Gulf War Illness: Unifying Hypothesis for a Continuing Health Problem
Abstract
1. Background
2. Potential Exposures
3. Multiple Vaccinations as the Initiating Cause of GWI
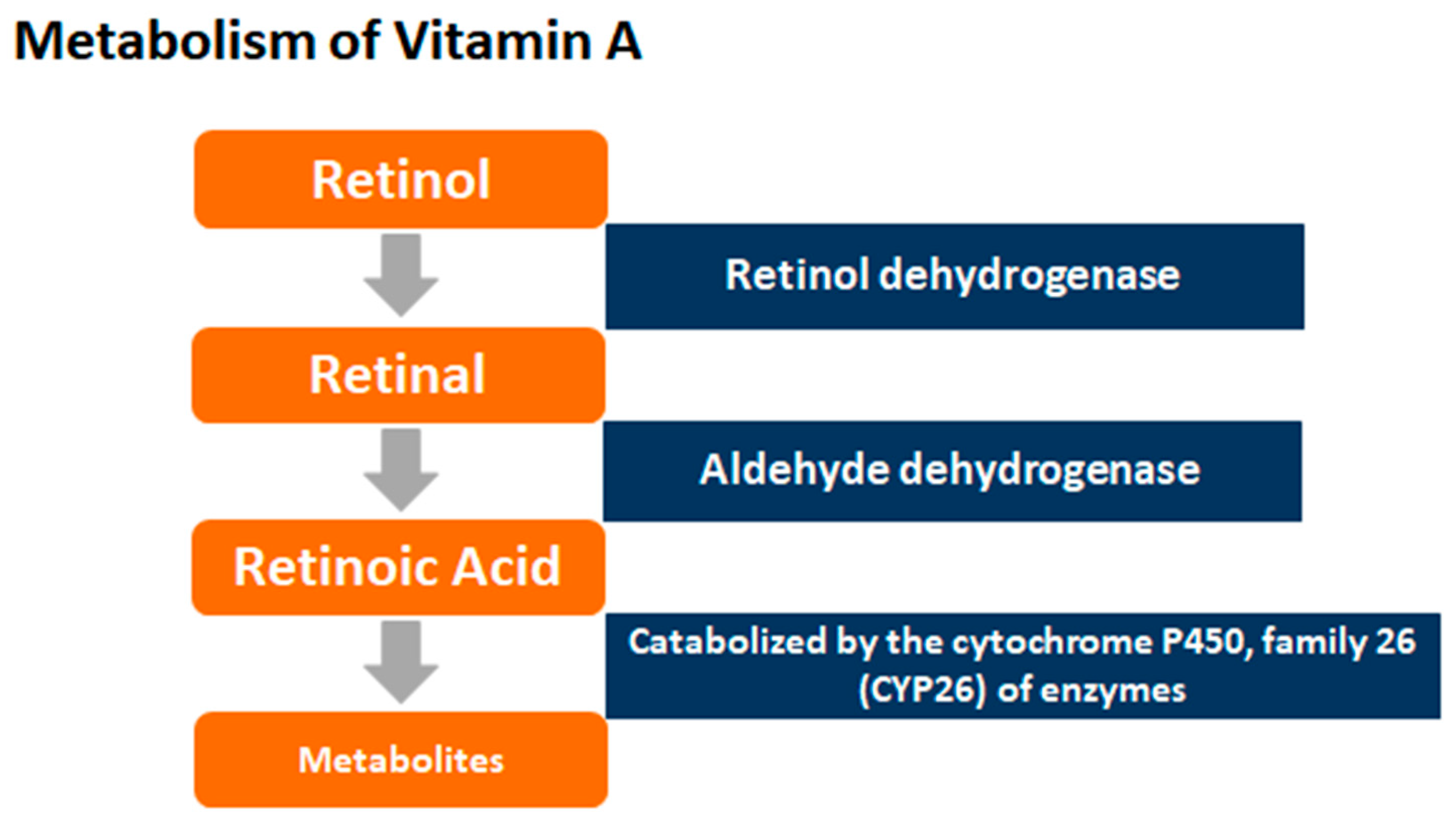
4. Clues to the Pathobiology of GWI
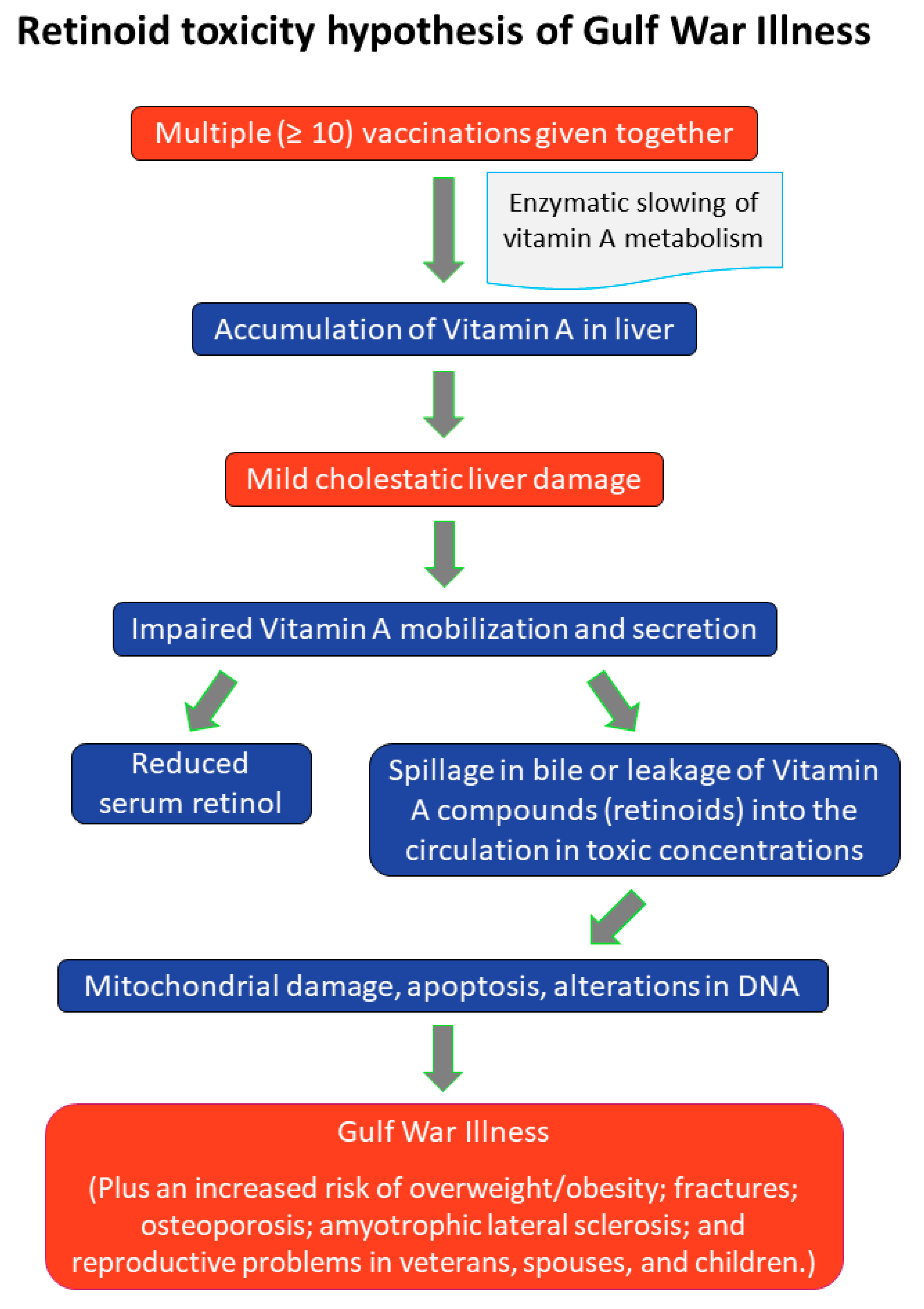
5. Synthesis of the Research Literature: New Hypothesis
6. Review of the Evidence
7. Discussion and Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Institute of Medicine. Gulf War and Health, Volume 8: Update of Health Effects of Serving in the Gulf War; The National Academies Press: Washington, DC, USA, 2010; ISBN 978-0-309-14921-1. [Google Scholar] [CrossRef]
- The Gulf War Illness Landscape. The 1990-91 Gulf War. Department of Defense, Gulf War Illness Research Program, October 2016. Available online: http://cdmrp.army.mil/gwirp/pdfs/GWIRP_Landscape.pdf (accessed on 27 December 2018).
