the Toxicity of Nanosized Zerovalent Iron. Environ. Sci. Technol. 2009, 43, 195–200. [Google Scholar] [CrossRef]
- Liang, B.; Xie, Y.; Fang, Z.; Tsang, E.P. Assessment of the transport of polyvinylpyrrolidone-stabilised zero-valent iron nanoparticles in a silica sand medium. J. Nanoparticle Res. 2014, 16, 2485. [Google Scholar] [CrossRef]
- He, F.; Zhao, D.; Liu, J.; Roberts, C.B. Stabilization of Fe−Pd Nanoparticles with Sodium Carboxymethyl Cellulose for Enhanced Transport and Dechlorination of Trichloroethylene in Soil and Groundwater. Ind. Eng. Chem. Res. 2007, 46, 29–34. [Google Scholar] [CrossRef]
- He, F.; Zhang, M.; Qian, T.; Zhao, D. Transport of carboxymethyl cellulose stabilized iron nanoparticles in porous media: Column experiments and modeling. J. Colloid Interface Sci. 2009, 334, 96–102. [Google Scholar] [CrossRef] [PubMed]
- Tiraferri, A.; Chen, K.L.; Sethi, R.; Elimelech, M. Reduced aggregation and sedimentation of zero-valent iron nanoparticles in the presence of guar gum. J. Colloid Interface Sci. 2008, 324, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Bardos, P.; Bone, B.; Daly, P.; Elliott, D.; Jones, S.; Lowry, G.; Merly, C. A Risk/Benefit Appraisal for the Application of Nano-Scale Zero Valent Iron (nZVI) for the Remediation of Contaminated Sites. NanoRem 2015, WP9, 1–76. [Google Scholar]
