A Meta-Regression Analysis of the Effectiveness of Mosquito Nets for Malaria Control: The Value of Long-Lasting Insecticide Nets
Abstract
1. Introduction
2. Methods
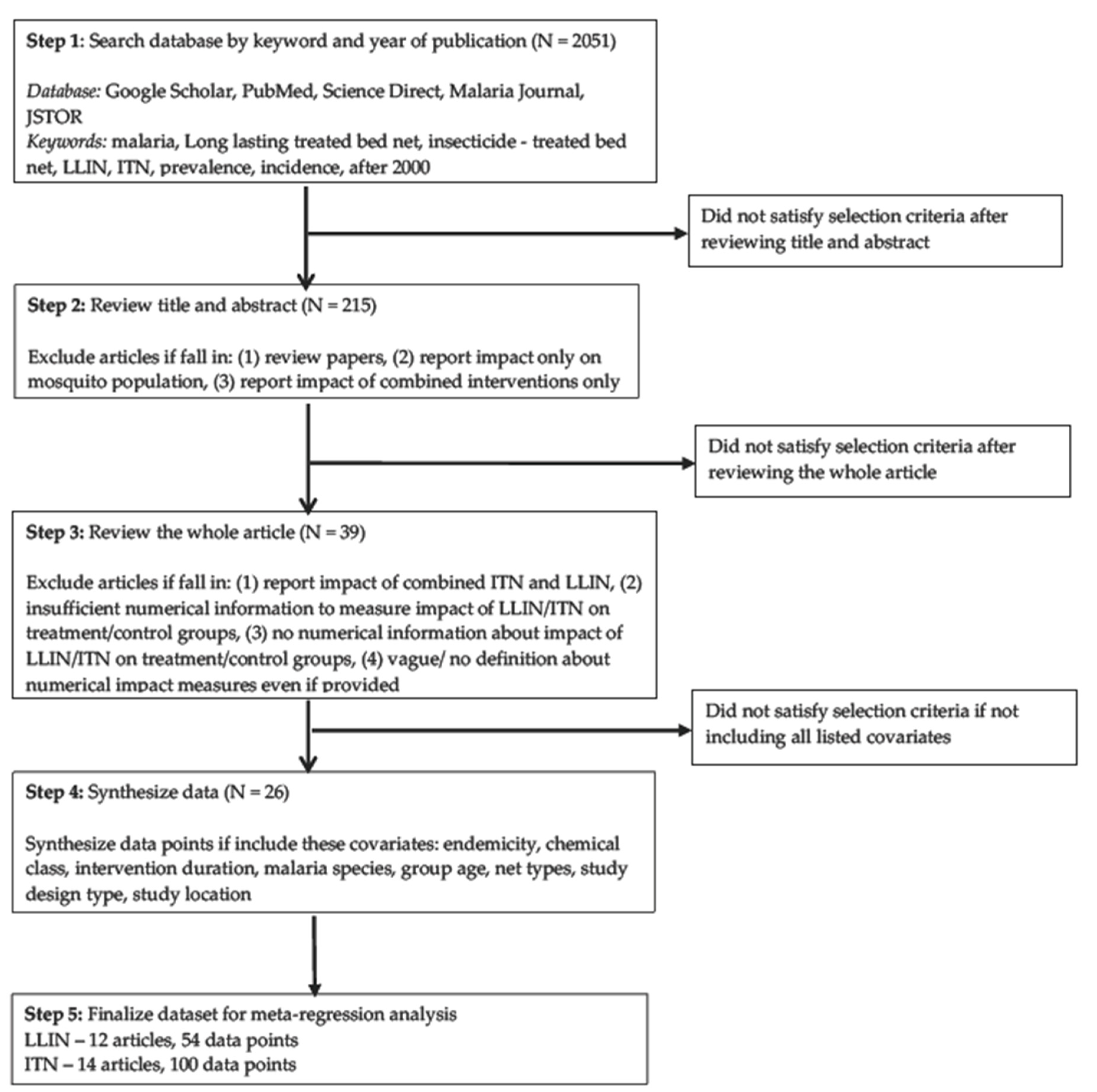
2.1. Database Search and Systematic Review
2.2. Data Collection and Analysis
3. Results
3.1 Description of Articles Included in the Final Data
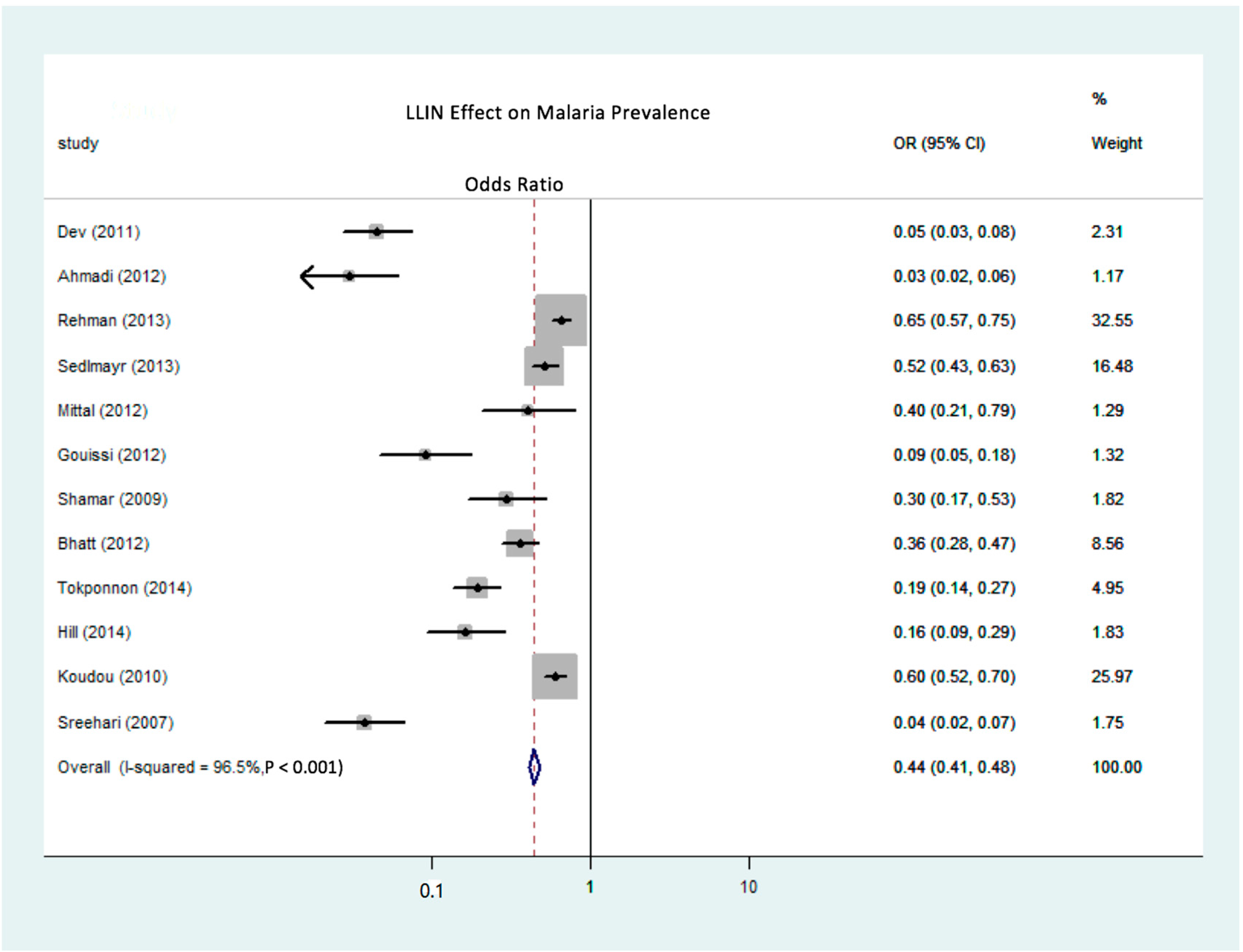
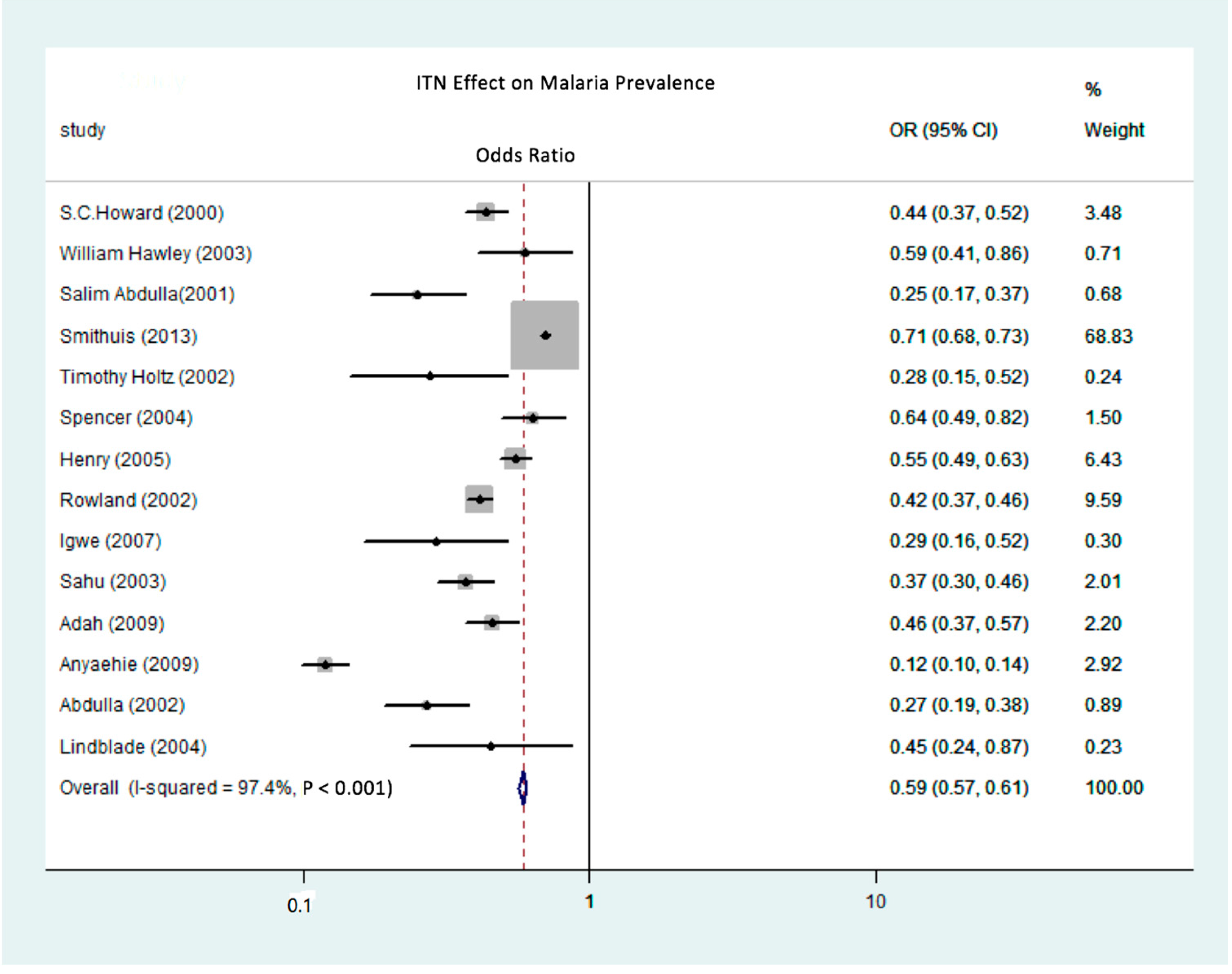
3.2. Meta-Regression Analysis
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- World Health Organization (WHO). World Malaria Report 2016; World Health Organization: Geneva, Switzerland, 2016. [Google Scholar]
- Abdulla, S.; Schellenberg, J.A.; Nathan, R.; Mukasa, O.; Marchant, T.; Smith, T.; Tanner, M.; Lengeler, C. Impact on malaria morbidity of a programme supplying insecticide treated nets in children aged under 2 years in Tanzania: Community cross sectional study. BMJ 2001, 322, 270–273. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Howard, S.C.; Omumbo, J.; Nevill, C.; Some, E.S.; Donnelly, C.A.; Snow, R.W. Evidence for a mass community effect of insecticide-treated bednets on the incidence of malaria on the Kenyan coast. Trans. R. Soc. Trop. Med. Hyg. 2000, 94, 357–360. [Google Scholar] [CrossRef]
- Lengeler, C. Insecticide-treated bed nets and curtains for preventing malaria. Cochrane Database Syst. Rev. 2004, CD000363. [Google Scholar] [CrossRef] [PubMed]
- Rowland, M.; Webster, J.; Saleh, P.; Chandramohan, D.; Freeman, T.; Pearcy, B.; Durrani, N.; Rab, A.; Mohammed, N. Prevention of malaria in Afghanistan through social marketing of insecticide-treated nets: Evaluation of coverage and effectiveness by cross-sectional surveys and passive surveillance. Trop. Med. Int. Health 2002, 7, 813–822. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.W.; Breman, J.G.; Teutsch, S.M.; Liu, S.; Hightower, A.W.; Sexton, J.D. The effectiveness of insecticide-impregnated bednets in reducing cases of malaria infection: A meta-analysis of published results. Am. J. Trop. Med. Hyg. 1995, 52, 377–382. [Google Scholar] [CrossRef] [PubMed]
- Hawley, W.A.; Phillips-Howard, P.A.; ter Kuile, F.O.; Terlouw, D.J.; Vulule, J.M.; Ombok, M.; Nahlen, B.L.; Gimnig, J.E.; Kariuki, S.K.; Kolczak, M.S.; et al. Community-wide effects of permethrin-treated bed nets on child mortality and malaria morbidity in western Kenya. Am. J. Trop. Med. Hyg. 2003, 68 (Suppl. 4), 121–127. [Google Scholar] [PubMed]
- World Health Organization (WHO). Instructions for Treatment and Use of Insecticide-Treated Mosquito Nets; World Health Organization: Geneva, Switzerland, 2002. [Google Scholar]
- Dev, V.; Phookan, S.; Padhan, K.; Tewari, G.G.; Khound, K. Laboratory wash-resistance and field evaluation of deltamethrin incorporated long-lasting polyethylene netting (Netprotect (R)) against malaria transmission in Assam, north-east India. Acta Trop. 2011, 119, 172–177. [Google Scholar] [CrossRef] [PubMed]
- Dabire, R.K.; Diabate, A.; Baldet, T.; Pare-Toe, L.; Guiguemde, R.T.; Ouedraogo, J.B.; Skovmand, O. Personal protection of long lasting insecticide-treated nets in areas of Anopheles gambiae s.s. resistance to pyrethroids. Malar. J. 2006, 5, 12. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Patipong, S.; Yongchaitrakul, S. Field efficacy and persistence of Long Lasting Insecticide treated mosquito Nets (LLINs) in comparison with conventional Insecticide Treated mosquito Nets (ITN) against malaria vector in Thailand. J. Vector Borne Dis. 2008, 5, 7–13. [Google Scholar]
- Guillet, P.; Alnwick, D.; Cham, M.K.; Neira, M.; Zaim, M.; Heymann, D.; Mukelabai, K. Long-lasting treated mosquito nets: A breakthrough in malaria prevention. Bull. World Health Organ. 2001, 79, 998. [Google Scholar] [PubMed]
- World Health Organization (WHO). WHO Recommendations for Achieving Universal Coverage with Long-Lasting Insecticidal Nets in Malaria Control; World Health Organization: Geneva, Switzerland, 2014; Available online: http://www.who.int/malaria/publications/atoz/who_recommendation_coverage_llin/en/ (accessed on 23 September 2017).
