Depressive Symptoms After PCB Exposure: Hypotheses for Underlying Pathomechanisms via the Thyroid and Dopamine System
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Study Population
2.3. Data Collection
2.3.1. Polychlorinated and Hydroxylated Biphenyls
2.3.2. Total Lipids
2.3.3. Free Thyroxine
2.3.4. Homovanillic Acid
2.3.5. Depressive Symptoms
2.4. Statistical Analyses
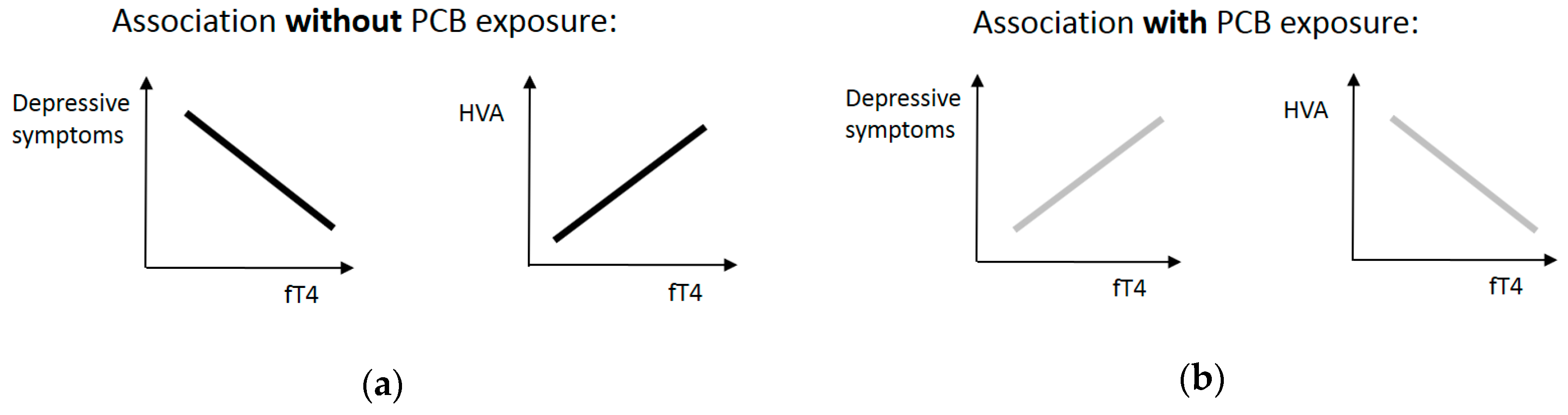
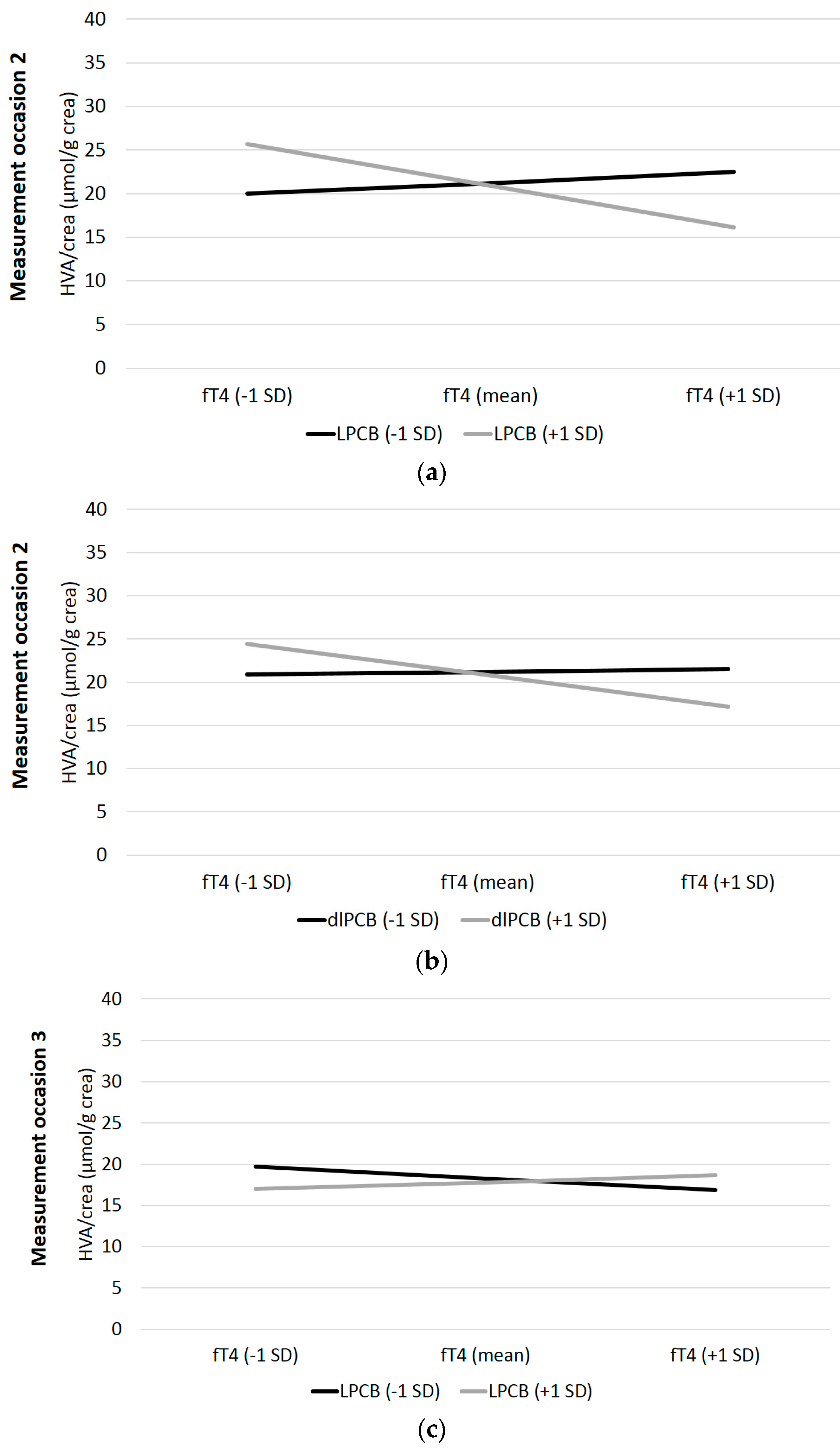
