Modulation of Endothelial Glycocalyx and Microcirculation in Healthy Young Men during High-Intensity Sprint Interval Cycling-Exercise by Supplementation with Pomegranate Extract. A Randomized Controlled Trial
Abstract
1. Introduction
2. Materials and Methods
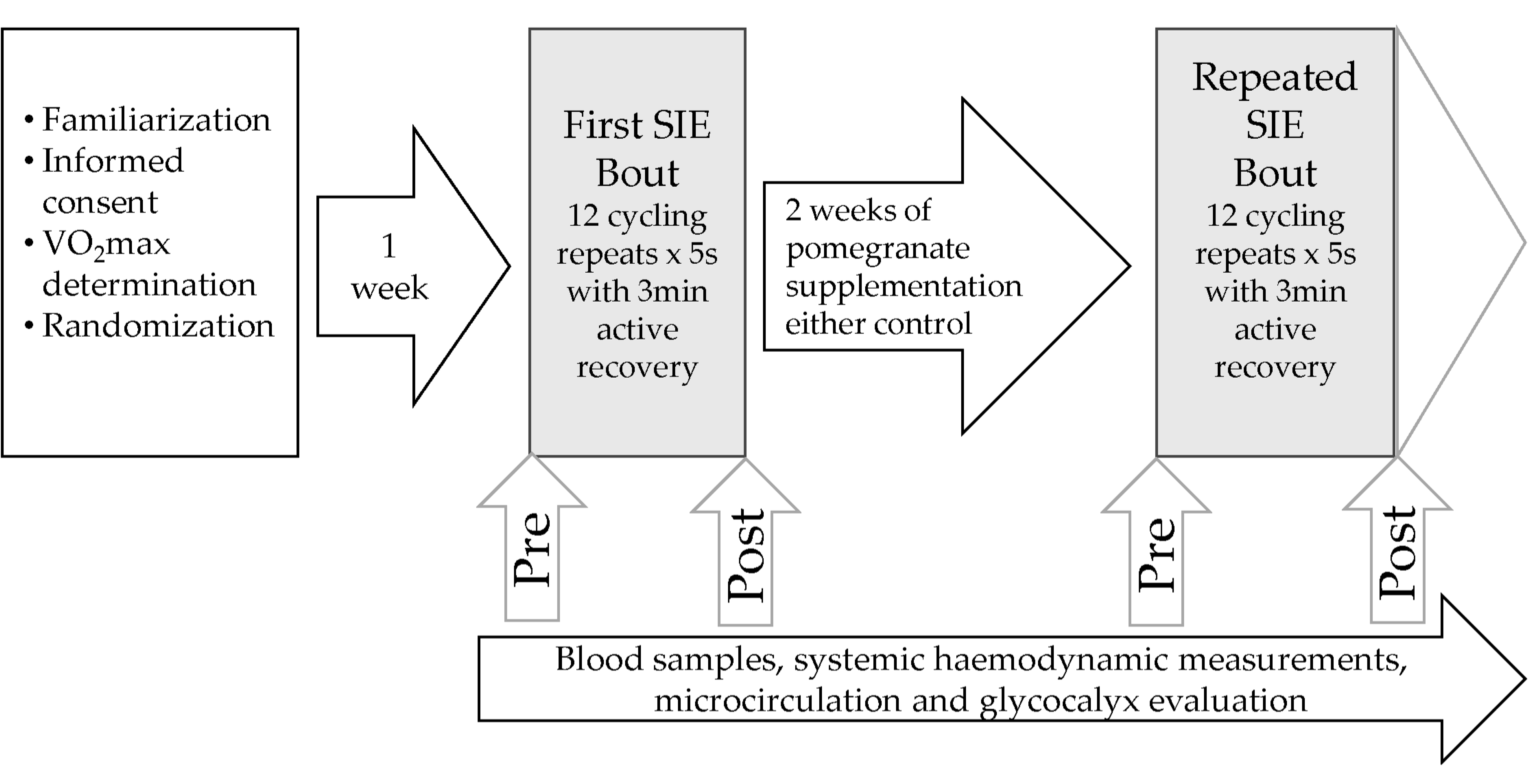
2.1. Experimental Design and Supplementation
2.2. Participants
2.3. Familiarization
2.4. Phenolic Compound Evaluation
2.5. Evaluating Microcirculation
2.6. Evaluating the Glycocalyx: Measuring the Perfused Boundary Region
2.7. Evaluating the Glycocalyx: Serum Measurements of a Glycocalyx Damage Marker
2.8. Lactate and Glucose Concentration
2.9. Statistics
3. Results
3.1. The Baseline Characteristics
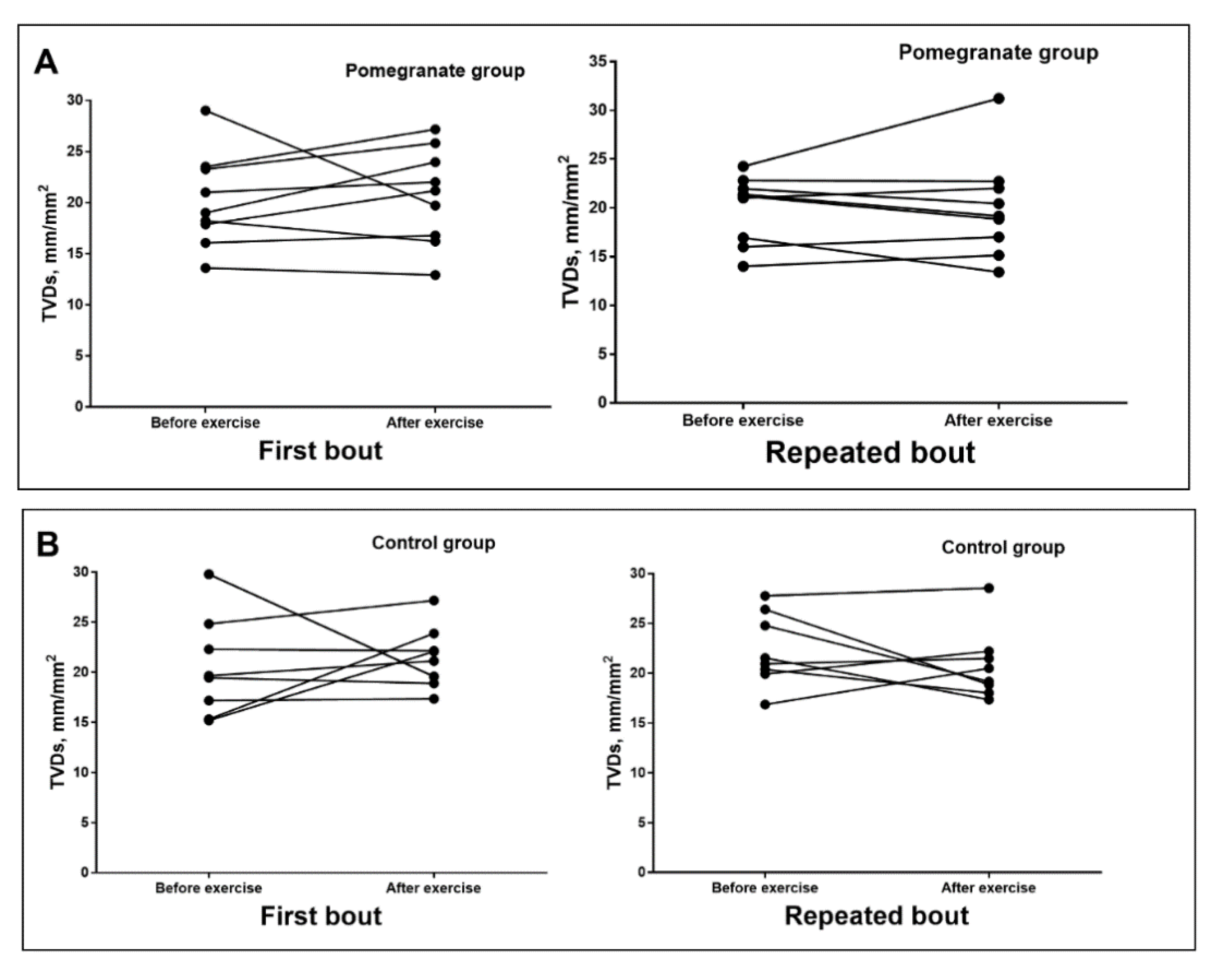
3.2. Glycocalyx Characteristics and Microcirculation
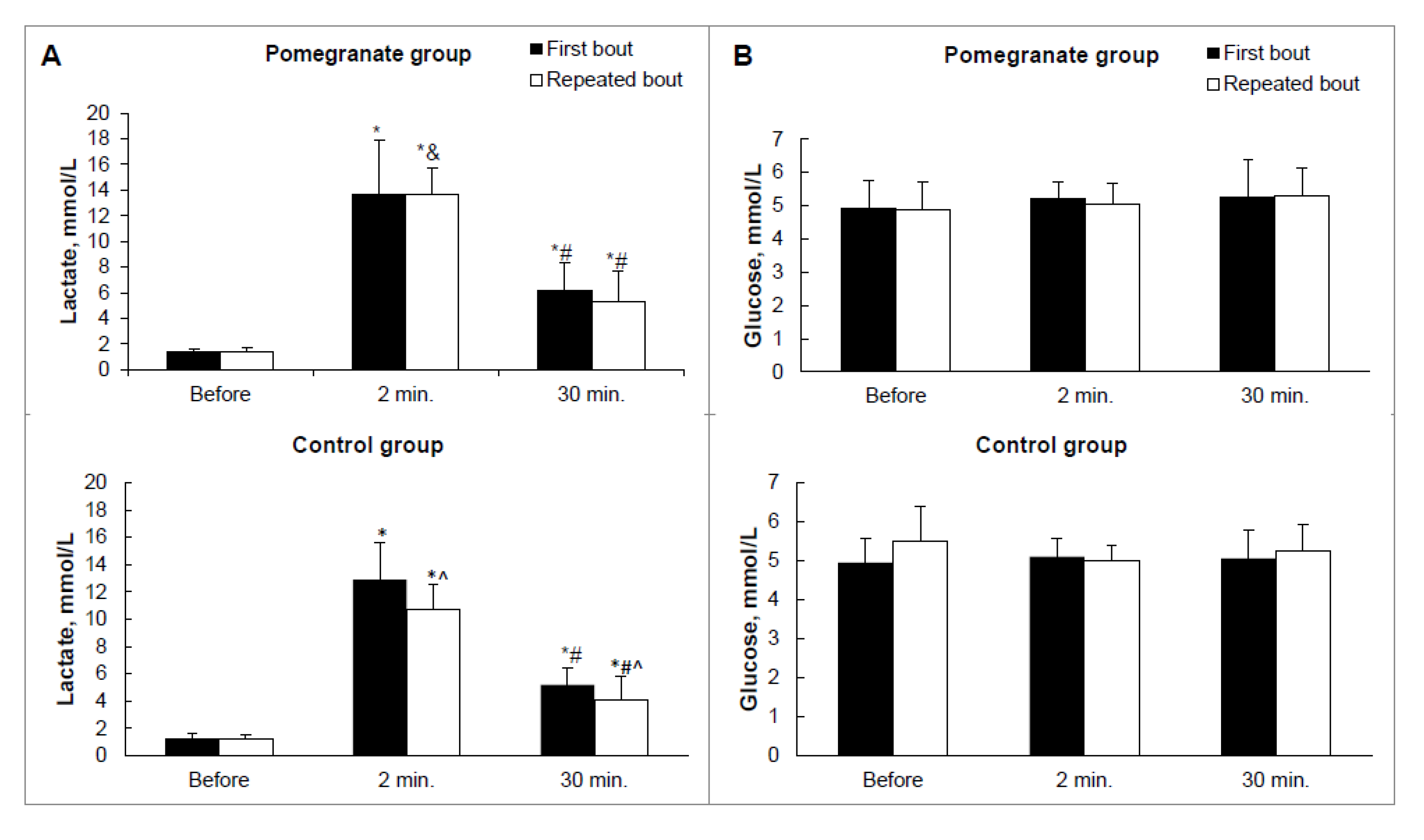
3.3. Blood Sample Analysis
3.4. Cycling Power and Systemic Hemodynamic Parameters
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Myburgh, K.H. Polyphenol supplementation: Benefits for exercise performance or oxidative stress? Sports Med. 2014, 44 (Suppl. 1), S57–S70. [Google Scholar] [CrossRef]
- Woodward, K.A.; Draijer, R.; Thijssen, D.H.J.; Low, D.A. Polyphenols and Microvascular Function in Humans: A Systematic Review. Curr. Pharm. Des. 2018, 24, 203–226. [Google Scholar] [CrossRef] [PubMed]
- Roelofs, E.J.; Smith-Ryan, A.E.; Trexler, E.T.; Hirsch, K.R.; Mock, M.G. Effects of pomegranate extract on blood flow and vessel diameter after high-intensity exercise in young, healthy adults. Eur. J. Sport Sci. 2017, 17, 317–325. [Google Scholar] [CrossRef] [PubMed]
- Asgary, S.; Sahebkar, A.; Afshani, M.R.; Keshvari, M.; Haghjooyjavanmard, S.; Rafieian-Kopaei, M. Clinical evaluation of blood pressure lowering, endothelial function improving, hypolipidemic and anti-inflammatory effects of pomegranate juice in hypertensive subjects. Phytother. Res. 2014, 28, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Trombold, J.R.; Reinfeld, A.S.; Casler, J.R.; Coyle, E.F. The effect of pomegranate juice supplementation on strength and soreness after eccentric exercise. J. Strength Cond. Res. 2011, 25, 1782–1788. [Google Scholar] [CrossRef] [PubMed]
- Chong, M.F.; Macdonald, R.; Lovegrove, J.A. Fruit polyphenols and CVD risk: A review of human intervention studies. Br. J. Nutr. 2010, 104 (Suppl. 3), S28–S39. [Google Scholar] [CrossRef]
