Antibacterial Activities of Crude Secondary Metabolite Extracts from Pantoea Species Obtained from the Stem of Solanum mauritianum and Their Effects on Two Cancer Cell Lines
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Collection
2.2. Endophytes Isolation and Identification
2.2.1. Morphological Identification of Bacterial Endophytes
2.2.2. Molecular Identification
Genomic DNA Extraction, Polymerase Chain Reaction, and Sequencing
Phylogenetic Analysis
2.3. Biological Activity Assay
2.3.1. Isolation of Secondary Metabolites from Endophytes
2.3.2. Extraction of the Solanum Mauritianum Plant Part
2.3.3. Determination of Anti-Bacterial Activity
2.3.4. Anticancer Assays
3. Results
3.1. Identification of Bacterial Endophytes
3.2. Antibacterial Activity of Bacterial Endophytes and Plant Part
3.3. Effects of Bacterial Endophytes and Plant Parts on Cancer Cells
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Okeke, I.N.; Laxminarayan, R.; Bhutta, Z.A.; Duse, A.G.; Jenkins, P.; O’Brien, T.F.; Pablos-Mendez, A.; Klugman, K.P. Antimicrobial resistance in developing countries. Part I: Recent trends and current status. Lancet Infect. Dis. 2005, 5, 481–493. [Google Scholar] [CrossRef]
- Cos, P.; Vlietinck, A.J.; Berghe, V.D.; Maes, L. Anti-infective potential of natural products: How to develop a stronger in vitro “proof-of-concept”. J. Ethnopharmacol. 2006, 106, 290–302. [Google Scholar] [CrossRef] [PubMed]
- Strobel, G.; Daisy, B. Bioprospecting for microbial endophytes and their natural products. Microbiol. Mol. Biol. Rev. 2003, 67, 491–502. [Google Scholar] [CrossRef]
- Strobel, G.A. Endophytes as sources of bioactive products. Microbes Infect. 2003, 5, 535–544. [Google Scholar] [CrossRef]
- Wilson, D. ‘Endophyte: The Evolution of a Term, and Clarification of Its Use and Definition’. Oikos 1995, 73, 274–276. [Google Scholar] [CrossRef]
- Azevedo, J.L.; Maccheroni, W., Jr.; Pereira, J.O.; de Araújo, W.L. Endophytic microorganisms: A review on insect control and recent advances on tropical plants. Electron. J. Biotechnol. 2000, 3, 15–16. [Google Scholar] [CrossRef]
- Stone, J.K.; Bacon, C.W.; White, J.F. An overview of endophytic microbes: Endophytism defined. In Microbial Endophytes; Bacon, C.W., White, J.F., Eds.; Marcel Decker Inc.: New York, NY, USA 2000; p. 3030. [Google Scholar]
