Changes in Thyroid and Glycemic Status and Food Intake in Children with Excess Weight Who Were Submitted for a Multi-Component School Intervention for 16 Months
Abstract
1. Introduction
2. Materials and Methods
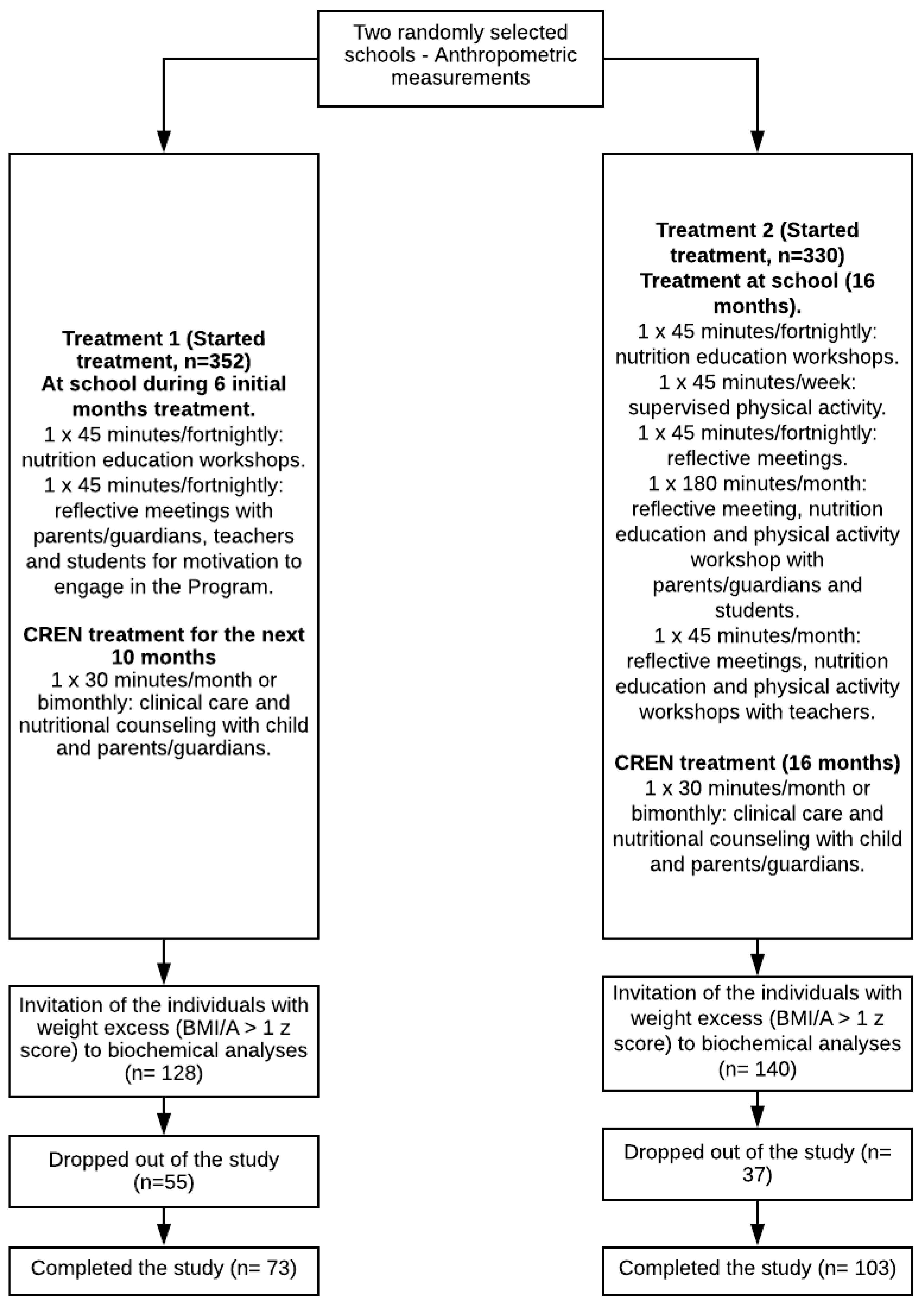
2.1. Population and Study Design
2.2. Anthropometry
2.3. Biochemistry
2.4. Dietary Evaluation
2.5. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Reinehr, T.; De Sousa, G.; Andler, W. Hyperthyrotropinemia in obese children is reversible after weight loss and is not related to lipids. J. Clin. Endocrinol. Metab. 2006, 91, 3088–3091. [Google Scholar] [CrossRef] [PubMed]
- Marwaha, R.K.; Tandon, N.; Garg, M.K.; Ganie, M.A.; Narang, A.; Mehan, N.; Bhadra, K. Impact of body mass index on thyroid functions in Indian children. Clin. Endocrinol. (Oxf.) 2013, 79, 424–428. [Google Scholar] [CrossRef] [PubMed]
- Rotondi, M.; Leporati, P.; La Manna, A.; Pirali, B.; Mondello, T.; Fonter, R.; Magri, F.; Chiovato, L. Raised serum TSH levels in patients with morbid obesity: Is it enough to diagnose subclinical hypothyroidism? Eur. J. Endocrinol. 2009, 160, 403–408. [Google Scholar] [CrossRef] [PubMed]
- Aeberli, I.; Jung, A.; Murer, S.B.; Wildhaber, J.; Wildhaber-Brooks, J.; Knöpfli, B.H.; Zimmermann, M.B. During rapid weight loss in obese children, reductions in TSH predict improvements in insulin sensitivity independent of changes in body weight or fat. J. Clin. Endocrinol. Metab. 2010, 95, 5412–5418. [Google Scholar] [CrossRef]
- Witkowska-Sȩdek, E.; Kucharska, A.; Rumińska, M.; Pyrzzak, B. Thyroid dysfunction in obese and overweight children. Endokrynol. Pol. 2017, 68, 54–60. [Google Scholar]
- Emokpae, M.A.; Obazelu, P.A. The Association of Triiodothyronine-to-Thyroxine Ratio with Body Mass Index in Obese Nigerian Children and Adolescents. Med. Sci. 2017, 5, 36. [Google Scholar] [CrossRef]
