Effect of Exergame Training and Detraining on Lower-Body Strength, Agility, and Cardiorespiratory Fitness in Women with Fibromyalgia: Single-Blinded Randomized Controlled Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Trial Design
2.2. Participants
- Female and aged between 30 and 75 years;
- able to communicate with research staff;
- had given their informed consent; and
- diagnosed with FM by a rheumatologist according to 2010 American College of Rheumatology criteria [1].
2.3. Intervention
- A warm-up where participants were guided by a video made by a kinesiologist;
- aerobic exercises based on dance steps shown by a dance teacher;
- postural control and coordination games where participants had to reach for an apple that came and went in different locations around them (the kinesiologist could manually control the body part that participants had to use to reach the apple); and
- walking training where participants had to follow a virtual trail of footprints. The type and amplitude of steps were controlled.
2.4. Outcomes
- Walking MET-minutes/week = 3.3 × walking minutes × walking days;
- moderate MET-minutes/week = 4.0 × moderate-intensity activity minutes × moderate-intensity days; and
- vigorous MET-minutes/week = 8.0 × vigorous-intensity activity minutes × vigorous-intensity days.
2.5. Statistical Analysis
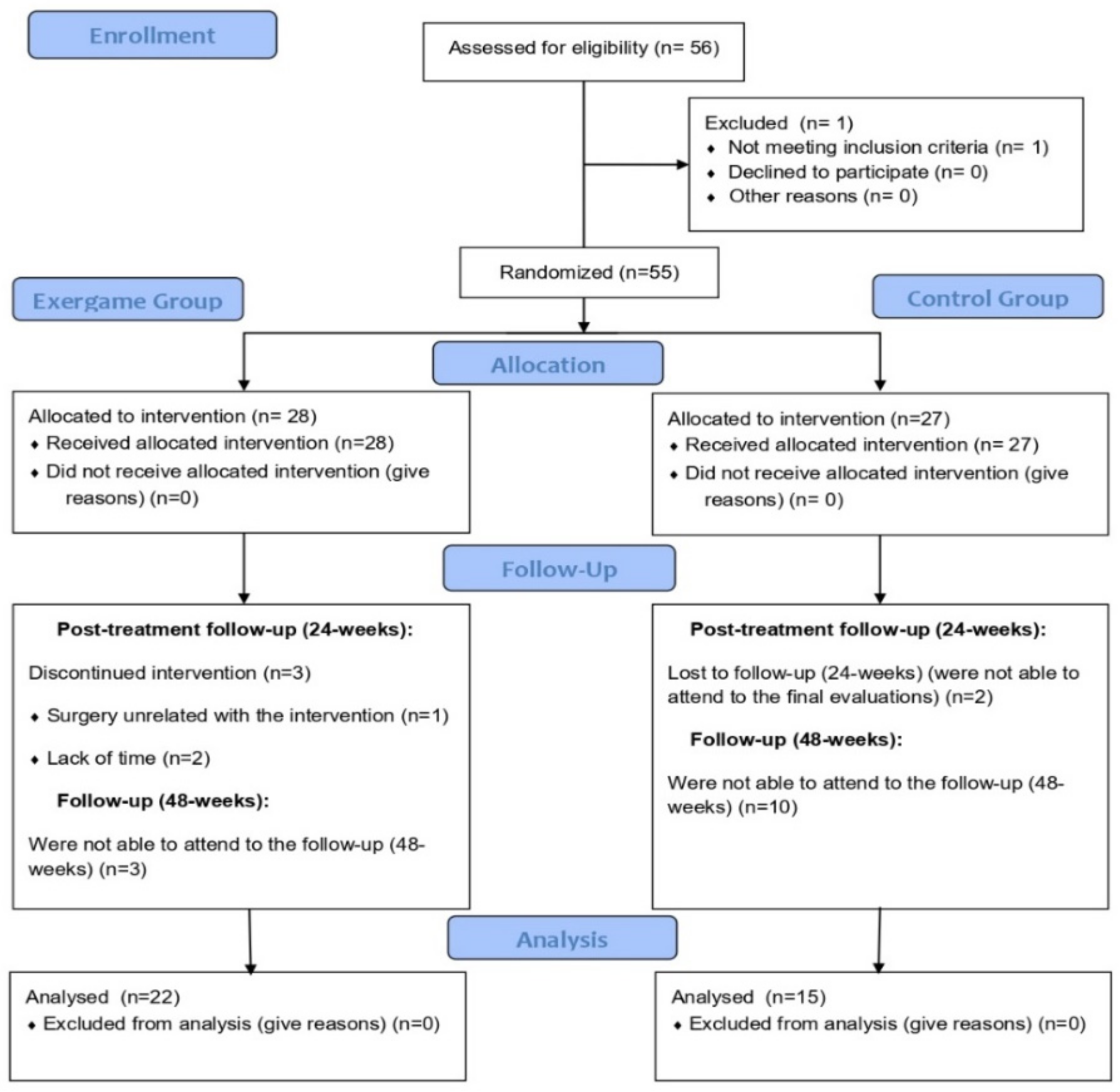
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wolfe, F.; Clauw, D.J.; Fitzcharles, M.A.; Goldenberg, D.L.; Katz, R.S.; Mease, P.; Russell, A.S.; Russell, I.J.; Winfield, J.B.; Yunus, M.B. The American College of Rheumatology Preliminary Diagnostic Criteria for Fibromyalgia and Measurement of Symptom Severity. Arthrit. Care Res. 2010, 62, 600–610. [Google Scholar] [CrossRef] [PubMed]
- Walker, J. Fibromyalgia: Clinical features, diagnosis and management. Nurs. Stand. 2016, 31, 51–63. [Google Scholar] [CrossRef] [PubMed]
- Verbunt, J.A.; Pernot, D.H.F.M.; Smeets, R.J.E.M. Disability and quality of life in patients with fibromyalgia. Health Qual. Out. 2008, 6, 8. [Google Scholar] [CrossRef] [PubMed]
- Macfarlane, G.J.; Kronisch, C.; Dean, L.E.; Atzeni, F.; Hauser, W.; Fluss, E.; Choy, E.; Kosek, E.; Amris, K.; Branco, J.; et al. EULAR revised recommendations for the management of fibromyalgia. Ann. Rheum. Dis. 2017, 76, 318–328. [Google Scholar] [CrossRef]