- Saikkonen, K.; Wali, P.; Helander, M.; Faeth, S.H. Evolution of endophyte-plant symbiosis. Trends Plant Sci. 2004, 9, 275–280. [Google Scholar] [CrossRef] [PubMed]
- Giménez, C.; Cabrera, R.; Reina, M.; González-Coloma, A. ‘Fungal Endophytes and their Role in Plant Protection’. Curr. Org. Chem. 2007, 11, 707–720. [Google Scholar] [CrossRef]
- Wani, Z.A.; Ashraf, N.; Mohiuddin, T.; Riyaz-Ul-Hassan, S. ‘Plant-endophyte symbiosis, an ecological perspective’. Appl. Microbiol. Biotechnol. 2015, 99, 2955–2965. [Google Scholar] [CrossRef]
- Gouda, S.; Das, G.; Sen, S.K.; Shin, H.S.; Patra, J.K. ‘Endophytes: A treasure house of bioactive compounds of medicinal importance’. Front. Microbiol. 2016, 7, 1–8. [Google Scholar] [CrossRef]
- Nchabeleng, E.K. Determination of biological activity of Celtis africana extracts and its endophytic microflora and mycoflora. Masters Thesis, Department of Biotechnology and Food Technology, University of Johannesburg, Johannesburg, South Africa, 2017. [Google Scholar]
- Guo, B.; Wang, Y.; Sun, X.; Tang, K. Bioactive natural products from endophytes: A review. Appl. Biochem. Microbiol. 2008, 44, 136–142. [Google Scholar] [CrossRef]
- Sadrati, N.; Daoud, H.; Zerroug, A.; Dahamna, S.; Bouharati, S. Screening of antimicrobial and antioxidant secondary metabolites from endophytic fungi isolated from wheat (Triticum durum). J. Plant Prot. Res. 2013, 53, 128–136. [Google Scholar] [CrossRef]
- Stierle, A.; Strobel, G.; Stierle, D. Taxol and taxane production by Taxomyces andreanae, an endophytic fungus of Pacific yew. Science 1993, 260, 214–216. [Google Scholar] [CrossRef] [PubMed]
- Owen, N.L.; Hundley, N. Endophytes the chemical synthesizers inside plants. Sci. Prog. 2004, 87, 79–99. [Google Scholar] [CrossRef] [PubMed]
- Jäger, A.K.; Hutchings, A.; Van Staden, J. ‘Screening of Zulu medicinal plants for prostaglandin-synthesis inhibitors’. J. Ethnopharmacol. 1996, 52, 95–100. [Google Scholar] [CrossRef]
- Lewu, F.B.; Afolayan, A.J. ‘Ethnomedicine in South Africa: The role of weedy species’. Afr. J. Biotechnol. 2009, 8, 929–934. [Google Scholar]