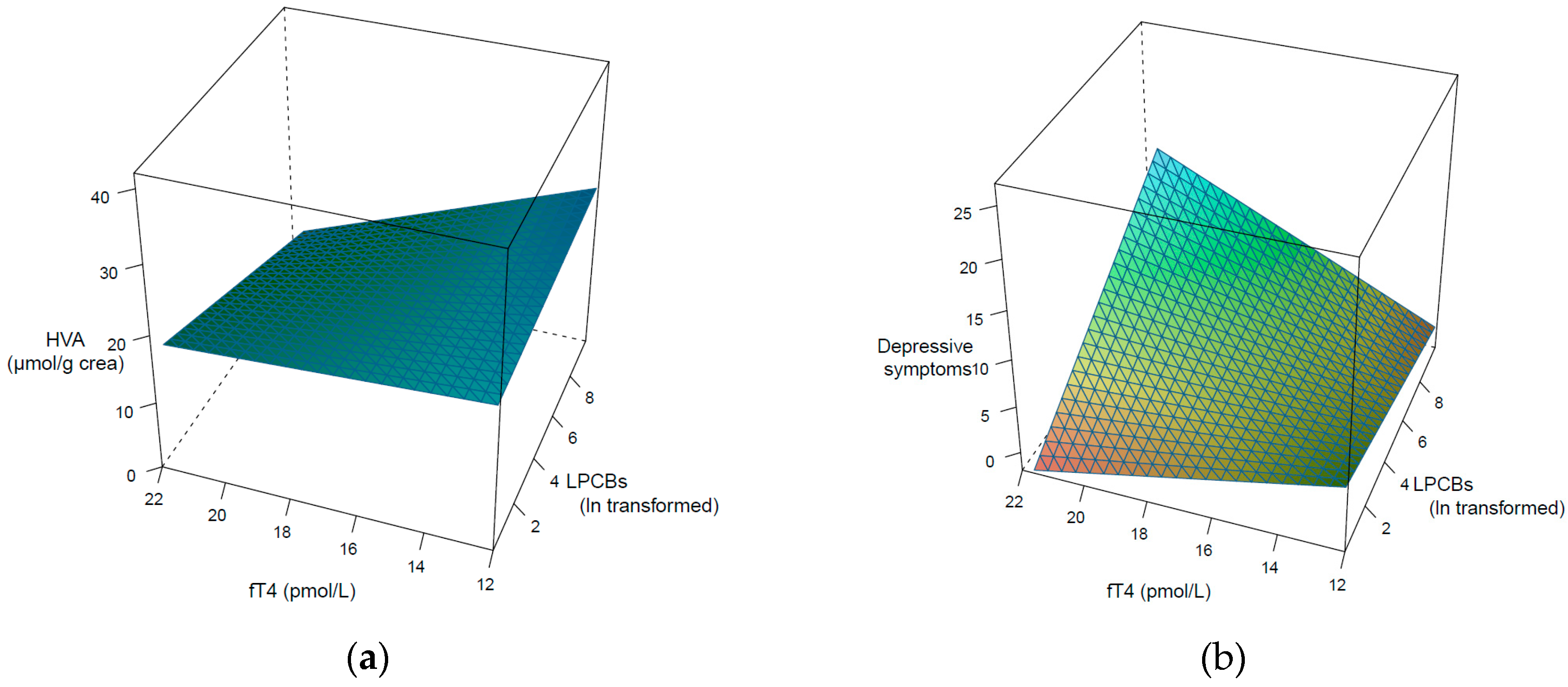
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
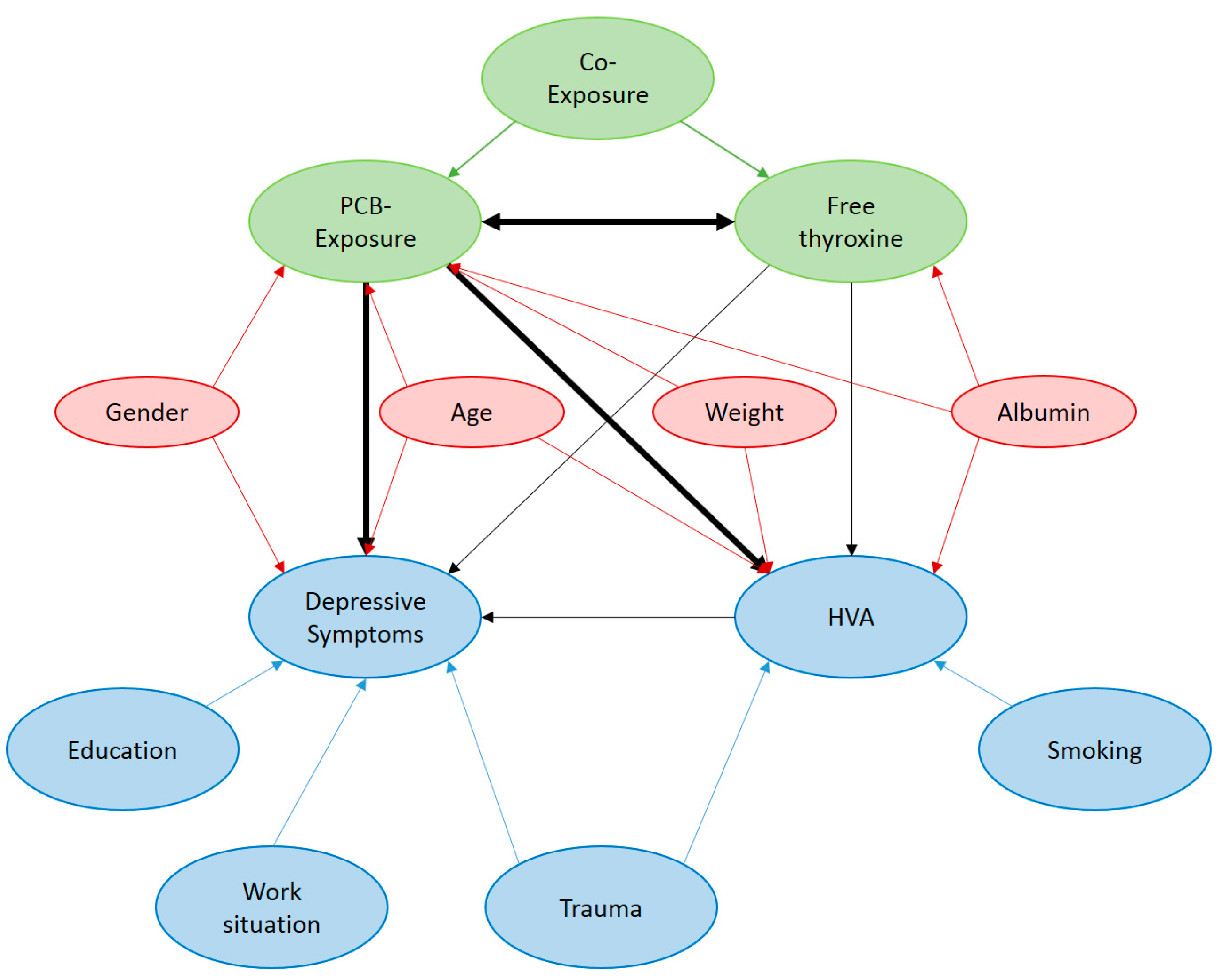
Appendix B
- Plasma was obtained by the centrifugation of blood samples (10 min at 2800 rpm), and 3.5 mL of supernatant was transferred to pre-cleaned glass vials and stored at −20 °C until analysis. Samples were thawed at room temperature before the analysis and then homogenized by vortex mixing for 1 min.