- Patil, S.S.; Shedbalkar, U.U.; Truskewycz, A.; Chopade, B.A.; Ball, A.S. Nanoparticles for environmental clean-up: A review of potential risks and emerging solutions. Environ. Technol. Innov. 2016, 5, 10–21. [Google Scholar] [CrossRef]
- Bardos, P.; Merly, C.; Kvapil, P.; Koschitzky, H.-P. Status of nanoremediation and its potential for future deployment: Risk-benefit and benchmarking appraisals. Remediat. J. 2018, 28, 43–56. [Google Scholar] [CrossRef]
- Qian, Y.; Qin, C.; Chen, M.; Lin, S. Nanotechnology in soil remediation—Applications vs. implications. Ecotoxicol. Environ. Saf. 2020, 201, 110815. [Google Scholar] [CrossRef]
- Latif, A.; Sheng, D.; Sun, K.; Si, Y.; Azeem, M.; Abbas, A.; Bilal, M. Remediation of heavy metals polluted environment using Fe-based nanoparticles: Mechanisms, influencing factors, and environmental implications. Environ. Pollut. 2020, 264, 114728. [Google Scholar] [CrossRef]
- Otero-González, L.; García-Saucedo, C.; Field, J.A.; Sierra-Álvarez, R. Toxicity of TiO2, ZrO2, Fe0, Fe2O3, and Mn2O3 nanoparticles to the yeast, Saccharomyces cerevisiae. Chemosphere 2013, 93, 1201–1206. [Google Scholar] [CrossRef]