- Kang, H.K.; Mahan, C.M.; Lee, K.Y.; Magee, C.A.; Murphy, F.M. Illnesses among United States veterans of the Gulf War: A population-based survey of 30,000 veterans. J. Occup. Environ. Med. 2000, 42, 491–501. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.K.; Li, B.; Mahan, C.M.; Eisen, S.A.; Engel, C.C. Health of US veterans of 1991 Gulf War: A follow-up survey in 10 years. J. Occup. Environ. Med. 2009, 51, 401–410. [Google Scholar] [CrossRef] [PubMed]
- Iowa Persian Gulf Study Group. Self-reported illness and health status among Gulf War veterans: A population-based study. JAMA 1997, 277, 238–245. [Google Scholar] [CrossRef]
- Institute of Medicine. Gulf War and Health: Volume 4: Health Effects of Serving in the Gulf War; Chapter 2; The National Academies Press: Washington, DC, USA, 2006. [CrossRef]
- Dursa, E.K.; Barth, S.K.; Schneiderman, A.I.; Bossarte, R.M. Physical and mental health status of Gulf War and Gulf Era veterans: Results from a large population-based epidemiological study. J. Occup. Environ. Med. 2016, 58, 41–46. [Google Scholar] [CrossRef] [PubMed]
- White, R.F.; Steele, L.; O’Callaghan, J.P.; Sullivan, K.; Binns, J.H.; Golomb, B.A.; Bloom, F.E.; Bunker, J.A.; Crawford, F.; Graves, J.C.; et al. Recent research on Gulf War illness and other health problems in veterans of the 1991 Gulf War: Effects of toxicant exposures during deployment. Cortex 2016, 74, 449–475. [Google Scholar] [CrossRef]
- Research Advisory Committee on Gulf War Veterans’ Illnesses Gulf War Illness and the Health of Gulf War Veterans: Scientific Findings and Recommendations; James H. Binns, Committee Chair; U.S. Government Printing Office: Washington, DC, USA, November 2008. Available online: https://www.va.gov/RAC-GWVI/docs/Committee_Documents/GWIandHealthofGWVeterans_RAC-GWVIReport_2008.pdf (accessed on 27 December 2018).
- Nevin, R.L.; Croft, A.M. Psychiatric effects of malaria and anti-malarial drugs: Historical and modern perspectives. Malar. J. 2016, 15, 332. [Google Scholar] [CrossRef]
- Kelsall, H.L.; Sim, M.R.; Forbes, A.B.; Glass, D.C.; McKenzie, D.P.; Ikin, D.P.; Abramson, M.J.; Blizzard, L.; Ittak, P. Symptoms and medical conditions in Australian veterans of the 1991 Gulf War: Relation to immunisations and other Gulf War exposures. Occup. Environ. Med. 2004, 61, 1006–1013. [Google Scholar] [CrossRef]
- U.S. Department of Veterans Affairs. Available online: https://www.publichealth.va.gov/exposures/gulfwar/sources/index.asp; (accessed on 27 December 2018).
- Unwin, C.; Blatchley, N.; Coker, W.; Ferry, S.; Hotopf, M.; Hull, L.; Ismail, K.; Palmer, I.; David, A.; Wessely, S. Health of UK servicemen who served in Persian Gulf War. Lancet 1999, 16, 169–178. [Google Scholar] [CrossRef]
- Steele, L. Prevalence and patterns of Gulf War illness in Kansas veterans: Association of symptoms with characteristics of person, place, and time of military service. Am. J. Epidemiol. 2000, 152, 992–1002. [Google Scholar] [CrossRef]
- Hotopf, M.; David, A.; Hull, L.; Ismail, K.; Unwin, C.; Wessely, S. Role of vaccinations as risk factors for ill health in veterans of the Gulf war: Cross sectional study. BMJ 2000, 320, 1363–1367. [Google Scholar] [CrossRef]
- Cherry, N.; Creed, F.; Silman, A.; Dunn, G.; Baxter, D.; Smedley, J.; Taylor, S.; Macfarlane, G.J. Health and exposures of United Kingdom Gulf war veterans. Part II: The relation of health to exposure. Occup. Environ. Med. 2001, 58, 299–306. [Google Scholar] [CrossRef] [PubMed]
- Nicolson, G.L. Gulf war illnesses: Causes and treatments. Armed Forces Med. Devel. 2001, 2, 41–44. Available online: http://www.immed.org/GWI%20Research%20docs/06.26.12.updates.pdfs.gwi/GWI-GLNetal-AFMD.pdf (accessed on 27 December 2018).
