Sarcopenia Is Associated with Cognitive Impairment Mainly Due to Slow Gait Speed: Results from the Korean Frailty and Aging Cohort Study (KFACS)
Abstract
1. Introduction
2. Materials and Methods
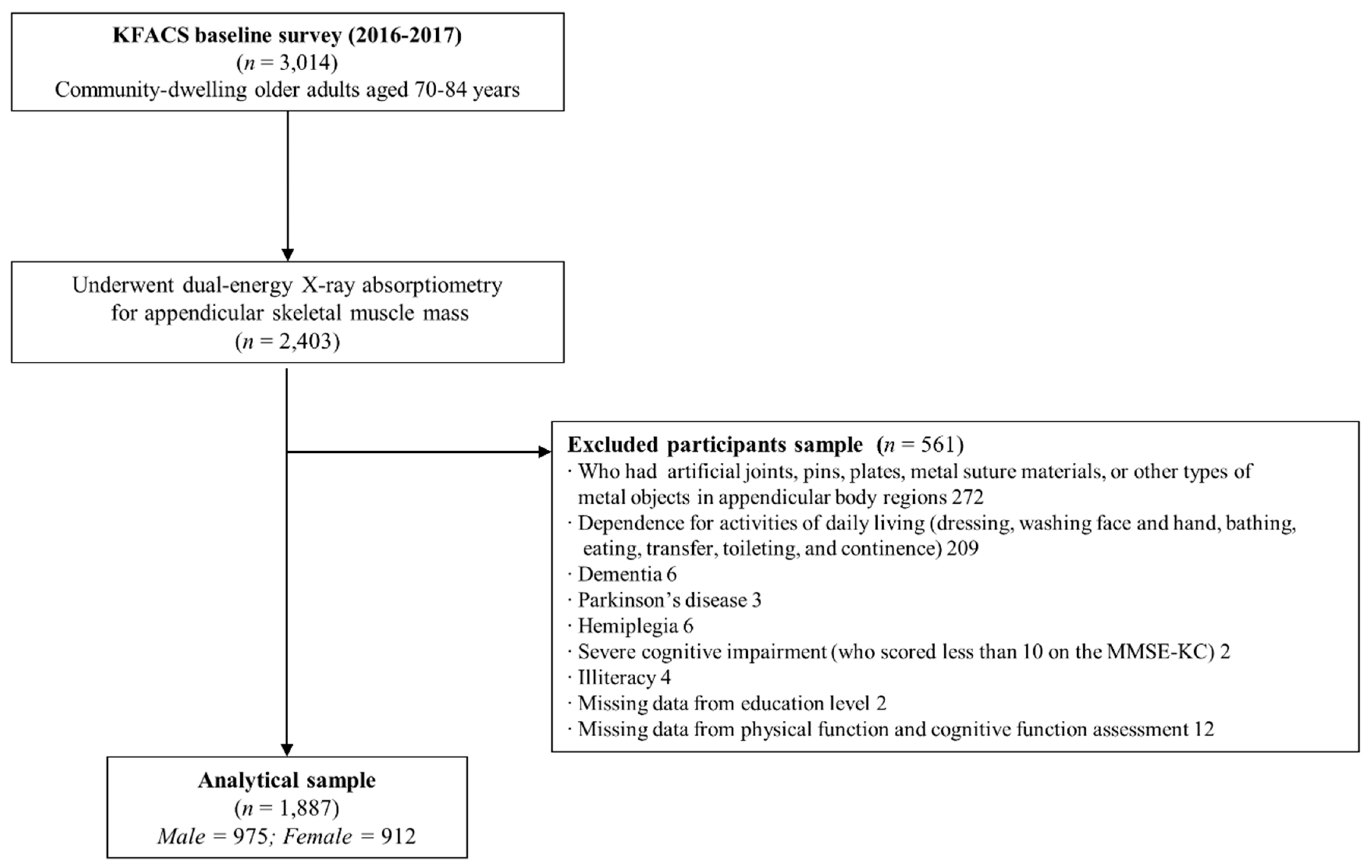
2.1. Study Population
2.2. Cognitive Function Assessment
2.3. Diagnosis of Sarcopenia
2.4. Other Measurements
2.5. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Plassman, B.L.; Langa, K.M.; McCammon, R.J.; Fisher, G.G.; Potter, G.G.; Burke, J.R.; Steffens, D.C.; Foster, N.L.; Giordani, B.; Unverzagt, F.W.; et al. Incidence of dementia and cognitive impairment, not dementia in the united states. Ann. Neurol. 2011, 70, 418–426. [Google Scholar] [CrossRef] [PubMed]
- Plassman, B.L.; Langa, K.M.; Fisher, G.G.; Heeringa, S.G.; Weir, D.R.; Ofstedal, M.B.; Burke, J.R.; Hurd, M.D.; Potter, G.G.; Rodgers, W.L.; et al. Prevalence of cognitive impairment without dementia in the united states. Ann. Intern. Med. 2008, 148, 427–434. [Google Scholar] [CrossRef]
- Lyketsos, C.G.; Lopez, O.; Jones, B.; Fitzpatrick, A.L.; Breitner, J.; DeKosky, S. Prevalence of neuropsychiatric symptoms in dementia and mild cognitive impairment: Results from the cardiovascular health study. JAMA 2002, 288, 1475–1483. [Google Scholar] [CrossRef] [PubMed]
- Tabert, M.H.; Albert, S.M.; Borukhova-Milov, L.; Camacho, Y.; Pelton, G.; Liu, X.; Stern, Y.; Devanand, D.P. Functional deficits in patients with mild cognitive impairment: Prediction of ad. Neurology 2002, 58, 758–764. [Google Scholar] [CrossRef]
- Jonsson, L.; Lindgren, P.; Wimo, A.; Jonsson, B.; Winblad, B. Costs of mini mental state examination-related cognitive impairment. Pharmacoeconomics 1999, 16, 409–416. [Google Scholar] [CrossRef] [PubMed]
- Gill, T.M.; Williams, C.S.; Richardson, E.D.; Tinetti, M.E. Impairments in physical performance and cognitive status as predisposing factors for functional dependence among nondisabled older persons. J. Gerontol. A Biol. Sci. Med. Sci. 1996, 51, M283–M288. [Google Scholar] [CrossRef] [PubMed]
- Baumgart, M.; Snyder, H.M.; Carrillo, M.C.; Fazio, S.; Kim, H.; Johns, H. Summary of the evidence on modifiable risk factors for cognitive decline and dementia: A population-based perspective. Alzheimers Dement. 2015, 11, 718–726. [Google Scholar] [CrossRef]
- Legdeur, N.; Heymans, M.W.; Comijs, H.C.; Huisman, M.; Maier, A.B.; Visser, P.J. Age dependency of risk factors for cognitive decline. BMC Geriatr. 2018, 18, 187. [Google Scholar] [CrossRef]
- Cruz-Jentoft, A.J.; Baeyens, J.P.; Bauer, J.M.; Boirie, Y.; Cederholm, T.; Landi, F.; Martin, F.C.; Michel, J.P.; Rolland, Y.; Schneider, S.M.; et al. Sarcopenia: European consensus on definition and diagnosis: Report of the european working group on sarcopenia in older people. Age Ageing 2010, 39, 412–423. [Google Scholar] [CrossRef]
- Fried, L.P.; Tangen, C.M.; Walston, J.; Newman, A.B.; Hirsch, C.; Gottdiener, J.; Seeman, T.; Tracy, R.; Kop, W.J.; Burke, G.; et al. Frailty in older adults: Evidence for a phenotype. J. Gerontol. A Biol. Sci. Med. Sci. 2001, 56, M146–M156. [Google Scholar] [CrossRef]
- Cao, L.; Morley, J.E. Sarcopenia is recognized as an independent condition by an international classification of disease, tenth revision, clinical modification (icd-10-cm) code. J. Am. Med. Dir. Assoc. 2016, 17, 675–677. [Google Scholar] [CrossRef]
- Cruz-Jentoft, A.J.; Bahat, G.; Bauer, J.; Boirie, Y.; Bruyere, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Sayer, A.A.; et al. Sarcopenia: Revised european consensus on definition and diagnosis. Age Ageing 2018, 48, 16–31. [Google Scholar] [CrossRef]
