Effects of Demographic Variables on Subjective Neurocognitive Complaints Using the Neurocognitive Questionnaire (NCQ) in an Aged Japanese Population
Abstract
1. Introduction
2. Methods
2.1. Participants
2.2. Measurements
2.3. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Jonker, C.; Launer, L.J.; Hooijer, C.; Lindeboom, J. Memory complaints and memory impairment in older individuals. J. Am. Geriatr. Soc. 1996, 44, 44–49. [Google Scholar] [CrossRef] [PubMed]
- Reisberg, B.; Shulman, M.B.; Torossian, C.; Leng, L.; Zhu, W. Outcome over seven years of healthy adults with and without subjective cognitive impairment. Alzheimers Dement. 2010, 6, 11–24. [Google Scholar] [CrossRef] [PubMed]
- Ronnlund, M.; Sundstrom, A.; Adolfsson, R.; Nilsson, L.G. Self-Reported Memory Failures: Associations with Future Dementia in a Population-Based Study with Long-Term Follow-Up. J. Am. Geriatr. Soc. 2015, 63, 1766–1773. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, A.J.; Beaumont, H.; Ferguson, D.; Yadegarfar, M.; Stubbs, B. Risk of dementia and mild cognitive impairment in older people with subjective memory complaints: Meta-analysis. Acta Psychiatr. Scand. 2014, 130, 439–451. [Google Scholar] [CrossRef] [PubMed]
- Rabin, L.A.; Smart, C.M.; Crane, P.K.; Amariglio, R.E.; Berman, L.M.; Boada, M.; Buckley, R.F.; Chételat, G.; Dubois, B.; Ellis, K.A.; et al. Subjective Cognitive Decline in Older Adults: An Overview of Self-Report Measures Used Across 19 International Research Studies. J. Alzheimers Dis. 2015, 48 (Suppl. 1), S63–S86. [Google Scholar] [CrossRef] [PubMed]
- Crumley, J.J.; Stetler, C.A.; Horhota, M. Examining the relationship between subjective and objective memory performance in older adults: A meta-analysis. Psychol. Aging 2014, 29, 250–263. [Google Scholar] [CrossRef] [PubMed]
- Luck, T.; Roehr, S.; Rodriguez, F.S.; Schroeter, M.L.; Witte, A.V.; Hinz, A.; Mehnert, A.; Engel, C.; Loeffler, M.; Thiery, J.; et al. Memory-related subjective cognitive symptoms in the adult population: Prevalence and associated factors—Results of the LIFE-Adult-Study. BMC Psychol. 2018, 6, 23. [Google Scholar] [CrossRef] [PubMed]