- Mattiello, T.; Trifiro, E.; Jotti, G.S.; Pulcinelli, F.M. Effects of pomegranate juice and extract polyphenols on platelet function. J. Med. Food 2009, 12, 334–339. [Google Scholar] [CrossRef] [PubMed]
- Gil, M.I.; Tomas-Barberan, F.A.; Hess-Pierce, B.; Holcroft, D.M.; Kader, A.A. Antioxidant activity of pomegranate juice and its relationship with phenolic composition and processing. J. Agric. Food Chem. 2000, 48, 4581–4589. [Google Scholar] [CrossRef] [PubMed]
- Crimi, E.; Ignarro, L.J.; Napoli, C. Microcirculation and oxidative stress. Free Radic. Res. 2007, 41, 1364–1375. [Google Scholar] [CrossRef]
- Shu, X.; Keller, T.C., IV; Begandt, D.; Butcher, J.T.; Biwer, L.; Keller, A.S.; Columbus, L.; Isakson, B.E. Endothelial nitric oxide synthase in the microcirculation. Cell. Mol. Life Sci. 2015, 72, 4561–4575. [Google Scholar] [CrossRef] [PubMed]
- Ammar, A.; Chtourou, H.; Souissi, N. Effect of Time-of-Day on Biochemical Markers in Response to Physical Exercise. J. Strength Cond. Res. 2017, 31, 272–282. [Google Scholar] [CrossRef] [PubMed]
- Pingitore, A.; Lima, G.P.; Mastorci, F.; Quinones, A.; Iervasi, G.; Vassalle, C. Exercise and oxidative stress: Potential effects of antioxidant dietary strategies in sports. Nutrition 2015, 31, 916–922. [Google Scholar] [CrossRef] [PubMed]
- Malaguti, M.; Angeloni, C.; Hrelia, S. Polyphenols in exercise performance and prevention of exercise-induced muscle damage. Oxid. Med. Cell. Longev. 2013, 2013, 825928. [Google Scholar] [CrossRef] [PubMed]
- Di Francescomarino, S.; Sciartilli, A.; Di Valerio, V.; Di Baldassarre, A.; Gallina, S. The effect of physical exercise on endothelial function. Sports Med. 2009, 39, 797–812. [Google Scholar] [CrossRef]
- Cerda, B.; Ceron, J.J.; Tomas-Barberan, F.A.; Espin, J.C. Repeated oral administration of high doses of the pomegranate ellagitannin punicalagin to rats for 37 days is not toxic. J. Agric. Food Chem. 2003, 51, 3493–3501. [Google Scholar] [CrossRef]
- Kingwell, B.A.; Sherrard, B.; Jennings, G.L.; Dart, A.M. Four weeks of cycle training increases basal production of nitric oxide from the forearm. Am. J. Physiol. 1997, 272, H1070–H1077. [Google Scholar] [CrossRef]
