A Comparison of Flavorless Electronic Cigarette-Generated Aerosol and Conventional Cigarette Smoke on the Survival and Growth of Common Oral Commensal Streptococci
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents and Supplies
2.2. Bacterial Strains
2.3. E-Liquid
2.4. Exposure Apparatus
2.5. Distribution of Aerosol and Smoke in the Exposure Chamber
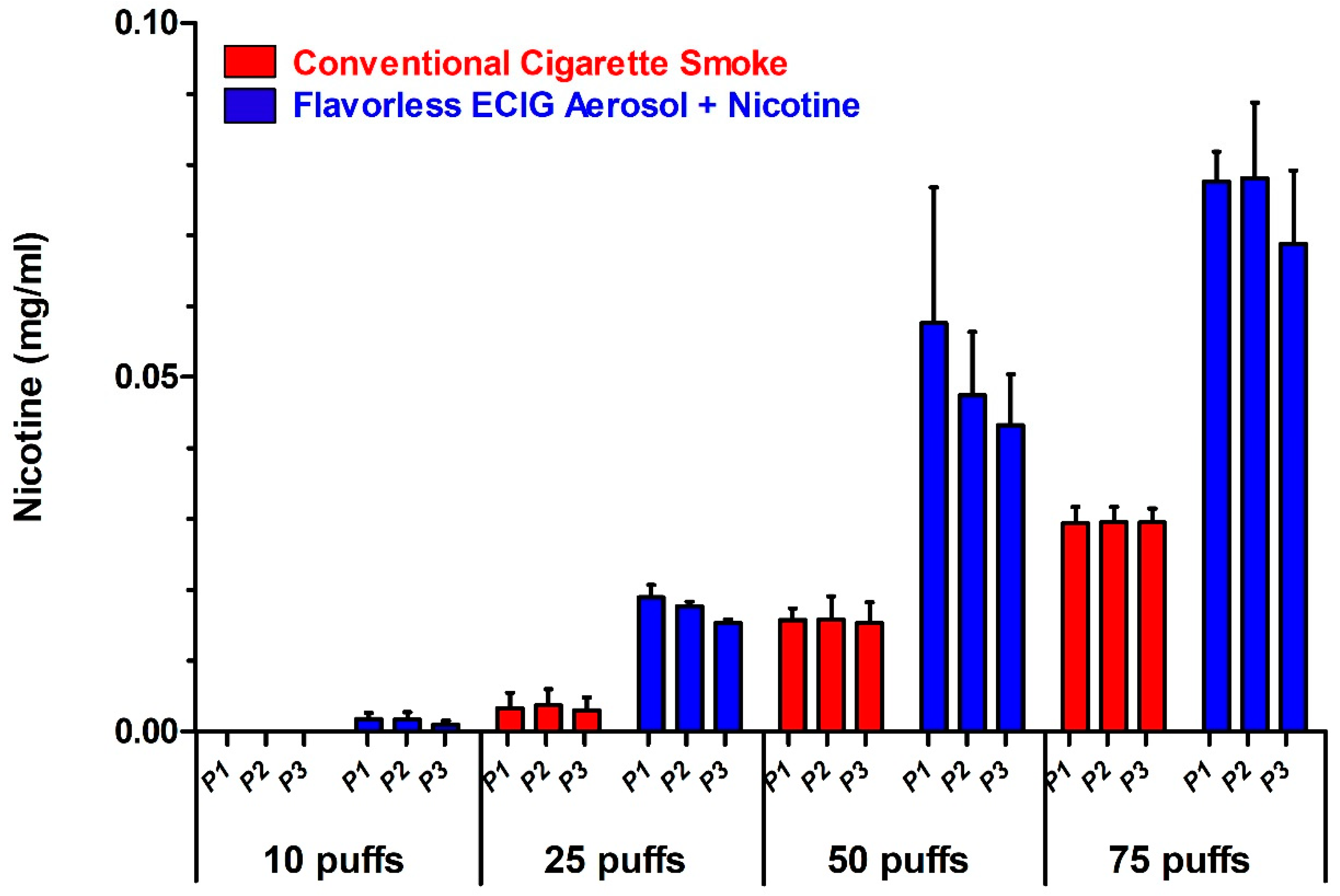
2.6. High-Performance Liquid Chromatography (HPLC) Determination of Nicotine
2.7. Colony-Forming Units (CFUs)
2.8. Biofilm Biomass
2.9. Biofilm Processing for Scanning Electron Microscope (SEM) Imaging
2.10. Statistical Analysis
3. Results
3.1. Distribution of Aerosol and Smoke in the Exposure Chamber
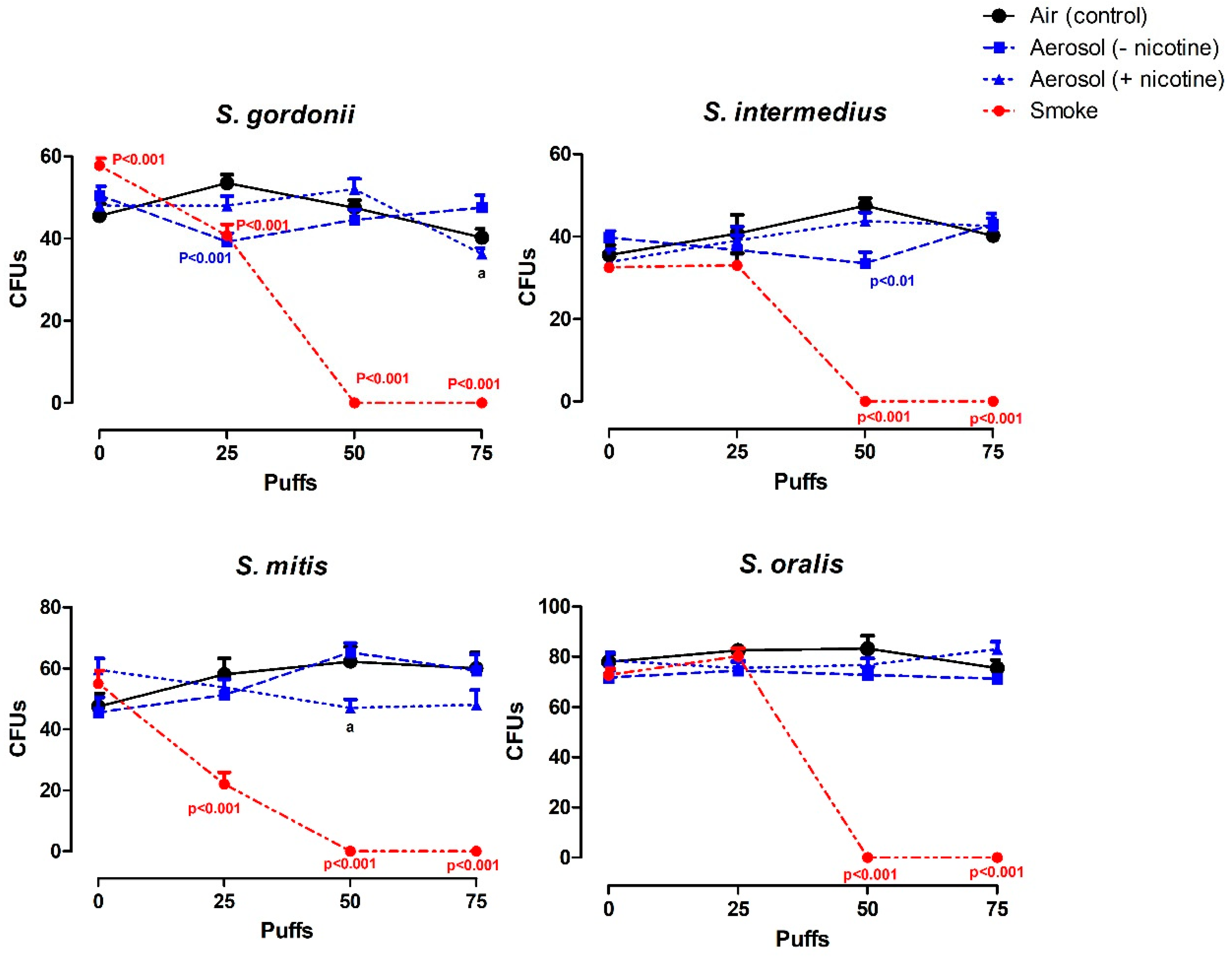
3.2. Effects of Electronic Cigarette (ECIG)-Generated Aerosol and Smoke on CFU Counts
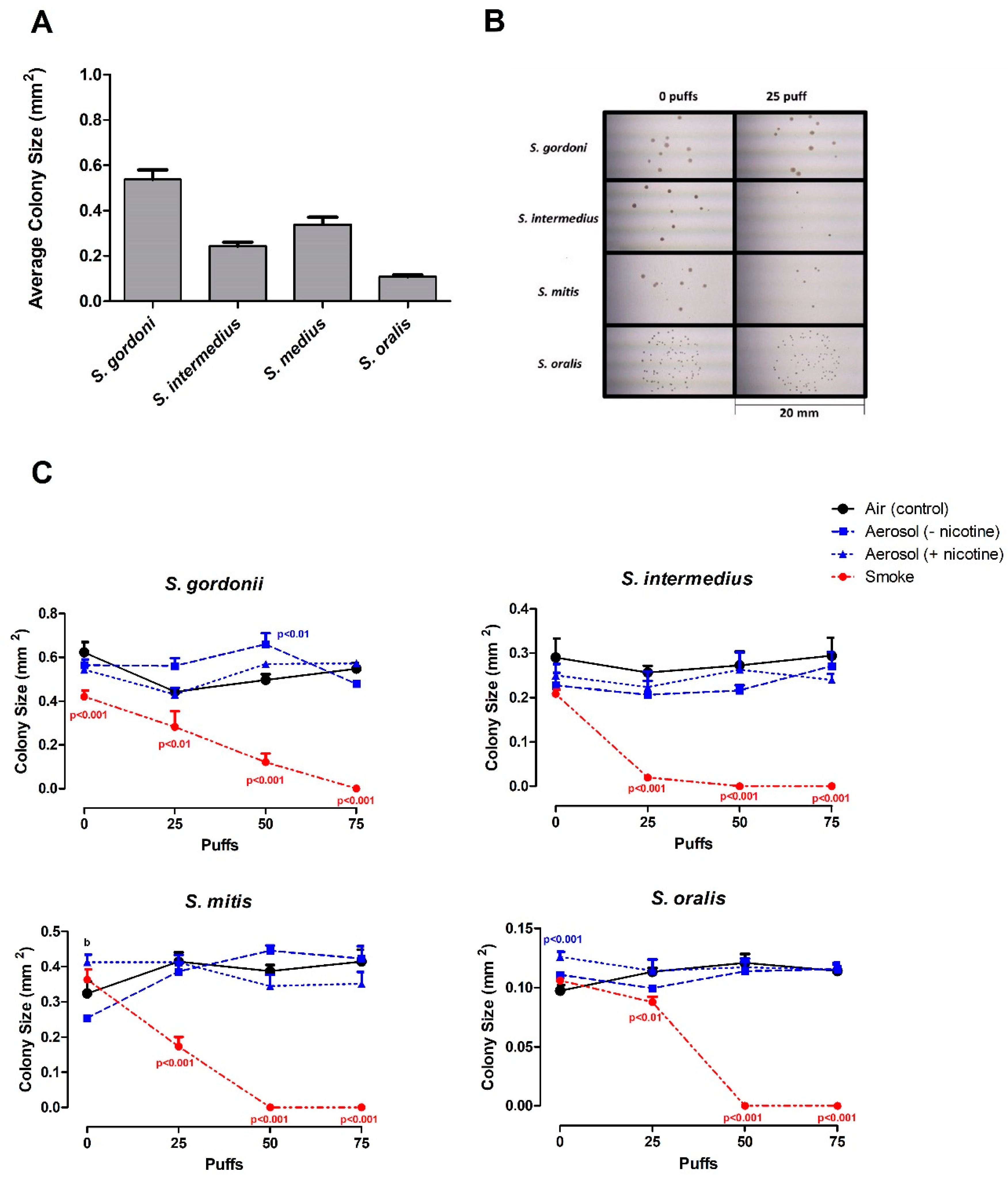
3.3. Effects of ECIG-Generated Aerosol and Smoke on Colony Size
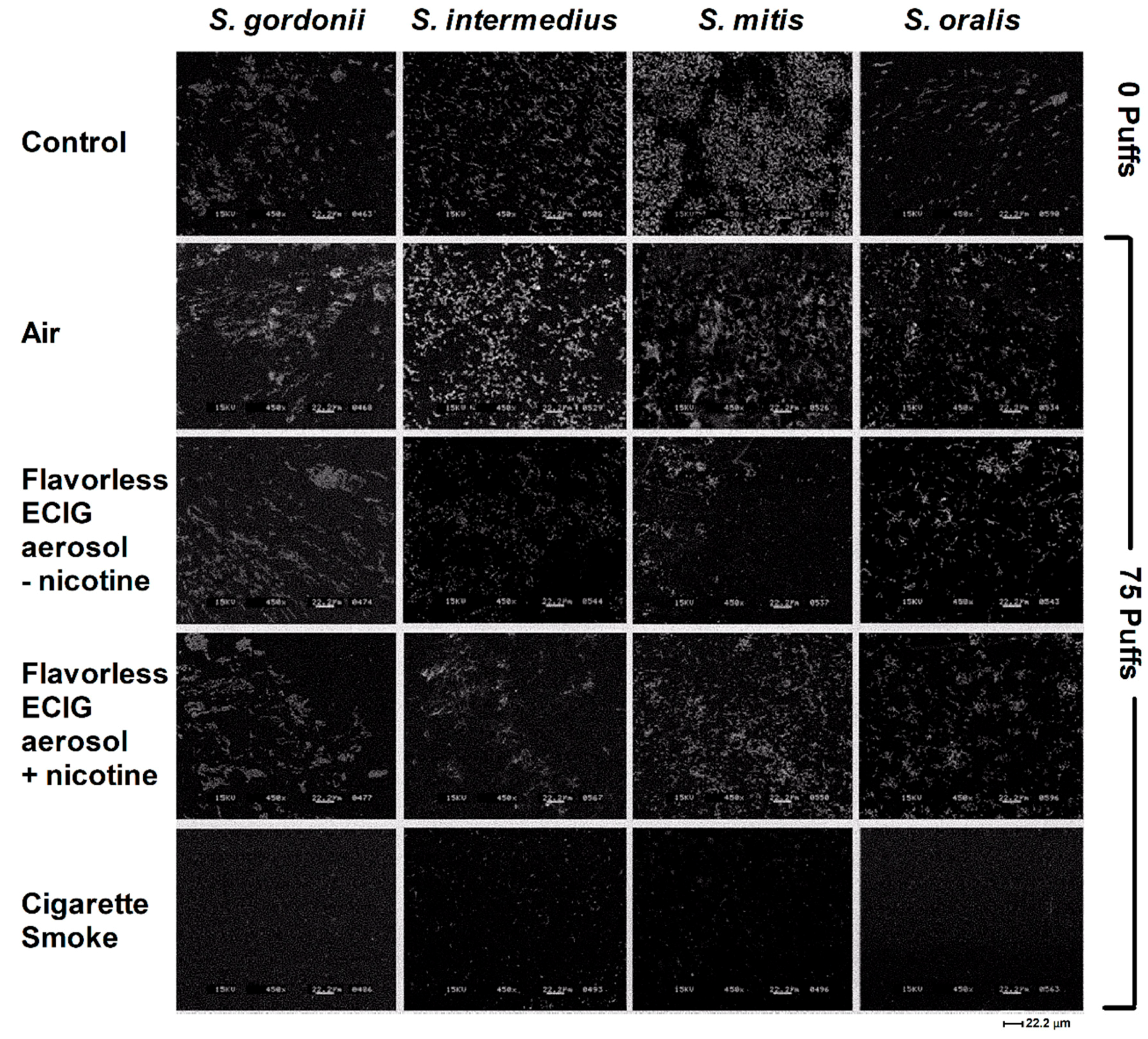
3.4. Effects of ECIG-Generated Aerosol and Smoke on Bacterial Biofilms
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Glantz, S.A.; Bareham, D.W. E-Cigarettes: Use, Effects on Smoking, Risks, and Policy Implications. Ann. Rev. Public Health 2018, 39, 215–235. [Google Scholar] [CrossRef]
- Hajek, P.; Etter, J.-F.; Benowitz, N.; Eissenberg, T.; McRobbie, H. Electronic cigarettes: Review of use, content, safety, effects on smokers and potential for harm and benefit. Addiction 2014, 109, 1801–1810. [Google Scholar] [CrossRef]
- Jensen, R.P.; Luo, W.; Pankow, J.F.; Strongin, R.M.; Peyton, D.H. Hidden formaldehyde in e-cigarette aerosols. N. Engl. J. Med. 2015, 372, 392–394. [Google Scholar] [CrossRef]
- Holliday, R.; Kist, R.; Bauld, L. E-cigarette vapour is not inert and exposure can lead to cell damage. Evid. Based Dent. 2016, 17, 2–3. [Google Scholar] [CrossRef]
- Yu, V.; Rahimy, M.; Korrapati, A.; Xuan, Y.; Zou, A.E.; Krishnan, A.R.; Tsui, T.; Aguilera, J.A.; Advani, S.; Alexander, L.E.C.; et al. Electronic cigarettes induce DNA strand breaks and cell death independently of nicotine in cell lines. Oral Oncol. 2016, 52, 58–65. [Google Scholar] [CrossRef]
- Muthumalage, T.; Prinz, M.; Ansah, K.O.; Gerloff, J.; Sundar, I.K.; Rahman, I. Inflammatory and Oxidative Responses Induced by Exposure to Commonly Used e-Cigarette Flavoring Chemicals and Flavored e-Liquids without Nicotine. Front. Physiol. 2017, 8, 1130. [Google Scholar] [CrossRef]
- Bahl, V.; Lin, S.; Xu, N.; Davis, B.; Wang, Y.; Talbot, P. Comparison of electronic cigarette refill fluid cytotoxicity using embryonic and adult models. Reprod. Toxicol. 2012, 34, 529–537. [Google Scholar] [CrossRef]
- Leigh, N.J.; Tran, P.L.; O’Connor, R.J.; Goniewicz, M.L. Cytotoxic effects of heated tobacco products (HTP) on human bronchial epithelial cells. Tob. Control 2018, 27, s26–s29. [Google Scholar] [CrossRef]
- Leigh, N.J.; Lawton, R.I.; Hershberger, P.A.; Goniewicz, M.L. Flavourings significantly affect inhalation toxicity of aerosol generated from electronic nicotine delivery systems (ENDS). Tob. Control 2016, 25, ii81–ii87. [Google Scholar] [CrossRef]
- Sundar, I.K.; Javed, F.; Romanos, G.E.; Rahman, I. E-cigarettes and flavorings induce inflammatory and pro-senescence responses in oral epithelial cells and periodontal fibroblasts. Oncotarget 2016, 7, 77196–77204. [Google Scholar] [CrossRef]
- Burne, R.A. Oral Streptococci... Products of Their Environment. J. Dent. Res. 1998, 77, 445–452. [Google Scholar] [CrossRef] [PubMed]
- Rosan, B.; Lamont, R.J. Dental plaque formation. Microbes Infect. 2000, 2, 1599–1607. [Google Scholar] [CrossRef]
