IQOSTM vs. e-Cigarette vs. Tobacco Cigarette: A Direct Comparison of Short-Term Effects after Overnight-Abstinence
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Materials
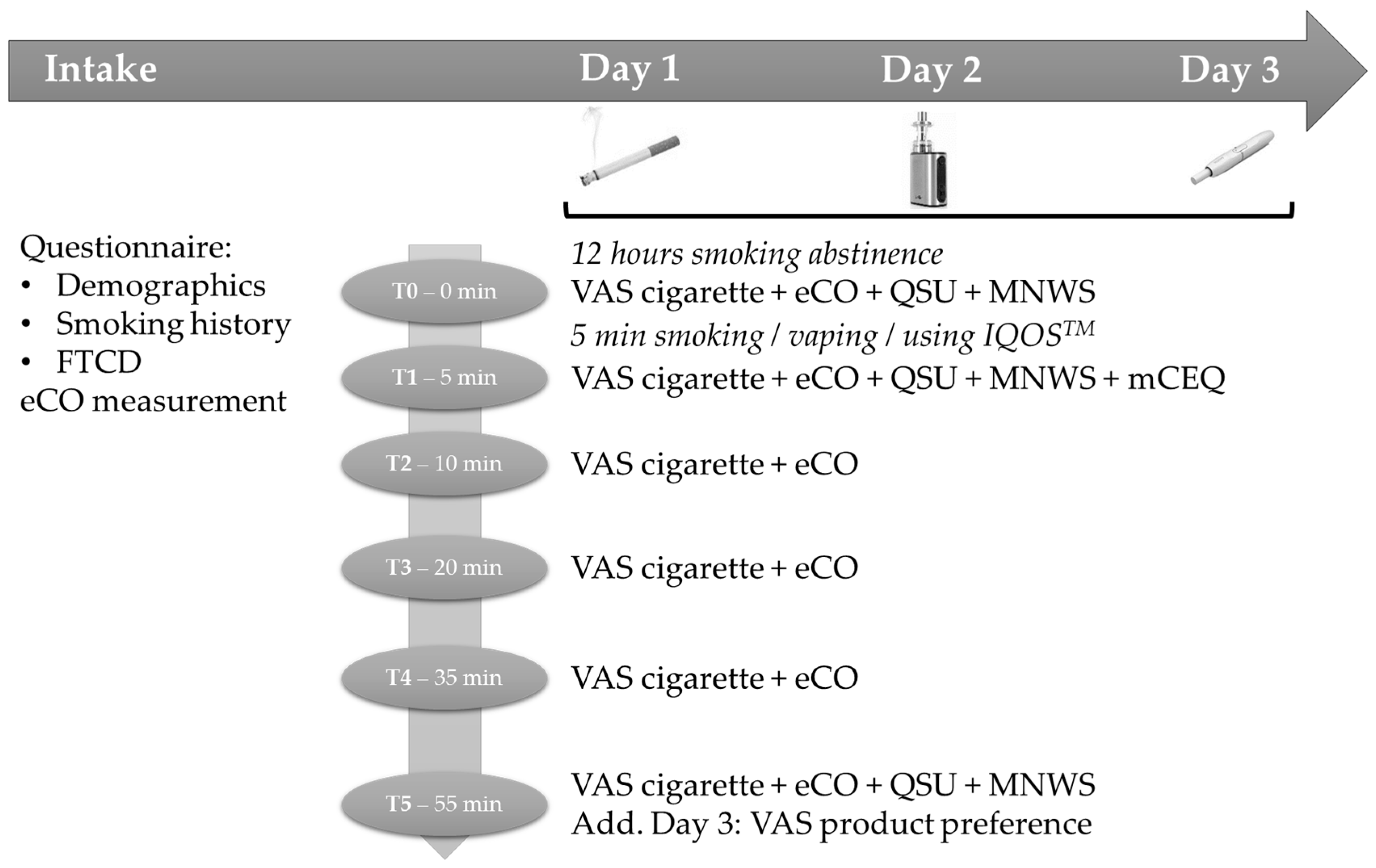
2.3. Study Design and Procedure
2.4. Outcome Measures
2.4.1. Physiological Measures
2.4.2. Subjective Effect Questionnaires
2.5. Statistical Analyses
3. Results
3.1. Participants
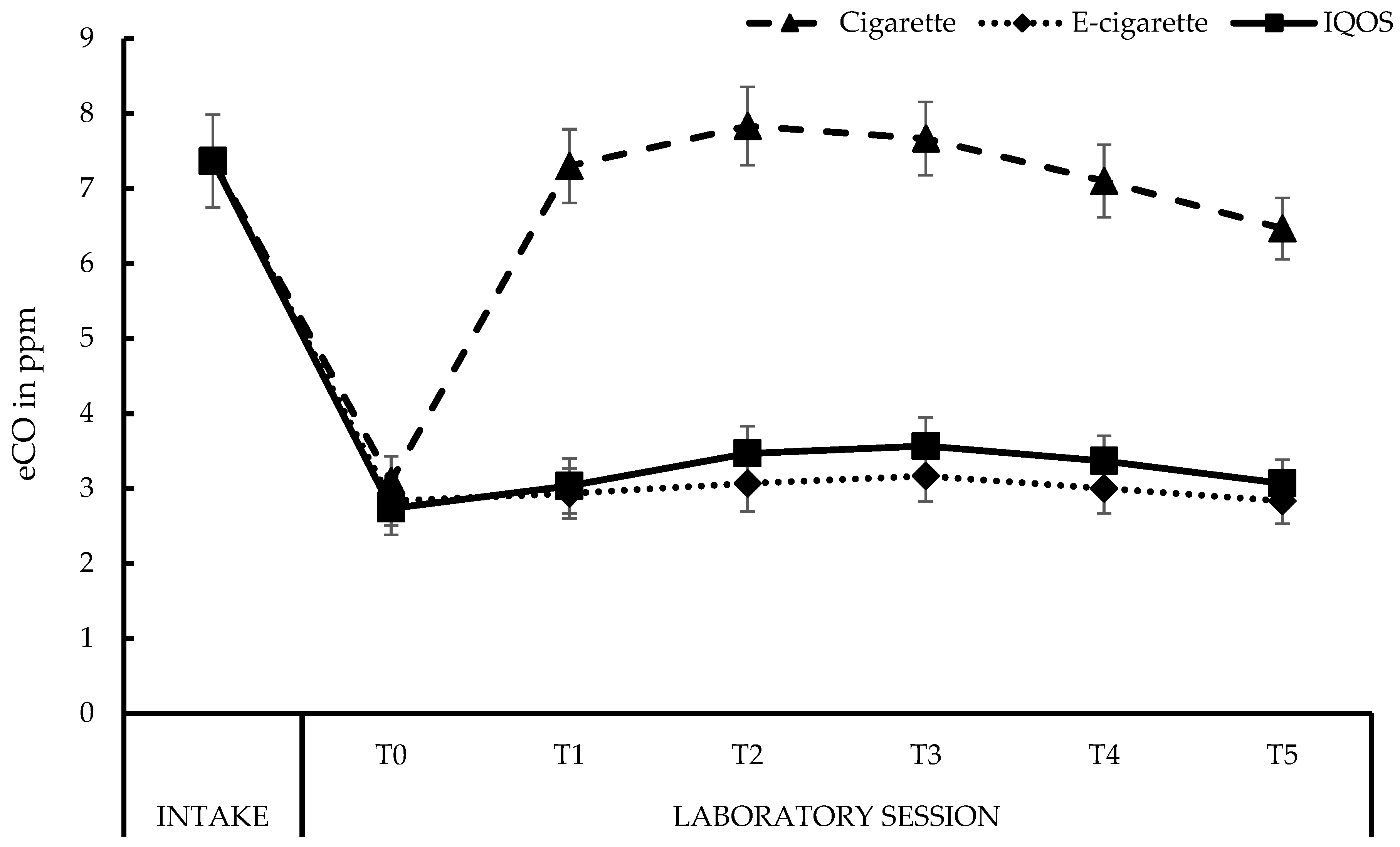
3.2. Physiological Measures
3.3. Subjective Effect Questionnaires
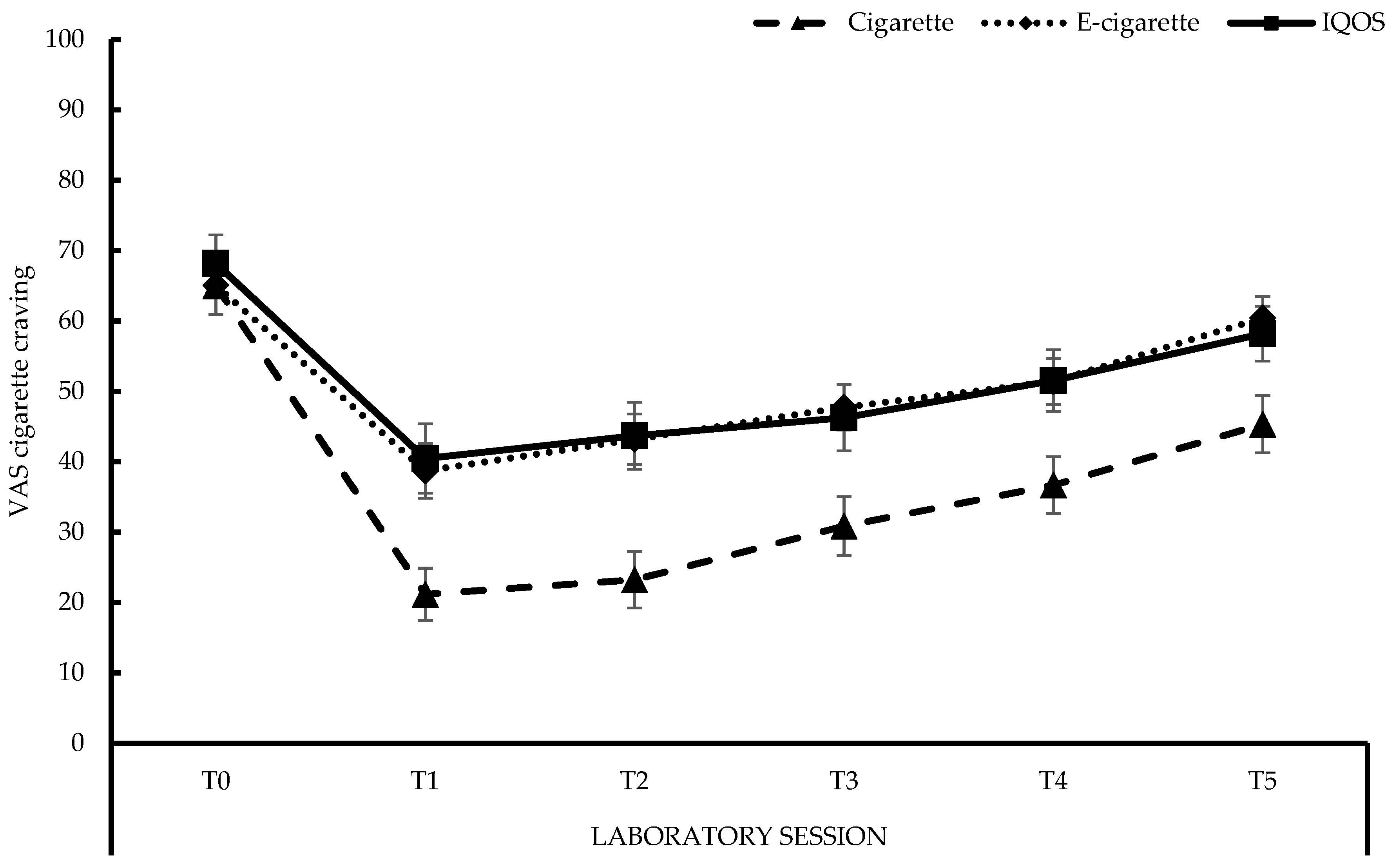
3.3.1. Cigarette Craving
3.3.2. Withdrawal Symptoms (MNWS-R)
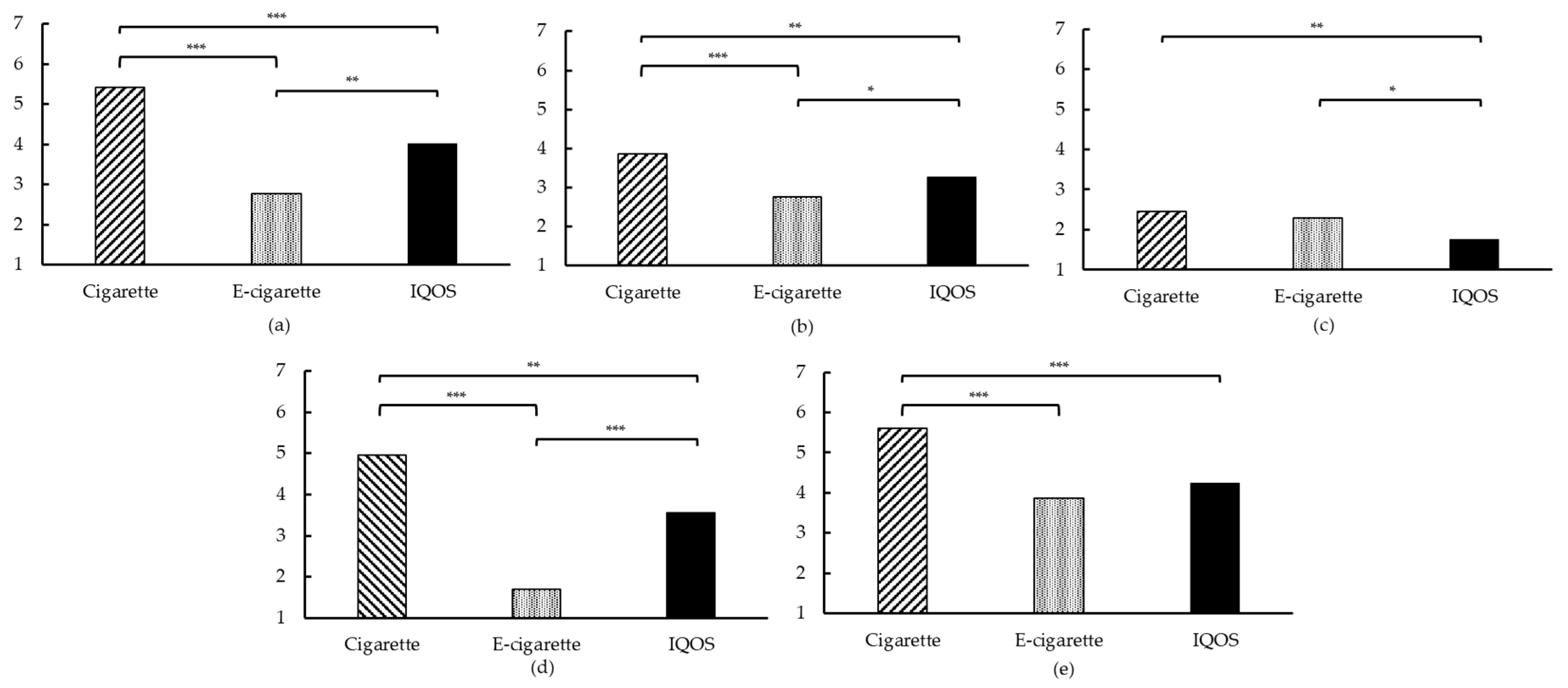
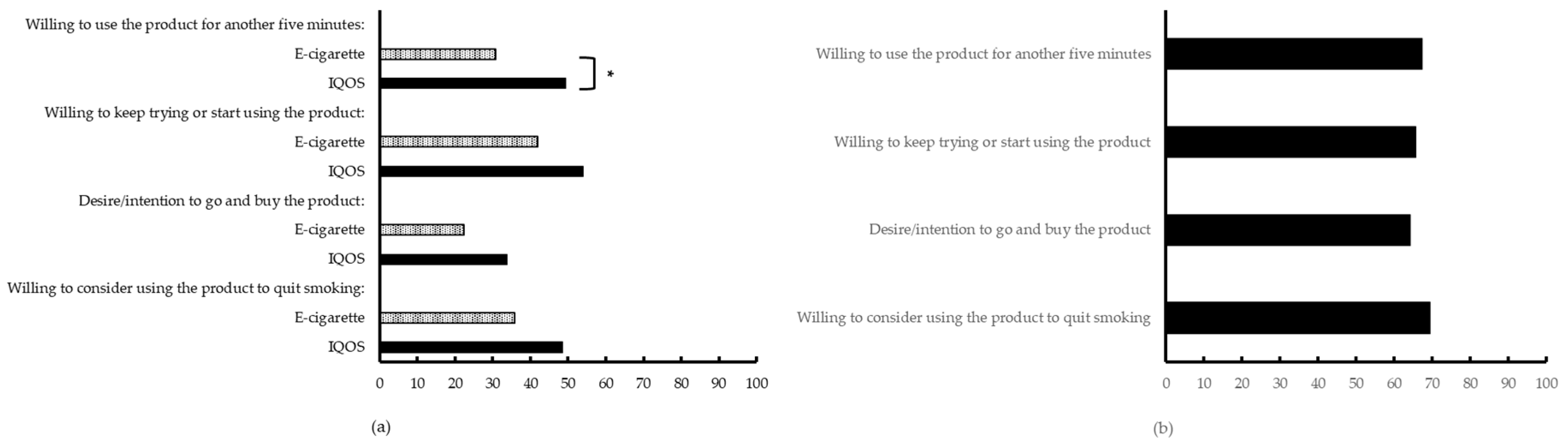
3.3.3. Product Evaluation and Preferences
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Variable | n | M (SD) or % |
|---|---|---|
| Smoking history | ||
| Age start smoking | 30 | 16.17 (1.84) |
| Age start smoking regularly | 30 | 17.90 (1.77) |
| Number of years smoking regularly | 30 | 4.47 (1.98) |
| Tried to quit smoking in the past (no/yes) | 20/10 | 66.67/33.33 |
| Number of past quit smoking attempts | 10 | 2.00 (0.94) |
| Quit-smoking aids used * | 10 | |
| E-cigarette | 1 | 10.00 |
| NRT | 0 | 0.00 |
| Smoking cessation medication | 0 | 0.00 |
| Counseling | 0 | 0.00 |
| None-willpower | 9 | 90.00 |
| Other (e.g., book) | 2 | 20.00 |
| Longest period quit smoking in months | 10 | 5.20 (9.02) |
| Quit-smoking aids used longest quit smoking period | 10 | |
| E-cigarette | 0 | 0.00 |
| NRT | 0 | 0.00 |
| Smoking cessation medication | 0 | 0.00 |
| Counseling | 0 | 0.00 |
| None-willpower | 10 | 100.00 |
| Other (e.g., book) | 0 | 0.00 |
| Current smoking behavior | ||
| CPD | 30 | 12.73 (3.62) |
| eCO | 30 | 7.37 (3.39) |
| Situations when smoking * | 30 | |
| Drinking alcohol | 29 | 96.67 |
| With others | 28 | 93.33 |
| After a meal | 26 | 86.67 |
| Alone | 25 | 83.33 |
| Going somewhere (car, walking, …) | 22 | 73.33 |
| At a café | 19 | 63.33 |
| At home | 18 | 60.00 |
| Drinking coffee | 14 | 46.67 |
