Methods for Evaluating the Combined Effects of Chemical and Nonchemical Exposures for Cumulative Environmental Health Risk Assessment
Abstract
1. Introduction
2. Psychosocial Stressors in Cumulative Risk Assessment
3. Understanding Biological Mechanisms: Animal Models and Human Biomonitoring
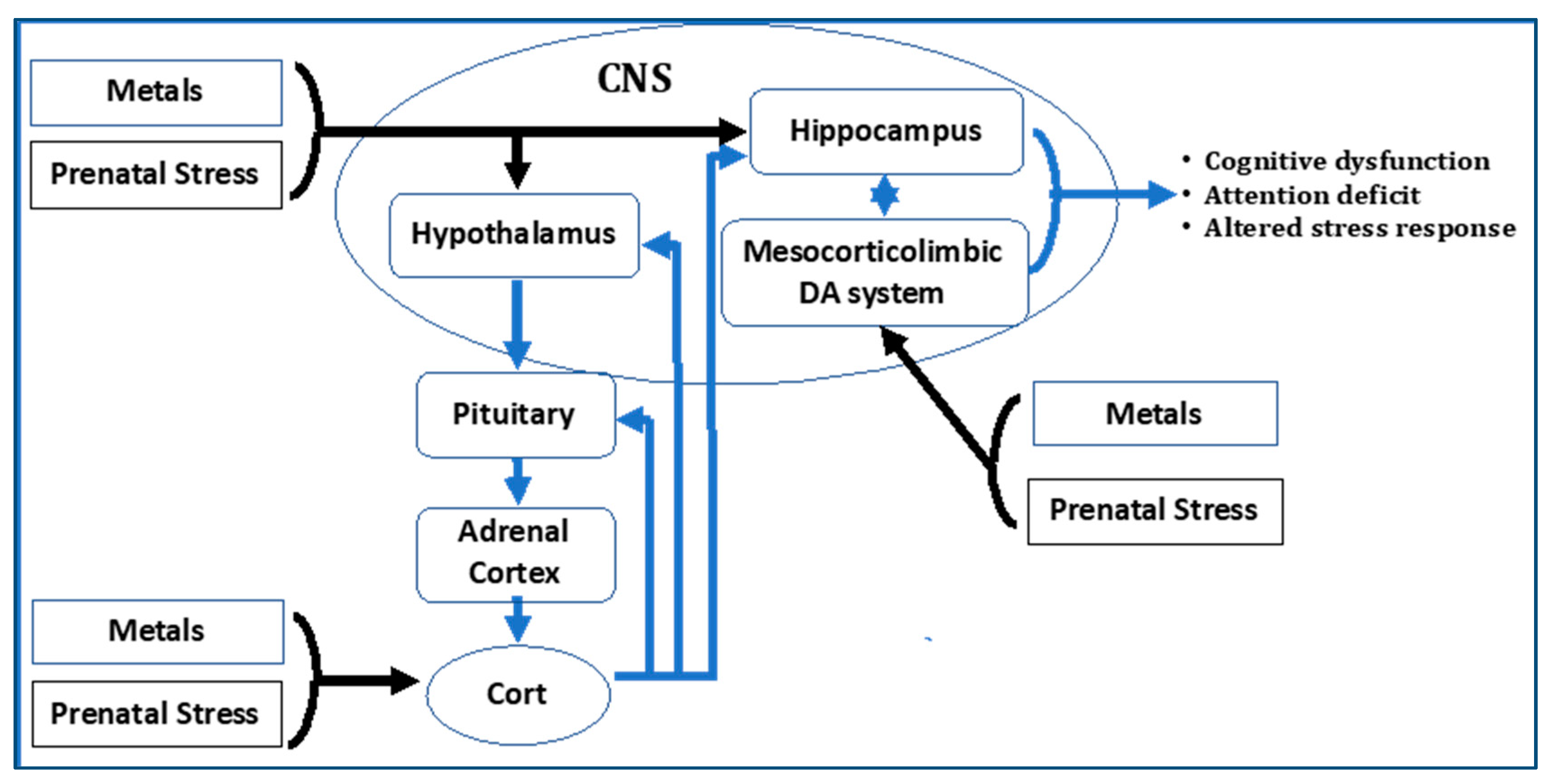
3.1. Animal Models to Examine Combined Effects
3.2. Human Biomarker Studies to Assess Combined Effects
4. Identifying Environmental Stressors Relevant to Communities
5. Challenges in Exposure Assessment for Nonchemical Stressors
5.1. Construct Validity and Publicly-Available Databases for Nonchemical Stressors
5.2. Spatial Scale and Chemical and Nonchemical Exposure Assessment
6. Statistical Methods for Assessing Combined Effects on Health
6.1. Latent Variable Modeling
6.2. Structural Equation Modeling (SEM)
6.3. Alternative Approaches for Evaluating Complex Interactions
7. Discussion
8. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Clougherty, J.E.; Shmool, J.L.C.; Kubzansky, L.D. The Role of Non-Chemical Stressors in Mediating Socioeconomic Susceptibility to Environmental Chemicals. Curr. Environ. Health Rep. 2014, 1, 302–313. [Google Scholar] [CrossRef]
- Hicken, M.T.; Gee, G.C.; Morenoff, J.; Connell, C.M.; Snow, R.C.; Hu, H. A novel look at racial health disparities: The interaction between social disadvantage and environmental health. Am. J. Public Health 2012, 102, 2344–2351. [Google Scholar] [CrossRef] [PubMed]
- Chari, R.; Burke, T.A.; White, R.H.; Fox, M.A. Integrating susceptibility into environmental policy: An analysis of the national ambient air quality standard for lead. Int. J. Environ. Res. Public Health 2012, 9, 1077–1096. [Google Scholar] [CrossRef] [PubMed]
- Lovasi, G.S.; Eldred-Skemp, N.; Quinn, J.W.; Chang, H.W.; Rauh, V.A.; Rundle, A.; Orjuela, M.A.; Perera, F.P. Neighborhood Social Context and Individual Polycyclic Aromatic Hydrocarbon Exposures Associated with Child Cognitive Test Scores. J. Child Fam. Stud. 2014, 23, 785–799. [Google Scholar] [CrossRef] [PubMed]
