Effectiveness of an Interdisciplinary Program Performed on Obese People Regarding Nutritional Habits and Metabolic Comorbidity: A Randomized Controlled Clinical Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Sample Size
2.3. Setting, Recruitment, and Selection
2.4. Intervention
2.4.1. The Interdisciplinary Intervention against Obesity
2.4.2. Control
2.5. Outcomes Measures
2.6. Statistical Analysis
2.7. Ethical Considerations
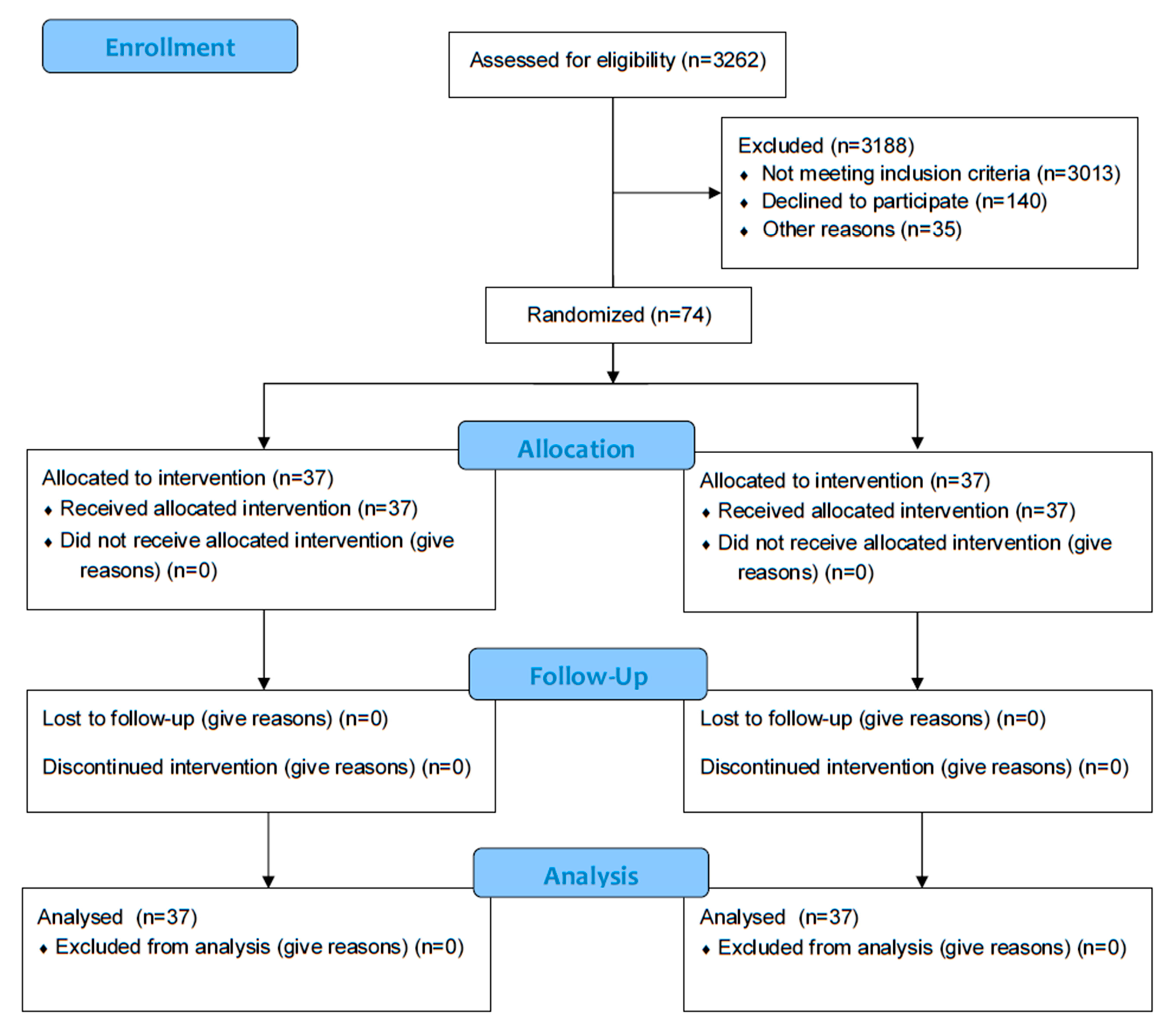
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Chooi, Y.C.; Ding, C.; Magkos, F. The epidemiology of obesity. Metabolism 2019, 92, 6–10. [Google Scholar] [CrossRef] [PubMed]
- Hernaez, A.; Zomeno, M.D.; Degano, I.R.; Perez-Fernandez, S.; Goday, A.; Vila, J.; Civeira, F.; Moure, R.; Marrugat, J. Excess Weight in Spain: Current Situation, Projections for 2030, and Estimated Direct Extra Cost for the Spanish Health System. Rev. Esp. Cardiol. 2019, 72, 916–924. [Google Scholar] [CrossRef] [PubMed]
- Grohmann, M.; Wiede, F.; Dodd, G.T.; Gurzov, E.N.; Ooi, G.J.; Butt, T.; Rasmiena, A.A.; Kaur, S.; Gulati, T.; Goh, P.K.; et al. Obesity Drives STAT-1-Dependent NASH and STAT-3-Dependent HCC. Cell 2018, 175, 1289–1306. [Google Scholar] [CrossRef] [PubMed]
- Al-Thani, M.H.; Al-Thani, A.A.M.; Cheema, S.; Sheikh, J.; Mamtani, R.; Lowenfels, A.B.; Al-Chetachi, W.F.; Almalki, B.A.; Hassan Khalifa, S.A.; Haj Bakri, A.O.; et al. Prevalence and determinants of metabolic syndrome in Qatar: Results from a National Health Survey. BMJ Open 2016, 6, e009514. [Google Scholar] [CrossRef]
- Tran, V.D.; Lee, A.H.; Jancey, J.; James, A.P.; Howat, P.; Thi Phuong Mai, L. Community-based physical activity and nutrition programme for adults with metabolic syndrome in Vietnam: Study protocol for a cluster-randomised controlled trial. BMJ Open 2016, 6, e011532. [Google Scholar] [CrossRef]
- Cordova Villalobos, J.A. Obesity: The real pandemic of the 21(st) century. Cir. Cir. 2016, 84, 351–355. [Google Scholar]
- Bloomfield, H.E.; Koeller, E.; Greer, N.; MacDonald, R.; Kane, R.; Wilt, T.J. Effects on Health Outcomes of a Mediterranean Diet With No Restriction on Fat Intake: A Systematic Review and Meta-analysis. Ann. Intern. Med. 2016, 165, 491–500. [Google Scholar] [CrossRef]
- Park, Y.-M.; Steck, S.E.; Fung, T.T.; Zhang, J.; Hazlett, L.J.; Han, K.; Merchant, A.T. Mediterranean diet and mortality risk in metabolically healthy obese and metabolically unhealthy obese phenotypes. Int. J. Obes. 2016, 40, 1541–1549. [Google Scholar] [CrossRef]