| At work/school | 13 | 43.33 |
| Immediately after getting up | 12 | 40.00 |
| During stress | 10 | 33.33 |
| Other (e.g., during breaks, when waiting) | 10 | 33.33 |
| Watching TV | 5 | 16.67 |
| Reasons why smoking * | 30 | |
| For relaxation | 26 | 86.67 |
| Feeling like having a cigarette | 25 | 83.33 |
| For atmosphere and sociability | 24 | 80.00 |
| Out of habit | 23 | 76.67 |
| For pleasure and enjoyment of it | 21 | 70.00 |
| For stress reduction | 20 | 66.67 |
| Out of boredom | 12 | 40.00 |
| Other (e.g., pastime, gesture/handling) | 9 | 30.00 |
| For nicotine | 8 | 26.67 |
| Most important cigarette | 30 | |
| First one of the day | 8 | 26.67 |
| After a meal | 6 | 20.00 |
| Last one of the day | 6 | 20.00 |
| After class/work | 4 | 13.33 |
| Other (e.g., with coffee, with friends) | 6 | 20.00 |
| Cigarette brand | 25 | |
| Lucky strike | 7 | 28.00 |
| Marlboro | 7 | 28.00 |
| Camel or Lucky strike | 3 | 12.00 |
| Camel | 3 | 12.00 |
| Other (e.g., Tigra, Wintson) | 5 | 20.00 |
| Cigarette type | 29 | |
| Filter cigarette | 23 | 79.31 |
| Self-rolled cigarette | 5 | 17.24 |
| Filter and self-rolled cigarettes | 1 | 3.45 |
| Currently trying to reduce smoking (no/yes) | 26/4 | 86.67/13.33 |
| Motivation to quit smoking | 30 | |
| No intention of quitting at all | 26 | 86.67 |
| No intention of quitting at all, but open for less harmful alternatives | 1 | 3.33 |
| Considering quitting smoking, but no concrete plans | 2 | 6.67 |
| Concrete plans to quit smoking in next 3 months | 1 | 3.33 |
| Experienced negative health effects of smoking | 30 | 2.23 (0.48) |
| FTCD-score | 30 | 3.50 (1.96) |
| Suffering from any psychological/psychiatric condition | 30 | |
| No | 29 | 96.67 |
| Yes | 0 | 0.00 |
| Prefer not to say | 1 | 3.33 |
| Source | df | MS | F | p |
|---|---|---|---|---|
| Intercept | 1 | 13897.91 | 215.56 | 0.0000 *** |
| Error | 29 | 64.47 | ||
| Product | 2 | 628.11 | 71.00 | 0.0000 *** |
| Error | 58 | 8.85 | ||
| Moment | 6 | 162.97 | 37.40 | 0.0000 *** |
| Error | 174 | 4.36 | ||