- Majerczak, J.; Duda, K.; Chlopicki, S.; Bartosz, G.; Zakrzewska, A.; Balcerczyk, A.; Smolenski, R.T.; Zoladz, J.A. Endothelial glycocalyx integrity is preserved in young, healthy men during a single bout of strenuous physical exercise. Physiol. Res. 2016, 65, 281–291. [Google Scholar] [CrossRef]
- Vink, H.; Duling, B.R. Identification of distinct luminal domains for macromolecules, erythrocytes, and leukocytes within mammalian capillaries. Circ. Res. 1996, 79, 581–589. [Google Scholar] [CrossRef]
- Laughlin, M.H.; Newcomer, S.C.; Bender, S.B. Importance of hemodynamic forces as signals for exercise-induced changes in endothelial cell phenotype. J. Appl. Physiol. (1985) 2008, 104, 588–600. [Google Scholar] [CrossRef]
- Place, N.; Ivarsson, N.; Venckunas, T.; Neyroud, D.; Brazaitis, M.; Cheng, A.J.; Ochala, J.; Kamandulis, S.; Girard, S.; Volungevičius, G.; et al. Ryanodine receptor fragmentation and sarcoplasmic reticulum Ca2+ leak after one session of high-intensity interval exercise. Proc. Natl. Acad. Sci. USA 2015, 112, 15492–15497. [Google Scholar] [CrossRef]
- Gibala, M.J.; Little, J.P.; van Essen, M.; Wilkin, G.P.; Burgomaster, K.A.; Safdar, A.; Raha, S.; Tarnopolsky, M.A. Short-term sprint interval versus traditional endurance training: Similar initial adaptations in human skeletal muscle and exercise performance. J. Physiol. 2006, 575, 901–911. [Google Scholar] [CrossRef] [PubMed]
- De Backer, D.; Hollenberg, S.; Boerma, C.; Goedhart, P.; Buchele, G.; Ospina-Tascon, G.; Dobbe, I.; Ince, C. How to evaluate the microcirculation: Report of a round table conference. Crit. Care 2007, 11, R101. [Google Scholar] [CrossRef] [PubMed]
- Verdant, C.L.; De Backer, D.; Bruhn, A.; Clausi, C.M.; Su, F.; Wang, Z.; Rodriguez, H.; Pries, A.R.; Vincent, J.L. Evaluation of sublingual and gut mucosal microcirculation in sepsis: A quantitative analysis. Crit. Care Med. 2009, 37, 2875–2881. [Google Scholar] [CrossRef]
- Gouverneur, M.; Spaan, J.A.; Pannekoek, H.; Fontijn, R.D.; Vink, H. Fluid shear stress stimulates incorporation of hyaluronan into endothelial cell glycocalyx. Am. J. Physiol. Heart Circ. Physiol. 2006, 290, H458–H462. [Google Scholar] [CrossRef]