- Greer, N.; Foman, N.A.; MacDonald, R.; Dorrian, J.; Fitzgerald, P.; Rutks, I.; Wilt, T.J. Advanced wound care therapies for nonhealing diabetic, venous, and arterial ulcers: A systematic review. Ann. Intern. Med. 2013, 159, 532–542. [Google Scholar] [CrossRef]
- Martinez-Gonzalez, M.A. Benefits of the Mediterranean diet beyond the Mediterranean Sea and beyond food patterns. BMC Med. 2016, 14, 157. [Google Scholar] [CrossRef]
- Estruch, R.; Ros, E.; Salas-Salvado, J.; Covas, M.-I.; Corella, D.; Aros, F.; Gomez-Gracia, E.; Ruiz-Gutierrez, V.; Fiol, M.; Lapetra, J.; et al. Primary prevention of cardiovascular disease with a Mediterranean diet. N. Engl. J. Med. 2013, 368, 1279–1290. [Google Scholar] [CrossRef] [PubMed]
- Shaw, K.; Gennat, H.; O’Rourke, P.; Del Mar, C. Exercise for overweight or obesity. Cochrane Database Syst. Rev. 2006, CD003817. [Google Scholar] [CrossRef] [PubMed]
- Clini, E.; Prato, F.; Nobile, M.; Bondi, M.; Serri, B.; Cilione, C.; Lugli, D. Interdisciplinary rehabilitation in morbidly obese subjects: An observational pilot study. Monaldi Arch. Chest Dis. Arch. Monaldi Per Le Mal. Del Torace 2006, 65, 89–95. [Google Scholar] [CrossRef][Green Version]
- Sakulsupsiri, A.; Sakthong, P.; Winit-Watjana, W. Cost-Effectiveness Analysis of the Self-Management Program for Thai Patients with Metabolic Syndrome. Value Heal. Reg. Issues 2016, 9, 28–35. [Google Scholar] [CrossRef]
- Zhang, Y.; Mei, S.; Yang, R.; Chen, L.; Gao, H.; Li, L. Effects of lifestyle intervention using patient-centered cognitive behavioral therapy among patients with cardio-metabolic syndrome: A randomized, controlled trial. BMC Cardiovasc. Disord. 2016, 16, 227. [Google Scholar] [CrossRef][Green Version]
- Gao, J.; Wang, J.; Zheng, P.; Haardörfer, R.; Kegler, M.C.; Zhu, Y.; Fu, H. Effects of self-care, self-efficacy, social support on glycemic control in adults with type 2 diabetes. BMC Fam. Pract. 2013, 14, 66. [Google Scholar] [CrossRef]
- Jezewska-Zychowicz, M.; Gębski, J.; Plichta, M.; Guzek, D.; Kosicka-Gębska, M. Diet-Related Factors, Physical Activity, and Weight Status in Polish Adults. Nutrients 2019, 11, 2532. [Google Scholar] [CrossRef]