- Chaithawiwat, K.; Vangnai, A.; McEvoy, J.M.; Pruess, B.; Krajangpan, S.; Khan, E. Impact of nanoscale zero valent iron on bacteria is growth phase dependent. Chemosphere 2016, 144, 352–359. [Google Scholar] [CrossRef] [PubMed]
- Fajardo, C.; Saccà, M.L.; Martinez-Gomariz, M.; Costa, G.; Nande, M.; Martin, M. Transcriptional and proteomic stress responses of a soil bacterium Bacillus cereus to nanosized zero-valent iron (nZVI) particles. Chemosphere 2013, 93, 1077–1083. [Google Scholar] [CrossRef] [PubMed]
- Lacalle, R.G.; Garbisu, C.; Becerril, J.M. Effects of the application of an organic amendment and nanoscale zero-valent iron particles on soil Cr(VI) remediation. Environ. Sci. Pollut. Res. 2020, 27, 31726–31736. [Google Scholar] [CrossRef] [PubMed]
- Saccà, M.L.; Fajardo, C.; Costa, G.; Lobo, C.; Nande, M.; Martin, M. Integrating classical and molecular approaches to evaluate the impact of nanosized zero-valent iron (nZVI) on soil organisms. Chemosphere 2014, 104, 184–189. [Google Scholar] [CrossRef] [PubMed]
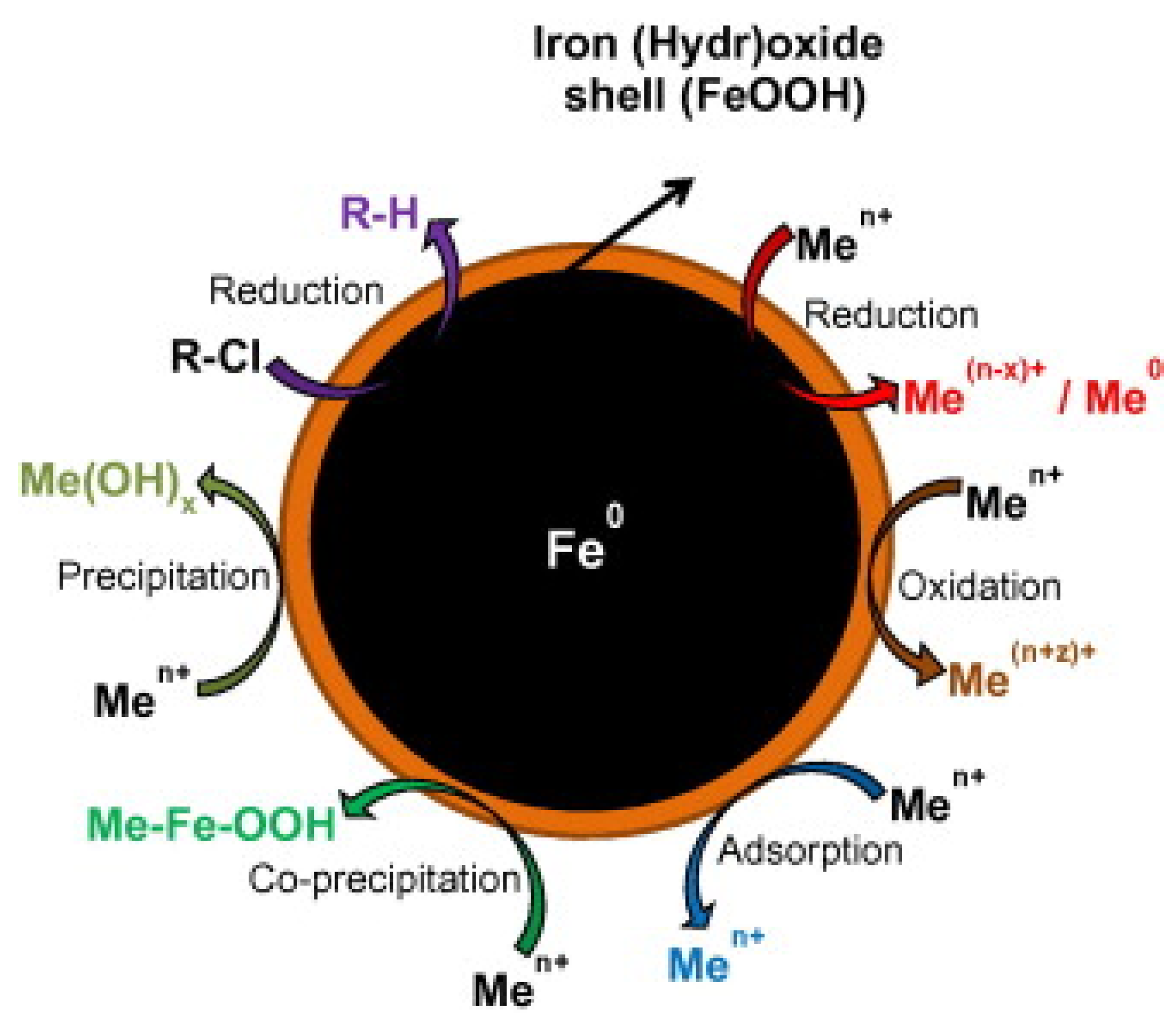
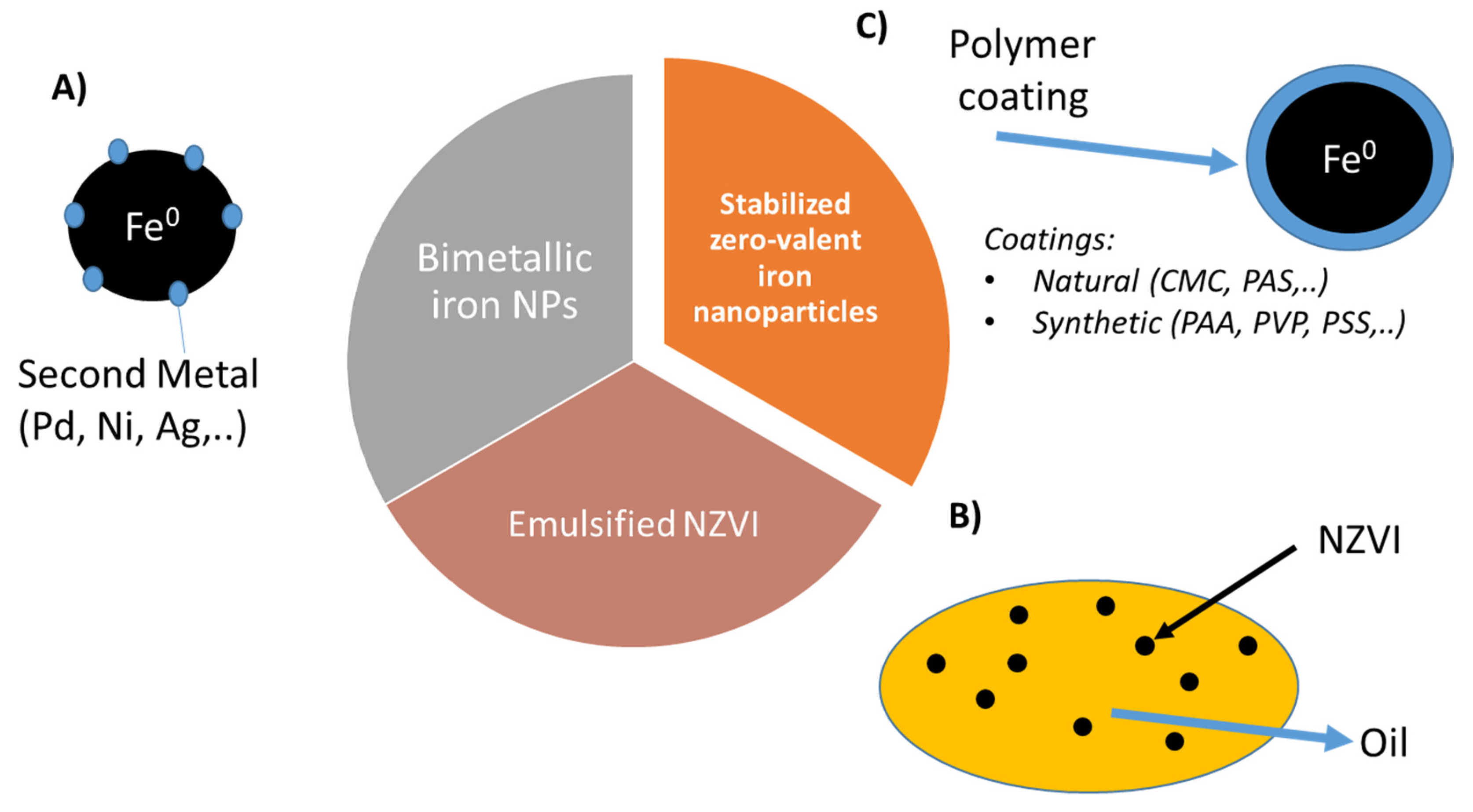
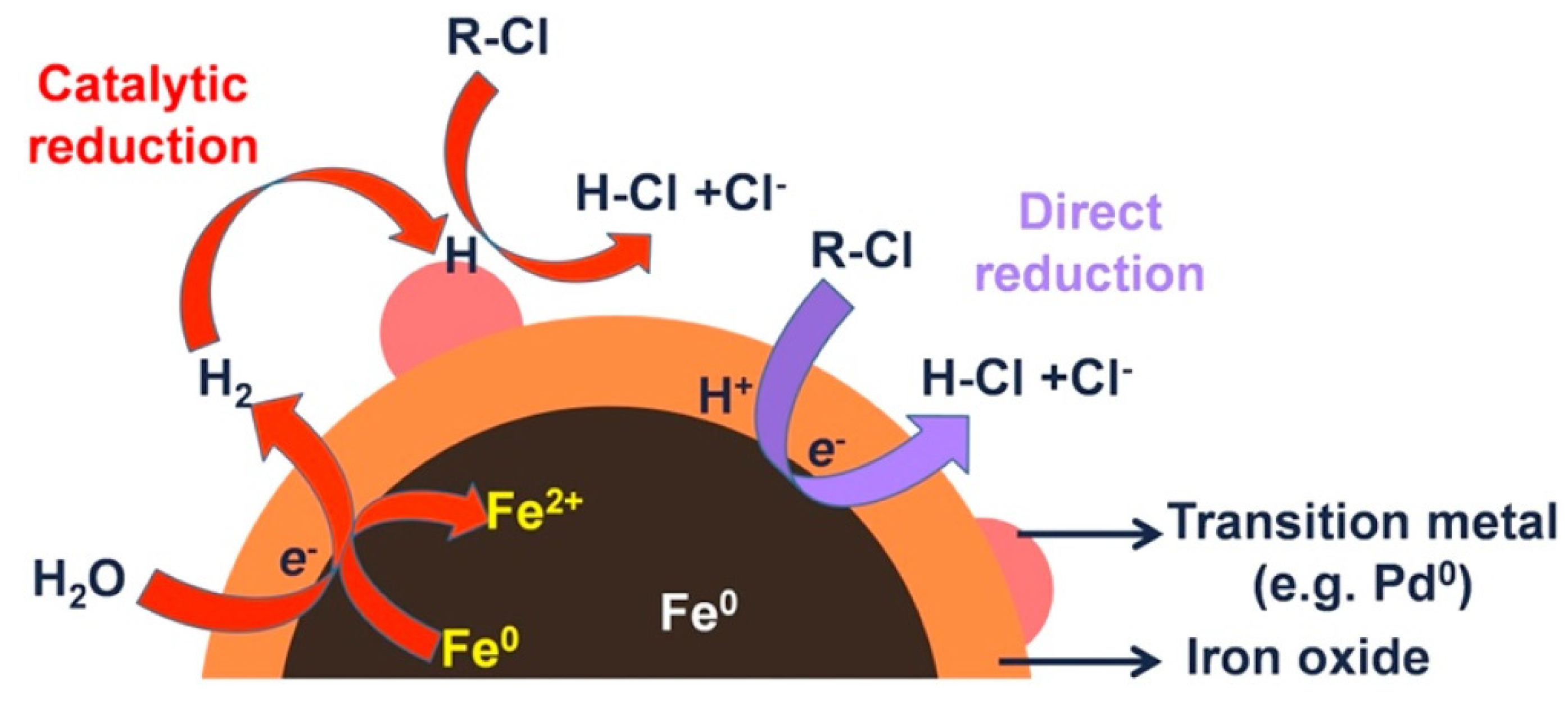
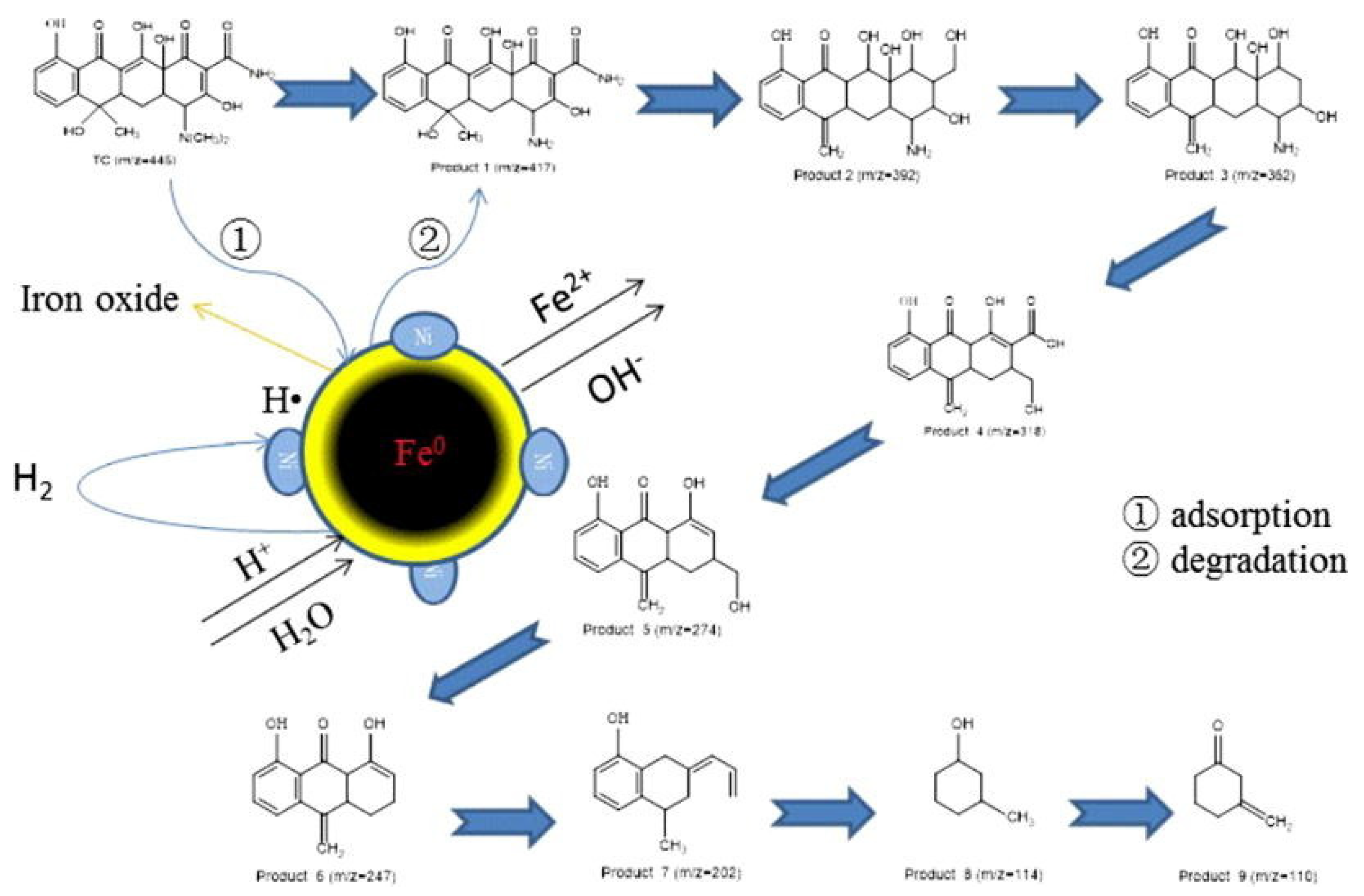