- Hocking, D.R.; Reeve, J.; Porter, M.A. Characterising the Profile of Everyday Executive Functioning and Relation to IQ in Adults with Williams Syndrome: Is the BRIEF Adult Version a Valid Rating Scale? PLoS ONE 2015, 10, e0137628. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.I.; Song, D.H.; Shin, D.W.; Kim, J.H.; Lee, Y.S.; Hwang, J.W.; Park, T.W.; Yook, K.H.; Lee, J.I.; Bahn, G.H.; et al. Efficacy and safety of atomoxetine hydrochloride in Korean adults with attention-deficit hyperactivity disorder. Asia-Pac. Psychiatry 2014, 6, 386–396. [Google Scholar] [CrossRef] [PubMed]
- Krull, K.R.; Gioia, G.; Ness, K.K.; Ellenberg, L.; Recklitis, C.; Leisenring, W.; Huang, S.; Stovall, M.; Robison, L.L.; Zeltzer, L. Reliability and validity of the Childhood Cancer Survivor Study Neurocognitive Questionnaire. Cancer 2008, 113, 2188–2197. [Google Scholar] [CrossRef]
- Roth, R.M.; Isquith, P.K.; Gioia, G.A. Behavior Inventory of Executive Function-Adult Version; Psychological Assessment Resources: Lutz, FL, USA, 2005. [Google Scholar]
- Roth, R.M.; Lance, C.E.; Isquith, P.K.; Fischer, A.S.; Giancola, P.R. Confirmatory factor analysis of the Behavior Rating Inventory of Executive Function-Adult version in healthy adults and application to attention-deficit/hyperactivity disorder. Arch. Clin. Neuropsychol. 2013, 28, 425–434. [Google Scholar] [CrossRef] [PubMed]
- Hoogendam, Y.Y.; Hofman, A.; van der Geest, J.N.; van der Lugt, A.; Ikram, M.A. Patterns of cognitive function in aging: The Rotterdam Study. Eur. J. Epidemiol. 2014, 29, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Cargin, J.W.; Collie, A.; Masters, C.; Maruff, P. The nature of cognitive complaints in healthy older adults with and without objective memory decline. J. Clin. Exp. Neuropsychol. 2008, 30, 245–257. [Google Scholar] [CrossRef] [PubMed]
- Jacqmin-Gadda, H.; Fabrigoule, C.; Commenges, D.; Dartigues, J.F. A 5-year longitudinal study of the Mini-Mental State Examination in normal aging. Am. J. Epidemiol. 1997, 145, 498–506. [Google Scholar] [CrossRef] [PubMed]
- Unger, J.M.; van Belle, G.; Heyman, A. Cross-sectional versus longitudinal estimates of cognitive change in nondemented older people: A CERAD study. Consortium to Establish a Registry for Alzheimer’s Disease. J. Am. Geriatr. Soc. 1999, 47, 559–563. [Google Scholar] [CrossRef] [PubMed]
- Donders, J.; Strong, C.A. Latent Structure of the Behavior Rating Inventory of Executive Function-Adult Version (BRIEF-A) After Mild Traumatic Brain Injury. Arch. Clin. Neuropsychol. 2016, 31, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Folstein, M.F.; Folstein, S.E.; McHugh, P.R. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr. Res. 1975, 12, 189–198. [Google Scholar] [CrossRef]