- For OH-PCBs, the sum of the free phenolic forms and the glucuronide and sulfate conjugates were determined by submitting the samples to enzymatic hydrolysis before extraction. The methodology has been previously described [38]. Briefly, 100 µL of plasma went through enzymatic hydrolysis to release the target compound from plasma, followed by protein precipitation together with 100 µL of ammonium acetate buffer 0.1mol/L (pH = 5.3) and 5 µL of ß-Glucuronidase/Arylsulfatase enzyme, which were added to a glass LC vial, mixed, and left overnight in a drying oven at 37 °C for enzymatic hydrolysis. After that, 50 µL of a mix of internal standards (10 ng/mL) and 600 µL of methanol were added, mixed by vortexing for 1 min, and centrifuged for 10 min at 4500 rpm for protein precipitation. The supernatant solutions were transferred into a vial and evaporated until 50 µL at 45 °C under a gentle stream of nitrogen. Finally, 50 µL of the 0.1 mol/L buffer was added to the remaining extract in order to make it up to 100 µL, which was analyzed by an online solid phase extraction method coupled to liquid chromatography-tandem mass spectrometry (LC-MS/MS) in multiple reactions monitoring mode (MRM). Recoveries for all compounds in plasma ranged from 71 to 134%, with an average exceeding 80%, which is available in Quinete et al. [38]. Concentrations of total OH-PCBs in plasma have been assessed, which included not only the free phenolic forms, but also the glucuronide and sulfate.
References
- Brown, J.S. Polybrominated Biphenyls and Polychlorinated Biphenyls. In Environmental and Chemical Toxins and Psychiatric Illness; Brown, J.S., Ed.; American Psychiatric Publishing, Inc.: Washington, DC, USA, 2002; pp. 251–259. ISBN 0-88048-954-5. [Google Scholar]
- Kraus, T.; Gube, M.; Lang, J.; Esser, A.; Sturm, W.; Fimm, B.; Willmes, K.; Neulen, J.; Baron, J.M.; Merk, H.; et al. Surveillance Program for Former PCB-Exposed Workers of a Transformer and Capacitor Recycling Company, Family Members, Empoyees of Surrounding Companies, and Area Residents—Executive Summary. J. Toxicol. Environ. Health Part A 2012, 75, 1241–1247. [Google Scholar] [CrossRef] [PubMed]
- Richter, S.; Steinhaeuser, K.-G.; Fiedler, H. Globaler Vertrag zur Regelung von POPs: Die Stockholm Konvention (Global Treaty for the regulation of POPs: The Stockholm convention). Z. Umweltchem Ökotox 2001, 13, 39–44. (In German) [Google Scholar] [CrossRef]
- Loganathan, B.G.; Kannan, K. Global organochlorine contamination trends: An overview. Ambio 1994, 23, 187–191. [Google Scholar]
- Schettgen, T.; Gube, M.; Esser, A.; Alt, A.; Kraus, T. Plasma Polychlorinated Biphenyls (PCB) Levels of Workers in a Transformer Recycling Company, Their Family Members, and Employees of Surrounding Companies. J. Toxicol. Environ. Health Part A 2012, 75, 414–422. [Google Scholar] [CrossRef]
- Kocan, A.; Drobna, B.; Petrik, J.; Jursa, S.; Chovancova, J.; Conka, K.; Balla, B.; Sovcikova, E.; Trnovec, T. Human exposure to PCBs and some other persistent organochlorines in Eastern Slovakia as a consequence of former PCB production. Organohalogen Compd. 2004, 66, 3490–3497. [Google Scholar]
- Longnecker, M.P.; Rogan, W.J.; Lucier, G. The Human Health Effects of DDT (Dichlorodiphenyltrichloroethane) and PCBs (Polychlorinated Biphenyls) and an Overview of Organochlorines in Public Health. Annu. Rev. Public Health 1997, 18, 211–244. [Google Scholar] [CrossRef] [PubMed]
- Gaum, P.M.; Lang, J.; Esser, A.; Schettgen, T.; Neulen, J.; Kraus, T.; Gube, M. Exposure to polychlorinated biphenyls and the thyroid gland—Examining and discussion possible longitudinal health effects in humans. Environ. Res. 2016, 148, 112–121. [Google Scholar] [CrossRef] [PubMed]
- Lauby-Secretan, B.; Loomis, D.; Grosse, Y.; Ghissassi, F.E.; Bouvard, V.; Benbrahim-Tallaa, L.; Guha, N.; Baan, R.; Mattock, H.; Straif, K.; et al. Carcinogenicity of polychlorinated biphenyls and polybrominated biphenyls. Lancet Oncol. 2013, 14, 287–288. [Google Scholar] [CrossRef]
- Gaum, P.M.; Esser, A.; Schettgen, T.; Gube, M.; Kraus, T.; Lang, J. Prevalence and incidence rates of mental syndromes after occupational exposure to polychlorinated biphenyls. Int. J. Hyg. Environ. Health 2014, 217, 765–774. [Google Scholar] [CrossRef]
- Kilburn, K.H.; Warsaw, R.H.; Shields, M.G. Neurobehavioral Dysfunction in Firemen Exposed to Polychlorinated Biphenyls (PCBs): Possible Improvement after Detoxification. Arch. Environ. Health 1989, 44, 345–350. [Google Scholar] [CrossRef]
- Fitzgerald, E.F.; Belanger, E.E.; Gomez, M.I.; Cayo, M.; McCaffrey, R.J.; Seegal, R.F.; Jansig, R.L.; Hwang, S.; Hicks, H.E. Polychlorinated Biphenyl Exposure and Neuropsychological Status among Older Residents of Upper Hudson River Communities. Environ. Health Perspect. 2008, 116, 209–215. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.S.; Gershon, S. Dopamine and depression. J. Neural Transm. 1993, 91, 75–109. [Google Scholar] [CrossRef]
- Richardson, J.R.; Miller, G.W. Acute exposure to Aroclor 1061 or 1260 differentially affects dopamine transporter and vesicular monoamine transporter 2 levels. Toxicol. Lett. 2004, 148, 29–40. [Google Scholar] [CrossRef] [PubMed]