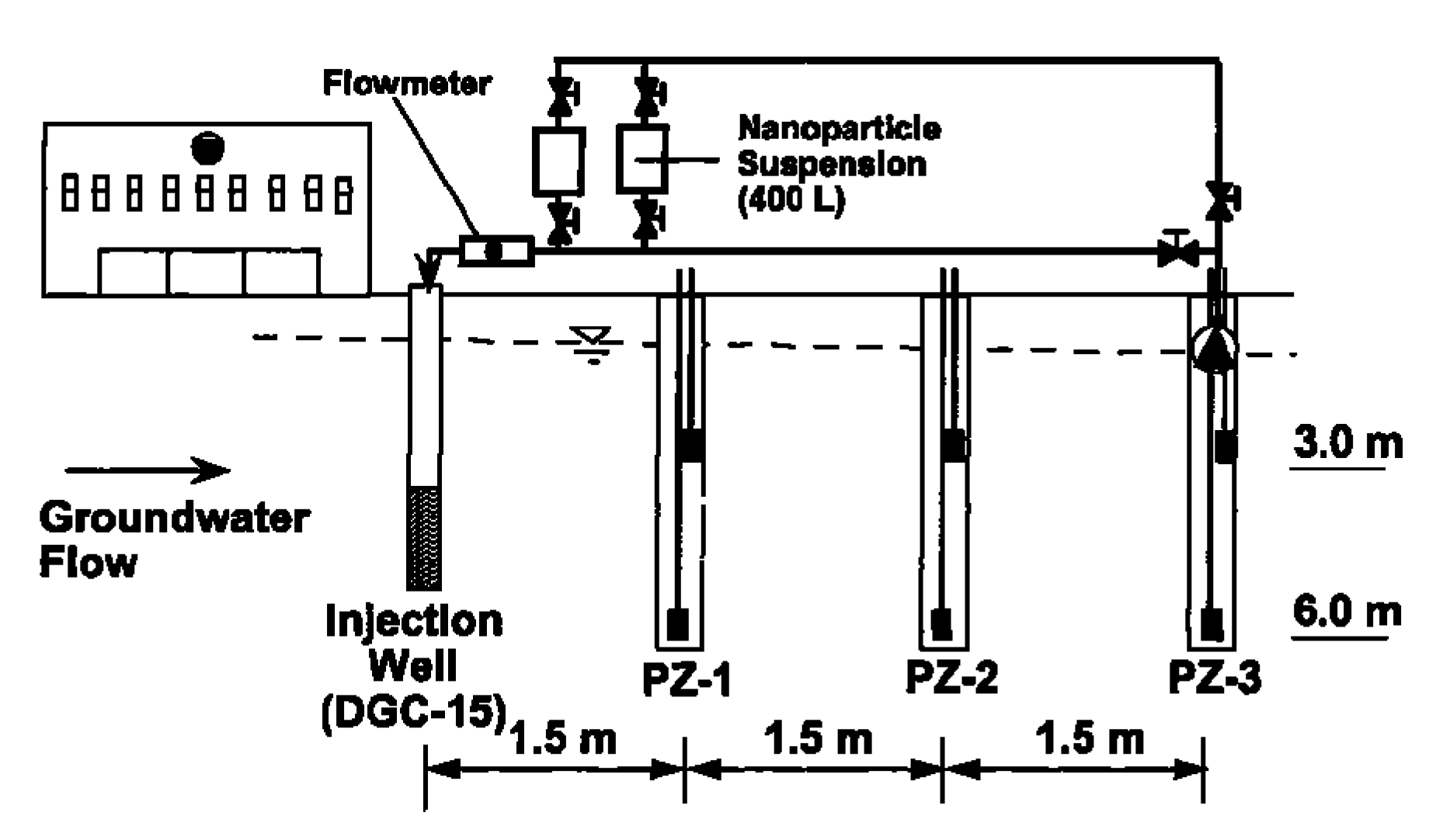
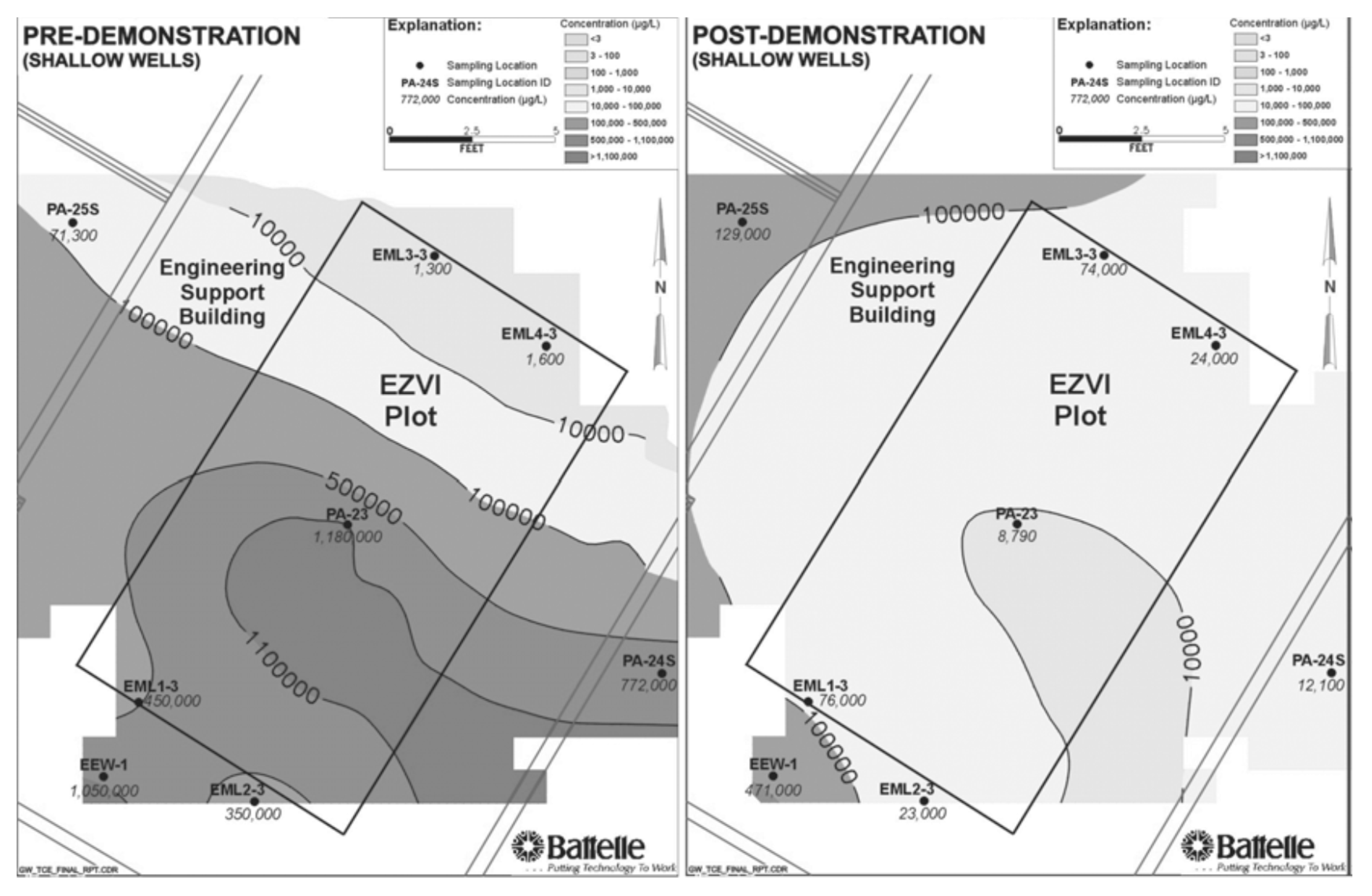
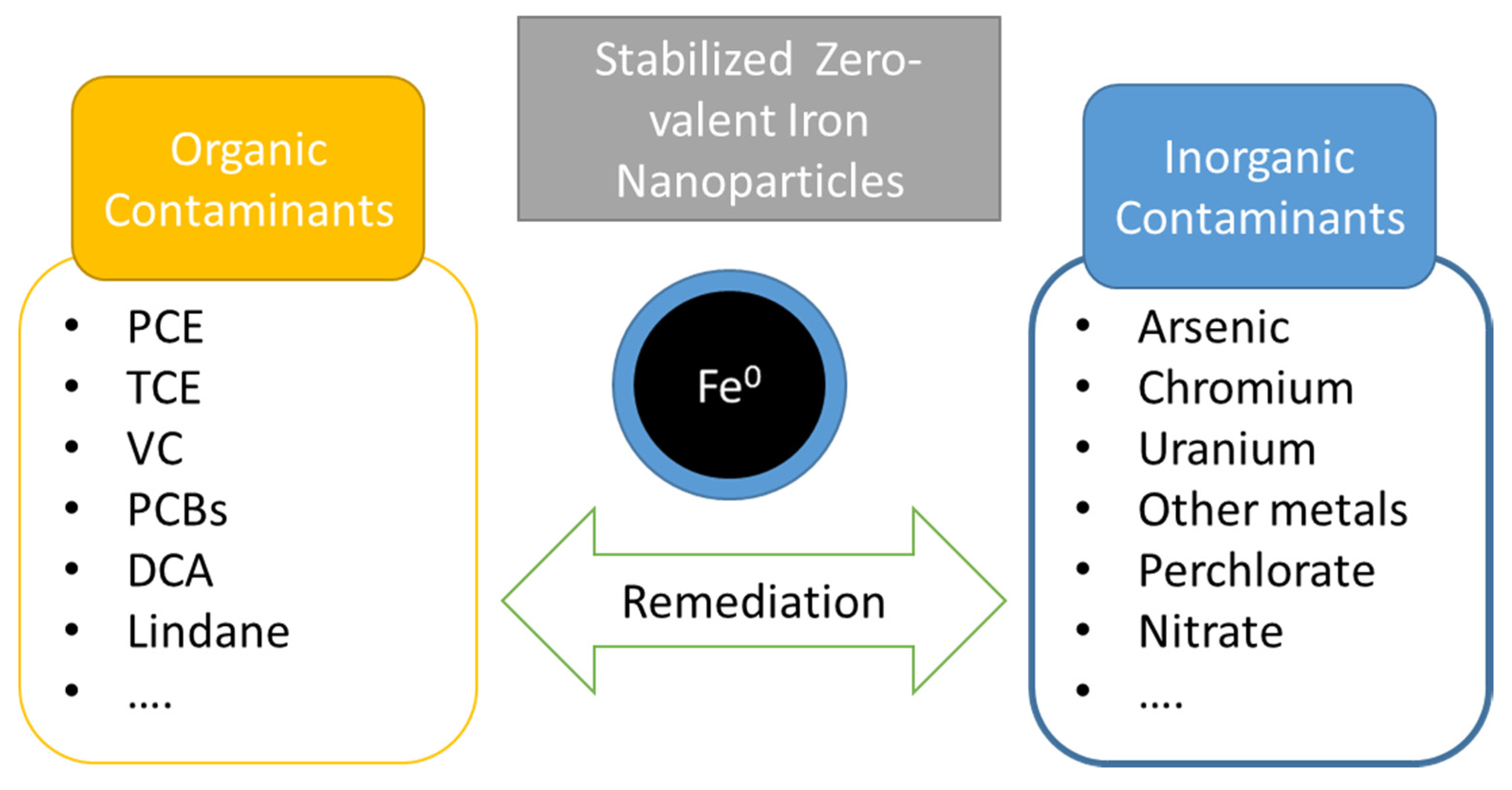
| Pollutants | NZVI Based Treatment | Reference |
|---|---|---|
| Heavy metals and metalloids | ||
| As | NZVI | [40,41,42] |
| Pb + Zn | NZVI | [43] |
| Pb | NZVI + citric acid | [44] |
| Cr(VI) | NZVI/Cu; NZVI/biochar; starch stabilized NZVI | [45,46,47,48] |
| Pb + Cd + Cr | NZVI + activated carbon | [48] |
| Sb(V) | NZVI + humic acid coated; NZVI | [49] |
| U | NZVI | [50] |
| Hg | NZVI | [51] |
| Ni | NZVI | [52] |
| Ag | NZVI | [52] |
| Cd | NZVI | [51] |
| Other inorganic compounds | ||
| Perchlorate | NZVI | [51] |
| Nitrate | NZVI | [51] |
| Organic compounds (pesticides, polychlorinated hydrocarbons, chlorobenzenes, colouring agents, etc.) | ||
| PAHs | NZVI; NZVI + SDS | [42,53,54,55] |
| TCE | CMC-NZVI | [56] |
| TCE + PCB | Starch-stabilized Fe/Pd | [57] |
| Ibuprofen | NZVI | [58] |
| DDT | NZVI | [59,60] |
| TNT | NZVI | [61] |
| 2,3,7,8-TCDD | NZVI + Tween 80 | [62] |
| TCC | NZVI | [51] |
| Bromoform | NZVI | [51] |
| Chloroform | NZVI | [51] |
| Dibromochloromethane | NZVI | [51] |
| Dichloromethane | NZVI | [51] |
| Dichlorobromomethane | NZVI | [51] |