- Mohs, R.C.; Rosen, W.G.; Davis, K.L. The Alzheimer’s disease assessment scale: An instrument for assessing treatment efficacy. Psychopharmacol. Bull. 1983, 19, 448–450. [Google Scholar]
- Jessen, F.; Amariglio, R.E.; van Boxtel, M.; Breteler, M.; Ceccaldi, M.; Chételat, G.; Dubois, B.; Dufouil, C.; Ellis, K.A.; Van Der Flier, W.M.; et al. A conceptual framework for research on subjective cognitive decline in preclinical Alzheimer’s disease. Alzheimers Dement. 2014, 10, 844–852. [Google Scholar] [CrossRef]
- Abdulrab, K.; Heun, R. Subjective Memory Impairment. A review of its definitions indicates the need for a comprehensive set of standardised and validated criteria. Eur. Psychiatry 2008, 23, 321–330. [Google Scholar] [CrossRef]
- Snitz, B.E.; Yu, L.; Crane, P.K.; Chang, C.C.; Hughes, T.F.; Ganguli, M. Subjective cognitive complaints of older adults at the population level: An item response theory analysis. Alzheimer Dis. Assoc. Disord. 2012, 26, 344–351. [Google Scholar] [CrossRef] [PubMed]
- Matthews, F.; Marioni, R.; Brayne, C.; Medical Research Council Cognitive Function and Ageing Study. Examining the influence of gender, education, social class and birth cohort on MMSE tracking over time: A population-based prospective cohort study. BMC Geriatr. 2012, 12, 45. [Google Scholar] [CrossRef] [PubMed]
- Wilson, R.S.; Hebert, L.E.; Scherr, P.A.; Barnes, L.L.; Mendes de Leon, C.F.; Evans, D.A. Educational attainment and cognitive decline in old age. Neurology 2009, 72, 460–465. [Google Scholar] [CrossRef] [PubMed]
- Hayat, S.A.; Luben, R.; Dalzell, N.; Moore, S.; Anuj, S.; Matthews, F.E.; Wareham, N.; Brayne, C.; Khaw, K.T. Cross Sectional Associations between Socio-Demographic Factors and Cognitive Performance in an Older British Population: The European Investigation of Cancer in Norfolk (EPIC-Norfolk) Study. PLoS ONE 2016, 11, e0166779. [Google Scholar] [CrossRef] [PubMed]
- Rexroth, D.F.; Tennstedt, S.L.; Jones, R.N.; Guey, L.T.; Rebok, G.W.; Marsiske, M.M.; Xu, Y.; Unverzagt, F.W. Relationship of demographic and health factors to cognition in older adults in the ACTIVE study. J. Aging Health 2013, 25, 128S–146S. [Google Scholar] [CrossRef] [PubMed]
- Kirton, J.W.; Dotson, V.M. The interactive effects of age, education, and BMI on cognitive functioning. Aging Neuropsychol. Cognit. 2016, 23, 253–262. [Google Scholar] [CrossRef]