- Skurvydas, A.; Verbickas, V.; Eimantas, N.; Baranauskiene, N.; Cernych, M.; Skrodeniene, E.; Daniuseviciute, L.; Brazaitis, M. Psychological and Physiological Biomarkers of Neuromuscular Fatigue after Two Bouts of Sprint Interval Exercise. Front. Psychol. 2017, 8, 2282. [Google Scholar] [CrossRef]
- Pries, A.R.; Secomb, T.W.; Gaehtgens, P. The endothelial surface layer. Pflugers Arch. 2000, 440, 653–666. [Google Scholar] [CrossRef]
- Ince, C.; Boerma, E.C.; Cecconi, M.; De Backer, D.; Shapiro, N.I.; Duranteau, J.; Pinsky, M.R.; Artigas, A.; Teboul, J.L.; Reiss, I.K.M.; et al. Cardiovascular Dynamics Section of the ESICM Second consensus on the assessment of sublingual microcirculation in critically ill patients: Results from a task force of the European Society of Intensive Care Medicine. Intensive Care Med. 2018, 44, 281–299. [Google Scholar] [CrossRef]
- Becker, B.F.; Chappell, D.; Jacob, M. Endothelial glycocalyx and coronary vascular permeability: The fringe benefit. Basic Res. Cardiol. 2010, 105, 687–701. [Google Scholar] [CrossRef]
- Verbickas, V.; Baranauskiene, N.; Eimantas, N.; Kamandulis, S.; Rutkauskas, S.; Satkunskiene, D.; Sadauskas, S.; Brazaitis, M.; Skurvydas, A. Effect of sprint cycling and stretch-shortening cycle exercises on the neuromuscular, immune and stress indicators in young men. J. Physiol. Pharmacol. 2017, 68, 125–132. [Google Scholar]
- Howley, E.T.; Bassett, D.R., Jr.; Welch, H.G. Criteria for maximal oxygen uptake: Review and commentary. Med. Sci. Sports Exerc. 1995, 27, 1292–1301. [Google Scholar] [CrossRef]
- Thompson, W.; Gordon, N.; Pescatello, L. American College of Sports Medicine. ACSM’s Guidelines for Exercise Testing and Prescription, 8th ed.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2010. [Google Scholar]
- Goedhart, P.T.; Khalilzada, M.; Bezemer, R.; Merza, J.; Ince, C. Sidestream Dark Field (SDF) imaging: A novel stroboscopic LED ring-based imaging modality for clinical assessment of the microcirculation. Opt. Express 2007, 15, 15101–15114. [Google Scholar] [CrossRef] [PubMed]
- Aykut, G.; Veenstra, G.; Scorcella, C.; Ince, C.; Boerma, C. Cytocam-IDF (incident dark field illumination) imaging for bedside monitoring of the microcirculation. Intensive Care Med. Exp. 2015, 3, 4. [Google Scholar] [CrossRef] [PubMed]