| Product × Moment | 12 | 38.95 | 70.39 | 0.0000 *** |
| Error | 348 | 0.55 |
| VAS Cigarette Craving | ||||
| Source | df | MS | F | p |
| Intercept | 1 | 1167987.03 | 243.41 | 0.000 *** |
| Error | 29 | 4798.49 | ||
| Product | 2 | 12092.99 | 12.49 | 0.000 *** |
| Error | 58 | 968.26 | ||
| Moment a | 5 | 13317.59 | 54.20 | 0.000 *** |
| Error | 145 | 245.73 | ||
| Product × Moment | 10 | 462.33 | 5.75 | 0.000 *** |
| Error | 290 | 80.45 | ||
| QSU-Brief–Total score | ||||
| Source | df | MS | F | p |
| Intercept | 1 | 2857.73 | 392.44 | 0.0000 *** |
| Error | 29 | 7.28 | ||
| Product | 2 | 9.23 | 10.29 | 0.0001*** |
| Error | 58 | 0.90 | ||
| Moment b | 2 | 88.68 | 82.98 | 0.0000 *** |
| Error | 58 | 1.07 | ||
| Product × Moment | 4 | 1.38 | 3.61 | 0.0082 ** |
| Error | 116 | 0.38 | ||
| QSU-Brief–Subscale “The desire and intention to smoke with an anticipation of pleasure from smoking” | ||||
| Source | df | MS | F | p |
| Intercept | 1 | 4612.80 | 518.69 | 0.0000 *** |
| Error | 29 | 8.89 | ||
| Product | 2 | 16.11 | 11.45 | 0.0001 *** |
| Error | 58 | 1.41 | ||
| Moment b | 2 | 154.21 | 107.43 | 0.0000 *** |
| Error | 58 | 1.44 | ||
| Product × Moment | 4 | 3.48 | 5.79 | 0.0002 *** |
| Error | 116 | 0.60 | ||
| QSU-Brief–Subscale “The relief from nicotine withdrawal or negative affect with an urgent and overwhelming desire to smoke” | ||||
| Source | df | MS | F | p |
| Intercept | 1 | 1266.06 | 179.51 | 0.0000 *** |
| Error | 29 | 7.05 | ||
| Product | 2 | 2.87 | 4.30 | 0.0182 * |
| Error | 58 | 0.67 | ||
| Moment b | 2 | 29.98 | 36.32 | 0.0000 *** |
| Error | 58 | 0.83 | ||
| Product × Moment | 4 | 0.16 | 0.60 | 0.6635 |
| Error | 116 | 0.27 | ||
| Source | df | MS | F | p |
|---|---|---|---|---|
| Intercept | 1 | 34138.13 | 129.52 | 0.0000 *** |
| Error | 29 | 263.58 | ||
| Product | 2 | 238.14 | 5.80 | 0.0050 ** |
| Error | 58 | 41.03 | ||
| Moment | 2 | 622.34 | 32.60 | 0.0000 *** |
| Error | 58 | 19.09 | ||
| Product × Moment | 4 | 42.52 | 3.78 | 0.0063 ** |
| Error | 116 | 11.24 |
| Subscale “Satisfaction” | ||||
