Influence of Age on Partial Clinical Remission among Children with Newly Diagnosed Type 1 Diabetes
Abstract
1. Introduction
Aim of the Study
2. Material and Methods
2.1. Definition of Partial Clinical Remission
2.2. Statistical Analysis
3. Results
3.1. Partial Clinical Remission Occurrence and Associated Variables at T1D Diagnosis
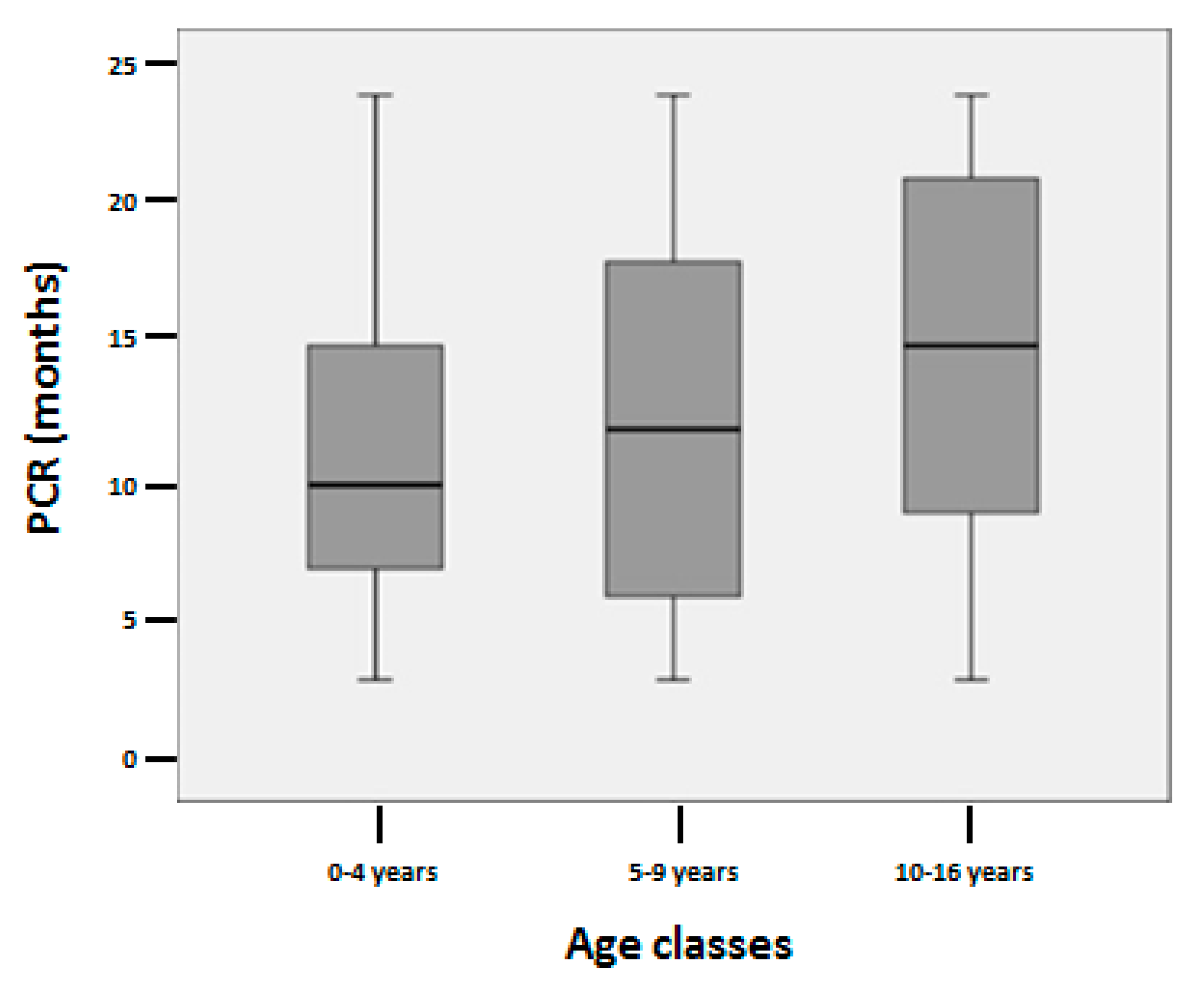
3.2. Partial Clinical Remission Duration
3.3. Anthropometric Parameters, HbA1c and Total Daily Insulin Dose at 1 and 2 Years after T1D Onset
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- American Diabetes Association. 2. Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes-2018. Diabetes Care 2018, 41, 13–27. [Google Scholar] [CrossRef]
- Pyziak, A.; Zmyslowska, A.; Bobeff, K.; Malachowska, B.; Fendler, W.; Wyka, K.; Baranowska-Jazwiecka, A.; Szymanska, M.; Szadkowska, A.; Mlynarski, W. Markers influencing the presence of partial clinical remission in patients with newly diagnosed type 1 diabetes. J. Pediatr. Endocrinol. Metab. 2017, 30, 1147–1153. [Google Scholar] [CrossRef] [PubMed]
- Lombardo, F.; Valenzise, M.; Wasniewska, M.; Messina, M.F.; Ruggeri, C.; Arrigo, T.; De Luca, F. Two-year prospective evaluation of the factors affecting honeymoon frequency and duration in children with insulin dependent diabetes mellitus: The key-role of age at diagnosis. Diabetes Nutr. Metab. 2002, 15, 246–251. [Google Scholar] [PubMed]
- Büyükgebiz, A.; Cemeroglu, A.P.; Böber, E.; Mohn, A.; Chiarelli, F. Factors Influencing Remission Phase in Children with Type 1 Diabetes Mellitus. J. Pediatr. Endocrinol. Metab. 2001, 14, 1585–1596. [Google Scholar] [CrossRef] [PubMed]
- Agner, Τ.; Damm, Ρ.; Binder, C. Remission in IDDM: Prospective study of basal C-peptide and insulin dose in 268 consecutive patients. Diabetes Care 1987, 10, 164–167. [Google Scholar] [CrossRef]
- Muhammad, B.J.; Swift, P.G.; Raymond, N.T.; Botha, J.L. Partial remission phase of diabetes in children younger than age 10 years. Arch. Dis. Child 1999, 80, 367–369. [Google Scholar] [CrossRef]
- Kordonouri, O.; Danne, T.; Enders, I.; Weber, B. Does the long-term clinical course of type 1 diabetes mellitus differ in patients with prepubertal and pubertal onset? Results of the Berlin Retinopathy Study. Eur. J. Pediatr. 1998, 157, 202–207. [Google Scholar] [CrossRef]