- Kolenbrander, P.E. Oral Microbial Communities: Biofilms, Interactions, and Genetic Systems. Annu. Rev. Microbiol. 2000, 54, 413–437. [Google Scholar] [CrossRef]
- Hajishengallis, G.; Lamont, R.J. Beyond the red complex and into more complexity: The polymicrobial synergy and dysbiosis (PSD) model of periodontal disease etiology. Mol. Oral Microbiol. 2012, 27, 409–419. [Google Scholar] [CrossRef]
- Aas, J.A.; Paster, B.J.; Stokes, L.N.; Olsen, I.; Dewhirst, F.E. Defining the Normal Bacterial Flora of the Oral Cavity. J. Clin. Microbiol. 2005, 43, 5721–5732. [Google Scholar] [CrossRef] [PubMed]
- Colombo, A.V.; da Silva, C.M.; Haffajee, A.; Colombo, A.P.V. Identification of intracellular oral species within human crevicular epithelial cells from subjects with chronic periodontitis by fluorescence in situ hybridization. J. Periodontal Res. 2007, 42, 236–243. [Google Scholar] [CrossRef]
- Garnier, F.; Gerbaud, G.; Courvalin, P.; Galimand, M. Identification of clinically relevant viridans group streptococci to the species level by PCR. J. Clin. Microbiol. 1997, 35, 2337–2341. [Google Scholar] [PubMed]
- Kolenbrander, P.E.; Andersen, R.N.; Blehert, D.S.; Egland, P.G.; Foster, J.S.; Palmer, R.J. Communication among oral bacteria. Microbiol. Mol. Biol. Rev. 2002, 66, 486–505. [Google Scholar] [CrossRef]
- Kolenbrander, P.E.; Egland, P.G.; Diaz, P.I.; Palmer, R.J. Genome–genome interactions: Bacterial communities in initial dental plaque. Trends Microbiol. 2005, 13, 11–15. [Google Scholar] [CrossRef] [PubMed]
- Teles, F.R.; Teles, R.P.; Uzel, N.G.; Song, X.Q.; Torresyap, G.; Socransky, S.S.; Haffajee, A.D. Early microbial succession in re-developing dental biofilms in periodontal health and disease. J. Periodontal Res. 2012, 47, 95–104. [Google Scholar] [CrossRef] [PubMed]