- Johns, D.J.; Hartmann-Boyce, J.; Jebb, S.A.; Aveyard, P.; Group, B.W.M.R. Diet or exercise interventions vs combined behavioral weight management programs: A systematic review and meta-analysis of direct comparisons. J. Acad. Nutr. Diet. 2014, 114, 1557–1568. [Google Scholar] [CrossRef]
- Mottalib, A.; Tomah, S.; Hafida, S.; Elseaidy, T.; Kasetty, M.; Ashrafzadeh, S.; Hamdy, O. Intensive multidisciplinary weight management in patients with type 1 diabetes and obesity: A one-year retrospective matched cohort study. Diabetes. Obes. Metab. 2019, 21, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Konig, D.; Hormann, J.; Predel, H.-G.; Berg, A. A 12-Month Lifestyle Intervention Program Improves Body Composition and Reduces the Prevalence of Prediabetes in Obese Patients. Obes. Facts 2018, 11, 393–399. [Google Scholar] [CrossRef]
- Lee, B.Y.; Bartsch, S.M.; Mui, Y.; Haidari, L.A.; Spiker, M.L.; Gittelsohn, J. A systems approach to obesity. Nutr. Rev. 2017, 75, 94–106. [Google Scholar] [CrossRef]
- Busetto, L.; Dicker, D.; Azran, C.; Batterham, R.L.; Farpour-Lambert, N.; Fried, M.; Hjelmesaeth, J.; Kinzl, J.; Leitner, D.R.; Makaronidis, J.M.; et al. Obesity Management Task Force of the European Association for the Study of Obesity Released “Practical Recommendations for the Post-Bariatric Surgery Medical Management”. Obes. Surg. 2018, 28, 2117–2121. [Google Scholar] [CrossRef] [PubMed]
- Steinberger, J.; Daniels, S.R.; Eckel, R.H.; Hayman, L.; Lustig, R.H.; McCrindle, B.; Mietus-Snyder, M.L. Progress and challenges in metabolic syndrome in children and adolescents: A scientific statement from the American Heart Association Atherosclerosis, Hypertension, and Obesity in the Young Committee of the Council on Cardiovascular Disease in the Young. Circulation 2009, 119, 628–647. [Google Scholar] [CrossRef] [PubMed]
- Murcian Health Service. Cartera de Servicios del Servicio Murciano de Salud; Murcian Health Service: Murcia, Spain, 2007; ISBN 978-84-95393-86-9. [Google Scholar]
- Rodriguez, N.R.; DiMarco, N.M.; Langley, S. Position of the American Dietetic Association, Dietitians of Canada, and the American College of Sports Medicine: Nutrition and athletic performance. J. Am. Diet. Assoc. 2009, 109, 509–527. [Google Scholar] [PubMed]