- Komulainen, J.; Lounamaa, R.; Knip, M.; Kaprio, E.A.; Akerblom, H.K. Ketoacidosis at the diagnosis of type 1 (insulin dependent) diabetes mellitus is related to poor residual beta cell function. Childhood Diabetes in Finland Study Group. Arch. Dis. Child 1996, 75, 410–415. [Google Scholar] [CrossRef]
- Ortqvist, E.; Falorni, A.; Scheynius, A.; Persson, B.; Lernmark, A. Age governs gender-dependent islet cell autoreactivity and predicts the clinical course in childhood IDDM. Acta Paediatr. 1997, 86, 1166–1171. [Google Scholar] [CrossRef]
- Couper, J.; Donaghue, K. International Society for Pediatric and Adolescent Diabetes. Phases of diabetes. Pediatr. Diabetes 2007, 8, 44–47. [Google Scholar] [CrossRef]
- Mortensen, H.B.; Hougaard, P.; Swift, P.; Hansen, L.; Holl, R.W.; Hoey, H.; Bjoerndalen, H.; de Beaufort, C.; Chiarelli, F.; Danne, T.; et al. New definition for the partial remission period in children and adolescents with type 1 diabetes. Diabetes Care 2009, 32, 1384–1390. [Google Scholar] [CrossRef] [PubMed]
- Max Andersen, M.L.C.; Hougaard, P.; Pörksen, S.; Nielsen, L.B.; Fredheim, S.; Svensson, J.; Thomsen, J.; Vikre-Jørgensen, J.; Hertel, T.; Petersen, J.S.; et al. Partial remission definition: Validation based on the insulin dose-adjusted HbA1c (IDAA1C) in 129 Danish children with new-onset type 1 diabetes. Pediatr. Diabetes 2014, 15, 469–476. [Google Scholar] [CrossRef] [PubMed]
- Visalli, N.; Cavallo, M.G.; Signore, A.; Baroni, M.G.; Buzzetti, R.; Fioriti, E.; Mesturino, C.; Fiori, R.; Lucentini, L.; Matteoli, M.C.; et al. A multi-centre randomized trial of two different doses of nicotinamide in patients with recent onset type 1 diabetes (the IMDIAB VI). Diabetes Metab. Res. Rev. 1999, 15, 181–185. [Google Scholar] [CrossRef]
- Passanisi, S.; Timpanaro, T.; Lo Presti, D.; Caruso-Nicoletti, M. From nephew to uncle: Diagnosis of a rare form of diabetes mellitus. Giornale Italiano Diabetologia Metabolismo 2013, 33, 147–149. [Google Scholar]
- Sørensen, J.S.; Johannesen, J.; Pociot, F.; Kristensen, K.; Thomsen, J.; Hertel, N.T.; Kjaersgaard, P.; Brorsson, C.; Birkebaek, N.H. Danish Society for Diabetes in Childhood and Adolescence. Residual β-Cell function 3–6 years after onset of type 1 diabetes reduces risk of severe hypoglycemia in children and adolescents. Diabetes Care 2013, 36, 3454–3459. [Google Scholar] [CrossRef]
- Hypoglycemia in the Diabetes Control and Complications Trial. The Diabetes Control and Complications Trial Research Group. Diabetes 1997, 46, 271–286. [Google Scholar] [CrossRef]