- Socransky, S.S.; Haffajee, A.D.; Cugini, M.A.; Smith, C.; Kent, R.L. Microbial complexes in subgingival plaque. J. Clin. Periodontol. 1998, 25, 134–144. [Google Scholar] [CrossRef]
- Heller, D.; Helmerhorst, E.J.; Gower, A.C.; Siqueira, W.L.; Paster, B.J.; Oppenheim, F.G. Microbial Diversity in the Early in Vivo-Formed Dental Biofilm. Appl. Environ. Microbiol. 2016, 82, 1881–1888. [Google Scholar] [CrossRef]
- Hasegawa, Y.; Mans, J.J.; Mao, S.; Lopez, M.C.; Baker, H.V.; Handfield, M.; Lamont, R.J. Gingival epithelial cell transcriptional responses to commensal and opportunistic oral microbial species. Infect. Immun. 2007, 75, 2540–2547. [Google Scholar] [CrossRef] [PubMed]
- Thurnheer, T.; Belibasakis, G.N. Streptococcus oralis maintains homeostasis in oral biofilms by antagonizing the cariogenic pathogen Streptococcus mutans. Mol. Oral Microbiol. 2018, 33, 234–239. [Google Scholar] [CrossRef]
- Gross, E.L.; Beall, C.J.; Kutsch, S.R.; Firestone, N.D.; Leys, E.J.; Griffen, A.L. Beyond Streptococcus mutans: Dental caries onset linked to multiple species by 16S rRNA community analysis. PLoS ONE 2012, 7, e47722. [Google Scholar] [CrossRef]
- Huang, X.; Browngardt, C.M.; Jiang, M.; Ahn, S.-J.; Burne, R.A.; Nascimento, M.M. Diversity in Antagonistic Interactions between Commensal Oral Streptococci and Streptococcus mutans. Caries Res. 2018, 52, 88–101. [Google Scholar] [CrossRef]
- Liu, Y.; Palmer, S.R.; Chang, H.; Combs, A.N.; Burne, R.A.; Koo, H. Differential oxidative stress tolerance of Streptococcus mutans isolates affects competition in an ecological mixed-species biofilm model. Environ. Microbiol. Rep. 2018, 10, 12–22. [Google Scholar] [CrossRef]
- Herrero, E.R.; Slomka, V.; Bernaerts, K.; Boon, N.; Hernandez-Sanabria, E.; Passoni, B.B.; Quirynen, M.; Teughels, W. Antimicrobial effects of commensal oral species are regulated by environmental factors. J. Dent. 2016, 47, 23–33. [Google Scholar] [CrossRef] [PubMed]