- Chen, L.K.; Liu, L.K.; Woo, J.; Assantachai, P.; Auyeung, T.W.; Bahyah, K.S.; Chou, M.Y.; Chen, L.Y.; Hsu, P.S.; Krairit, O.; et al. Sarcopenia in asia: Consensus report of the asian working group for sarcopenia. J. Am. Med. Dir. Assoc. 2014, 15, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Studenski, S.A.; Peters, K.W.; Alley, D.E.; Cawthon, P.M.; McLean, R.R.; Harris, T.B.; Ferrucci, L.; Guralnik, J.M.; Fragala, M.S.; Kenny, A.M.; et al. The fnih sarcopenia project: Rationale, study description, conference recommendations, and final estimates. J. Gerontol. A Biol. Sci. Med. Sci. 2014, 69, 547–558. [Google Scholar] [CrossRef]
- Mayhew, A.J.; Amog, K.; Phillips, S.; Parise, G.; McNicholas, P.D.; de Souza, R.J.; Thabane, L.; Raina, P. The prevalence of sarcopenia in community-dwelling older adults, an exploration of differences between studies and within definitions: A systematic review and meta-analyses. Age Ageing 2018, 48, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Larsson, L.; Degens, H.; Li, M.; Salviati, L.; Lee, Y.I.; Thompson, W.; Kirkland, J.L.; Sandri, M. Sarcopenia: Aging-related loss of muscle mass and function. Physiol. Rev. 2019, 99, 427–511. [Google Scholar] [CrossRef] [PubMed]
- Hsu, Y.H.; Liang, C.K.; Chou, M.Y.; Liao, M.C.; Lin, Y.T.; Chen, L.K.; Lo, Y.K. Association of cognitive impairment, depressive symptoms and sarcopenia among healthy older men in the veterans retirement community in southern taiwan: A cross-sectional study. Geriatr. Gerontol. Int. 2014, 14 (Suppl. 1), 102–108. [Google Scholar] [CrossRef]
- Tolea, M.I.; Galvin, J.E. Sarcopenia and impairment in cognitive and physical performance. Clin. Interv. Aging 2015, 10, 663–671. [Google Scholar] [CrossRef]
- Canon, M.E.; Crimmins, E.M. Sex differences in the association between muscle quality, inflammatory markers, and cognitive decline. J. Nutr. Health Aging 2011, 15, 695–698. [Google Scholar] [CrossRef]
- Chang, K.V.; Hsu, T.H.; Wu, W.T.; Huang, K.C.; Han, D.S. Association between sarcopenia and cognitive impairment: A systematic review and meta-analysis. J. Am. Med. Dir. Assoc. 2016, 17, 1164.e7–1164.e15. [Google Scholar] [CrossRef] [PubMed]
- Won, C.W.; Lee, Y.; Choi, J.; Kim, K.W.; Park, Y.; Park, H.; Oh, I.-H.; Ga, H.; Kim, Y.S.; Jang, H.C. Starting construction of frailty cohort for elderly and intervention study. Ann. Geriatr. Med. Res. 2016, 20, 114–117. [Google Scholar] [CrossRef]
- Lee, J.H.; Lee, K.U.; Lee, D.Y.; Kim, K.W.; Jhoo, J.H.; Kim, J.H.; Lee, K.H.; Kim, S.Y.; Han, S.H.; Woo, J.I. Development of the korean version of the consortium to establish a registry for alzheimer’s disease assessment packet (cerad-k): Clinical and neuropsychological assessment batteries. J. Gerontol. B Psychol. Sci. Soc. Sci. 2002, 57, P47–P53. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.Y.; Lee, K.U.; Lee, J.H.; Kim, K.W.; Jhoo, J.H.; Kim, S.Y.; Yoon, J.C.; Woo, S.I.; Ha, J.; Woo, J.I. A normative study of the cerad neuropsychological assessment battery in the korean elderly. J. Int. Neuropsychol. Soc. 2004, 10, 72–81. [Google Scholar] [CrossRef]
- Kim, T.H.; Huh, Y.; Choe, J.Y.; Jeong, J.W.; Park, J.H.; Lee, S.B.; Lee, J.J.; Jhoo, J.H.; Lee, D.Y.; Woo, J.I.; et al. Korean version of frontal assessment battery: Psychometric properties and normative data. Dement. Geriatr. Cogn. Disord. 2010, 29, 363–370. [Google Scholar] [CrossRef]
- Morris, J.C.; Heyman, A.; Mohs, R.C.; Hughes, J.P.; van Belle, G.; Fillenbaum, G.; Mellits, E.D.; Clark, C. The consortium to establish a registry for alzheimer’s disease (cerad). Part I. Clinical and neuropsychological assessment of alzheimer’s disease. Neurology 1989, 39, 1159–1165. [Google Scholar]
