Sex-Dependent Effects of PM2.5 Maternal Exposure and Quercetin Intervention on Offspring’s Short Chain Fatty Acids
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of PM2.5 and Chemicals
2.2. Dose Information
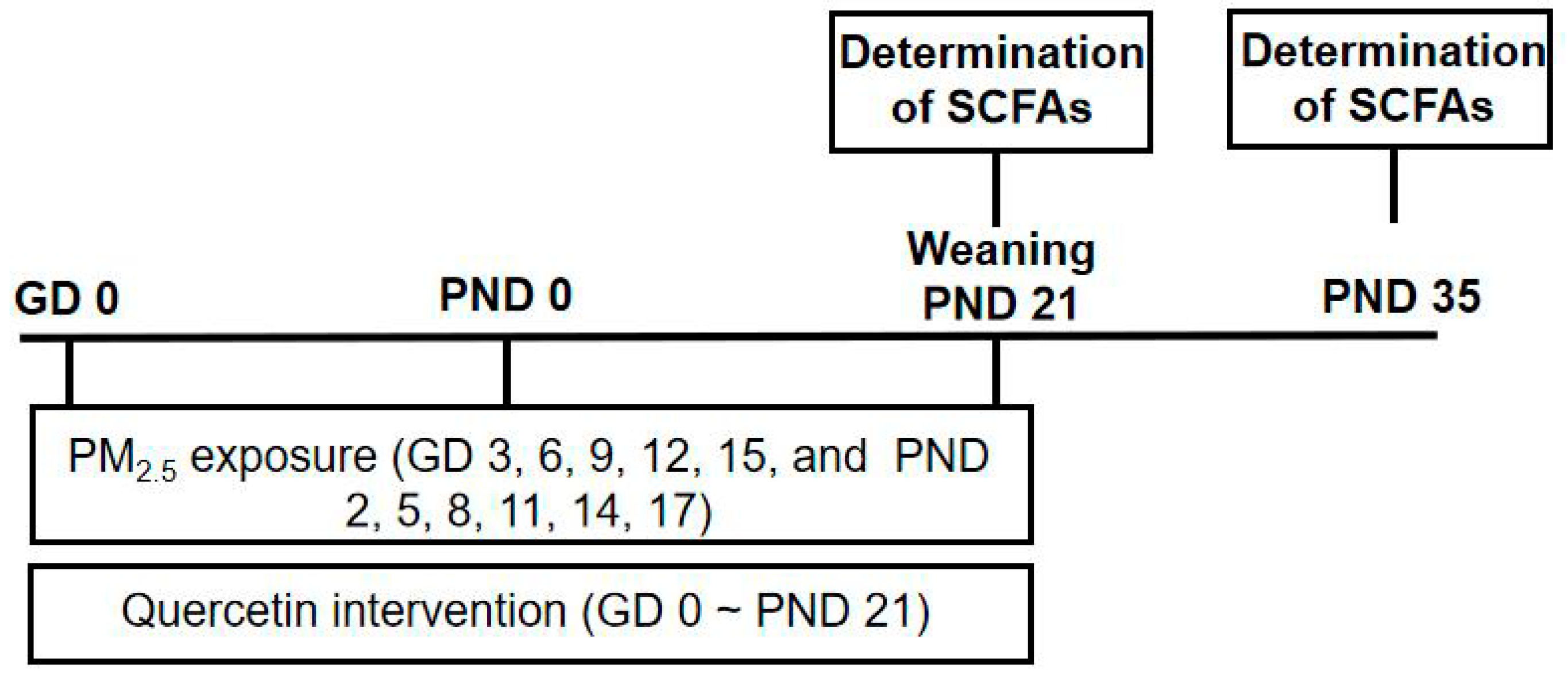
2.3. Animals and Treatment
2.4. Determination of SCFAs
2.5. Statistical Analysis
3. Results
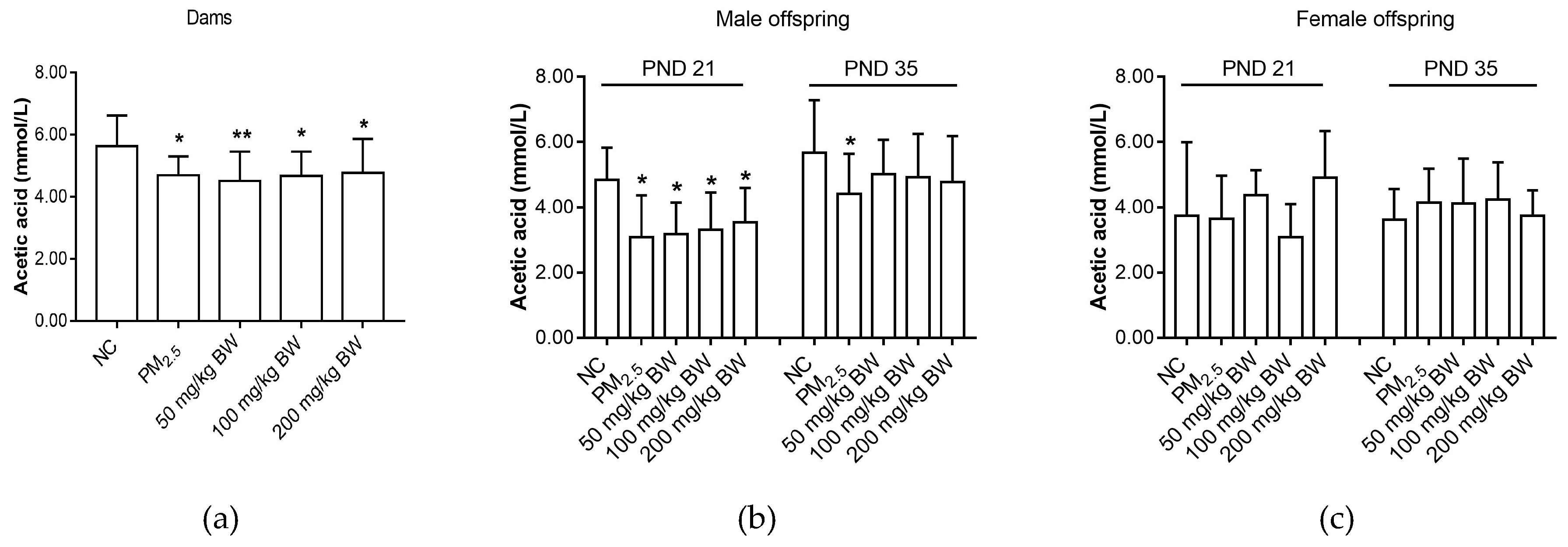
3.1. Acetic Acid
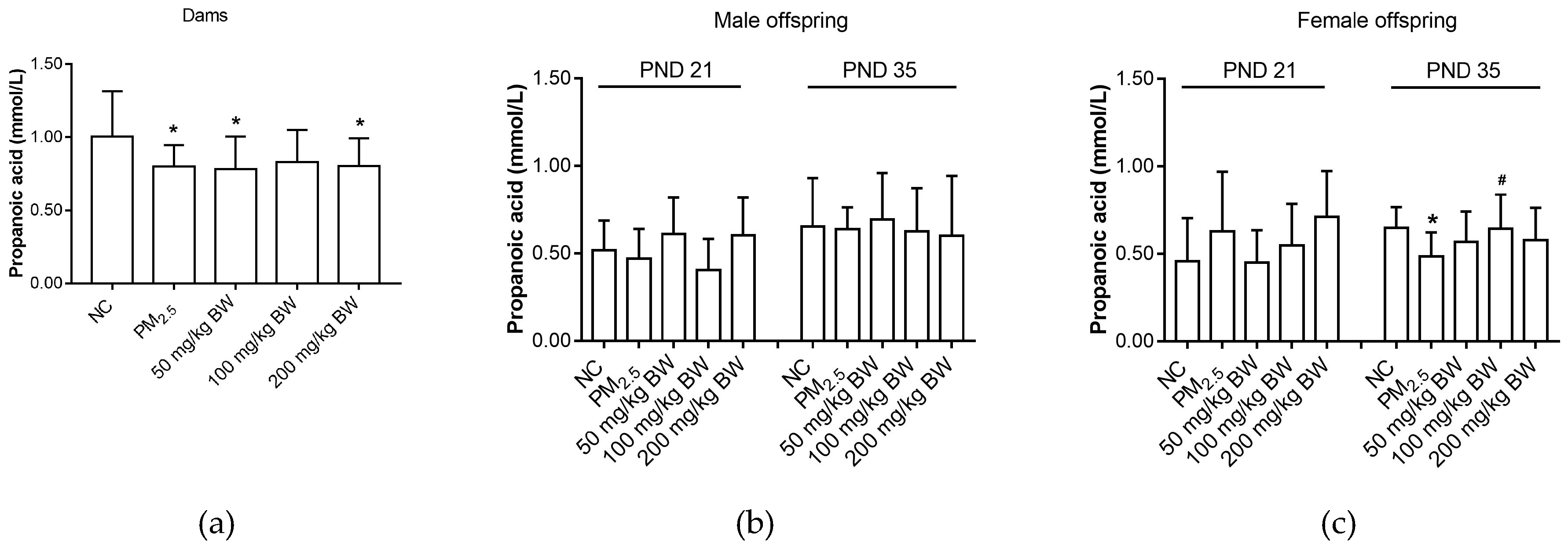
3.2. Propanoic Acid
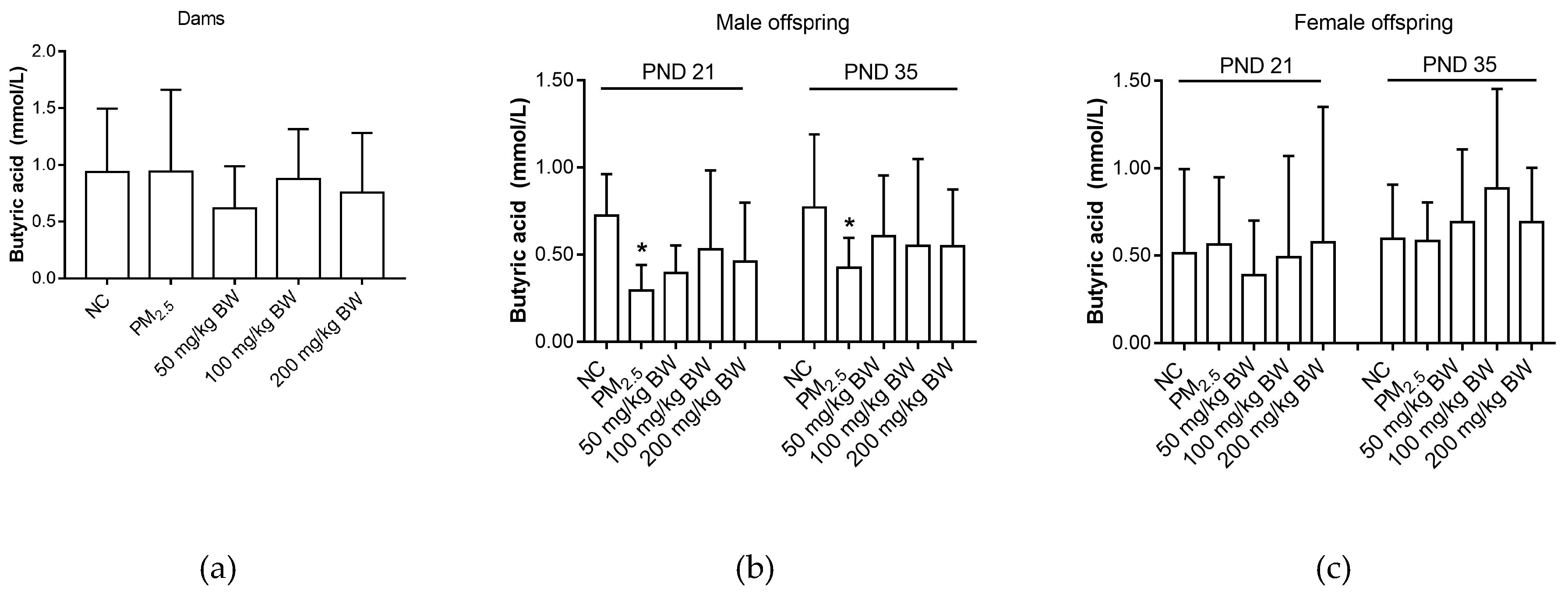
3.3. Butyric Acid
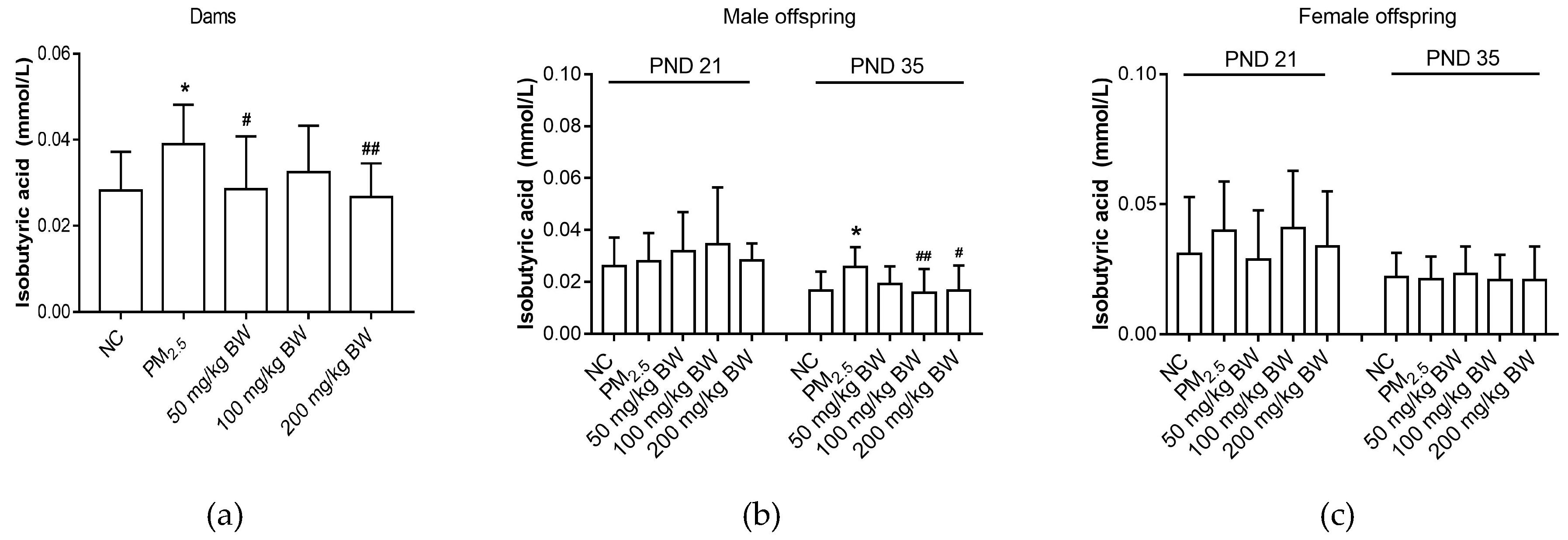
3.4. Isobutyric Acid
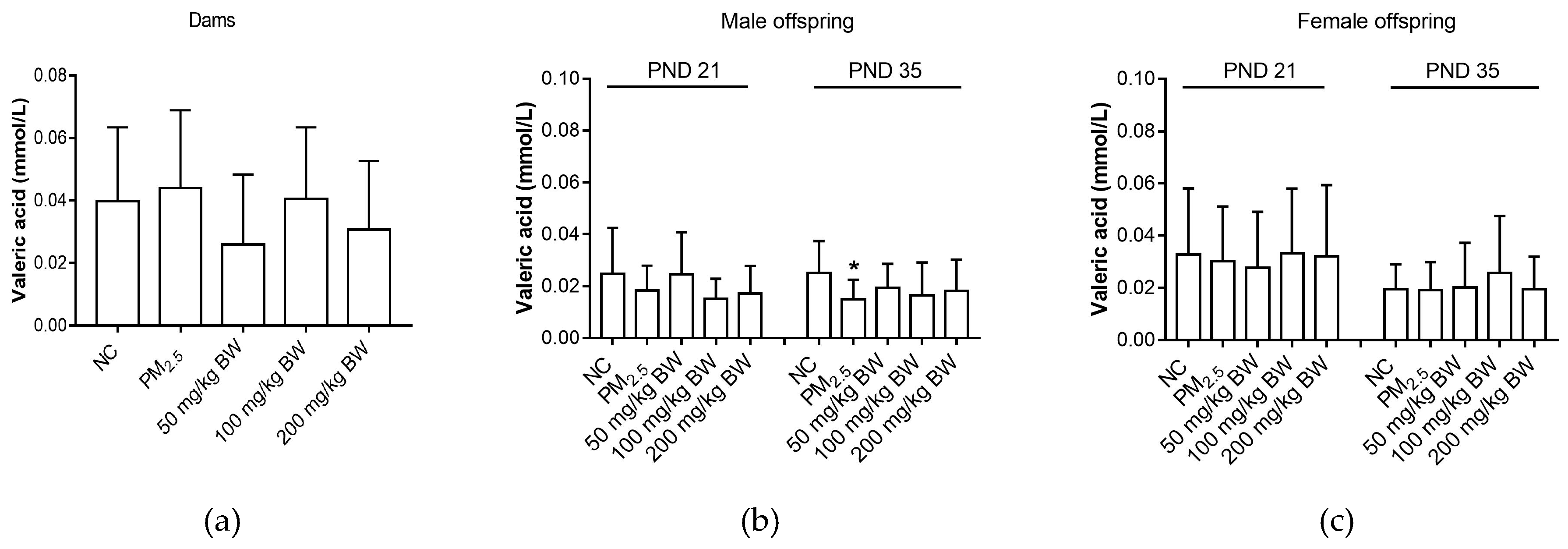
3.5. Valeric Acid
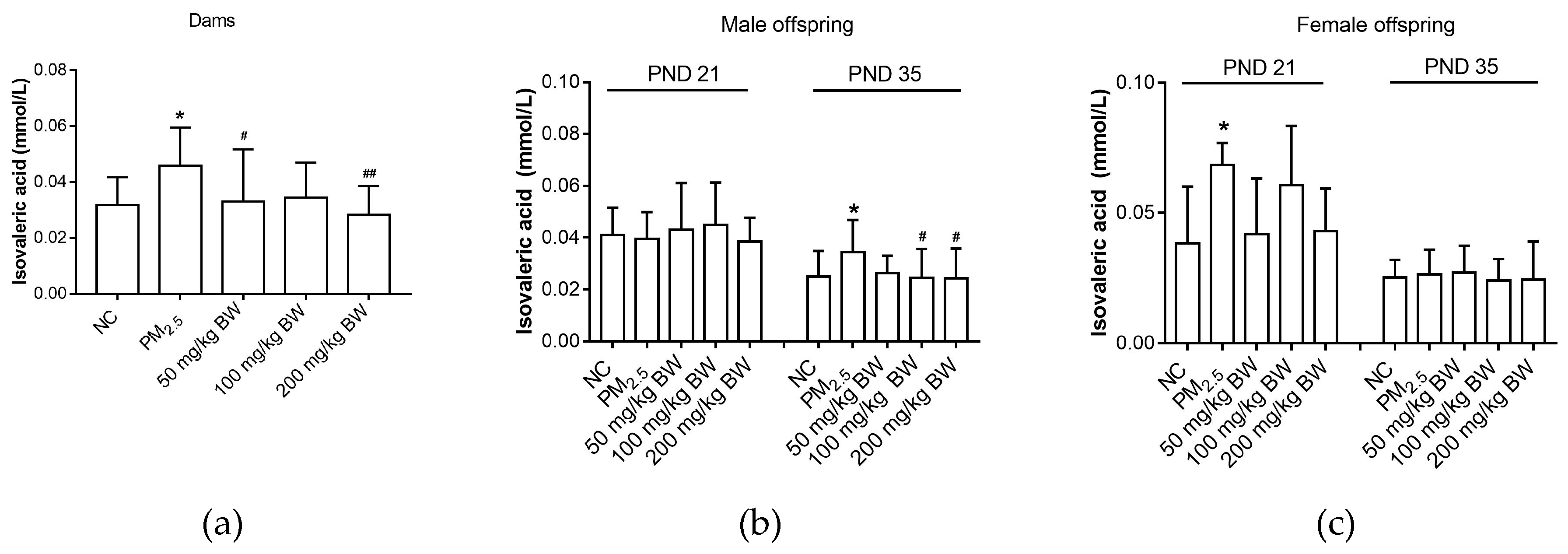
3.6. Isovaleric Acid
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ailshire, J.A.; Crimmins, E.M. Fine particulate matter air pollution and cognitive function among older US adults. Am. J. Epidemiol. 2014, 180, 359–366. [Google Scholar] [CrossRef] [PubMed]
- Liang, R.; Zhang, B.; Zhao, X.; Ruan, Y.; Lian, H.; Fan, Z. Effect of exposure to PM2.5 on blood pressure: A systematic review and meta-analysis. J. Hypertens. 2014, 32, 2130–2140. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Deng, F.; Hao, Y.; Wang, X.; Zheng, C.; Lv, H.; Lu, X.; Wei, H.; Huang, J.; Qin, Y. Fine particulate matter, temperature, and lung function in healthy adults: Findings from the HVNR study. Chemosphere 2014, 108, 168–174. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, G.G.; James, H.; Joshua, K.; Sands, B.E.; Remo, P.; Subrata, G.; Wheeler, A.J.; Villeneuve, P.J. The inflammatory bowel diseases and ambient air pollution: A novel association. Am. J. Gastroenterol. 2010, 105, 2412–2419. [Google Scholar] [CrossRef] [PubMed]
- Ananthakrishnan, A.N.; McGinley, E.L.; Binion, D.G.; Saeian, K. Ambient air pollution correlates with hospitalizations for inflammatory bowel disease: An ecologic analysis. Inflamm. Bowel Dis. 2011, 17, 1138–1145. [Google Scholar] [CrossRef] [PubMed]
- Mutlu, E.A.; Comba, I.Y.; Cho, T.; Engen, P.A.; Yazici, C.; Soberanes, S.; Hamanaka, R.B.; Nigdelioglu, R.; Meliton, A.Y.; Ghio, A.J.; et al. Inhalational exposure to particulate matter air pollution alters the composition of the gut microbiome. Environ. Pollut. 2018, 240, 817–830. [Google Scholar] [CrossRef] [PubMed]
- Byrne, C.S.; Chambers, E.S.; Morrison, D.J.; Frost, G. The role of short chain fatty acids in appetite regulation and energy homeostasis. Int. J. Obes. (Lond.) 2015, 39, 1331–1338. [Google Scholar] [CrossRef] [PubMed]
- Topping, D.L.; Clifton, P.M. Short-chain fatty acids and human colonic function: Roles of resistant starch and nonstarch polysaccharides. Physiol. Rev. 2001, 81, 1031–1064. [Google Scholar] [CrossRef] [PubMed]
- Jian, T.; Mckenzie, C.; Potamitis, M.; Thorburn, A.N.; Mackay, C.R.; Macia, L. Chapter Three—The Role of Short-Chain Fatty Acids in Health and Disease. Adv. Immunol. 2014, 121, 91–119. [Google Scholar]
- Kendrick, S.F.W.; Graeme, O.; Jelena, M.; Mujdat, Z.; Jeremy, P.; Jones, D.E.J.; Day, C.P. Acetate, the key modulator of inflammatory responses in acute alcoholic hepatitis. Hepatology 2010, 51, 1988–1997. [Google Scholar] [CrossRef] [PubMed]
- Petra, L.; Flint, H.J. Diversity, metabolism and microbial ecology of butyrate-producing bacteria from the human large intestine. FEMS Microbiol. Lett. 2010, 294, 1–8. [Google Scholar]
- Wong, J.M.; de Souza, R.; Kendall, C.W.; Emam, A.; Jenkins, D.J. Colonic health: Fermentation and short chain fatty acids. J. Clin. Gastroenterol. 2006, 40, 235–243. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, N.A.; Gassull, M.A.; Remy, M. Short-chain fatty acids: Ready for prime time? Nutr. Clin. Pract. 2006, 21, 351–366. [Google Scholar]
- Liu, W.; Zhang, M.; Feng, J.; Fan, A.; Zhou, Y.; Xu, Y. The Influence of Quercetin on Maternal Immunity, Oxidative Stress, and Inflammation in Mice with Exposure of Fine Particulate Matter during Gestation. Int. J. Environ. Res. Public Health 2017, 14, 592. [Google Scholar] [CrossRef] [PubMed]
- Gruse, J.; Kanitz, E.; Weitzel, J.M.; Tuchscherer, A.; Stefaniak, T.; Jawor, P.; Wolffram, S.; Hammon, H.M. Quercetin Feeding in Newborn Dairy Calves Cannot Compensate Colostrum Deprivation: Study on Metabolic, Antioxidative and Inflammatory Traits. PLoS ONE 2016, 11, e146932. [Google Scholar] [CrossRef] [PubMed]
- Djouossi, M.G.; Tamokou, J.D.; Ngnokam, D.; Kuiate, J.R.; Tapondjou, L.A.; Harakat, D.; Voutquennenazabadioko, L. Antimicrobial and antioxidant flavonoids from the leaves of Oncoba spinosa Forssk. (Salicaceae). BMC Complement. Altern. Med. 2015, 15, 134. [Google Scholar] [CrossRef] [PubMed]
- Upadhyay, N.K.; Ratan, K.; Siddiqui, M.S.; Asheesh, G. Mechanism of Wound-Healing Activity of Hippophae rhamnoides L. Leaf Extract in Experimental Burns. Evid.-Based Complement. Altern. Med. eCAM 2011, 2011, 659705. [Google Scholar] [CrossRef] [PubMed]
- Mouat, M.F.; Kolli, K.; Orlando, R.; Hargrove, J.L.; Grider, A. The effects of quercetin on SW480 human colon carcinoma cells: A proteomic study. Nutr. J. 2005, 4, 11. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Choi, J.W.; Sohng, J.K.; Pandey, R.P.; Park, Y.I. The immunostimulating activity of quercetin 3-O-xyloside in murine macrophages via activation of the ASK1/MAPK/NF-κB signaling pathway. Int. Immunopharmacol. 2015, 31, 88–97. [Google Scholar] [CrossRef] [PubMed]
- Parkar, S.G.; Stevenson, D.E.; Skinner, M.A. The potential influence of fruit polyphenols on colonic microflora and human gut health. Int. J. Food Microbiol. 2008, 124, 295–298. [Google Scholar] [CrossRef] [PubMed]
- Parkar, S.G.; Trower, T.M.; Stevenson, D.E. Fecal microbial metabolism of polyphenols and its effects on human gut microbiota. Anaerobe 2013, 23, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Liu, W.; Zhou, Y.; Li, Y.; Xu, Y. Neurodevelopmental toxicity induced by maternal PM2.5 exposure and protective effects of quercetin and Vitamin C. Chemosphere 2018, 213, 182–196. [Google Scholar] [CrossRef] [PubMed]
- Huang, F.; Li, X.; Wang, C.; Xu, Q.; Wang, W.; Luo, Y.; Tao, L.; Gao, Q.; Guo, J.; Chen, S.; et al. PM2.5 Spatiotemporal Variations and the Relationship with Meteorological Factors during 2013–2014 in Beijing, China. PLoS ONE 2015, 10, e141642. [Google Scholar] [CrossRef] [PubMed]
- Jian, S.; Oran, K.; Shenglin, C.; Rushad, D.; Peter, E.; Park, J.B.; Mark, L. Flavonoid inhibition of sodium-dependent vitamin C transporter 1 (SVCT1) and glucose transporter isoform 2 (GLUT2), intestinal transporters for vitamin C and Glucose. J. Biol. Chem. 2002, 277, 15252–15260. [Google Scholar]
- Maryam, Z.; Reza, G.; Awat, F.; Gholamreza, A.; Leila, D. Does Quercetin Improve Cardiovascular Risk factors and Inflammatory Biomarkers in Women with Type 2 Diabetes: A Double-blind Randomized Controlled Clinical Trial. Int. J. Prev. Med. 2013, 4, 777–785. [Google Scholar]
- Takuya, S.; Shoko, Y.; Hiroshi, H. Physiological concentrations of short-chain fatty acids immediately suppress colonic epithelial permeability. Br. J. Nutr. 2008, 100, 297–305. [Google Scholar]
- Cruz-Bravo, R.K.; Guevara-González, R.G.; Ramos-Gómez, M.; Oomah, B.D.; Wiersma, P.; Campos-Vega, R.; Loarca-Piña, G. The fermented non-digestible fraction of common bean (Phaseolus vulgaris L.) triggers cell cycle arrest and apoptosis in human colon adenocarcinoma cells. Genes Nutr. 2013, 9, 359. [Google Scholar] [CrossRef] [PubMed]
- Chaput, J.; Jensen, M.G.; Thivierge, M.C.; Tremblay, A. Chapter 43—Metabolic Effects of Propionic Acid-Enriched Breads. In Flour and Breads and Their Fortification in Health and Disease Prevention; Preedy, V.R., Watson, R.R., Patel, V.B., Eds.; Academic Press: San Diego, CA, USA, 2011; pp. 475–484. [Google Scholar]
- Wang, W.; Ji, Z.; Chen, M.; Huang, X.; Xie, X.; Li, W.; Qi, C.; Kan, H.; Xu, Y.; Ying, Z. Exposure to concentrated ambient PM 2.5 alters the composition of gut microbiota in a murine model. Part. Fibre Toxicol. 2018, 15, 17. [Google Scholar] [CrossRef] [PubMed]
- Faisal, A.; Amin, I.; Sander, K. Molecular mechanisms underlying the potential antiobesity-related diseases effect of cocoa polyphenols. Mol. Nutr. Food Res. 2014, 58, 33–48. [Google Scholar]
- De, L.I.R.; Milagro, F.I.; Campión, J.; Boqué, N.; Martínez, J.A. Healthy properties of proanthocyanidins. Biofactors 2010, 36, 159–168. [Google Scholar]
- Cardona, F.; Andrés-Lacueva, C.; Tulipani, S.; Tinahones, F.J.; Queipo-Ortuño, M.I. Benefits of polyphenols on gut microbiota and implications in human health. J. Nutr. Biochem. 2013, 24, 1415–1422. [Google Scholar] [CrossRef] [PubMed]
- Willhite, C.C. Teratogenic potential of quercetin in the rat. Food Chem. Toxicol. 1982, 20, 75–79. [Google Scholar] [CrossRef]
- Etxeberria, U.; Arias, N.; Boqué, N.; Macarulla, M.T.; Portillo, M.P.; Martínez, J.A.; Milagro, F.I. Reshaping faecal gut microbiota composition by the intake of trans-resveratrol and quercetin in high-fat sucrose diet-fed rats. J. Nutr. Biochem. 2015, 26, 651–660. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Wu, Z.H.; Xu, Y.J. Effects of quercetin in pregnant and lactation period on weight and expression of insulin-like growth factors-1 mRNA of obese female rats offspring. Beijing Da Xue Xue Bao Yi Xue Ban 2014, 46, 347–354. [Google Scholar] [PubMed]
- Liu, W.; Zhang, M.; Zhou, Y.; Li, Y.; Xu, Y. The Influence of Quercetin on Organs of Mice with Tracheal Exposure of Fine Particulate Matter during Gestation and Lactation. Chin. J. Reprod. Health 2018, 29, 146–153. [Google Scholar]
| Group | N | PM2.5 (mg/kg) | Quercetin (mg/kg) |
|---|---|---|---|
| NC | 12 | - | - |
| PM2.5 | 12 | 15 | - |
| 50 mg/kg BW quercetin | 12 | 15 | Quercetin (50) |
| 100 mg/kg BW quercetin | 12 | 15 | Quercetin (100) |
| 200 mg/kg BW quercetin | 12 | 15 | Quercetin (200) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, W.; Zhou, Y.; Qin, Y.; Li, Y.; Yu, L.; Li, R.; Chen, Y.; Xu, Y. Sex-Dependent Effects of PM2.5 Maternal Exposure and Quercetin Intervention on Offspring’s Short Chain Fatty Acids. Int. J. Environ. Res. Public Health 2019, 16, 4371. https://doi.org/10.3390/ijerph16224371
Liu W, Zhou Y, Qin Y, Li Y, Yu L, Li R, Chen Y, Xu Y. Sex-Dependent Effects of PM2.5 Maternal Exposure and Quercetin Intervention on Offspring’s Short Chain Fatty Acids. International Journal of Environmental Research and Public Health. 2019; 16(22):4371. https://doi.org/10.3390/ijerph16224371
Chicago/Turabian StyleLiu, Wei, Yalin Zhou, Yong Qin, Yong Li, Lanlan Yu, Ruijun Li, Yuhan Chen, and Yajun Xu. 2019. "Sex-Dependent Effects of PM2.5 Maternal Exposure and Quercetin Intervention on Offspring’s Short Chain Fatty Acids" International Journal of Environmental Research and Public Health 16, no. 22: 4371. https://doi.org/10.3390/ijerph16224371
APA StyleLiu, W., Zhou, Y., Qin, Y., Li, Y., Yu, L., Li, R., Chen, Y., & Xu, Y. (2019). Sex-Dependent Effects of PM2.5 Maternal Exposure and Quercetin Intervention on Offspring’s Short Chain Fatty Acids. International Journal of Environmental Research and Public Health, 16(22), 4371. https://doi.org/10.3390/ijerph16224371