| Source | df | MS | F | p |
| Intercept | 1 | 1491.11 | 765.65 | 0.0000 *** |
| Error | 29 | 1.95 | ||
| Product | 2 | 52.06 | 37.87 | 0.0000 *** |
| Error | 58 | 1.37 | ||
| Subscale “Psychological reward” | ||||
| Source | df | MS | F | p |
| Intercept | 1 | 978.78 | 405.23 | 0.0000 *** |
| Error | 29 | 2.42 | ||
| Product | 2 | 9.21 | 14.01 | 0.0000 *** |
| Error | 58 | 0.66 | ||
| Subscale “Aversion” | ||||
| Source | df | MS | F | p |
| Intercept | 1 | 422.50 | 165.20 | 0.0000 *** |
| Error | 29 | 2.56 | ||
| Product | 2 | 4.08 | 4.62 | 0.0138 * |
| Error | 58 | 0.88 | ||
| Subscale “Enjoyment of respiratory tract sensations” | ||||
| Source | df | MS | F | p |
| Intercept | 1 | 1047.21 | 513.67 | 0.0000 *** |
| Error | 29 | 2.04 | ||
| Product | 2 | 80.58 | 36.65 | 0.0000 *** |
| Error | 58 | 2.20 | ||
| Subscale “Craving reduction” | ||||
| Source | df | MS | F | p |
| Intercept | 1 | 1876.90 | 673.92 | 0.0000 *** |
| Error | 29 | 2.79 | ||
| Product | 2 | 25.03 | 11.78 | 0.0001 *** |
| Error | 58 | 2.13 | ||
| Variable–VAS | E-CIG M (SD) | HnB Product M (SD) | Statistic | p-Value |
|---|---|---|---|---|
| Willing to use the product for another five minutes | 30.70 (27.87) | 49.47 (29.34) | t(29) = −2.36 | 0.0251 * |
| Willing to keep trying or start using the product | 41.93 (30.82) | 53.90 (29.52) | t(29) = −1.42 | 0.1673 |
| Desire/intention to go and buy the product | 22.43 (26.90) | 33.83 (27.38) | t(29) = −1.79 | 0.0842 |
| Willing to consider using the product to (try to) quit smoking | 35.83 (29.64) | 48.60 (34.63) | t(29) = −1.56 | 0.1286 |
References
- Smoking & Tobacco Use—Fast Facts. Available online: https://www.cdc.gov/tobacco/data_statistics/fact_sheets/fast_facts/index.htm (accessed on 15 October 2018).
- Utter Innumeracy: Six Impossible Claims about Tobacco Most “Public Health” People Believe before Breakfast. Available online: https://antithrlies.com/2015/12/23/utter-innumeracy-six-impossible-claims-about-tobacco-most-public-health-people-believe-before-breakfast/ (accessed on 15 October 2018).