- Rauh, V.A.; Whyatt, R.M.; Garfinkel, R.; Andrews, H.; Hoepner, L.; Reyes, A.; Diaz, D.; Camann, D.; Perera, F.P. Developmental effects of exposure to environmental tobacco smoke and material hardship among inner-city children. Neurotoxicol. Teratol. 2004, 26, 373–385. [Google Scholar] [CrossRef] [PubMed]
- Vishnevetsky, J.; Tang, D.; Chang, H.W.; Roen, E.L.; Wang, Y.; Rauh, V.; Wang, S.; Miller, R.L.; Herbstman, J.; Perera, F.P. Combined effects of prenatal polycyclic aromatic hydrocarbons and material hardship on child IQ. Neurotoxicol. Teratol. 2015, 49, 74–80. [Google Scholar] [CrossRef] [PubMed]
- Cory-Slechta, D.A. Studying toxicants as single chemicals: Does this strategy adequately identify neurotoxic risk? Neurotoxicology 2005, 26, 491–510. [Google Scholar] [CrossRef]
- Padula, A.M.; Yang, W.; Carmichael, S.L.; Tager, I.B.; Lurmann, F.; Hammond, S.K.; Shaw, G.M. Air Pollution, Neighbourhood Socioeconomic Factors, and Neural Tube Defects in the San Joaquin Valley of California. Paediatr. Perinat. Epidemiol. 2015, 29, 536–545. [Google Scholar] [CrossRef]
- US Environmental Protection Agency. Framework for Cumulative Risk Assessment; EPA/600/P-02/001F; Office of Research and Development National Center for Environmental Assessment: Washington, DC, USA, 2003.
- National Environmental Justice Advisory Council. Recommendations for Integrating Environmental Justice into the EPA’s Research Enterprise; National Environmental Justice Advisory Council: Washington, DC, USA, 2014.
- National Environmental Justice Advisory Council. Ensuring Risk Reduction in Communities with Multiple Stressors: Environmental Justice and Cumulative Risks/Impact; National Environmental Justice Advisory Council: Washington, DC, USA, 2004.
- National Research Council. Phthalates and Cumulative Risk Assessment the Task Ahead; National Academy of Sciences: Washington, DC, USA, 2008.
- National Research Council. Sceince and Decisions; National Academies of Sciences: Washington, DC, USA, 2009.