- Fitzcharles, M.A.; Ste-Marie, P.A.; Goldenberg, D.L.; Pereira, J.X.; Abbey, S.; Choiniere, M.; Ko, G.; Moulin, D.E.; Panopalis, P.; Proulx, J.; et al. 2012 Canadian Guidelines for the diagnosis and management of fibromyalgia syndrome: Executive summary. Pain Res. Manag. 2013, 18, 119–126. [Google Scholar] [CrossRef]
- Bidonde, J.; Busch, A.J.; Schachter, C.L.; Overend, T.J.; Kim, S.Y.; Goes, S.M.; Boden, C.; Foulds, H.J. Aerobic exercise training for adults with fibromyalgia. Cochrane Database Syst. Rev. 2017, 6, Cd012700. [Google Scholar] [CrossRef]
- Wang, C.; Schmid, C.H.; Fielding, R.A.; Harvey, W.F.; Reid, K.F.; Price, L.L.; Driban, J.B.; Kalish, R.; Rones, R.; McAlindon, T. Effect of tai chi versus aerobic exercise for fibromyalgia: Comparative effectiveness randomized controlled trial. BMJ 2018, 360, k851. [Google Scholar] [CrossRef]
- Busch, A.; Barber, K.; Overend, T.; Peloso, P.; Schachter, C. Exercise for treating fibromyalgia syndrome. Cochrane Database Syst. Rev. 2007. [Google Scholar] [CrossRef]
- Júnior, J.C.A.; de Almeida Silva, H.J.; da Silva, J.F.C.; da Silva Cruz, R.; de Almeida Lins, C.A.; de Souza, M.C. Zumba dancing can improve the pain and functional capacity in women with fibromyalgia. J. Bodyw. Mov. Ther. 2018, 22, 455–459. [Google Scholar] [CrossRef]
- Gavi, M.; Vassalo, D.V.; Amaral, F.T.; Macedo, D.C.F.; Gava, P.L.; Dantas, E.M.; Valim, V. Strengthening Exercises Improve Symptoms and Quality of Life but Do Not Change Autonomic Modulation in Fibromyalgia: A Randomized Clinical Trial. PLoS ONE 2014, 9, e90767. [Google Scholar] [CrossRef]
- Kayo, A.H.; Peccin, M.S.; Sanches, C.M.; Trevisani, V.F.M. Effectiveness of physical activity in reducing pain in patients with fibromyalgia: A blinded randomized clinical trial. Rheumatol. Int. 2012, 32, 2285–2292. [Google Scholar] [CrossRef] [PubMed]
- Palstam, A.; Larsson, A.; Lofgren, M.; Ernberg, M.; Bjersing, J.; Bileviciute-Ljungar, I.; Gerdle, B.; Kosek, E.; Mannerkorpi, K. Decrease of fear avoidance beliefs following person-centered progressive resistance exercise contributes to reduced pain disability in women with fibromyalgia: Secondary exploratory analyses from a randomized controlled trial. Arthritis Res. Ther. 2016, 18. [Google Scholar] [CrossRef] [PubMed]