- Benn, C.S.; Netea, M.G.; Selin, L.K.; Aaby, P. A small jab—A big effect: Nonspecific immunomodulation by vaccines. Trends Immunol. 2013, 34, 431–439. [Google Scholar] [CrossRef] [PubMed]
- Jensen, K.J.; Benn, C.S.; van Crevel, R. Unravelling the nature of non-specific effects of vaccines—A challenge for innate immunologists. Semin. Immunol. 2016, 28, 377–383. [Google Scholar] [CrossRef] [PubMed]
- Mogensen, S.W.; Andersen, A.; Rodrigues, A.; Benn, C.S.; Aaby, P. The introduction of diphtheria-tetanus-pertussis and oral polio vaccine among young infants in an urban African community: A natural experiment. EBio Med. 2017, 17, 192–198. [Google Scholar] [CrossRef] [PubMed]
- Mawson, A.R.; Ray, B.D.; Bhuiyan, A.R.; Jacob, B. Pilot comparative study on the health of vaccinated and unvaccinated 6- to 12-year-old U.S. children. J. Transl. Sci. 2017, 3, 1–12. [Google Scholar] [CrossRef]
- Mawson, A.R.; Bhuiyan, A.Z.; Jacob, B.; Ray, B.D. Preterm birth, vaccination and neurodevelopmental disorders: A cross-sectional study of vaccinated and unvaccinated children. J. Transl. Sci. 2017, 3, 1–8. [Google Scholar] [CrossRef]
- Koslik, H.J.; Hamilton, G.; Golomb, B.A. Mitochondrial dysfunction in Gulf War illness revealed by 31Phosphorus Magnetic Resonance Spectroscopy: A case-control study. PLoS ONE 2014, 9, e92887. [Google Scholar] [CrossRef]
- Fostel, J.; Boneva, R.; Lloyd, A. Exploration of the gene expression correlates of chronic unexplained fatigue using factor analysis. Pharmacogenomics 2006, 7, 441–454. [Google Scholar] [CrossRef]
- Rogal, S.S.; Bielefeldt, K.; Wasan, A.D.; Szigethy, E.; Lotrich, F.; DiMartini, A.F. Fibromyalgia symptoms and cirrhosis. Dig. Dis. Sci. 2015, 60, 1482–1489. [Google Scholar] [CrossRef]
- Al-Harthy, N.; Kumagi, T.; Coltescu, C.; Hirschfield, G.M. The specificity of fatigue in primary biliary cirrhosis: Evaluation of a large clinic practice. Hepatology 2010, 52, 562–570. [Google Scholar] [CrossRef] [PubMed]
- Kelsall, H.L.; McKenzie, D.P.; Sim, M.R.; Leder, K.; Forbes, A.B.; Dwyer, T. Physical, psychological, and functional comorbidities of multisymptom illness in Australian male veterans of the 1991 Gulf war. Am. J. Epidemiol. 2009, 170, 1048–1056. [Google Scholar] [CrossRef]
- Maden, M. Retinoid signaling in the development of the central nervous system. Nat. Rev. Neurosci. 2002, 3, 843–853. [Google Scholar] [CrossRef] [PubMed]
- D’Ambrosio, D.N.; Clugston, R.D.; Blaner, W.S. Vitamin A metabolism: An update. Nutrients 2011, 3, 63–103. [Google Scholar] [CrossRef] [PubMed]
- Russell, R.M. Vitamin and trace mineral deficiency and excess. In Harrison’s Principles of Internal Medicine, 16th ed.; Kasper, D.L., Braunwald, E., Fauci, A.S., Hauser, S.L., Long, D.L., Jameson, J.L., Eds.; McGraw Hill: New York, NY, USA, 2005; pp. 403–411. [Google Scholar]
- Penniston, K.L.; Tanumihardjo, S.A. The acute and chronic toxic effects of vitamin A. Am. J. Clin. Nutr. 2006, 83, 191–201. [Google Scholar] [CrossRef] [PubMed]
- Theodosiou, M.; Laudet, V.; Schubert, M. From carrot to clinic: An overview of the retinoic acid signaling pathway. Cell. Mol. Life Sci. 2010, 67, 1423–1445. [Google Scholar] [CrossRef] [PubMed]
- Blomhoff, R.; Blomhoff, H.K. Overview of retinoid metabolism and function. J. Neurobiol. 2006, 66, 606–630. [Google Scholar] [CrossRef]
- Alecron-Corredor, O.M.; Alfonso, R. Clinical and biochemical alterations in rats treated with high doses of vitamin A. Arch. Latinoam. Nutr. 2007, 57, 224–230. [Google Scholar]
- Shirakami, Y.; Lee, S.A.; Clugston, R.D.; Blaner, W.S. Hepatic metabolism of retinoids and disease associations. Biochim. Biophys. Acta 2012, 1821, 124–136. [Google Scholar] [CrossRef]