- Martins, V.J.B.; Toledo Florêncio, T.M.M.; Grillo, L.P.; Franco, M.d.C.P.; Martins, P.A.; Clemente, A.P.G.; Santos, C.D.L.; Vieria, M.d.F.A.; Sawaya, A.L. Long-lasting effects of undernutrition. Int. J. Environ. Res. Public Health 2011, 8, 1817–1846. [Google Scholar] [CrossRef]
- Dahl, M.; Ohrt, J.D.; Fonvig, C.E.; Kloppenborg, J.T.; Pedersen, O.; Hansen, T.; Holm, J.C. Subclinical hypothyroidism in danish lean and obese children and adolescents. JCRPE J. Clin. Res. Pediatr. Endocrinol. 2017, 9, 8–16. [Google Scholar] [CrossRef]
- Peeters, R.P. Subclinical hypothyroidism. N. Engl. J. Med. 2017, 376, 2556–2565. [Google Scholar] [CrossRef]
- Romualdo, M.C.D.S.; De Nóbrega, F.J.; Escrivão, M.A.M.S. Insulin resistance in obese children and adolescents. J. Pediatr. (Rio. J.) 2014, 90, 600–607. [Google Scholar] [CrossRef]
- Frithioff-Bøjsøe, C.; Lund, M.A.V.; Kloppenborg, J.T.; Nielsen, T.T.H.; Fonvig, C.E.; Lausten-Thomsen, U.; Hedley, P.L.; Hansen, T.; Pedersen, O.B.; Christiansen, M.; et al. Glucose metabolism in children and adolescents: Population-based reference values and comparisons to children and adolescents enrolled in obesity treatment. Pediatr. Diabetes 2019, 20, 538–548. [Google Scholar] [CrossRef] [PubMed]
- Fontenelle, L.C.; Feitosa, M.M.; Severo, J.S.; Freitas, T.E.C.; Morais, J.B.S.; Torres-Leal, F.L.; Henriques, G.S.; Do Nascimento Marreiro, D. Thyroid Function in Human Obesity: Underlying Mechanisms. Horm. Metab. Res. 2016, 48, 787–794. [Google Scholar] [CrossRef] [PubMed]
- Brufani, C.; Manco, M.; Nobili, V.; Fintini, D.; Barbetti, F.; Cappa, M. Thyroid function tests in obese prepubertal children: Correlations with insulin sensitivity and body fat distribution. Horm. Res. Paediatr. 2012, 78, 100–105. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization Obesity and Overweight. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 28 November 2019).