- Won, C.W.; Lee, Y.; Kim, S.; Yoo, J.; Kim, M.; Ng, T.P.; Kim, H.; Son, S.J. Modified criteria for diagnosing “cognitive frailty”. Psychiatry Investig. 2018, 15, 839–842. [Google Scholar] [CrossRef]
- Kim, M.; Jeong, M.J.; Yoo, J.; Song, D.Y.; Won, C.W. Calf circumference as a screening tool for cognitive frailty in community-dwelling older adults: The korean frailty and aging cohort study (kfacs). J. Clin. Med. 2018, 7, 332. [Google Scholar] [CrossRef] [PubMed]
- Petersen, R.C.; Smith, G.E.; Waring, S.C.; Ivnik, R.J.; Tangalos, E.G.; Kokmen, E. Mild cognitive impairment: Clinical characterization and outcome. Arch. Neurol. 1999, 56, 303–308. [Google Scholar] [CrossRef]
- Jessen, F.; Amariglio, R.E.; van Boxtel, M.; Breteler, M.; Ceccaldi, M.; Chetelat, G.; Dubois, B.; Dufouil, C.; Ellis, K.A.; van der Flier, W.M.; et al. A conceptual framework for research on subjective cognitive decline in preclinical alzheimer’s disease. Alzheimers Dement. 2014, 10, 844–852. [Google Scholar] [CrossRef]
- Lee, S.C.; Kim, W.H.; Chang, S.M.; Kim, B.S.; Lee, D.W.; Bae, J.N.; Cho, M.J. The use of the korean version of short form geriatric depression scale (sgds-k)in the community dwelling elderly in korea. J. Korean Geriatr. Psychiatry 2013, 17, 37–43. [Google Scholar]
- Rubenstein, L.Z.; Harker, J.O.; Salva, A.; Guigoz, Y.; Vellas, B. Screening for undernutrition in geriatric practice: Developing the short-form mini-nutritional assessment (mna-sf). J. Gerontol. A Biol. Sci. Med. Sci. 2001, 56, M366–M372. [Google Scholar] [CrossRef]
- Jeon, S.Y.; Won, C.W.; Choi, H.R.; Kim, B.S.; Kim, S.Y.; Hur, J.H. Physical frailty predicts cognitive decline in elderly people: Prospective findings from the living profiles of older people survey in Korea. Korean J. Fam. Pract. 2015, 5, 702–707. [Google Scholar]
- Guralnik, J.M.; Ferrucci, L.; Pieper, C.F.; Leveille, S.G.; Markides, K.S.; Ostir, G.V.; Studenski, S.; Berkman, L.F.; Wallace, R.B. Lower extremity function and subsequent disability: Consistency across studies, predictive models, and value of gait speed alone compared with the short physical performance battery. J. Gerontol. A Biol. Sci. Med. Sci. 2000, 55, M221–M231. [Google Scholar] [CrossRef] [PubMed]
- Guralnik, J.M.; Simonsick, E.M.; Ferrucci, L.; Glynn, R.J.; Berkman, L.F.; Blazer, D.G.; Scherr, P.A.; Wallace, R.B. A short physical performance battery assessing lower extremity function: Association with self-reported disability and prediction of mortality and nursing home admission. J. Gerontol. 1994, 49, M85–M94. [Google Scholar] [CrossRef] [PubMed]
- Podsiadlo, D.; Richardson, S. The timed “up & go”: A test of basic functional mobility for frail elderly persons. J. Am. Geriatr. Soc. 1991, 39, 142–148. [Google Scholar] [PubMed]
- Kim, J.K.; Choi, S.R.; Choi, M.J.; Kim, S.G.; Lee, Y.K.; Noh, J.W.; Kim, H.J.; Song, Y.R. Prevalence of and factors associated with sarcopenia in elderly patients with end-stage renal disease. Clin. Nutr. 2014, 33, 64–68. [Google Scholar] [CrossRef]
- Huang, C.Y.; Hwang, A.C.; Liu, L.K.; Lee, W.J.; Chen, L.Y.; Peng, L.N.; Lin, M.H.; Chen, L.K. Association of dynapenia, sarcopenia, and cognitive impairment among community-dwelling older taiwanese. Rejuvenation Res. 2016, 19, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, A.J. A meta-analysis of the accuracy of the mini-mental state examination in the detection of dementia and mild cognitive impairment. J. Psychiatr. Res. 2009, 43, 411–431. [Google Scholar] [CrossRef]