- Niedzwiecki, P.; Pilacinski, S.; Uruska, A.; Adamska, A.; Naskret, D.; Zozulinska-Ziolkiewicz, D. Influence of remission and its duration on development of early microvascular complications in young adults with type 1 diabetes. J. Diabetes Complicat. 2015, 29, 1105–1111. [Google Scholar] [CrossRef]
- The absence of a glycemic threshold for the development of long-term complications: The perspective of the Diabetes Control and Complications Trial. Diabetes 1996, 45, 1289–1298. [CrossRef]
- Ludvigsson, J. Combination therapy for preservation of beta cell function in Type 1 diabetes: New attitudes and strategies are needed! Immunol. Lett. 2014, 159, 30–35. [Google Scholar] [CrossRef]
- Schloot, N.C.; Hanifi-Moghaddam, P.; Aabenhus-Andersen, N.; Alizadeh, B.Z.; Saha, M.T.; Knip, M.; Devendra, D.; Wilkin, T.; Bonifacio, E.; Roep, B.O.; et al. Association of immune mediators at diagnosis of type 1 diabetes with later clinical remission. Diabet. Med. 2007, 24, 512–520. [Google Scholar] [CrossRef]
- Abdul-Rasoul, M.; Habib, H.; Al-Khouly, M. The honeymoon phase in children with type 1 diabetes mellitus: Frequency, duration, and influential factors. Pediatr. Diabetes 2006, 7, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Moole, H.; Moole, V.; Mamidipalli, A.; Dharmapuri, S.; Boddireddy, R.; Taneja, D.; Sfeir, H.; Gajula, S. Spontaneous complete remission of type 1 diabetes mellitus in an adult—Review and case report. J. Community Hosp. Intern. Med. Perspect. 2015, 5, 28709. [Google Scholar] [CrossRef] [PubMed]
- Mayer-Davis, E.J.; Kahkoska, A.R.; Jefferies, C.; Dabelea, D.; Balde, N.; Gong, C.X.; Aschner, P.; Craig, M.E. ISPAD Clinical Practice Consensus Guidelines 2018: Definition, epidemiology, and classification of diabetes in children and adolescents. Pediatr. Diabetes 2018, 19 (Suppl. 27), 7–19. [Google Scholar] [CrossRef]
- Wolfsdorf, J.I.; Glaser, N.; Agus, M.; Fritsch, M.; Hanas, R.; Rewers, A.; Sperling, M.A.; Codner, E. ISPAD Clinical Practice Consensus Guidelines 2018: Diabetic ketoacidosis and the hyperglycemic hyperosmolar state. Pediatr. Diabetes 2018, 19 (Suppl. 27), 155–177. [Google Scholar] [CrossRef] [PubMed]
- Nagl, K.; Hermann, J.M.; Plamper, M.; Schröder, C.; Dost, A.; Kordonouri, O.; Rami-Merhar, B.; Holl, R.W. Factors contributing to partial remission in type 1 diabetes: Analysis based on the insulin dose-adjusted HbA1c in 3657 children and adolescents from Germany and Austria. Pediatr. Diabetes 2017, 18, 428–434. [Google Scholar] [CrossRef]