- González-Muniesa, P.; Mártinez-González, M.A.; Hu, F.B.; Després, J.P.; Matsuzawa, Y.; Loos, R.J.F.; Moreno, L.A.; Bray, G.A.; Martinez, J.A. Obesity. Nat. Rev. Dis. Prim. 2017, 3, 1–18. Available online: https://www.nature.com/articles/nrdp201734 (accessed on 28 November 2019).
- Biener, A.; Cawley, J.; Meyerhoefer, C. The impact of obesity on medical care costs and labor market outcomes in the US. Clin. Chem. 2018, 64, 108–117. [Google Scholar] [CrossRef]
- Lanigan, J. Prevention of overweight and obesity in early life. Proc. Nutr. Soc. 2018, 77, 247–256. [Google Scholar] [CrossRef]
- Moxley, E.; Habtzghi, D.; Klinkhamer, N.; Wang, H.; Donnelly, S.; Dykhuizen, J. Prevention and treatment of pediatric obesity: A strategy involving children, adolescents and the family for improved body composition. J. Pediatr. Nurs. 2019, 45, 13–19. [Google Scholar] [CrossRef]
- Licenziati, M.R.; Valerio, G.; Vetrani, I.; De Maria, G.; Liotta, F.; Radetti, G. Altered Thyroid Function and Structure in Children and Adolescents Who Are Overweight and Obese: Reversal after Weight Loss. J. Clin. Endocrinol. Metab. 2019, 104, 2757–2765. [Google Scholar] [CrossRef]
- Kirschenbaum, D.S.; Gierut, K. Treatment of childhood and adolescent obesity: An integrative review of recent recommendations from five expert groups. J. Consult. Clin. Psychol. 2013, 81, 347–360. [Google Scholar] [CrossRef]
- Matthews, D.R.; Hosker, J.P.; Rudenski, A.S.; Naylor, B.A.; Treacher, D.F.; Turner, R.C. Homeostasis model assessment: Insulin resistance and ?-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985, 28, 412–419. [Google Scholar] [CrossRef]
- Araujo, M.C.; Yokoo, E.M.; Pereira, R.A. Validation and Calibration of a Semiquantitative Food Frequency Questionnaire Designed for Adolescents. J. Am. Diet. Assoc. 2010, 110, 1170–1177. [Google Scholar] [CrossRef] [PubMed]
- Brito, A.P.; Araujo, M.C.; Guimarães, C.P.; Pereira, R.A. Relative validity of a food frequency questionnaire supported by images. Cienc. e Saude Coletiva 2017, 22, 457–468. [Google Scholar] [CrossRef][Green Version]