- Fact Sheets—Tobacco. Available online: http://www.who.int/news-room/fact-sheets/detail/tobacco (accessed on 27 August 2018).
- Bauld, L.; Hiscock, R.; Dobbie, F.; Aveyard, P.; Coleman, T.; Leonardi-Bee, J.; McRobbie, H.; McEwen, A. English stop-smoking services: One-year outcomes. Int. J. Environ. Res. Public Health 2016, 13, 1175. [Google Scholar] [CrossRef] [PubMed]
- Cahill, K.; Stevens, S.; Lancaster, T. Pharmacological treatments for smoking cessation. JAMA 2014, 311, 193–194. [Google Scholar] [CrossRef] [PubMed]
- Hughes, J.R.; Keely, J.; Naud, S. Shape of the relapse curve and long-term abstinence among untreated smokers. Addiction 2004, 99, 29–38. [Google Scholar] [CrossRef] [PubMed]
- Schlam, T.T.; Baker, T.B. Interventions for tobacco smoking. Annu. Rev. Clin. Psychol. 2013, 9, 675–702. [Google Scholar] [CrossRef] [PubMed]
- McNeill, A.; Munafò, M.R. Reducing harm from tobacco use. J. Psychopharmacol. 2013, 27, 13–18. [Google Scholar] [CrossRef] [PubMed]
- What Is Tobacco Harm Reduction? Available online: https://antithrlies.com/2017/09/18/what-is-tobacco-harm-reduction/ (accessed on 15 October 2018).
- Rodu, B.; Godshall, W.T. Tobacco harm reduction: An alternative cessation strategy for inveterate smokers. Harm Reduct. J. 2006, 3. [Google Scholar] [CrossRef] [PubMed]
- Warner, K.E. How to think—Not feel—About tobacco harm reduction. Nicotine Tob. Res. 2018, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Rodu, B. The scientific foundation for tobacco harm reduction, 2006-2011. Harm Reduct. J. 2011, 8. [Google Scholar] [CrossRef]
- McNeill, A.; Brose, L.S.; Calder, R.; Bauld, L.; Robson, D. Evidence Review of E-Cigarettes and Heated Tobacco Products 2018; A report commissioned by Public Health England London; Public Health England: London, UK, 2018. [Google Scholar]
- Public Health Consequences of E-Cigarettes; The National Academies Press: Washington, DC, USA; Available online: https://www.nap.edu/catalog/24952/public-health-consequences-of-e-cigarettes (accessed on 27 August 2018).