- Xie, H.; Mayo, N.; Koski, L. Predictors of future cognitive decline in persons with mild cognitive impairment. Dement. Geriatr. Cogn. Disord. 2011, 32, 308–317. [Google Scholar] [CrossRef]
- O’Bryant, S.E.; Humphreys, J.D.; Smith, G.E.; Ivnik, R.J.; Graff-Radford, N.R.; Petersen, R.C.; Lucas, J.A. Detecting dementia with the mini-mental state examination in highly educated individuals. Arch. Neurol. 2008, 65, 963–967. [Google Scholar] [CrossRef]
- Spering, C.C.; Hobson, V.; Lucas, J.A.; Menon, C.V.; Hall, J.R.; O’Bryant, S.E. Diagnostic accuracy of the mmse in detecting probable and possible alzheimer’s disease in ethnically diverse highly educated individuals: An analysis of the nacc database. J. Gerontol. A Biol. Sci. Med. Sci. 2012, 67, 890–896. [Google Scholar] [CrossRef]
- Xu, X.; Chong, E.; Hilal, S.; Ikram, M.K.; Venketasubramanian, N.; Chen, C. Beyond screening: Can the mini-mental state examination be used as an exclusion tool in a memory clinic? Diagnostics 2015, 5, 475–486. [Google Scholar] [CrossRef]
- Wu, Y.H.; Liu, L.K.; Chen, W.T.; Lee, W.J.; Peng, L.N.; Wang, P.N.; Chen, L.K. Cognitive function in individuals with physical frailty but without dementia or cognitive complaints: Results from the i-lan longitudinal aging study. J. Am. Med. Dir. Assoc. 2015, 16, 899.e9–899.e16. [Google Scholar] [CrossRef]
- Mielke, M.M.; Roberts, R.O.; Savica, R.; Cha, R.; Drubach, D.I.; Christianson, T.; Pankratz, V.S.; Geda, Y.E.; Machulda, M.M.; Ivnik, R.J.; et al. Assessing the temporal relationship between cognition and gait: Slow gait predicts cognitive decline in the mayo clinic study of aging. J. Gerontol. A Biol. Sci. Med. Sci. 2013, 68, 929–937. [Google Scholar] [CrossRef]
- Callisaya, M.L.; Blizzard, C.L.; Wood, A.G.; Thrift, A.G.; Wardill, T.; Srikanth, V.K. Longitudinal relationships between cognitive decline and gait slowing: The tasmanian study of cognition and gait. J. Gerontol. A Biol. Sci. Med. Sci. 2015, 70, 1226–1232. [Google Scholar] [CrossRef]
- Lipnicki, D.M.; Crawford, J.; Kochan, N.A.; Trollor, J.N.; Draper, B.; Reppermund, S.; Maston, K.; Mather, K.A.; Brodaty, H.; Sachdev, P.S.; et al. Risk factors for mild cognitive impairment, dementia and mortality: The sydney memory and ageing study. J. Am. Med. Dir. Assoc. 2017, 18, 388–395. [Google Scholar] [CrossRef]
- Callisaya, M.L.; Beare, R.; Phan, T.G.; Blizzard, L.; Thrift, A.G.; Chen, J.; Srikanth, V.K. Brain structural change and gait decline: A longitudinal population-based study. J. Am. Geriatr. Soc. 2013, 61, 1074–1079. [Google Scholar] [CrossRef]
- Killane, I.; Donoghue, O.A.; Savva, G.M.; Cronin, H.; Kenny, R.A.; Reilly, R.B. Relative association of processing speed, short-term memory and sustained attention with task on gait speed: A study of community-dwelling people 50 years and older. J. Gerontol. A Biol. Sci. Med. Sci. 2014, 69, 1407–1414. [Google Scholar] [CrossRef]
- Taylor, M.E.; Lasschuit, D.A.; Lord, S.R.; Delbaere, K.; Kurrle, S.E.; Mikolaizak, A.S.; Kvelde, T.; Close, J.C.T. Slow gait speed is associated with executive function decline in older people with mild to moderate dementia: A one year longitudinal study. Arch. Gerontol. Geriatr. 2017, 73, 148–153. [Google Scholar] [CrossRef]
- Watson, N.L.; Rosano, C.; Boudreau, R.M.; Simonsick, E.M.; Ferrucci, L.; Sutton-Tyrrell, K.; Hardy, S.E.; Atkinson, H.H.; Yaffe, K.; Satterfield, S.; et al. Executive function, memory, and gait speed decline in well-functioning older adults. J. Gerontol. A Biol. Sci. Med. Sci. 2010, 65, 1093–1100. [Google Scholar] [CrossRef]