- Filgueiras, A.R.; Pires de Almeida, V.B.; Koch Nogueira, P.C.; Alvares Domene, S.M.; Eduardo da Silva, C.; Sesso, R.; Sawaya, A.L. Exploring the consumption of ultra-processed foods and its association with food addiction in overweight children. Appetite 2019, 135, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Häkkänen, P.; Ketola, E.; Laatikainen, T. Screening and treatment of obesity in school health care—The gap between clinical guidelines and reality. Scand. J. Caring Sci. 2018, 32, 1332–1341. [Google Scholar]
- Kothandan, S.K. School based interventions versus family based interventions in the treatment of childhood obesity—A systematic review. Arch. Public Heal. 2014, 72, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Johannsen, D.L.; Marlatt, K.L.; Conley, K.E.; Smith, S.R.; Ravussin, E. Metabolic adaptation is not observed after 8 weeks of overfeeding but energy expenditure variability is associated with weight recovery. Am. J. Clin. Nutr. 2019, 110, 805–813. [Google Scholar] [CrossRef] [PubMed]
- Shao, S.S.; Zhao, Y.F.; Song, Y.F.; Xu, C.; Yang, J.M.; Xuan, S.M.; Yan, H.L.; Yu, C.X.; Zhao, M.; Xu, J.; et al. Dietary high-fat lard intake induces thyroid dysfunction and abnormal morphology in rats. Acta Pharmacol. Sin. 2014, 35, 1411–1420. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Chen, W.; Shao, S.; Xu, G.; Song, Y.; Xu, C.; Gao, L.; Hu, C.; Zhao, J. A High-Fat Diet Rich in Saturated and Mono-Unsaturated Fatty Acids Induces Disturbance of Thyroid Lipid Profile and Hypothyroxinemia in Male Rats. Mol. Nutr. Food Res. 2018, 62, 1–9. [Google Scholar] [CrossRef]
- Štrbák, V.; Tatár, P.; Angyal, R.; Štrec, V.; Aksamitová, K.; Vigaš, M.; Jánošová, H. Effects of sauna and glucose intake on TSH and thyroid hormone levels in plasma of euthyroid subjects. Metabolism 1987, 36, 426–431. [Google Scholar] [CrossRef]
- Zhao, C.H.; Campbell, E.S.; Tschiffely, A.E.; Castonguay, T.W. Overnight Access to Sugar Solutions Affects mRNA Expression of Several Neuropeptides in Different Hypothalamic Regions in Rats. J. Food Nutr. Res. 2015, 3, 69–76. [Google Scholar] [CrossRef][Green Version]
- Lundbäck, V.; Ekbom, K.; Hagman, E.; Dahlman, I.; Marcus, C. Thyroid-Stimulating Hormone, Degree of Obesity, and Metabolic Risk Markers in a Cohort of Swedish Children with Obesity. Horm. Res. Paediatr. 2017, 88, 140–146. [Google Scholar] [CrossRef] [PubMed]