- Seegal, R.F.; Marek, K.L.; Seibyl, J.P.; Jennings, D.L.; Molho, E.S.; Higgins, D.S.; Factor, S.A.; Fitzgerald, E.F.; Hills, E.A.; Korrick, S.A.; et al. Occupational exposure to PCBs reduces striatial dopamine transporter densities only in women: A β-CIT imaging study. Neurobiol. Dis. 2010, 38, 219–225. [Google Scholar] [CrossRef] [PubMed]
- Choksi, N.Y.; Kondavanti, P.R.S.; Tilson, H.A.; Booth, R.G. Effects of polychlorinated biphenyls (PCBs) on brain tyrosine hydroxylase activity and dopamine synthesis in rats. Fundam. Appl. Toxicol. 1997, 39, 76–80. [Google Scholar] [CrossRef]
- Putschögl, F.M.; Gaum, P.M.; Schettgen, T.; Gube, M.; Kraus, T.; Lang, J. Effects of Occupational Exposure to Polychlorinated Biphenyls on Urinary Metabolites of Neurotransmitters: A Cross-Sectional and Longitudinal Perspective. Int. J. Hyg. Environ. Health 2015, 218, 452–460. [Google Scholar] [CrossRef]
- Fonnum, F.; Mariussen, E. Mechanisms involved in the neurotoxic effects of environmental toxicants such as polychlorinated biphenyls and brominated flame retardants. J. Neurochem. 2009, 111, 1327–1347. [Google Scholar] [CrossRef]
- Gaum, P.M.; Gube, M.; Schettgen, T.; Putschögl, F.M.; Kraus, T.; Fimm, B.; Lang, J. Polychlorinated biphenyls and depression: Cross-sectional and longitudinal investigation of a dopamine-related neurochemical path in the German HELPcB surveillance program. Environ. Health 2017, 16, 1–11. [Google Scholar] [CrossRef]
- Bloom, M.S.; Jansing, R.L.; Kannan, K.; Rej, R.; Fitzgerald, E.F. Thyroid hormones are associated with exposure to persistent organic pollutants in aging residents of upper Hudson River communities. Int. J. Hyg. Environ. Health 2014, 217, 473–482. [Google Scholar] [CrossRef]
- Xu, P.; Lou, X.; Ding, G.; Shen, H.; Wu, L.; Chen, Z.; Han, J.; Wang, X. Effects of PCBs and PBDEs on thyroid hormone, lymphocyte proliferation, hematology and kidney injury markers in residents of an e-waste dismantling area in Zhejiang, China. Sci. Total Environ. 2015, 536, 215–222. [Google Scholar] [CrossRef]
- Bloom, M.S.; Vena, J.E.; Olson, J.R.; Kostyniak, P.J. Assessment of polychlorinated biphenyl congeners, thyroid stimulating hormone, and free thyroxine among New York State anglers. Int. J. Hyg. Environ. Health 2009, 212, 599–611. [Google Scholar] [CrossRef]
- Hansen, J.M.; Siersbaek-Nielsen, S. Cerebrospinal Fluid Thyroxine. J. Clin. Endocr. 1969, 29, 1023–1026. [Google Scholar] [CrossRef]
- Gulseren, S.; Gulseren, L.; Hekimsoy, Z.; Cetinay, P.; Ozen, C.; Tokatlioglu, B. Depression, anxiety, health-related quality of life, and disability in patients with overt and subclinical thyroid dysfunction. Arch. Med. Res. 2006, 37, 133–139. [Google Scholar] [CrossRef]
- Berent, D.; Zboralski, K.; Orzechowska, A.; Galecki, P. Thyroid hormones association with depression severity and clinical outcome in patients with major depressive disorder. Mol. Biol. Rep. 2014, 41, 2419–2425. [Google Scholar] [CrossRef]
- Hage, M.P.; Azar, S.T. The Link between Thyroid function and Depression. J. Thyroid. Res. 2012. [Google Scholar] [CrossRef]
- Maayan, M.L.; Sellitto, R.V.; Volpert, E.M. Dopamine and L-Dopa: Inhibition of Thyrotropin-Stimulated Thyroidal Thyroxine Release. Endocrinology 1986, 118, 632–636. [Google Scholar] [CrossRef]
- Engstöm, G.; Svensson, T.H.; Waldeck, B. Thyroxine and brain catecholamines: Increased transmitter synthesis and increased receptor sensitivity. Brain Res. 1974, 77, 471–483. [Google Scholar] [CrossRef]
- Hassan, W.A.; Aly, M.S.; Rahman, T.A.; Shahat, A.S. Impact of experimental hypothyroidism on monoamine level in discrete brain regions and other peripheral tissues of young and adult male rats. Int. J. Dev. Neurosci. 2013, 31, 225–233. [Google Scholar] [CrossRef]
- Hassan, W.A.; Rahman, T.A.; Aly, M.S.; Shahat, A.S. Alterations in monoamines level in discrete brain regions and other peripheral tissues in young and adult male rats during experimental hyperthyroidism. Int. J. Dev. Neurosci. 2013, 31, 311–318. [Google Scholar] [CrossRef]
- Mansourian, A.R. Abnormal serum thyroid hormones concentration with healthy functional gland: A Review on the metabolic role of thyroid hormones transporter proteins. Pak. J. Biol. Sci. 2011, 14, 313–326. [Google Scholar] [CrossRef]
- Schreiber, G.; Southwell, B.R.; Richardson, S.J. Hormone delivery system to the brain—Transthyretin. Exp. Clin. Endocrinol. Diabetes 1995, 103, 75–80. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, J.A.; Benson, M.D. Transthyretin: A review from structural perspective. Cell. Mol. Life Sci. 2001, 58, 1491–1521. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, G.M.; Hatterer, J.A.; Herbert, J.; Chen, X.; Roose, S.P.; Attia, E.; Mann, J.; Maragell, L.B.; Goetz, R.R.; Gorman, J.M. Low levels of transthyretin in the CSF of depressed patients. Am. J. Psychiatry 1999, 156, 710–715. [Google Scholar] [CrossRef]
- Chauhan, K.R.; Kodavanti, P.R.S.; McKinney, J.D. Assessing the role of ortho-substitution on polychlorinated biphenyl binding to transthyretin, a thyroxine transport protein. Toxicol. Appl. Pharmacol. 2000, 162, 10–21. [Google Scholar] [CrossRef] [PubMed]