- Yukich, J.; Tediosi, F.; Lengeler, C. Comparative Cost-Effectiveness of ITNs or IRS in Sub-Saharan Africa; Newsletter July 2007; Swiss Tropical Institute: Basel, Switzerland, 2007. [Google Scholar]
- Programme, G.M. Insecticide-Treated Mosquito Nets: A WHO Position Statement; World Health Organization: Geneva, Switzerland, 2007. [Google Scholar]
- Yukich, J.O.; Lengeler, C.; Tediosi, F.; Brown, N.; Mulligan, J.A.; Chavasse, D.; Stevens, W.; Justino, J.; Conteh, L.; Maharaj, R.; et al. Costs and consequences of large-scale vector control for malaria. Malar. J. 2008, 7, 258. [Google Scholar] [CrossRef] [PubMed]
- Graham, K.; Kayedi, M.H.; Maxwell, C.; Kaur, H.; Rehman, H.; Malima, R.; Curtis, C.F.; Lines, J.D.; Rowland, M.W. Multi-country field trials comparing wash-resistance of PermaNet and conventional insecticide-treated nets against anopheline and culicine mosquitoes. Med. Vet. Entomol. 2005, 19, 72–83. [Google Scholar] [CrossRef] [PubMed]
- Sreehari, U.; Raghavendra, K.; Rizvi, M.M.; Dash, A.P. Wash resistance and efficacy of three long-lasting insecticidal nets assessed from bioassays on Anopheles culicifacies and Anopheles stephensi. Trop. Med. Int. Health 2009, 14, 597–602. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.; Fedak, K.; Kramer, R. Reduction of malaria prevalence by indoor residual spraying: A meta-regression analysis. Am. J. Trop. Med. Hyg. 2012, 87, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Fullman, N.; Burstein, R.; Lim, S.S.; Medlin, C.; Gakidou, E. Nets, spray or both? The effectiveness of insecticide-treated nets and indoor residual spraying in reducing malaria morbidity and child mortality in sub-Saharan Africa. Malar. J. 2013, 12, 62. [Google Scholar] [CrossRef] [PubMed]
- Haidich, A.B. Meta-analysis in medical research. Hippokratia 2010, 14 (Suppl. 1), 29. [Google Scholar] [PubMed]
- Thompson, S.G.; Higgins, J. How should meta-regression analyses be undertaken and interpreted? Stat. Med. 2002, 21, 1559–1573. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Children: Reducing Mortality; Retrieved 3 March 2018; World Health Organization: Geneva, Switzerland, 2017; Available online: http://www.who.int/mediacentre/factsheets/fs178/en/ (accessed on 3 March 2018).
- Sreehari, U.; Razdan, R.K.; Mittal, P.K.; Ansari, M.A.; Rizvi, M.M.; Dash, A.P. Impact of Olyset nets on malaria transmission in India. J. Vector Borne Dis. 2007, 44, 137–144. [Google Scholar] [PubMed]
- Adah, P.O.; Mafiana, C.F.; Sam-Wobo, S.O. Impact assessment of the use of insecticide-treated bed nets on parasitaemia and anaemia for malaria control in children, Ogun State, Nigeria. Public Health 2009, 123, 390–392. [Google Scholar] [CrossRef] [PubMed]
- Holtz, T.H.; Marum, L.H.; Mkandala, C.; Chizani, N.; Roberts, J.M.; Macheso, A.; Parise, M.E.; Kachur, S.P. Insecticide-treated bednet use, anaemia, and malaria parasitaemia in Blantyre District, Malawi. Trop. Med. Int. Health 2002, 7, 220–230. [Google Scholar] [CrossRef] [PubMed]
- Lindblade, K.A.; Eisele, T.P.; Gimnig, J.E.; Alaii, J.A.; Odhiambo, F.; ter Kuile, F.O.; Hawley, W.A.; Wannemuehler, K.A.; Phillips-Howard, P.A.; Rosen, D.H.; et al. Sustainability of reductions in malaria transmission and infant mortality in western Kenya with use of insecticide-treated bednets: 4 to 6 years of follow-up. J. Am. Med. Assoc. 2004, 291, 2571–2580. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sahu, S.S.; Jambulingam, P.; Vijayakumar, T.; Subramanian, S.; Kalyanasundaram, M. Impact of alphacypermethrin treated bed nets on malaria in villages of Malkangiri district, Orissa, India. Acta Trop. 2003, 89, 55–66. [Google Scholar] [CrossRef] [PubMed]