- Gruartmoner, G.; Mesquida, J.; Ince, C. Microcirculatory monitoring in septic patients: Where do we stand? Med. Intensiva. 2017, 41, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Boerma, E.C.; Mathura, K.R.; van der Voort, P.H.; Spronk, P.E.; Ince, C. Quantifying bedside-derived imaging of microcirculatory abnormalities in septic patients: A prospective validation study. Crit. Care 2005, 9, R601–R606. [Google Scholar] [CrossRef]
- Pranskunas, A.; Arstikyte, J.; Pranskuniene, Z.; Bernatoniene, J.; Kiudulaite, I.; Vaitkaitiene, E.; Vaitkaitis, D.; Brazaitis, M. Time Evolution of Sublingual Microcirculatory Changes in Recreational Marathon Runners. Biomed. Res. Int. 2017, 2017, 7120785. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.H.; Dane, M.J.; van den Berg, B.M.; Boels, M.G.; van Teeffelen, J.W.; de Mutsert, R.; den Heijer, M.; Rosendaal, F.R.; van der Vlag, J.; van Zonneveld, A.J.; et al. NEO study group Deeper penetration of erythrocytes into the endothelial glycocalyx is associated with impaired microvascular perfusion. PLoS ONE 2014, 9, e96477. [Google Scholar]
- Martens, R.J.; Vink, H.; van Oostenbrugge, R.J.; Staals, J. Sublingual microvascular glycocalyx dimensions in lacunar stroke patients. Cerebrovasc. Dis. 2013, 35, 451–454. [Google Scholar] [CrossRef]
- Rovas, A.; Lukasz, A.H.; Vink, H.; Urban, M.; Sackarnd, J.; Pavenstadt, H.; Kumpers, P. Bedside analysis of the sublingual microvascular glycocalyx in the emergency room and intensive care unit—The GlycoNurse study. Scand. J. Trauma. Resusc. Emerg. Med. 2018, 26, 16. [Google Scholar] [CrossRef]
- Nunes, J.C.; Radbruch, H.; Walz, R.; Lin, K.; Stenzel, W.; Prokop, S.; Koch, A.; Heppner, F.L. The most fulminant course of the Marburg variant of multiple sclerosis-autopsy findings. Mult. Scler. 2015, 21, 485–487. [Google Scholar] [CrossRef]
- Trexler, E.T.; Smith-Ryan, A.E.; Melvin, M.N.; Roelofs, E.J.; Wingfield, H.L. Effects of pomegranate extract on blood flow and running time to exhaustion. Appl. Physiol. Nutr. Metab. 2014, 39, 1038–1042. [Google Scholar] [CrossRef]
- Ristow, M.; Zarse, K.; Oberbach, A.; Kloting, N.; Birringer, M.; Kiehntopf, M.; Stumvoll, M.; Kahn, C.R.; Bluher, M. Antioxidants prevent health-promoting effects of physical exercise in humans. Proc. Natl. Acad. Sci. USA 2009, 106, 8665–8670. [Google Scholar] [CrossRef] [PubMed]