- Ericsson, A.; Palstam, A.; Larsson, A.; Lofgren, M.; Bileviciute-Ljungar, I.; Bjersing, J.; Gerdle, B.; Kosek, E.; Mannerkorpi, K. Resistance exercise improves physical fatigue in women with fibromyalgia: A randomized controlled trial. Arthritis Res. Ther. 2016, 18. [Google Scholar] [CrossRef] [PubMed]
- Assumpcao, A.; Matsutani, L.A.; Yuan, S.L.; Santo, A.S.; Sauer, J.; Mango, P.; Marques, A.P. Muscle stretching exercises and resistance training in fibromyalgia: Which is better? A three-arm randomized controlled trial. Eur. J. Phys. Rehabil. Med. 2018, 54, 663–670. [Google Scholar] [CrossRef] [PubMed]
- Wuest, S.; van de Langenberg, R.; de Bruin, E.D. Design considerations for a theory-driven exergame-based rehabilitation program to improve walking of persons with stroke. Eur. Rev. Aging Phys. A 2014, 11, 119–129. [Google Scholar] [CrossRef]
- Collado-Mateo, D.; Dominguez-Munoz, F.J.; Adsuar, J.C.; Garcia-Gordillo, M.A.; Gusi, N. Effects of Exergames on Quality of Life, Pain, and Disease Effect in Women With Fibromyalgia: A Randomized Controlled Trial. Arch. Phys. Med. Rehab. 2017, 98, 1725–1731. [Google Scholar] [CrossRef]
- Collado-Mateo, D.; Dominguez-Muñoz, F.J.; Adsuar, J.C.; Merellano-Navarro, E.; Gusi, N. Exergames for women with fibromyalgia: A randomised controlled trial to evaluate the effects on mobility skills, balance and fear of falling. PeerJ 2017, 5, e3211. [Google Scholar] [CrossRef]
- Villafaina, S.; Collado-Mateo, D.; Dominguez-Munoz, F.J.; Fuentes-Garcia, J.P.; Gusi, N. Benefits of 24-Week Exergame Intervention on Health-Related Quality of Life and Pain in Women with Fibromyalgia: A Single-Blind, Randomized Controlled Trial. Games Health J. 2019. [Google Scholar] [CrossRef]
- Martin-Martinez, J.P.; Villafaina, S.; Collado-Mateo, D.; Perez-Gomez, J.; Gusi, N. Effects of 24-wk exergame intervention on physical function under single- and dual-task conditions in fibromyalgia: A randomized controlled trial. Scand. J. Med. Sci. Spor. 2019. [Google Scholar] [CrossRef]
- Villafaina, S.; Collado-Mateo, D.; Fuentes, J.P.; Rohlfs-Dominguez, P.; Gusi, N. Effects of Exergames on Brain Dynamics in Women with Fibromyalgia: A Randomized Controlled Trial. J. Clin. Med. 2019, 8, 1015. [Google Scholar] [CrossRef]