- Royal College of Physicians. Nicotine without Smoke: Tobacco Harm Reduction; RCP: London, UK, 2016. [Google Scholar]
- Burstyn, I. Peering through the mist: Systematic review of what the chemistry of contaminants in electronic cigarettes tells us about health risks. BMC Public Health 2014, 14, 18. [Google Scholar] [CrossRef] [PubMed]
- Goniewicz, M.L.; Knysak, J.; Gawron, M.; Kosmider, L.; Sobczak, A.; Kurek, J.; Prokopowicz, A.; Jablonska-Czapla, M.; Rosik-Dulewska, C.; Havel, C.; et al. Levels of selected carcinogens and toxicants in vapor from electronic cigarettes. Tob. Control 2014, 23, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Shahab, S.; Goniewicz, M.L.; Blount, B.C.; Brown, J.; McNeill, A.; Alwis, U.; Feng, J.; Wang, L.; West, R. Nicotine, carcinogen, and toxin exposure in long-term e-cigarette and nicotine replacement therapy users. Ann. Int. Med. 2017, 166, 390–400. [Google Scholar] [CrossRef] [PubMed]
- Beard, E.; West, R.; Michie, S.; Brown, J. Association between electronic cigarette use and changes in quit attempts, success of quit attempts, use of smoking cessation pharmacotherapy, and use of stop smoking services in England: Time series analysis of population trends. BMJ 2016, 354. [Google Scholar] [CrossRef] [PubMed]
- Hartmann-Boyce, J.; McRobbie, H.; Bullen, C.; Begh, R.; Stead, L.F.; Hajek, P. Can electronic cigarettes help people stop smoking, and are they safe to use for this purpose? Cochrane Database Syst. Rev. 2016, 9. [Google Scholar] [CrossRef]
- Malas, M.; van der Tempel, J.; Schwartz, R.; Minichiello, A.; Lightfoot, C.; Noormohamed, A.; Andrews, J.; Zawertailo, L.; Ferrence, R. Electronic cigarettes for smoking cessation: A systematic review. Nicotine Tob. Res. 2016, 18, 1926–1936. [Google Scholar] [CrossRef] [PubMed]
- Berry, K.M.; Reynolds, L.M.; Collins, J.M.; Siegel, M.B.; Fetterman, J.L.; Hamburg, N.M.; Bhatnagar, A.; Benjamin, E.J.; Stokes, A. E-cigarette initiation and associated changes in smoking cessation and reduction: The Population Assessment of Tobacco and Health Study, 2013–2015. Tob. Control 2018, 1–7. [Google Scholar] [CrossRef]
- Biener, L.; Hargraves, J.L. A longitudinal study of electronic cigarette use among a population-based sample of adult smokers: Association with smoking cessation and motivation to quit. Nicotine Tob. Res. 2015, 127–133. [Google Scholar] [CrossRef]
- Hitchman, S.C.; Brose, L.S.; Brown, J.; Robson, D.; McNeill, A. Associations between e-cigarette type, frequency of use, and quitting smoking: Findings from a longitudinal online panel survey in Great Britain. Nicotine Tob. Res. 2015, 17, 1187–1194. [Google Scholar] [CrossRef]
- Zhuang, Y.; Cummins, S.E.; Sun, J.Y.; Zhu, S. Long-term e-cigarette use and smoking cessation: A longitudinal study with US population. Tob. Control 2016, 25, i90–i95. [Google Scholar] [CrossRef]
- ASH Factsheet: Use of E-Cigarettes (Vapourisers) among Adults in Great Britain. Available online: http://ash.org.uk/category/information-and-resources/fact-sheets/ (accessed on 15 October 2018).
- Farsalinos, K.E.; Poulas, K.; Voudris, V.; Le Houezec, J. Electronic cigarette use in the European Union: Analysis of a representative sample of 27,460 Europeans from 28 countries. Addiction 2016, 111, 2032–2040. [Google Scholar] [CrossRef]
- Farsalinos, K.E.; Poulas, K.; Voudris, V.; Le Houezec, J. Prevalence and correlates of current daily use of electronic cigarettes in the European Union: Analysis of the 2014 Eurobarometer survey. Int. Emerg. Med. 2017, 12, 757–763. [Google Scholar] [CrossRef]
- Pasquereau, A.; Gautier, A.; Andler, R.; Guignard, R.; Richard, J.-B.; Nguyen-Thanh, V. Tabac et e-cigarette en France: Niveaux d’usage d’après les premiers résultats du Baromètre Santé 2016. Bull. Epidémiol. Hebd. 2017, 12, 214–222. [Google Scholar]
- Special Eurobarometer 458: Attitudes of Europeans towards Tobacco and Electronic Cigarettes. Available online: http://ec.europa.eu/commfrontoffice/publicopinion/index.cfm/Survey/getSurveyDetail/instruments/SPECIAL/surveyKy/2146 (accessed on 27 August 2018).
- Wang, T.W.; Asman, K.; Gentzke, A.S.; Cullen, K.A.; Holder-Hayes, E.; Reyes-Guzman, C.; Jamal, A.; Neff, L.; King, B.A. Tobacco product use among adults—United States, 2017. MMWR Morb. Mortal. Wkly. Rep. 2018, 67, 1225–1232. [Google Scholar] [CrossRef] [PubMed]
- 2016 CDC Data Shows E-Cigarette Use Declines Again. Available online: https://rodutobaccotruth.blogspot.com/2017/09/2016-cdc-data-shows-e-cigarette-use.html (accessed on 27 August 2018).
- Inoue-Choi, M.; Hartge, P.; Liao, L.M.; Caporaso, N.; Freedman, N.D. Association between long-term low-intensity cigarette smoking and incidence of smoking-related cancer in the national institutes of health-AARP cohort. Int. J. Cancer 2018, 142, 271–280. [Google Scholar] [CrossRef] [PubMed]
- Inoue-Choi, M.; Liao, L.M.; Reyes-Guzman, C.; Hartge, P.; Caporaso, N.; Freedman, N.D. Association of long-term, low-intensity smoking with all-cause and cause-specific mortality in the national institutes of health-AARP diet and health study. JAMA Int. Med. 2016. [Google Scholar] [CrossRef] [PubMed]
- McKeganey, N.; Dickson, T. Why don’t more smokers switch to using e-cigarettes: The views of confirmed smokers. Int. J. Environ. Res. Public Health 2017, 14, 647. [Google Scholar] [CrossRef] [PubMed]
- Robertson, L.; Hoek, J.; Blank, M.-L.; Richards, R.; Ling, P.; Popova, L. Dual use of electronic nicotine delivery systems (ENDS) and smoked tobacco; A qualitative analysis. Tob. Control 2018, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Tobacco Heating Products. Available online: http://www.bat-science.com/groupms/sites/BAT_9GVJXS.nsf/vwPagesWebLive/DO9PRKM9?opendocument (accessed on 27 August 2018).
- Smith, M.; Haziza, C.; Hoeng, J.; Lüdicke, F.; Maeder, S.; Vanscheeuwijck, P.; Peitsch, M.C. The Science behind the Tobacco Heating System: A Summary of Published Scientific Articles. Available online: https://www.pmiscience.com/resources/docs/default-source/Library_Documents_latest/pmi-science-ths-executive-summary20e4a5852f88696a9e88ff040043f5e9.pdf?sfvrsn=f06ccc06_0 (accessed on 27 August 2018).