- Paulsen, G.; Cumming, K.T.; Holden, G.; Hallen, J.; Ronnestad, B.R.; Sveen, O.; Skaug, A.; Paur, I.; Bastani, N.E.; Ostgaard, H.N.; et al. Vitamin C and E supplementation hampers cellular adaptation to endurance training in humans: A double-blind, randomised, controlled trial. J. Physiol. 2014, 592, 1887–1901. [Google Scholar] [CrossRef]
- Seeram, N.P.; Lee, R.; Heber, D. Bioavailability of ellagic acid in human plasma after consumption of ellagitannins from pomegranate (Punica granatum L.) juice. Clin. Chim. Acta 2004, 348, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Matthaiou, C.M.; Goutzourelas, N.; Stagos, D.; Sarafoglou, E.; Jamurtas, A.; Koulocheri, S.D.; Haroutounian, S.A.; Tsatsakis, A.M.; Kouretas, D. Pomegranate juice consumption increases GSH levels and reduces lipid and protein oxidation in human blood. Food Chem. Toxicol. 2014, 73, 1–6. [Google Scholar] [CrossRef]
- Fuster-Munoz, E.; Roche, E.; Funes, L.; Martinez-Peinado, P.; Sempere, J.M.; Vicente-Salar, N. Effects of pomegranate juice in circulating parameters, cytokines, and oxidative stress markers in endurance-based athletes: A randomized controlled trial. Nutrition 2016, 32, 539–545. [Google Scholar] [CrossRef] [PubMed]
- Adeva-Andany, M.; Lopez-Ojen, M.; Funcasta-Calderon, R.; Ameneiros-Rodriguez, E.; Donapetry-Garcia, C.; Vila-Altesor, M.; Rodriguez-Seijas, J. Comprehensive review on lactate metabolism in human health. Mitochondrion 2014, 17, 76–100. [Google Scholar] [CrossRef]
- Copp, S.W.; Schwagerl, P.J.; Hirai, D.M.; Poole, D.C.; Musch, T.I. Acute ascorbic acid and hindlimb skeletal muscle blood flow distribution in old rats: Rest and exercise. Can. J. Physiol. Pharmacol. 2012, 90, 1498–1505. [Google Scholar] [CrossRef] [PubMed]
- Brooks, G.A.; Dubouchaud, H.; Brown, M.; Sicurello, J.P.; Butz, C.E. Role of mitochondrial lactate dehydrogenase and lactate oxidation in the intracellular lactate shuttle. Proc. Natl. Acad. Sci. USA 1999, 96, 1129–1134. [Google Scholar] [CrossRef] [PubMed]
| Variables | Pomegranate Group | Control Group | p |
|---|---|---|---|
| Age (yr) | 24 (22–25) | 23 (22–28) | 0.893 |
| Mass (kg) | 78.0 (68.2–84.6) | 70.2 (65.0–81.1) | 0.426 |
| Height (cm) | 180.0 (178.0–184.5) | 180.0 (177.0–183.5) | 0.929 |
| Body mass index (kg/m2) | 21.3 (20.7–27.3) | 21.9 (19.8–25.3) | 0.566 |
| Fat (%) | 11.9 (10.1–18.8) | 11.2 (9.3–17.5) | 0.700 |