- Murias, J.M.; Edwards, J.A.; Paterson, D.H. Effects of short-term training and detraining on VO2 kinetics: Faster VO2 kinetics response after one training session. Scand. J. Med. Sci. Sports 2016, 26, 620–629. [Google Scholar] [CrossRef] [PubMed]
- St-Amand, J.; Yoshioka, M.; Nishida, Y.; Tobina, T.; Shono, N.; Tanaka, H. Effects of mild-exercise training cessation in human skeletal muscle. Eur. J. Appl. Physiol. 2012, 112, 853–869. [Google Scholar] [CrossRef] [PubMed]
- Tomas-Carus, P.; Hakkinen, A.; Gusi, N.; Leal, A.; Hakkinen, K.; Ortega-Alonso, A. Aquatic training and detraining on fitness and quality of life in fibromyalgia. Med. Sci. Sports Exerc. 2007, 39, 1044–1050. [Google Scholar] [CrossRef] [PubMed]
- Romero-Zurita, A.; Carbonell-Baeza, A.; Aparicio, V.A.; Ruiz, J.R.; Tercedor, P.; Delgado-Fernandez, M. Effectiveness of a tai-chi training and detraining on functional capacity, symptomatology and psychological outcomes in women with fibromyalgia. Evid. Based Complement. Alternat. Med. 2012, 2012, 614196. [Google Scholar] [CrossRef]
- Sanudo, B.; Carrasco, L.; de Hoyo, M.; McVeigh, J.G. Effects of exercise training and detraining in patients with fibromyalgia syndrome: A 3-yr longitudinal study. Am. J. Phys. Med. Rehabil. 2012, 91, 561–569. [Google Scholar] [CrossRef]
- Lewis, G.N.; Rosie, J.A. Virtual reality games for movement rehabilitation in neurological conditions: How do we meet the needs and expectations of the users? Disabil. Rehabil. 2012, 34, 1880–1886. [Google Scholar] [CrossRef]
- Bidonde, J.; Jean Busch, A.; Bath, B.; Milosavljevic, S. Exercise for adults with fibromyalgia: An umbrella systematic review with synthesis of best evidence. Curr. Rheumatol. Rev. 2014, 10, 45–79. [Google Scholar] [CrossRef]
- Rikli, R.E.; Jones, C.J. Development and validation of a functional fitness test for community-residing older adults. J. Aging Phys. Activ. 1999, 7, 129–161. [Google Scholar] [CrossRef]
- Carbonell-Baeza, A.; Alvarez-Gallardo, I.C.; Segura-Jiménez, V.; Castro-Pinero, J.; Ruiz, J.R.; Delgado-Fernández, M.; Aparicio, V.A. Reliability and feasibility of physical fitness tests in female fibromyalgia patients. Int. J. Sports Med. 2015, 36, 157–162. [Google Scholar] [CrossRef]