- Lundberg, R.L.; Marino, K.R.; Jasrotia, A.; Maranda, L.S.; Barton, B.A.; Alonso, L.C.; Nwosu, B.U. Partial clinical remission in type 1 diabetes: A comparison of the accuracy of total daily dose of insulin of <0.3 units/kg/day to the gold standard insulin-dose adjusted hemoglobin A1c of ≤9 for the detection of partial clinical remission. J. Pediatr. Endocrinol. Metab. 2017, 30, 823–830. [Google Scholar] [CrossRef] [PubMed]
- Marino, K.R.; Lundberg, R.L.; Jasrotia, A.; Maranda, L.S.; Thompson, M.J.; Barton, B.A.; Alonso, L.C.; Nwosu, B.U. A predictive model for lack of partial clinical remission in new-onset pediatric type 1 diabetes. PLoS ONE 2017, 12, e0176860. [Google Scholar] [CrossRef]
- Nwosu, B.U.; Zhang, B.; Ayyoub, S.S.; Choi, S.; Villalobos-Ortiz, T.R.; Alonso, L.C.; Barton, B.A. Children with type 1 diabetes who experienced a honeymoon phase had significantly lower LDL cholesterol 5 years after diagnosis. PLoS ONE 2018, 13, e0196912. [Google Scholar] [CrossRef]
- Chobot, A.; Stompór, J.; Szyda, K.; Sokołowska, M.; Deja, G.; Polańska, J.; Jarosz-Chobot, P. Remission phase in children diagnosed with type 1 diabetes in years 2012 to 2013 in Silesia, Poland: An observational study. Pediatr. Diabetes 2019, 20, 286–292. [Google Scholar] [CrossRef]
- Chiavaroli, V.; Derraik, J.G.B.; Jalaludin, M.Y.; Albert, B.B.; Ramkumar, S.; Cutfield, W.S.; Hofman, P.L.; Jefferies, C.A. Partial remission in type 1 diabetes and associated factors: Analysis based on the insulin dose-adjusted hemoglobin A1c in children and adolescents from a regional diabetes center, Auckland, New Zealand. Pediatr. Diabetes 2019, 20, 892–900. [Google Scholar] [CrossRef]
- Fonolleda, M.; Murillo, M.; Vázquez, F.; Bel, J.; Vives-Pi, M. Remission phase in paediatric type 1 diabetes: New understanding and emerging biomarkers. Horm. Res. Paediatr. 2017, 88, 307–315. [Google Scholar] [CrossRef] [PubMed]
- Wilkin, T.J. The accelerator hypothesis: Weight gain as the missing link between Type I and Type II diabetes. Diabetologia 2001, 44, 914–922. [Google Scholar] [CrossRef] [PubMed]
- Bowden, S.A.; Duck, M.M.; Hoffman, R.P. Young children (<5 yr) and adolescents (>12 yr) with type 1 diabetes mellitus have low rate of partial remission: Diabetic ketoacidosis is an important risk factor. Pediatr. Diabetes 2008, 9, 197–201. [Google Scholar] [CrossRef] [PubMed]
- Bowden, S.A. Partial Remission (honeymoon phase) in Type 1 Diabetes Mellitus. In Frontiers in Clinical Drug Research—Diabetes and Obesity; The Ohio State University: Columbus, OH, USA, 2017; Volume 4, pp. 1–20. [Google Scholar]