- Hamers, T.; Kamstra, J.H.; Cenijn, P.H.; Pencikova, K.; Palkova, L.; Simeckova, P.; Vondracek, J.; Andersson, P.L.; Stenberg, M.; Machala, M. In vitro toxicity profiling of ultrapure non-dioxin-like polychlorinated biphenyl congeners and their relative toxic contribution to PCB mixtures in humans. Toxicol. Sci. 2011, 121, 88–100. [Google Scholar] [CrossRef]
- Lans, M.C.; Klasson-Wehler, E.; Willemsen, M.; Meussen, E.; Safe, S.; Brouwer, A. Structure-dependent, competitive interaction of hydroxy-polychlorobiphenyls, -dibenzo-p-dioxins and—dibenzofurans with human transthyretin. Chem.-Biol. Interact. 1993, 88, 7–21. [Google Scholar] [CrossRef]
- Quinete, N.; Kraus, T.; Belov, V.N.; Aretz, C.; Esser, A.; Schettgen, T. Fast determination of hydroxylated polychlorinated biphenyls in human plasma by online solid phase extraction coupled to liquid chromatography-tandem mass spectrometry. Anal. Chim. Acta 2015, 888, 94–102. [Google Scholar] [CrossRef] [PubMed]
- Schettgen, T.; Gube, M.; Alt, A.; Fromme, H.; Kraus, T. Pilot study on the exposure of the German general population to non-dioxin-like and dioxin-like PCBs. Int. J. Hyg. Environ. Health 2011, 214, 319–325. [Google Scholar] [CrossRef]
- Schettgen, T.; Alt, A.; Esser, A.; Kraus, T. Current data on the background burden to the persistent organochlorine pollutants HCB, p,p’-DDE as well as PCB 138, PCB 153 and PCB 180 in plasma of the general population in Germany. Int. J. Hyg. Environ. Health 2015, 218, 380–385. [Google Scholar] [CrossRef] [PubMed]
- Bernert, J.T.; Turner, W.E.; Patterson, D.G., Jr.; Needham, L.L. Calculation of serum “total lipid” concentrations for the adjustment of persistent organohalogen toxicant measurements in human samples. Chemosphere 2007, 68, 824–831. [Google Scholar] [CrossRef] [PubMed]
- Quinete, N.; Esser, A.; Kraus, T.; Schettgen, T. Determination of hydroxylated polychlorinated biphenyls (OH-PCBs) in human urine in a highly occupationally exposed German cohort: New prospects for urinary biomarkers of PCB exposure. Environ. Int. 2016, 97, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Eisenhofer, G.; Kopin, I.J.; Goldstein, D.S. Catecholamine Metabolism: A contemporary view with implications for physiology and medicine. Pharmacol. Rev. 2004, 56, 333–349. [Google Scholar] [CrossRef] [PubMed]
- Amin, F.; Davidson, M.; Davis, K.L. Homovanillic Acid Measurement in Clinical Research: A Review of Methodology. Schizophr. Bull. 1992, 18, 123–148. [Google Scholar] [CrossRef] [PubMed]
- Commission “Human Biomonitoring” of the German Federal Environment Agency. Normierung von Stoffgehalten im Urin—Kreatinin [Standardization of substance contents in urine—Creatinine]. Bundesgesundheitsbl Gesundheitsforsch Gesundheitsschutz 2005, 48, 616–618. (In German) [Google Scholar] [CrossRef] [PubMed]
- Loewe, B.; Spitzer, R.L.; Zipfel, S.; Herzog, W. PHQ-D—Gesundheitsfragebogen für Patienten, Manual—Komplettversion und Kurzform—Autorisierte deutsche Version des “Prime MD Patient Health Questionnaire (PHQ)”; Pfizer: Heidelberg, Germany, 2002; (In German). Available online: https://www.klinikum.uni-heidelberg.de/fileadmin/Psychosomatische_Klinik/download/PHQ_Manual1.pdf (accessed on 15 March 2019).
- Greenland, S.; Pearl, J.; Robins, J.M. Causal diagrams for epidemiologic research. Epidemiology 1999, 10, 37–48. [Google Scholar] [CrossRef] [PubMed]
- Textor, J.; Hardt, J.; Knueppel, S. DAGitty: A graphical tool for analyzing causal diagrams. Epidemiology 2011, 22, 745. [Google Scholar] [CrossRef]
- Bray, G.A.; Fisher, D.A.; Chopra, I.J. Relation of thyroid hormones to body-weight. Lancet 1976, 307, 1206–1208. [Google Scholar] [CrossRef]
- Thapa, B.R.; Walia, A. Liver function tests and their interpretation. Indian J. Pediatr. 2007, 74, 663–671. [Google Scholar] [CrossRef]
- Jacobi, F.; Wittchen, H.-U.; Hölting, C.; Höfler, M.; Pfister, H.; Müller, N.; Lieb, R. Prevalence, co-morbidity and correlates of metnal disorders in the general population: Results from the German health interview and examination survey (GHS). Psychol. Med. 2004, 34, 1–15. [Google Scholar] [CrossRef]
- Abdelouahab, N.; Mergler, D.; Takser, L.; Vanier, C.; St-Jean, M.; Baldwin, M.; Spear, P.A.; Chan, H.M. Gender differences in the effects of organochlorines, mercury, and lead on thyroid hormone levels in lakeside communitiers of Quebec (Canada). Environ. Res. 2008, 107, 380–392. [Google Scholar] [CrossRef]
- IBM Corp. IBM SPSS Statistics for Windows, version 25.0; IBM Corp: Armonk, NY, USA, 2017. [Google Scholar]
- Hayes, A.F. Introduction to Mediation, Moderation, and Conditional Process Analysis—A Regression Based Approach, 2nd ed.; Guilford Press: New York, NY, USA, 2017; ISBN 9781462534654. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2017. Available online: https://www.R-project.org/ (accessed on 15 March 2019).
- RStudio Team. RStudio: Integrated Development for R; RStudio Inc.: Boston, MA, USA, 2016; Available online: http://www.rstudio.com/ (accessed on 15 March 2019).