- Santos, M.I.; Limbert, C.; Marques, F.C.; Rosário, F.; Lopes, L. Childhood obesity, thyroid function, and insulin resistance-is there a link? A longitudinal study. J. Pediatr. Endocrinol. Metab. 2015, 28, 557–562. [Google Scholar] [CrossRef] [PubMed]
- Brunetto, E.L.; Teixeira, S.D.S.; Giannocco, G.; MacHado, U.F.; Nunes, M.T. T 3 rapidly increases SLC2A4 gene expression and glut4 trafficking to the plasma membrane in skeletal muscle of rat and improves glucose homeostasis. Thyroid 2012, 22, 70–79. [Google Scholar] [CrossRef] [PubMed]
- Stanická, S.; Vondra, K.; Pelikánová, T.; Vlček, P.; Hill, M.; Zamrazil, V. Insulin sensitivity and counter-regulatory hormones in hypothyroidism and during thyroid hormone replacement therapy. Clin. Chem. Lab. Med. 2005, 43, 715–720. [Google Scholar] [CrossRef]
- Chatzitomaris, A.; Hoermann, R.; Midgley, J.E.; Hering, S.; Urban, A.; Dietrich, B.; Abood, A.; Klein, H.H.; Dietrich, J.W. Thyroid allostasis-adaptive responses of thyrotropic feedback control to conditions of strain, stress, and developmental programming. Front. Endocrinol. (Lausanne) 2017, 8, 1–28. [Google Scholar] [CrossRef]
- Martins, V.; Neves, A.; Garcia, M.; Spadari, R.; Clemente, A.; de Albuquerque, M.; Hoffman, D.; Sawaya, A. Normal cortisol response to cold pressor test, but lower free thyroxine, after recovery from undernutrition. Br. J. Nutr. 2016, 115, 14–23. [Google Scholar] [CrossRef]
- Poddar, M.; Chetty, Y.; Chetty, V.T. How does obesity affect the endocrine system? A narrative review. Clin. Obes. 2017, 7, 136–144. [Google Scholar] [CrossRef]
- Bianco, A.C.; Dumitrescu, A.; Gereben, B.; Ribeiro, M.O.; Fonseca, T.L.; Fernandes, G.W.; Bocco, B.M.L.C. Paradigms of Dynamic Control of Thyroid Hormone Signaling. Endocr. Rev. 2019, 40, 1000–1047. [Google Scholar] [CrossRef]
- Reinehr, T. Obesity and thyroid function. Mol. Cell. Endocrinol. 2010, 316, 165–171. [Google Scholar] [CrossRef]
| Excess Weight (BMI ≥ 1 z Score) | Treatment 1 (n = 73; B 27, G46) | Treatment 2 (n = 103; B 51, G 52) | pb |
|---|---|---|---|
| Age (years) | 9.83 ± 0.85 | 9.40 ± 0.90 | 0.001 |
| HAZ (z score) | 0.60 ± 0.89 | 0.68 ± 0.93 | 0.557 |
| BMI/A (z score) | 2.04 ± 0.72 | 1.98 ± 0.70 | 0.588 |