- Jaccard, G.; Djoko, D.T.; Moennikes, O.; Jeannet, C.; Kondylis, A.; Belushkin, M. Comparative assessment of HPHC yields in the tobacco heating system THS2.2 and commercial cigarettes. Regul. Toxicol. Pharm. 2017, 90, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Kogel, U.; Titz, B.; Schlage, W.K.; Nury, C.; Martin, F.; Oviedo, A.; Lebrun, S.; Elamin, A.; Guedj, E.; Trivedi, K.; et al. Evaluation of the tobacco heating system 2.2. part 7: Systems toxicological assessment of a mentholated version revealed reduced cellular and molecular exposure effects compared with mentholated and non-mentholated cigarette smoke. Regul. Toxicol. Pharm. 2016, 81, S123–S138. [Google Scholar] [CrossRef] [PubMed]
- Pratte, P.; Cosandey, S.; Ginglinger, G. Investigation of solid particles in the mainstream aerosol of the tobacco heating system THS2.2 and mainstream smoke of a 3R4F reference cigarette. Hum. Exp. Toxicol. 2017, 36, 1115–1120. [Google Scholar] [CrossRef]
- Schaller, J.-P.; Keller, D.; Poget, L.; Pratte, P.; Kaelin, E.; McHugh, D.; Cudazzo, G.; Smart, D.; Tricker, A.R.; Gautier, L.; et al. Evaluation of the tobacco heating system 2.2. Part 2: Chemical composition, genotoxicity, cytotoxicity, and physical properties of the aerosol. Regul. Toxicol. Pharm. 2016, 81, S27–S47. [Google Scholar] [CrossRef]
- Wong, E.T.; Kogel, U.; Veljkovic, E.; Martin, F.; Xiang, Y.; Boue, S.; Vuillaume, G.; Leroy, P.; Guedj, E.; Rodrigo, G.; et al. Evaluation of the tobacco heating system 2.2. part 4: 90-day OECD 413 rat inhalation study with systems toxicology endpoints demonstrates reduced exposure effects compared with cigarette smoke. Regul. Toxicol. Pharm. 2016, 81, S59–S81. [Google Scholar] [CrossRef] [PubMed]
- Brossard, P.; Weitkunat, R.; Poux, V.; Lama, N.; Haziza, C.; Picavet, P.; Baker, G.; Lüdicke, F. Nicotine pharmacokinetic profiles of the tobacco heating system 2.2, cigarettes and nicotine gum in Japanese smokers. Regul. Toxicol. Pharm. 2017, 89, 193–199. [Google Scholar] [CrossRef]
- Picavet, P.; Haziza, C.; Lama, N.; Weitkunat, R.; Lüdicke, F. Comparison of the pharmacokinetics of nicotine following single and ad libitum use of a tobacco heating system or combustible cigarettes. Nicotine Tob. Res. 2016, 557–563. [Google Scholar] [CrossRef] [PubMed]
- Haziza, C.; de La Bourdonnaye, G.; Merlet, S.; Benzimra, M.; Ancerewicz, J.; Donelli, A.; Baker, G.; Picavet, P.; Lüdicke, F. Assessment of the reduction in levels of exposure to harmful and potentially harmful constituents in Japanese subjects using a novel tobacco heating system compared with conventional cigarettes and smoking abstinence: A randomized controlled study in confinement. Regul. Toxicol. Pharm. 2016, 81, 489–499. [Google Scholar] [CrossRef]
- Haziza, C.; de La Bourdonnaye, G.; Skiada, D.; Ancerewicz, J.; Baker, G.; Picavet, P.; Lüdicke, F. Evaluation of the tobacco heating system 2.2. Part 8: 5-Day randomized reduced exposure clinical study in Poland. Regul. Toxicol. Pharm. 2016, 81, S139–S150. [Google Scholar] [CrossRef]
- Lüdicke, F.; Picavet, P.; Baker, G.; Haziza, C.; Poux, V.; Lama, N.; Weitkunat, R. Effects of switching to the tobacco heating system 2.2 menthol, smoking abstinence, or continued cigarette smoking on biomarkers of exposure: A randomized, controlled, open-label, multicenter study in sequential confinement and ambulatory settings (part 1). Nicotine Tob. Res. 2016, 5, 161–172. [Google Scholar] [CrossRef] [PubMed]
- Lüdicke, F.; Picavet, P.; Baker, G.; Haziza, C.; Poux, V.; Lama, N.; Weitkunat, R. Effects of switching to the menthol tobacco heating system 2.2 menthol, smoking abstinence, or continued cigarette smoking on clinically relevant risk markers: A randomized, controlled, open-label, multicenter study in sequential confinement and ambulatory settings (part 2). Nicotine Tob. Res. 2017, 5, 173–182. [Google Scholar] [CrossRef]
- Simonavicius, E.; McNeill, A.; Shahab, L.; Brose, L.S. Heat-not-burn tobacco products: A systematic literature review. Tob. Control 2018, 1–13. [Google Scholar] [CrossRef]
- Auer, R.; Concha-Lozano, N.; Jacot-Sadowski, I.; Cornuz, J.; Berthet, A. Heat-not-burn tobacco cigarettes: Smoke by any other name. JAMA Int. Med. 2017, 177, 1050–1052. [Google Scholar] [CrossRef]
- Bekki, K.; Inaba, Y.; Uchiyama, S.; Kunugita, N. Comparison of chemicals in mainstream smoke in heat-not-burn tobacco and combustion cigarettes. J. UOEH 2017, 39, 201–207. [Google Scholar] [CrossRef]
- Farsalinos, K.E.; Yannovits, N.; Sarri, T.; Voudris, V.; Poulas, K. Nicotine delivery to the aerosol of a heat-not-burn tobacco product: Comparison with a tobacco cigarette and e-cigarettes. Nicotine Tob. Res. 2018, 20, 1004–1009. [Google Scholar] [CrossRef] [PubMed]
- Farsalinos, K.E.; Yannovits, N.; Sarri, T.; Voudris, V.; Poulas, K.; Leischow, S.J. Carbonyl emissions from a novel heated tobacco product (IQOS): Comparison with an e-cigarette and a tobacco cigarette. Addiction 2018, 113, 2099–2106. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Luo, Y.; Jiang, X.; Zhang, H.; Zhu, F.; Hu, S.; Hou, H.; Hu, Q.; Pang, Y. Chemical analysis and simulated pyrolysis of tobacco heating system 2.2 compared to conventional cigarettes. Nicotine Tob. Res. 2018, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Leigh, N.J.; Palumbo, M.N.; Marino, A.M.; O’Connor, R.J.; Goniewicz, M.L. Tobacco-specific nitrosamines (TSNA) in heated tobacco product IQOS. Tob. Control 2018, 27, s37–s38. [Google Scholar] [CrossRef]
- Mallock, N.; Böss, L.; Burk, R.; Danziger, M.; Welsch, T.; Hahn, H.; Trieu, H.-L.; Hahn, J.; Pieper, E.; Henkler-Stephani, F.; et al. Levels of selected analytes in the emissions of “heat not burn” tobacco products that are relevant to assess human health risks. Arch. Toxicol. 2018, 92, 2145–2149. [Google Scholar] [CrossRef]
- Pieper, E.; Mallock, N.; Henkler-Stephani, F.; Luch, A. Tabakerhitzer als neues product der tabakindustrie: Gesundheitliche risiken. Bundesgesundheitsblatt Gesundh. Gesundh. 2018, 61, 1422–1428. [Google Scholar] [CrossRef]
- Leigh, N.J.; Tran, P.L.; O’Connor, R.J.; Coniewicz, M.L. Cytotoxic effects of heated tobacco products (HTP) on human bronchial epithelial cells. Tob. Control 2018, 27, s26–s29. [Google Scholar] [CrossRef]
- Caponnetto, P.; Maglia, M.; Prosperini, G.; Busà, B.; Polosa, R. Carbon monoxide levels after inhalation from new generation heated tobacco products. Respir. Res. 2018, 19. [Google Scholar] [CrossRef]
- Adriaens, K.; Van Gucht, D.; Declerk, P.; Baeyens, F. Effectiveness of the electronic cigarette: An eight-week Flemish study with six-month follow-up on smoking reduction, craving and experienced benefits and complaints. Int. J. Environ. Res. Public Health 2014, 11, 11220–11248. [Google Scholar] [CrossRef]
- Experiment Management System (Computer software); Sona Systems, Ltd.: Tallinn, Estonia, 2018.