- Nanri, A.; Tomita, K.; Matsushita, Y.; Ichikawa, F.; Yamamoto, M.; Nagafuchi, Y.; Kakumoto, Y.; Mizoue, T. Effect of six months lifestyle intervention in Japanese men with metabolic syndrome: Randomized controlled trial. J. Occup. Health 2012, 54, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Dapcich, V.; Salvador Caste, G.; Ribas Barba, L.; Pérez Rodrigo, C.; Aranceta Bartrina, J.; Serra Majem, L. Guía de la Alimentación Saludable; Editado por la Sociedad Espanolade Nutricion Comunitaria, Ed.; Sociedad Española de Nutrición Comunitaria: Madrid, Spain, 2004. [Google Scholar]
- Simonelli Muñoz, A.J. Esquizofrenia y Obesidad: Influencia de Los Hábitos Alimenticios, Tratamiento Farmacológico y Aspectos Sociofamiliares. Ph.D. Thesis, The University Catholic San Antonio, San Antonio, TX, USA, 2008. [Google Scholar]
- Soler Marín, A. Metodología de Intervención Nutricional en un Colectivo Especial. Personas con Síndrome de Down. Ph.D. Thesis, The University Catholic San Antonio, San Antonio, TX, USA, 2004. [Google Scholar]
- Calderone, A.; Calabro, P.F.; Lippi, C.; Jaccheri, R.; Vitti, J.; Santini, F. Psychopathological Behaviour and Cognition in Morbid Obesity. Recent Pat. Endocr. Metab. Immune Drug Discov. 2017, 10, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Martins, A.D.; Majzoub, A.; Agawal, A. Metabolic Syndrome and Male Fertility. World J. Mens. Health 2019, 37, 113–127. [Google Scholar] [CrossRef]
- Jinnett, K.; Kyle, T.; Parry, T.; Stevenin, B.; Ramasamy, A. Insights into the Role of Employers Supporting Obesity Management in People with Obesity: Results of the National ACTION Study. Popul. Health Manag. 2019, 22, 308–314. [Google Scholar] [CrossRef]
- Sureshchandra, S.; Marshall, N.E.; Wilson, R.M.; Barr, T.; Rais, M.; Purnell, J.Q.; Thornburg, K.L.; Messaoudi, I. Inflammatory Determinants of Pregravid Obesity in Placenta and Peripheral Blood. Front. Physiol. 2018, 9, 1089. [Google Scholar] [CrossRef]
- Mehta, L.S.; Watson, K.E.; Barac, A.; Beckie, T.M.; Bittner, V.; Cruz-Flores, S.; Dent, S.; Kondapalli, L.; Ky, B.; Okwuosa, T.; et al. Cardiovascular Disease and Breast Cancer: Where These Entities Intersect: A Scientific Statement From the American Heart Association. Circulation 2018, 137, e30–e66. [Google Scholar] [CrossRef]
- Montesi, L.; El Ghoch, M.; Brodosi, L.; Calugi, S.; Marchesini, G.; Dalle Grave, R. Long-term weight loss maintenance for obesity: A multidisciplinary approach. Diabetes. Metab. Syndr. Obes. 2016, 9, 37–46. [Google Scholar] [PubMed]
- Barnhill, A.; Palmer, A.; Weston, C.M.; Brownell, K.D.; Clancy, K.; Economos, C.D.; Gittelsohn, J.; Hammond, R.A.; Kumanyika, S.; Bennett, W.L. Grappling With Complex Food Systems to Reduce Obesity: A US Public Health Challenge. Public Health Rep. 2018, 133, 44S–53S. [Google Scholar] [CrossRef] [PubMed]