- Watt, J.M.; Breyer-Brandwijk, M.G. The Medicinal and Poisonous Plants of Southern and Eastern Africa, 2nd ed.; Livingstone: London, UK, 1962. [Google Scholar]
- Mabogo, D.E.N. The Ethnobotany of the VhaVenda. Master’s Thesis, Department of Botany, University of Pretoria, Pretoria, South Africa, 1990. [Google Scholar]
- Jasim, B.; Joseph, A.A.; John, C.J.; Mathew, J.; Radhakrishnan, E.K. ‘Isolation and characterization of plant growth promoting endophytic bacteria from the rhizome of Zingiber officinale’. 3 Biotech 2014 2014, 4, 197–204. [Google Scholar] [CrossRef] [PubMed]
- Collins, M.D.; Falsen, E.; Brownlee, K.; Lawson, P.A. ‘Helcococcus sueciensis sp. nov., isolated from a human wound’. Int. J. Syst. Evol. Microbiol. 2004, 54, 1557–1560. [Google Scholar] [CrossRef] [PubMed]
- Yeates, C.; Gillings, M.R.; Davison, A.D.; Altavilla, N.; Veal, D.A. PCR amplification of crude microbial DNA extracted from soil. Lett. Appl. Microbiol. 1997, 25, 303–307. [Google Scholar] [CrossRef] [PubMed]
- Maloney, K.N.; MacMillan, J.B.; Kauffman, C.A.; Jensen, P.R.; DiPasquale, A.G.; Rheingold, A.L.; Fenical, W. Lodopyridone, a structurally unprecedented alkaloid from a marine actinomycete. Org. Lett. 2009, 11, 5422–5424. [Google Scholar] [CrossRef]
- Fomogne-Fodjo, M.C.Y.; Van Vuuren, S.; Ndinteh, D.T.; Krause, R.W.M.; Olivier, D.K. Antibacterial activities of plants from Central Africa used traditionally by the Bakola pygmies for treating respiratory and tuberculosis-related symptoms. J. Ethnopharmacol. 2014, 155, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Uche-Okereafor, N.C. Phytochemical screening, elemental analysis and antibacterial investigation of Rhoicissus tomentosa: A medicinal plant used in South African traditional medicine. Masters. Thesis, Department of Biotechnology and Food Technology, University of Johannesburg, Johannesburg, South Africa, 2016. [Google Scholar]
- Andrews, J.M. Determination of minimum inhibitory concentrations. J. Antimicrob. Chemother. 2001, 48 (Suppl. 1), 5–16. [Google Scholar] [CrossRef] [PubMed]
- McCauley, J.; Zivanovic, A.; Skropeta, D. Bioassays for anticancer activities. Methods Mol. Biol. 2013, 1055, 191–205. [Google Scholar] [PubMed]