- piCOTM Smokerlyzer® (Apparatus and Software); Bedftont Scientific Ltd.: Maidstone, UK, 2017.
- Affect 5 (Computer Software). 2018. Available online: https://ppw.kuleuven.be/apps/clep/affect5/ (accessed on 6 November 2017).
- Fagerström, K. Determinants of tobacco use and renaming the FTND to the Fagerström Test for Cigarette Dependence. Nicotine Tob. Res. 2011, 14, 75–78. [Google Scholar] [CrossRef]
- Little, M.; Franken, I.H.A.; Muris, P. Psychometric properties of the brief Questionnaire on Smoking Urges (QSU-Brief) in a Dutch smoker population. Neth. J. Psychol. 2011, 66, 44–49. [Google Scholar]
- Etter, J.-F.; Ussher, M.; Hughes, J.R. A test of proposed new tobacco withdrawal symptoms. Addiction 2012, 108, 50–59. [Google Scholar] [CrossRef] [PubMed]
- Toll, B.A.; O’Malley, S.S.; McKee, S.A.; Salovey, P.; Krishnan-Sarin, S. Confirmatory factor analysis of the Minnesota Nicotine Withdrawal Scale. Psychol. Addict. Behav. 2007, 21, 216–225. [Google Scholar] [CrossRef] [PubMed]
- Cappelleri, J.C.; Bushmakin, A.G.; Baker, C.L.; Merikle, E.; Olufade, A.O.; Gilbert, D.G. Confirmatory factor analyses and reliability of the modified cigarette evaluation questionnaire. Addict. Behav. 2007, 32, 912–923. [Google Scholar] [CrossRef] [PubMed]
- Glass, G.V.; Peckham, P.D.; Sanders, J.R. Consequences of failure to meet assumptions underlying the fixed effects analyses of variance and covariance. Rev. Educ. Res. 1972, 42, 237–288. [Google Scholar] [CrossRef]
- Statistica (Version 13) (Computer Software); TIBCO Software Inc.: Palo Alto, CA, USA, 2018.
- Baker, R.R. The formation of the oxides of carbon by the pyrolysis of tobacco. Beiträge Tab. 1975, 8, 16–27. [Google Scholar] [CrossRef]
- Herod, A.A.; Kandiyoti, R. Pyrolysis. 2011. Available online: http://www.thermopedia.com/content/1068/ (accessed on 15 October 2018).
- Maeder, S.; Peitsch, M. Comments on the Article Entitled “Heat-Not-Burn Tobacco Cigarettes: Smoke by Any Other Name” by Auer R. 2017. Available online: https://www.pmiscience.com/resources/docs/default-source/news-documents/review_of_auer_et_al-_-_jama_2017_-_pc-final.pdf?sfvrsn=afc8cb06_2 (accessed on 15 October 2018).
- Farsalinos, K.E.; Spyrou, A.; Stefopoulos, C.; Tsimopoulou, K.; Kourkoveli, P.; Tsiapras, D.; Kyrzopoulos, S.; Poulas, K.; Voudris, V. Nicotine absorption from electronic cigarette use: Comparison between experienced consumers (vapers) and naïve users (smokers). Sci. Rep. 2015, 5. [Google Scholar] [CrossRef]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Adriaens, K.; Van Gucht, D.; Baeyens, F. IQOSTM vs. e-Cigarette vs. Tobacco Cigarette: A Direct Comparison of Short-Term Effects after Overnight-Abstinence. Int. J. Environ. Res. Public Health 2018, 15, 2902. https://doi.org/10.3390/ijerph15122902
Adriaens K, Van Gucht D, Baeyens F. IQOSTM vs. e-Cigarette vs. Tobacco Cigarette: A Direct Comparison of Short-Term Effects after Overnight-Abstinence. International Journal of Environmental Research and Public Health. 2018; 15(12):2902. https://doi.org/10.3390/ijerph15122902
Chicago/Turabian StyleAdriaens, Karolien, Dinska Van Gucht, and Frank Baeyens. 2018. "IQOSTM vs. e-Cigarette vs. Tobacco Cigarette: A Direct Comparison of Short-Term Effects after Overnight-Abstinence" International Journal of Environmental Research and Public Health 15, no. 12: 2902. https://doi.org/10.3390/ijerph15122902
APA StyleAdriaens, K., Van Gucht, D., & Baeyens, F. (2018). IQOSTM vs. e-Cigarette vs. Tobacco Cigarette: A Direct Comparison of Short-Term Effects after Overnight-Abstinence. International Journal of Environmental Research and Public Health, 15(12), 2902. https://doi.org/10.3390/ijerph15122902