| Waist hip ratio | 0.82 ± 0.04 | 0.82 ± 0.04 | 0.688 |
| Waist-to-height ratio | 0.49 ± 0.05 | 0.49 ± 0.04 | 0.949 |
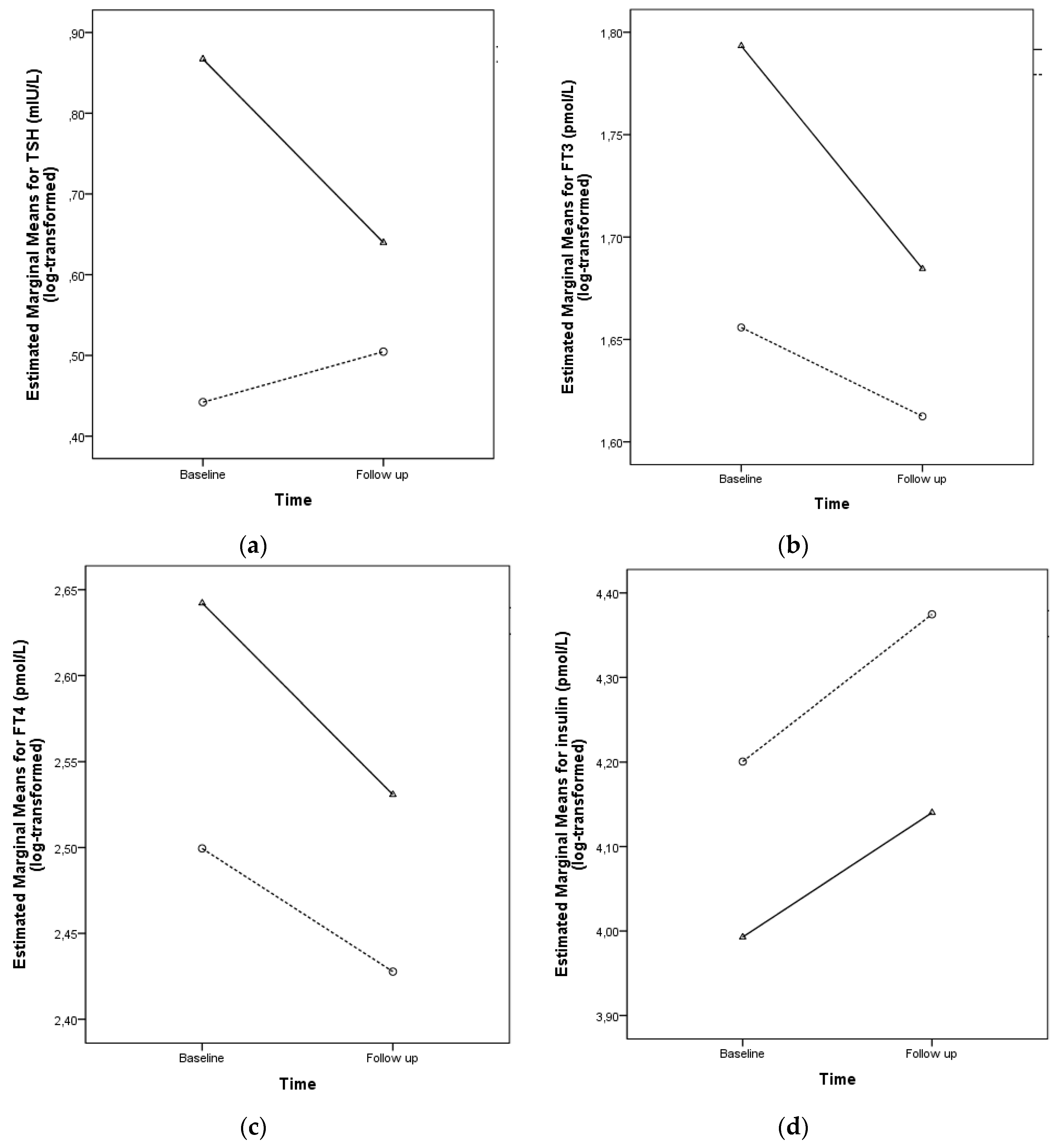
| TSH (mIU/L) † | 2.39 (2.12–2.69) | 1.48 (1.34–1.62) | <0.001 |
| FT3 (pmol/L) † | 5.93 (5.75–6.11) | 5.26 (5.10–5.36) | <0.001 |
| FT4 (pmol/L) † | 14.00 (13.59–14.29) | 12.17 (11.93–12.42) | <0.001 |
| Insulin (pmol/L) † | 54.03 (48.40–60.31) | 65.99 (59.71–72.20) | 0.008 |
| Glucose (mmol/L) | 5.14 ± 0.40 | 5.14 ± 0.33 | 0.903 |
| HOMA-IR † | 2.05 (1.82–2.31) | 2.50 (2.27–2.77) | 0.014 |
| Excess Weight (BMI ≥ 1 z Score) | Treatment 1 (n = 55, B18, G37) | Treatment 2 (n = 87, B43, G44) | pb |
|---|---|---|---|
| HAZ (z score) | −0.02 (−0.80–1.00) | 0.05 (−1.17–0.92) | 0.156 |
| BMI/A (z score) | −0.19 (−1.48–0.71) | −0.19 (−1.19–0.61) | 0.717 |
| Waist hip ratio | −0.01 (−0.07–0.06) | −0.01 (−0.09–0.09) | 0.650 |
| Waist-to-height ratio | −0.01 (−0.05–0.04) | −0.01 (−0.08–0.04) | 0.081 |
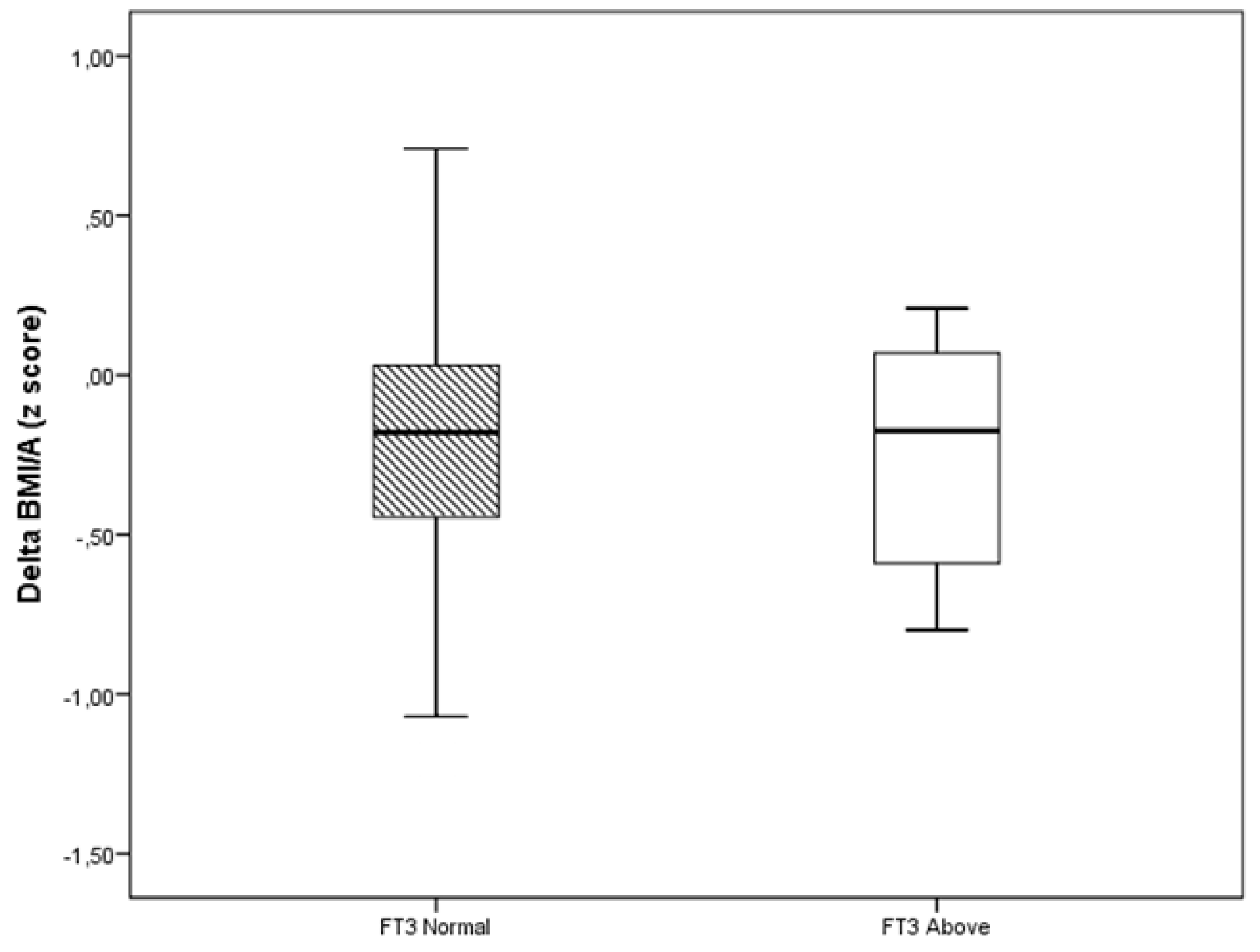
| TSH (mIU/L) | −0.45 (−3.19–2.17) | 0.06 (−4.57–1.63) | 0.001 |
| FT3 (pmol/L) | −0.46 (−2.92–1.54) | −0.15 (−2.46–1.38) | 0.038 |
| FT4 (pmol/L) | −1.41 (−6.18–3.47) | −0.90 (−4.89–2.96) | 0.018 |
| Insulin (pmol/L) | 8.13 (−91.62–80.82) | 11.40 (−87.00–150.00) | 0.688 |
| Glucose (mmol/L) | −0.16 (−0.72–0.94) | −0.05 (−1.17–1.33) | 0.227 |
| HOMA-IR | 0.20 (−3.79–3.10) | 0.40 (−3.92–7.13) | 0.473 |
| Type of Treatment | Baseline | |||
| FT3 Normal | FT3 Above | Total | p | |
| Treatment 1 n (%) | 41 (23.3) | 32 (18.1) | 73 (41.4) | |
| Treatment 2 n (%) | 89 (50.6) | 14 (8.0) | 103 (58.6) | 0.001 |
| Total | 130 (73.9) | 46 (26.1) | 176 (100) | |
| Type of treatment | Follow-up | |||
| FT3 Normal | FT3 Above | Total | p | |
| Treatment 1 n (%) | 49 (34.0) | 9 (6.2) | 58 (40.3) | |
| Treatment 2 n (%) | 83 (57.6) | 3 (2.1) | 86 (59.7) | 0.014 |
| Total | 132 (91.7) | 12 (8.3) | 144 (100) | |
| Total | Baseline | 16 Months | p Treatment | p Time | p Treatment × Time |
|---|---|---|---|---|---|
| Energy (kJ) | |||||
| Treatment 1 | 8575 (8116–9443) | 7304 (6806–7840) | 0.439 | <0.001 | <0.001 |
| Treatment 2 | 8233 (7815–8677) | 8267 (7739–8832) | |||
| Total Fat (g) | |||||
| Treatment 1 | 77 (70–84) | 62 (58–68) | 0.163 | 0.002 | <0.001 |