- Holmlund, A.; Lampa, E.; Lind, L. Oral health and cardiovascular disease risk in a cohort of periodontitis patients. Atherosclerosis 2017, 262, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Huang, R.; Zhou, X.; Zhang, K.; Zheng, X.; Gregory, R.L. Effect of nicotine on dual-species biofilms of Streptococcus mutans and Streptococcus sanguinis. FEMS Microbiol. Lett. 2014, 350, 125–132. [Google Scholar] [CrossRef]
- Huang, R.; Li, M.; Ye, M.; Yang, K.; Xu, X.; Gregory, R.L. Effects of Nicotine on Streptococcus gordonii Growth, Biofilm Formation, and Cell Aggregation. Appl. Environ. Microbiol. 2014, 80, 7212–7218. [Google Scholar] [CrossRef]
- Tomar, S.L.; Asma, S. Smoking-Attributable Periodontitis in the United States: Findings from NHANES III. J. Periodontol. 2000, 71, 743–751. [Google Scholar] [CrossRef] [PubMed]
- Bagaitkar, J.; Daep, C.A.; Patel, C.K.; Renaud, D.E.; Demuth, D.R.; Scott, D.A. Tobacco smoke augments Porphyromonas gingivalis-Streptococcus gordonii biofilm formation. PLoS ONE 2011, 6, e27386. [Google Scholar] [CrossRef] [PubMed]
- Moon, J.-H.; Lee, J.-H.; Lee, J.-Y. Subgingival microbiome in smokers and non-smokers in Korean chronic periodontitis patients. Mol. Oral Microbiol. 2015, 30, 227–241. [Google Scholar] [CrossRef]
- Calafat, A.M. Determination of tar, nicotine, and carbon monoxide yields in the mainstream smoke of selected international cigarettes. Tob. Control 2004, 13, 45–51. [Google Scholar] [CrossRef]
- Cobb, E.; Hall, J.; Palazzolo, D.L. Induction of Metallothionein Expression After Exposure to Conventional Cigarette Smoke but Not Electronic Cigarette (ECIG)-Generated Aerosol in Caenorhabditis elegans. Front. Physiol. 2018, 9, 426. [Google Scholar] [CrossRef]