- Li, H.; Hu, J.; Wu, S.; Wang, L.; Cao, X.; Zhang, X.; Dai, B.; Cao, M.; Shao, R.; Zhang, R.; et al. Auranofin-mediated inhibition of PI3K/AKT/mTOR axis and anticancer activity in non-small cell lung cancer cells. Oncotarget 2015, 7, 3548–3558. [Google Scholar] [CrossRef] [PubMed]
- Roder, C.; Thomson, M.J. Auranofin: Repurposing an Old Drug for a Golden New Age. Drugs R&D 2015, 15, 13–20. [Google Scholar]
- Dujiff, B.J.; Gianinazzi-Pearson, V.; Lemanceau, P. Involvement of the outer membrane lipopolysaccharides in the endophytic colonization of tomato roots by biocontrol Pseudomonas fluorescens strain WCS417r. New Phytol. 1997, 135, 325–334. [Google Scholar] [CrossRef]
- Hallmann, J.; Ha Umann, A.Q.; Mahaffee, W.F.; Kloepper, J.W. Bacterial endophytes in agricultural crops. Can. J. Microbiol. 1997, 43, 895–914. [Google Scholar]
- Akinsanya, M.A.; Goh, J.K.; Lim, S.P.; Ting, A.S.Y. Diversity, antimicrobial and antioxidant activities of culturable bacterial endophyte communities in Aloe vera. FEMS Microbiol. Lett. 2015, 362, 184. [Google Scholar] [CrossRef]
- Ferreira, A.; Quecine, M.C.; Lacava, P.T.; Oda, S.; Azevedo, J.L.; Araújo, W.L. Diversity of endophytic bacteria from Eucalyptus species seeds and colonization of seedlings by Pantoea agglomerans. FEMS Microbiol. Lett. 2008, 287, 8–14. [Google Scholar] [CrossRef]
- Medrano, E.G.; Bell, A.A. Role of Pantoea agglomerans in opportunistic bacterial seed and boll rot of cotton (Gossypium hirsutum) grown in the field. J. Appl. Microbiol. 2007, 102, 134–143. [Google Scholar] [CrossRef]
- Wright, S.A.I.; Zumoff, C.H.; Schneider, L.; Beer, S.V. Pantoea agglomerans strain EH318 produces two antibiotics that inhibit Erwinia amylovora in vitro. Appl. Environ. Microbiol. 2001, 67, 284–292. [Google Scholar] [CrossRef] [PubMed]
- Procópio, R.E.L. Diversidade bacteriana endofítica de Eucalyptus spp. e avaliação do seu potencial biotecnológico. Ph.D. Thesis, Universidade de São Paulo, São Paulo, Brazil, 2004. [Google Scholar]
- Feng, Y.; Shen, D.; Song, W. Rice endophyte Pantoea agglomerans YS19 promotes host plant growth and affects allocations of host photosynthates. J. Appl. Microbiol. 2006, 100, 938–945. [Google Scholar] [CrossRef] [PubMed]
