E-Cigarettes Increase Candida albicans Growth and Modulate its Interaction with Gingival Epithelial Cells
Abstract
1. Introduction
2. Materials and Methods
2.1. Candida Strain
2.2. E-Cigarettes
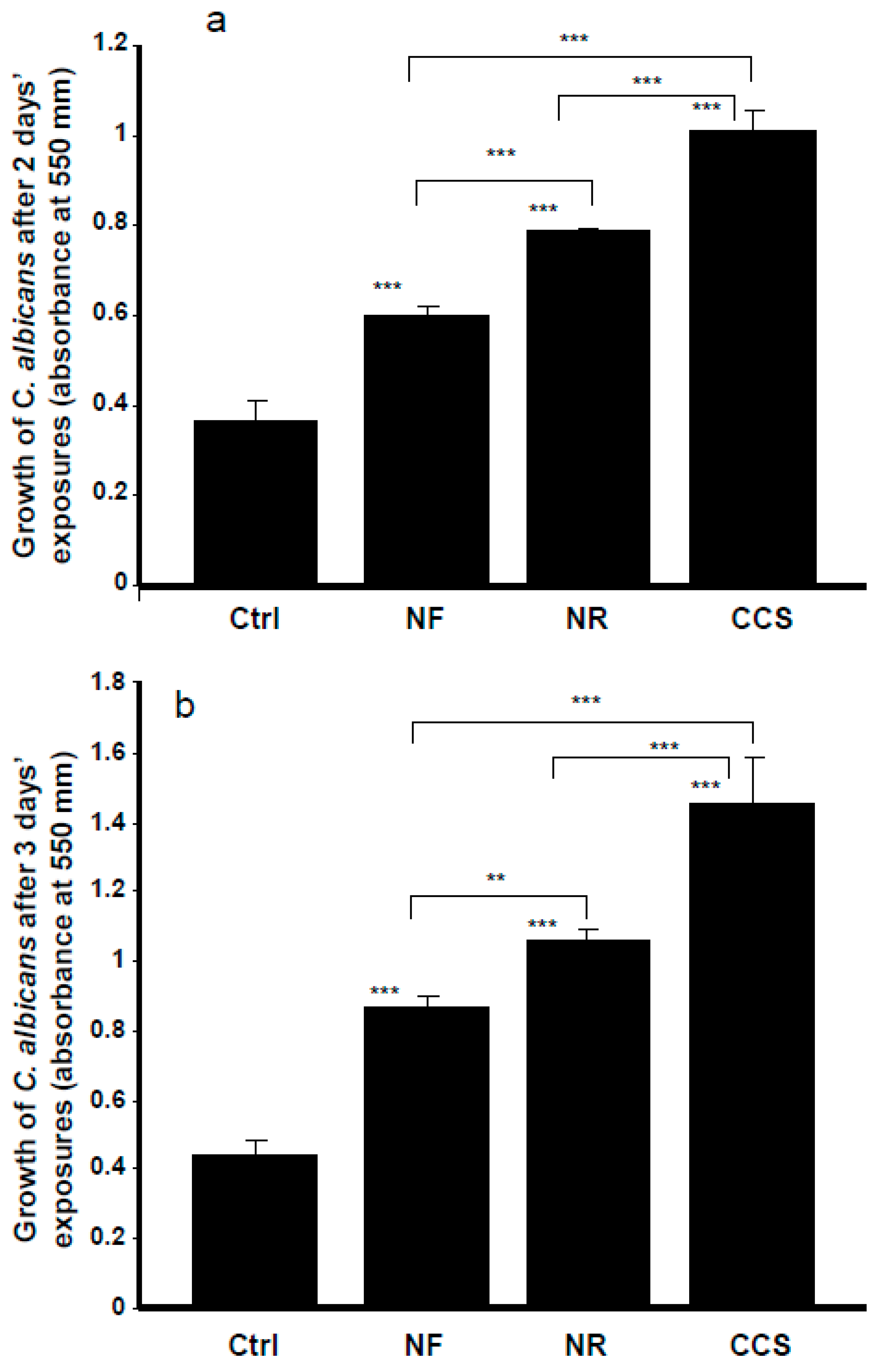
2.3. Effect of e-Vapor on C. albicans Growth
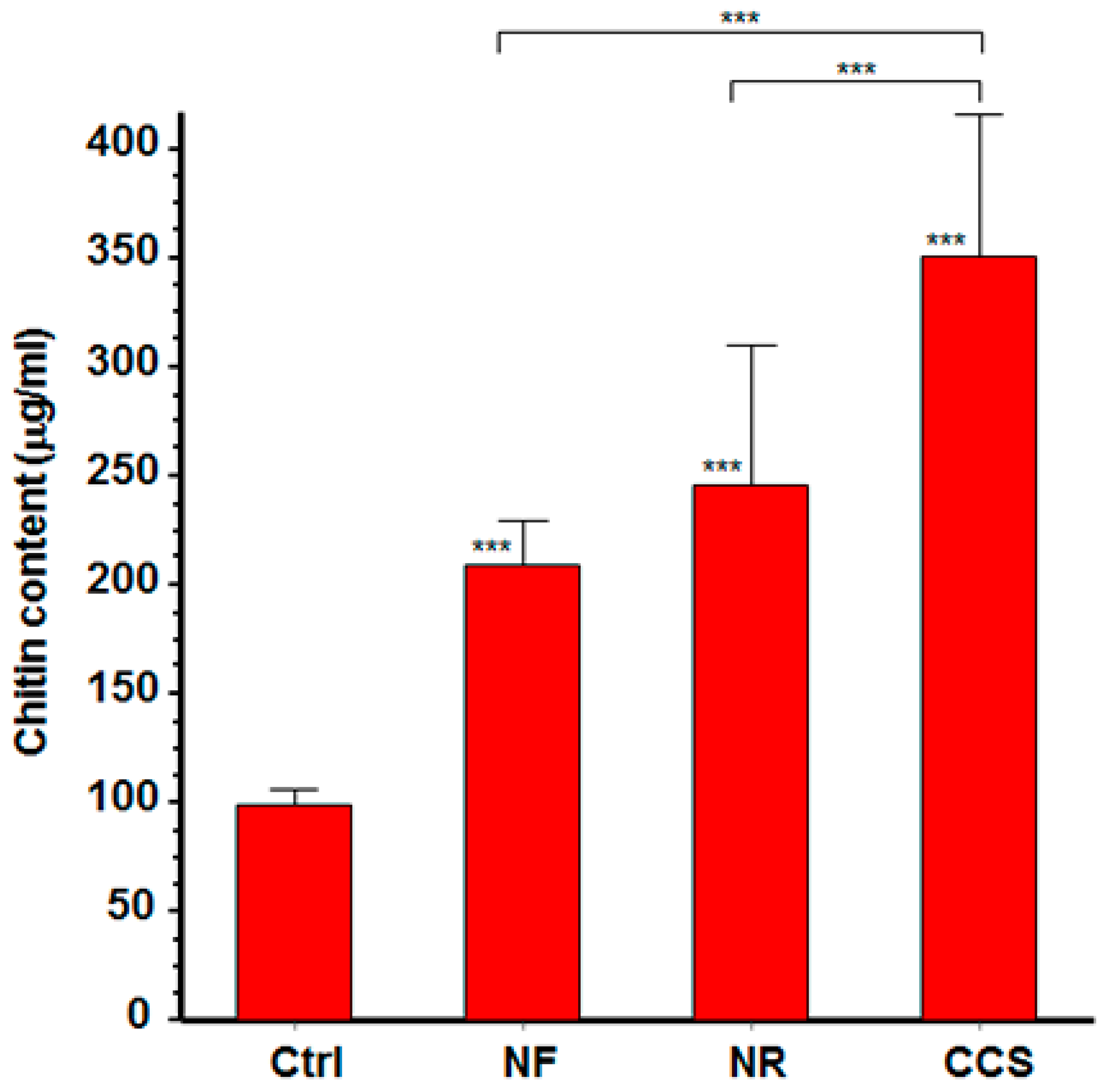
2.4. Effect of e-Vapor on C. albicans Cell Wall Chitin Content
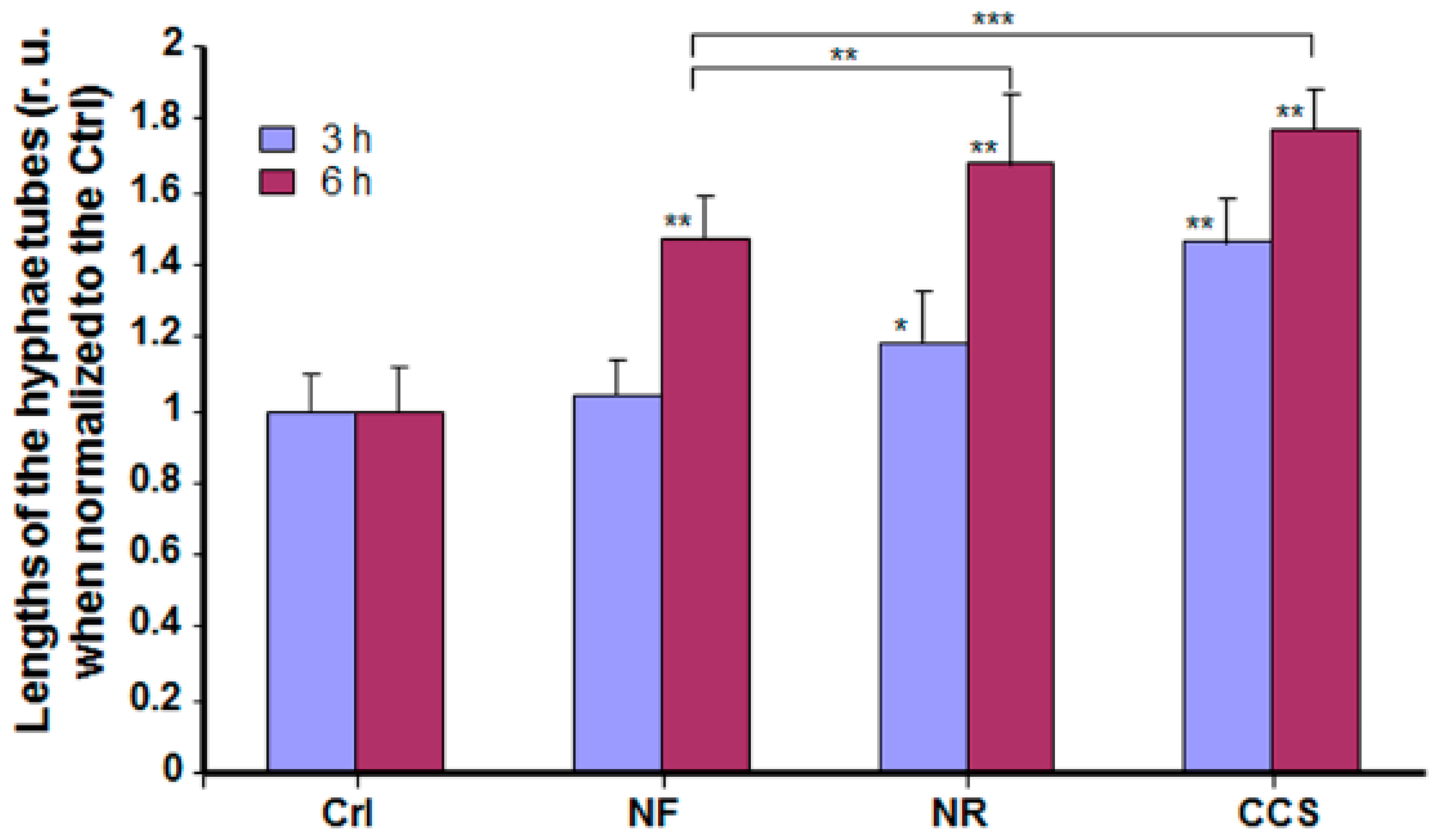
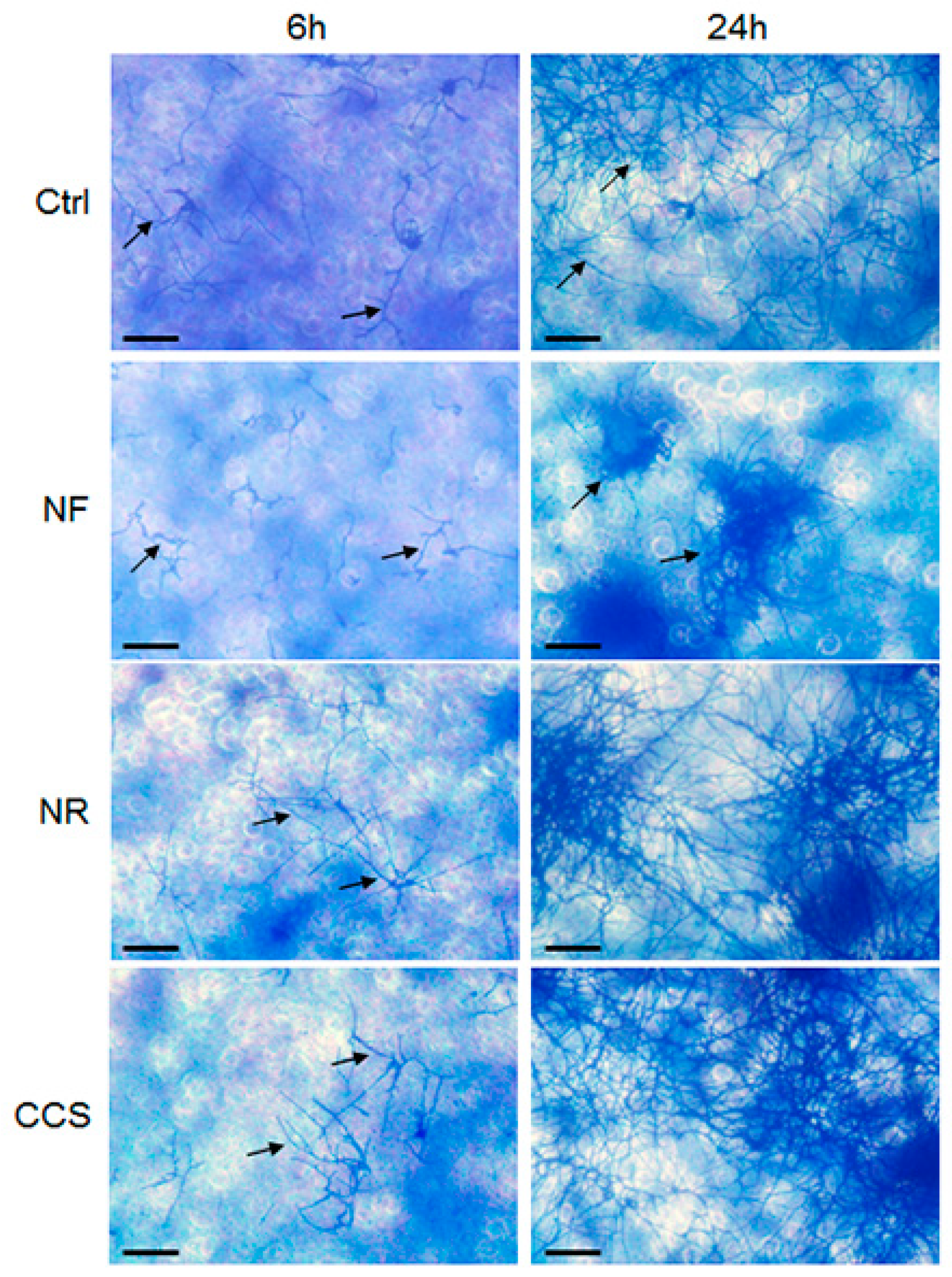
2.5. Effect of e-Vapor on C. albicans Transition from Blastospore to Hyphal Form
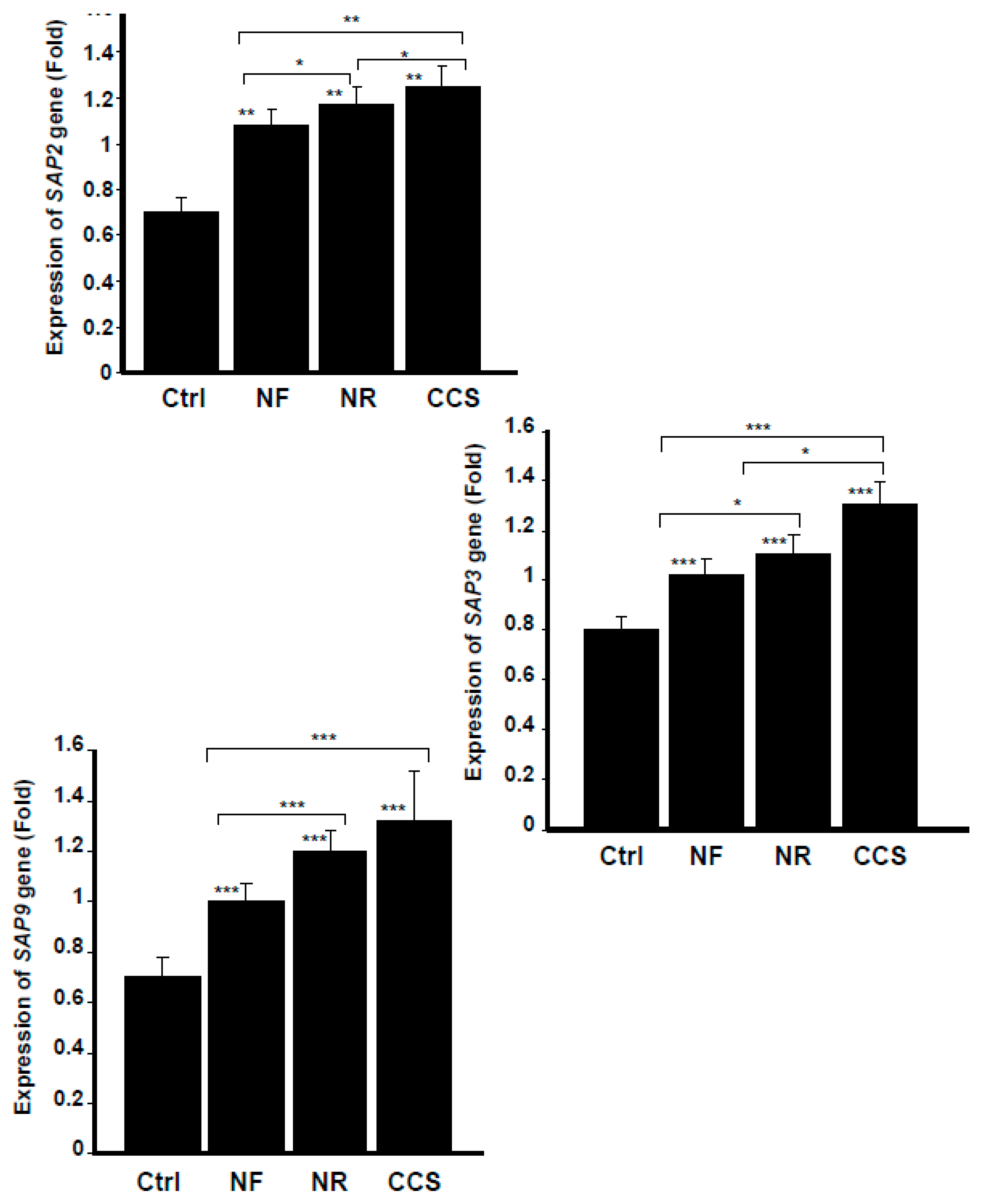
2.6. Effect of e-Vapor on the Expression of SAP2, SAP3, and SAP9 genes by C. albicans
2.7. Adhesion of e-Vapor-Exposed C. albicans to Gingival Epithelial Cells
2.8. Growth of Epithelial Cells Following Indirect Contact with e-Vapor-Exposed C. albicans
2.9. Statistical Analysis
3. Results and Discussion
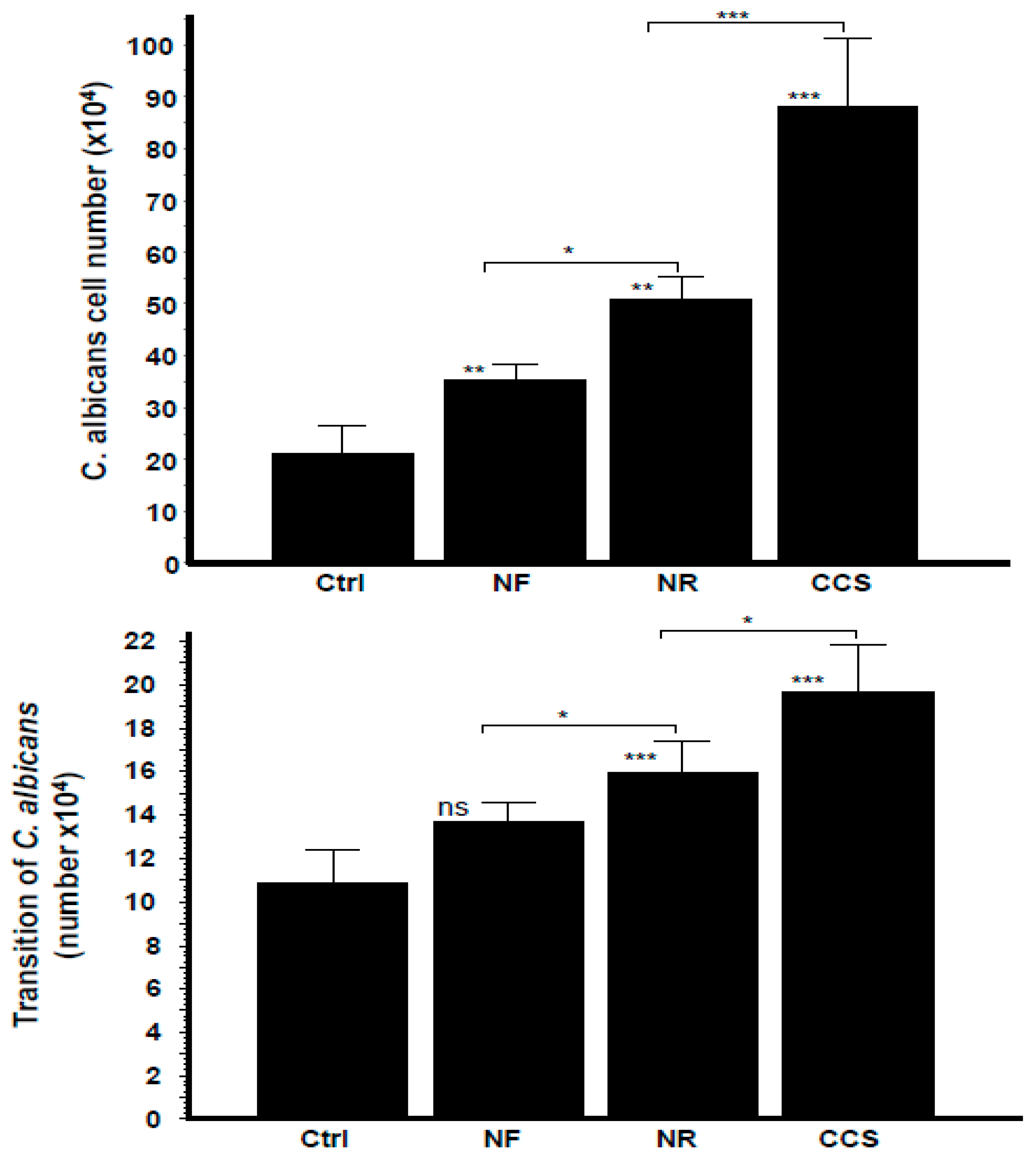
3.1. E-Cigarette Vapor Promoted C. albicans Growth
3.2. Chitin Content was High in e-Cigarette Vapor-Exposed C. albicans
3.3. E-Vapor-Exposed C. albicans Displayed an Increase in Hyphal Length
3.4. E-Vapor-Exposed C. albicans Expressed High Virulent Gene Levels
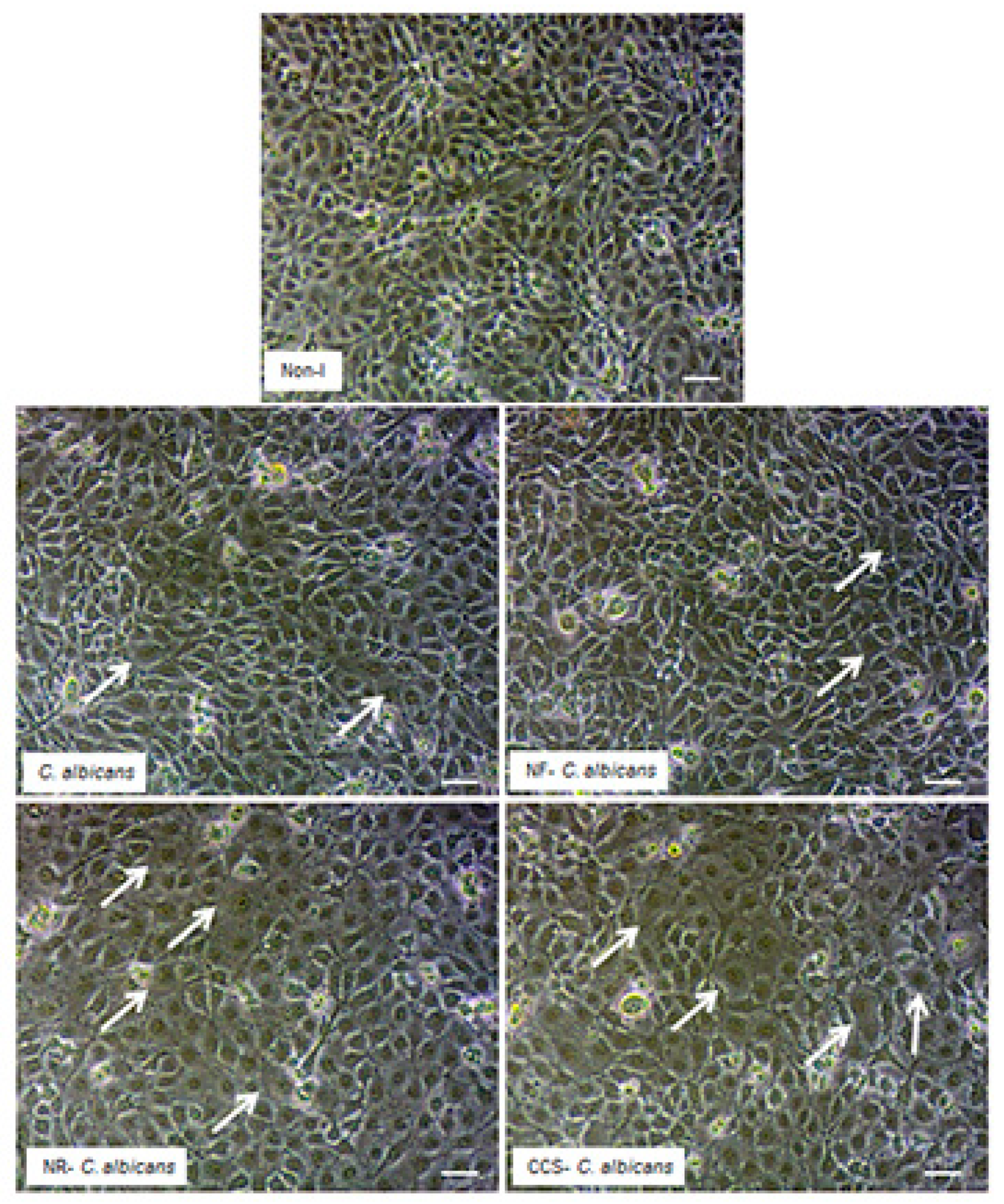
3.5. E-Vapor-Exposed C. albicans Adhered Better to Gingival Epithelial Cells
3.6. Crosstalk Interactions between e-Vapor-Exposed C. albicans and Epithelial Cells Promoted the Yeast’s Growth and Morphological Changes
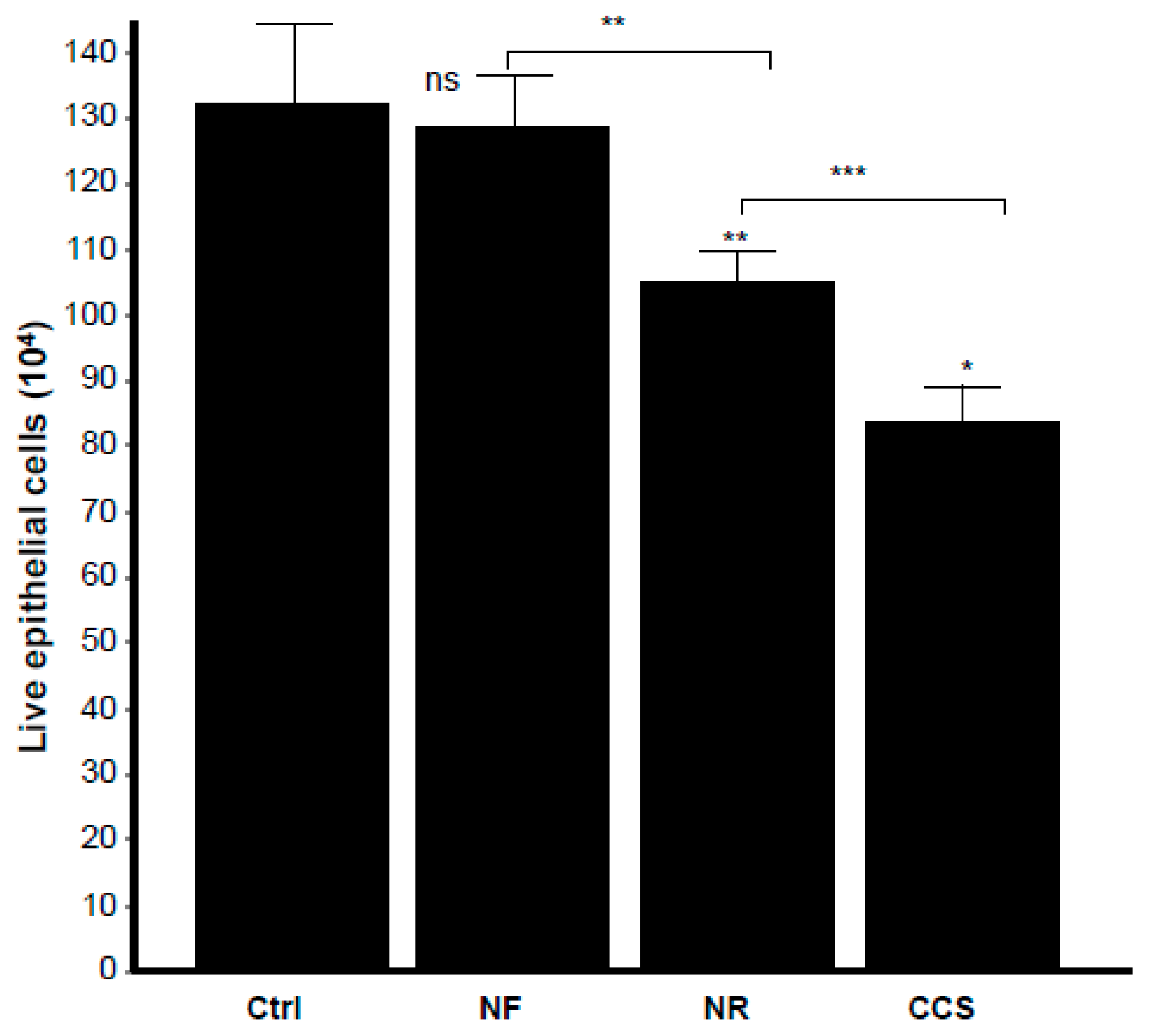
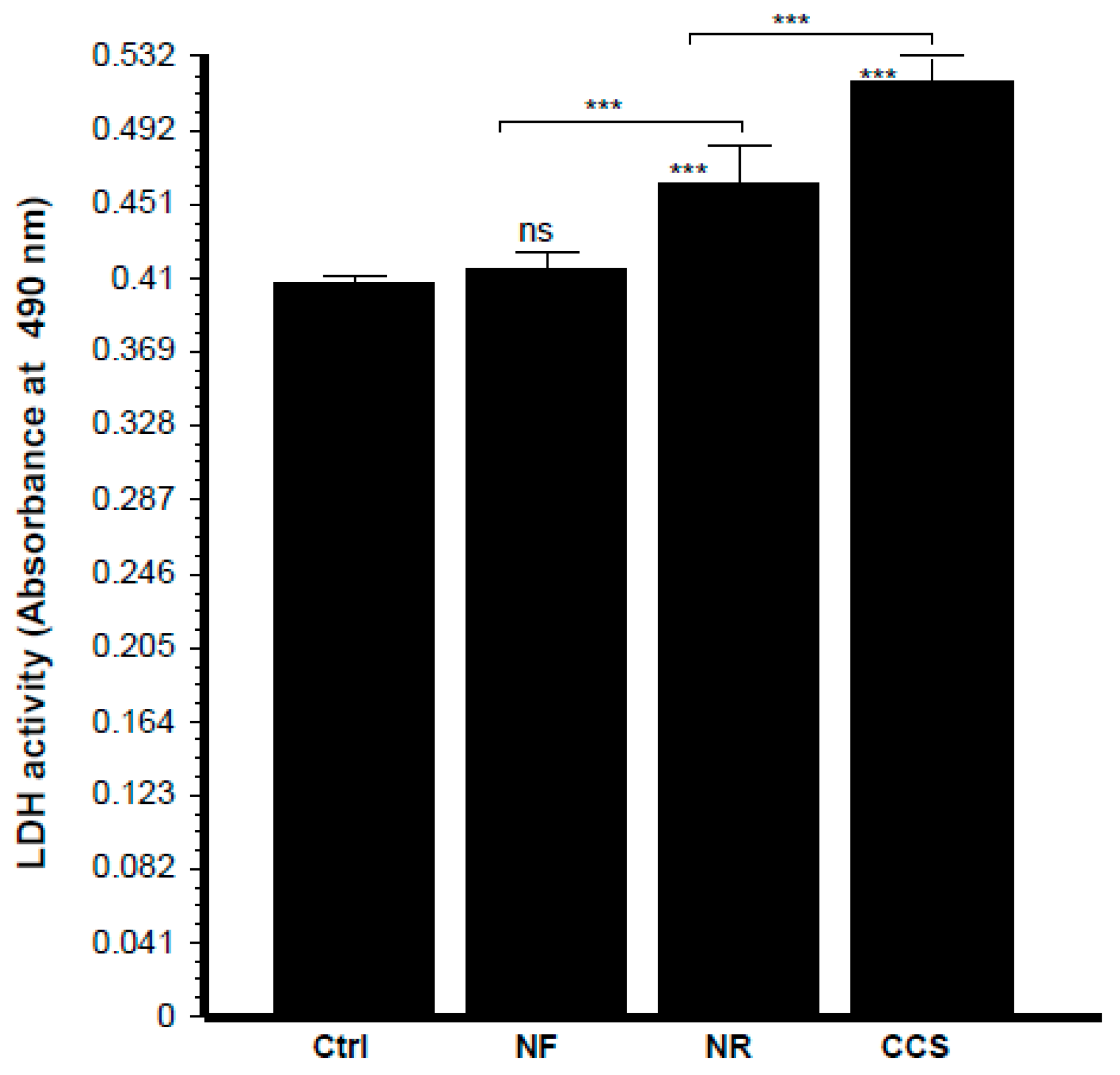
3.7. E-Vapor-Exposed C. albicans Promoted Morphological Changes in Epithelial Cells and Reduced Their Growth
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Feldman, C.; Anderson, R. Cigarette smoking and mechanisms of susceptibility to infections of the respiratory tract and other organ systems. J. Infect. 2013, 67, 169–184. [Google Scholar] [CrossRef] [PubMed]
- Vellappally, S.; Fiala, Z.; Smejkalová, J.; Jacob, V.; Shriharsha, P. Influence of tobacco use in dental caries development. Cent. Eur. J. Public Health 2007, 15, 116–121. [Google Scholar] [CrossRef] [PubMed]
- Baboni, F.B.; Barp, D.; Izidoro, A.C.; Samaranayake, L.P.; Rosa, E.A. Enhancement of Candida albicans virulence after exposition to cigarette mainstream smoke. Mycopathologia 2009, 168, 227–235. [Google Scholar] [CrossRef] [PubMed]
- Bagaitkar, J.; Williams, L.R.; Renaud, D.E.; Bemakanakere, M.R.; Martin, M.; Scott, D.A.; Demuth, D.R. Tobacco-induced alterations to Porphyromonas gingivalis-host interactions. Environ. Microbiol. 2009, 11, 1242–1253. [Google Scholar] [CrossRef] [PubMed]
- Canabarro, A.; Valle, C.; Farias, M.R.; Santos, F.B.; Lazera, M.; Wanke, B. Association of subgingival colonization of Candida albicans and other yeasts with severity of chronic periodontitis. J. Periodontal Res. 2013, 48, 428–432. [Google Scholar] [CrossRef] [PubMed]
- Dahlen, G. Role of suspected periodontopathogens in microbiological monitoring of periodontitis. Adv. Dent. Res. 1993, 7, 163–174. [Google Scholar] [CrossRef]
- Karkowska-Kuleta, J.; Bartnicka, D.; Zawrotniak, M.; Zielinska, G.; Kieronska, A.; Bochenska, O.; Ciaston, I.; Koziel, J.; Potempa, J.; Baster, Z.; et al. The activity of bacterial peptidylarginine deiminase is important during formation of dual-species biofilm by periodontal pathogen Porphyromonas gingivalis and opportunistic fungus Candida albicans. Pathog. Dis. 2018, 76, fty033. [Google Scholar] [CrossRef]
- Al Mubarak, S.; Robert, A.A.; Baskaradoss, J.K.; Al-Zoman, K.; Al Sohail, A.; Alsuwyed, A.; Ciancio, S. The prevalence of oral Candida infections in periodontitis patients with type 2 diabetes mellitus. J. Infect. Public Health 2013, 6, 296–301. [Google Scholar] [CrossRef]
- Rubio, N.A.; Puia, S.; Toranzo, S.; Brusca, M.I. Fungal invasion of connective tissue in patients with gingival-periodontal disease. Rev. Iberoam. Micol. 2015, 32, 20–24. [Google Scholar] [CrossRef]
- Semlali, A.; Killer, K.; Alanazi, H.; Chmielewski, W.; Rouabhia, M. Cigarette smoke condensate increases C. albicans adhesion, growth, biofilm formation, and EAP1, HWP1 and SAP2 gene expression. BMC Microbiol. 2014, 14, 61. [Google Scholar] [CrossRef]
- Wigginton, B.; Gartner, C.; Rowlands, I.J. Is It Safe to Vape? Analyzing online forums discussing e-cigarette use during pregnancy. Womens Health Issues 2017, 27, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Margham, J.; McAdam, K.; Forster, M.; Liu, C.; Wright, C.; Mariner, D.; Proctor, C. Chemical Composition of Aerosol from an E-Cigarette: A Quantitative Comparison with Cigarette Smoke. Chem. Res. Toxicol. 2016, 29, 1662–1678. [Google Scholar] [CrossRef] [PubMed]
- Lerner, C.A.; Sundar, I.K.; Watson, R.M.; Elder, A.; Jones, R.; Done, D.; Kurtzman, R.; Ossip, D.J.; Robinson, R.; McIntosh, S.; et al. Environmental health hazards of e-cigarettes and their components: Oxidants and copper in e-cigarette aerosols. Environ. Pollut. 2015, 198, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Lerner, C.A.; Rutagarama, P.; Ahmad, T.; Sundar, I.K.; Elder, A.; Rahman, I. Electronic cigarette aerosols and copper nanoparticles induce mitochondrial stress and promote DNA fragmentation in lung fibroblasts. Biochem. Biophys. Res. Commun. 2016, 477, 620–625. [Google Scholar] [CrossRef] [PubMed]
- Belmadani, A.; Semlali, A.; Rouabhia, M. Dermaseptin-S1 decreases Candida albicans growth, biofilm formation and the expression of hyphal wall protein 1 and aspartic protease genes. J. Appl. Microbiol. 2018, 125, 72–83. [Google Scholar] [CrossRef] [PubMed]
- Alanazi, H.; Semlali, A.; Perraud, L.; Chmielewski, W.; Zakrzewski, A.; Rouabhia, M. Cigarette smoke-exposed Candida albicans increased chitin production and modulated human fibroblast cell responses. Biomed. Res. Int. 2014, 2014, 963156. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Imamura, K.; Kokubu, E.; Kita, D.; Ota, K.; Yoshikawa, K.; Ishihara, K.; Saito, A. Role of mitogen-activated protein kinase pathways in migration of gingival epithelial cells in response to stimulation by cigarette smoke condensate and infection by Porphyromonas gingivalis. J. Periodontal Res. 2016, 51, 613–621. [Google Scholar] [CrossRef] [PubMed]
- Camenga, D.R.; Tindle, H.A. Weighing the risks and benefits of electronic cigarette use in high-risk Populations. Med. Clin. N. Am. 2018, 102, 765–779. [Google Scholar] [CrossRef]
- Rouabhia, M.; Park, H.J.; Semlali, A.; Zakrzewski, A.; Chmielewski, W.; Chakir, J. E-Cigarette vapor induces an apoptotic response in human gingival epithelial cells through the caspase-3 pathway. J. Cell. Physiol. 2017, 232, 1539–1547. [Google Scholar] [CrossRef]
- Brown, A.J.P.; Budge, S.; Kaloriti, D.; Tillmann, A.; Jacobsen, M.D.; Yin, Z.; Ene, I.V.; Bohovych, I.; Sandai, D.; Kastora, S.; et al. Stress adaptation in a pathogenic fungus. J. Exp. Biol. 2014, 217, 144–155. [Google Scholar] [CrossRef] [PubMed]
- Walker, L.A.; Munro, C.A.; De Bruijn, I.; Lenardon, M.D.; McKinnon, A.; Gow, N.A.R. Stimulation of chitin synthesis rescues Candida albicans from echinocandins. PLoS Pathog. 2008, 4, e1000040. [Google Scholar] [CrossRef] [PubMed]
- Saville, S.P.; Lazzell, A.L.; Monteagudo, C.; Lopez-Ribot, J.L. Engineered control of cell morphology in vivo reveals distinct roles for yeast and filamentous forms of Candida albicans during infection. Eukaryot. Cell 2003, 2, 1053–1060. [Google Scholar] [CrossRef]
- Hirakawa, M.P.; Martinez, D.A.; Sakthikumar, S.; Anderson, M.Z.; Berlin, A.; Gujja, S.; Zeng, Q.; Zisson, E.; Wang, J.M.; Greenberg, J.M.; et al. Genetic and phenotypic intra-species variation in Candida albicans. Genome Res. 2015, 25, 413–425. [Google Scholar] [CrossRef] [PubMed]
- Vogtmann, E.; Graubard, B.; Loftfield, E.; Chaturvedi, A.; Dye, B.A.; Abnet, C.C.; Freedman, N.D. Contemporary impact of tobacco use on periodontal disease in the USA. Tob. Control 2017, 26, 237–238. [Google Scholar] [CrossRef]
- Mathe, L.; Van Dijck, P. Recent insights into Candida albicans biofilm resistance. Curr. Genet. 2013, 59, 251–264. [Google Scholar] [CrossRef] [PubMed]
- Monod, M.; Hube, B.; Hess, D.; Sanglard, D. Differential regulation of SAP8 and SAP9, which encode two new members of the secreted aspartic proteinase family in Candida albicans. Microbiology 1998, 144 Pt 10, 2731–2737. [Google Scholar] [CrossRef]
- Cavalcanti, Y.W.; Wilson, M.; Lewis, M.; Del-Bel-Cury, A.A.; da Silva, W.J.; Williams, D.W. Modulation of Candida albicans virulence by bacterial biofilms on titanium surfaces. Biofouling 2016, 32, 123–134. [Google Scholar] [CrossRef] [PubMed]
- Naglik, J.R.; Moyes, D.; Makwana, J.; Kanzaria, P.; Tsichlaki, E.; Weindl, G.; Tappuni, A.R.; Rodgers, C.A.; Woodman, A.J.; Challacombe, S.J.; et al. Quantitative expression of the Candida albicans secreted aspartyl proteinase gene family in human oral and vaginal candidiasis. Microbiology 2008, 154 Pt 11, 3266–3280. [Google Scholar] [CrossRef]
- Morrow, B.; Srikantha, T.; Anderson, J.; Soll, D.R. Coordinate regulation of two opaque-phase-specific genes during white-opaque switching in Candida albicans. Infect. Immun. 1993, 61, 1823–1828. [Google Scholar] [PubMed]
- Solis, N.V.; Park, Y.N.; Swidergall, M.; Daniels, K.J.; Filler, S.G.; Soll, D.R. Candida albicans white-opaque switching influences virulence but not mating during oropharyngeal candidiasis. Infect. Immun. 2018, 86, e00774-17. [Google Scholar] [CrossRef] [PubMed]
- Schild, L.; Heyken, A.; de Groot, P.W.; Hiller, E.; Mock, M.; de Koster, C.; Horn, U.; Rupp, S.; Hube, B. Proteolytic cleavage of covalently linked cell wall proteins by Candida albicans SAP9 and SAP10. Eukaryot. Cell 2011, 10, 98–109. [Google Scholar] [CrossRef] [PubMed]
- Joo, M.Y.; Shin, J.H.; Jang, H.C.; Song, E.S.; Kee, S.J.; Shin, M.G.; Suh, S.P.; Ryang, D.W. Expression of SAP5 and SAP9 in Candida albicans biofilms: Comparison of bloodstream isolates with isolates from other sources. Med. Mycol. 2013, 51, 892–896. [Google Scholar] [CrossRef] [PubMed]
- Albrecht, A.; Felk, A.; Pichova, I.; Naglik, J.R.; Schaller, M.; de Groot, P.; Maccallum, D.; Odds, F.C.; Schäfer, W.; Klis, F.; et al. Glycosylphosphatidyinositol-anchored proteases of Candida albicans target proteins necessary for both cellular processes and host-pathogen interactions. J. Biol. Chem. 2006, 281, 688–694. [Google Scholar] [CrossRef]
- Rapala-Kozik, M.; Bochenska, O.; Zawrotniak, M.; Wolak, N.; Trebacz, G.; Gogol, M.; Ostrowska, D.; Aoki, W.; Ueda, M.; Kozik, A. Inactivation of the antifungal and immunomodulatory properties of human cathelicidin LL-37 by aspartic proteases produced by the pathogenic yeast Candida albicans. Infect. Immun. 2015, 83, 2518–2530. [Google Scholar] [CrossRef]
- Teughels, W.; Van Eldere, J.; van Steenberghe, D.; Cassiman, J.J.; Fives-Taylor, P.; Quirynen, M. Influence of nicotine and cotinine on epithelial colonization by periodontopathogens. J. Periodontol. 2005, 76, 1315–1322. [Google Scholar] [CrossRef]
- Plaine, A.; Walker, L.; Da Costa, G.; Mora-Montes, H.M.; McKinnon, A.; Gow, N.A.; Gaillardin, C.; Munro, C.A.; Richard, M.L. Functional analysis of Candida albicans GPI-anchored proteins: Roles in cell wall integrity and caspofungin sensitivity. Fungal Genet. Biol. 2008, 45, 1404–1414. [Google Scholar] [CrossRef]
- Pâquet, I.; Chouinard, N.; Rouabhia, M. Cutaneous cell and extracellular matrix responses to ultraviolet-B irradiation. J. Cell. Physiol. 1996, 166, 296–304. [Google Scholar] [CrossRef]
- Alanazi, H.; Park, H.J.; Chakir, J.; Semlali, A.; Rouabhia, M. Comparative study of the effects of cigarette smoke and electronic cigarettes on human gingival fibroblast proliferation, migration and apoptosis. Food Chem. Toxicol. 2018, 118, 390–398. [Google Scholar] [CrossRef]
- Anderson, C.; Majeste, A.; Hanus, J.; Wang, S. E-Cigarette aerosol exposure induces reactive oxygen species, DNA damage, and cell death in vascular endothelial cells. Toxicol. Sci. 2016, 154, 332–340. [Google Scholar] [CrossRef]
- Rouabhia, M.; Alanazi, H.; Park, H.J.; Gonçalves, R.B. Cigarette smoke and E-cigarette vapor dysregulate osteoblast interaction with titanium dental implant surface. J. Oral Implantol. 2018. [Google Scholar] [CrossRef] [PubMed]

| Gene | Primer Sequence (5′ à 3′) | Tm-m | Tm-Exp | Amp Size (Bp) |
|---|---|---|---|---|
| ACT1 | Forward: GACAATTTCTCTTTCAGCACTAGTAGTGA | 57 °C | 60 °C | 87 |
| Reverse: GCTGGTAGAGACTTGACCAACCA | 59 °C | 60 °C | ||
| SAP2 | Forward: TCCTGATGTTAATGTTGATTGTCAAG | 54 °C | 60 °C | 82 |
| Reverse: TGGATCATATGTCCCCTTTTGTT | 54 °C | 60 °C | ||
| SAP3 | Forward: GGACCAGTAACATTTTTATGAGTTTTGAT | 55 °C | 60 °C | 87 |
| Reverse: TGCTACTCCAACAACTTTCAACAAT | 56 °C | 60 °C | ||
| SAP9 | Forward: ATTTACTCCACAGTTTATCACTGAAGGT | 57 °C | 60 °C | 86 |
| Reverse: CCACAAGAACCACCCTCAGTT | 57 °C | 60 °C |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alanazi, H.; Semlali, A.; Chmielewski, W.; Rouabhia, M. E-Cigarettes Increase Candida albicans Growth and Modulate its Interaction with Gingival Epithelial Cells. Int. J. Environ. Res. Public Health 2019, 16, 294. https://doi.org/10.3390/ijerph16020294
Alanazi H, Semlali A, Chmielewski W, Rouabhia M. E-Cigarettes Increase Candida albicans Growth and Modulate its Interaction with Gingival Epithelial Cells. International Journal of Environmental Research and Public Health. 2019; 16(2):294. https://doi.org/10.3390/ijerph16020294
Chicago/Turabian StyleAlanazi, Humidah, Abdelhabib Semlali, Witold Chmielewski, and Mahmoud Rouabhia. 2019. "E-Cigarettes Increase Candida albicans Growth and Modulate its Interaction with Gingival Epithelial Cells" International Journal of Environmental Research and Public Health 16, no. 2: 294. https://doi.org/10.3390/ijerph16020294
APA StyleAlanazi, H., Semlali, A., Chmielewski, W., & Rouabhia, M. (2019). E-Cigarettes Increase Candida albicans Growth and Modulate its Interaction with Gingival Epithelial Cells. International Journal of Environmental Research and Public Health, 16(2), 294. https://doi.org/10.3390/ijerph16020294






