Associations of Various Nighttime Noise Exposure Indicators with Objective Sleep Efficiency and Self-Reported Sleep Quality: A Field Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population and Procedures
2.2. Sleep Outcomes
2.3. Noise Measurements and Modeling
2.4. Data Analysis
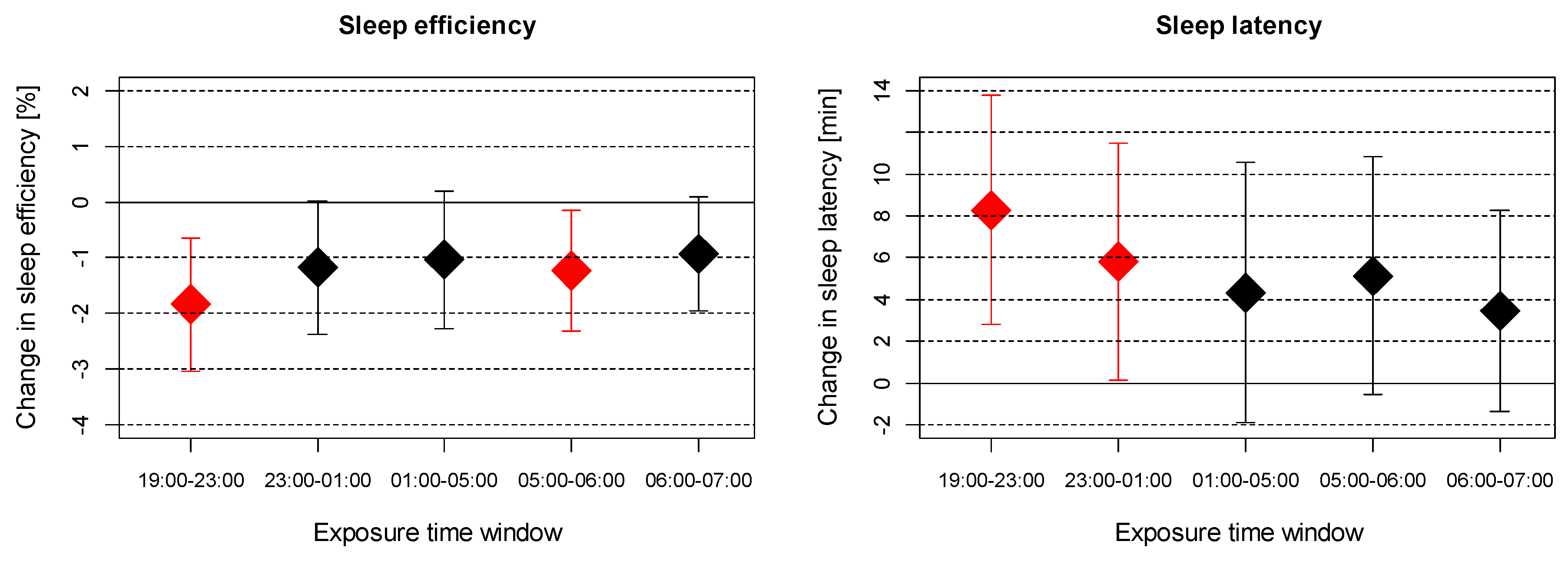
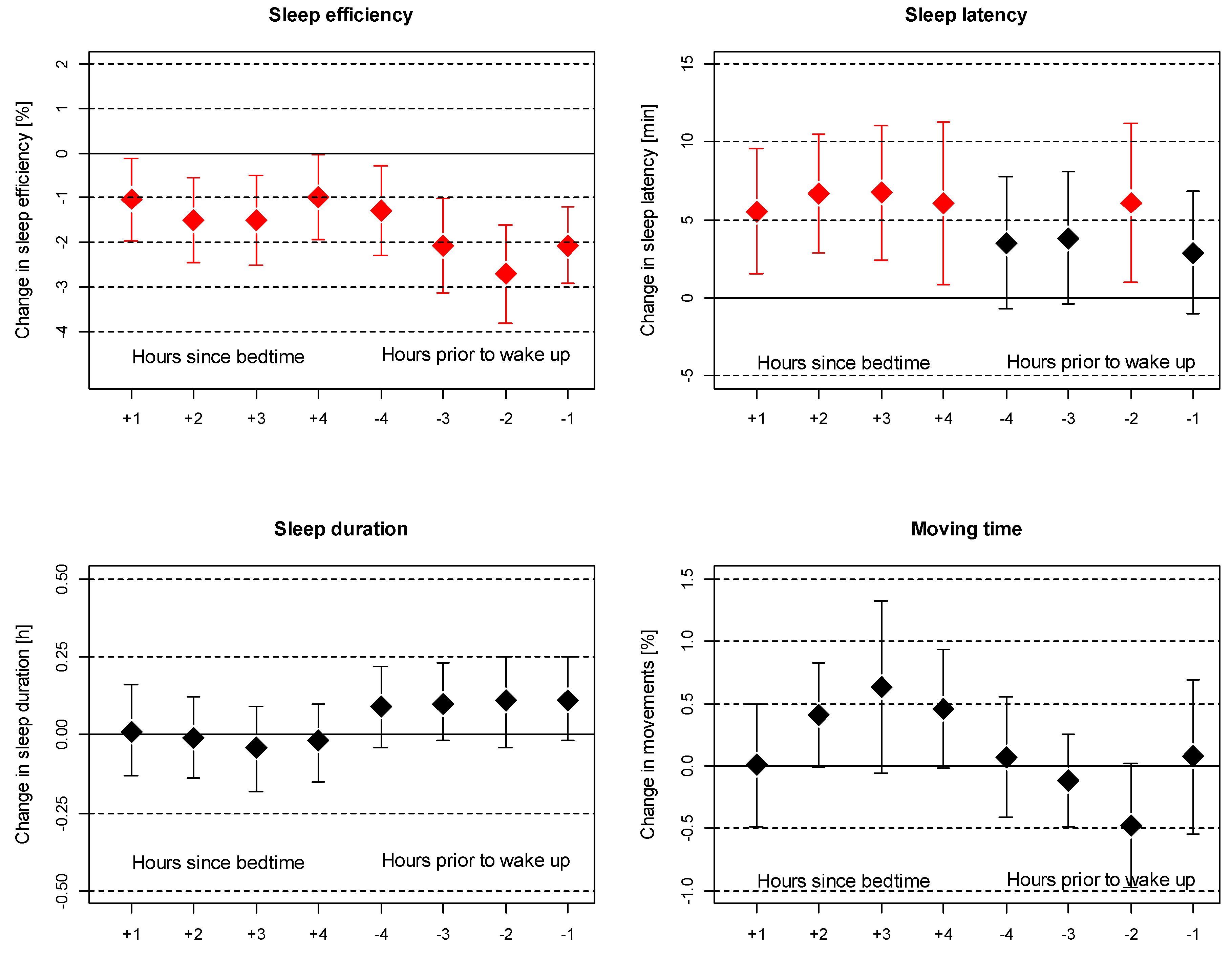
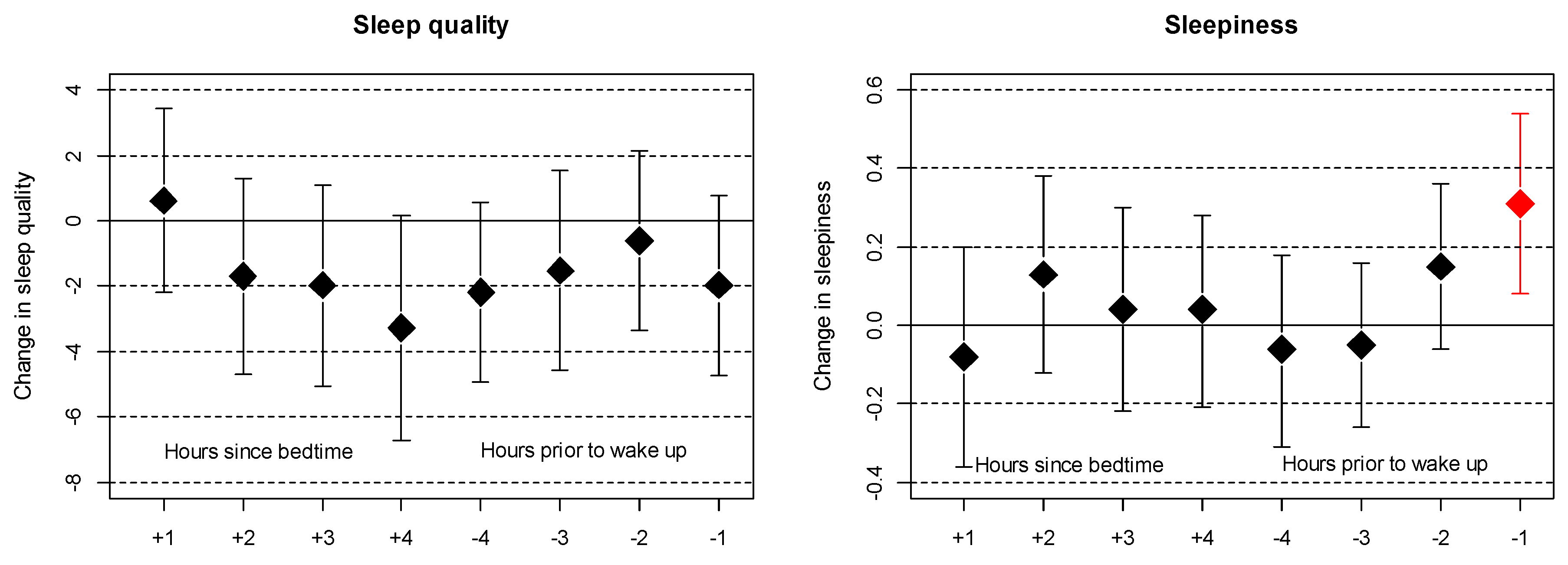
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Heritier, H.; Vienneau, D.; Foraster, M.; Eze, I.C.; Schaffner, E.; de Hoogh, K.; Thiesse, L.; Rudzik, F.; Habermacher, M.; Kopfli, M.; et al. A systematic analysis of mutual effects of transportation noise and air pollution exposure on myocardial infarction mortality: A nationwide cohort study in Switzerland. Eur. Heart J. 2019, 40, 598–603. [Google Scholar] [CrossRef]
- Sorensen, M.; Andersen, Z.J.; Nordsborg, R.B.; Jensen, S.S.; Lillelund, K.G.; Beelen, R.; Schmidt, E.B.; Tjonneland, A.; Overvad, K.; Raaschou-Nielsen, O. Road traffic noise and incident myocardial infarction: A prospective cohort study. PLoS ONE 2012, 7, e39283. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.; Hodgson, S.; Blangiardo, M.; Gulliver, J.; Morley, D.; Fecht, D.; Vienneau, D.; de Hoogh, K.; Key, T.; Hveem, K.; et al. Road traffic noise, air pollution and incident cardiovascular disease: A joint analysis of the hunt, epic-oxford and uk biobank cohorts. Environ. Int. 2018, 114, 191–201. [Google Scholar] [CrossRef]
- Hansell, A.L.; Blangiardo, M.; Fortunato, L.; Floud, S.; de Hoogh, K.; Fecht, D.; Ghosh, R.E.; Laszlo, H.E.; Pearson, C.; Beale, L.; et al. Aircraft noise and cardiovascular disease near heathrow airport in London: Small area study. BMJ 2013, 347, f5432. [Google Scholar] [CrossRef] [PubMed]
- Foraster, M.; Eze, I.C.; Vienneau, D.; Schaffner, E.; Jeong, A.; Heritier, H.; Rudzik, F.; Thiesse, L.; Pieren, R.; Brink, M.; et al. Long-term exposure to transportation noise and its association with adiposity markers and development of obesity. Environ. Int. 2018, 121, 879–889. [Google Scholar] [CrossRef]
- Christensen, J.S.; Hjortebjerg, D.; Raaschou-Nielsen, O.; Ketzel, M.; Sorensen, T.I.; Sorensen, M. Pregnancy and childhood exposure to residential traffic noise and overweight at 7years of age. Environ. Int. 2016, 94, 170–176. [Google Scholar] [CrossRef] [PubMed]
- Eze, I.C.; Foraster, M.; Schaffner, E.; Vienneau, D.; Heritier, H.; Rudzik, F.; Thiesse, L.; Pieren, R.; Imboden, M.; von Eckardstein, A.; et al. Long-term exposure to transportation noise and air pollution in relation to incident diabetes in the sapaldia study. Int. J. Epidemiol. 2017, 46, 1115–1125. [Google Scholar] [CrossRef] [PubMed]
- Ohlwein, S.; Hennig, F.; Lucht, S.; Matthiessen, C.; Pundt, N.; Moebus, S.; Jockel, K.H.; Hoffmann, B. Indoor and outdoor road traffic noise and incident diabetes mellitus: Results from a longitudinal german cohort study. Environ. Epidemiol. 2019, 3, e037. [Google Scholar] [CrossRef]
- Sorensen, M.; Andersen, Z.J.; Nordsborg, R.B.; Becker, T.; Tjonneland, A.; Overvad, K.; Raaschou-Nielsen, O. Long-term exposure to road traffic noise and incident diabetes: A cohort study. Environ. Health Perspect. 2013, 121, 217–222. [Google Scholar] [CrossRef] [PubMed]
- Beutel, M.E.; Junger, C.; Klein, E.M.; Wild, P.; Lackner, K.; Blettner, M.; Binder, H.; Michal, M.; Wiltink, J.; Brahler, E.; et al. Noise annoyance is associated with depression and anxiety in the general population—The contribution of aircraft noise. PLoS ONE 2016, 11, e0155357. [Google Scholar] [CrossRef] [PubMed]
- Orban, E.; McDonald, K.; Sutcliffe, R.; Hoffmann, B.; Fuks, K.B.; Dragano, N.; Viehmann, A.; Erbel, R.; Jockel, K.H.; Pundt, N.; et al. Residential road traffic noise and high depressive symptoms after five years of follow-up: Results from the heinz nixdorf recall study. Environ. Health Perspect. 2016, 124, 578–585. [Google Scholar] [CrossRef] [PubMed]
- Seidler, A.; Hegewald, J.; Seidler, A.L.; Schubert, M.; Wagner, M.; Droge, P.; Haufe, E.; Schmitt, J.; Swart, E.; Zeeb, H. Association between aircraft, road and railway traffic noise and depression in a large case-control study based on secondary data. Environ. Res. 2017, 152, 263–271. [Google Scholar] [CrossRef] [PubMed]
- Clark, C.; Crombie, R.; Head, J.; van Kamp, I.; van Kempen, E.; Stansfeld, S.A. Does traffic-related air pollution explain associations of aircraft and road traffic noise exposure on children’s health and cognition? A secondary analysis of the United Kingdom sample from the ranch project. Am. J. Epidemiol. 2012, 176, 327–337. [Google Scholar] [CrossRef] [PubMed]
- Stansfeld, S.A.; Berglund, B.; Clark, C.; Lopez-Barrio, I.; Fischer, P.; Ohrstrom, E.; Haines, M.M.; Head, J.; Hygge, S.; van Kamp, I.; et al. Aircraft and road traffic noise and children’s cognition and health: A cross-national study. Lancet 2005, 365, 1942–1949. [Google Scholar] [CrossRef]
- Van Kempen, E.; van Kamp, I.; Lebret, E.; Lammers, J.; Emmen, H.; Stansfeld, S. Neurobehavioral effects of transportation noise in primary schoolchildren: A cross-sectional study. Environ. Health 2010, 9, 25. [Google Scholar] [CrossRef] [PubMed]
- Sygna, K.; Aasvang, G.M.; Aamodt, G.; Oftedal, B.; Krog, N.H. Road traffic noise, sleep and mental health. Environ. Res. 2014, 131, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Munzel, T.; Schmidt, F.P.; Steven, S.; Herzog, J.; Daiber, A.; Sorensen, M. Environmental noise and the cardiovascular system. J. Am. Coll. Cardiol. 2018, 71, 688–697. [Google Scholar] [CrossRef] [PubMed]
- McEwen, B.S. Sleep deprivation as a neurobiologic and physiologic stressor: Allostasis and allostatic load. Metabolism 2006, 55, S20–S23. [Google Scholar] [CrossRef]
- Cappuccio, F.P.; Cooper, D.; D’Elia, L.; Strazzullo, P.; Miller, M.A. Sleep duration predicts cardiovascular outcomes: A systematic review and meta-analysis of prospective studies. Eur. Heart J. 2011, 32, 1484–1492. [Google Scholar] [CrossRef]
- Schmid, S.M.; Hallschmid, M.; Schultes, B. The metabolic burden of sleep loss. Lancet Diabetes Endocrinol. 2015, 3, 52–62. [Google Scholar] [CrossRef]
- DePorter, D.P.; Coborn, J.E.; Teske, J.A. Partial sleep deprivation reduces the efficacy of orexin-a to stimulate physical activity and energy expenditure. Obesity 2017, 25, 1716–1722. [Google Scholar] [CrossRef] [PubMed]
- Basner, M.; Muller, U.; Elmenhorst, E.M. Single and combined effects of air, road, and rail traffic noise on sleep and recuperation. Sleep 2011, 34, 11–23. [Google Scholar] [CrossRef] [PubMed]
- Elmenhorst, E.M.; Griefahn, B.; Rolny, V.; Basner, M. Comparing the effects of road, railway, and aircraft noise on sleep: Exposure(-)response relationships from pooled data of three laboratory studies. Int. J. Environ. Res. Public Health 2019, 16, 1073. [Google Scholar] [CrossRef] [PubMed]
- Rudzik, F.; Thiesse, L.; Pieren, R.; Wunderli, J.M.; Brink, M.; Foraster, M.; Heritier, H.; Eze, I.C.; Garbazza, C.; Vienneau, D.; et al. Sleep spindle characteristics and arousability from nighttime transportation noise exposure in healthy young and older individuals. Sleep 2018, 41, zsy077. [Google Scholar] [CrossRef] [PubMed]
- Elmenhorst, E.M.; Pennig, S.; Rolny, V.; Quehl, J.; Mueller, U.; Maass, H.; Basner, M. Examining nocturnal railway noise and aircraft noise in the field: Sleep, psychomotor performance, and annoyance. Sci. Total Environ. 2012, 424, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Frei, P.; Mohler, E.; Röösli, M. Effect of nocturnal road traffic noise exposure and annoyance on objective and subjective sleep quality. Int. J. Hyg. Environ. Health 2014, 217, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Evandt, J.; Oftedal, B.; Hjertager Krog, N.; Nafstad, P.; Schwarze, P.E.; Marit Aasvang, G. A population-based study on nighttime road traffic noise and insomnia. Sleep 2017, 40. [Google Scholar] [CrossRef] [PubMed]
- Basner, M.; McGuire, S. Who environmental noise guidelines for the european region: A systematic review on environmental noise and effects on sleep. Int. J. Environ. Res. Public Health 2017, 14, 1539. [Google Scholar] [CrossRef]
- Janssen, S.A.; Centen, M.R.; Vos, H.; van Kamp, I. The effect of the number of aircraft noise events on sleep quality. Appl. Acoust. 2014, 84, 9–16. [Google Scholar] [CrossRef]
- Basner, M.; Samel, A.; Isermann, U. Aircraft noise effects on sleep: Application of the results of a large polysomnographic field study. J. Acoust. Soc. Am. 2006, 119, 2772–2784. [Google Scholar] [CrossRef]
- Brink, M.; Lercher, P.; Eisenmann, A.; Schierz, C. Influence of slope of rise and event order of aircraft noise events on high resolution actimetry parameters. Somnologie 2008, 12, 118–128. [Google Scholar] [CrossRef]
- Wunderli, J.M.; Pieren, R.; Habermacher, M.; Vienneau, D.; Cajochen, C.; Probst-Hensch, N.; Röösli, M.; Brink, M. Intermittency ratio: A metric reflecting short-term temporal variations of transportation noise exposure. J. Expo. Sci. Environ. Epidemiol. 2016, 26, 575–585. [Google Scholar] [CrossRef] [PubMed]
- Heritier, H.; Vienneau, D.; Foraster, M.; Eze, I.C.; Schaffner, E.; Thiesse, L.; Rudzik, F.; Habermacher, M.; Kopfli, M.; Pieren, R.; et al. Transportation noise exposure and cardiovascular mortality: A nationwide cohort study from switzerland. Eur. J. Epidemiol. 2017, 32, 307–315. [Google Scholar] [CrossRef] [PubMed]
- Griefahn, B.; Marks, A.; Robens, S. Experiments on the time frame of temporally limited traffic curfews to prevent noise induced sleep disturbances. Somnologie 2008, 12, 140–148. [Google Scholar] [CrossRef]
- Brink, M.; Schaffer, B.; Vienneau, D.; Foraster, M.; Pieren, R.; Eze, I.C.; Cajochen, C.; Probst-Hensch, N.; Röösli, M.; Wunderli, J.M. A survey on exposure-response relationships for road, rail, and aircraft noise annoyance: Differences between continuous and intermittent noise. Environ. Int. 2019, 125, 277–290. [Google Scholar] [CrossRef] [PubMed]
- Fields, J.M.; De Jong, R.G.; Gjestland, T.; Flindell, I.H.; Job, R.F.S.; Kurra, S.; Lercher, P.; Vallet, M.; Yano, T.; Guski, R.; et al. Standardized general-purpose noise reaction questions for community noise surveys: Research and a recommendation. J. Sound Vib. 2001, 242, 641–679. [Google Scholar] [CrossRef]
- Griefahn, B.; Marks, A.; Gjestland, T.; Preis, A. Annoyance and Noise Sensitivity in Urban Areas; ICA: Madrid, Spain, 2007. [Google Scholar]
- Akerstedt, T.; Gillberg, M. Subjective and objective sleepiness in the active individual. Int. J. Neurosci. 1990, 52, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Heutschi, K. Sonroad: New swiss road traffic noise model. Acta Acust. United Acust. 2004, 903, 548–554. [Google Scholar]
- Karipidis, I.; Vienneau, D.; Habermacher, M.; Köpflii, M.; Brink, M.; Probst-Hensch, N.; Röösli, M.; Wunderli, J.-M. Reconstruction of historical noise exposure data for environmental epidemiology in switzerland within the sirene project. Noise Mapp. 2014, 1, 3–14. [Google Scholar] [CrossRef]
- Schlatter, F.; Piquerez, A.; Habermacher, M.; Ragettli, M.S.; Röösli, M.; Brink, M.; Cajochen, C.; Probst-Hensch, N.; Foraster, M.; Wunderli, J.-M. Validation of large scale noise exposure modeling by long-term measurements. Noise Mapp. 2017, 4, 75–86. [Google Scholar] [CrossRef]
- Locher, B.; Piquerez, A.; Habermacher, M.; Ragettli, M.; Röösli, M.; Brink, M.; Cajochen, C.; Vienneau, D.; Foraster, M.; Müller, U.; et al. Differences between outdoor and indoor sound levels for open, tilted, and closed windows. Int. J. Environ. Res. Public Health 2018, 15, 149. [Google Scholar] [CrossRef] [PubMed]
- Halonen, J.I.; Vahtera, J.; Stansfeld, S.; Yli-Tuomi, T.; Salo, P.; Pentti, J.; Kivimaki, M.; Lanki, T. Associations between nighttime traffic noise and sleep: The finnish public sector study. Environ. Health Perspect. 2012, 120, 1391–1396. [Google Scholar] [CrossRef] [PubMed]
- Brink, M.; Schaeffer, B.; Pieren, R.; Wunderli, J.M. Conversion between noise exposure indicators leq(24h), l-day, l-evening, l-night, l-dn and l-den: Principles and practical guidance. Int. J. Hyg. Environ. Health 2018, 221, 54–63. [Google Scholar] [CrossRef] [PubMed]
- Heritier, H.; Vienneau, D.; Foraster, M.; Eze, I.C.; Schaffner, E.; Thiesse, L.; Ruzdik, F.; Habermacher, M.; Kopfli, M.; Pieren, R.; et al. Diurnal variability of transportation noise exposure and cardiovascular mortality: A nationwide cohort study from switzerland. Int. J. Hyg. Environ. Health 2018, 221, 556–563. [Google Scholar] [CrossRef] [PubMed]
- Brink, M.; Schäffer, B.; Vienneau, D.; Pieren, R.; Foraster, M.; Eze, I.C.; Rudzik, F.; Thiesse, L.; Cajochen, C.; Probst-Hensch, N.; et al. Self-reported sleep disturbance from road, rail and aircraft noise: Exposure-response relationships and effect modifiers in the swiss SiRENE study. Int. J. Environ. Res. Public Health 2019. submitted. [Google Scholar]
- Foraster, M.; Kunzli, N.; Aguilera, I.; Rivera, M.; Agis, D.; Vila, J.; Bouso, L.; Deltell, A.; Marrugat, J.; Ramos, R.; et al. High blood pressure and long-term exposure to indoor noise and air pollution from road traffic. Environ. Health Perspect. 2014, 122, 1193–1200. [Google Scholar] [CrossRef]
- Foraster, M.; Eze, I.C.; Schaffner, E.; Vienneau, D.; Heritier, H.; Endes, S.; Rudzik, F.; Thiesse, L.; Pieren, R.; Schindler, C.; et al. Exposure to road, railway, and aircraft noise and arterial stiffness in the sapaldia study: Annual average noise levels and temporal noise characteristics. Environ. Health Perspect. 2017, 125, 097004. [Google Scholar] [CrossRef]
| Outcome | N | Mean | SD | Min | Max |
|---|---|---|---|---|---|
| ACT sleep efficiency [%] | 634 | 88.4 | 7.9 | 46.3 | 98.2 |
| ACT sleep latency [min] | 634 | 29.4 | 33 | 2 | 303 |
| ACT sleep duration [h] | 634 | 7.0 | 1.2 | 4.1 | 11.7 |
| ACT moving time [%] | 634 | 7.2 | 4.7 | 0.8 | 35.9 |
| SR sleep quality [0–100] | 639 | 65 | 20 | 4 | 100 |
| SR sleepiness [1–9] | 633 | 4.1 | 1.8 | 1 | 9 |
| Outcome | N | Mean | SD | Min | Max |
|---|---|---|---|---|---|
| Indoor Leq,night | 685 | 30.2 | 7.6 | 20.0 | 55.3 |
| Outdoor Leq,night | 685 | 47.0 | 6.9 | 29.6 | 62.7 |
| Outdoor Leq,19–23 | 685 | 51.2 | 6.6 | 33.2 | 68.6 |
| Outdoor Leq,23–01 | 685 | 46.1 | 7.4 | 29.4 | 62.9 |
| Outdoor Leq,01–05 | 685 | 41.8 | 7.5 | 27.8 | 62.7 |
| Outdoor Leq,05–06 | 685 | 46.3 | 8.1 | 28.8 | 64.7 |
| Outdoor Leq,06–07 | 685 | 50.6 | 8.1 | 29.5 | 70.3 |
| Outcome | N | Coefficient | Confidence Interval | p-Value |
|---|---|---|---|---|
| ACT sleep efficiency [%] | 634 | −1.11 | −2.44 to 0.21 | 0.10 |
| ACT sleep latency [min] | 634 | 5.67 | −1.00 to 12.34 | 0.10 |
| ACT sleep duration [h] | 634 | 0.01 | −0.17 to 0.19 | 0.94 |
| ACT moving time [%] | 634 | −0.41 | −1.13 to 0.31 | 0.27 |
| SR sleep quality [0–100] | 639 | −1.09 | −4.96 to 2.78 | 0.58 |
| SR sleepiness [1–9] | 633 | 0.02 | −0.29 to 0.32 | 0.91 |
| Outcome | N | Coefficient | Confidence Interval | p-Value |
|---|---|---|---|---|
| ACT sleep efficiency [%] | 634 | −1.06 | −2.86 to 0.74 | 0.25 |
| ACT sleep latency [min] | 634 | 4.39 | −5.54 to 14.32 | 0.39 |
| ACT sleep duration [h] | 634 | −0.06 | −0.21 to 0.10 | 0.48 |
| ACT moving time [%] | 634 | −0.24 | −0.90 to 0.42 | 0.47 |
| SR sleep quality [0–100] | 639 | 0.21 | −3.46 to 3.88 | 0.91 |
| SR sleepiness [1–9] | 633 | −0.01 | −0.28 to 0.26 | 0.95 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Röösli, M.; Brink, M.; Rudzik, F.; Cajochen, C.; Ragettli, M.S.; Flückiger, B.; Pieren, R.; Vienneau, D.; Wunderli, J.-M. Associations of Various Nighttime Noise Exposure Indicators with Objective Sleep Efficiency and Self-Reported Sleep Quality: A Field Study. Int. J. Environ. Res. Public Health 2019, 16, 3790. https://doi.org/10.3390/ijerph16203790
Röösli M, Brink M, Rudzik F, Cajochen C, Ragettli MS, Flückiger B, Pieren R, Vienneau D, Wunderli J-M. Associations of Various Nighttime Noise Exposure Indicators with Objective Sleep Efficiency and Self-Reported Sleep Quality: A Field Study. International Journal of Environmental Research and Public Health. 2019; 16(20):3790. https://doi.org/10.3390/ijerph16203790
Chicago/Turabian StyleRöösli, Martin, Mark Brink, Franziska Rudzik, Christian Cajochen, Martina S. Ragettli, Benjamin Flückiger, Reto Pieren, Danielle Vienneau, and Jean-Marc Wunderli. 2019. "Associations of Various Nighttime Noise Exposure Indicators with Objective Sleep Efficiency and Self-Reported Sleep Quality: A Field Study" International Journal of Environmental Research and Public Health 16, no. 20: 3790. https://doi.org/10.3390/ijerph16203790
APA StyleRöösli, M., Brink, M., Rudzik, F., Cajochen, C., Ragettli, M. S., Flückiger, B., Pieren, R., Vienneau, D., & Wunderli, J.-M. (2019). Associations of Various Nighttime Noise Exposure Indicators with Objective Sleep Efficiency and Self-Reported Sleep Quality: A Field Study. International Journal of Environmental Research and Public Health, 16(20), 3790. https://doi.org/10.3390/ijerph16203790