| VO2max (mL∙kg−1∙min−1) | 45.9 (41.0–46.5) | 42.2 (39.4–44.9) | 0.172 |
| Mean arterial pressure (mmHg) | 94 (93–101) | 97 (94–100) | 0.756 |
| Heart rate (beats/min) | 76 (70–81) | 79 (66–86) | 0.690 |
| Cardiac index (L/min/m2) | 3.6 (3.1–4.4) | 3.8 (2.9–4.4) | 0.690 |
| Lactate (mmol/L) | 1.4 (1.3–1.6) | 1.1 (1.0–1.9) | 0.478 |
| Syndecan-1 (ng/mL) | 8.7 (2.7–34.7) | 7.7 (1.7–16.8) | 0.685 |
| PBR (µm) | 1.87 (1.63–2.05) | 1.94 (1.66–2.00) | 0.635 |
| TVDs (mm/mm2) | 19.0 (17.0–23.4) | 19.7 (16.3–27.3) | 0.222 |
| PVDs (1/mm) | 9.7 (8.8–13.6) | 11.1 (9.7–15.7) | 0.080 |
| PPVs (%) | 95.7 (93.0–99.3) | 98.6 (95.3–98.8) | 0.386 |
| MFIs | 3.00 (3.00–3.00) | 3.00 (3.00–3.00) | 0.955 |
| Baseline | After 2 Weeks | |||||
|---|---|---|---|---|---|---|
| Before | After | Before | After | p | ||
| Syndecan-1 (ng/mL) | PG | 8.7(2.7–34.7) | 7.1(4.0–31.9) | 8.7(4.9–36.7) | 9.3(4.7–37.5) | 0.672 |
| Con | 7.7(1.7–16.8) | 7.4(1.7–17.2) | 6.0(2.5–19.3) | 5.8(4.7–16.5) | 0.798 | |
| PBR (µm) | PG | 1.87(1.63–2.05) | 1.90(1.72–1.95) | 1.97(1.81–2.10) | 1.73(1.52–1.89) | 0.563 |
| Con | 1.94(1.66–2.00) | 1.87(1.73–1.95) | 1.82(1.63–1.98) | 1.81(1.68–1.95) | 0.496 | |
| Baseline | After 2 Weeks | |||||
|---|---|---|---|---|---|---|
| Before | After | Before | After | p | ||
| TVDs (mm/mm2) | PG | 19.0(17.0–23.4) | 21.2(16.8–25.8) | 21.4(16.9–22.8) | 19.2(15.1–22.7) | 0.932 |
| Con | 19.7(16.3–27.3) | 21.2(19.2–23.0) | 21.5(20.2–27.1) | 20.5(18.4–25.4) | 0.978 | |
| PVDs (1/mm) | PG | 9.7(8.8–13.6) | 11.0(9.4–14.5) | 11.9(9.3–12.4) | 10.8(8.6–12.2) | 0.508 |
| Con | 11.1(9.7–15.7) | 11.4(10.7–13.5) | 12.0(10.4–15.9) | 12.1(11.1–15.0) | 0.996 | |
| PPVs (%) | PG | 95.7(93.0–99.3) | 96.1(93.8–98.8) | 95.6(93.8–97.3) | 95.6(95.0–100.0) | 0.745 |
| Con | 98.6(95.3–98.8) | 96.4(91.4–98.9) | 95.9(93.15–98.4) | 95.8(91.7–98.3) | 0.472 | |
| MFIs | PG | 3.00(3.00–3.00) | 3.00(2.84–3.00) | 3.00(3.00–3.00) | 3.00(3.00–3.00) | 0.437 |
| Con | 3.00(3.00–3.00) | 3.00(2.81–3.00) | 3.00(2.75–3.00) | 3.00(2.81–3.00) | 0.395 | |
| Baseline | After 2 Weeks | |||||
|---|---|---|---|---|---|---|
| Before | After | Before | After | p | ||
| MAP (mmHg) | PG | 94(93–101) | 107(96–116) a | 98(92–101) | 104(94–107) | 0.026 |
| Con | 97(94–100) | 103(99–115) a | 97(90–100) | 100(95–107) | 0.030 | |