- Martin-Martinez, J.P.; Collado-Mateo, D.; Dominguez-Munoz, F.J.; Villafaina, S.; Gusi, N.; Perez-Gomez, J. Reliability of the 30 s Chair Stand Test in Women with Fibromyalgia. Int. J. Environ. Res. Pub. Health 2019, 16, 2344. [Google Scholar] [CrossRef]
- Collado-Mateo, D.; Domínguez-Muñoz, F.J.; Olivares, P.R.; Adsuar, J.C.; Gusi, N. Stair negotiation in women with fibromyalgia: A descriptive correlational study. Medicine 2017, 96, e8364. [Google Scholar] [CrossRef] [PubMed]
- King, S.; Wessel, J.; Bhambhani, Y.; Maikala, R.; Sholter, D.; Maksymowych, W. Validity and reliability of the 6 min walk in persons with fibromyalgia. J. Rheumatol. 1999, 26, 2233–2237. [Google Scholar] [PubMed]
- Bennett, R.M.; Friend, R.; Jones, K.D.; Ward, R.; Han, B.K.; Ross, R.L. The revised fibromyalgia impact questionnaire (FIQR): Validation and psychometric properties. Arthritis Res. Ther. 2009, 11, R120. [Google Scholar] [CrossRef] [PubMed]
- Salgueiro, M.; Garcia-Leiva, J.M.; Ballesteros, J.; Hidalgo, J.; Molina, R.; Calandre, E.P. Validation of a Spanish version of the Revised Fibromyalgia Impact Questionnaire (FIQR). Health Qual. Life Outcomes 2013, 11, 132. [Google Scholar] [CrossRef]
- Craig, C.L.; Marshall, A.L.; Sjostrom, M.; Bauman, A.E.; Booth, M.L.; Ainsworth, B.E.; Pratt, M.; Ekelund, U.; Yngve, A.; Sallis, J.F.; et al. International physical activity questionnaire: 12-country reliability and validity. Med. Sci. Sports Exerc. 2003, 35, 1381–1395. [Google Scholar] [CrossRef]
- Committee, I.R. Guidelines for Data Processing and Analysis of the International Physical Activity Questionnaire (IPAQ)-Short and Long Forms. Available online: http://www.ipaq.ki.se/scoring.pdf (accessed on 25 June 2005).
- Bennett, R. The Fibromyalgia Impact Questionnaire (FIQ): A review of its development, current version, operating characteristics and uses. Clin. Exp. Rheumatol. 2005, 23, S154–S162. [Google Scholar]
- Bennett, R.M.; Bushmakin, A.G.; Cappelleri, J.C.; Zlateva, G.; Sadosky, A.B. Minimal clinically important difference in the fibromyalgia impact questionnaire. J. Rheumatol. 2009, 36, 1304–1311. [Google Scholar] [CrossRef]
- Esteve-Vives, J.; Redondo, J.R.; Salvat, M.I.S.; de Gracia Blanco, M.; de Miquele, C.A. Proposal for a consensus version of the Fibromyalgia Impact Questionnaire (FIQ) for the Spanish population. Reumatol. Clin. 2007, 3, 21–24. [Google Scholar] [CrossRef]