- Doi, T.; Shimada, H.; Makizako, H.; Tsutsumimoto, K.; Uemura, K.; Anan, Y.; Suzuki, T. Cognitive function and gait speed under normal and dual-task walking among older adults with mild cognitive impairment. BMC Neurol. 2014, 14, 67. [Google Scholar] [CrossRef]
- Rosano, C.; Studenski, S.A.; Aizenstein, H.J.; Boudreau, R.M.; Longstreth, W.T., Jr.; Newman, A.B. Slower gait, slower information processing and smaller prefrontal area in older adults. Age Ageing 2012, 41, 58–64. [Google Scholar] [CrossRef]
- Boripuntakul, S.; Lord, S.R.; Brodie, M.A.; Smith, S.T.; Methapatara, P.; Wongpakaran, N.; Sungkarat, S. Spatial variability during gait initiation while dual tasking is increased in individuals with mild cognitive impairment. J. Nutr. Health Aging 2014, 18, 307–312. [Google Scholar] [CrossRef]
- Horak, F.B. Postural orientation and equilibrium: What do we need to know about neural control of balance to prevent falls? Age Ageing 2006, 35 (Suppl. 2), ii7–ii11. [Google Scholar] [CrossRef]
- Albinet, C.T.; Boucard, G.; Bouquet, C.A.; Audiffren, M. Processing speed and executive functions in cognitive aging: How to disentangle their mutual relationship? Brain Cogn. 2012, 79, 1–11. [Google Scholar] [CrossRef]
- Caetano, M.J.D.; Menant, J.C.; Schoene, D.; Pelicioni, P.H.S.; Sturnieks, D.L.; Lord, S.R. Sensorimotor and cognitive predictors of impaired gait adaptability in older people. J. Gerontol. A Biol. Sci. Med. Sci. 2017, 72, 1257–1263. [Google Scholar] [CrossRef] [PubMed]
- Fritz, N.E.; McCarthy, C.J.; Adamo, D.E. Handgrip strength as a means of monitoring progression of cognitive decline—A scoping review. Ageing Res. Rev. 2017, 35, 112–123. [Google Scholar] [CrossRef]
- Pentikäinen, H.; Savonen, K.; Komulainen, P.; Kiviniemi, V.; Paajanen, T.; Kivipelto, M.; Soininen, H.; Rauramaa, R. Muscle strength and cognition in ageing men and women: The dr’s extra study. Eur. Geriatr. Med. 2017, 8, 275–277. [Google Scholar] [CrossRef]
- Wang, L.; Larson, E.B.; Bowen, J.D.; van Belle, G. Performance-based physical function and future dementia in older people. Arch. Intern. Med. 2006, 166, 1115–1120. [Google Scholar] [CrossRef]
- Won, H.; Abdul Manaf, Z.; Mat Ludin, A.F.; Shahar, S. Wide range of body composition measures are associated with cognitive function in community-dwelling older adults. Geriatr. Gerontol. Int. 2017, 17, 554–560. [Google Scholar] [CrossRef]
- Abellan van Kan, G.; Cesari, M.; Gillette-Guyonnet, S.; Dupuy, C.; Nourhashemi, F.; Schott, A.M.; Beauchet, O.; Annweiler, C.; Vellas, B.; Rolland, Y. Sarcopenia and cognitive impairment in elderly women: Results from the epidos cohort. Age Ageing 2013, 42, 196–202. [Google Scholar] [CrossRef] [PubMed]
- Matousek, R.H.; Sherwin, B.B. Sex steroid hormones and cognitive functioning in healthy, older men. Horm. Behav. 2010, 57, 352–359. [Google Scholar] [CrossRef][Green Version]
- Gibbs, R.B. Estrogen therapy and cognition: A review of the cholinergic hypothesis. Endocr. Rev. 2010, 31, 224–253. [Google Scholar] [CrossRef] [PubMed]
- Smith, E.; Bailey, P.E.; Crawford, J.; Samaras, K.; Baune, B.T.; Campbell, L.; Kochan, N.; Menant, J.; Sturnieks, D.L.; Brodaty, H.; et al. Adiposity estimated using dual energy X-ray absorptiometry and body mass index and its association with cognition in elderly adults. J. Am. Geriatr. Soc. 2014, 62, 2311–2318. [Google Scholar] [CrossRef] [PubMed]
| Variable | Overall (n = 1887) | Without Sarcopenia (n = 1724) | With Sarcopenia (n = 163) |
|---|---|---|---|
| Age (years) | 75.8 ± 3.9 | 75.5 ± 3.8 | 78.5 ± 3.8 |
| Female sex | 912 (48.3) | 843 (48.9) | 69 (42.3) |