- Jaensson-Gyllenbäck, E.; Kotarsky, K.; Zapata, F.; Persson, E.K.; Gundersen, T.E.; Blomhoff, R.; Agace, W.W. Bile retinoids imprint intestinal CD103+ dendritic cells with the ability to generate gut-tropic T cells. Mucosal Immunol. 2011, 4, 438–447. [Google Scholar] [CrossRef]
- Leo, M.A.; Lieber, C.S. New pathway of retinol metabolism in liver microsomes. J. Biol. Chem. 1985, 260, 5228–5231. [Google Scholar]
- Fukuda, K.; Nisenbaum, R.; Stewart, G.; Thompson, W.W.; Robin, L.; Washko, R.M.; Noah, D.L.; Barrett, D.H.; Randall, B.; Herwaldt, B.L.; et al. Chronic multisymptom illness affecting Air Force veterans of the Gulf War. JAMA 1998, 280, 981–988. [Google Scholar] [CrossRef]
- Simmons, R.; Maconochie, N.; Doyle, P. Self-reported ill health in male UK Gulf War veterans: A retrospective cohort study. BMC Public Health 2004, 4, 27. [Google Scholar] [CrossRef] [PubMed]
- Maule, A.L.; Janulewicz, P.A.; Sullivan, K.A.; Krengel, M.H.; Yee, M.K.; McClean, M.; White, R.F. Meta-analysis of self-reported health symptoms in 1990–1991 Gulf War and Gulf War-era veterans. BMJ Open 2018, 8, e016086. [Google Scholar] [CrossRef] [PubMed]
- DermNet. Available online: https://www.dermnetnz.org/topics/vitamin-a-toxicity?utm_source=TrendMD&utm_medium=cpc&utm_campaign=DermNet_NZ_TrendMD_1 (accessed on 29 December 2017).
- Medscape: Vitamin A Toxicity Clinical Presentation. Available online: https://emedicine.medscape.com/article/819426-clinical (accessed on 29 December 2017).
- Myhre, A.M.; Carlsen, M.H.; Bøhn, S.K.; Wold, H.L.; Laake, P.; Blomhoff, R. Water-miscible, emulsified, and solid forms of retinol supplements are more toxic than oil-based preparations. Am. J. Clin. Nutr. 2003, 78, 1152–1159. [Google Scholar] [CrossRef] [PubMed]
- Hull, P.R.; D’Arcy, C. Isotretinoin use and subsequent depression and suicide: Presenting the evidence. Am. J. Clin. Dermatol. 2004, 4, 493–505. [Google Scholar] [CrossRef] [PubMed]
- Bremner, J.D.; Shearer, K.D.; McCaffery, P.J. Retinoic acid and affective disorders: The evidence for an association. J. Clin. Psychiatr. 2012, 73, 37–50. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, J. Polar hysteria: An expression of hypervitaminosis A. Am. J. Ther. 2004, 11, 507–516. [Google Scholar] [CrossRef]
- De Oliveira, M.R.; Silvestrin, R.B.; Mello e Souza, T.; Moreira, J.C. Therapeutic vitamin A doses increase the levels of markers of oxidative insult in substantia nigra and decrease locomotor and exploratory activity in rats after acute and chronic supplementation. Neurochem. Res. 2008, 33, 378–383. [Google Scholar] [CrossRef]
- Hanson, N.; Leachman, S. Safety issues in isotretinoin therapy. Semin. Cutan. Med. Surg. 2001, 20, 166–183. [Google Scholar] [CrossRef]
- Crockett, S.D.; Porter, C.Q.; Martin, C.F.; Sandler, R.S.; Kappelman, M.D. Isotretinoin use and the risk of inflammatory bowel disease: A case-control study. Am. J. Gastroenterol. 2010, 105, 1986–1993. [Google Scholar] [CrossRef] [PubMed]
- Machner, B.; Neppert, B.; Paulsen, M.; Hofmann, C.; Sander, T.; Helmchen, C. Pseudotumor cerebri as a reversible side effect of all-trans retinoic acid treatment in acute promyelocytic leukaemia. Eur. J. Neurol. 2008, 15, e68–e69. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, H.K.; Berezin, S.; Heyman, S.; Witzleben, C.; Watkins, J.B. Pleural effusion and ascites: Unusual presenting features in a pediatric patient with vitamin A intoxication. Clin. Pediatr. 1982, 21, 435–440. [Google Scholar] [CrossRef] [PubMed]
- Reid, S.; Hotopf, M.; Hull, L.; Ismail, K.; Unwin, C.; Wessely, S. Multiple chemical sensitivity and chronic fatigue syndrome in British Gulf War veterans. Am. J. Epidemiol. 2001, 153, 604–609. [Google Scholar] [CrossRef] [PubMed]
- Reid, S.; Chalder, T.; Cleare, A.; Hotopf, M.; Wessely, S. Chronic fatigue syndrome. Clin. Evid. 2002, 7, 966–978. [Google Scholar]