- Mortensen, H.B.; Swift, P.G.F.; Holl, R.W.; Hougaard, P.; Hansen, L.; Bjoerndalen, H.; de Beaufort, C.E.; Knip, M. Hvidoere Study Group on Childhood Diabetes. Multinational study in children and adolescents with newly diagnosed type 1 diabetes: Association of age, ketoacidosis, HLA status, and autoantibodies on residual beta-cell function and glycemic control 12 months after diagnosis. Pediatr. Diabetes 2010, 11, 218–226. [Google Scholar] [PubMed]
- Atkinson, M.A.; von Herrath, M.; Powers, A.C.; Clare-Salzler, M. Current concepts on the pathogenesis of type 1 diabetes--considerations for attempts to prevent and reverse the disease. Diabetes Care 2015, 38, 979–988. [Google Scholar] [CrossRef] [PubMed]
- Kara, Ö.; Esen, İ.; Tepe, D. Factors influencing frequency and duration of remission in children and adolescents newly diagnosed with type 1 diabetes. Med. Sci. Monit. 2018, 24, 5996–6001. [Google Scholar] [CrossRef]
- Rosenbloom, A.L. The variable but inevitable loss of beta cells in overt type 1 diabetes. Pediatr. Diabetes 2003, 4, 1–3. [Google Scholar] [CrossRef]
- Camilo, D.S.; Pradella, F.; Paulino, M.F.; Baracat, E.C.E.; Marini, S.H.; Guerra, G., Jr.; Pavin, E.J.; Parisi, C.; Longhini, A.L.F.; Marques, S.B. Partial remission in Brazilian children and adolescents with type 1 diabetes. Association with a haplotype of class II human leukocyte antigen and synthesis of autoantibodies. Pediatr. Diabetes 2020, 21, 606–614. [Google Scholar] [CrossRef]
- Pozzilli, P.; Manfrini, S.; Buzzetti, R.; Lampeter, E.; Leeuw, I.D.; Iafusco, D.; Prisco, M.; Ionescu-Tirgoviste, C.; Kolouskovà, S.; Linn, T. Glucose evaluation trial for remission (GETREM) in type 1 diabetes: A European multicentre study. Diabetes Res. Clin. Pract. 2005, 68, 258–264. [Google Scholar] [CrossRef]
- Ozen, G.; Zanfardino, A.; Confetto, S.; Piscopo, A.; Casaburo, F.; Tinto, N.; Iafusco, F.; Ozen, G.; Miraglia del Giudice, E.; Aysin Tasar, M.; et al. The Association of Autoimmune Diseases with Type 1 Diabetes Mellitus in Children Depends Also by the Length of Partial Clinical Remission Phase (Honeymoon). Int. J. Endocrinol. 2020, 2020, 2630827. [Google Scholar] [CrossRef]
- Bonfanti, R.; Bognetti, E.; Meschi, F.; Bruneiii, A.; Riva, M.C.; Pastore, M.R.; Calori, G.; Chiumello, G. Residual beta-cell function and spontaneous clinical remission in type 1 diabetes mellitus: The role of puberty. Acta Diabetol. 1998, 35, 91–95. [Google Scholar] [CrossRef] [PubMed]