- Saboya, P.P.; Bodanese, L.C.; Zimmermann, P.R.; Gustavo, A.d.S.; Macagnan, F.E.; Feoli, A.P.; Oliveira, M.D.S. Lifestyle Intervention on Metabolic Syndrome and its Impact on Quality of Life: A Randomized Controlled Trial. Arq. Bras. Cardiol. 2017, 108, 60–69. [Google Scholar] [PubMed]
- Wang, Y.; Xue, H.; Huang, Y.; Huang, L.; Zhang, D. A Systematic Review of Application and Effectiveness of mHealth Interventions for Obesity and Diabetes Treatment and Self-Management. Adv. Nutr. 2017, 8, 449–462. [Google Scholar] [CrossRef]
- Kim, Y.J.; Hwang, J.-Y.; Kim, H.; Park, S.; Kwon, O. Diet quality, physical activity, and their association with metabolic syndrome in Korean adults. Nutrition 2019, 59, 138–144. [Google Scholar] [CrossRef]
- Wing, R.R.; Lang, W.; Wadden, T.A.; Safford, M.; Knowler, W.C.; Bertoni, A.G.; Hill, J.O.; Brancati, F.L.; Peters, A.; Wagenknecht, L. Benefits of modest weight loss in improving cardiovascular risk factors in overweight and obese individuals with type 2 diabetes. Diabetes Care 2011, 34, 1481–1486. [Google Scholar] [CrossRef]
- Salas-Salvado, J.; Diaz-Lopez, A.; Ruiz-Canela, M.; Basora, J.; Fito, M.; Corella, D.; Serra-Majem, L.; Warnberg, J.; Romaguera, D.; Estruch, R.; et al. Effect of a Lifestyle Intervention Program With Energy-Restricted Mediterranean Diet and Exercise on Weight Loss and Cardiovascular Risk Factors: One-Year Results of the PREDIMED-Plus Trial. Diabetes Care 2019, 42, 777–788. [Google Scholar] [CrossRef]
- Berzigotti, A.; Albillos, A.; Villanueva, C.; Genesca, J.; Ardevol, A.; Augustin, S.; Calleja, J.L.; Banares, R.; Garcia-Pagan, J.C.; Mesonero, F.; et al. Effects of an intensive lifestyle intervention program on portal hypertension in patients with cirrhosis and obesity: The SportDiet study. Hepatology 2017, 65, 1293–1305. [Google Scholar] [CrossRef]
- Champagne, C.M.; Broyles, S.T.; Moran, L.D.; Cash, K.C.; Levy, E.J.; Lin, P.-H.; Batch, B.C.; Lien, L.F.; Funk, K.L.; Dalcin, A.; et al. Dietary intakes associated with successful weight loss and maintenance during the Weight Loss Maintenance trial. J. Am. Diet. Assoc. 2011, 111, 1826–1835. [Google Scholar] [CrossRef]
- Estruch, R.; Salas-Salvado, J. Towards an even healthier Mediterranean diet. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 1163–1166. [Google Scholar] [CrossRef]