| Chloromethane | NZVI | [51] |
| N-nitrosodimethylamine | NZVI | [52] |
| Hexachlorobencene | NZVI | [52] |
| PCB | NZVI + saponin | [63] |
| DDT | NZVI | [52] |
| Lindane | NZVI | [64,65] |
| Pentachlorophenol | NZVI | [52] |
| Chrysoidin | NZVI | [51] |
| Tropaeolin O | NZVI | [51] |
| Components | Contaminant | Reference |
|---|---|---|
| Fe/Pd | Chlorobenzenes | [75] |
| Fe/Pd | Polychlorinated biphenyls (PCB) | [76,77] |
| Fe/Pd and Fe/Ni | Chlorinated aliphatics (PCE, TCE, VC, …) | [28,59,78,79] |
| Fe/Pd and Fe/Ni | Polybrominated diphenyl ethers (PBDEs) | [80,81] |
| Fe/Cu, Fe/Al and Fe/Ni | Chromium, Copper | [45,82,83] |
| Fe/Pd, Fe/Zn and Fe/Ni | Dyes (Orange G, Congo red, Orange II, …) | [84,85,86,87,88] |
| Fe/Ni | Tetracycline | [89] |
| Fe/Ni | Triclosan | [82] |
| Pollutants | Concentration Decrease | Addition Method | Site | Comments | Time for Study | Reference |
|---|---|---|---|---|---|---|
| TCE | 96% | Gravity-fed | Groundwater | High dosage of BNPs needed | n/a | [99] |
| PCE, TCE; Others | n/a | n/a | Soil and groundwater | n/a | n/a | [98] |
| VOCs | 74% | n/a | n/a | n/a | Six months | [98] |
| VOCs (ethane, TCE, TCA, DCE, DCA) | 65–96% | Gravity-fed and recirculate | Groundwater | Pd/Fe BNPs | 1 year | [101] |