- Rodloff, A.; Bauer, T.; Ewig, S.; Kujath, P.; Müller, E. Susceptible, Intermediate, and Resistant—The Intensity of Antibiotic Action. Dtsch. Arztebl. Int. 2008, 105, 657–662. [Google Scholar] [CrossRef] [PubMed]
- Tan, R.X.; Zou, W.X. Endophytes: A rich source of functional metabolites. Nat. Prod. Rep. 2001, 18, 448–459. [Google Scholar] [CrossRef] [PubMed]
- Pomini, A.M.; Araújo, W.L.; Marsaioli, A.J. Structural Elucidation and Biological Activity of Acyl-homoserine Lactones from the Phytopathogen Pantoea ananatis Serrano 1928. J. Chem. Ecol. 2006, 32, 1769–1778. [Google Scholar] [CrossRef] [PubMed]
- Kamber, T.; Lansdell, T.A.; Stockwell, V.O.; Ishimaru, C.A.; Smits, T.H.M.; Duffy, B. Characterization of the biosynthetic operon for the antibacterial peptide herbicolin in Pantoea vagans biocontrol strain C9-1 and incidence in Pantoea species. Appl. Environ. Microbiol. 2012, 78, 4412–4419. [Google Scholar] [CrossRef] [PubMed]
- Chah, K.F.; Muko, K.N.; Oboegbulem, S.I. Antimicrobial activity of methanolic extract of Solanum torvum fruit. Fitoterapia 2000, 71, 187–189. [Google Scholar] [CrossRef]
- Aliero, A.A.; Afolayan, A.J. Antimicrobial activity of Solanum tomentosum. Afr. J. Biotechnol. 2006, 5, 369–372. [Google Scholar]
- Koduru, S.; Grierson, D.S.; Afolayan, A.J. Antimicrobial Activity of Solanum aculeastrum. Pharm. Biol. 2006, 44, 283–286. [Google Scholar] [CrossRef]
- Niño, J.; Correa, Y.M.; Mosquera, O.M. Antibacterial, Antifungal, and Cytotoxic Activities of 11 Solanaceae Plants from Colombian Biodiversity. Pharm. Biol. 2006, 44, 14–18. [Google Scholar] [CrossRef]
- Rana, S.; Prakash, V.; Sagar, A. Antibacterial Activity of Solanum xanthocarpum Leaf Extract. Int. J. Curr. Microbiol. Appl. Sci. 2016, 5, 323–328. [Google Scholar] [CrossRef]
- Van Vuuren, S.F. Antimicrobial activity of South African medicinal plants. J. Ethnopharmacol. 2008, 119, 462–472. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Wang, J.; Li, G.; Zheng, Z.; Su, W. Antitumor and antifungal activities in endophytic fungi isolated from pharmaceutical plants Taxus mairei, Cephalataxus fortunei and Torreya grandis. FEMS Immunol. Med. Microbiol. 2001, 31, 163–167. [Google Scholar] [CrossRef]
