Distribution and Release Characteristics of Phosphorus in a Reservoir in Southwest China
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Site
2.2. Sample Collection
2.3. Analysis of Overlying Water and Surface Sediments
2.4. Laboratory Experiments of the Rate and Flux of P
2.5. Statistical Analysis
3. Results
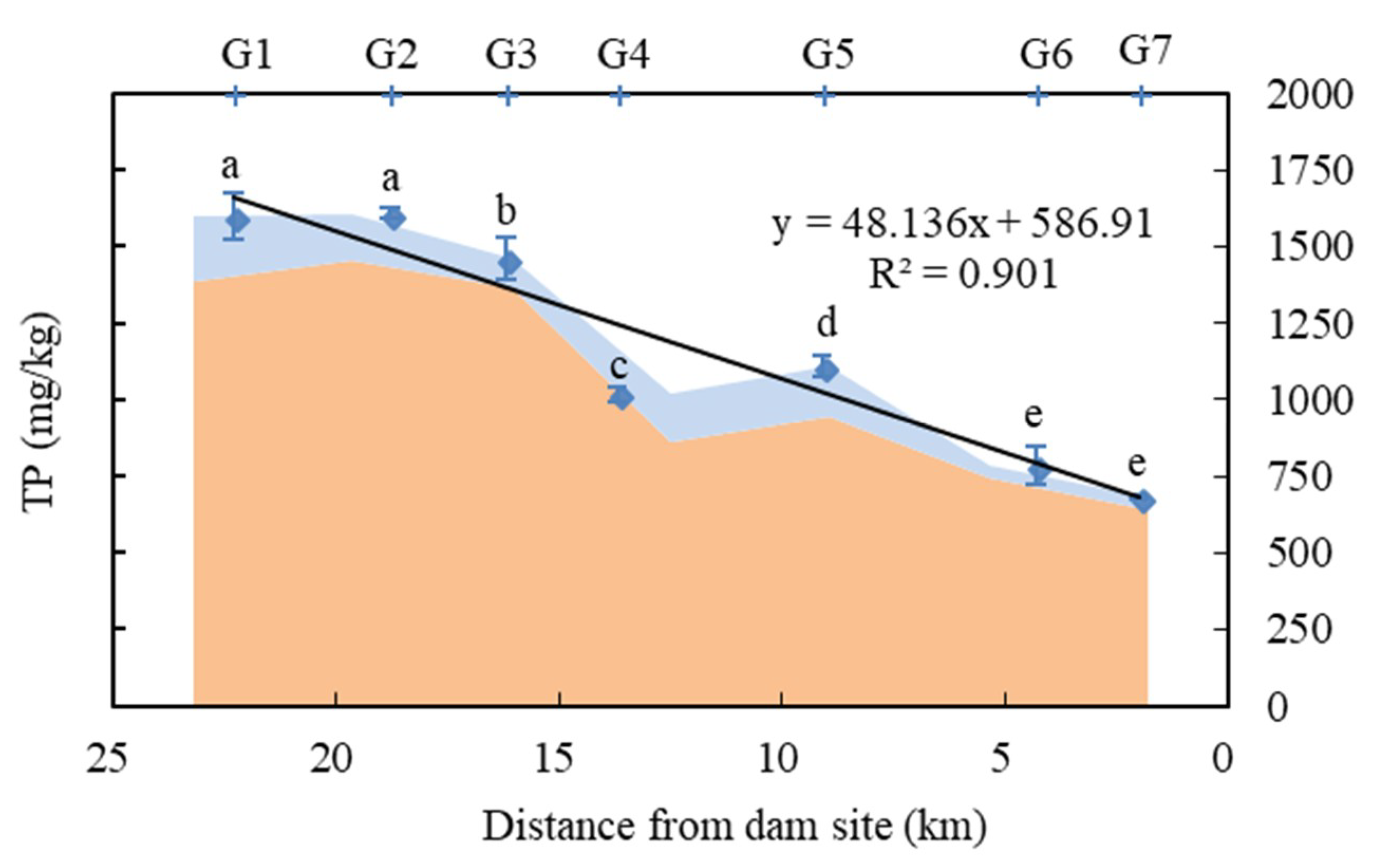
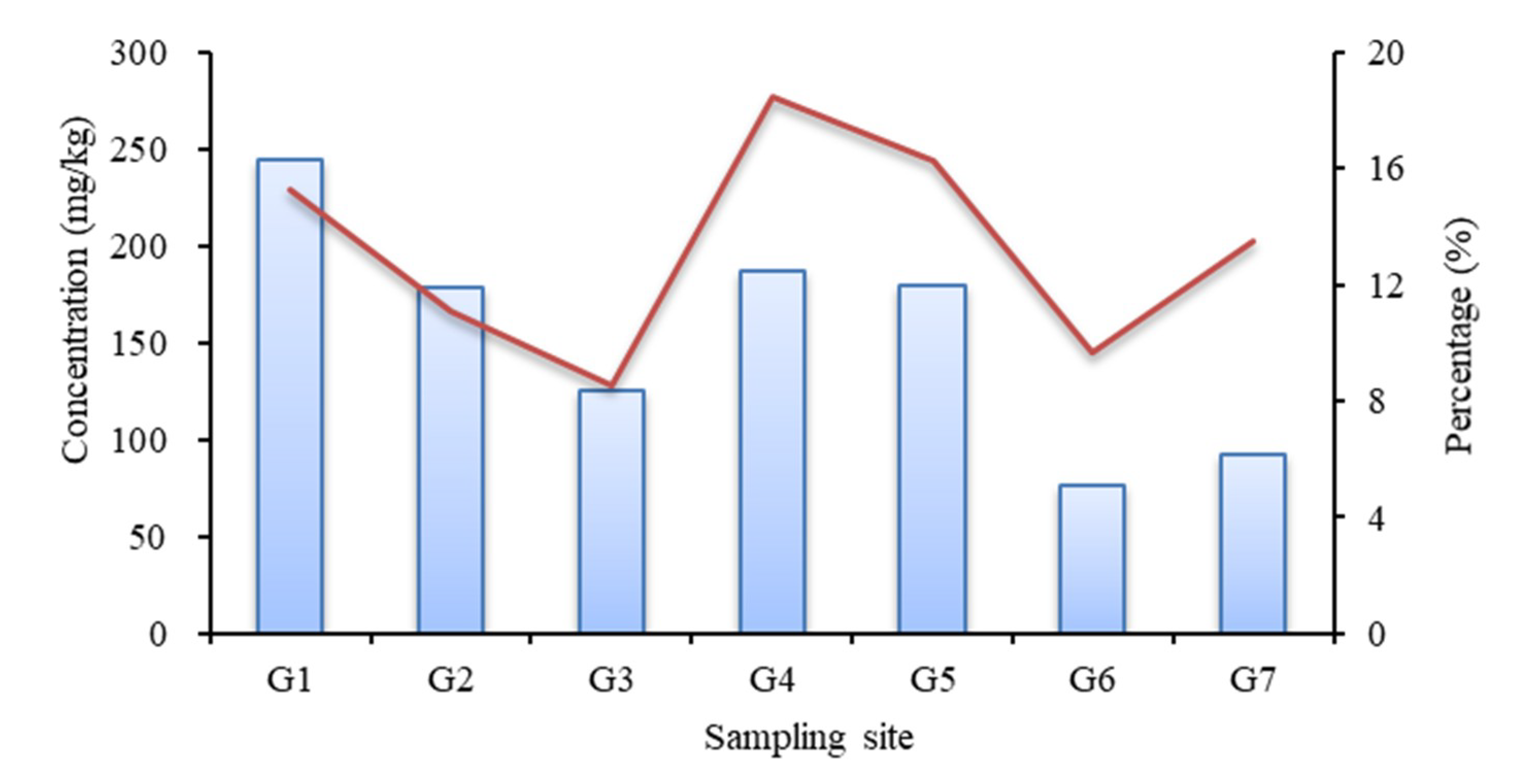
3.1. Spatial-Temporal Distribution of Phosphorus in Sediments
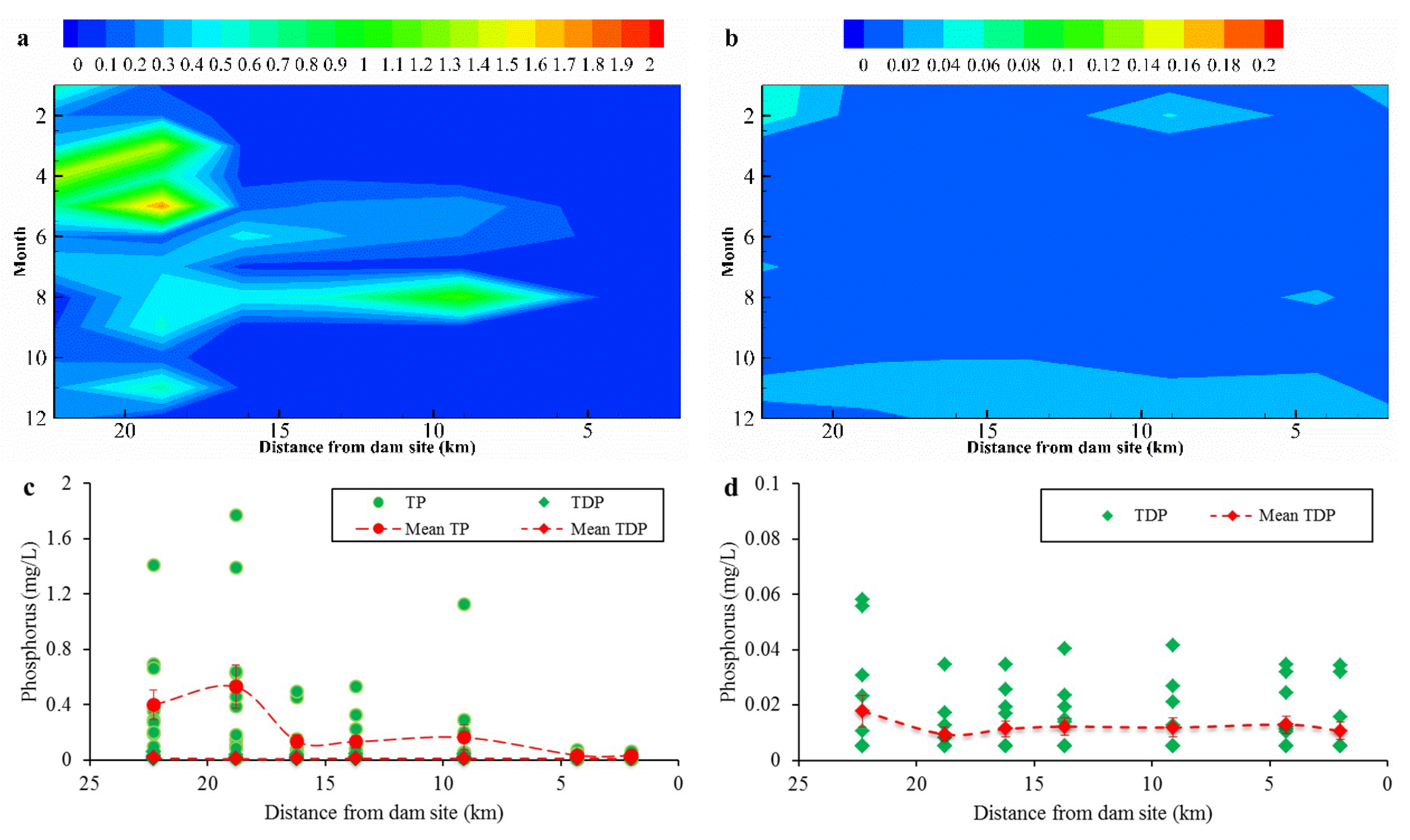
3.2. Spatial-Temporal Distribution of Phosphorus in Overlying Water
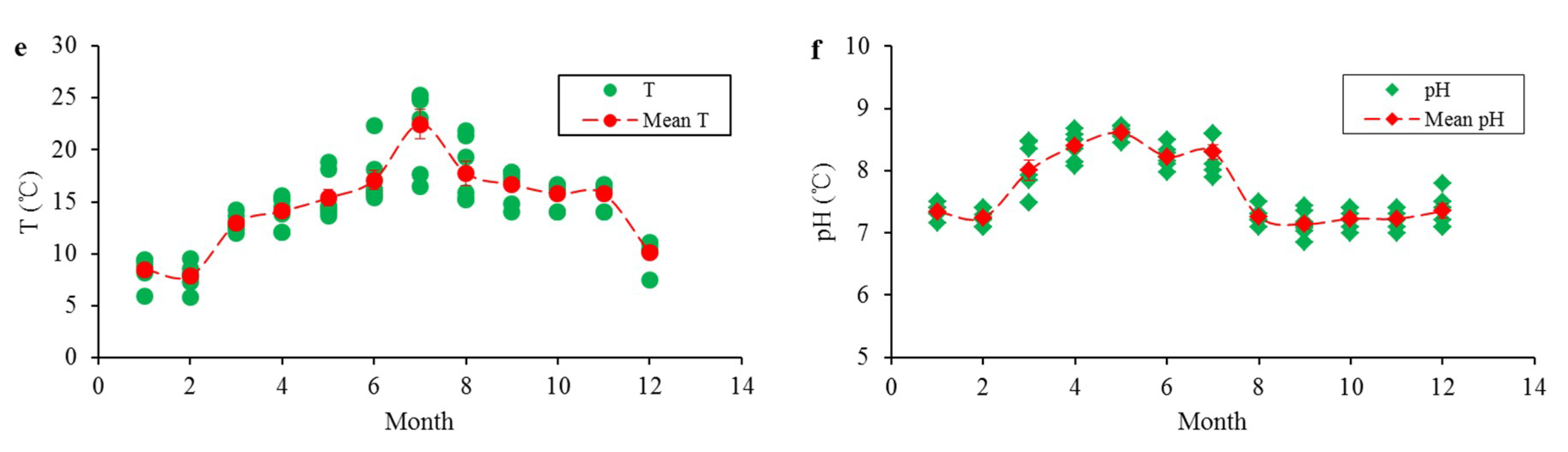
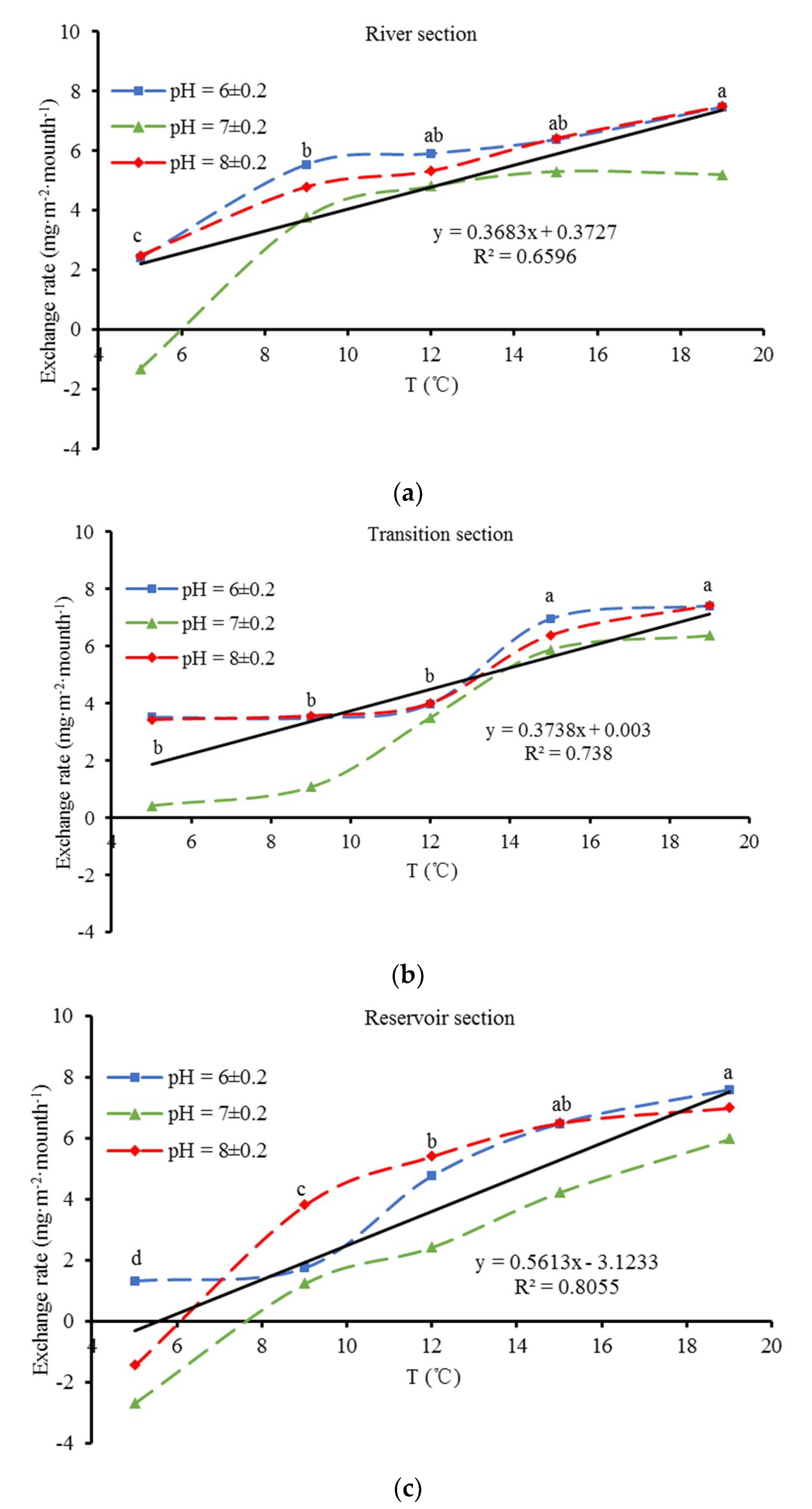
3.3. Characteristics of Phosphorus Exchange at the Sediment-Water Interface
4. Discussion
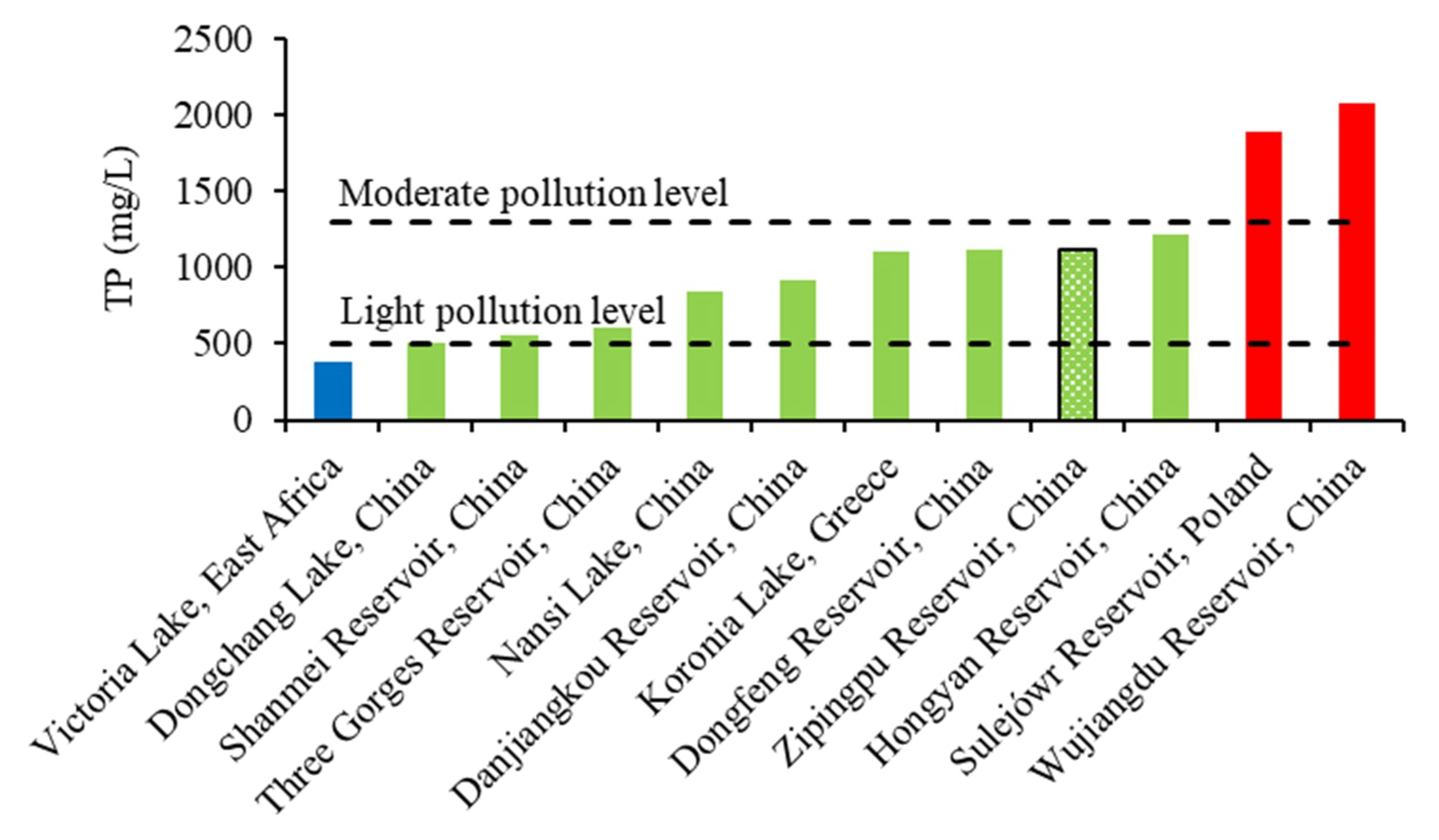
4.1. Phosphorus in Sediments and Overlying Water
4.2. Exchange Characteristics of Phosphorus at the Sediment-Water Interface
5. Conclusions
- (1)
- The TP content of surface sediments in reservoirs decreased from the tail to the front of the dam, overall. The IP was the main form of phosphorus in surface sediments.
- (2)
- PP was the main form of phosphorus in the overlying water of the reservoirs and was strongly affected by hydrodynamic conditions. The TDP content in the overlying water was relatively low.
- (3)
- Both water temperature and pH significantly affected the release rate of the sediment, but the influence of water temperature was more significant. Acidic and alkaline conditions were conducive to the release of phosphorus from sediments, while a neutral environment was not. The release rate significantly increased with increasing water temperature. Sediment exhibited absorption characteristics when the water temperature was extremely low and releasing characteristics at a high temperature.