| VC | 50–99% | Injection | Groundwater | Pd/Fe BNPs | Six months | [102] |
| Pollutants | Concentration Decrease | Addition Method | Site | Comments | Location | Reference |
|---|---|---|---|---|---|---|
| TCE, TCA, DCE, DCA | TCE and TCA > 65–96% | Gravity-fed and recirculate | groundwater | Polymer coated nanoparticles | Naval Air Station Jacksonville (FL, USA) | [101] |
| PCE, TCE | PCE > 85% TCE > 85% | Pneumatic injection, direct injection | groundwater | Uncertainties in the estimations | Parrick Island (SC, USA) | [109] |
| TCE | TCE > 80 (DNAPL) TCE > 60% (groundwater) | n/a | DNAPL, groundwater | n/a | Cape Canaveral Air Force Station (FL, USA) | [105] |
| TCE | TCE > 95% | n/a | n/a | n/a | Patrick Air Force Base (FL, USA) | [110] |
| Chlorinated VOCs | >86% | Pneumatic injection | Soil and groundwater | 2.5 years monitoring | Marine Corps Recruit Depo. Parris Island (SC, USA) | [111] |
| Coated Polymer | Improvement | Contaminant | Reference |
|---|---|---|---|
| Synthetic Polymers | |||
| PAA | Transportability | TCE, Lindane | [64,66] |
| PV3A | Stability | TCE | [119] |
| PEG | Stability | Lindane | [65] |
| PTHF | Stability | Lindane | [65] |