- Kuznetsova, A.; Brockhoff, P.B.; Christensen, R.H.B. lmerTest Package: Tests in Linear Mixed Effects Models. J. Stat. Softw. 2017, 82, 1–6. [Google Scholar] [CrossRef]
- Sarkar, D. Lattice: Multivariate Data Visualization with R; Springer: New York, NY, USA, 2008; ISBN 978-0-387-75968-5. [Google Scholar]
- Baron, R.M.; Kenny, D.A. The moderator-mediator variable Distinction in social psychological research: Conceptual, strategic, and statistical considerations. J. Pers. Soc. Psychol. 1986, 51, 1173–1182. [Google Scholar] [CrossRef] [PubMed]
- Lambert, G.; Johansson, M.; Ågren, H.; Friberg, P. Reduced brain norepinephrine and dopamine release in treatment-refractory depressive illness. Arch. Gen. Psychiatry 2000, 57, 787–793. [Google Scholar] [CrossRef] [PubMed]
- Lans, M.C.; Spiertz, C.; Brouwer, A.; Koeman, J.H. Different competition of thyroxine binding to transthyretin and thyroxine-binding globulin by hydroxyl-PCBs, PCDs and PCDFs. Eur. J. Pharm.-Environ. 1994, 270, 129–136. [Google Scholar] [CrossRef]
- Bitman, J.; Cecil, H.C. Estrogenic activity of DDT analogs and polychlorinated biphenyls. J. Agric. Food Chem. 1970, 18, 1108–1112. [Google Scholar] [CrossRef] [PubMed]
- Connor, K.; Ramamoorthy, K.; Moore, M.; Mustain, M.; Chen, I.; Safe, S.; Zacherewski, T.; Gillesby, B.; Joyeux, A.; Balaguer, P. Hydroxylated Polychlorinated Biphenyls (PCBs) as Estrogens and Antiestrogens: Structure–Activity Relationships. Toxicol. Appl. Pharmacol. 1997, 111–123. [Google Scholar] [CrossRef]
- Arafah, B.M. Increased need for thyroxine in women with hypothyroidism during estrogen therapy. N. Engl. J. Med. 2001, 344, 1743–1749. [Google Scholar] [CrossRef] [PubMed]
- Toney, T.W.; Katzenellenbogen, B.S. Antiestrogen Action in the Medial Basal Hypothalamus and Pituitary of Immature Female Rats: Insights Concerning Relationships among Estrogen, Dopamine, and Prolactin. Endocrinology 1986, 119, 2661–2669. [Google Scholar] [CrossRef]
- Dirinck, E.; Dirtu, A.C.; Malarvannan, G.; Covaci, A.; Jorens, P.G.; Van Gaal, L.F. A preliminary link between hydroxylated metabolites of polychlorinated biphenyls and free thyroxine in humans. Int. J. Environ. Res. Public Health 2016, 13, 421. [Google Scholar] [CrossRef]
- Langer, P.; Kocan, A.; Tajtakova, M.; Radikova, Z.; Petrik, J.; Koska, J.; Ksinantova, L.; Imrich, R.; Huckova, M.; Chovancova, J.; et al. Possible effects of persistent organochlorinated pollutants cocktail on thyroid hormone levels and pituiary-thyroid interrelations. Chemosphere 2007, 70, 110–118. [Google Scholar] [CrossRef]
- Dewailly, É.; Mulvad, G.; Pedersen, H.S.; Ayotte, P.; Demers, A.; Weber, J.-P.; Hansen, J.C. Concentration of organochlorines in human brain, liver, and adipose tissue autopsy samples from Greenland. Environ. Health Perspect. 1999, 107, 823–828. [Google Scholar] [CrossRef] [PubMed]