- Smithuis, F.M.; Kyaw, M.K.; Phe, U.O.; van der Broek, I.; Katterman, N.; Rogers, C.; Almeida, P.; Kager, P.A.; Stepniewska, K.; Lubell, Y.; et al. The effect of insecticide-treated bed nets on the incidence and prevalence of malaria in children in an area of unstable seasonal transmission in western Myanmar. Malar. J. 2013, 12, 363. [Google Scholar] [CrossRef] [PubMed]
- Abdulla, S.; Schellenberg, J.R.; Mukasa, O.; Lengeler, C. Usefulness of a dispensary-based case-control study for assessing morbidity impact of a treated net programme. Int. J. Epidemiol. 2002, 31, 175–180. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Igwe, P.C.; Inem, V.; Ebuehi, O.M.; Afolabi, B.M. The effect of insecticide treated bed net use on malaria episodes, parasitaemia and haemoglobin concentration among primigravidae in a peri-urban settlement in southeast Nigeria. J. Rural Trop. Public Health 2007, 6, 24–32. [Google Scholar]
- Spencer, S.; Grant, A.D.; Piola, P.; Tukpo, K.; Okia, M.; Garcia, M.; Salignon, P.; Genevier, C.; Kiguli, J.; Guthmann, J.P. Malaria in camps for internally-displaced persons in Uganda: Evaluation of an insecticide-treated bednet distribution programme. Trans. R. Soc. Trop. Med. Hyg. 2004, 98, 719–727. [Google Scholar] [CrossRef] [PubMed]
- Anyaehie, U.; Nwagha, U.I.; Aniebue, P.N.; Nwagha, T.U. The effect of free distribution of insecticide-treated nets on asymptomatic Plasmodium parasitemia in pregnant and nursing mothers in a rural Nigerian community. Niger. J. Clin. Pract. 2011, 14, 19–22. [Google Scholar] [PubMed]
- Henry, M.C.; Assi, S.B.; Rogier, C.; Dossou-Yovo, J.; Chandre, F.; Guillet, P.; Carnevale, P. Protective efficacy of lambda-cyhalothrin treated nets in Anopheles gambiae pyrethroid resistance areas of Cote d'Ivoire. Am. J. Trop. Med. Hyg. 2005, 73, 859–864. [Google Scholar] [PubMed]
- Bhatt, R.M.; Sharma, S.N.; Uragayala, S.; Dash, A.P.; Kamaraju, R. Effectiveness and durability of Interceptor(R) long-lasting insecticidal nets in a malaria endemic area of central India. Malar. J. 2012, 11, 189. [Google Scholar] [CrossRef] [PubMed]
- Hill, N.; Zhou, H.N.; Wang, P.; Guo, X.; Carneiro, I.; Moore, S.J. A household randomized, controlled trial of the efficacy of 0.03% transfluthrin coils alone and in combination with long-lasting insecticidal nets on the incidence of Plasmodium falciparum and Plasmodium vivax malaria in Western Yunnan Province, China. Malar. J. 2014, 13, 208. [Google Scholar] [CrossRef] [PubMed]
- Mittal, P.K.; Sood, R.D.; Kapoor, N.; Razdan, R.K.; Dash, A.P. Field evaluation of Icon(R)Life, a long-lasting insecticidal net (LLIN) against Anopheles culicifacies and transmission of malaria in District Gautam Budh Nagar (Uttar Pradesh), India. J. Vector Borne Dis. 2012, 49, 181–187. [Google Scholar] [PubMed]
- Sedlmayr, R.; Fink, G.; Miller, J.M.; Earle, D.; Steketee, R.W. Health impact and cost-effectiveness of a private sector bed net distribution: Experimental evidence from Zambia. Malar. J. 2013, 12, 102. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.K.; Tyagi, P.K.; Upadhyay, A.K.; Haque, M.A.; Mohanty, S.S.; Raghavendra, K.; Dash, A.P. Efficacy of permethrin treated long-lasting insecticidal nets on malaria transmission and observations on the perceived side effects, collateral benefits and human safety in a hyperendemic tribal area of Orissa, India. Acta Trop. 2009, 112, 181–187. [Google Scholar] [CrossRef] [PubMed]