- Benjamini, Y.; Hochberg, Y. Controlling the false discovery rate: A practical and powerful approach to multiple testing. J. R. Stat. Soc. B 1995, 57, 289–300. [Google Scholar] [CrossRef]
- Fritz, C.O.; Morris, P.E.; Richler, J.J. Effect size estimates: Current use, calculations, and interpretation. J. Exp. Psychol. Gen. 2012, 141, 2–18. [Google Scholar] [CrossRef]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences; Routledge: Abingdon, UK, 2013. [Google Scholar]
- Gavilan-Carrera, B.; Segura-Jimenez, V.; Mekary, R.A.; Borges-Cosic, M.; Acosta-Manzano, P.; Estevez-Lopez, F.; Alvarez-Gallardo, I.C.; Geenen, R.; Delgado-Fernandez, M. Substituting Sedentary Time With Physical Activity in Fibromyalgia and the Association with Quality of Life and Impact of the Disease: The al-andalus Project. Arthritis Care Res. 2019, 71, 281–289. [Google Scholar] [CrossRef] [PubMed]
- Borges-Cosic, M.; Aparicio, V.A.; Estevez-Lopez, F.; Soriano-Maldonado, A.; Acosta-Manzano, P.; Gavilan-Carrera, B.; Delgado-Fernandez, M.; Geenen, R.; Segura-Jimenez, V. Sedentary time, physical activity, and sleep quality in fibromyalgia: The al-andalus project. Scand. J. Med. Sci. Sports 2019, 29, 266–274. [Google Scholar] [CrossRef] [PubMed]
- Meekes, W.; Stanmore, E.K. Motivational determinants of exergame participation for older people in assisted living facilities: Mixed-methods study. J. Med. Internet Res. 2017, 19, e238. [Google Scholar] [CrossRef] [PubMed]
- Lange, B.; Flynn, S.M.; Rizzo, A.A. Game-based telerehabilitation. Eur. J. Phys. Rehabil. Med. 2009, 45, 143–151. [Google Scholar]
- Stanmore, E.K.; Mavroeidi, A.; de Jong, L.D.; Skelton, D.A.; Sutton, C.J.; Benedetto, V.; Munford, L.A.; Meekes, W.; Bell, V.; Todd, C. The effectiveness and cost-effectiveness of strength and balance Exergames to reduce falls risk for people aged 55 years and older in UK assisted living facilities: A multi-centre, cluster randomised controlled trial. BMC Med. 2019, 17, 49. [Google Scholar] [CrossRef]
- Gowans, S.E.; deHueck, A. Effectiveness of exercise in management of fibromyalgia. Curr. Opin. Rheumatol. 2004, 16, 138–142. [Google Scholar] [CrossRef]