- Seelbach, M.; Chen, L.; Powell, A.; Choi, Y.J.; Zhang, B.; Henning, B.; Toborek, M. Polychlorinated biphenyls disrupt blood-brain barrier integrity and promote brain metastasis formation. Environ. Health Perspect. 2009, 118, 479–484. [Google Scholar] [CrossRef] [PubMed]
- Kroenke, K.; Spitzer, R.L.; Williams, J.B.W. The PHQ-9: Validity of a brief depression severity measure. J. Gen. Intern. Med. 2001, 16, 606–613. [Google Scholar] [CrossRef] [PubMed]
| t1 | t2 | t3 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Ref | M (SD) | Mdn | Range | M (SD) | Mdn | Range | M (SD) | Mdn | Range | |
| ∑LPCBs (µg/L plasma) | 0.02 1 | 4.6 (21.0) | 0.2 | <LOD–190.0 | 3.5 (16.3) | 0.1 | <LOD–126.4 | 2.6 (13.1) | 0.1 | <LOD–114.8 |
| ∑HPCBs (µg/L plasma) | 2.36 ² | 8.9 (19.2) | 2.6 | 0.2–169.1 | 8.0 (16.8) | 2.6 | 0.3–152.1 | 8.2 (15.7) | 3.0 | 0.3–135.5 |
| ∑dlPCBs (µg/L plasma) | 0.05 1 | 3.4 (8.4) | 0.5 | 0.1–62.4 | 2.9 (7.1) | 0.5 | 0.1–49.6 | 2.4 (5.7) | 0.4 | 0.1–36.7 |
| ∑OH-PCBs (µg/L plasma) | - ³ | 5.2 (10.3) | 1.7 | 0.2–71.4 | 4.6 (10.4) | 1.5 | 0.1–88.8 | 4.2 (6.0) | 1.5 | 0.1–35.8 |
| fT4 (pmol/L) | 12–22 4 | 15.8 (2.0) | 15.7 | 10.7–20.8 | 16.0 (2.0) | 16.0 | 11.9–21.6 | 15.7 (2.0) | 15.6 | 12.2–21.6 |
| HVA/crea (µmol/g crea) | <42 4 | 19.4 (9.9) | 17.9 | 6.3–79.0 | 21.0 (8.0) | 19.1 | 11.8–55.6 | 17.8 (5.7) | 17.5 | 7.1–37.0 |
| Depressive symptoms | 0–27 | 3.2 (3.6) | 2.0 | 0–20 | 3.5 (3.6) | 2.0 | 0–19 | 3.7 (4.4) | 2.0 | 0–22 |
| Age (years) | - | 44.3 (12.8) | 45.0 | 22–83 | 45.3 (12.8) | 46.0 | 23–84 | 46.3 (12.8) | 47.0 | 24–85 |
| Total lipids (g/L serum) | 8.6 5 | 7.6 (2.2) | 7.4 | 4.4–18.9 | 7.6 (1.9) | 7.4 | 4.2–13.2 | 7.7 (2.1) | 7.3 | 4.7–15.2 |
| Albumin (g/L serum) | 35–52 4 | 48.1 (2.7) | 47.8 | 41.4–55.4 | 49.1 (2.7) | 48.8 | 43.4–57.2 | 48.0 (2.9) | 47.7 | 41.6–54.7 |
| Weight (kg) | - | 88.6 (16.5) | 85.0 | 54–140 | 88.6 (16.0) | 85.5 | 57–145 | 87.5 (15.5) | 85.0 | 58–125 |
| PCB | Spearmans Rho | p |
|---|---|---|
| t1 | ||
| lip156 | 0.170 | 0.034 |
| t3 | ||
| lip123 | 0.187 | 0.023 |
| OH-PCB | ||
| t1 | ||
| 3OHCB153 | 0.208 | 0.013 |
| 4OHCB172_180 | 0.198 | 0.017 |
| t3 | ||
| 4OHCB9 | 0.201 | 0.015 |
| HVA/Crea | Depressive Symptoms | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Moderation | β | S.E. | t | p | ΔR² | β | S.E. | t | p | ΔR² |
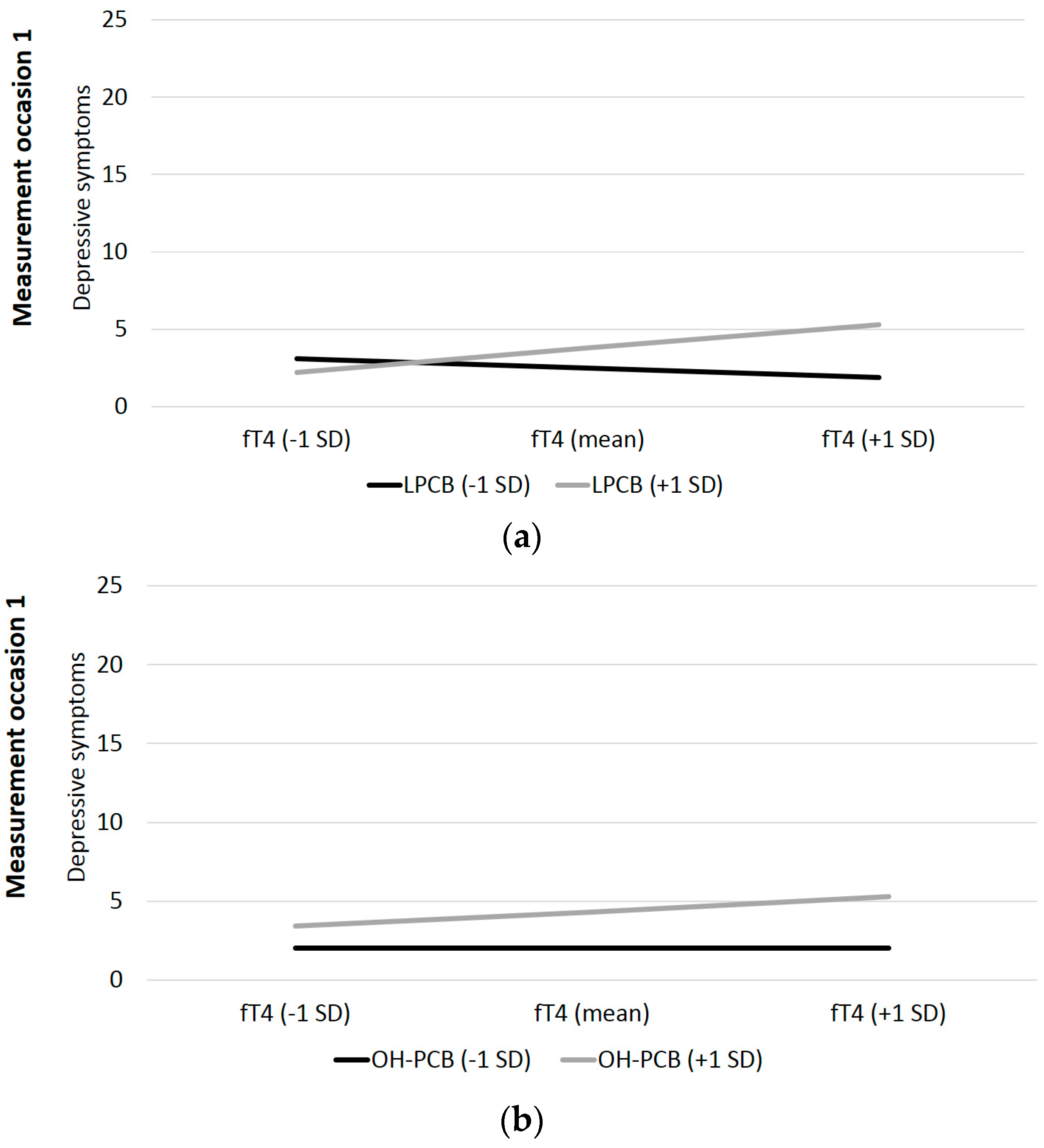
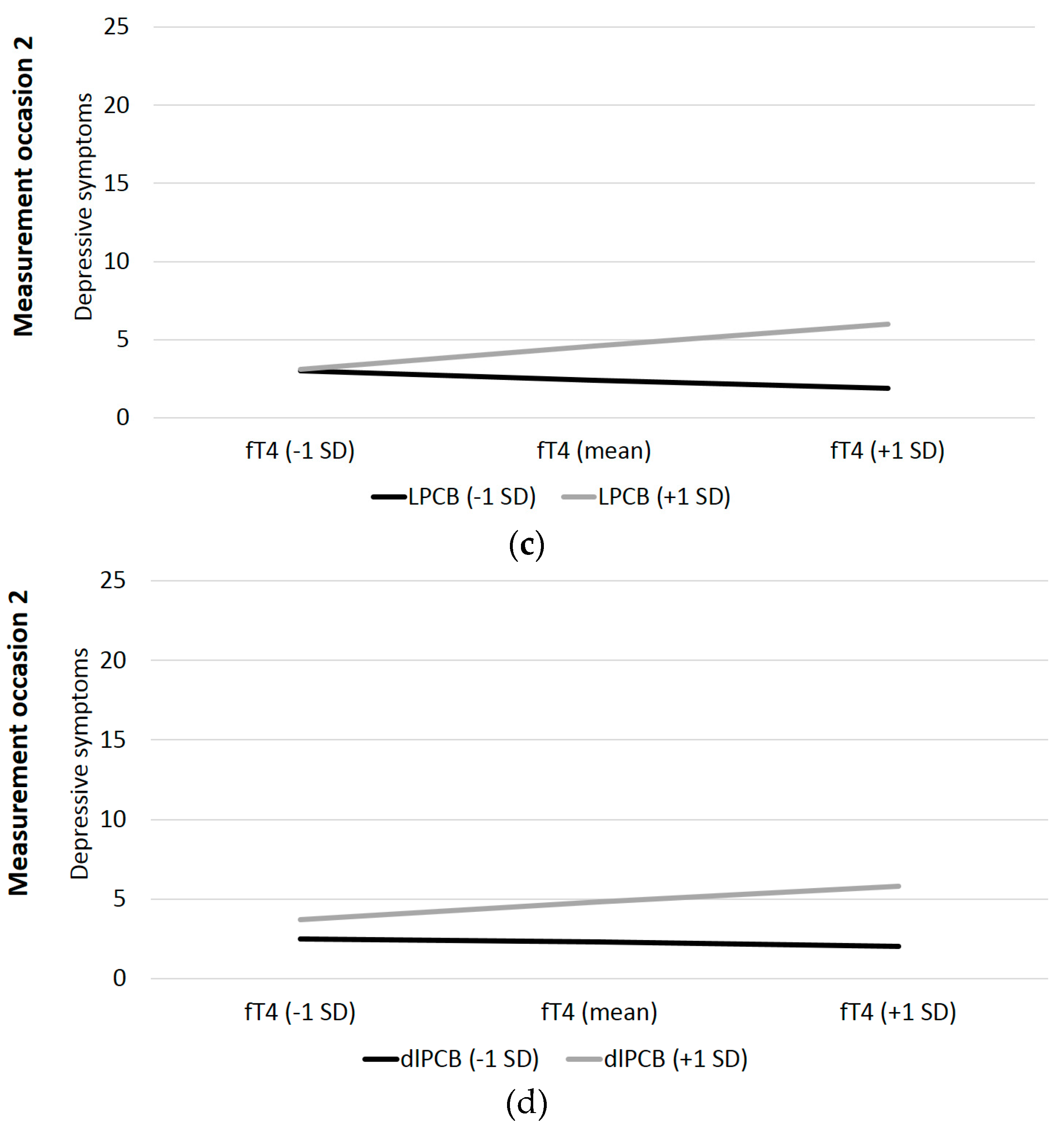
| t1: LPCB*fT4 | −0.03 | 0.11 | −0.30 | 0.38 | 0.001 | 0.32 | 0.10 | 3.38 | 0.001 | 0.091 |
| t2: LPCB*fT4 | −0.34 | 0.09 | −3.77 | <0.001 | 0.101 | 0.31 | 0.13 | 2.44 | 0.01 | 0.053 |
| t3: LPCB*fT4 | 0.35 | 0.18 | 1.98 | 0.03 | 0.045 | −0.12 | 0.13 | −0.89 | 0.19 | 0.009 |
| t1: HPCB*fT4 | −0.11 | 0.12 | −0.86 | 0.19 | 0.007 | 0.07 | 0.10 | 0.69 | 0.25 | 0.004 |
| t2: HPCB*fT4 | −0.16 | 0.09 | −1.68 | 0.05 | 0.022 | 0.13 | 0.12 | 1.02 | 0.16 | 0.009 |