- Li, J.Y.; Strobel, G.A. Jesterone and hydroxy-jesterone antioomycete cyclohexenenone epoxides from the endophytic fungus Pestalotiopsis jesteri. Phytochemistry 2001, 57, 261–265. [Google Scholar] [CrossRef]
- Salton, M.R.J.; Kim, K.S. Structure. In Medical Microbiology, 4th ed.; Baron, S., Ed.; University of Texas Medical Branch at Galveston: Galveston TX, USA, 1996; Chapter 2; Available online: https://www.ncbi.nlm.nih.gov/books/NBK8477/ (accessed on 8 November 2018).
- Aboul-Enein, A.M.; El-Ela, F.A.; Shalaby, E.; El-Shemy, H. Potent Anticancer and Antioxidant Activities of Active Ingredients Separated from Solanum nigrum and Cassia italica Extracts. J. Arid Land Stud. 2014, 24, 145–152. [Google Scholar]
- Koduru, S.; Grierson, D.S.; van de Venter, M.; Afolayan, A.J. Anticancer Activity of Steroid Alkaloids Isolated from Solanum aculeastrum. Pharm. Biol. 2007, 45, 613–618. [Google Scholar] [CrossRef]
- Chen, Y.T.; Yuan, Q.; Shan, L.T.; Lin, M.A.; Cheng, D.Q.; Li, C.Y. Antitumor activity of bacterial exopolysaccharides from the endophyte Bacillus amyloliquefaciens sp.isolated from Ophiopogon japonicus. Oncol. Lett. 2013, 5, 1787–1792. [Google Scholar] [CrossRef] [PubMed]
- Cui, J.L.; Guo, S.X.; Xiao, P.G. Antitumor and antimicrobial activities of endophytic fungi from medicinal parts of Aquilaria sinensis. J. Zhejiang Univ.-Sci. B (Biomed. & Biotechnol.) 2011, 12, 385–392. [Google Scholar]
- Lakshmi, P.J.; Selvi, K.V. Anticancer potentials of secondary metabolites from endophytes of Barringtonia acutangula and its molecular characterization. Int. J. Curr. Microbiol. App. Sci. 2013, 2, 44–45. [Google Scholar]
- Wu, Y.; Girmay, S.; da Silva, V.M.; Perry, B.; Hu, X.; Tan, G.T. The Role of Endophytic Fungi in the Anticancer Activity of Morinda citrifolia Linn. (Noni). Evid.-Based Complement. Alternative Med. 2015, 2015, 393960:1–393960:8. [Google Scholar]
- Noch, E.K.; Ramakrishna, R.; Magge, R. Challenges in the Treatment of Glioblastoma: Multisystem Mechanisms of Therapeutic Resistance. World Neurosurg. 2018, 116, 505–517. [Google Scholar] [CrossRef] [PubMed]