| Variables | Percentages and Mean ± SDS | Median (IQR) |
|---|---|---|
| Gender | ||
| Male | 52.70% | |
| Female | 47.30% | |
| Age at the onset (years) | 8.6 ± 3.8 | 8.8 (5.9; 11.5) |
| Age classes at the onset | ||
| 0–4 years | 21% | |
| 5–9 years | 41.30% | |
| 10–16 years | 37.70% | |
| BMI at the onset (SDS) | −0.08 ± 1.21 | −0.04 (−0.91; 0.79) |
| pH | 7.28 ± 0.14 | 7.31 (7.22; 7.37) |
| Presence of DKA | ||
| Yes | 49% | |
| No | 51% | |
| HbA1c at the onset (%) | 10.7 ± 1.9 | 10.4 (9.5; 12.1) |
| HbA1c at the onset (mmol/mol) | 93 ± 21 | 90 (80; 109) |
| Basal c-peptide (ng/mL) | 0.43 ± 0.38 | 0.26 (0.10; 0.50) |
| Stimulated c-peptide (ng/mL) | 0.77 ± 0.64 | 0.58 (0.39; 0.97) |
| HLA predisposition | ||
| Yes | 87.40% | |
| No | 12.60% | |
| GADA positivity | ||
| Yes | 65.60% | |
| No | 34.40% | |
| ICA positivity | ||
| Yes | 50.70% | |
| No | 49.30% | |
| Total daily insulin dose at the onset (IU/Kg) | 0.75 ± 0.31 | 0.73 (0.52; 0.96) |
| PCR occurrence | ||
| Yes | 63.50% | |
| No | 36.50% | |
| BMI at 1-year-follow-up (SDS) | 0.15 ± 0.98 | 0.9 (−0.5; 0.85) |
| Last year mean value HbA1c at 1-year-follow-up (%) | 6.8 ± 0.7 | 6.8 (6.4; 7.3) |
| Last year mean value HbA1c at 1-year-follow-up (mmol/mol) | 51 ± 6.9 | 51 (46; 56) |
| Total daily insulin dose at 1-year-follow-up (IU/Kg) | 0.57 ± 0.31 | 0.45 (0.40; 0.65) |
| BMI at 2-years-follow-up (SDS) | 0.16 ± 0.98 | 0.1 (−0.45; 0.85) |
| Last year mean value HbA1c at 2-years-follow-up (%) | 7.2 ± 0.8 | 7.1 (6.7; 7.5) |
| Last year mean value HbA1c at 2-years-follow-up (mmol/mol) | 55 ± 8.4 | 54 (50; 58) |
| Total daily insulin dose at 2-years-follow-up (IU/Kg) | 0.71 ± 0.28 | 0.65 (0.51; 0.78) |
| Remitters | Non-Remitters | ||||
|---|---|---|---|---|---|
| Variables | Frequency or Mean ± SDS | Median (IQR) | Frequency or Mean ± SDS | Median (IQR) | p-Value |
| N | 63.5% | 36.5% | |||
| Sex | 0.49 | ||||
| Male | 54.70% | 49.20% | |||
| Female | 45.30% | 50.80% | |||
| Age at the onset (years) | 9.3 ± 3.7 | 9.4 (6.8; 12.3) | 7.4 ± 3.7 | 7.9 (3.9; 10.2) | 0.002 |
| Age classes | 0.011 | ||||
| 0–4 years | 14.20% | 32.80% | |||
| 5–9 years | 42.50% | 39.30% | |||
| 10–16 years | 43.40% | 27.90% | |||
| BMI at the onset (SDS) | −0.04 ± 1.10 | 0.01 (−0.81; 1.06) | −0.31 ± 1.3 | −0.08 (−1.23; 0.54) | 0.145 |
| Basal c-peptide (ng/mL) at the onset | 0.51 ± 0.42 | 0.30 (0.12; 0.51) | 0.29 ± 0.22 | 0.20 (0.10; 0.37) | <0.001 |
