Comparison between the Bouchama and Japanese Association for Acute Medicine Heatstroke Criteria with Regard to the Diagnosis and Prediction of Mortality of Heatstroke Patients: A Multicenter Observational Study
Abstract
1. Background
2. Methods
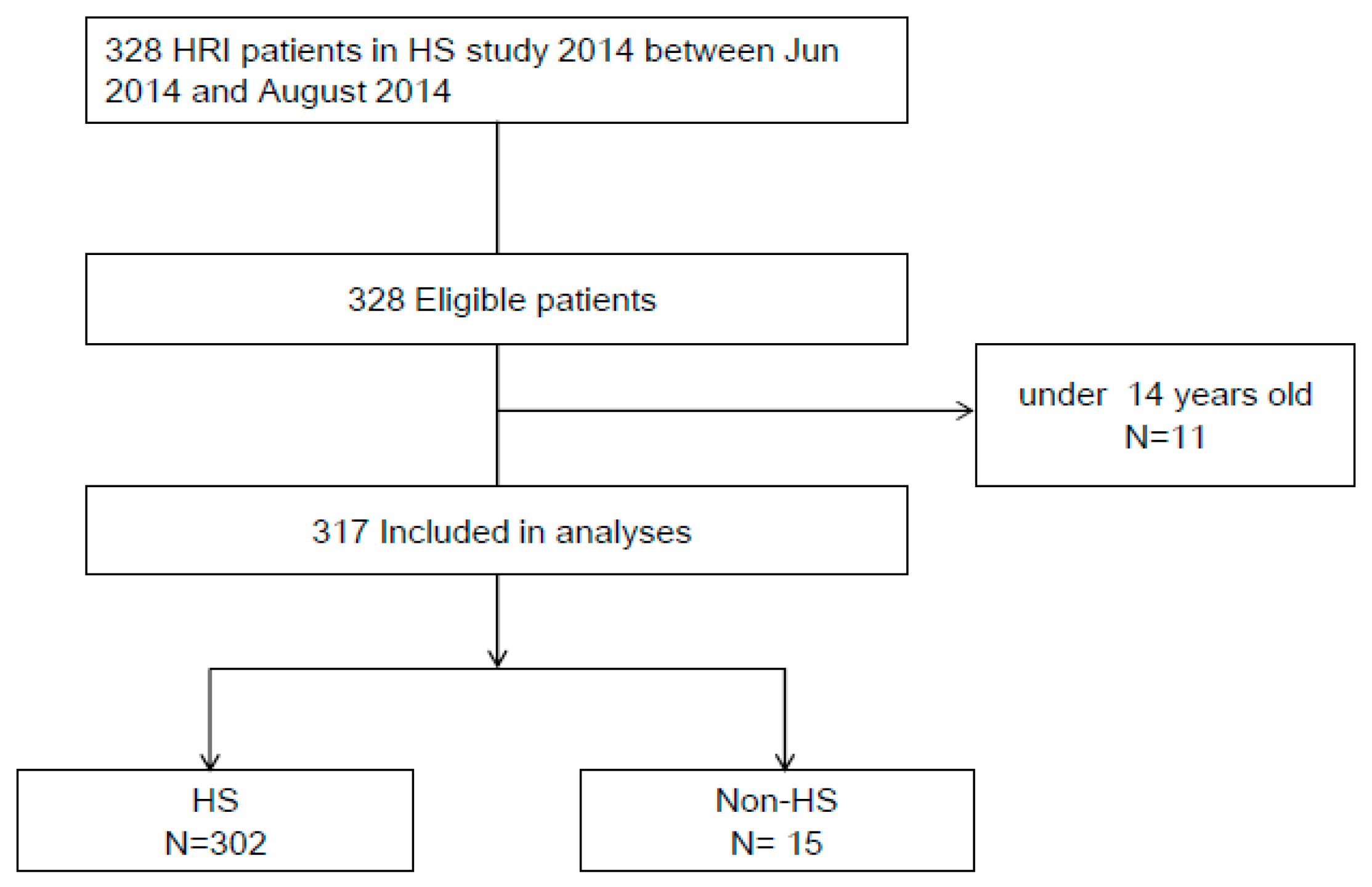
2.1. Study Design and Patients’ Enrolment
2.2. Definitions
2.3. Data Sampling
2.4. Statistical Analysis
3. Results
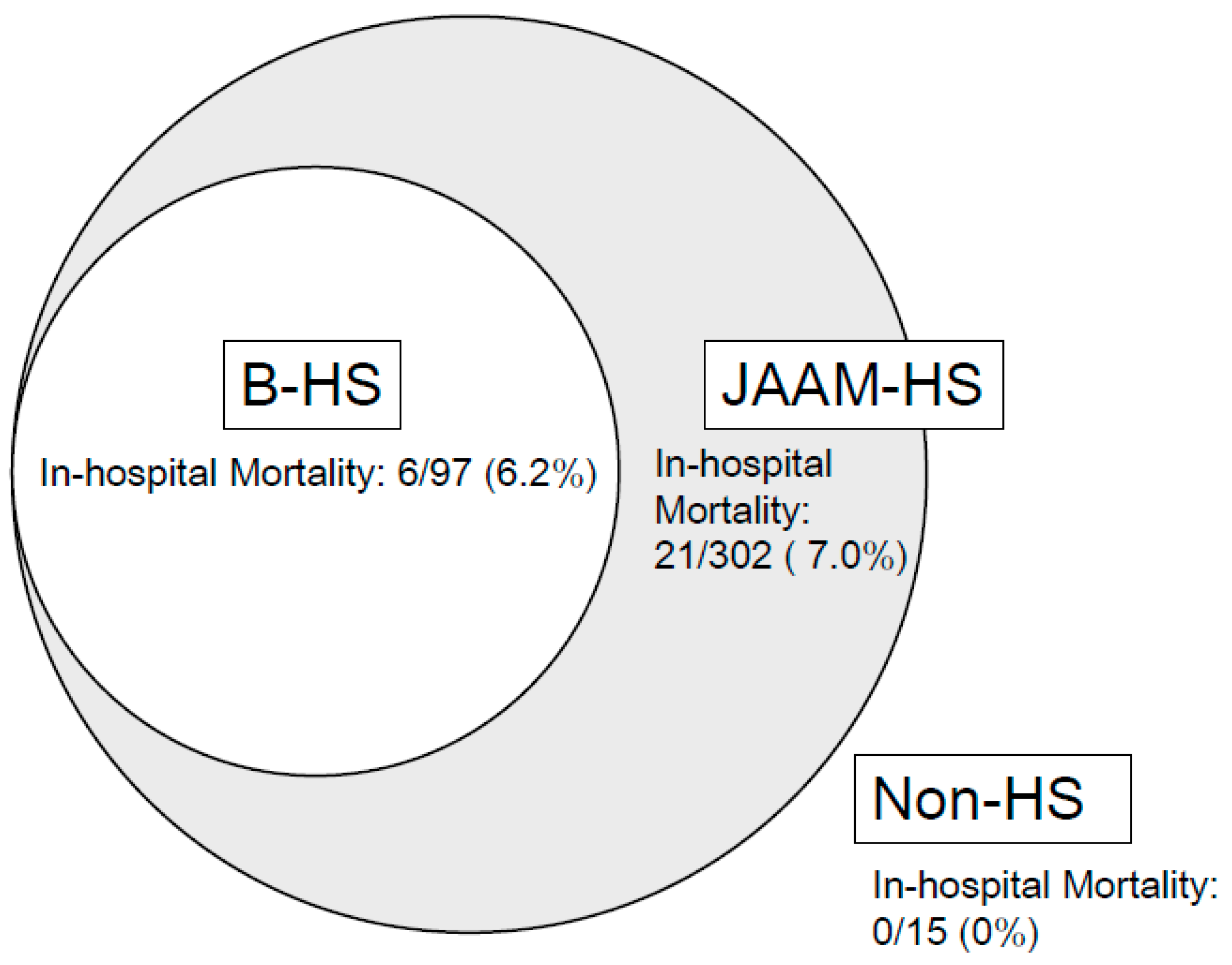
3.1. Baseline Characteristics of The Study Population
3.2. Clinical Manifestations of B-HS, JAAM-HS, and Non-HS Patients
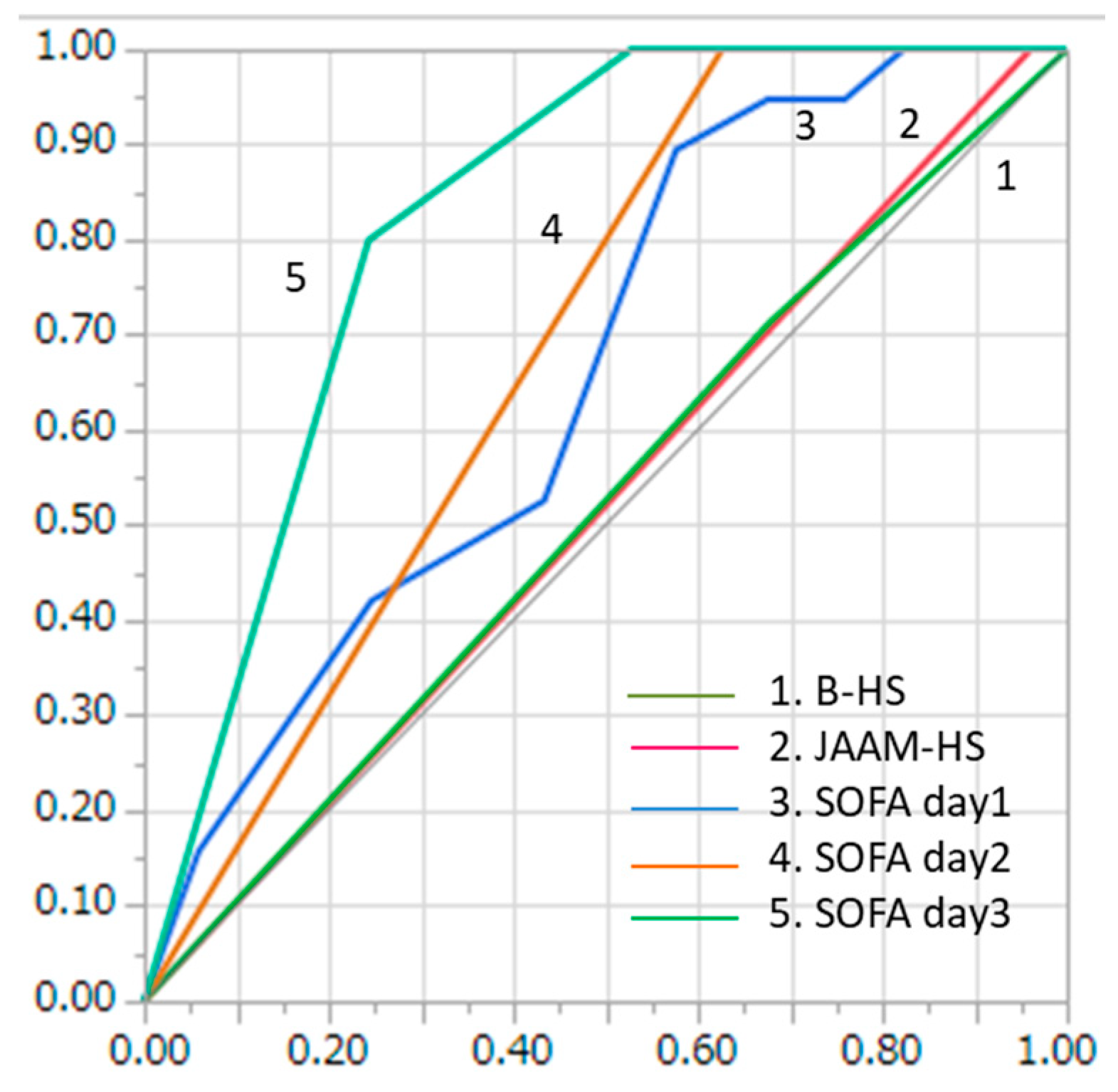
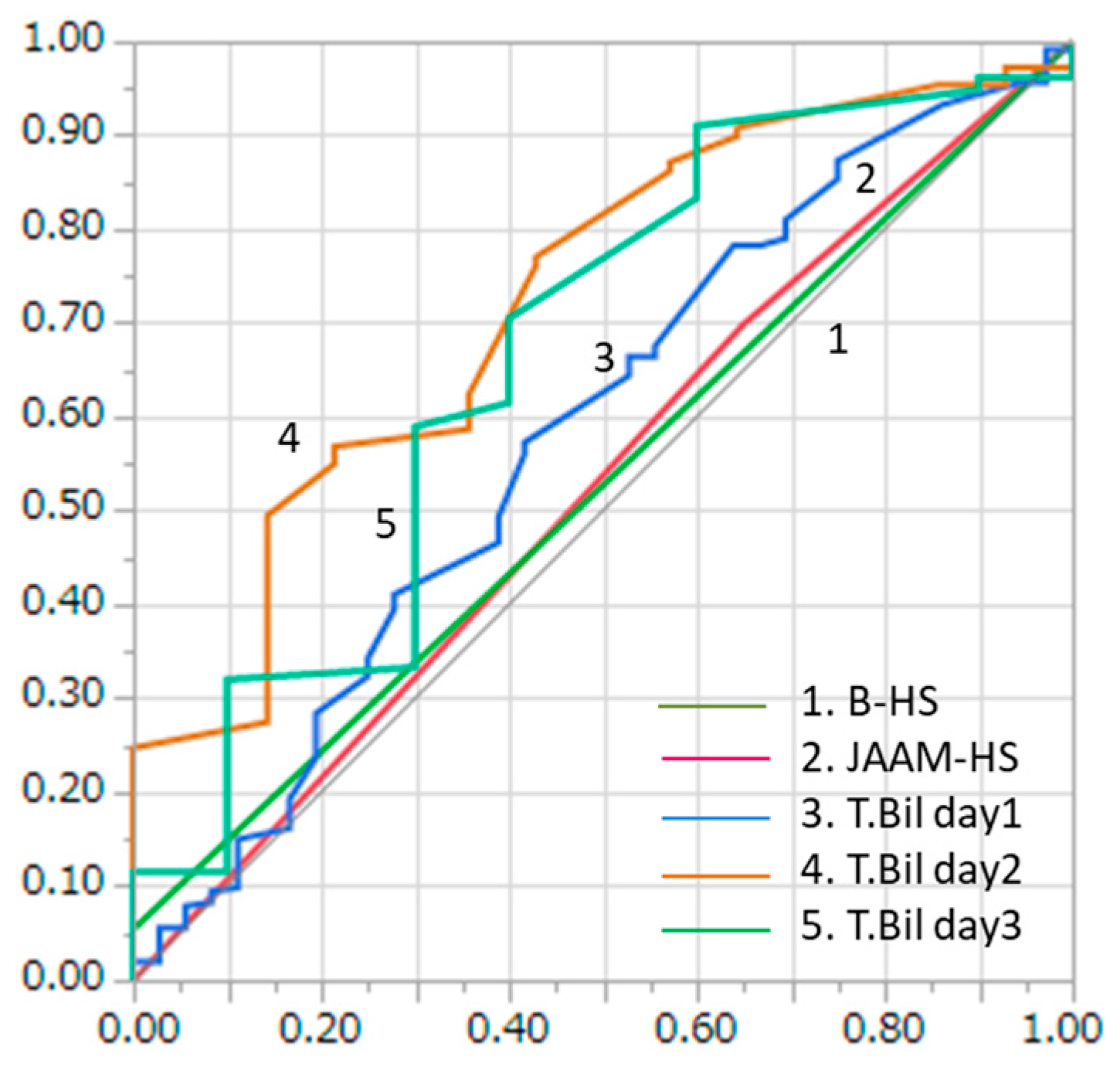
3.3. Prediction of Mortality among HRI Patients
3.4. Prediction of Neurological Function among HRI Patients
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Ethics Approval and Consent to Participate
Abbreviations
| AUC | area under the curve |
| B-HS | Bouchama heatstroke |
| BT | body temperature |
| BUN | blood urea nitrogen |
| CI | confidence interval |
| Cre | creatinine |
| DIC | disseminated intravascular coagulation |
| ED | emergency department |
| FDP | fibrin/fibrinogen degradation product |
| GCS | Glasgow coma score |
| HRI | heat-related illness |
| HS | heatstroke |
| JAAM-HS | Japanese Association for Acute Medicine heatstroke |
| MOF | multiple organ failure |
| mRS | modified Rankin scale |
| PNS | poor neurological status |
| PT | prothrombin time |
| ROC | receiver operating characteristic |
| SOFA | sequential organ |
| T.Bil | total bilirubin |
References
- Wang, Y.; Bobb, J.F.; Papi, B.; Wang, Y.; Kosheleva, A.; Di, Q.; Schwartz, J.D.; Dominici, F. Heat stroke admissions during heat waves in 1,916 US counties for the period from 1999 to 2010 and their effect modifiers. Environ. Health A Glob. Access Sci. Source 2016, 15, 83. [Google Scholar] [CrossRef] [PubMed]
- Argaud, L.; Ferry, T.; Le, Q.H.; Marfisi, A.; Ciorba, D.; Achache, P.; Ducluzeau, R.; Robert, D. Short- and long-term outcomes of heatstroke following the 2003 heat wave in Lyon, France. Arch. Intern. Med. 2007, 167, 2177–2183. [Google Scholar] [CrossRef] [PubMed]
- Jia, Y.A.; Rowlinson, S.; Ciccarelli, M. Climatic and psychosocial risks of heat illness incidents on construction site. Appl. Ergon. 2016, 53, 25–35. [Google Scholar] [CrossRef] [PubMed]
- Bouchama, A.; Knochel, J.P. Heat stroke. N. Engl. J. Med. 2002, 346, 1978–1988. [Google Scholar] [CrossRef] [PubMed]
- Fuse, A.; Yamashiro, K.; Oji, Y.; Furuya, T.; Noda, K.; Hattori, N.; Okuma, Y. Reversible focal cerebral cortical lesions in a patient with heat stroke. Intern. Med. 2013, 52, 377–380. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Carvalho, A.S.; Rodeia, S.C.; Silvestre, J.; Povoa, P. Exertional heat stroke and acute liver failure: A late dysfunction. BMJ Case Rep. 2016, 2016. [Google Scholar] [CrossRef] [PubMed]
- Ramanathan, M.; Pedersen, M.; Ramsey, R.; Seetharam, A. Diagnostic value of coagulation factor and intracranial pressure monitoring in acute liver failure from heat stroke: Case report and review of the literature. Transplant. Proc. 2015, 47, 817–819. [Google Scholar] [CrossRef] [PubMed]
- Savic, S.; Pavlekic, S.; Alempijevic, D.; Dragan, J. Death caused by heat stroke: Case report. Srp. Arh. Celok. Lek. 2014, 142, 360–364. [Google Scholar] [CrossRef] [PubMed]
- Pease, S.; Bouadma, L.; Kermarrec, N.; Schortgen, F.; Regnier, B.; Wolff, M. Early organ dysfunction course, cooling time and outcome in classic heatstroke. Intensive Care Med. 2009, 35, 1454–1458. [Google Scholar] [CrossRef]
- Akieda, K.; Yamamoto, R.; Tamura, K.; Morita, S.; Amino, M.; Sakurai, K.; Otsuka, H.; Motojuku, M.; Inokuchi, S. Successful treatment of a case with acute hepatic failure following hot bath immersion. Tokai J. Exp. Clin. Med. 2008, 33, 65–69. [Google Scholar] [PubMed]
- Hamaya, H.; Hifumi, T.; Kawakita, K.; Okazaki, T.; Kiridume, K.; Shinohara, N.; Abe, Y.; Takano, K.; Hagiike, M.; Kuroda, Y. Successful management of heat stroke associated with multiple-organ dysfunction by active intravascular cooling. Am. J. Emerg. Med. 2015, 33, e5–e7. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, Y.; Sakemi, T.; Nishihara, G.; Nakamura, M.; Fujisaki, T.; Koh, T.; Tomiyoshi, Y.; Emura, S.; Taki, K. Efficacy of blood purification therapy for heat stroke presenting rapid progress of multiple organ dysfunction syndrome: A comparison of five cases. Intensive Care Med. 1999, 25, 315–318. [Google Scholar] [CrossRef] [PubMed]
- Miyake, Y. Characteristics of elderly heat illness patients in Japan--analysis from Heatstroke STUDY 2010. Nihon Rinsho Jpn. J. Clin. Med. 2013, 71, 1065–1073. [Google Scholar]
- Nakamura, S. Sequelae secondary to heat-related illness. Nihon Rinsho Jpn. J. Clin. Med. 2012, 70, 969–974. [Google Scholar]
- Gando, S.; Saitoh, D.; Ishikura, H.; Ueyama, M.; Otomo, Y.; Oda, S.; Kushimoto, S.; Tanjoh, K.; Mayumi, T.; Ikeda, T.; et al. A randomized, controlled, multicenter trial of the effects of antithrombin on disseminated intravascular coagulation in patients with sepsis. Crit. Care 2013, 17, R297. [Google Scholar] [CrossRef]
- Singh, R.K.; Baronia, A.K.; Sahoo, J.N.; Sharma, S.; Naval, R.; Pandey, C.M.; Poddar, B.; Azim, A.; Gurjar, M. Prospective comparison of new Japanese Association for Acute Medicine (JAAM) DIC and International Society of Thrombosis and Hemostasis (ISTH) DIC score in critically ill septic patients. Thromb. Res. 2012, 129, e119–e125. [Google Scholar] [CrossRef]
- Iwai, K.; Uchino, S.; Endo, A.; Saito, K.; Kase, Y.; Takinami, M. Prospective external validation of the new scoring system for disseminated intravascular coagulation by Japanese Association for Acute Medicine (JAAM). Thromb. Res. 2010, 126, 217–221. [Google Scholar] [CrossRef]
- Kushimoto, S.; Gando, S.; Saitoh, D.; Ogura, H.; Mayumi, T.; Koseki, K.; Ikeda, T.; Ishikura, H.; Iba, T.; Ueyama, M.; et al. Clinical course and outcome of disseminated intravascular coagulation diagnosed by Japanese Association for Acute Medicine criteria. Comparison between sepsis and trauma. Thromb. Haemost. 2008, 100, 1099–1105. [Google Scholar]
- Quinn, T.J.; Dawson, J.; Walters, M.R.; Lees, K.R. Reliability of the modified Rankin Scale: A systematic review. Stroke 2009, 40, 3393–3395. [Google Scholar] [CrossRef]
- Cincura, C.; Pontes-Neto, O.M.; Neville, I.S.; Mendes, H.F.; Menezes, D.F.; Mariano, D.C.; Pereira, I.F.; Teixeira, L.A.; Jesus, P.A.; De Queiroz, D.C.; et al. Validation of the National Institutes of Health Stroke Scale, modified Rankin Scale and Barthel Index in Brazil: The role of cultural adaptation and structured interviewing. Cerebrovasc. Dis. 2009, 27, 119–122. [Google Scholar] [CrossRef]
- Sulter, G.; Steen, C.; De Keyser, J. Use of the Barthel index and modified Rankin scale in acute stroke trials. Stroke 1999, 30, 1538–1541. [Google Scholar] [CrossRef] [PubMed]
- Rankin, J. Cerebral vascular accidents in patients over the age of 60. II. Prognosis. Scott. Med. J. 1957, 2, 200–215. [Google Scholar] [CrossRef] [PubMed]
- Cheng, B.; Forkert, N.D.; Zavaglia, M.; Hilgetag, C.C.; Golsari, A.; Siemonsen, S.; Fiehler, J.; Pedraza, S.; Puig, J.; Cho, T.H.; et al. Influence of stroke infarct location on functional outcome measured by the modified rankin scale. Stroke 2014, 45, 1695–1702. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Xue, Y.; Tan, M.T.; Chen, P. Efficient statistical tests to compare Youden index: Accounting for contingency correlation. Stat. Med. 2015, 34, 1560–1576. [Google Scholar] [CrossRef] [PubMed]
- Xu, T.; Wang, J.; Fang, Y. A model-free estimation for the covariate-adjusted Youden index and its associated cut-point. Stat. Med. 2014, 33, 4963–4974. [Google Scholar] [CrossRef] [PubMed]
- Inayat, F.; Virk, H.U. Liver Transplantation after Exertional Heatstroke-Induced Acute Liver Failure. Cureus 2016, 8, e768. [Google Scholar] [CrossRef] [PubMed]
- Lim, C.L.; Mackinnon, L.T. The roles of exercise-induced immune system disturbances in the pathology of heat stroke: The dual pathway model of heat stroke. Sports Med. 2006, 36, 39–64. [Google Scholar] [CrossRef]
- Phillips, N.A.; Welc, S.S.; Wallet, S.M.; King, M.A.; Clanton, T.L. Protection of intestinal injury during heat stroke in mice by interleukin-6 pretreatment. J. Physiol. 2015, 593, 739–752. [Google Scholar] [CrossRef]
- Casa, D.J.; Armstrong, L.E.; Kenny, G.P.; O’Connor, F.G.; Huggins, R.A. Exertional heat stroke: New concepts regarding cause and care. Curr. Sports Med. Rep. 2012, 11, 115–123. [Google Scholar] [CrossRef]
- Armstrong, L.E.; Casa, D.J.; Millard-Stafford, M.; Moran, D.S.; Pyne, S.W.; Roberts, W.O. American College of Sports Medicine position stand. Exertional heat illness during training and competition. Med. Sci. Sports Exerc. 2007, 39, 556–572. [Google Scholar] [CrossRef]
- Misset, B.; De Jonghe, B.; Bastuji-Garin, S.; Gattolliat, O.; Boughrara, E.; Annane, D.; Hausfater, P.; Garrouste-Orgeas, M.; Carlet, J. Mortality of patients with heatstroke admitted to intensive care units during the 2003 heat wave in France: A national multiple-center risk-factor study. Crit. Care Med. 2006, 34, 1087–1092. [Google Scholar] [CrossRef] [PubMed]
- Yankelson, L.; Sadeh, B.; Gershovitz, L.; Werthein, J.; Heller, K.; Halpern, P.; Halkin, A.; Adler, A.; Steinvil, A.; Viskin, S. Life-threatening events during endurance sports: Is heat stroke more prevalent than arrhythmic death? J. Am. Coll. Cardiol. 2014, 64, 463–469. [Google Scholar] [CrossRef] [PubMed]
- Zeller, L.; Novack, V.; Barski, L.; Jotkowitz, A.; Almog, Y. Exertional heatstroke: Clinical characteristics, diagnostic and therapeutic considerations. Eur. J. Intern. Med. 2011, 22, 296–299. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, F.L.; Bota, D.P.; Bross, A.; Melot, C.; Vincent, J.L. Serial evaluation of the SOFA score to predict outcome in critically ill patients. JAMA 2001, 286, 1754–1758. [Google Scholar] [CrossRef] [PubMed]
- Safari, S.; Shojaee, M.; Rahmati, F.; Barartloo, A.; Hahshemi, B.; Forouzanfar, M.M.; Mohammadi, E. Accuracy of SOFA score in prediction of 30-day outcome of critically ill patients. Turk. J. Emerg. Med. 2016, 16, 146–150. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.X.; Wang, J.Y.; Guo, S.B. Use of CRB-65 and quick Sepsis-related Organ Failure Assessment to predict site of care and mortality in pneumonia patients in the emergency department: A retrospective study. Crit. Care 2016, 20, 167. [Google Scholar] [CrossRef]
- Murakami, S.; Miyatake, N.; Sakano, N. Changes in air temperature and its relation to ambulance transports due to heat stroke in all 47 prefectures of Japan. J. Prev. Med. Public Health 2012, 45, 309–315. [Google Scholar] [CrossRef]
| Criteria | Definition |
|---|---|
| B-HS | It is diagnosed by meeting all three below criteria |
| 1. Core temperature > 40 °C | |
| 2. Central nervous system abnormalities (such as delirium, convulsions, or coma) or documented evidence of cooling before the first record temperature | |
| 3. A reliable history of compatible environmental exposure (hot, dry or flushed skin) | |
| JAAM-HS | Physician suspect heat disorder and show one or more below criteria |
| 1. Central nervous system abnormalities (GCS ≤ 14) | |
| 2. Hepatic/Renal dysfunction (T.Bil ≥ 1.2, Cre ≥ 1.2) | |
| 3. Coagulation disorder (DIC, JAAM criteria *) | |
| Variables | B-HS | JAAM-HS | Non-HS | p-Value |
|---|---|---|---|---|
| Age | 71 (52–82) | 66 (41–81) | 20 (15–53) | <0.001 *** |
| Gender (male/female) | 66/31 | 217/85 | 12/3 | 0.76 |
| sBP (mmHg) | 121 (102.8–145.3) | 123 (107–143) | 138 (127–147) | 0.20 |
| dBP (mmHg) | 70 (57–81) | 71 (62–87) | 75 (70–85) | 0.26 |
| HR (bpm) | 114 (82.5–141.5) | 98 (77–120) | 78 (68–103) | 0.04 * |
| RR (/min) | 24 (20–35) | 21 (17–30) | 18 (15–24) | 0.12 |
| SpO2 (%) | 98 (96–99) | 98 (96–99) | 98 (97–99) | 0.4 |
| BT (°C, On-scene) | 40.1 (38.2–41) | 38.3 (36.7–40) | 37.0 (36.6–39.7) | 0.17 |
| BT (°C, ER, superficial) | 39.1 (37.3–40.3) | 37.1 (36.4–38.7) | 36.8 (36.4–37.6) | 0.28 |
| BT (°C, ER, core body) | 40.1 (38.8–41) | 38.8 (37.4–40.1) | 38.8 (37.2–40) | 0.82 |
| GCS | 10 (3–15) | 14 (10–15) | 15 | <0.001 *** |
| pH | 7.43 (7.38–7.48) | 7.42 (7.38–7.46) | 7.46 (7.42–7.48) | 0.35 |
| PaO2 (mmHg) | 109.7 (72–186.9) | 94.8 (67.1–148.5) | 65.2 (32–94.5) | 0.28 |
| PaCO2 (mmHg) | 32.2 (25.8–39.1) | 35.3 (32.5–39.9) | 35.3 (32.5–39.9) | 0.99 |
| WBC (/μL) | 10,200 (7400–13,420) | 10,415 (7882.5–14,000) | 9450 (6260–10,185) | 0.08 |
| Hgb (g/dl) | 13.6 (11.8–15.4) | 14.3 (12.5–16.3) | 14.8 (13.2–15.6) | 0.76 |
| Hct (%) | 40.4 (35.3–45.9) | 42.5 (37.1–47.3) | 42.8 (37.7–44.2) | 0.19 |
| Plt (×103/μL) | 20.1 (15.8–24.9) | 22.1 (4.6–50.8) | 20.3 (19.7–35.7) | 0.75 |
| BUN (mg/dl) | 23.2 (17.6–34.6) | 24.5 (18–34) | 14.4 (10.1–19.2) | 0.04 * |
| Cre (mg/dl) | 1.41 (1.04–2.02) | 1.51 (1.03–2.29) | 0.81 (0.65–0.97) | <0.01 ** |
| T.Bil (mg/dl) | 0.9 (0.7–1.4) | 0.9 (0.7–1.3) | 0.8 (0.7–0.9) | 0.24 |
| AST (U/L) | 46 (28–107) | 33 (26–62.5) | 27 (21–40) | 0.33 |
| ALT (U/L) | 30 (18–70) | 25 (17–51) | 15 (13–35) | 0.45 |
| CK (U/L) | 337 (142–853) | 290.5 (133.5–621.3) | 166 (107.5–259) | 0.46 |
| Na (mmol/L) | 140 (135–144) | 140 (136–143) | 138.5 (134–140.8) | 0.04 * |
| K (mmol/L) | 4.1 (3.7–4.7) | 4.1 (3.7–4.7) | 3.8 (3.6–4.3) | 0.20 |
| Cl (mmol/L) | 103 (99–108) | 102 (99–106) | 103 (98–107) | 0.33 |
| Glucose (mg/dl) | 160 (124–205) | 145 (51–183) | 115 (100–136) | 0.86 |
| PT ratio | 1.1 (1.03–1.24) | 1.08 (1–1.17) | 1.05 (1.03–1.11) | 0.36 |
| FDP (μg/mL) | 9.3 (3.3–46.7) | 5.15 (2.7–13.8) | 3 (3–3.75) | 0.35 |
| D-dimer (μg/mL) | 5 (1.2–17.9) | 2.2 (0.8–7.65) | 0.6 (0.5–4.7) | 0.35 |
| AT Ⅲ (%) | 97.5 (80.8–109) | 101 (86.9–111.4) | 95.5 (84.3–142) | 0.5 |
| DIC score | 1 (0–4) | 0 (0–1) | 0 (0–1) | 0.14 |
| SOFA score | 5 (2–9) | 3 (2–5) | 0 (0–1) | <0.001 *** |
| median, (interquartile range) | JAAM-HS vs. non-HS |
| Symptom | B-HS | JAAM-HS | Non-HS | Total | p-Value |
|---|---|---|---|---|---|
| thirsty | 8 (8.2) | 36 (11.9) | 2 (13.3) | 38 (12) | 0.86 |
| muscle pain | 6 (6.2) | 40 (13.2) | 3 (20) | 43 (13.6) | 0.56 |
| muscle cramp | 10 (9.7) | 64 (21.2) | 2 (13.3) | 66 (20.8) | 0.46 |
| dizziness | 6 (6.2) | 27 (8.9) | 1 (6.7) | 28 (8.8) | 0.76 |
| general fatigue | 22 (22.7) | 95 (31.5) | 8 (53.3) | 103 (32.5) | 0.08 |
| syncope | 0 (0) | 11 (3.6) | 1 (6.7) | 12 (3.8) | 0.55 |
| headache | 5 (5.2) | 19 (6.3) | 5 (33.3) | 24 (7.6) | <0.001 *** |
| agitation | 6 (6.2) | 14 (4.6) | 1 (6.7) | 15 (4.7) | 0.72 |
| sweat | 31 (32) | 113 (37.4) | 8 (53.3) | 121 (38.2) | 0.37 |
| vomit | 14 (14.4) | 52 (17.2) | 3 (20) | 55 (17.4) | 0.78 |
| diarrhea | 2 (2.1) | 8 (2.6) | 0 (0) | 8 (2.5) | 0.52 |
| abdominal pain | 2 (2.1) | 6 (2) | 1 (6.7) | 7 (2.2) | 0.23 |
| incontinence | 5 (5.2) | 9 (3) | 0 (0) | 9 (2.8) | 0.5 |
| N, (%) |
| B-HS | JAAM-HS | |
|---|---|---|
| Mortality, Number (%) | 6/97 (6.2) | 21/302 (7.0) |
| Sensitivity of death (95% CI) | 0.29 (0.14–0.49) | 1.0 (0.87–1.0) |
| Specificity of death (95% CI) | 0.69 (0.68–0.71) | 0.05 (0.04–0.05) |
| LR+ | 0.93 (0.44–1.68) | 1.05 (0.91–1.05) |
| LR− | 1.03 (0.72–1.26) | 0 (0–3.19) |
| Youden index | −0.22 (−0.18–0.2) | 0.05 (−0.09–0.05) |
| AUC | 0.52 | 0.52 |
| OR (95% CI) | 0.90 (0.35–2.33) | Inf |
| B-HS | JAAM-HS | |
|---|---|---|
| PNS ([mRS, 3–6]), Number (%) | 14/97 (14.4) | 40/302 (13.2) |
| Sensitivity of PNS (95% CI) | 0.35 (0.23–0.49) | 1.0 (0.93–1.0) |
| Specificity of PNS (95% CI) | 0.70 (0.68–0.72) | 0.05 (0.04–0.05) |
| LR+ | 1.17 (0.72–1.77) | 1.06 (0.97–1.06) |
| LR− | 0.93 (0.70–1.13) | 0 (0–1.66) |
| Youden index | 0.05 (−0.09–0.21) | 0.05 (−0.03–0.05) |
| AUC | 0.52 | 0.53 |
| OR (95% CI) | 1.26 (0.63–2.51) | Inf |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kondo, Y.; Hifumi, T.; Shimazaki, J.; Oda, Y.; Shiraishi, S.-i.; Hayashida, K.; Fukuda, T.; Wakasugi, M.; Kanda, J.; Moriya, T.; et al. Comparison between the Bouchama and Japanese Association for Acute Medicine Heatstroke Criteria with Regard to the Diagnosis and Prediction of Mortality of Heatstroke Patients: A Multicenter Observational Study. Int. J. Environ. Res. Public Health 2019, 16, 3433. https://doi.org/10.3390/ijerph16183433
Kondo Y, Hifumi T, Shimazaki J, Oda Y, Shiraishi S-i, Hayashida K, Fukuda T, Wakasugi M, Kanda J, Moriya T, et al. Comparison between the Bouchama and Japanese Association for Acute Medicine Heatstroke Criteria with Regard to the Diagnosis and Prediction of Mortality of Heatstroke Patients: A Multicenter Observational Study. International Journal of Environmental Research and Public Health. 2019; 16(18):3433. https://doi.org/10.3390/ijerph16183433
Chicago/Turabian StyleKondo, Yutaka, Toru Hifumi, Junya Shimazaki, Yasutaka Oda, Shin-ichiro Shiraishi, Kei Hayashida, Tatsuma Fukuda, Masahiro Wakasugi, Jun Kanda, Takashi Moriya, and et al. 2019. "Comparison between the Bouchama and Japanese Association for Acute Medicine Heatstroke Criteria with Regard to the Diagnosis and Prediction of Mortality of Heatstroke Patients: A Multicenter Observational Study" International Journal of Environmental Research and Public Health 16, no. 18: 3433. https://doi.org/10.3390/ijerph16183433
APA StyleKondo, Y., Hifumi, T., Shimazaki, J., Oda, Y., Shiraishi, S.-i., Hayashida, K., Fukuda, T., Wakasugi, M., Kanda, J., Moriya, T., Yagi, M., Kawahara, T., Tonouchi, M., Yokobori, S., Yokota, H., Miyake, Y., & Shimizu, K. (2019). Comparison between the Bouchama and Japanese Association for Acute Medicine Heatstroke Criteria with Regard to the Diagnosis and Prediction of Mortality of Heatstroke Patients: A Multicenter Observational Study. International Journal of Environmental Research and Public Health, 16(18), 3433. https://doi.org/10.3390/ijerph16183433