- Atkins, R.J.; Styllia, S.S.; Kurganovs, N.; Mangiola, S.; Nowell, C.J.; Ware, T.M.; Corcoran, N.M.; Brown, D.V.; Kaye, A.H.; Morokoff, A.; et al. Cell quiescence correlates with enhanced glioblastoma cell invasion and cytotoxic resistance. Exp. Cell Res. 2019, 374, 353–364. [Google Scholar] [CrossRef] [PubMed]
- Shanker, M.; Willcutts, D.; Roth, J.A.; Ramesh, R. Drug resistance in lung cancer. Lung Cancer Targets Ther. 2010, 1, 23–36. [Google Scholar]
- Bradshaw, A.; Wickremsekera, A.; Tan, S.T.; Peng, L.; Davis, P.F.; Itinteang, T. Cancer stem cell hierarchy in glioblastoma multiforme. Front. Surg. 2016, 3, 21. [Google Scholar] [CrossRef]
- Van Schaijik, B.; Wickremesekera, A.C.; Mantamadiotis, T.; Kaye, A.H.; Tan, S.T.; Stylli, S.S.; Itinteang, T. Circulating tumor stem cells and glioblastoma: A review. J. Clin. Neurosci. 2018. [Google Scholar] [CrossRef] [PubMed]
- Tsvetkova, E.; Goss, G.D. Drug resistance and its significance for treatment decisions in non-small-cell lung cancer. Curr. Oncol. 2012, 19, S45–S51. [Google Scholar] [PubMed]
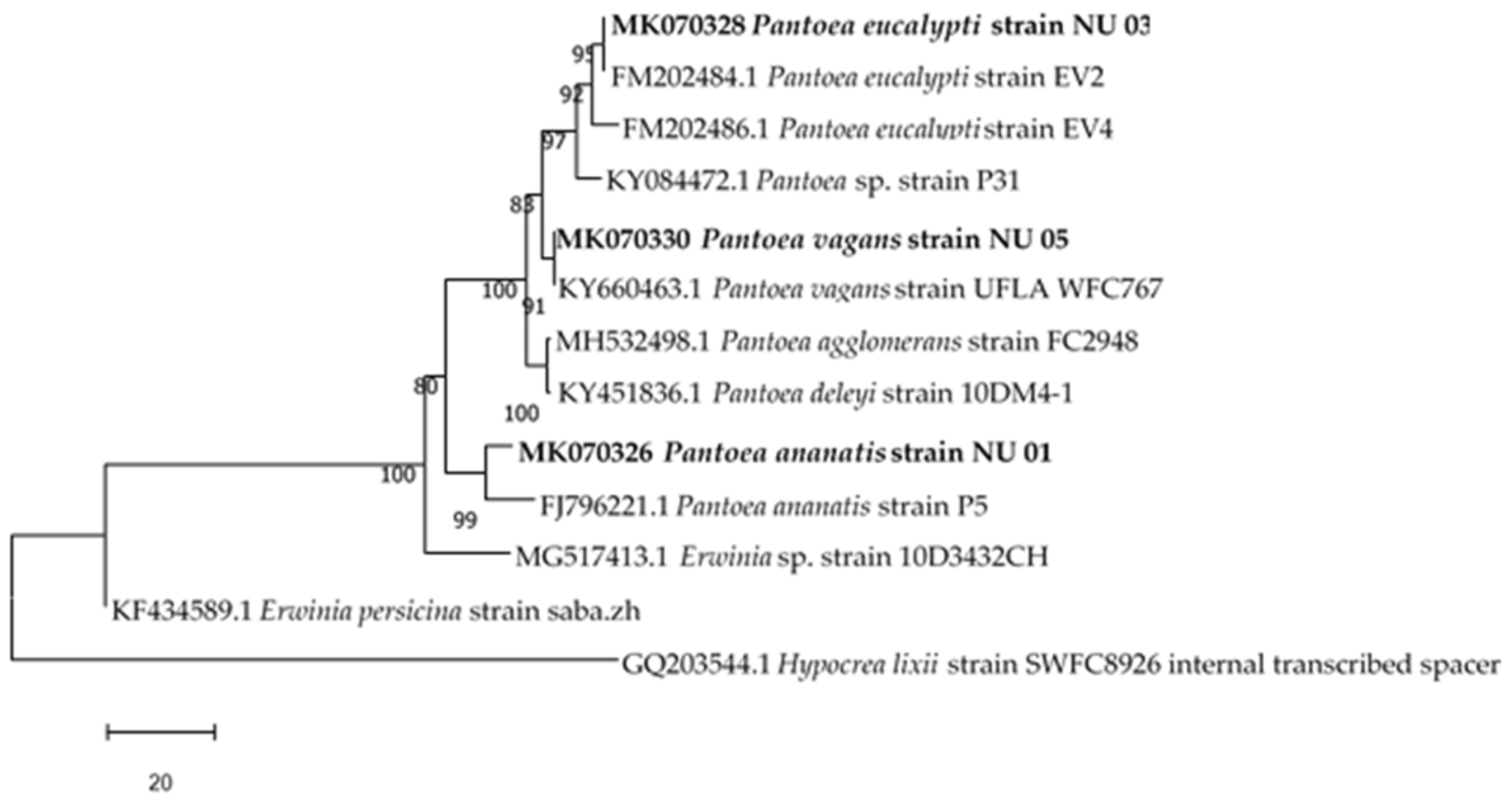
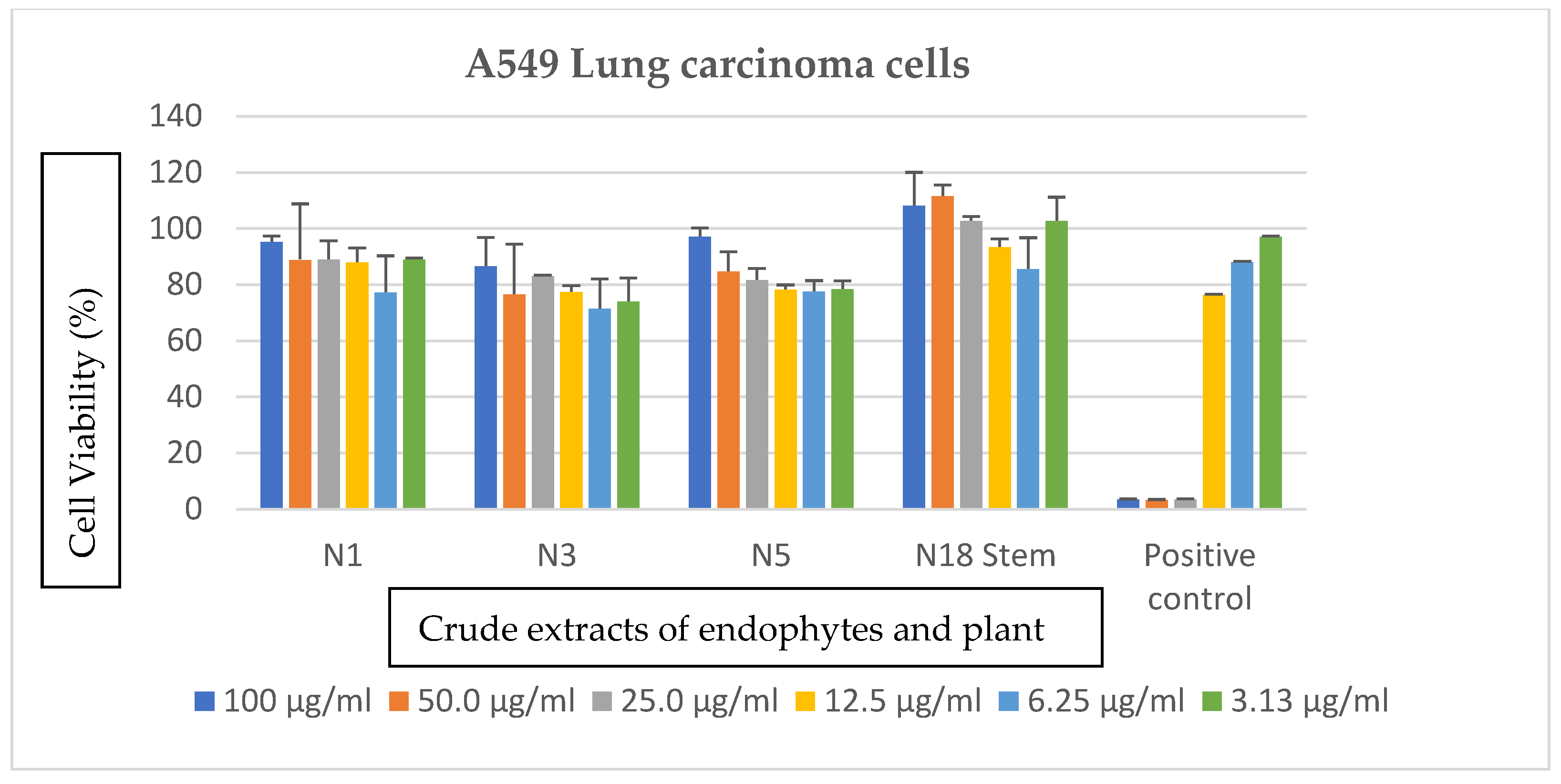
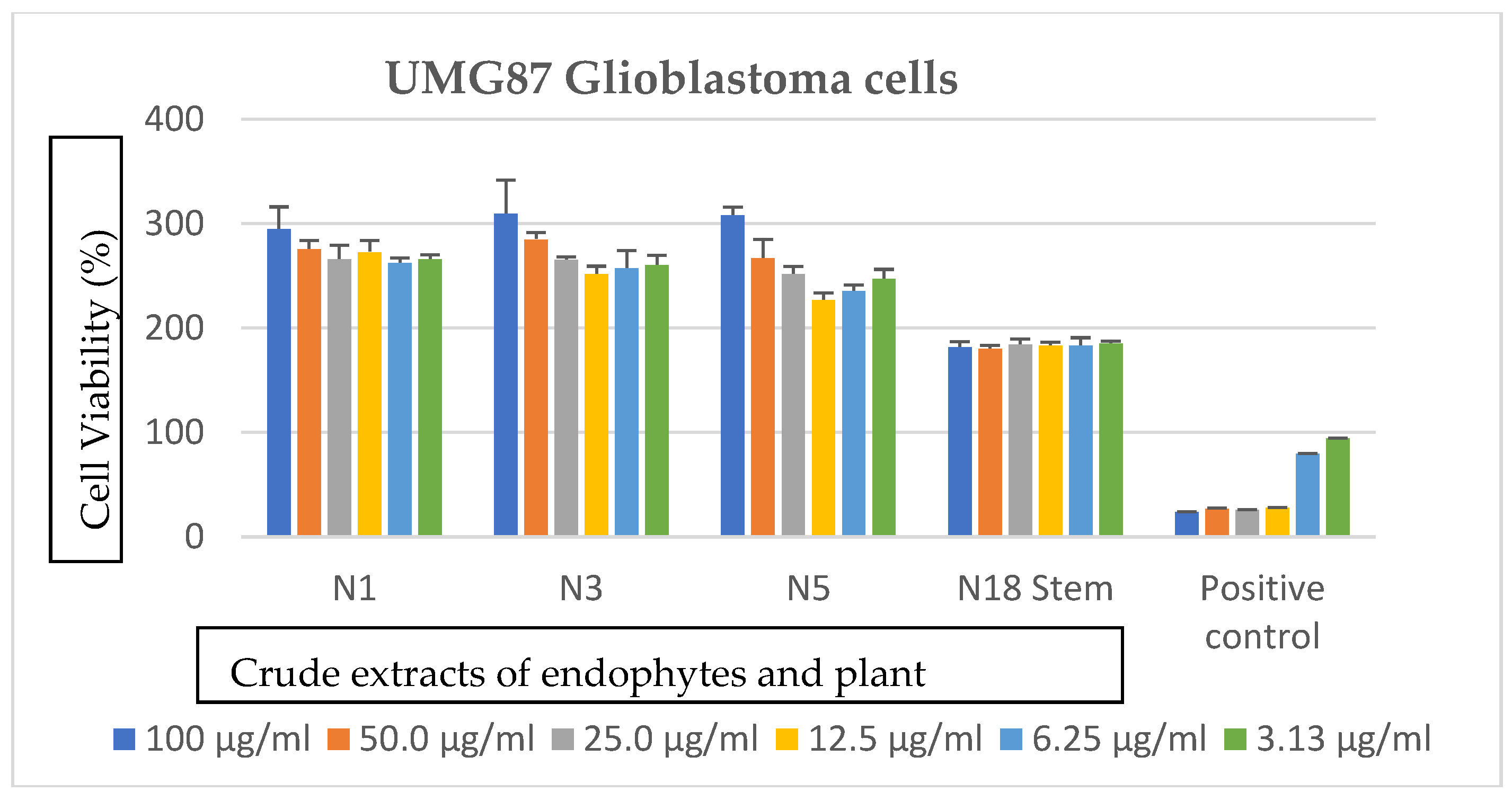
| Bacterial Isolate | Accession Number | Closest Relatives in the National Center for Biotechnology Information (NCBI) | Percentage Similarity | Classification | Macroscopic Identification | Microscopic Identification |
|---|---|---|---|---|---|---|
| NU 01 | MK070326 | Pantoea ananatis strain P5 (FJ796221) | 99 | Pantoea ananatis | Round, white, raised and entire colonies | Gram negative, rods |
| NU 03 | MK070328 | Pantoea eucalypti strain EV2 (FM202484) | 99 | Pantoea eucalypti | Round, Creamy, irregular and raised colonies | Gram negative, rods |
| NU 05 | MK070330 | Pantoea vagans strain UFLA WFC767 (KY660463) | 99 | Pantoea vagans | Round, translucent, convex and entire colonies | Gram negative, rods |
| Test Organism | Gram Reaction of Test Organisms | P. vagans MIC (mg/mL) | P. ananatis MIC (mg/mL) | P. eucalypti MIC (mg/mL) | Plant Part (Stem) MIC (mg/mL) | Positive Control (Streptomycin) MIC (µg/mL) |
|---|---|---|---|---|---|---|
| B. cereus | Positive | 0.5000 | 0.1250 | 0.5000 | 0.1250 | 0.03125 |
| B. subtilis | Positive | 0.1250 | 0.2500 | 0.5000 | 0.1250 | 0.03125 |
| E. aerogenes | Negative | 8.0000 | 4.0000 | 4.0000 | 2.0000 | 0.12500 |