| Stimulated c-peptide (ng/mL) at the onset | 1.01 ± 0.73 | 0.70 (0.55; 1.70) | 0.49 ± 0.35 | 0.40 (0.28; 0.58) | <0.001 |
| pH | 7.3 ± 0.12 | 7.32 (7.27; 7.38) | 7.23 ± 0.16 | 7.28 (7.15; 7.36) | 0.005 |
| DKA at the onset | 0.044 | ||||
| Yes | 43.20% | 58.90% | |||
| No | 56.80% | 41.10% | |||
| Severity of DKA | 0.015 | ||||
| Mild | 70% | 35.50% | |||
| Moderate | 15% | 32.30% | |||
| Severe | 15% | 32.30% | |||
| HbA1c at the onset (%) | 10.7 ± 1.9 | 10.4 (9.5; 12.3) | 10.6 ± 2.0 | 10.5 (9.3; 12.1) | 0.972 |
| HbA1c at the onset (mmol/mol) | 93 ± 21 | 90 (80; 111) | 92 ± 23 | 91 (78; 109) | 0.972 |
| HLA predisposition | 0.526 | ||||
| Yes | 84.70% | 91.70% | |||
| No | 15.30% | 8.30% | |||
| ICA positivity | 0.116 | ||||
| Yes | 45.70% | 60% | |||
| No | 54.30% | 40% | |||
| GADA positivity | 0.232 | ||||
| Yes | 61.40% | 73.80% | |||
| No | 38.60% | 26.20% | |||
| Total daily insulin dose at the onset (IU/Kg) | 0.71 ± 0.27 | 0.72 (0.50; 0.93) | 0.83 ± 0.35 | 0.77 (0.57; 1.16) | 0.103 |
| BMI at 1-year-follow-up (SDS) | 0.13 ± 1 | 0.1 (−0.59; 0.92) | 0.19 ± 0.95 | 0.23 (−0.35; 0.79) | 0.651 |
| Last year mean value HbA1c at 1-year-follow-up (%) | 6.6 ± 0.6 | 6.7 (6.2; 6.9) | 7.2 ± 0.6 | 7.3 (6.7; 7.7) | <0.001 |
| Last year mean value HbA1c at 1-year-follow-up (mmol/mol) | 49 ± 6.5 | 50 (44; 52) | 55 ± 6.1 | 56 (50; 61) | <0.001 |
| Total daily insulin dose at 1-year-follow-up (IU/Kg) | 0.45 ± 0.25 | 0.35 (0.25; 0.50) | 0.78 ± 0.3 | 0.70 (0.50; 0.90) | <0.001 |
| BMI at 2-years-follow-up (SDS) | 0.18 ± 0.96 | 0.01 (−0.45; 0.86) | 0.13 ± 1.01 | 0.2 (−0.43; 0.74) | 0.899 |
| Last year mean value HbA1c at 2-year-follow-up (%) | 7.0 ± 0.7 | 7.0 (6.5; 7.5) | 7.4 ± 0.8 | 7.3 (6.9; 7.7) | 0.017 |
| Last year mean value HbA1c at 2-year-follow-up (mmol/mol) | 53 ± 8.1 | 53 (48; 58) | 57 ± 8.6 | 56 (52; 61) | 0.017 |
| Total daily insulin dose at 2-years-follow-up (IU/Kg) | 0.63 ± 0.27 | 0.48 (0.35; 0.69) | 0.87 ± 0.23 | 0.80 (0.60; 0.90) | <0.001 |
| Univariate | Multivariate | |||||
|---|---|---|---|---|---|---|
| Variables | Crude OR | 95% C.I. | p-Value | Adjusted OR | 95% C.I. | p-Value |
| Age at the onset | 1.14 | 1.05–1.25 | 0.002 | 1.15 | 0.96–1.39 | 0.139 |
| Gender (male) | 1.25 | 0.67–2.35 | 0.490 | 1.85 | 0.48–7.17 | 0.377 |
| BMI | 1.11 | 1.01–1.22 | 0.032 | 1.1 | 0.85–1.43 | 0.478 |
| pH | 49.02 | 3.63–662.1 | 0.003 | 0.51 | 0.01–328.35 | 0.838 |