- Cupitt, J.M. A case for steroids in acute lung injury associated with the retinoic acid syndrome. Anaesth. Intensive Care 2000, 28, 202–204. [Google Scholar] [PubMed]
- Takada, S.; Matumoto, K.; Sakura, T.; Shiozaki, H.; Miyawaki, S. Sweet’s syndrome followed by retinoic acid syndrome during the treatment of acute promyelocytic leukemia with all-trans retinoic acid. Int. J. Hematol. 1999, 70, 26–29. [Google Scholar]
- Ferrer, M.; Kaplan, A.P. Progress and challenges in the understanding of chronic urticaria. Allergy Asthma Clin. Immunol. 2007, 3, 31–35. [Google Scholar] [CrossRef]
- Babina, M.; Guhl, S.; Motakis, E.; Artuc, M.; Hazzan, T.; Worm, M.; Forrest, A.R.; Zuberbier, T. Retinoic acid potentiates inflammatory cytokines in human mast cells: Identification of mast cells as prominent constituents of the skin retinoid network. Mol. Cell. Endocrinol. 2015, 406, 49–59. [Google Scholar] [CrossRef]
- Babina, M.; Artuc, M.; Guhl, S.; Zuberbier, T. Retinoic acid negatively impacts proliferation and MCtc specific attributes of human skin derived mast cells, but reinforces allergic stimulability. Int. J. Mol. Sci. 2017, 18, 525. [Google Scholar] [CrossRef]
- Bono, M.R.; Tejon, G.; Flores-Santibañez, F.; Fernandez, D.; Rosenblatt, M.; Sauma, D. Retinoic acid as a modulator of T cell immunity. Nutrients 2016, 8, 349. [Google Scholar] [CrossRef] [PubMed]
- Freemont, A.J. Bone and the Gulf War. J. Clin. Pathol. 2002, 55, 884. [Google Scholar] [CrossRef] [PubMed]
- Wu, A.M.; Huang, C.Q.; Lin, Z.K.; Tian, N.F.; Ni, W.F.; Wang, X.Y.; Xu, H.Z.; Chi, Y.L. The relationship between vitamin A and risk of fracture: Meta-analysis of prospective studies. J. Bone Miner. Res. 2014, 29, 2032–2039. [Google Scholar] [CrossRef] [PubMed]
- Horner, R.D.; Kamins, K.G.; Feussner, J.R.; Grambow, S.C.; Hoff-Lindquist, J.; Harati, Y.; Mitsumoto, H.; Pascuzzi, R.; Spencer, P.S.; Tim, R.; et al. Military personnel who were deployed to the Gulf region during the Gulf War period experienced a greater postwar risk of ALS than those who were not deployed to the Gulf. Neurology 2003, 61, 742–749. [Google Scholar] [CrossRef] [PubMed]
- Crochemore, C.; Virgili, M.; Bonamassa, B.; Canistro, D.; Pena-Altamira, E.; Paolini, M.; Contestabile, A. Long-term dietary administration of valproic acid does not affect, while retinoic acid decreases, the lifespan of G93A mice, a model for amyotrophic lateral sclerosis. Muscle Nerve 2009, 39, 548–552. [Google Scholar] [CrossRef]
- Araneta, M.R.; Moore, C.A.; Olney, R.S.; Edmonds, L.D.; Karcher, J.A.; McDonough, C.; Hiliopoulos, K.M.; Schlangen, K.M.; Gray, G.C. Goldenhar syndrome among infants born in military hospitals to Gulf War veterans. Teratology 1997, 56, 244–251. [Google Scholar] [CrossRef]
- Araneta, M.R.; Kamens, D.R.; Zau, A.C.; Gastañaga, V.M.; Schlangen, K.M.; Hiliopoulos, K.M.; Gray, G.C. Conception and pregnancy during the Persian Gulf War: The risk to women veterans. Ann. Epidemiol. 2004, 14, 109–116. [Google Scholar] [CrossRef]
- Tzimas, G.; Nau, H. The role of metabolism and toxicokinetics in retinoid teratogenesis. Curr. Pharm. Des. 2001, 7, 803–831. [Google Scholar] [CrossRef]
- D’Aniello, E.; Waxman, J.S. Input overload: Contributions of retinoic acid signaling feedback mechanisms to heart development and teratogenesis. Dev. Dyn. An Off. Publ. Am. Assoc. Anat. 2015, 244, 513–523. [Google Scholar] [CrossRef]
- Institute of Medicine. Gulf War and Health: Volume 10: Update of Health Effects of Serving in the Gulf War, 2016. Available online: http://www.covvha.net/2016/02/11/institute-medicine-gulf-war-health-volume-10-update-health-effects-serving-gulf-war-2016/ (accessed on 10 December 2018).