- Palazzolo, D.L.; Nelson, J.M.; Ely, E.A.; Crow, A.P.; Distin, J.; Kunigelis, S.C. The Effects of Electronic Cigarette (ECIG)-Generated Aerosol and Conventional Cigarette Smoke on the Mucociliary Transport Velocity (MTV) Using the Bullfrog (R. catesbiana) Palate Paradigm. Front. Physiol. 2017, 8, 1023. [Google Scholar] [CrossRef]
- Trehy, M.L.; Ye, W.; Hadwiger, M.E.; Moore, T.W.; Allgire, J.F.; Woodruff, J.T.; Ahadi, S.; Black, J.C.; Westenberger, B.J. Analysis of Electronic Cigarette Cartridges, Refill Solutions, and Smoke for Nicotine and Nicotine Related Impurities. J. Liq. Chromatogr. Relat. Technol. 2011, 34, 1442–1458. [Google Scholar] [CrossRef]
- Murray, J. Nicotine and What Else? HPLC Elution Optimization for the Analysis of Alkaloids Found in Electronic Cigarettes. Honors Theses, 2014. Available online: https://scholar.utc.edu/honors-theses/3 (accessed on 21 March 2019).
- Palazzolo, D.L.; Crow, A.P.; Nelson, J.M.; Johnson, R.A. Trace Metals Derived from Electronic Cigarette (ECIG) Generated Aerosol: Potential Problem of ECIG Devices That Contain Nickel. Front Physiol. 2017, 7. [Google Scholar] [CrossRef]
- Sutton, S. Accuracy of plate counts. J. Valid. Technol. 2011, 17, 42–46. [Google Scholar]
- Kolenbrander, P.E. Multispecies communities: Interspecies interactions influence growth on saliva as sole nutritional source. Int. J. Oral Sci. 2011, 3, 49–54. [Google Scholar] [CrossRef]
- Shah, S.A.; Ganesan, S.M.; Varadharaj, S.; Dabdoub, S.M.; Walters, J.D.; Kumar, P.S. The making of a miscreant: Tobacco smoke and the creation of pathogen-rich biofilms. NPJ Biofilms Microbiomes 2017, 3, 26. [Google Scholar] [CrossRef]
- Zonuz, A.T.; Rahmati, A.; Mortazavi, H.; Khashabi, E.; Farahani, R.M.Z. Effect of cigarette smoke exposure on the growth of Streptococcus mutans and Streptococcus sanguis: An in vitro study. Nicotine Tob. Res. 2008, 10, 63–67. [Google Scholar] [CrossRef]