| Treatment 2 | 74 (69–78) | 74 (69–81) | |||
| Total carbohydrate (g) | |||||
| Treatment 1 | 268 (251–287) | 228 (211–246) | 0.774 | <0.001 | 0.004 |
| Treatment 2 | 252 (239–265) | 248 (233–264) | |||
| Total Protein (g) | |||||
| Treatment 1 | 84 (77–91) | 68 (64–73) | 0.761 | 0.010 | <0.001 |
| Treatment 2 | 75 (71–79) | 78 (73–84) | |||
| Sodium (mg) | |||||
| Treatment 1 | 3315 (3056–3595) | 2746 (2590–2911) | 0.176 | 0.004 | <0.001 |
| Treatment 2 | 3150 (2998–3310) | 3217 (2972–3482) | |||
| Sugar (g) | |||||
| Treatment 1 | 107 (98–117) | 84 (75–95) | 0.078 | <0.001 | 0.007 |
| Treatment 2 | 89 (82–96) | 83 (77–90) |
| Variables | Age (Years) | TSH (mIU/L) | FT3 (pmol/L) | FT4 (pmol/L) | Insulin (pmol/L) | Glucose (mmol/L) | HOMA-IR | HAZ (z Score) | BMI/A (z Score) | WHR | Waist to Height Ratio |
|---|---|---|---|---|---|---|---|---|---|---|---|
| TSH (mIU/L) † | −0.027 | ||||||||||
| FT3 (pmol/L)† | −0.152 * | 0.279 ** | |||||||||
| FT4 (pmol/L) † | −0.020 | 0.333 * | 0.364 ** | ||||||||
| Insulin (pmol/L) † | 0.053 | 0.075 | 0.114 | −0.155 * | |||||||
| Glucose (mmol/L) | 0.082 | 0.108 | 0.191 * | 0.078 | 0.307 ** | ||||||
| HOMA-IR † | 0.061 | 0.085 | 0.133 | −0.138 | 0.992 ** | 0.424 ** | |||||
| HAZ (z score) | −0.088 | −0.110 | 0.066 | −0.128 | 0.273** | 0.004 | 0.264 ** | ||||
| BMI/A (z score) | −0.051 | 0.087 | 0.212 ** | 0.047 | 0.463 ** | 0.116 | 0.460 ** | 0.311 ** | |||
| WHR | −0.236 ** | 0.103 | 0.104 | 0.041 | 0.201 ** | 0.031 | 0.195 * | −0.037 | 0.431 ** | ||
| Waist to height ratio | −0.077 | 0.139 | 0.230 ** | 0.071 | 0.451 ** | 0.083 | 0.443 ** | 0.037 | 0.844 ** | 0.700 ** | |
| Sugar (mg) | −0.039 | 0.183 * | 0.124 | 0.034 | −0.087 | −0.083 | −0.101 | 0.044 | 0.066 | 0.057 | 0.011 |
| Variables | Age (Years) | TSH (mIU/L) | FT3 (pmol/L) | FT4 (pmol/L) | Insulin (pmol/L) | Glucose (mmol/L) | HOMA-IR | HAZ (z Score) | BMI/A (z Score) | WHR | Waist to Height Ratio |
|---|---|---|---|---|---|---|---|---|---|---|---|
| TSH (mIU/L) † | 0.085 | ||||||||||
| FT3 (pmol/L) † | −0.227 ** | 0.118 | |||||||||
| FT4 (pmol/L) † | −0.030 | 0.119 | 0.270 ** | ||||||||
| Insulin (pmol/L) † | 0.119 | 0.079 | 0.041 | −0.221 ** | |||||||