- Institute of Medicine. Long-Term Health Consequences of Exposure to Burn Pits in Iraq and Afghanistan. 31 October 2011. Available online: http://www.nationalacademies.org/hmd/Reports/2011/Long-Term-Health-Consequences-of-Exposure-to-Burn-Pits-in-Iraq-and-Afghanistan.aspx (accessed on 10 December 2018).
- Georgopoulos, A.P.; James, L.M.; Carpenter, A.F.; Engdahl, B.E.; Leuthold, A.C.; Lewis, S.M. Gulf War illness (GWI) as a neuroimmune disease. Exp. Brain Res. 2017, 235, 3217–3225. [Google Scholar] [CrossRef]
- Coughlin, S.S. A neuroimmune model of Gulf War Illness. J. Environ. Health Sci. 2017, 3. [Google Scholar] [CrossRef] [PubMed]
- Coughlin, S.S.; Krengel, M.; Sullivan, K.; Pierce, P.F.; Heboyan, V.; Wilson, L.C.C. A review of epidemiologic studies of the health of Gulf War women veterans. J. Environ. Health Sci. 2017, 3. [Google Scholar] [CrossRef] [PubMed]
- McGarvey, P.B.; Suzek, B.E.; Baraniuk, J.N.; Rao, S.; Conkright, B.; Lababid, S.; Sutherland, A.; Forshee, R.; Madhavan, S. In silico analysis of autoimmune diseases and genetic relationships to vaccination against infectious diseases. BMC Immunol. 2014, 15, 61. [Google Scholar] [CrossRef] [PubMed]
- Golomb, B.A. Acetylcholinesterase inhibitors and Gulf War illnesses. Proc. Natl. Acad. Sci. USA 2008, 105, 4295–4300. [Google Scholar] [CrossRef] [PubMed]
- Elia, M.; Lanham-New, S.A. Nutrition. In Kumar & Clark’s Clinical Medicine, 9th ed.; Kumar, P., Clark, M., Eds.; Elsevier Saunders: London, UK, 2017; pp. 183–218. [Google Scholar]
| Condition | GW Veterans vs. Gulf Era Veterans (% Difference) | Adjusted Odds Ratio and 95% Confidence Interval |
|---|---|---|
| Chronic multisymptom illness | 43.9% vs. 20.3% | 2.36; 1.94, 2.86 |
| Chronic fatigue syndrome | 11.8% vs. 5.3% | 2.36; 1.94, 2.86 |
| Neuralgia | 9.4% vs. 6.3% | 1.65; 1.40, 1.95 |
| Gastritis | 20.2% vs. 14.3% | 1.59; 1.35, 1.73 |
| Chronic obstructive pulmonary disease | 8.4% vs. 6.3% | 1.48; 1.23, 1.78 |
| Fibromyalgia | 3.7% vs. 2.9% | 1.48; 1.15, 1.91 |
| Tachycardia | 8.1% vs. 5.9% | 1.47; 1.20, 1.79 |
| Dermatitis | 27.4% vs. 21.1% | 1.44; 1.27, 1.63 |
| Rheumatoid arthritis | 9.9% vs. 7.9% | 1.40; 1.17, 1.67 |
| Seizures | 2.7% vs. 2.0% | 1.38; 1.03, 1.85 |
| Coronary heart disease | 5.6% vs. 5.3% | 1.32; 1.09, 1.59 |
| Migraine headaches | 20.3% vs. 16.1% | 1.30; 1.15, 1.47 |
| Hypertension | 43.0% vs. 40.0% | 1.22; 1.10, 1.35 |
| Asthma | 10.2% vs. 9.0% | 1.22; 1.04, 1.44 |
| Unspecified arthritis | 33.9% vs. 31.8% | 1.16; 1.05, 1.29 |
| Irritable bowel syndrome | 24.4% vs. 14.3% | 2.10; 1.79, 2.45 |
| Functional dyspepsia: | 27.7% vs. 15.9% | 1.94; 1.75, 2.17 |
| PTSD in the past 4 weeks | 20.9% vs. 11.5% | 1.93; 1.67, 2.24 |
| Major depressive disorder, past 2 weeks | 32.9% vs. 22.9% | 1.56; 1.41, 1.73 |
| Other depressive disorder, past 2 weeks | 23.5% vs. 19.1% | 1.24; 1.08, 1.38 |