- Dalle Grave, R.; Calugi, S.; Marchesini, G. The influence of cognitive factors in the treatment of obesity: Lessons from the QUOVADIS study. Behav. Res. Ther. 2014, 63, 157–161. [Google Scholar] [CrossRef] [PubMed]
- Thom, G.; Lean, M. Is There an Optimal Diet for Weight Management and Metabolic Health? Gastroenterology 2017, 152, 1739–1751. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.M.; Wallace, E.; O’Dowd, T.; Fortin, M. Interventions for improving outcomes in patients with multimorbidity in primary care and community settings. Cochrane Database Syst. Rev. 2016, 3. [Google Scholar] [CrossRef] [PubMed]
| CG n = 37 | EG n = 37 | Test | |||||
|---|---|---|---|---|---|---|---|
| M/N | SD/% | M/N | SD/% | Statistic | Df | P | |
| Age (years) | 62.8 | 8.9 | 59.4 | 9.1 | 1.646 ª | 72 | 0.104 |
| Gender (male) | 18 | 48.6 | 19 | 51.4 | 0.000 b | 1 | 1.000 |
| BMI (kg/m2) | 34.3 | 4.5 | 32.4 | 3.8 | 1.979 ª | 72 | 0.052 |
| Smoking | |||||||
| Non-smoker | 26 | 70.3 | 20 | 54.1 | 2.177 b | 2 | 0.337 |
| Former smoker | 9 | 24.3 | 13 | 35.1 | |||
| Smoker | 2 | 5.4 | 4 | 10.8 | |||
| Alcohol | 22 | 59.5 | 21 | 56.8 | 0.000 b | 1 | 1.000 |
| DM | 23 | 62.2 | 16 | 43.2 | 1.952 b | 1 | 0.162 |
| HTN | 32 | 86.5 | 29 | 78.4 | 0.373 b | 1 | 0.541 |
| DLP | 21 | 56.8 | 19 | 51.4 | 0.054 b | 1 | 0.816 |
| HD | 1 | 2.7 | 1 | 2.7 | 0.000 b | 1 | 1.000 |
| Control n = 37 | Intervention n = 37 | Interaction | Moment | Group | ||||
|---|---|---|---|---|---|---|---|---|
| M | SD | M | SD | F2;144 p η2 | F2;144 p η2 | F1;72 p η2 | ||
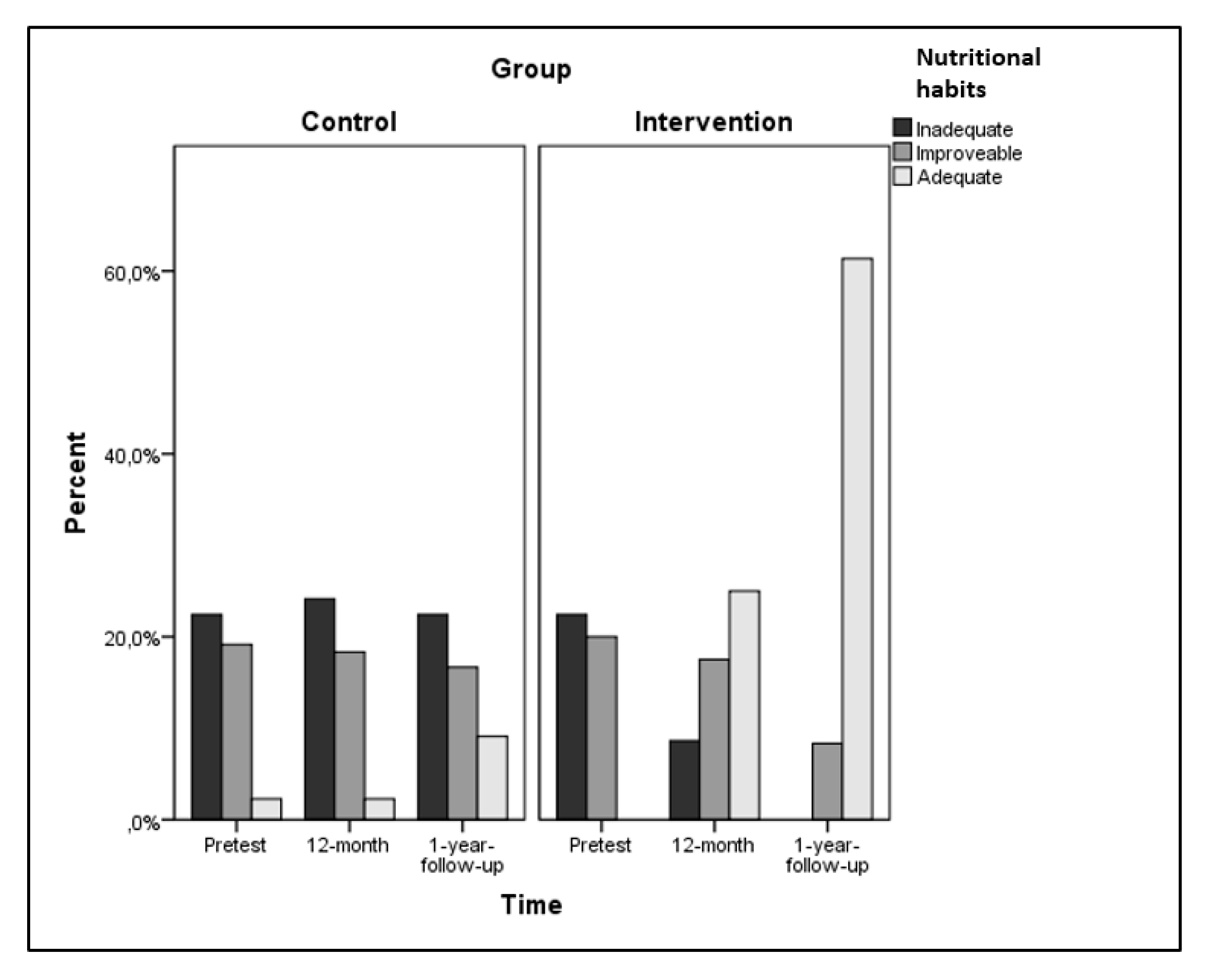
| Nutritional Habits a | Pretest | 28.5 | 5.3 | 28.5 | 6.2 | 52.029 | 63.928 | 28.183 |
| 12-month | 27.9 | 5.3 | 33.2 | 7.1 | <0.001 | <0.001 | <0.001 | |
| Follow-up | 29.1 | 7.4 | 43.2 | 6.2 | 0.419 | 0.470 | 0.281 | |
| FG1 b | Pretest | 3.1 | 0.4 | 3.0 | 0.4 | 0.286 | 0.087 | 0.670 |
| 12-month | 3.1 | 0.3 | 3.0 | 0.3 | 0.752 | 0.917 | 0.416 | |
| Follow-up | 3.1 | 0.4 | 3.0 | 0.3 | 0.004 | 0.001 | 0.009 | |
| FG2 c | Pretest | 3.2 | 0.4 | 3.1 | 0.4 | 1.679 | 2.480 | 0.837 |
| 12-month | 3.2 | 0.4 | 3.1 | 0.4 | 0.190 | 0.087 | 0.363 | |
| Follow-up | 3.2 | 0.5 | 3.2 | 0.3 | 0.023 | 0.033 | 0.011 | |
| FG3 d | Pretest | 3.5 | 0.6 | 3.0 | 0.5 | 26.034 | 15.588 | 0.254 |
| 12-month | 3.5 | 0.6 | 3.5 | 0.5 | <0.001 | <0.001 | 0.616 | |