| PVP | Stability | TC, TCE | [120,121,122] |
| PSS | Stability | n/a | [123,124,125] |
| PAM | Stability | n/a | [123] |
| PMAA-b-PMMA-b-PSS | Stability | TCE | [124,126] |
| OMA | Transportability | n/a | [127] |
| Natural Polymers | |||
| CMC | Stability, Transportability | TCE, PCB, Lindane, Cr(VI) | [64,100,122,125,128,129] |
| PAS | Stability | Lindane | [64,125] |
| XG | Stability, Transportability | n/a | [116,130] |
| GG | Stability, Reactivity | TCE | [116,122] |
| Pollutants | Conc. Decrease | Addition Method | Site | Comments | Location | Reference |
|---|---|---|---|---|---|---|
| Chlorinated compounds | >90% | Injection in two phases | 30 days | Hamilton Township, New Jersey (USA) | [32] | |
| TCA, DCE, TCE, PCE | 80–90% | n/a | Soil | n/a | Naval Air Engineering Station of Lakehurst (USA) | [98] |
| TCA, DCE, TCE, PCE | 80–90% | n/a | Soil | n/a | Naval Air Station of Jacksonville (USA) | [98] |
| PCE | 90% | n/a | Soil | 2 years after, more reduction | Bornheim, Germany (Europe) | [137] |
| PCE, TCE, DCE | 60–75% for Horice and 90% for Pisecna | Injection (82 injection wells) | Soil | n/a | Czech Republic (Horice and Pisecna) | [137] |