| Education (≥7 years) | 1221 (64.7) | 1119 (64.9) | 102 (62.6) |
| Live alone | 382 (20.2) | 343 (19.9) | 39 (23.9) |
| BMI (kg/m2) | 24.2 ± 2.8 | 24.4 ± 2.8 | 22.5 ± 2.9 |
| Appendicular lean mass index (kg/m2) | 6.46 ± 1.00 | 6.53 ± 1.00 | 5.6 ± 0.76 |
| Appendicular lean mass to BMI ratio | 0.685 ± 0.157 | 0.690 ± 0.157 | 0.632 ± 1.148 |
| Current smoker | 119 (6.3) | 104 (6.0) | 15 (9.2) |
| Alcohol (≥2–3 time/week) | 360 (19.1) | 326 (18.9) | 34 (20.9) |
| Low physical activity | 174 (9.3) | 143 (8.4) | 31 (19.3) |
| Number of comorbidities | 1.6 ± 1.2 | 1.6 ± 1.2 | 1.9 ± 1.3 |
| Fair/poor self-perceived health | 453 (24.0) | 381 (22.1) | 72 (44.2) |
| Depressive symptoms (GDS score ≥6) | 370 (19.6) | 312 (18.1) | 58 (35.6) |
| MNA Screening score (≤11) | 144 (7.6) | 116 (6.7) | 28 (17.2) |
| Physical function | |||
| Handgrip strength (kg) | 27.1 ± 7.4 | 27.7 ± 7.3 | 21.0 ± 4.9 |
| Usual gait speed (m/s) | 1.14 ± 0.25 | 1.16 ± 0.25 | 0.96 ± 0.25 |
| Timed get up and go test (s) | 10.1 ± 2.4 | 10.0 ± 2.3 | 11.8 ± 2.6 |
| Five Times Sit-to-Stand (s) | 11.0 ± 3.5 | 10.8 ± 3.3 | 13.0 ± 4.6 |
| Short Physical Performance Battery (score) | 11.0 ± 1.4 | 11.1 ± 1.3 | 10.0 ± 1.8 |
| AWGS-defined sarcopenia | |||
| Low muscle mass | 709 (37.6) | 546 (31.7) | 163 (100.0) |
| Weakness | 281 (14.9) | 144 (8.4) | 137 (84.0) |
| Slow gait speed | 116 (6.1) | 68 (3.9) | 48 (29.4) |
| Variable | Male | Female | ||||
|---|---|---|---|---|---|---|
| Without Sarcopenia (n = 881) | With Sarcopenia (n = 94) | p-Value | Without Sarcopenia (n = 843) | With Sarcopenia (n = 69) | p-Value | |
| Global cognitive function | ||||||
| Mini-Mental State Examination score | 26.5 ± 2.5 | 25.7 ± 3.1 | 0.020 | 25.6 ± 3.2 | 24.6 ± 3.4 | 0.021 |
| Mini-Mental State Examination score < 24 | 148 (16.8) | 27 (28.7) | 0.007 | 247 (29.3) | 26 (37.7) | 0.171 |
| Cognitive dysfunction a | 34 (3.9) | 6 (6.4) | 0.267 | 27 (3.2) | 2 (2.9) | 0.890 |
| Neuropsychological tests | ||||||
| Word list learning score | 16.9 ± 3.9 | 14.7 ± 4.3 | 0.000 | 17.7 ± 4.2 | 16.7 ± 4.0 | 0.078 |
| Word list recall score | 5.7 ± 1.9 | 4.5 ± 2.4 | 0.000 | 5.7 ± 2.1 | 5.5 ± 2.0 | 0.298 |
| Word list recognition score | 8.7 ± 1.7 | 8.0 ± 2.4 | 0.006 | 8.7 ± 1.8 | 8.3 ± 1.8 | 0.028 |
| Trail making test A, s | 59.8 ± 32.9 | 82.2 ± 53.9 | 0.000 | 86.8 ± 64.9 | 105.2 ± 79.7 | 0.025 |
| Digit span forward score | 6.3 ± 1.4 | 6.0 ± 1.5 | 0.043 | 5.8 ± 1.4 | 5.2 ± 1.4 | 0.001 |
| Digit span backward score | 3.6 ± 1.0 | 3.5 ± 1.2 | 0.242 | 3.3 ± 1.1 | 3.1 ± 1.2 | 0.234 |
| Frontal Assessment Battery score | 14.5 ± 2.5 | 13.2 ± 3.3 | 0.000 | 13.4 ± 2.9 | 12.7 ± 2.8 | 0.019 |
| Cognitive impairment by domains | ||||||
| Processing speed | 18 (2.0) | 8 (8.5) | 0.002 | 37 (4.4) | 7 (10.1) | 0.042 |
| Executive function | 55 (6.2) | 18 (19.1) | 0.000 | 65 (7.7) | 5 (7.2) | 0.561 |
| Verbal episodic memory | 77 (8.7) | 11 (11.7) | 0.217 | 103 (12.2) | 12 (17.4) | 0.146 |
| Working memory | 44 (5.0) | 9 (9.6) | 0.060 | 35 (4.2) | NA | |
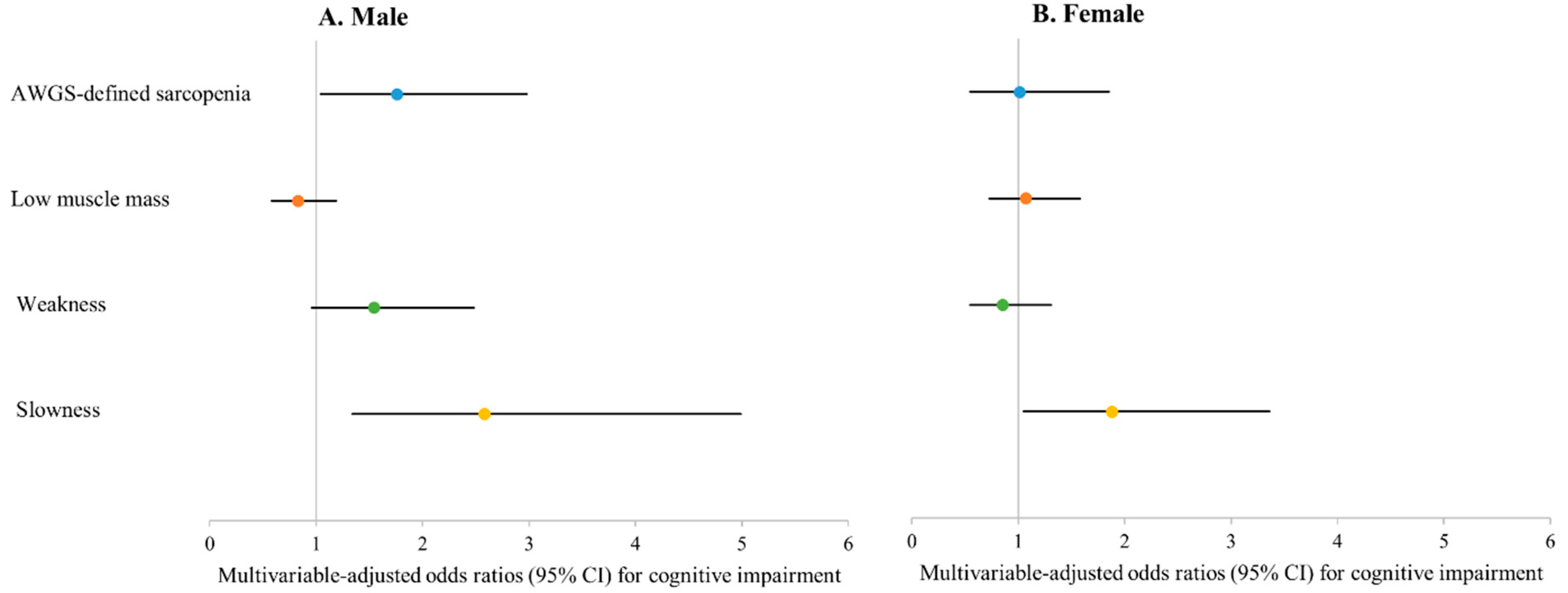
| Cognitive impairment b | 161 (18.3) | 28 (29.8) | 0.013 | 188 (22.3) | 18 (26.1) | 0.457 |
| Variable | Model 1 | Model 2 | Model 3 |
|---|---|---|---|
| Odds Ratio (95% Confidence Interval) | |||
| Impaired processing speed | |||
| AWGS-defined sarcopenia | |||
| Low muscle mass (n = 485) | 1.13 (0.52–2.47) | 1.09 (0.98–1.20) | 0.89 (0.35–2.21) |
| Weakness (n = 118) | 4.08 (1.78–9.34) | 3.44 (1.42–8.34) | 3.11 (1.22–7.90) |
| Slow gait speed (n = 47) | 6.64(2.53–17.43) | 5.44 (1.96–15.1) | 5.54 (1.84–16.64) |
| Sarcopenia (n = 94) | 4.46 (1.88–10.56) | 3.74 (1.45–9.61) | 3.46 (1.24–9.65) |
| FNIH-defined sarcopenia | |||
| Sarcopenia (n = 76) | 3.78 (1.47–9.69) | 3.02 (1.10–8.26) | 3.00 (1.08–8.42) |
| Sarcopenia (slow gait speed) (n = 11) | 8.70 (1.78–42.46) | 5.77 (1.09–30.5) | 4.47 (0.79–25.69) |
| EWGSOP2-defined sarcopenia | |||
| Sarcopenia (n = 111) | 3.65 (1.55–8.61) | 3.00 (1.17–7.66) | 2.75 (1.01–7.65) |
| Severe Sarcopenia (n = 13) | 7.11 (1.49–33.8) | 4.87 (0.95–24.92) | 3.56 (0.59–21.37) |
| Impaired executive function | |||
| AWGS-defined sarcopenia | |||
| Low muscle mass | 1.68 (1.04–2.73) | 1.01 (0.95–1.07) | 1.11 (0.65–1.90) |
| Weakness | 2.63 (1.48–4.65) | 2.64 (1.45–4.83) | 2.25 (1.19–4.25) |
| Slow gait speed | 3.20 (1.48–6.90) | 3.11 (1.40–6.91) | 3.33 (1.39–7.94) |
| Sarcopenia | 3.58 (1.99–6.36) | 3.86 (2.04–7.29) | 2.98 (1.51–5.89) |
| FNIH-defined sarcopenia | |||
| Sarcopenia | 1.75 (0.84–3.68) | 1.68 (0.78–3.63) | 1.63 (0.72–3.69) |
| Sarcopenia (slow gait speed) | 4.79 (1.24–18.46) | 4.41 (1.10–17.7) | 5.06 (1.07–23.89) |
| EWGSOP2-defined sarcopenia | |||
| Sarcopenia | 3.36 (1.93–5.88) | 3.65 (1.98–6.73) | 2.78 (1.45–5.31) |
| Severe Sarcopenia | 5.75 (1.73–19.15) | 5.46 (1.58–18.87) | 4.80 (1.17–19.70) |
| Impaired working memory | |||
| AWGS-defined sarcopenia | |||
| Low muscle mass | 0.62 (0.39–0.97) | 1.06 (1.00–1.13) | 0.54 (0.32–0.90) |
| Weakness | 1.73 (0.72–1.92) | 1.14 (0.60–2.15) | 1.01 (0.58–2.14) |
| Slow gait speed | 2.17 (0.98–4.81) | 1.67 (0.74–3.86) | 1.69 (0.71–4.00) |
| Sarcopenia | 1.38 (0.71–2.71) | 1.07 (0.53–2.18) | 1.06 (0.50–2.23) |
| FNIH-defined sarcopenia | |||
| Sarcopenia | 1.20 (0.56–2.06) | 0.93 (0.42–2.07) | 0.88 (0.39–1.99) |
| Sarcopenia (slow gait speed) | 3.88 (1.01–14.89) | 2.58 (0.64–10.41) | 2.19 (0.51–9.43) |
| EWGSOP2-defined sarcopenia | |||
| Sarcopenia | 1.12 (0.58–2.20) | 0.85 (0.42–1.72) | 0.81 (0.39–1.68) |
| Severe Sarcopenia | 1.85 (0.40–8.49) | 1.24 (0.26–5.98) | 1.01 (0.19–5.29) |
| Impaired verbal episodic memory | |||
| AWGS-defined sarcopenia | |||
| Low muscle mass | 1.18 (0.68–2.06) | 1.01 (0.94–1.08) | 0.88 (0.46–1.67) |
| Weakness | 1.75 (0.86–3.59) | 1.96 (0.92–4.16) | 1.85 (0.84–4.06) |
| Slow gait speed | 3.36 (1.43–7.90) | 3.86 (1.57–9.45) | 3.71 (1.43–9.62) |
| Sarcopenia | 2.01 (0.95–4.27) | 2.28 (1.01–5.11) | 2.00 (0.84–4.75) |
| FNIH-defined sarcopenia | |||
| Sarcopenia | 1.55 (0.64–3.76) | 1.67 (0.67–4.19) | 1.70 (0.66–4.39) |
| Sarcopenia (slow gait speed) | 3.98 (0.84–18.89) | 4.96 (0.98–25.10) | 3.89 (0.69–21.78) |
| EWGSOP2-defined sarcopenia | |||
| Sarcopenia | 1.89 (0.92–3.88) | 2.15 (0.99–4.67) | 1.92 (0.84–4.38) |
| Severe Sarcopenia | 4.37 (0.94–20.31) | 3.71 (0.76–18.02) | 2.36 (0.44–12.85) |
| Variable | Model 1 | Model 2 | Model 3 |
|---|---|---|---|
| Odds Ratio (95% Confidence Interval | |||
| Impaired processing speed | |||
| AWGS-defined sarcopenia | |||
| Low muscle mass (n = 251) | 0.87 (0.43–1.75) | 0.96 (1.05–1.23) | 0.97 (0.44–2.16) |
| Weakness (n = 163) | 2.01 (1.03–3.93) | 1.37 (0.68–2.78) | 1.19 (0.56–1.23) |
| Slow gait speed (n = 69) | 8.79 (4.48–17.24) | 4.46 (2.11–9.44) | 4.71 (2.00–11.09) |
| Sarcopenia (n = 69) | 2.46 (1.05–5.74) | 1.82 (0.74–4.48) | 1.67 (0.62–4.47) |
| FNIH-defined sarcopenia | |||
| Sarcopenia (n = 38) | 0.55 (0.70–3.90) | 0.28 (0.04–2.16) | 0.24 (0.03–1.99) |
| Sarcopenia (slow gait speed) (n = 11) | 2.00 (0.25–15.95) | 0.67 (0.80–5.53) | 0.55 (0.06–5.23) |
| EWGSOP2-defined sarcopenia | |||
| Sarcopenia (n = 57) | 2.53 (1.02–6.26) | 1.62 (0.63–4.12) | 1.18 (0.41–3.37) |
| Severe Sarcopenia (n = 14) | 8.58 (2.58–28.55) | 2.91 (0.80–10.57) | 1.79 (0.42–7.61) |
| Impaired executive function | |||
| AWGS-defined sarcopenia | |||
| Low muscle mass | 0.98 (0.57–1.69) | 0.99 (0.93–1.06) | 1.05 (0.58–1.91) |
| Weakness | 0.95 (0.50–1.81) | 0.95 (0.49–1.84) | 0.83 (0.42–1.65) |
| Slow gait speed | 2.85 (1.45–5.61) | 3.89 (1.61–7.15) | 2.97 (1.29–6.83) |
| Sarcopenia | 0.94 (0.36–2.41) | 0.96 (0.37–2.49) | 0.97 (0.36–2.61) |
| FNIH-defined sarcopenia | |||
| Sarcopenia | 0.66 (0.16–2.79) | 0.66 (0.15–2.85) | 0.57 (0.13–2.58) |
| Sarcopenia (slow gait speed) | 2.72 (0.58–12.85) | 2.94 (0.59–14.70) | 2.27 (0.42–12.37) |
| EWGSOP2-defined sarcopenia | |||
| Sarcopenia | 0.90 (0.32–2.57) | 0.91 (0.32–2.63) | 0.70 (0.23–2.12) |
| Severe Sarcopenia | 3.38 (0.92–12.42) | 3.93 (0.98–15.76) | 2.77 (0.61–12.52) |
| Impaired working memory | |||
| AWGS-defined sarcopenia | |||
| Low muscle mass | 1.02 (0.66–1.57) | 1.04 (0.99–1.09) | 1.13 (0.67–1.83) |
| Weakness | 1.42 (0.78–2.61) | 1.00 (0.60–1.66) | 0.88 (0.52–1.49) |
| Slow gait speed | 2.06 (1.12–3.79) | 1.40 (0.73–2.70) | 1.49 (0.72–2.90) |
| Sarcopenia | 1.51 (0.79–2.91) | 1.37 (0.70–2.69) | 1.36 (0.67–2.75) |
| FNIH-defined sarcopenia | |||
| Sarcopenia | 1.60 (0.69–3.73) | 1.25 (0.53–2.96) | 1.19 (0.48–2.91) |
| Sarcopenia (slow gait speed) | 5.99 (1.78–20.00) | 3.85 (1.11–13.37) | 3.36 (0.95–12.60) |
| EWGSOP2-defined sarcopenia | |||
| Sarcopenia | 1.52 (0.75–3.10) | 1.25 (0.60–2.61) | 1.02 (0.47–2.21) |
| Severe Sarcopenia | 5.43 (1.85–15.94) | 3.49 (1.12–10.86) | 2.70 (0.81–8.96) |
| Impaired verbal episodic memory | |||
| AWGS-defined sarcopenia | |||
| Low muscle mass | 0.33 (0.12–0.94) | 1.10 (1.01–1.20) | 0.33 (0.10–1.07) |
| Weakness | 2.51 (1.22–5.15) | 1.87 (0.88–3.69) | 1.75 (0.81–3.80) |
| Slow gait speed | 2.12 (0.79–5.64) | 0.93 (0.33–2.65) | 0.95 (0.31–2.95) |
| Sarcopenia | NA | NA | NA |
| FNIH-defined sarcopenia | |||
| Low muscle mass | 0.98 (0.64–2.07) | 0.88 (0.41–1.90) | 0.93 (0.41–2.10) |
| Sarcopenia | 1.42 (0.32–6.13) | 0.88 (0.20–3.92) | 0.79 (0.16–3.83) |
| Sarcopenia (slow gait speed) | 2.55 (0.32–20.49) | 1.01 (0.12–8.49) | 0.86 (0.09–8.32) |
| EWGSOP2-defined sarcopenia | |||
| Sarcopenia | 2.15 (0.99–4.67) | 1.85 (0.66–5.17) | 1.75 (0.57–5.53) |
| Severe Sarcopenia | 4.37 (0.94–20.31) | 1.68 (0.33–8.49) | 1.43 (0.24–8.60) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, M.; Won, C.W. Sarcopenia Is Associated with Cognitive Impairment Mainly Due to Slow Gait Speed: Results from the Korean Frailty and Aging Cohort Study (KFACS). Int. J. Environ. Res. Public Health 2019, 16, 1491. https://doi.org/10.3390/ijerph16091491
Kim M, Won CW. Sarcopenia Is Associated with Cognitive Impairment Mainly Due to Slow Gait Speed: Results from the Korean Frailty and Aging Cohort Study (KFACS). International Journal of Environmental Research and Public Health. 2019; 16(9):1491. https://doi.org/10.3390/ijerph16091491
Chicago/Turabian StyleKim, Miji, and Chang Won Won. 2019. "Sarcopenia Is Associated with Cognitive Impairment Mainly Due to Slow Gait Speed: Results from the Korean Frailty and Aging Cohort Study (KFACS)" International Journal of Environmental Research and Public Health 16, no. 9: 1491. https://doi.org/10.3390/ijerph16091491
APA StyleKim, M., & Won, C. W. (2019). Sarcopenia Is Associated with Cognitive Impairment Mainly Due to Slow Gait Speed: Results from the Korean Frailty and Aging Cohort Study (KFACS). International Journal of Environmental Research and Public Health, 16(9), 1491. https://doi.org/10.3390/ijerph16091491