- Modeste Gouissi, F.; Salifou, S.; Patrick Edorh, A.; Anges Yadouleton, W.; Djenontin, A.; Bio-Banganna, S.; Geoffroy Augustin Gouissi, S.; Akogbeto, M. Assessment of Long-Lasting Insecticidal Nets (LLINs) on Vectors and Malaria Transmission in the Commune of Aguegues, Benin. BioImpacts 2012, 2, 159–166. [Google Scholar] [PubMed]
- Rehman, A.M.; Mann, A.G.; Schwabe, C.; Reddy, M.R.; Roncon Gomes, I.; Slotman, M.A.; Yellott, L.; Matias, A.; Caccone, A.; Nseng Nchama, G.; et al. Five years of malaria control in the continental region, Equatorial Guinea. Malar. J. 2013, 12, 154. [Google Scholar] [CrossRef] [PubMed]
- Tokponnon, F.T.; Ogouyemi, A.H.; Sissinto, Y.; Sovi, A.; Gnanguenon, V.; Cornelie, S.; Adeothy, A.A.; Osse, R.; Wakpo, A.; Gbenou, D.; et al. Impact of long-lasting, insecticidal nets on anaemia and prevalence of Plasmodium falciparum among children under five years in areas with highly resistant malaria vectors. Malar. J. 2014, 13, 76. [Google Scholar] [CrossRef] [PubMed]
- Koudou, B.G.; Ghattas, H.; Esse, C.; Nsanzabana, C.; Rohner, F.; Utzinger, J.; Faragher, B.E.; Tschannen, A.B. The use of insecticide-treated nets for reducing malaria morbidity among children aged 6–59 months, in an area of high malaria transmission in central Cote d’Ivoire. Parasit. Vectors 2010, 3, 91. [Google Scholar] [CrossRef] [PubMed]
- Soleimani-Ahmadi, M.; Vatandoost, H.; Shaeghi, M.; Raeisi, A.; Abedi, F.; Eshraghian, M.R.; Madani, A.; Safari, R.; Oshaghi, M.A.; Abtahi, M.; et al. Field evaluation of permethrin long-lasting insecticide treated nets (Olyset (R)) for malaria control in an endemic area, southeast of Iran. Acta Trop. 2012, 123, 146–153. [Google Scholar] [CrossRef] [PubMed]
- Jadad, A.R.; Enkin, M.W. Bias in Randomized Controlled Trials. In Randomized Controlled Trials: Questions, Answers, and Musings, 2nd ed.; Blackwell Publishing Ltd.: Oxford, UK, 2007. [Google Scholar]
- Lam, Y.; Harvey, S.A.; Monroe, A.; Muhangi, D.; Loll, D.; Kabali, A.T.; Weber, R. Decision-making on intra-household allocation of bed nets in Uganda: Do households prioritize the most vulnerable members? Malar. J. 2014, 13, 183. [Google Scholar] [CrossRef] [PubMed]
- Ricotta, E.; Koenker, H.; Kilian, A.; Lynch, M. Are pregnant women prioritized for bed nets? An assessment using survey data from 10 African countries. Glob. Health Sci. Pract. 2014, 2, 165–172. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Toe, L.P.; Skovmand, O.; Dabire, K.R.; Diabate, A.; Diallo, Y.; Guiguemde, T.R.; Doannio, J.M.; Akogbeto, M.; Baldet, T.; Gruenais, M.E. Decreased motivation in the use of insecticide-treated nets in a malaria endemic area in Burkina Faso. Malar. J. 2009, 8, 175. [Google Scholar] [CrossRef] [PubMed]
- Frey, C.; Traore, C.; De Allegri, M.; Kouyate, B.; Muller, O. Compliance of young children with ITN protection in rural Burkina Faso. Malar. J. 2006, 5, 70. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Nuwaha, F. People’s perception of malaria in Mbarara, Uganda. Trop. Med. Int. Health 2002, 7, 462–470. [Google Scholar] [CrossRef] [PubMed]
- Ye, Y.; Patton, E.; Kilian, A.; Dovey, S.; Eckert, E. Can universal insecticide-treated net campaigns achieve equity in coverage and use? the case of northern Nigeria. Malar. J. 2012, 11, 32. [Google Scholar] [CrossRef] [PubMed]