| HR (beats/min) | PG | 76(70–81) | 106(96–116) a | 75(62–89) | 104(91–116) a | <0.001 |
| Con | 79(66–86) | 106(92–115) a | 77(63–84) | 104(92–116) a | <0.001 | |
| CI (L/min/m2) | PG | 3.6(3.1–4.4) | 4.4(3.5–4.9) a | 3.7(3.5–4.0) | 4.5(3.7–4.7) a | 0.045 |
| Con | 3.8(2.9–4.4) | 4.2(3.4–5.1) a | 3.3(3.2–3.7) | 3.9(3.5–4.9) a | 0.037 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pranskuniene, Z.; Belousoviene, E.; Baranauskiene, N.; Eimantas, N.; Vaitkaitiene, E.; Bernatoniene, J.; Brazaitis, M.; Pranskunas, A. Modulation of Endothelial Glycocalyx and Microcirculation in Healthy Young Men during High-Intensity Sprint Interval Cycling-Exercise by Supplementation with Pomegranate Extract. A Randomized Controlled Trial. Int. J. Environ. Res. Public Health 2020, 17, 4405. https://doi.org/10.3390/ijerph17124405
Pranskuniene Z, Belousoviene E, Baranauskiene N, Eimantas N, Vaitkaitiene E, Bernatoniene J, Brazaitis M, Pranskunas A. Modulation of Endothelial Glycocalyx and Microcirculation in Healthy Young Men during High-Intensity Sprint Interval Cycling-Exercise by Supplementation with Pomegranate Extract. A Randomized Controlled Trial. International Journal of Environmental Research and Public Health. 2020; 17(12):4405. https://doi.org/10.3390/ijerph17124405
Chicago/Turabian StylePranskuniene, Zivile, Egle Belousoviene, Neringa Baranauskiene, Nerijus Eimantas, Egle Vaitkaitiene, Jurga Bernatoniene, Marius Brazaitis, and Andrius Pranskunas. 2020. "Modulation of Endothelial Glycocalyx and Microcirculation in Healthy Young Men during High-Intensity Sprint Interval Cycling-Exercise by Supplementation with Pomegranate Extract. A Randomized Controlled Trial" International Journal of Environmental Research and Public Health 17, no. 12: 4405. https://doi.org/10.3390/ijerph17124405
APA StylePranskuniene, Z., Belousoviene, E., Baranauskiene, N., Eimantas, N., Vaitkaitiene, E., Bernatoniene, J., Brazaitis, M., & Pranskunas, A. (2020). Modulation of Endothelial Glycocalyx and Microcirculation in Healthy Young Men during High-Intensity Sprint Interval Cycling-Exercise by Supplementation with Pomegranate Extract. A Randomized Controlled Trial. International Journal of Environmental Research and Public Health, 17(12), 4405. https://doi.org/10.3390/ijerph17124405