| Chlorinated compounds | >90% | n/a | n/a | 30 days | Hamilton Township, New Jersey (USA) | [32] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Galdames, A.; Ruiz-Rubio, L.; Orueta, M.; Sánchez-Arzalluz, M.; Vilas-Vilela, J.L. Zero-Valent Iron Nanoparticles for Soil and Groundwater Remediation. Int. J. Environ. Res. Public Health 2020, 17, 5817. https://doi.org/10.3390/ijerph17165817
Galdames A, Ruiz-Rubio L, Orueta M, Sánchez-Arzalluz M, Vilas-Vilela JL. Zero-Valent Iron Nanoparticles for Soil and Groundwater Remediation. International Journal of Environmental Research and Public Health. 2020; 17(16):5817. https://doi.org/10.3390/ijerph17165817
Chicago/Turabian StyleGaldames, Alazne, Leire Ruiz-Rubio, Maider Orueta, Miguel Sánchez-Arzalluz, and José Luis Vilas-Vilela. 2020. "Zero-Valent Iron Nanoparticles for Soil and Groundwater Remediation" International Journal of Environmental Research and Public Health 17, no. 16: 5817. https://doi.org/10.3390/ijerph17165817
APA StyleGaldames, A., Ruiz-Rubio, L., Orueta, M., Sánchez-Arzalluz, M., & Vilas-Vilela, J. L. (2020). Zero-Valent Iron Nanoparticles for Soil and Groundwater Remediation. International Journal of Environmental Research and Public Health, 17(16), 5817. https://doi.org/10.3390/ijerph17165817