- Tucker-Drob, E.M.; Johnson, K.E.; Jones, R.N. The cognitive reserve hypothesis: A longitudinal examination of age-associated declines in reasoning and processing speed. Dev. Psychol. 2009, 45, 431–446. [Google Scholar] [CrossRef]
- Yamada, M.; Landes, R.D.; Mimori, Y.; Nagano, Y.; Sasaki, H. Trajectories of cognitive function in dementia-free subjects: Radiation Effects Research Foundation Adult Health Study. J. Neurol. Sci. 2015, 351, 115–119. [Google Scholar] [CrossRef]
- Robitaille, A.; Piccinin, A.M.; Muniz-Terrera, G.; Hoffman, L.; Johansson, B.; Deeg, D.J.; Aartsen, M.J.; Comijs, H.C.; Hofer, S.M. Longitudinal mediation of processing speed on age-related change in memory and fluid intelligence. Psychol. Aging 2013, 28, 887–901. [Google Scholar] [CrossRef]
| # | NCQ Item | Factor 1 | Factor 2 | Factor 3 | Factor 4 | a BRIEF-A | a CCSS NCQ |
|---|---|---|---|---|---|---|---|
| 24 | Trouble solving math problems | 0.78 | 0.08 | −0.05 | 0.01 | - | |
| 20 | Trouble remembering things | 0.68 | 0.04 | 0.06 | 0.01 | metacognition | memory |
| 22 | Reads slowly | 0.55 | 0.03 | −0.02 | 0.19 | task | |
| 21 | Trouble prioritizing activities | 0.51 | −0.02 | 0.40 | −0.08 | metacognition | task |
| 17 | Trouble with multitasking | 0.47 | −0.03 | 0.24 | 0.23 | metacognition | task |
| 25 | Does not work well under pressure | 0.44 | 0.29 | −0.03 | 0.22 | task | |
| 10 | Different ways to solve a problem | 0.44 | 0.16 | 0.29 | 0.04 | - | |
| 13 | Forgot what doing in middle of things | 0.42 | 0.05 | 0.28 | −0.02 | metacognition | memory |
| 5 | Forgets instructions easily | 0.40 | 0.09 | 0.28 | 0.06 | metacognition | memory |
| 1 | Gets upset easily | 0.01 | 0.74 | −0.12 | 0.12 | emotional | |
| 9 | Mood changes frequently | 0.02 | 0.72 | 0.24 | −0.07 | behavioral | emotional |
| 8 | Gets frustrated easily | 0.16 | 0.70 | 0.02 | −0.10 | emotional | |
| 16 | Easily overwhelmed | 0.16 | 0.50 | 0.01 | 0.19 | task | |
| 11 | Impulsive | −0.10 | 0.46 | 0.36 | 0.02 | behavioral | - |
| 6 | Problems completing my work | 0.00 | 0.04 | 0.67 | 0.21 | metacognition | task |
| 4 | Disorganized | 0.03 | 0.03 | 0.48 | 0.27 | metacognition | organization |
| 14 | Problems with self-motivation | 0.35 | −0.01 | 0.46 | 0.11 | behavioral | task |
| 12 | Trouble finding things in bedroom | 0.29 | 0.04 | 0.40 | 0.15 | metacognition | organization |
| 23 | Slower than others | 0.19 | −0.07 | 0.13 | 0.69 | task | |
| 2 | Takes longer to complete work | −0.06 | 0.06 | 0.27 | 0.62 | task | |
| 3 | Does not think of consequences | 0.12 | 0.22 | 0.31 | 0.09 | behavioral | - |
| 7 | Difficulty recalling things learned before | 0.35 | 0.18 | 0.12 | 0.04 | memory | |
| 15 | Underachiever | 0.36 | 0.14 | 0.09 | 0.26 | - | |
| 18 | Blurts things out | 0.32 | 0.27 | 0.08 | −0.08 | - | |
| 19 | Desk/workspace a mess | 0.11 | 0.04 | 0.29 | 0.23 | organization | |
| Mean factor score | 1.34 | 1.26 | 1.25 | 1.38 | |||
| Standard deviation of factor score | 0.42 | 0.38 | 0.42 | 0.55 | |||
| Cronbach’s | 0.911 | 0.851 | 0.842 | 0.817 |
| Score | Correlations with | ||
|---|---|---|---|
| Factor 2 | Factor 3 | Factor 4 | |
| Factor 1 | 0.708 | 0.798 | 0.707 |
| Factor 2 | -- | 0.641 | 0.533 |
| Factor 3 | -- | -- | 0.728 |
| Administration | NCQ | Factor 1 | Factor 2 | Factor 3 | Factor 4 |
|---|---|---|---|---|---|
| Total | 555 | 614 | 612 | 626 | 630 |
| Only 1st administration | 189 b | 177 | 168 | 165 | 151 |
| Only 2nd administration | 52 | 29 | 18 | 23 | 13 |
| Both administrations | 314 a,b | 408 | 426 | 438 | 466 |
| Sample | N | City (%) | Sex (%) | a Education (%) | b Age | ||||
|---|---|---|---|---|---|---|---|---|---|
| Hiroshima | Nagasaki | Female | Primary School | High School | Junior University | University | Mean ± SD | ||
| CC | 314 | 56.0 | 44.0 | 52.9 | 20.1 | 60.4 | 6.8 | 12.7 | 76.2 ± 5.8 |
| EFA | 503 | 53.7 | 46.3 | 58.7 | 22.8 | 60.5 | 6.9 | 9.8 | 77.3 ± 5.8 |
| AA | 649 | 55.2 | 44.8 | 61.6 | 23.7 | 61.0 | 7.3 | 8.0 | 77.6 ± 5.9 |
| a Score | b Age-Linear | b Age-Quadratic | c Male | d Education | ||
|---|---|---|---|---|---|---|
| HS vs. P | JU vs. P | U vs. P | ||||
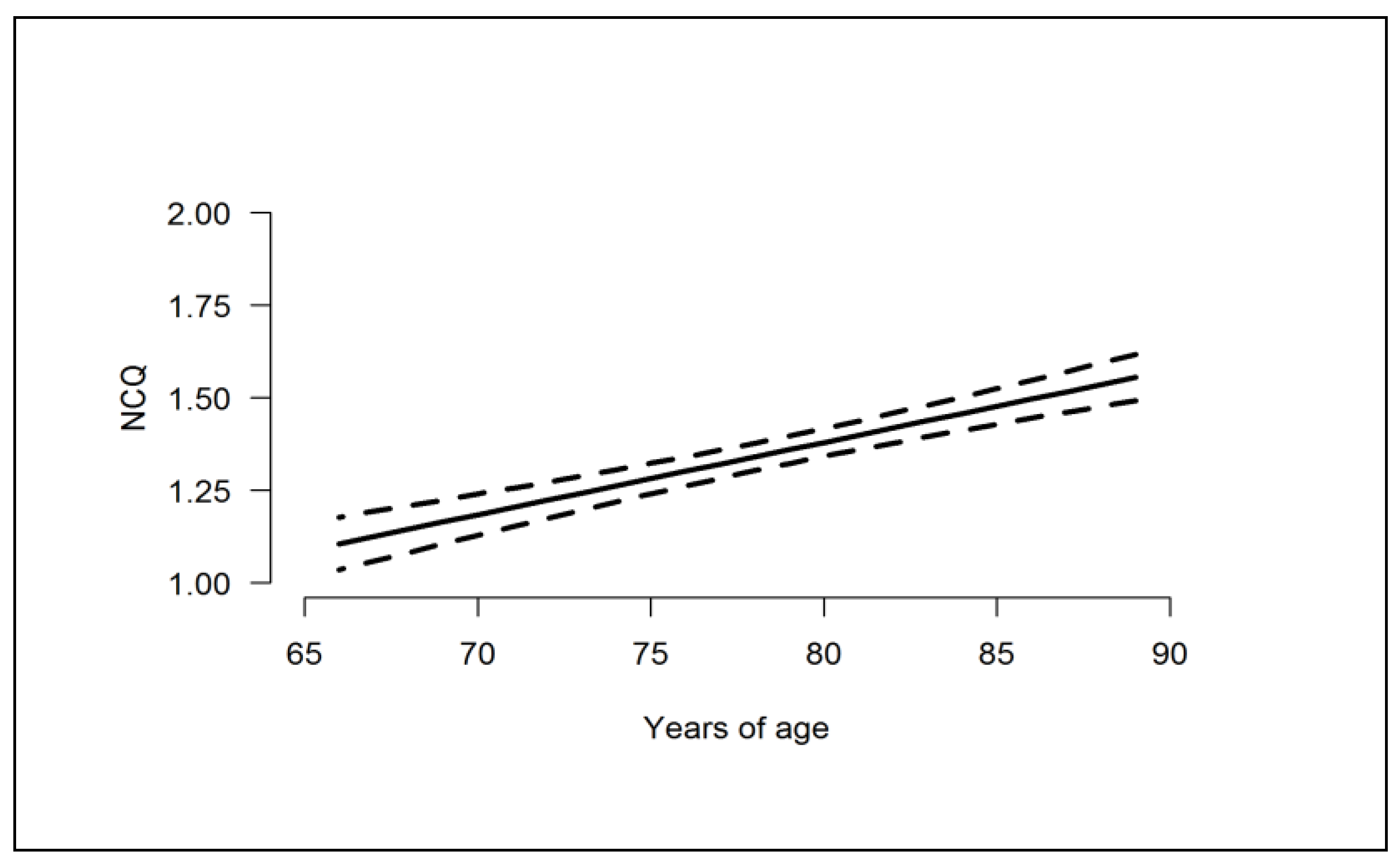
| NCQ | 0.195; (0.147, 0.243) | -- | -- | −0.065; (−0.138, 0.008) | −0.152; (−0.280, 0.023) | −0.111; (−0.226, 0.005) |
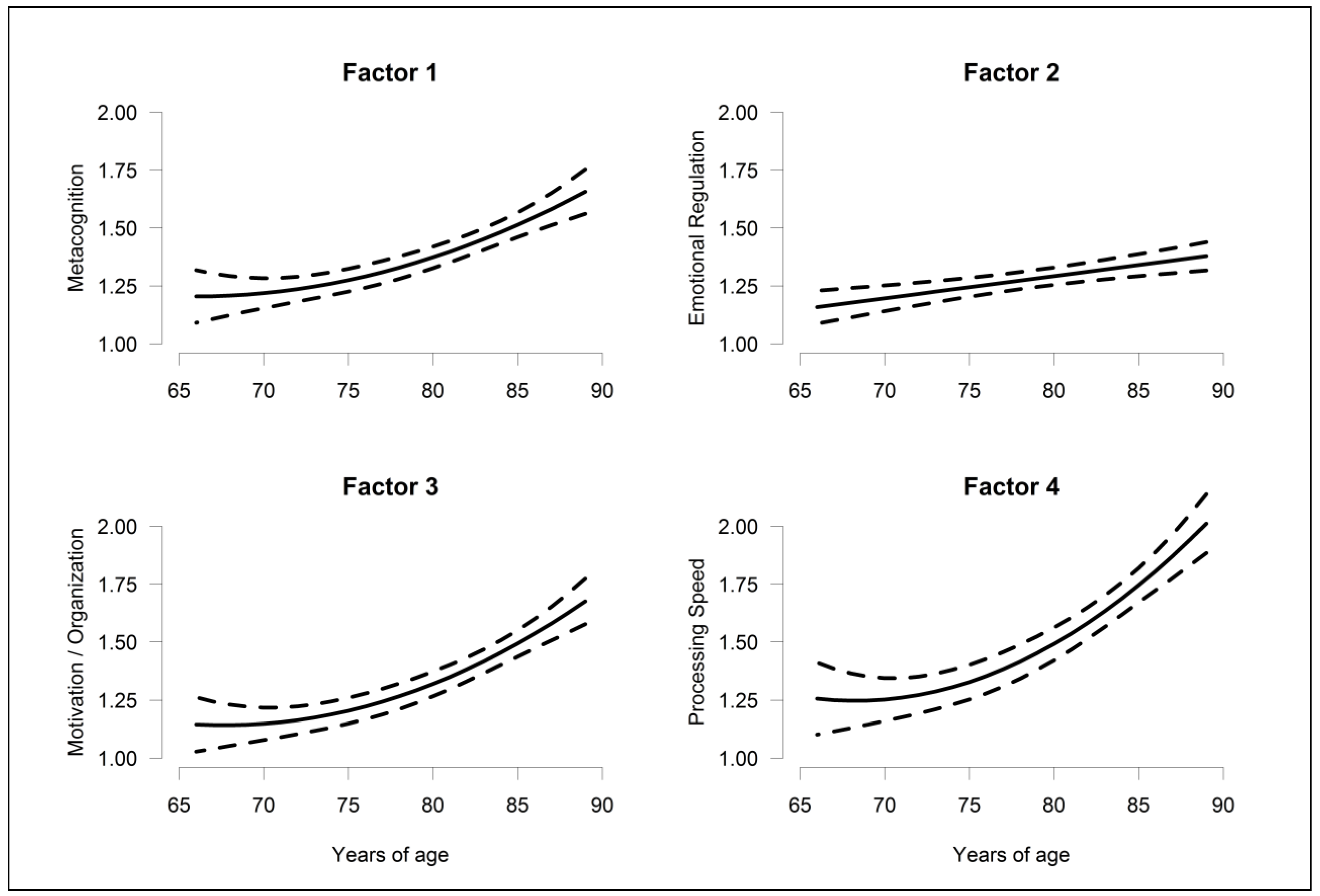
| Factor 1 | 0.199; (0.146, 0.251) | 0.085; (0.008, 0.161) | -- | −0.104; (−0.183, −0.026) | −0.180; (−0.318, −0.042) | −0.176; (−0.305, −0.048) |
| Factor 2 | 0.096; (0.048, 0.143) | -- | -- | −0.092; (−0.164, −0.020) | −0.208; (−0.330, −0.085) | −0.149; (−0.266, −0.033) |
| Factor 3 | 0.233; (0.180, 0.286) | 0.118; (0.038, 0.198) | −0.062; (−0.130, 0.006) | −0.088; (−0.165, −0.010) | −0.130; (−0.265, 0.005) | −0.106; (−0.238, 0.026) |
| Factor 4 | 0.332; (0.262, 0.402) | 0.178; (0.074, 0.282) | −0.089; (−0.181, 0.003) | −0.118; (−0.220, −0.015) | −0.067; (−0.248, 0.114) | −0.189; (−0.364, −0.013) |
| Score | Correlation | Differences in Scores by Age Group | |||||
|---|---|---|---|---|---|---|---|
| All CC | [65, 70) | [70, 75) | [75, 80) | [80, 85) | [85, 90) | ||
| NCQ | 0.81 | 0.05; (0.02, 0.07) | 0.04; (−0.02, 0.09) | 0.05; (0.01, 0.10) | −0.01; (−0.06, 0.05) | 0.07; (0.02, 0.12) | 0.08; (−0.02, 0.18) |
| Factor 1 | 0.77 | 0.05; (0.02, 0.08) | 0.03; (−0.03, 0.09) | 0.07; (0.02, 0.13) | −0.01; (−0.07, 0.05) | 0.08; (0.01, 0.15) | 0.07; (−0.07, 0.21) |
| Factor 2 | 0.73 | 0.02; (−0.01, 0.05) | 0.06; (−0.00, 0.12) | 0.04; (−0.02, 0.10) | −0.05; (−0.11, 0.02) | 0.01; (−0.05, 0.07) | 0.05; (−0.04, 0.14) |
| Factor 3 | 0.73 | 0.06; (0.02, 0.09) | 0.05; (−0.01, 0.11) | 0.06; (0.01, 0.11) | 0.00; (−0.07, 0.08) | 0.07; (0.00, 0.13) | 0.18; (−0.04, 0.39) |
| Factor 4 | 0.70 | 0.11; (0.06, 0.15) | 0.01; (−0.07, 0.09) | 0.06; (−0.02, 0.13) | 0.07; (−0.06, 0.19) | 0.20; (0.11, 0.29) | 0.25; (0.03, 0.47) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yamada, M.; Landes, R.D.; Hida, A.; Ishihara, K.; Krull, K.R. Effects of Demographic Variables on Subjective Neurocognitive Complaints Using the Neurocognitive Questionnaire (NCQ) in an Aged Japanese Population. Int. J. Environ. Res. Public Health 2019, 16, 421. https://doi.org/10.3390/ijerph16030421
Yamada M, Landes RD, Hida A, Ishihara K, Krull KR. Effects of Demographic Variables on Subjective Neurocognitive Complaints Using the Neurocognitive Questionnaire (NCQ) in an Aged Japanese Population. International Journal of Environmental Research and Public Health. 2019; 16(3):421. https://doi.org/10.3390/ijerph16030421
Chicago/Turabian StyleYamada, Michiko, Reid D. Landes, Ayumi Hida, Kayoko Ishihara, and Kevin R. Krull. 2019. "Effects of Demographic Variables on Subjective Neurocognitive Complaints Using the Neurocognitive Questionnaire (NCQ) in an Aged Japanese Population" International Journal of Environmental Research and Public Health 16, no. 3: 421. https://doi.org/10.3390/ijerph16030421
APA StyleYamada, M., Landes, R. D., Hida, A., Ishihara, K., & Krull, K. R. (2019). Effects of Demographic Variables on Subjective Neurocognitive Complaints Using the Neurocognitive Questionnaire (NCQ) in an Aged Japanese Population. International Journal of Environmental Research and Public Health, 16(3), 421. https://doi.org/10.3390/ijerph16030421