| Presence of DKA | 0.53 | 0.27–1.03 | 0.062 | 0.8 | 0.13–4.88 | 0.812 |
| HbA1c | 1.03 | 0.86–1.22 | 0.775 | 0.9 | 0.64–1.28 | 0.563 |
| c-peptide levels | 12.8 | 2.54–64.47 | 0.002 | 6.72 | 0.12–372.3 | 0.352 |
| HLA predisposition | 0.51 | 0.13–2.01 | 0.331 | 0.19 | 0.01–2.69 | 0.218 |
| ICA positivity | 0.56 | 0.28-1.13 | 0.104 | 0.24 | 0.02–3.01 | 0.266 |
| GADA positivity | 0.57 | 0.25–1.28 | 0.566 | 24.65 | 1.6–380.66 | 0.022 |
| Total daily insulin dose | 0.27 | 0.08–0.87 | 0.028 | 0.06 | 0.01–0.77 | 0.031 |
| Variables | B | 95% C.I. | p-Value |
|---|---|---|---|
| Age at the onset | 0.84 | 0.25;1.43 | 0.005 |
| Gender | 1.21 | −2.69; 5.11 | 0.543 |
| BMI | 0.29 | −1.73; 2.32 | 0.776 |
| pH | 5.04 | −17.05; 27.12 | 0.655 |
| Presence of DKA | −0.40 | −5.87; 5.07 | 0.885 |
| C-peptide | −3.06 | −8.35; 2.23 | 0.257 |
| HLA predisposition | −3.74 | −9.46; 1.98 | 0.200 |
| ICA positivity | −0.516 | −6.85; 5.82 | 0.873 |
| GADA positivity | 4.15 | −2.73; 11.04 | 0.237 |
| Total daily insulin dose | −5.38 | −12.94; 2.17 | 0.162 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Passanisi, S.; Salzano, G.; Gasbarro, A.; Urzì Brancati, V.; Mondio, M.; Pajno, G.B.; Alibrandi, A.; Lombardo, F. Influence of Age on Partial Clinical Remission among Children with Newly Diagnosed Type 1 Diabetes. Int. J. Environ. Res. Public Health 2020, 17, 4801. https://doi.org/10.3390/ijerph17134801
Passanisi S, Salzano G, Gasbarro A, Urzì Brancati V, Mondio M, Pajno GB, Alibrandi A, Lombardo F. Influence of Age on Partial Clinical Remission among Children with Newly Diagnosed Type 1 Diabetes. International Journal of Environmental Research and Public Health. 2020; 17(13):4801. https://doi.org/10.3390/ijerph17134801
Chicago/Turabian StylePassanisi, Stefano, Giuseppina Salzano, Albino Gasbarro, Valentina Urzì Brancati, Matilde Mondio, Giovanni Battista Pajno, Angela Alibrandi, and Fortunato Lombardo. 2020. "Influence of Age on Partial Clinical Remission among Children with Newly Diagnosed Type 1 Diabetes" International Journal of Environmental Research and Public Health 17, no. 13: 4801. https://doi.org/10.3390/ijerph17134801
APA StylePassanisi, S., Salzano, G., Gasbarro, A., Urzì Brancati, V., Mondio, M., Pajno, G. B., Alibrandi, A., & Lombardo, F. (2020). Influence of Age on Partial Clinical Remission among Children with Newly Diagnosed Type 1 Diabetes. International Journal of Environmental Research and Public Health, 17(13), 4801. https://doi.org/10.3390/ijerph17134801