| Follow-up | 3.4 | 0.5 | 3.6 | 0.4 | 0.266 | 0.178 | 0.004 | |
| FG4 e | Pretest | 2.6 | 0.6 | 2.4 | 0.5 | 2.280 | 0.233 | 1.713 |
| 12-month | 2.5 | 0.5 | 2.5 | 0.4 | 0.106 | 0.792 | 0.195 | |
| Follow-up | 2.6 | 0.5 | 2.5 | 0.4 | 0.031 | 0.003 | 0.023 | |
| FG5 f | Pretest | 2.6 | 0.6 | 2.3 | 0.5 | 35.440 | 5.845 | 25.981 |
| 12-month | 2.6 | 0.7 | 2.0 | 0.4 | <0.001 | 0.004 | <0.001 | |
| Follow-up | 2.9 | 0.6 | 1.9 | 0.3 | 0.330 | 0.075 | 0.265 | |
| FG6 g | Pretest | 2.5 | 0.8 | 2.5 | 0.8 | 1.641 | 0.061 | 0.027 |
| 12-month | 2.5 | 0.9 | 2.5 | 0.6 | 0.197 | 0.941 | 0.870 | |
| Follow-up | 2.4 | 0.9 | 2.5 | 0.6 | 0.022 | 0.001 | 0.000 | |
| FG7 h | Pretest | 1.7 | 0.9 | 1.7 | 1.1 | 8.114 | 2.874 | 4.000 |
| 12-month | 1.6 | 1.0 | 2.2 | 1.4 | <0.001 | 0.060 | 0.049 | |
| Follow-up | 1.5 | 1.1 | 2.4 | 1.4 | 0.101 | 0.038 | 0.053 | |
| CG n = 37 | EG n = 37 | Interaction | Moment | Group | ||||
|---|---|---|---|---|---|---|---|---|
| M | SD | M | SD | F3;216 p η2 | F3;216 p η2 | F1;72 p η2 | ||
| SBP a (mmHg) | Pretest | 152.3 | 11.8 | 150.6 | 12.9 | 43.948 | 168.376 | 47.033 |
| 12-Month | 144.1 | 7.3 | 127.3 | 7.0 | <0.001 | <0.001 | <0.001 | |
| Follow-up | 145.5 | 6.6 | 127.6 | 5.8 | 0.379 | 0.700 | 0.395 | |
| DBP b (mmHg) | Pretest | 87.3 | 6.8 | 87.6 | 5.8 | 87.778 | 347.916 | 39.925 |
| 12-Month | 80.3 | 4.7 | 71.5 | 4.2 | <0.001 | <0.001 | <0.001 | |
| Follow-up | 83.0 | 5.3 | 70.9 | 3.9 | 0.549 | 0.829 | 0.357 | |
| TC c (mg/dL) | Pretest | 214.5 | 26.4 | 217.6 | 40.4 | 94.240 | 79.434 | 46.577 |
| 12-Month | 207.4 | 32.2 | 157.2 | 24.0 | <0.001 | <0.001 | <0.001 | |
| Follow-up | 223.4 | 33.3 | 145.9 | 19.2 | 0.567 | 0.525 | 0.393 | |
| TG d (mg/dL) | Pretest | 147.5 | 84.4 | 171.5 | 81.9 | 30.066 | 18.293 | 2.009 |
| 12-Month | 143.2 | 62.7 | 115.5 | 36.0 | <0.001 | <0.001 | 0.161 | |
| Follow-up | 164.9 | 69.7 | 110.3 | 28.9 | 0.295 | 0.203 | 0.027 | |
| Glucose e (mg/dL) | Pretest | 139.1 | 33.4 | 132.9 | 36.0 | 27.584 | 46.603 | 40.406 |
| 12-Month | 129.1 | 27.2 | 88.4 | 9.7 | <0.001 | <0.001 | <0.001 | |
| Follow-up | 136.6 | 31.4 | 86.8 | 6.8 | 0.277 | 0.393 | 0.359 | |
| HbA1c f (mg/dL) | Pretest | 7.2 | 1.1 | 6.7 | 1.2 | 54.957 | 65.294 | 64.781 |
| 12-Month | 7.0 | 1.1 | 5.1 | 0.3 | <0.001 | <0.001 | <0.001 | |
| Follow-up | 7.3 | 1.1 | 5.1 | 0.2 | 0.433 | 0.476 | 0.474 | |
| GOT g (U/L) | Pretest | 22.7 | 9.7 | 23.0 | 10.7 | 8.248 | 4.192 | 4.565 |