| Other anxiety disorder, past 4 weeks | 18.7% vs. 14.4% | 1.34; 1.17, 1.54 |
| Somatic symptom severity, past 4 weeks | 16.1% vs. 8.3% | 2.10; 1.79, 2.45 |
| Vaccine | Military Personnel | Schedule/Dose |
|---|---|---|
| Adenovirus | All recruits | 1 oral dose |
| Influenza | All recruits and active duty | Annual shot |
| Measles | All recruits | 1 shot |
| Meningococcal | All recruits, active duty as required | 1st shot, then booster every 3–5 years |
| Plague | All Marines; Army and Navy special forces, others in at-risk occupations or deploying to high risk areas | 5 shots over 12 months, then booster every 1–2 years |
| Polio | All recruits | 1 oral dose |
| Rabies | Special forces, at-risk occupations | 3 shot series |
| Rubella | All recruits | 1 shot |
| Smallpox vaccine or booster | New recruits through the late 1980s | 1 dose |
| Tetanus-diphtheria | All recruits, active duty, and reserve | Booster every 10 years |
| Typhoid | Army and Air Force alert forces and for deployment to high risk areas | 2 doses in 2 months, then booster every 3 years |
| Yellow fever | All Navy and Marine Corps, Army and Air Force alert forces and for deployment to high risk areas | 1st shot, then booster every 10 years |
| Major Signs/Symptoms | Gulf War Illness | Chronic Hypervitaminosis A |
|---|---|---|
| Mood swings, irritability | + | + |
| Memory loss, lack of concentration | + | + |
| Anxiety, stress, sleep disturbance | + | + |
| Depression | + | + |
| Other psychiatric disorders | + | + |
| Muscular pain, weakness | + | + |
| General Fatigue | + | + |
| Fevers/Night sweats | + | + |
| Headaches | + | + |
| Numbness, tingling, dizziness | + | + |
| Sinus congestion | + | + |
| Chronic, frequent infection | + | + |
| Skin allergies, other allergies | + | + |
| Respiratory problems | + | + |
| Digestive, stomach and intestinal disorders | + | + |
| Weight gain/loss | + | + |
| Reproductive disorders | + | + |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mawson, A.R.; Croft, A.M. Gulf War Illness: Unifying Hypothesis for a Continuing Health Problem. Int. J. Environ. Res. Public Health 2019, 16, 111. https://doi.org/10.3390/ijerph16010111
Mawson AR, Croft AM. Gulf War Illness: Unifying Hypothesis for a Continuing Health Problem. International Journal of Environmental Research and Public Health. 2019; 16(1):111. https://doi.org/10.3390/ijerph16010111
Chicago/Turabian StyleMawson, Anthony R., and Ashley M. Croft. 2019. "Gulf War Illness: Unifying Hypothesis for a Continuing Health Problem" International Journal of Environmental Research and Public Health 16, no. 1: 111. https://doi.org/10.3390/ijerph16010111
APA StyleMawson, A. R., & Croft, A. M. (2019). Gulf War Illness: Unifying Hypothesis for a Continuing Health Problem. International Journal of Environmental Research and Public Health, 16(1), 111. https://doi.org/10.3390/ijerph16010111