- Zollner, C.; De Allegri, M.; Louis, V.R.; Ye, M.; Sie, A.; Tiendrebeogo, J.; Jahn, A.; Muller, O. Insecticide-treated mosquito nets in rural Burkina Faso: Assessment of coverage and equity in the wake of a universal distribution campaign. Health Policy Plan 2015, 30, 171–180. [Google Scholar] [CrossRef] [PubMed]
- Roll Back Malaria-World Health Organization (RBM-WHO). World Malaria Report; World Health Organization: Geneva, Switzerland; United Nations Children Fund: Geneva, Switzerland, 2005. [Google Scholar]
- Tarozzi, A.; Mahajan, A.; Blackburn, B.; Kopf, D.; Krishnan, L.; Yoong, J. Micro-Loans, Insecticide-Treated Bednets, and Malaria: Evidence from a Randomized Controlled Trial in Orissa, India. Am. Econ. Rev. 2014, 104, 1909–1941. [Google Scholar] [CrossRef] [PubMed]
- Koenker, H.; Kilian, A. Recalculating the Net Use Gap: A Multi-Country Comparison of ITN Use versus ITN Access. PLoS ONE 2014, 9, e97496. [Google Scholar] [CrossRef] [PubMed]
| Covariates | ITN | LLIN | |
|---|---|---|---|
| Total Articles | 14 | 12 | |
| Study types | Randomized Control Trials | 6 | 5 |
| Cross-sectional | 6 | 4 | |
| Cohort | 2 | 3 | |
| Study locations | Africa | 11 | 5 |
| Asia | 3 | 7 | |
| Malaria species | P. falciparum only | 8 | 7 |
| P. falciparum and P. vivax | 4 | 4 | |
| Chemical class | Permethrin | 6 | 5 |
| Deltamethrin | 5 | 5 | |
| Alphapermethrin | 1 | 2 | |
| Age | Children under 5 | 9 | 4 |
| Covariate (Dependent Variable = log OR) | Coefficient (95% Confidence Interval) |
|---|---|
| Use of LLIN (only) | −0.954 (−1.419 to −0.49) *** |
| Log of initial prevalence (malaria proportion before intervention) | −0.224 (−0.355 to −0.092) *** |
| Total sample size (thousands) | −0.0066 (−0.076 to 0.062) |
| Study duration (more than 12 months) | −0.451 (−0.868 to −0.034) ** |
| Use of deltamethrin | 0.228 (−0.195 to 0.653) |
| Use of alphapermethrin | 0.399 (−0.263 to 1.063) |
| Children under the age of five | 1.313 (0.821 to 1.804) *** |
| Plasmodium falciparum (only) | −0.274 (−0.661 to 0.112) |
| Cohort study design | −0.336 (−0.845 to 0.171) |
| Cross-sectional study design | −0.7 (−1.301 to −0.099 ) ** |
| Constant | −0.980 (−1.594 to −0.367) *** |
| Adjusted R-square | 0.33 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, G.-g.; Kim, D.; Pham, A.; Paul, C.J. A Meta-Regression Analysis of the Effectiveness of Mosquito Nets for Malaria Control: The Value of Long-Lasting Insecticide Nets. Int. J. Environ. Res. Public Health 2018, 15, 546. https://doi.org/10.3390/ijerph15030546
Yang G-g, Kim D, Pham A, Paul CJ. A Meta-Regression Analysis of the Effectiveness of Mosquito Nets for Malaria Control: The Value of Long-Lasting Insecticide Nets. International Journal of Environmental Research and Public Health. 2018; 15(3):546. https://doi.org/10.3390/ijerph15030546
Chicago/Turabian StyleYang, Gi-geun, Dohyeong Kim, Anh Pham, and Christopher John Paul. 2018. "A Meta-Regression Analysis of the Effectiveness of Mosquito Nets for Malaria Control: The Value of Long-Lasting Insecticide Nets" International Journal of Environmental Research and Public Health 15, no. 3: 546. https://doi.org/10.3390/ijerph15030546
APA StyleYang, G.-g., Kim, D., Pham, A., & Paul, C. J. (2018). A Meta-Regression Analysis of the Effectiveness of Mosquito Nets for Malaria Control: The Value of Long-Lasting Insecticide Nets. International Journal of Environmental Research and Public Health, 15(3), 546. https://doi.org/10.3390/ijerph15030546