- Nweke, O.C.; Payne-Sturges, D.; Garcia, L.; Lee, C.; Zenick, H.; Grevatt, P.; Sanders, W.H., 3rd; Case, H.; Dankwa-Mullan, I. Symposium on integrating the science of environmental justice into decision-making at the Environmental Protection Agency: An overview. Am. J. Public Health 2011, 101 (Suppl. 1), S19–S26. [Google Scholar] [CrossRef]
- National Research Council. Risk Assessment in the Federal Government: Managing the Process; The National Academies Press: Washington, DC, USA, 1983.
- Risk Assessment Forum. Framework for Cumulative Risk Assessment; EPA/630/P-02/001F; U.S. Environmental Protection Agency: Washington, DC, USA, 2003.
- Payne-Sturges, D.C.; Korfmacher, K.S.; Cory-Slechta, D.A.; Jimenez, M.; Symanski, E.; Carr Shmool, J.L.; Dotson-Newman, O.; Clougherty, J.E.; French, R.; Levy, J.I.; et al. Engaging Communities in Research on Cumulative Risk and Social Stress-Environment Interactions: Lessons Learned from EPA’s STAR Program. Environ. Justice 2015, 8, 203–212. [Google Scholar] [CrossRef]
- Clougherty, J.E.; Levy, J.I. Chapter 17: Psychosocial and chemical stressors. In Chemical Mixtures and Combined Chemical and Nonchemical Stressors; Rider, C., Simmons, J.E., Eds.; Springer: Berlin, Germany, 2018. [Google Scholar]
- Cohen, S.; Kessler, R.C.; Gordon, L.U. Measuring Stress: A Guide of Health and Social Scientists; Oxford University Press: New York, NY, USA, 1995. [Google Scholar]
- de Kloet, E.R.; Joels, M.; Holsboer, F. Stress and the brain: From adaptation to disease. Nat. Rev. Neurosci. 2005, 6, 463–475. [Google Scholar] [CrossRef] [PubMed]
- Glover, V.; O’Connor, T.G.; O’Donnell, K. Prenatal stress and the programming of the HPA axis. Neurosci. Biobehav. Rev. 2010, 35, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Selye, H. Stress and the general adaptation syndrome. Br. Med. J. 1950, 1, 1383–1392. [Google Scholar] [CrossRef] [PubMed]
- Gamble, K.L.; Berry, R.; Frank, S.J.; Young, M.E. Circadian clock control of endocrine factors. Nat. Rev. Endocrinol. 2014, 10, 466–475. [Google Scholar] [CrossRef] [PubMed]
- McEwen, B.S. Physiology and neurobiology of stress and adaptation: Central role of the brain. Physiol. Rev. 2007, 87, 873–904. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira, A.R.; Reimer, A.E.; Reis, F.M.; Brandao, M.L. Conditioned fear response is modulated by a combined action of the hypothalamic-pituitary-adrenal axis and dopamine activity in the basolateral amygdala. Eur. Neuropsychopharmacol. 2013, 23, 379–389. [Google Scholar] [CrossRef] [PubMed]
- Kasahara, M.; Groenink, L.; Olivier, B.; Sarnyai, Z. Corticotropin-releasing factor (CRF) over-expression down-regulates hippocampal dopamine receptor protein expression and CREB activation in mice. Neuro Endocrinol. Lett. 2011, 32, 193–198. [Google Scholar]
- Young, A.H. The effects of HPA axis function on cognition and its implications for the pathophysiology of bipolar disorder. Harv. Rev. Psychiatry 2014, 22, 331–333. [Google Scholar] [CrossRef]
- Belda, X.; Armario, A. Dopamine D1 and D2 dopamine receptors regulate immobilization stress-induced activation of the hypothalamus-pituitary-adrenal axis. Psychopharmacology 2009, 206, 355–365. [Google Scholar] [CrossRef]
- McEwen, B.S. Protective and damaging effects of stress mediators. N. Engl. J. Med. 1998, 338, 171–179. [Google Scholar] [CrossRef]
- Miller, G.E.; Chen, E.; Sze, J.; Marin, T.; Arevalo, J.M.; Doll, R.; Ma, R.; Cole, S.W. A functional genomic fingerprint of chronic stress in humans: Blunted glucocorticoid and increased NF-kappaB signaling. Biol. Psychiatry 2008, 64, 266–272. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.; Janicki-Deverts, D.; Doyle, W.J.; Miller, G.E.; Frank, E.; Rabin, B.S.; Turner, R.B. Chronic stress, glucocorticoid receptor resistance, inflammation, and disease risk. Proc. Natl. Acad. Sci. USA 2012, 109, 5995–5999. [Google Scholar] [CrossRef] [PubMed]
- Seeman, T.E.; McEwen, B.S.; Rowe, J.W.; Singer, B.H. Allostatic load as a marker of cumulative biological risk: MacArthur studies of successful aging. Proc. Natl. Acad. Sci. USA 2001, 98, 4770–4775. [Google Scholar] [CrossRef] [PubMed]
- Hadley, M.E. Endocrinology; Prentice Hall: Englewood Cliffs, NJ, USA, 1992. [Google Scholar]
- Brunner, E.J. Toward a new social biology. In Social Epidemiology; Berkman, L.F., Kawachi, I., Eds.; Oxford University Press: New York, NY, USA, 2000; pp. 306–331. [Google Scholar]
- Heim, C.; Ehlert, U.; Hellhammer, D.H. The potential role of hypocortisolism in the pathophysiology of stress-related bodily disorders. Psychoneuroendocrinology 2000, 25, 1–35. [Google Scholar] [CrossRef]
- Bellingrath, S.; Rohleder, N.; Kudielka, B.M. Effort-reward-imbalance in healthy teachers is associated with higher LPS-stimulated production and lower glucocorticoid sensitivity of interleukin-6 in vitro. Biol. Psychol. 2013, 92, 403–409. [Google Scholar] [CrossRef] [PubMed]
- Miller, G.E.; Cohen, S.; Ritchey, A.K. Chronic psychological stress and the regulation of pro-inflammatory cytokines: A glucocorticoid-resistance model. Health Psychol. 2002, 21, 531–541. [Google Scholar] [CrossRef] [PubMed]
- Miller, G.E.; Chen, E.; Zhou, E.S. If it goes up, must it come down? Chronic stress and the hypothalamic-pituitary-adrenocortical axis in humans. Psychol. Bull. 2007, 133, 25–45. [Google Scholar] [CrossRef]
- McEwen, B.S.; Seeman, T. Protective and damaging effects of mediators of stress. Elaborating and testing the concepts of allostasis and allostatic load. Ann. N. Y. Acad. Sci. 1999, 896, 30–47. [Google Scholar] [CrossRef]
- Clougherty, J.E.; Kubzansky, L.D. A framework for examining social stress and susceptibility to air pollution in respiratory health. Environ. Health Perspect. 2009, 117, 1351–1358. [Google Scholar] [CrossRef]
- Morello-Frosch, R.; Shenassa, E.D. The environmental “riskscape” and social inequality: Implications for explaining maternal and child health disparities. Environ. Health Perspect. 2006, 114, 1150–1153. [Google Scholar] [CrossRef]
- Wright, R.J. Health effects of socially toxic neighborhoods: The violence and urban asthma paradigm. Clin. Chest Med. 2006, 27, 413–421. [Google Scholar] [CrossRef] [PubMed]
- Allen, R.W.; Davies, H.; Cohen, M.A.; Mallach, G.; Kaufman, J.D.; Adar, S.D. The spatial relationship between traffic-generated air pollution and noise in 2 US cities. Environ. Res. 2009, 109, 334–342. [Google Scholar] [CrossRef] [PubMed]
- Anisman, H.; Matheson, K. Stress, depression, and anhedonia: Caveats concerning animal models. Neurosci. Biobehav. Rev. 2005, 29, 525–546. [Google Scholar] [CrossRef] [PubMed]
- Blanchard, R.J.; McKittrick, C.R.; Blanchard, D.C. Animal models of social stress: Effects on behavior and brain neurochemical systems. Physiol. Behav. 2001, 73, 261–271. [Google Scholar] [CrossRef]
- Campos, A.C.; Fogaca, M.V.; Aguiar, D.C.; Guimaraes, F.S. Animal models of anxiety disorders and stress. Rev. Bras. Psiquiatr. 2013, 35 (Suppl. 2), S101–S111. [Google Scholar] [CrossRef]
- Cory-Slechta, D.A.; Virgolini, M.B.; Thiruchelvam, M.; Weston, D.D.; Bauter, M.R. Maternal stress modulates the effects of developmental lead exposure. Environ. Health Perspect. 2004, 112, 717–730. [Google Scholar] [CrossRef] [PubMed]
- Cory-Slechta, D.A.; Virgolini, M.B.; Rossi-George, A.; Thiruchelvam, M.; Lisek, R.; Weston, D. Lifetime consequences of combined maternal lead and stress. Basic Clin. Pharmacol. Toxicol. 2008, 102, 218–227. [Google Scholar] [CrossRef]
- Cory-Slechta, D.A.; Stern, S.; Weston, D.; Allen, J.L.; Liu, S. Enhanced learning deficits in female rats following lifetime pb exposure combined with prenatal stress. Toxicol. Sci. 2010, 117, 427–438. [Google Scholar] [CrossRef]
- Rossi-George, A.; Virgolini, M.B.; Weston, D.; Cory-Slechta, D.A. Alterations in glucocorticoid negative feedback following maternal Pb, prenatal stress and the combination: A potential biological unifying mechanism for their corresponding disease profiles. Toxicol. Appl. Pharmacol. 2009, 234, 117–127. [Google Scholar] [CrossRef]
- Virgolini, M.B.; Rossi-George, A.; Lisek, R.; Weston, D.D.; Thiruchelvam, M.; Cory-Slechta, D.A. CNS effects of developmental Pb exposure are enhanced by combined maternal and offspring stress. Neurotoxicology 2008, 29, 812–827. [Google Scholar] [CrossRef]
- Weston, H.I.; Sobolewski, M.E.; Allen, J.L.; Weston, D.; Conrad, K.; Pelkowski, S.; Watson, G.E.; Zareba, G.; Cory-Slechta, D.A. Sex-dependent and non-monotonic enhancement and unmasking of methylmercury neurotoxicity by prenatal stress. Neurotoxicology 2014, 41, 123–140. [Google Scholar] [CrossRef] [PubMed]
- Cory-Slechta, D.A.; Virgolini, M.B.; Liu, S.; Weston, D. Enhanced stimulus sequence-dependent repeated learning in male offspring after prenatal stress alone or in conjunction with lead exposure. Neurotoxicology 2012, 33, 1188–1202. [Google Scholar] [CrossRef] [PubMed]
- Cory-Slechta, D.A.; Merchant-Borna, K.; Allen, J.; Liu, S.; Weston, D.; Conrad, K. Variations in the Nature of Behavioral Experience Can Differentially Alter the Consequences of Developmental Exposures to Lead, Prenatal Stress and the Combination. Toxicol. Sci. 2013, 131, 194–205. [Google Scholar] [CrossRef] [PubMed]
- Cory-Slechta, D.A.; Weston, D.; Liu, S.; Allen, J.L. Brain hemispheric differences in the neurochemical effects of lead, prenatal stress, and the combination and their amelioration by behavioral experience. Toxicol. Sci. 2013, 132, 419–430. [Google Scholar] [CrossRef] [PubMed]
- Virgolini, M.B.; Chen, K.; Weston, D.D.; Bauter, M.R.; Cory-Slechta, D.A. Interactions of chronic lead exposure and intermittent stress: Consequences for brain catecholamine systems and associated behaviors and HPA axis function. Toxicol. Sci. 2005, 87, 469–482. [Google Scholar] [CrossRef] [PubMed]
- Virgolini, M.B.; Bauter, M.R.; Weston, D.D.; Cory-Slechta, D.A. Permanent alterations in stress responsivity in female offspring subjected to combined maternal lead exposure and/or stress. Neurotoxicology 2006, 27, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Virgolini, M.B.; Rossi-George, A.; Weston, D.; Cory-Slechta, D.A. Influence of low level maternal Pb exposure and prenatal stress on offspring stress challenge responsivity. Neurotoxicology 2008, 29, 928–939. [Google Scholar] [CrossRef] [PubMed]
- Weston, H.I.; Weston, D.D.; Allen, J.L.; Cory-Slechta, D.A. Sex-dependent impacts of low-level lead exposure and prenatal stress on impulsive choice behavior and associated biochemical and neurochemical manifestations. Neurotoxicology 2014, 44, 169–183. [Google Scholar] [CrossRef] [PubMed]
- Sobolewski, M.; Conrad, K.; Marvin, E.; Allen, J.L.; Cory-Slechta, D.A. Endocrine active metals, prenatal stress and enhanced neurobehavioral disruption. Horm. Behav. 2018, 101, 36–49. [Google Scholar] [CrossRef]
- Bale, T.L. The placenta and neurodevelopment: Sex differences in prenatal vulnerability. Dialogues Clin. Neurosci. 2016, 18, 459–464. [Google Scholar]
- Bale, T.L.; Epperson, C.N. Sex differences and stress across the lifespan. Nat. Neurosci. 2015, 18, 1413–1420. [Google Scholar] [CrossRef] [PubMed]
- Bale, T.L. Sex differences in prenatal epigenetic programming of stress pathways. Stress 2011, 14, 348–356. [Google Scholar] [CrossRef] [PubMed]
- Sobolewski, M.; Allen, J.L.; Morris-Schaffer, K.; Klocke, C.; Conrad, K.; Cory-Slechta, D.A. A novel, ecologically relevant, highly preferred, and non-invasive means of oral substance administration for rodents. Neurotoxicol. Teratol. 2016, 56, 75–80. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.H.; Fan, Z.; Lioy, P.J.; Baptista, A.; Greenberg, M.; Laumbach, R.J. A novel mobile monitoring approach to characterize spatial and temporal variation in traffic-related air pollutants in an urban community. Atmos. Environ. 2016, 141, 161–173. [Google Scholar] [CrossRef]
- Ji, N.; Baptista, A.; Greenberg, M.; Mincey, I.; Cepeda, C.; Ohman-Strickland, P.A.; Black, C.; Kipen, H.M.; Laumbach, R.J. Personal Exposure to Black Carbon, Nitrogen Dioxide, and Chronic Psychosocial Stress: Impacts on Childhood Asthma Exacerbation in a Seaport-Adjacent Community. Am. J. Respir. Crit. Care Med. 2017, 195, A4803. [Google Scholar]
- Scammell, M.K. Qualitative environmental health research: An analysis of the literature, 1991–2008. Environ. Health Perspect. 2010, 118, 1146–1154. [Google Scholar] [CrossRef] [PubMed]
- Brown, P.; Mikkelsen, E.J. No Safe Place: Toxic Waste, Leukemia, and Community Action; University of California Press: Berkeley, CA, USA, 1990. [Google Scholar]
- Corburn, J. Street Science: Community Knowledge and Environmental Health Justice; MIT Press: Cambridge, UK, 2005. [Google Scholar]
- Levine, A.G. Love Canal: Science, Politics, and People; Heath: Lexington, MA, USA, 1982. [Google Scholar]
- Reich, M.R. Toxic Politics: Responding to Chemical Disasters; Cornell University Press: Ithaca, NY, USA, 1991. [Google Scholar]
- Sze, J. Noxious New York: The Racial Politics of Urban Health and Environmental Justice; MIT Press: Cambridge, MA, USA, 2007. [Google Scholar]
- Shmool, J.L.; Yonas, M.A.; Newman, O.D.; Kubzansky, L.D.; Joseph, E.; Parks, A.; Callaway, C.; Chubb, L.G.; Shepard, P.; Clougherty, J.E. Identifying Perceived Neighborhood Stressors Across Diverse Communities in New York City. Am. J. Community Psychol. 2015, 56, 145–155. [Google Scholar] [CrossRef]
- Green, L.; Fullilove, M.; Evans, D.; Shepard, P. “Hey, mom, thanks!”: Use of focus groups in the development of place-specific materials for a community environmental action campaign. Environ. Health Perspect. 2002, 110 (Suppl. 2), 265–269. [Google Scholar] [CrossRef]
- Symanski, E.; Karpman, M.; Jimenez, M.; Lopez, D.S.; Felknor, S.A.; Upadhyaya, M.; Strom, S.S.; Bondy, M.L. Using a Community-Engaged Approach to Develop a Bilingual Survey about Psychosocial Stressors among Individuals of Mexican Origin. J. Health Care Poor Underserved 2015, 26, 1456–1471. [Google Scholar] [CrossRef]
- Chow, W.H.; Chrisman, M.; Daniel, C.R.; Ye, Y.; Gomez, H.; Dong, Q.; Anderson, C.E.; Chang, S.; Strom, S.; Zhao, H.; et al. Cohort Profile: The Mexican American Mano a Mano Cohort. Int. J. Epidemiol. 2017, 46, e3. [Google Scholar] [CrossRef]
- Rammah, A.; Walker Whitworth, K.; Han, I.; Chan, W.; Jimenez, M.D.; Strom, S.S.; Bondy, M.L.; Symanski, E. A Mixed-Methods Study to Examine the Role of Psychosocial Stress and Air Pollution on Hypertension in Mexican-Origin Hispanics. J. Racial Ethn. Health Dispar. 2018. [Google Scholar] [CrossRef] [PubMed]
- Ou, J.Y.; Levy, J.I.; Peters, J.L.; Bongiovanni, R.; Soto, J.G.; Medina, R.; Scammell, M.K. A walk in the park: The influence of urban parks and community violence on physical activity in Chelsea, MA. Int. J. Environ. Res. Public Health 2015, 13, 97. [Google Scholar] [CrossRef] [PubMed]
- Scammell, M.K.; Ou, J.Y.; Carlin, C.; Olortegui, R.M.; Peters, J.L.; Bongiovanni, R. Chelsea STAR (Science to Achieve Results) Annotated Interview Guide. Community Engaged Scholarship for Health (CES4H), Product ID#W5SKDYJB: 2016. Available online: http://ces4health.info/find-products/view-product.aspx?code=W5SKDYJB (accessed on 6 December 2018).
- Karner, A.A.; Eisinger, D.S.; Niemeier, D.A. Near-roadway air quality: Synthesizing the findings from real-world data. Environ. Sci. Technol. 2010, 44, 5334–5344. [Google Scholar] [CrossRef] [PubMed]
- Lazarus, R.S.; Folkman, S. Stress, Appraisal, and Coping; Springer Publishing Company: New York, NY, USA, 1984. [Google Scholar]
- Neckerman, K.M.; Lovasi, G.S.; Davies, S.; Purciel, M.; Quinn, J.; Feder, E.; Raghunath, N.; Wasserman, B.; Rundle, A. Disparities in urban neighborhood conditions: Evidence from GIS measures and field observation in New York City. J. Public Health Policy 2009, 30 (Suppl. 1), S264–S285. [Google Scholar] [CrossRef] [PubMed]
- Bangia, K.S.; Symanski, E.; Strom, S.S.; Bondy, M. A cross-sectional analysis of polycyclic aromatic hydrocarbons and diesel particulate matter exposures and hypertension among individuals of Mexican origin. Environ. Health 2015, 14, 51. [Google Scholar] [CrossRef] [PubMed]
- Shmool, J.L.C.; Johnson, I.L.; Dodson, Z.; Keene, R.; Gradeck, B.; Clougherty, J.E. Developing a GIS-based online survey tool to elicit self-report neighborhood geography: A pilot study in New York City and Pittsburgh. Prof. Geographer. 2018, 70, 423–433. [Google Scholar] [CrossRef]
- Shmool, J.L.; Kubzansky, L.D.; Newman, O.D.; Spengler, J.; Shepard, P.; Clougherty, J.E. Social stressors and air pollution across New York City communities: A spatial approach for assessing correlations among multiple exposures. Environ. Health 2014, 13, 91. [Google Scholar] [CrossRef] [PubMed]
- Levy, J.I.; Fabian, M.P.; Peters, J.L. Community-wide health risk assessment using geographically resolved demographic data: A synthetic population approach. PLoS ONE 2014, 9, e87144. [Google Scholar] [CrossRef]
- United States Census Bureau. American Community Survey. Available online: https://www.census.gov/programs-surveys/acs/ (accessed on 28 November 2018).
- Fabian, M.P.; Levy, J.I.; Vieira, V.M.; Peters, J.L.; Korrick, S.A. Behavioral and Sociodemographic Predictors of Exposure to Multiple Chemicals Associated with ADHD-Related Behavior in a Low Income Community; NIEHS Fest: Durham, NC, USA, 2016. [Google Scholar]
- Basra, K.; Fabian, M.P.; Holberger, R.R.; French, R.; Levy, J.I. Community-Engaged Modeling of Geographic and Demographic Patterns of Multiple Public Health Risk Factors. Int. J. Environ. Res. Public Health 2017, 14, 730. [Google Scholar] [CrossRef]
- Juster, R.P.; McEwen, B.S.; Lupien, S.J. Allostatic load biomarkers of chronic stress and impact on health and cognition. Neurosci. Biobehav. Rev. 2010, 35, 2–16. [Google Scholar] [CrossRef]
- Skrondal, A.; Rabe-Hesketh, S. Generalized Latent Variable Modeling: Multilevel, