Impact of Carers’ Smoking Status on Childhood Obesity in the Growing up in Ireland Cohort Study
Abstract
1. Introduction
2. Materials
2.1. Participants
2.2. Data Collection Procedures
2.3. Outcome Measures: Childhood Obesity
2.4. Exposure: Carers’ Smoking Status
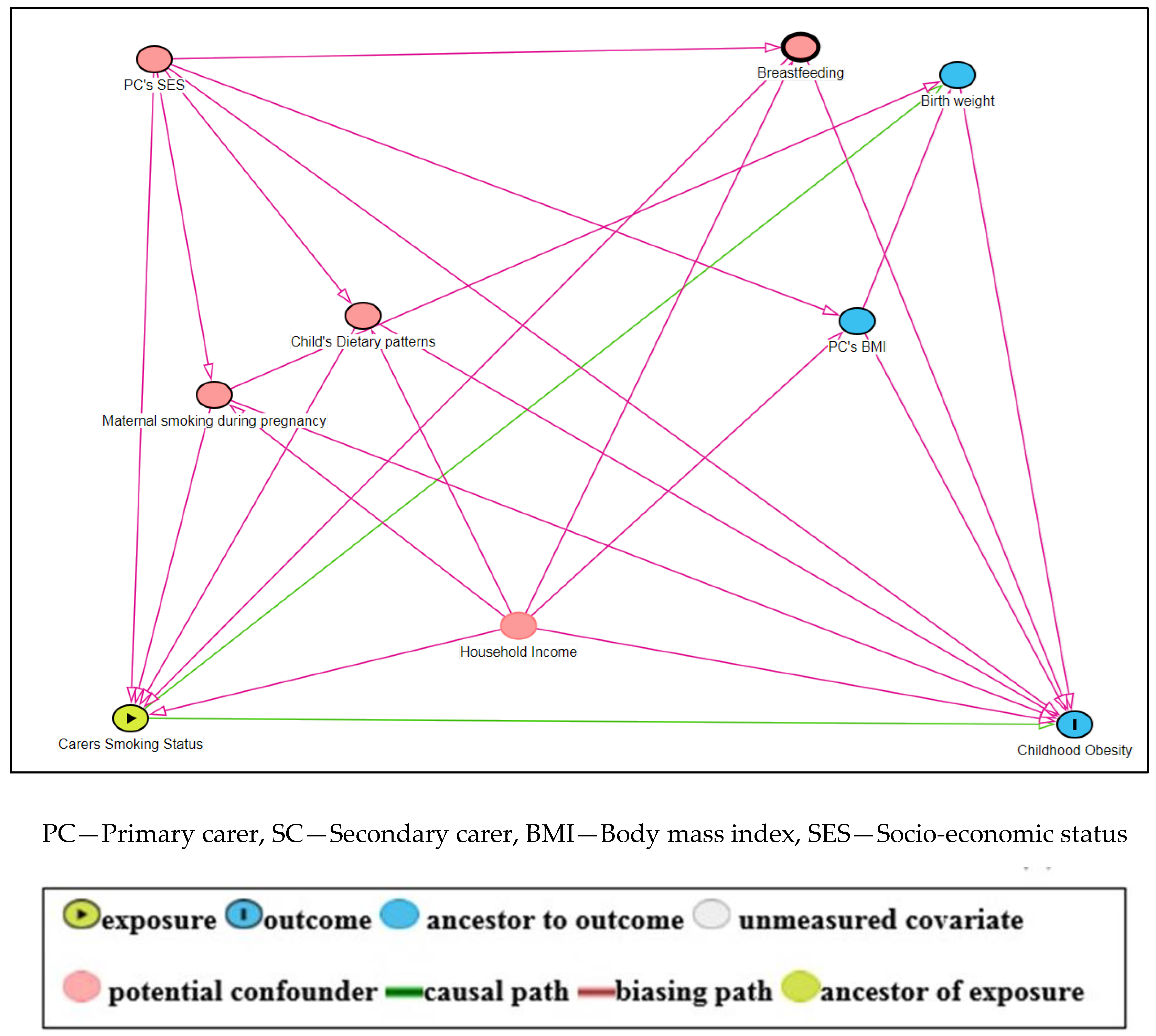
2.5. Potential Confounders
2.6. Statistical Analysis
3. Results
3.1. Overview of Infant and Carers’ Characteristics and Smoking Status
3.2. Obesity Estimates in Children at Age Three (Wave 2) and at Age Five (Wave 3)
4. Discussion
4.1. Strengths
4.2. Limitations
4.3. Future Recommendations and Policy Implications
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Primary Carers’ Smoking | Wave 2 (9793) | Wave 3 (9001) |
|---|---|---|
| Yes | 2558 (26.4%) | 2069 (23.4%) |
| No | 7149 (73.6%) | 6784 (76.6%) |
| Prevalence of Overweight/Obesity | ||
|---|---|---|
| Wave 2 | Wave 3 | |
| Normal | 7267 (65.3%) | 7066 (79.7%) |
| Overweight | 1752 (15.7%) | 1353 (15.3%) |
| Obese | 514 (4.6%) | 445 (5.0%) |
| Carers’ Smoking at Three Years | Risk of Overweight or Obesity at Three Years | ||
| OR (95%CI) | 95% CI | p | |
| Primary Carer Smoking | 1.47 | 1.23, 1.76 | 0.00 |
| Carers’ Smoking at Five Years | Risk of Overweight or Obesity at Five Years | ||
| OR (95%CI) | 95% CI | p | |
| Primary Carer Smoking | 1.48 | 1.22, 1.82 | 0.00 |
References
- World Health Organization. Childhood Overweight and Obesity; World Health Organization: Geneva, Switzerland, 2018; Available online: http://www.who.int/dietphysicalactivity/childhood/en/ (accessed on 25 July 2019).
- Ng, M.; Fleming, T.; Robinson, M.; Thomson, B.; Graetz, N.; Margono, C.; Mullany, E.C.; Biryukov, S.; Abbafati, C.; Abera, S.F.; et al. Global, regional, and national prevalence of overweight and obesity in children and adults during 1980–2013: A systematic analysis for the Global Burden of Disease Study 2013. Lancet 2014, 384, 766–781. [Google Scholar] [CrossRef]
- Van Jaarsveld, C.H.; Gulliford, M.C. Childhood obesity trends from primary care electronic health records in England between 1994 and 2013: Population-based cohort study. Arch. Dis. Child. 2015, 100, 214–219. [Google Scholar] [CrossRef]
- Ogden, C.L.; Carroll, M.D.; Kit, B.K.; Flegal, K.M. Prevalence of Childhood and Adult Obesity in the United States, 2011–2012. JAMA 2014, 311, 806–814. [Google Scholar] [CrossRef] [PubMed]
- Olds, T.S.; Tomkinson, G.R.; Ferrar, K.; Maher, C. Trends in the prevalence of childhood overweight and obesity in Australia between 1985 and 2008. Int. J. Obes. 2009, 34, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Heinen, M.M.; Concannon, M.; Farrell, D.; Flood, C.; Mehegan, J.; Murrin, C.M.; Kelleher, C.C. Overweight and obesity trends are stabilizing among children aged 7 years: Results of the Childhood Obesity Surveillance Initiative in the Republic of Ireland. Proc. Nutr. Soc. 2016, 75. [Google Scholar] [CrossRef]
- Bel-Serrat, S.; Heinen, M.M.; Murrin, C.M.; Daly, L.; Mehegan, J.; Concannon, M.; Flood, C.; Farrell, D.; O’Brien, S.; Eldin, N.; et al. The Childhood Obesity Surveillance Initiative (COSI) in the Republic of Ireland: Findings from 2008, 2010, 2012 and 2015; Health Service Executive: Dublin, Ireland, 2017. [Google Scholar]
- Lakshman, R.; Elks, C.; Ong, K. Childhood Obesity. Circulation 2012, 126, 1770–1779. [Google Scholar] [CrossRef]
- World Health Organization. Commission on Ending Childhood Obesity; World Health Organization: Geneva, Switzerland, 2018; Available online: http://www.who.int/en/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 9 January 2019).
- Lissau, I. Overweight and obesity epidemic among children. Answer from European countries. Int. J. Obes. 2004, 28, S10–S15. [Google Scholar] [CrossRef][Green Version]
- Whelton, H.; Harrington, J.; Crowley, E.; Kelleher, V.; Cronin, M.; Perry, I. Prevalence of overweight and obesity on the island of Ireland: Results from the North South Survey of Children’s Height, Weight and Body Mass Index, 2002. BMC Public Health 2007, 7, 187. [Google Scholar] [CrossRef] [PubMed]
- Kabir, Z.; Manning, P.; Holohan, J.; Goodman, P.; Clancy, L. Active smoking and second-handsmoke exposure at home among Irish children, 1995–2007. Arch. Dis. Child. 2009, 95, 42–45. [Google Scholar] [CrossRef]
- Lobstein, T.; Baur, L.; Uauy, R. Obesity in children and young people: A crisis in public health. Obes. Rev. 2004, 5, 4–85. [Google Scholar] [CrossRef]
- Health Gov.ie. 2019. Available online: https://health.gov.ie/wp-content/uploads/2014/03/HealthyIrelandBrochureWA2.pdf (accessed on 14 July 2019).
- Khan, L.K.; Freedman, D.S.; Serdula, M.K.; Dietz, W.H.; Srinivasan, S.R.; Berenson, G.S. The Relation of Childhood BMI to Adult Adiposity: The Bogalusa Heart Study. Pediatrics 2005, 115, 22–27. [Google Scholar]
- Stateofobesity Org. Obesity Rates & Trends Overview—The State of Obesity. 2018. Available online: https://stateofobesity.org/obesity-rates-trends-overview/ (accessed on 9 January 2019).
- Rutter, H.; Savona, N.; Glonti, K.; Bibby, J.; Cummins, S.; Finegood, D.T.; Greaves, F.; Harper, L.; Hawe, P.; Moore, L.; et al. The need for a complex systems model of evidence for public health. Lancet 2017, 390, 2602–2604. [Google Scholar] [CrossRef]
- Behl, M.; Rao, D.; Aagaard, K.; Davidson, T.L.; Levin, E.D.; Slotkin, T.A.; Srinivasan, S.; Wallinga, D.; White, M.F.; Walker, V.R.; et al. Evaluation of the Association between Maternal Smoking, Childhood Obesity, and Metabolic Disorders: A National Toxicology Program Workshop Review. Environ. Health Perspect. 2012, 121, 170–180. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Report of a Joint WHO/FAO Expert Consultation. In Diet Nutrition and the Prevention of Chronic Diseases; WHO Technical Report Series no. 916; WHO: Geneva, Switzerland, 2002. [Google Scholar]
- Central Statistics Office. Quarterly National Household Survey—Childcare. In First Quarter 2005; Stationery Office: Dublin, Ireland, 2006. [Google Scholar]
- Berman, B.; Wong, G.C.; Bastani, R.; Hoang, T.; Jones, C.; Goldstein, D.R.; Bernert, J.; Hammond, K.S.; Tashkin, D.; Lewis, M.A. Household smoking behavior and ETS exposure among children with asthma in low-income, minority households. Addict. Behav. 2003, 28, 111–128. [Google Scholar] [CrossRef]
- Hilliard, M.E.; Riekert, K.A.; Hovell, M.F.; Rand, C.S.; Welkom, J.S.; Eakin, M.N. Family Beliefs and Behaviors About Smoking and Young Children’s Secondhand Smoke Exposure. Nicotine Tob. Res. 2014, 17, 1067–1075. [Google Scholar] [CrossRef] [PubMed]
- Squillacioti, G.; Bellisario, V.; Grignani, E.; Mengozzi, G.; Bardaglio, G.; Dalmasso, P.; Bono, R. The Asti Study: The Induction of Oxidative Stress in A Population of Children According to Their Body Composition and Passive Tobacco Smoking Exposure. Int. J. Environ. Res. Public Health 2019, 16, 490. [Google Scholar] [CrossRef] [PubMed]
- Ng, M.; Freeman, M.K.; Fleming, T.D.; Robinson, M.; Dwyer-Lindgren, L.; Thomson, B.; Wollum, A.; Sanman, E.; Wulf, S.; Lopez, A.D.; et al. Smoking Prevalence and Cigarette Consumption in 187 Countries, 1980–2012. JAMA 2014, 311, 183. [Google Scholar] [CrossRef] [PubMed]
- Marcon, A.; Pesce, G.; Calciano, L.; Bellisario, V.; Dharmage, S.C.; Garcia-Aymerich, J.; Gislasson, T.; Heinrich, J.; Holm, M.; Janson, C.; et al. Trends in smoking initiation in Europe over 40 years: A retrospective cohort study. PLoS ONE 2018, 13, e0201881. [Google Scholar] [CrossRef]
- WHO. Exposure to Second-Hand Smoke, Who Int. 2018. Available online: http://www.who.int/gho/phe/secondhand_smoke/exposure/en/. (accessed on 25 July 2019).
- Öberg, M.; Jaakkola, M.S.; Woodward, A.; Peruga, A.; Prüss-Ustün, A. Worldwide burden of disease from exposure to second-hand smoke: A retrospective analysis of data from 192 countries. Lancet 2011, 377, 139–146. [Google Scholar] [CrossRef]
- Thayer, K.; Heindel, J.; Bucher, J.; Gallo, M. Role of Environmental Chemicals in Diabetes and Obesity: A National Toxicology Program Workshop Review. Environ. Health Perspect. 2012, 120, 779–789. [Google Scholar] [CrossRef]
- Yang, S.; Decker, A.; Kramer, M.S. Exposure to parental smoking and child growth and development: A cohort study. BMC Pediatr. 2013, 13, 104. [Google Scholar] [CrossRef] [PubMed]
- Robinson, O.; Martínez, D.; Aurrekoetxea, J.J.; Estarlich, M.; Somoano, A.F.; Íñiguez, C.; Santa-Marina, L.; Tardón, A.; Torrent, M.; Sunyer, J.; et al. The association between passive and active tobacco smoke exposure and child weight status among Spanish children. Obesity 2016, 24, 1767–1777. [Google Scholar] [CrossRef] [PubMed]
- McConnell, R.; Shen, E.; Gilliland, F.D.; Jerrett, M.; Wolch, J.; Chang, C.C.; Lurmann, F.; Berhane, K. A Longitudinal Cohort Study of Body Mass Index and Childhood Exposure to Secondhand Tobacco Smoke and Air Pollution: The Southern California Children’s Health Study. Environ. Health Perspect. 2014, 123, 360–366. [Google Scholar] [CrossRef] [PubMed]
- Møller, S.E.; Ajslev, T.A.; Andersen, C.S.; Dalgård, C.; Sørensen, T.I.A. Risk of Childhood Overweight after Exposure to Tobacco Smoking in Prenatal and Early Postnatal Life. PLoS ONE 2014, 9, e109184. [Google Scholar] [CrossRef] [PubMed]
- Beyerlein, A.; Rückinger, S.; Toschke, A.M.; Rosario, A.S.; Von Kries, R. Is low birth weight in the causal pathway of the association between maternal smoking in pregnancy and higher BMI in the offspring? Eur. J. Epidemiol. 2011, 26, 413–420. [Google Scholar] [CrossRef] [PubMed]
- Health Gov.ie. 2019. Available online: https://health.gov.ie/wp-content/uploads/2016/12/DoH-SoS-2016-2019-Final-En.pdf (accessed on 15 July 2019).
- Quail, A.; Williams, J.; McCrory, C.; Murray, A.; Thornton, M. A Summary Guide to Wave 1 of the Infant Cohort (at 9 Months) of Growing Up in Ireland Ucd.ie. 2011. Available online: http://www.ucd.ie/issda/static/documentation/esri/GUISummaryGuideInfants.pdf (accessed on 22 July 2019).
- Quail, A.; Williams, J.; McCrory, C.; Murray, A.; Thornton, M. A Summary Guide to Wave 2 of the Infant Cohort (at 9 Months) of Growing Up in Ireland Ucd.ie. 2011. Available online: https://www.ucd.ie/t4cms/SUMMARY%20GUIDE%20%20INFANT%20-%20WAVE%202.pdf (accessed on 22 July 2019).
- Quail, A.; Williams, J.; McCrory, C.; Murray, A.; Thornton, M. A Summary Guide to Wave 3 of the Infant Cohort (at 9 Months) of Growing Up in Ireland Ucd.ie. 2011. Available online: https://www.ucd.ie/t4cms/Summary%20Guide%20%20Infant%20Cohort%20Wave%203.pdf (accessed on 22 July 2019).
- Layte, R.; McCrory, C. Maternal Health Behaviors and Child Growth in Infancy Analyses of the Infant Cohort of the Growing Up in Ireland Study. 2014. Available online: https://www.esri.ie/pubs/BKMNEXT286.pdf (accessed on 19 July 2019).
- World Obesity Federation. New Child Cut-Offs Worldobesity org. 2018. Available online: https://www.worldobesity.org/data/cut-pointsused/newchildcutoffs/ (accessed on 2 August 2019).
- Cole, T.J.; Lobstein, T. Extended international (IOTF) body mass index cut-offs for thinness, overweight and obesity. Pediatr. Obes. 2012, 7, 284–294. [Google Scholar] [CrossRef]
- Hofhuis, W.; De Jongste, J.C.; Merkus, P.J.F.M. Adverse health effects of prenatal and postnatal tobacco smoke exposure on children. Arch. Dis. Child. 2003, 88, 1086–1090. [Google Scholar] [CrossRef] [PubMed]
- Maughan, B. Unravelling prenatal influences: The case of smoking in pregnancy. Int. J. Epidemiol. 2009, 38, 619–621. [Google Scholar] [CrossRef][Green Version]
- Textor, J.; van der Zander, B.; Gilthorpe, M.S.; Liśkiewicz, M.; Ellison, G.T. Robust causal inference using directed acyclic graphs: The R package ‘dagitty’. Int. J. Epidemiol. 2016, 45, 1887–1894. [Google Scholar] [CrossRef] [PubMed]
- Raum, E.; Lamerz, A.; Hebebrand, J.; Brenner, H.; Küpper-Nybelen, J.; Herpertz-Dahlmann, B. Tobacco Smoke Exposure Before, During, and After Pregnancy and Risk of Overweight at Age 6. Obesity 2011, 19, 2411–2417. [Google Scholar] [CrossRef]
- Danielzik, S.; Czerwinski-Mast, M.; Langnäse, K.; Dilba, B.; Müller, M.J. Parental overweight, socioeconomic status and high birth weight are the major determinants of overweight and obesity in 5–7 y-old children: Baseline data of the Kiel Obesity Prevention Study (KOPS). Int. J. Obes. 2004, 28, 1494–1502. [Google Scholar] [CrossRef] [PubMed]
- Florath, I.; Kohler, M.; Weck, M.N.; Brandt, S.; Rothenbacher, D.; Schöttker, B.; Mos, A.; Gottmann, P.; Wabitsch, M.; Brenner, H. Association of pre- and post-natal parental smoking with offspring body mass index: An 8-year follow-up of a birth cohort. Pediatr. Obes. 2013, 9, 121–134. [Google Scholar] [CrossRef] [PubMed]
- Pagani, L.S.; Nguyen, A.K.D.; Fitzpatrick, C. Prospective Associations Between Early Long-Term Household Tobacco Smoke Exposure and Subsequent Indicators of Metabolic Risk at Age 10. Nicotine Tob. Res. 2015, 18, 1250–1257. [Google Scholar] [CrossRef] [PubMed]
- Steur, M.; Smit, H.A.; Schipper, C.M.A.; Scholtens, S.; Kerkhof, M.; De Jongste, J.C.; Haveman-Nies, A.; Brunekreef, B.; Wijga, A.H. Predicting the risk of newborn children to become overweight later in childhood: The PIAMA birth cohort study. Pediatr. Obes. 2011, 6, e170–e178. [Google Scholar] [CrossRef] [PubMed]
- Harris, H.R.; Willett, W.C.; Michels, K.B. Parental smoking during pregnancy and risk of overweight and obesity in the daughter. Int. J. Obes. 2013, 37, 1356–1363. [Google Scholar] [CrossRef] [PubMed]
- Durmuş, B.; Kruithof, C.J.; Gillman, M.H.; Willemsen, S.P.; Hofman, A.; Raat, H.; Eilers, P.H.; Ap Steegers, E.; Jaddoe, V.W. Parental smoking during pregnancy, early growth, and risk of obesity in preschool children: The Generation R Study. Am. J. Clin. Nutr. 2011, 94, 164–171. [Google Scholar] [CrossRef] [PubMed]
- Nagelhout, G.E.; De Vries, H.; Boudreau, C.; Allwright, S.; McNeill, A.; Putte, B.V.D.; Fong, G.T.; Willemsen, M.C. Comparative impact of smoke-free legislation on smoking cessation in three European countries. Eur. J. Public Health 2012, 22 (Suppl. 1), 4–9. [Google Scholar] [CrossRef] [PubMed]
- Mak, Y.W.; Loke, A.Y.; Lam, T.H.; Abdullah, A.S.M. Validity of self-reports and reliability of spousal proxy reports on the smoking behavior of Chinese parents with young children. Addict. Behav. 2005, 30, 841–845. [Google Scholar] [CrossRef] [PubMed]
- Soulakova, J.N.; Hartman, A.M.; Liu, B.; Willis, G.B.; Augustine, S. Reliability of Adult Self-Reported Smoking History: Data from the Tobacco Use Supplement to the Current Population Survey 2002–2003 Cohort. Nicotine Tob. Res. 2012, 14, 952–960. [Google Scholar] [CrossRef]
- Passaro, K.; Noss, J.; Savitz, D.; Little, R. Agreement between self and partner reports of paternal drinking and smoking. The ALSPAC Study Team. Avon Longitudinal Study of Pregnancy and Childhood. Int. J. Epidemiol. 1997, 26, 315–320. [Google Scholar] [CrossRef]
- England, L.; Tong, V.T.; Rockhill, K.; Hsia, J.; McAfee, T.; Patel, D.; Rupp, K.; Conrey, E.J.; Valdivieso, C.; Davis, K.C. Evaluation of a federally funded mass media campaign and smoking cessation in pregnant women: A population-based study in three states. BMJ Open 2017, 7, e016826. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Thornton, J.C.; Russell, M.; Burastero, S.; Heymsfield, S.; Pierson, R.N. Asians have lower body mass index (BMI) but higher percent body fat than do whites: Comparisons of anthropometric measurements. Am. J. Clin. Nutr. 1994, 60, 23–28. [Google Scholar] [CrossRef] [PubMed]
| Variables | Neither Carer Smoked | Only Primary Carer Smoked | Both Carer Smoked | P | |
|---|---|---|---|---|---|
| n = 6037 (%) a | n = 1339 (%) b | n = 1084 (%) c | |||
| Prevalence | 60.5% | 13.4% | 10.9% | ||
| Infant Characteristics | |||||
| Gender | Boy | 3082 (51.1) | 691 (51.6) | 558 (51.5) | 0.973 |
| Girl | 2955 (49.0) | 648 (48.4) | 526 (48.5) | ||
| Birth weight | <2500 | 324 (5.4) | 95 (7.1) | 75 (6.9) | 0.011 |
| ≥2500 | 5713 (94.6) | 1244 (92.9) | 1009 (93.1) | ||
| BMI | 18.3 (2.7) | 18.2 (2.70) | 18.1 ± 2.8 | ||
| Breastfed | Yes No | 4033 (66.8) | 521 (38.9) | 537 (49.5) | <0.001 |
| 2002 (33.2) | 818 (61.1) | 547 (50.5) | |||
| Primary Carers’ Characteristics | |||||
| Mean Age (years) | 32 ± 5.0 | 28 ± 6.0 | 30 ± 5.5 | ||
| BMI | Healthy | 3051 (52.9) | 614 (48.1) | 514 (49.1) | <0.001 |
| Overweight | 1731 (30.0) | 360 (28.2) | 320 (30.6) | ||
| Obese | 872 (15.1) | 236 (18.5) | 166 (15.9) | ||
| Economic status | Pre-school | 0 | 0 | 0 | <0.001 |
| School/Education | 96 (1.6) | 37 (2.8) | 14 (1.3) | ||
| At work/training | 3667 (60.7) | 569 (42.5) | 590 (54.4) | ||
| Unemployed | 181 (3.0) | 97 (7.2) | 48 (4.4) | ||
| Home duties | 2035 (33.7) | 619 (46.2) | 416 (38.4) | ||
| Other | 56 (0.9) | 17 (1.3) | 13 (1.2) | ||
| Primary Carers’ Smoking at Three Years | Risk of Overweight or Obesity at Three Years | ||
| (yes/no) | OR | 95% CI | P |
| Unadjusted Model | 1.37 | 1.24, 1.52 | 0.00 |
| Adjusted Model * | 1.30 | 1.17, 1.46 | 0.00 |
| Primary Carers’ Smoking at Five years | Risk of Overweight or Obesity at Five Years | ||
| (yes/no) | OR | 95% CI | P |
| Unadjusted Model | 1.36 | 1.21, 1.53 | 0.00 |
| Adjusted Model ** | 1.31 | 1.16, 1.49 | 0.00 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sunday, S.; Kabir, Z. Impact of Carers’ Smoking Status on Childhood Obesity in the Growing up in Ireland Cohort Study. Int. J. Environ. Res. Public Health 2019, 16, 2759. https://doi.org/10.3390/ijerph16152759
Sunday S, Kabir Z. Impact of Carers’ Smoking Status on Childhood Obesity in the Growing up in Ireland Cohort Study. International Journal of Environmental Research and Public Health. 2019; 16(15):2759. https://doi.org/10.3390/ijerph16152759
Chicago/Turabian StyleSunday, Salome, and Zubair Kabir. 2019. "Impact of Carers’ Smoking Status on Childhood Obesity in the Growing up in Ireland Cohort Study" International Journal of Environmental Research and Public Health 16, no. 15: 2759. https://doi.org/10.3390/ijerph16152759
APA StyleSunday, S., & Kabir, Z. (2019). Impact of Carers’ Smoking Status on Childhood Obesity in the Growing up in Ireland Cohort Study. International Journal of Environmental Research and Public Health, 16(15), 2759. https://doi.org/10.3390/ijerph16152759