- Busch, A.J.; Overend, T.J.; Schachter, C.L. Fibromyalgia treatment: The role of exercise and physical activity. Int. J. Clin. Rheumtol. 2009, 4, 343–380. [Google Scholar] [CrossRef]
- Burke, S.M.; Carron, A.V.; Eys, M.A.; Ntoumanis, N.; Estabrooks, P.A. Group versus individual approach? A meta-analysis of the effectiveness of interventions to promote physical activity. Sport Exerc. Psychol. Rev. 2006, 2, 19–35. [Google Scholar]
- Segura-Jimenez, V.; Munguia-Izquierdo, D.; Camiletti-Moiron, D.; Alvarez-Gallardo, I.C.; Ortega, F.B.; Ruiz, J.R.; Delgado-Fernandez, M. Comparison of the International Physical Activity Questionnaire (IPAQ) with a multi-sensor armband accelerometer in women with fibromyalgia: The al-Andalus project. Clin. Exp. Rheumatol. 2013, 31, S94–S101. [Google Scholar]
| Exergame Group Mean (SD) | Control Group Mean (SD) | p-Value | Effect Size | |
|---|---|---|---|---|
| Sample size (N) | 22 | 15 | ||
| Age (years) | 54.27 (9.29) | 53.44 (9.47) | 0.788 | 0.002 |
| FIQ-r | 52.62 (17.12) | 54.97 (20.34) | 0.702 | 0.004 |
| METs (per week) | 2667.3 (3704) | 4422.71(4291) | 0.174 | −0.046 |
| Sitting time (min per day) | 264.09 (171.94) | 276 (166.34) | 0.836 | <0.001 |
| BMI (kg/m2) | 27.11 (2.90) | 28.19 (3.88) | 0.332 | 0.026 |
| Fat mass (kg) | 26.99 (5.95) | 26.9 (7.60) | 0.969 | <0.001 |
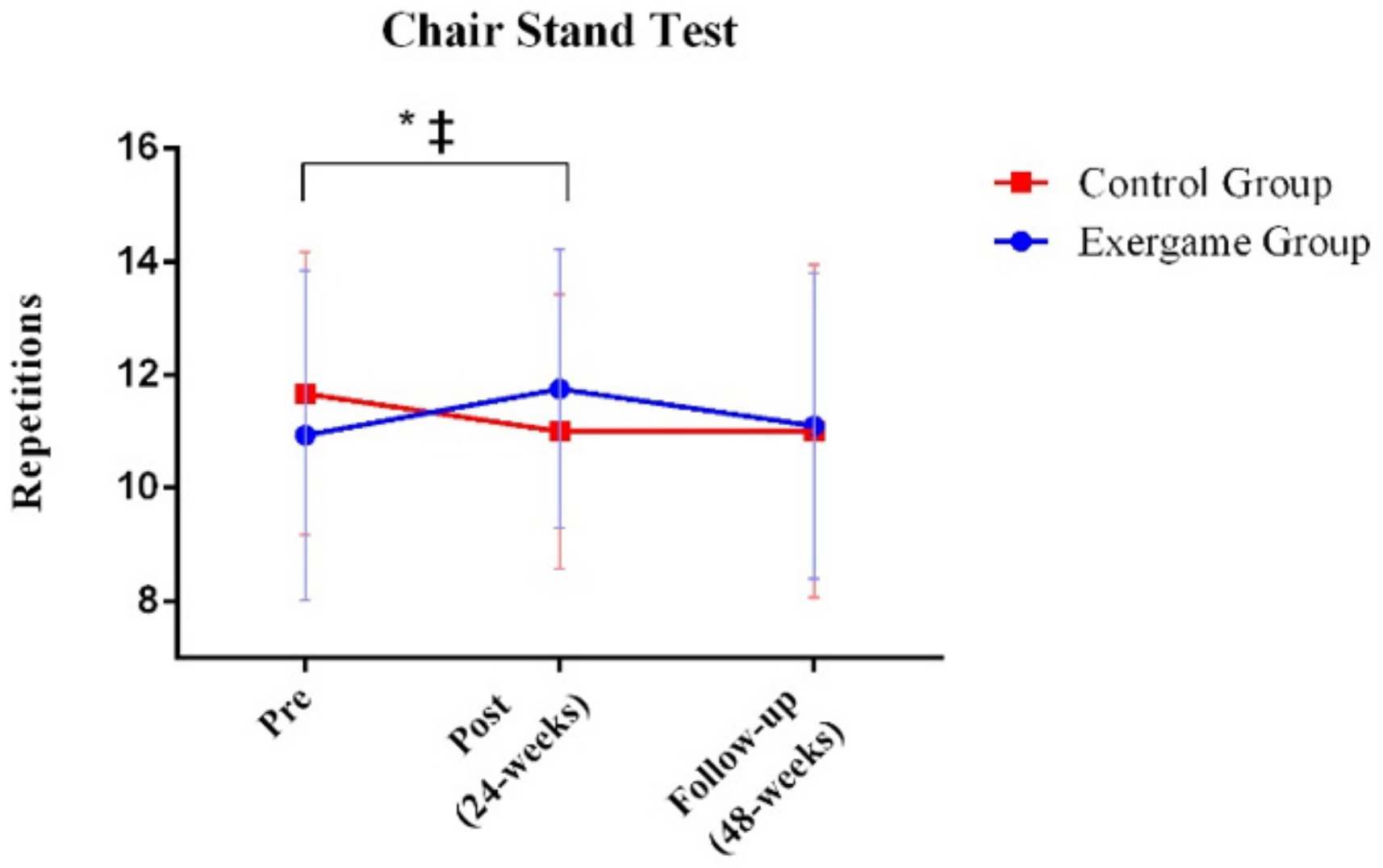
| Chair–stand test (Rep) | 10.93 (2.91) | 11.31 (2.80) | 0.688 | 0.005 |
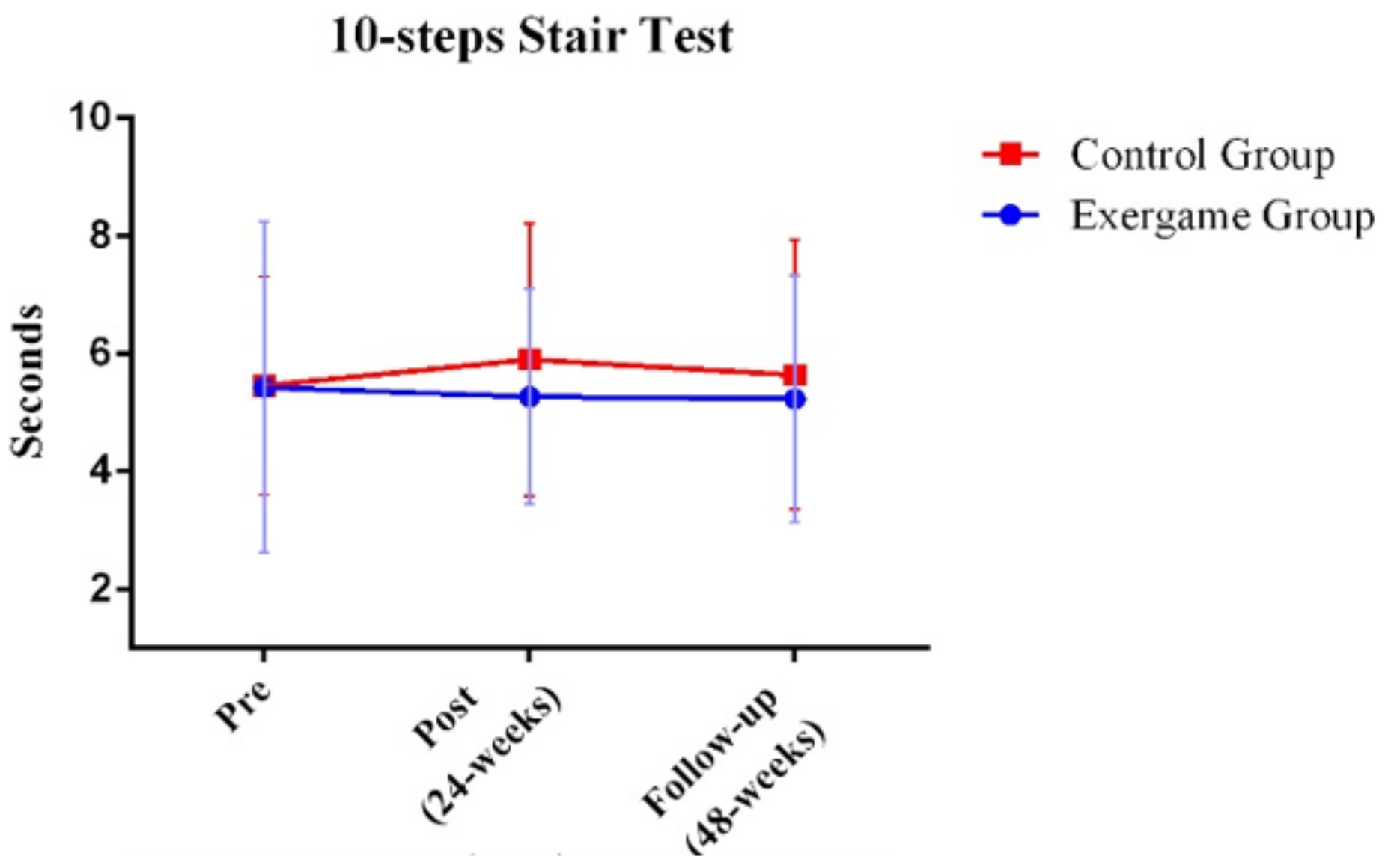
| Stairs (s) | 5.43 (2.81) | 5.46 (1.86) | 0.980 | <0.001 |
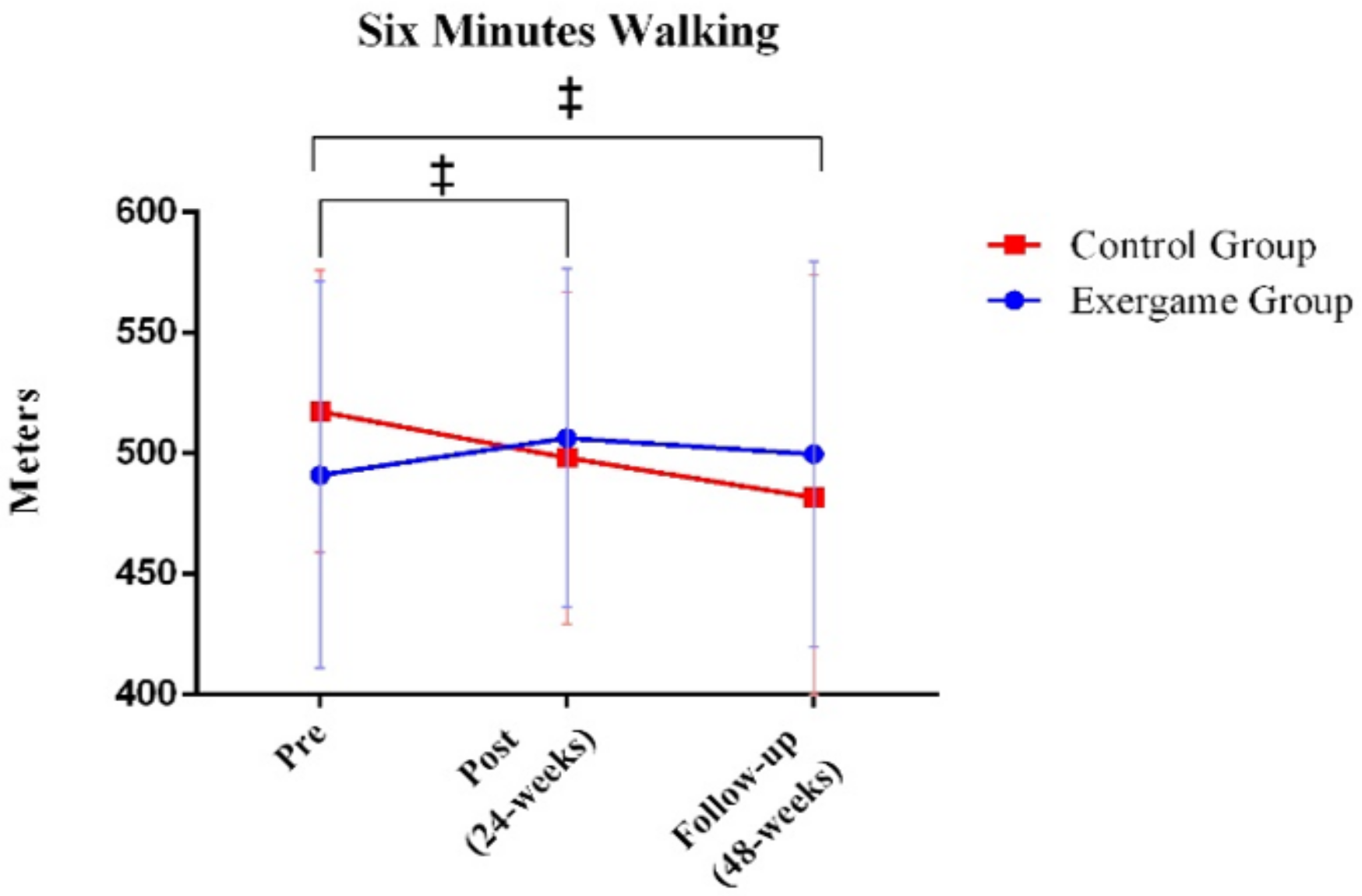
| Six-minute walk test (M) | 491.15 (80.21) | 517.45 (58.42) | 0.285 | 0.032 |
| Test | Groups | Comparisons Between Groups | |||||
|---|---|---|---|---|---|---|---|
| Preintervention Mean (SD) | Post-Treatment (24 Weeks) Mean (SD) | Follow-Up (48 Weeks) Mean (SD) | F | p-Value * | Effect Size | ||
| FIQ-r | Exergames (N = 22) | 53.17 (16.93) | 51.60 (18.25) | 51.10 (18.99) | 0.295 | 0.746 | 0.016 |
| Control (N = 15) | 54.15 (19.98) | 55.35 (20.11) | 52.07 (18.37) | ||||
| METs (per week) | Exergames (N = 22) | 2667.35 (3704.18) | 2990.17 (3090.58) | 3194.14 (3356.59) | 0.624 | 0.541 | 0.034 |
| Control (N = 15) | 4422.71 (4291.30) | 3406.94 (6315.86) | 3063.29 (2942.56) | ||||
| Sitting time (min/day) | Exergames (N = 22) | 262.38 (178.04) | 277.14 (162.36) | 364.28 (205.07) | 0.369 | 0.694 | 0.023 |