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- López-Tarazón, J.A.; López, P.; Lobera, G.; Batalla, R.J. Suspended sediment, carbon and nitrogen transport in a regulated Pyrenean river. Sci. Total Environ. 2016, 540, 133–143. [Google Scholar] [CrossRef] [PubMed]
- Maavara, T.; Parsons, C.T.; Ridenour, C.; Stojanovic, S.; Dürr, H.H.; Powley, H.R.; Van Cappellen, P. Global phosphorus retention by river damming. Proc. Natl. Acad. Sci. USA 2015, 112, 15603–15608. [Google Scholar] [CrossRef] [PubMed]
- Lau, S.S.S.; Lane, S.N. Biological and chemical factors influencing shallow lake eutrophication: A long-term study. Sci. Total Environ. 2002, 288, 167–181. [Google Scholar] [CrossRef]
- Conley, D.J.; Paerl, H.W.; Howarth, R.W.; Boesch, D.F.; Seitzinger, S.P.; Havens, K.E.; Likens, G.E. Controlling eutrophication: Nitrogen and phosphorus. Science 2009, 323, 1014–1015. [Google Scholar] [CrossRef] [PubMed]
- Vink, S.; Chambers, R.M.; Smith, S.V. Distribution of phosphorus in sediments from Tomales Bay, California. Mar. Geol. 1997, 139, 157–179. [Google Scholar] [CrossRef]
- Dong, L.; Yang, Z.; Liu, X. Phosphorus fractions, sorption characteristics, and its release in the sediments of Baiyangdian Lake, China. Environ. Monit. Assess. 2011, 179, 335–345. [Google Scholar] [CrossRef] [PubMed]
- Ruttenberg, K.C. Development of a sequential extraction method for different forms of phosphorus in marine sediments. Limnol. Oceanogr. 1992, 37, 1460–1482. [Google Scholar] [CrossRef]