- Abiko, Y.; Sato, T.; Mayanagi, G.; Takahashi, N. Profiling of subgingival plaque biofilm microflora from periodontally healthy subjects and from subjects with periodontitis using quantitative real-time PCR. J. Periodont. Res. 2010, 45, 389–395. [Google Scholar] [CrossRef]
- Genco, C.A.; Van Dyke, T.; Amar, S. Animal models for Porphyromonas gingivalis-mediated periodontal disease. Trends Microbiol. 1998, 6, 444–449. [Google Scholar] [CrossRef]
- Olsen, I.; Progulske-Fox, A. Invasion of Porphyromonas gingivalis strains into vascular cells and tissue. J. Oral Microbiol. 2015, 7. [Google Scholar] [CrossRef] [PubMed]
- Pussinen, P.J.; Tuomisto, K.; Jousilahti, P.; Havulinna, A.S.; Sundvall, J.; Salomaa, V. Endotoxemia, immune response to periodontal pathogens, and systemic inflammation associate with incident cardiovascular disease events. Arterioscler. Thromb. Vasc. Biol. 2007, 27, 1433–1439. [Google Scholar] [CrossRef] [PubMed]
- Sosnowski, T.R.; Odziomek, M. Particle Size Dynamics: Toward a Better Understanding of Electronic Cigarette Aerosol Interactions with the Respiratory System. Front. Physiol. 2018, 9, 853. [Google Scholar] [CrossRef]
- Kaufman, J.W.; Farahmand, K. In vivo measurements of human oral cavity heat and water vapor transport. Respir. Physiol. Neurobiol. 2006, 150, 261–277. [Google Scholar] [CrossRef]

| Smoke Pump/Chamber | Aerosol Pump/Chamber | |
|---|---|---|
| Pump Flow Rate (mL/min) | 400 | 400 |
| Puff Duration (s) | 5 | 5 |
| Puff Volume (mL) | 33.3 | 33.3 |
| Nicotine (mg/cigarette) a | 0.92 | 2.80 |
| Nicotine (mg/puff) b | 0.06 | 0.19 |
| Nicotine Flow (µg/puff/mL) into Chamber | 1.8 | 5.7 |
| Exposure Chamber Volume (cm3) | 2126 | 2126 |
| Nicotine Delivered to Chamber (mg) | ||
| 0 puffs (0.0 cigarettes) | 0.0 | 0.0 |
| 25 puffs (1.7 cigarettes) | 1.5 | 4.8 |