| t3: HPCB*fT4 | 0.07 | 0.13 | 0.49 | 0.32 | 0.003 | −0.09 | 0.10 | −0.89 | 0.19 | 0.008 |
| t1: dlPCB*fT4 | −0.08 | 0.12 | −0.64 | 0.26 | 0.004 | 0.13 | 0.09 | 1.37 | 0.09 | 0.016 |
| t2: dlPCB*fT4 | −0.23 | 0.09 | −2.51 | 0.01 | 0.048 | 0.20 | 0.12 | 1.69 | <0.05 | 0.025 |
| t3: dlPCB*fT4 | 0.13 | 0.15 | 0.84 | 0.20 | 0.008 | −0.14 | 0.11 | −1.23 | 0.11 | 0.015 |
| t1: OH-PCB*fT4 | −0.05 | 0.12 | 0.41 | 0.35 | 0.002 | 0.16 | 0.09 | 1.73 | 0.04 | 0.025 |
| t2: OH-PCB*fT4 | −0.09 | 0.07 | −1.22 | 0.11 | 0.012 | 0.15 | 0.10 | 1.55 | 0.06 | 0.022 |
| t3: OH-PCB*fT4 | 0.15 | 0.15 | 1.03 | 0.15 | 0.013 | −0.17 | 0.11 | −1.51 | 0.07 | 0.023 |
| Homovanillic Acid | Depressive Symptoms | |||||
|---|---|---|---|---|---|---|
| β | t | p | β | t | p | |
| Fixed Effect | ||||||
| fT4 | −0.13 | −2.00 | 0.03 | 0.04 | 0.65 | 0.26 |
| LPCBs | −0.08 | −1.32 | 0.10 | 0.18 | 3.35 | <0.001 |
| Age | 0.11 | 1.68 | 0.05 | −0.06 | −1.14 | 0.13 |
| Weight | −0.27 | −4.55 | <0.001 | −0.02 | −0.23 | 0.39 |
| Albumin | −0.04 | −0.59 | 0.28 | −0.07 | −1.23 | 0.11 |
| Gender | 0.47 | 2.06 | 0.02 | 0.44 | 2.23 | 0.02 |
| fT4*PCBs | −0.11 | −1.72 | 0.04 | 0.19 | 3.37 | <0.001 |
| R² | 0.140 | 0.104 | ||||
| Random Effect (for measurement occasion) | ||||||
| Variance | 0.05 | 0.00 | ||||
| Standard deviation | 0.23 | 0.00 | ||||
| R² | 0.050 | 0.000 | ||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gaum, P.M.; Gube, M.; Esser, A.; Schettgen, T.; Quinete, N.; Bertram, J.; Putschögl, F.M.; Kraus, T.; Lang, J. Depressive Symptoms After PCB Exposure: Hypotheses for Underlying Pathomechanisms via the Thyroid and Dopamine System. Int. J. Environ. Res. Public Health 2019, 16, 950. https://doi.org/10.3390/ijerph16060950
Gaum PM, Gube M, Esser A, Schettgen T, Quinete N, Bertram J, Putschögl FM, Kraus T, Lang J. Depressive Symptoms After PCB Exposure: Hypotheses for Underlying Pathomechanisms via the Thyroid and Dopamine System. International Journal of Environmental Research and Public Health. 2019; 16(6):950. https://doi.org/10.3390/ijerph16060950
Chicago/Turabian StyleGaum, Petra Maria, Monika Gube, André Esser, Thomas Schettgen, Natalia Quinete, Jens Bertram, Franziska Maria Putschögl, Thomas Kraus, and Jessica Lang. 2019. "Depressive Symptoms After PCB Exposure: Hypotheses for Underlying Pathomechanisms via the Thyroid and Dopamine System" International Journal of Environmental Research and Public Health 16, no. 6: 950. https://doi.org/10.3390/ijerph16060950
APA StyleGaum, P. M., Gube, M., Esser, A., Schettgen, T., Quinete, N., Bertram, J., Putschögl, F. M., Kraus, T., & Lang, J. (2019). Depressive Symptoms After PCB Exposure: Hypotheses for Underlying Pathomechanisms via the Thyroid and Dopamine System. International Journal of Environmental Research and Public Health, 16(6), 950. https://doi.org/10.3390/ijerph16060950