| 12-Month | 22.0 | 6.5 | 19.1 | 2.8 | <0.001 | 0.017 | 0.036 | |
| Follow-up | 24.6 | 6.1 | 18.5 | 2.1 | 0.103 | 0.055 | 0.060 | |
| GPT h (U/L) | Pretest | 24.8 | 13.8 | 28.5 | 18.3 | 9.394 | 11.180 | 1.253 |
| 12-Month | 23.1 | 8.4 | 19.6 | 5.9 | <0.001 | <0.001 | 0.267 | |
| Follow-up | 25.1 | 6.7 | 18.1 | 2.6 | 0.115 | 0.134 | 0.017 | |
| GGT i (U/L) | Pretest | 25.2 | 14.0 | 30.8 | 19.7 | 16.587 | 12.844 | 1.796 |
| 12-Month | 24.4 | 11.3 | 19.0 | 4.5 | <0.001 | <0.001 | 0.184 | |
| Follow-up | 27.4 | 11.2 | 17.9 | 1.7 | 0.187 | 0.151 | 0.024 | |
| TB j (mg/dL) | Pretest | 0.5 | 0.2 | 0.5 | 0.2 | 20.822 | 5.964 | 1.104 |
| 12-Month | 0.5 | 0.2 | 0.6 | 0.3 | <0.001 | 0.003 | 0.297 | |
| Follow-up | 0.5 | 0.2 | 0.5 | 0.1 | 0.224 | 0.076 | 0.015 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fernández-Ruiz, V.E.; Ramos-Morcillo, A.J.; Solé-Agustí, M.; Paniagua-Urbano, J.A.; Armero-Barranco, D. Effectiveness of an Interdisciplinary Program Performed on Obese People Regarding Nutritional Habits and Metabolic Comorbidity: A Randomized Controlled Clinical Trial. Int. J. Environ. Res. Public Health 2020, 17, 336. https://doi.org/10.3390/ijerph17010336
Fernández-Ruiz VE, Ramos-Morcillo AJ, Solé-Agustí M, Paniagua-Urbano JA, Armero-Barranco D. Effectiveness of an Interdisciplinary Program Performed on Obese People Regarding Nutritional Habits and Metabolic Comorbidity: A Randomized Controlled Clinical Trial. International Journal of Environmental Research and Public Health. 2020; 17(1):336. https://doi.org/10.3390/ijerph17010336
Chicago/Turabian StyleFernández-Ruiz, Virginia Esperanza, Antonio Jesús Ramos-Morcillo, María Solé-Agustí, José Antonio Paniagua-Urbano, and David Armero-Barranco. 2020. "Effectiveness of an Interdisciplinary Program Performed on Obese People Regarding Nutritional Habits and Metabolic Comorbidity: A Randomized Controlled Clinical Trial" International Journal of Environmental Research and Public Health 17, no. 1: 336. https://doi.org/10.3390/ijerph17010336
APA StyleFernández-Ruiz, V. E., Ramos-Morcillo, A. J., Solé-Agustí, M., Paniagua-Urbano, J. A., & Armero-Barranco, D. (2020). Effectiveness of an Interdisciplinary Program Performed on Obese People Regarding Nutritional Habits and Metabolic Comorbidity: A Randomized Controlled Clinical Trial. International Journal of Environmental Research and Public Health, 17(1), 336. https://doi.org/10.3390/ijerph17010336