| 50 puffs (3.3 cigarettes) | 3.0 | 9.5 |
| 75 puffs (5.0 cigarettes) | 4.5 | 14.3 |
| Nicotine Concentration in Chamber (µg/cm3) c | ||
| 0 puffs (0.0 cigarettes) | 0.0 | 0.0 |
| 25 puffs (1.7 cigarettes) | 0.7 | 2.2 |
| 50 puffs (3.3 cigarettes) | 1.4 | 4.5 |
| 75 puffs (5.0 cigarettes) | 2.1 | 6.7 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cuadra, G.A.; Smith, M.T.; Nelson, J.M.; Loh, E.K.; Palazzolo, D.L. A Comparison of Flavorless Electronic Cigarette-Generated Aerosol and Conventional Cigarette Smoke on the Survival and Growth of Common Oral Commensal Streptococci. Int. J. Environ. Res. Public Health 2019, 16, 1669. https://doi.org/10.3390/ijerph16101669
Cuadra GA, Smith MT, Nelson JM, Loh EK, Palazzolo DL. A Comparison of Flavorless Electronic Cigarette-Generated Aerosol and Conventional Cigarette Smoke on the Survival and Growth of Common Oral Commensal Streptococci. International Journal of Environmental Research and Public Health. 2019; 16(10):1669. https://doi.org/10.3390/ijerph16101669
Chicago/Turabian StyleCuadra, Giancarlo A., Maxwell T. Smith, John M. Nelson, Emma K. Loh, and Dominic L. Palazzolo. 2019. "A Comparison of Flavorless Electronic Cigarette-Generated Aerosol and Conventional Cigarette Smoke on the Survival and Growth of Common Oral Commensal Streptococci" International Journal of Environmental Research and Public Health 16, no. 10: 1669. https://doi.org/10.3390/ijerph16101669
APA StyleCuadra, G. A., Smith, M. T., Nelson, J. M., Loh, E. K., & Palazzolo, D. L. (2019). A Comparison of Flavorless Electronic Cigarette-Generated Aerosol and Conventional Cigarette Smoke on the Survival and Growth of Common Oral Commensal Streptococci. International Journal of Environmental Research and Public Health, 16(10), 1669. https://doi.org/10.3390/ijerph16101669