| E. coli | Negative | 1.0000 | 0.5000 | 0.5000 | 0.5000 | 0.12500 |
| K. pneumoniae | Negative | 8.0000 | 2.0000 | 4.0000 | 0.5000 | 0.12500 |
| M. marinum | Positive | 0.5000 | 0.1250 | 0.5000 | 0.5000 | 0.06250 |
| M. smegmatis | Positive | 0.1250 | 0.1250 | 0.1250 | 0.1250 | 0.06250 |
| P. vulgaris | Negative | 4.0000 | 0.5000 | 4.0000 | 0.5000 | 0.12500 |
| P. aeruginosa | Negative | 4.0000 | 1.0000 | 2.0000 | 0.1250 | 0.12500 |
| S. aureus | Positive | 0.1250 | 0.0625 | 0.1250 | 0.0625 | 0.03125 |
| S. epidermidis | Positive | 0.5000 | 0.1250 | 0.1250 | 0.1250 | 0.06250 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Uche-Okereafor, N.; Sebola, T.; Tapfuma, K.; Mekuto, L.; Green, E.; Mavumengwana, V. Antibacterial Activities of Crude Secondary Metabolite Extracts from Pantoea Species Obtained from the Stem of Solanum mauritianum and Their Effects on Two Cancer Cell Lines. Int. J. Environ. Res. Public Health 2019, 16, 602. https://doi.org/10.3390/ijerph16040602
Uche-Okereafor N, Sebola T, Tapfuma K, Mekuto L, Green E, Mavumengwana V. Antibacterial Activities of Crude Secondary Metabolite Extracts from Pantoea Species Obtained from the Stem of Solanum mauritianum and Their Effects on Two Cancer Cell Lines. International Journal of Environmental Research and Public Health. 2019; 16(4):602. https://doi.org/10.3390/ijerph16040602
Chicago/Turabian StyleUche-Okereafor, Nkemdinma, Tendani Sebola, Kudzanai Tapfuma, Lukhanyo Mekuto, Ezekiel Green, and Vuyo Mavumengwana. 2019. "Antibacterial Activities of Crude Secondary Metabolite Extracts from Pantoea Species Obtained from the Stem of Solanum mauritianum and Their Effects on Two Cancer Cell Lines" International Journal of Environmental Research and Public Health 16, no. 4: 602. https://doi.org/10.3390/ijerph16040602
APA StyleUche-Okereafor, N., Sebola, T., Tapfuma, K., Mekuto, L., Green, E., & Mavumengwana, V. (2019). Antibacterial Activities of Crude Secondary Metabolite Extracts from Pantoea Species Obtained from the Stem of Solanum mauritianum and Their Effects on Two Cancer Cell Lines. International Journal of Environmental Research and Public Health, 16(4), 602. https://doi.org/10.3390/ijerph16040602