| Control (N = 15) | 270 (171.92) | 265.71 (129.89) | 409.28 (205.71) | ||||
| Chair–stand test (Rep) | Exergames (N = 22) | 10.93 (2.91) | 11.75 (2.46) | 11.10 (2.70) | 4.593 | 0.017 | 0.213 |
| Control (N = 15) | 11.67 (2.50) | 11.00 (2.42) | 11.00 (2.94) | ||||
| Stairs (s) | Exergames (N = 22) | 5.43 (2.81) | 5.27 (1.83) | 5.23 (2.10) | 0.412 | 0.666 | 0.024 |
| Control (N = 14) | 5.46 (1.86) | 5.90 (2.32) | 5.64 (2.29) | ||||
| Six-minute walk test (m) | Exergames (N = 22) | 491.15 (80.21) | 506.47 (70.15) | 499.74 (80.05) | 5.191 | 0.011 | 0.234 |
| Control (N = 15) | 517.45 (58.42) | 498.21 (68.80) | 481.70 (92.30) | ||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Villafaina, S.; Borrega-Mouquinho, Y.; Fuentes-García, J.P.; Collado-Mateo, D.; Gusi, N. Effect of Exergame Training and Detraining on Lower-Body Strength, Agility, and Cardiorespiratory Fitness in Women with Fibromyalgia: Single-Blinded Randomized Controlled Trial. Int. J. Environ. Res. Public Health 2020, 17, 161. https://doi.org/10.3390/ijerph17010161
Villafaina S, Borrega-Mouquinho Y, Fuentes-García JP, Collado-Mateo D, Gusi N. Effect of Exergame Training and Detraining on Lower-Body Strength, Agility, and Cardiorespiratory Fitness in Women with Fibromyalgia: Single-Blinded Randomized Controlled Trial. International Journal of Environmental Research and Public Health. 2020; 17(1):161. https://doi.org/10.3390/ijerph17010161
Chicago/Turabian StyleVillafaina, Santos, Yolanda Borrega-Mouquinho, Juan Pedro Fuentes-García, Daniel Collado-Mateo, and Narcis Gusi. 2020. "Effect of Exergame Training and Detraining on Lower-Body Strength, Agility, and Cardiorespiratory Fitness in Women with Fibromyalgia: Single-Blinded Randomized Controlled Trial" International Journal of Environmental Research and Public Health 17, no. 1: 161. https://doi.org/10.3390/ijerph17010161
APA StyleVillafaina, S., Borrega-Mouquinho, Y., Fuentes-García, J. P., Collado-Mateo, D., & Gusi, N. (2020). Effect of Exergame Training and Detraining on Lower-Body Strength, Agility, and Cardiorespiratory Fitness in Women with Fibromyalgia: Single-Blinded Randomized Controlled Trial. International Journal of Environmental Research and Public Health, 17(1), 161. https://doi.org/10.3390/ijerph17010161