| Glucose (mmol/L) | −0.015 | 0.137 | −0.053 | −0.209 * | 0.298 ** | ||||||
| HOMA-IR † | 0.067 | 0.062 | 0.006 | −0.260 ** | 0.922 ** | 0.373 ** | |||||
| HAZ (z score) | −0.126 | −0.104 | 0.092 | −0.140 | 0.318 ** | 0.149 | 0.274 ** | ||||
| BMI/A (z score) | −0.012 | 0.042 | 0.222 ** | 0.085 | 0.429 ** | −0.028 | 0.364 ** | 0.235 ** | |||
| WHR | −0.251 ** | 0.068 | 0.182 * | 0.065 | 0.190 * | −0.023 | 0.164 * | −0.069 | 0.420 ** | ||
| Waist to height ratio | 0.011 | 0.123 | 0.250 ** | 0.151 | 0.407 ** | −0.055 | 0.345 ** | −0.043 | 0.887 ** | 0.677 ** | |
| Sugar (mg) | 0.001 | 0.058 | −0.008 | 0.044 | −0.157 | −0.134 | −0.187 | 0.002 | −0.075 | −0.001 | −0.095 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martins, V.J.B.; Filgueiras, A.R.; Almeida, V.B.P.; de Moraes, R.C.S.; Sawaya, A.L. Changes in Thyroid and Glycemic Status and Food Intake in Children with Excess Weight Who Were Submitted for a Multi-Component School Intervention for 16 Months. Int. J. Environ. Res. Public Health 2020, 17, 3825. https://doi.org/10.3390/ijerph17113825
Martins VJB, Filgueiras AR, Almeida VBP, de Moraes RCS, Sawaya AL. Changes in Thyroid and Glycemic Status and Food Intake in Children with Excess Weight Who Were Submitted for a Multi-Component School Intervention for 16 Months. International Journal of Environmental Research and Public Health. 2020; 17(11):3825. https://doi.org/10.3390/ijerph17113825
Chicago/Turabian StyleMartins, Vinicius J. B., Andrea R. Filgueiras, Viviane B. P. Almeida, Rúbia C. S. de Moraes, and Ana L. Sawaya. 2020. "Changes in Thyroid and Glycemic Status and Food Intake in Children with Excess Weight Who Were Submitted for a Multi-Component School Intervention for 16 Months" International Journal of Environmental Research and Public Health 17, no. 11: 3825. https://doi.org/10.3390/ijerph17113825
APA StyleMartins, V. J. B., Filgueiras, A. R., Almeida, V. B. P., de Moraes, R. C. S., & Sawaya, A. L. (2020). Changes in Thyroid and Glycemic Status and Food Intake in Children with Excess Weight Who Were Submitted for a Multi-Component School Intervention for 16 Months. International Journal of Environmental Research and Public Health, 17(11), 3825. https://doi.org/10.3390/ijerph17113825