- Filippelli, G.M.; Delaney, M.L. Phosphorus geochemistry of equatorial Pacific sediments. Geochim. Cosmochim. Acta 1996, 60, 1479–1495. [Google Scholar] [CrossRef]
- Richerson, P.J.; Suchanek, T.H.; Why, S.J. The Causes and Control of Algal Blooms in Clear Lake: Clean Lakes Diagnostic/Feasibility Study for Clear Lake, California; University of California, Division of Environmental Studies: Santa Cruz, CA, USA, 1994. [Google Scholar]
- Li, S. Internal phosphorus of Chagan Lake release and impact on eutrophication in low temperature period. Ph.D. Thesis, Jilin University, Changchun, China, 2013. [Google Scholar]
- Kim, L.H.; Choi, E.; Gil, K.I.; Stenstrom, M.K. Phosphorus release rates from sediments and pollutant characteristics in Han River, Seoul, Korea. Sci. Total Environ. 2004, 321, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Liang, T. Distribution characteristics of phosphorus in the sediments and overlying water of Poyang Lake. PLoS ONE 2015, 10, e0125859. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Fang, H.; He, G.; Jiang, H.; Wang, C. Effects of internal loading on phosphorus distribution in the Taihu Lake driven by wind waves and lake currents. Environ. Pollut. 2016, 219, 760–773. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Fang, F.; Guo, J.; Chen, Y.; Li, Z.; Guo, S. Phosphorus fractions and phosphate sorption-release characteristics relevant to the soil composition of water-level-fluctuating zone of Three Gorges Reservoir. Ecol. Eng. 2012, 40, 153–159. [Google Scholar] [CrossRef]
- Jin, X.; He, Y.; Kirumba, G.; Hassan, Y.; Li, J. Phosphorus fractions and phosphate sorption-release characteristics of the sediment in the Yangtze River estuary reservoir. Ecol. Eng. 2013, 55, 62–66. [Google Scholar] [CrossRef]
- Huang, H.; Yan, Z. Present situation and future prospect of hydropower in China. Renew. Sustain. Energy Rev. 2009, 13, 1652–1656. [Google Scholar] [CrossRef]
- Fang, Y.; Deng, W. The critical scale and section management of cascade hydropower exploitation in Southwestern China. Energy 2011, 36, 5944–5953. [Google Scholar] [CrossRef]
- Tang, X.J.; Zhou, J. A future role for cascade hydropower in the electricity system of China. Energy Policy 2012, 51, 358–363. [Google Scholar] [CrossRef]
- Yong, X.D.; Tuo, Y.C.; Li, R.; Li, J.; Li, K.F.; Deng, Y.; Long, Q.J.; Teng, H. Deep water sampling system. Chinese Patent CN102128735A, 2010. [Google Scholar]
- Jin, X.; Tu, Q. Investigation Specification of Lake Eutrophication; China Environmental Science Press: Beijing, China, 1990. [Google Scholar]
- Giblin, A.E.; Hopkinson, C.S.; Tucker, J. Benthic metabolism and nutrient cycling in Boston Harbor, Massachusetts. Estuaries 1997, 20, 346–364. [Google Scholar] [CrossRef]
- Xu, Q.; Liu, X.; Wang, H.; Liu, L.; Liu, J. Study on the endogenous phosphorus load of sediments in Miyun reservoir. Sci. China Ser. D (Earth Sci.) 2005, 48, 333–340. [Google Scholar]
- Jin, X.; Wang, S.; Pang, Y.; Wu, F.C. Phosphorus fractions and the effect of pH on the phosphorus release of the sediments from different trophic areas in Taihu Lake, China. Environ. Pollut. 2006, 139, 288–295. [Google Scholar] [CrossRef]
- Peng, J.F.; Wang, B.Z.; Song, Y.H.; Yuan, P.; Liu, Z. Adsorption and release of phosphorus in the surface sediment of a wastewater stabilization pond. Ecol. Eng. 2007, 31, 92–97. [Google Scholar] [CrossRef]
- Fan, C.X.; Zhang, L.; Bao, X.M.; You, B.S.; Zhong, J.C.; Wang, J.J.; Ding, S.M. Migration mechanism of biogenic elements and their quantification on the sediment-water Interface of Lake Taihu: II. Chemical thermodynamic mechanism of phosphorus release and its source-sink transiton. J. Lake Sci. 2006, 18, 207–217. (In Chinese) [Google Scholar]
- Wang, Y.; Shen, Z.Y.; Hu, L.J.; Niu, J.F. Adsorption and release of phosphorus from sediments from the main branches of the Three-Gorges Reservoir. Acta Sci. Circumst. 2008, 28, 1654–1661. (In Chinese) [Google Scholar]
- Yin, D.Q.; Qin, Q.R.; Yan, H. Effects of environmental factors on the release of phosphorus from sediments of Wuli Lake. J. Lake Sci. 1994, 6, 240–244. (In Chinese) [Google Scholar]
- Andersen, F.Ø.; Ring, P. Comparison of phosphorus release from littoral and profundal sediments in a shallow, eutrophic lake. In Shallow Lakes’ 98; Springer: Dordrecht, The Netherlands, 1999; pp. 175–183. [Google Scholar]
- Ribeiro, D.C.; Martins, G.; Nogueira, R.; Cruz, J.V.; Brito, A.G. Phosphorus fractionation in volcanic lake sediments (Azores–Portugal). Chemosphere 2008, 70, 1256–1263. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.F. Study on Phosphorus Forms and Characteristics of Phosphorus Adsorption and Release of Sediments in Dongchang Lake. Master’s Thesis, Ocean University of China, Qingdao, China, 2013. [Google Scholar]
- Zhou, Z.M.; Shen, C.H.; Zhao, Z.L.; Tu, F. Distribution and pollution of total phosphorus and total nitrogen in surface sediments from Shanmei reservoir valley. J. Fuzhou Univ. 2011, 39, 608–612. (In Chinese) [Google Scholar]
- Song, W.C. Polluted Characteristics Sediments and Phosphate Adsorption and Release Characteristics at the Sediment-Water Inerface in Nansi Lake, China, and Its Main Estuaries. Ph.D. Thesis, Shandong University, Changchun, China, 2008. [Google Scholar]
- Wang, W.W.; Wang, S.H.; Zhao, L. Identification of inorganic and organic species of phosphorus and its bio-availability by aequential extraction method in surface sediments of Danjiangkou Reservoir. China Environ. Sci. 2016, 36, 808–818. (In Chinese) [Google Scholar]
- Kaiserli, A.; Voutsa, D.; Samara, C. Phosphorus fractionation in lake sediments–Lakes Volvi and Koronia, N. Greece. Chemosphere 2002, 46, 1147–1155. [Google Scholar] [CrossRef]
- Yin, R.; Wang, F.S.; Mei, H.Y.; Yao, C.C.; Guo, M.Y. Distribution of phosphorus forms in the sediments of cascade reservoirs with different trophic states in Wujiang catchment. Chin. J. Ecol. 2010, 1, 91–97. (In Chinese) [Google Scholar]
- Frankowski, L.; Bolałek, J.; Szostek, A. Phosphorus in bottom sediments of pomeranian bay (Southern Baltic-Poland). Estuar. Coast. Shelf Sci. 2002, 54, 1027–1038. [Google Scholar] [CrossRef]
- Owens, P.N.; Walling, D.E. The phosphorus content of fluvial sediment in rural and industrialized river basins. Water Res. 2002, 36, 685–701. [Google Scholar] [CrossRef]
- Wang, S.; Jin, X.; Zhao, H.; Wu, F. Phosphorus fractions and its release in the sediments from the shallow lakes in the middle and lower reaches of Yangtze River area in China. Colloids Surf. A Physicochem. Eng. Aspects 2006, 273, 109–116. [Google Scholar] [CrossRef]
- Dai, J.C.; Song, J.M.; Li, X.G.; Yuan, H.M.; Zheng, G.X.; Li, N. Sediment record of phosphorus and the primary study of its bioavailability in Jiaozhou Bay sediment. Environ. Sci. 2007, 28, 929–936. (In Chinese) [Google Scholar]
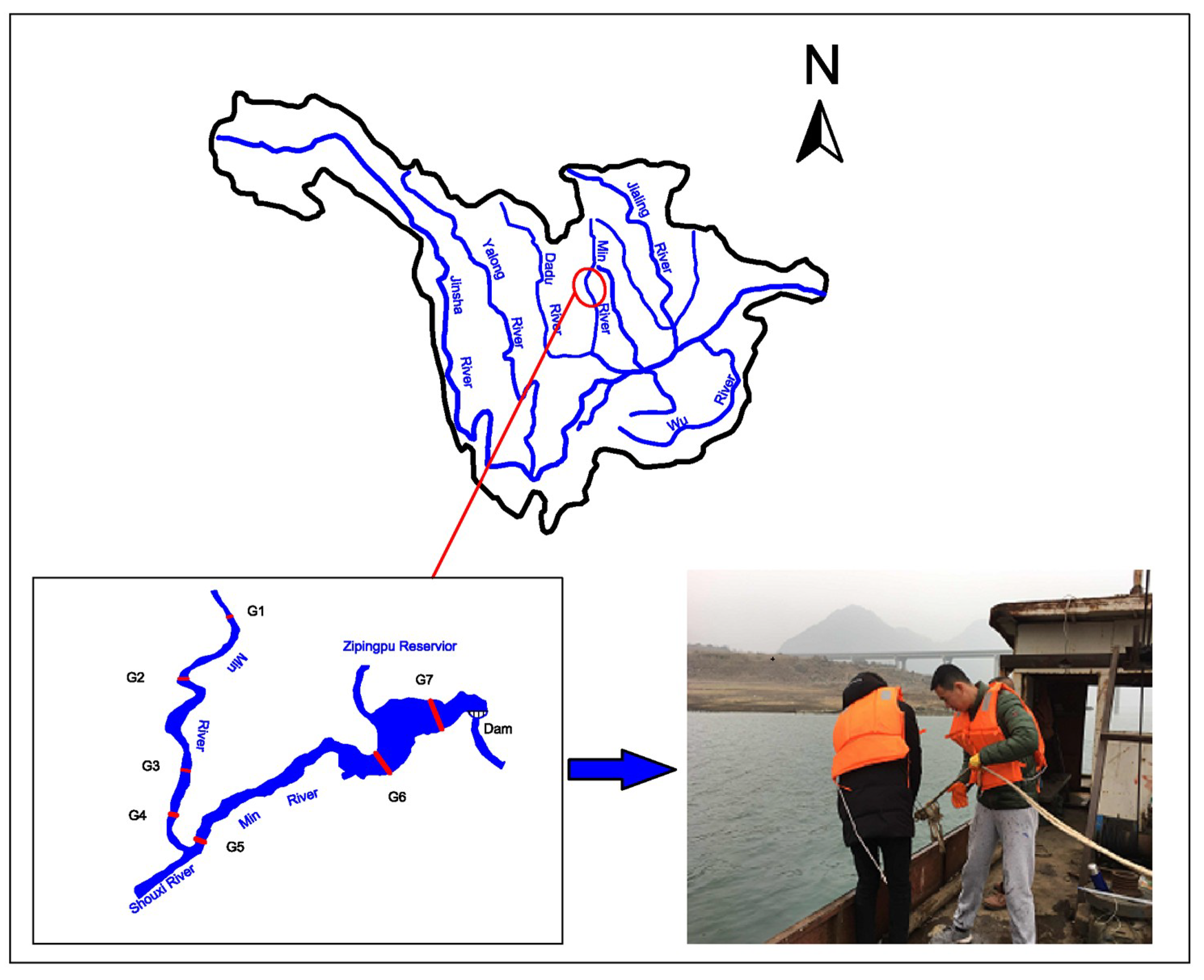
| Sampling Site | Distance from Dam Site (km) | Geographic Coordinates | Notes |
|---|---|---|---|
| G1 | 22.3 | N 31′04′03.33″ E 103’29’21.63″ | Control section at the reservoir tail |
| G2 | 18.8 | N 31′02′44.44″ E 103′28′25.93″ | Downstream of Yingxiu county |
| G3 | 16.2 | N 31°00′38.38″ E 103°28′25.16″ | The middle of the dam and upstream of the Shouxi River, a tributary of the Zipingpu Reservoir |
| G4 | 13.7 | N 31°00′08.32″ E 103°28′21.56″ | The middle of the dam and upstream of the Shouxi tributary |
| G5 | 9.1 | N 31°00′25.89″ E 103°29″35.78″ | The middle of the dam and downstream of the Shouxi River |
| G6 | 4.3 | N 31°01′18.66″ E 103°32′47.16′ | The middle of the dam and downstream the Shouxi River |
| G7 | 2.0 | N 31°01′58.90′ E 103°33′37.53′ | The front of the dam |
| Sampling Site | G1 | G2 | G3 | G4 | G5 | G6 | G7 |
|---|---|---|---|---|---|---|---|
| Ex-P (mg/kg) | 14.50 | 15.82 | 9.09 | 16.36 | 11.66 | 22.61 | 7.52 |
| Al-P (mg/kg) | 4.26 | 2.81 | 5.35 | 2.04 | 2.16 | 2.98 | 4.63 |
| Fe-P (mg/kg) | 8.68 | 7.66 | 9.22 | 12.09 | 2.34 | 7.42 | 39.51 |
| Ca-P (mg/kg) | 1223.16 | 1264.72 | 1171.02 | 638.72 | 740.04 | 489.09 | 392.80 |
| O-P (mg/kg) | 132.68 | 165.86 | 167.14 | 190.23 | 188.22 | 222.92 | 197.65 |
| Sources of Variation | df | F | Sig. | Partial Squared |
|---|---|---|---|---|
| Sampling site | 6 | 10.29 | <0.001 | 0.638 |
| Phosphorus form | 4 | 416.78 | <0.001 | 0.979 |
| Sampling site * Phosphorus form | 24 | 12.85 | <0.001 | 0.898 |
| Sources of Variation | df | F | Sig. | Partial Squared | |
|---|---|---|---|---|---|
| River section | pH | 2 | 11.45 | 0.004 | 0.741 |
| T | 4 | 26.97 | <0.001 | 0.931 | |
| Transition section | pH | 2 | 9.43 | 0.008 | 0.702 |
| T | 4 | 30.92 | <0.001 | 0.939 | |
| Reservoir section | pH | 2 | 7.94 | 0.013 | 0.665 |
| T | 4 | 30.04 | <0.001 | 0.939 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.; Li, K.; Liang, R.; Han, S.; Li, Y. Distribution and Release Characteristics of Phosphorus in a Reservoir in Southwest China. Int. J. Environ. Res. Public Health 2019, 16, 303. https://doi.org/10.3390/ijerph16030303
Wang Y, Li K, Liang R, Han S, Li Y. Distribution and Release Characteristics of Phosphorus in a Reservoir in Southwest China. International Journal of Environmental Research and Public Health. 2019; 16(3):303. https://doi.org/10.3390/ijerph16030303
Chicago/Turabian StyleWang, Yuanming, Kefeng Li, Ruifeng Liang, Shiqing Han, and Yong Li. 2019. "Distribution and Release Characteristics of Phosphorus in a Reservoir in Southwest China" International Journal of Environmental Research and Public Health 16, no. 3: 303. https://doi.org/10.3390/ijerph16030303
APA StyleWang, Y., Li, K., Liang, R., Han, S., & Li, Y. (2019). Distribution and Release Characteristics of Phosphorus in a Reservoir in Southwest China. International Journal of Environmental Research and Public Health, 16(3), 303. https://doi.org/10.3390/ijerph16030303






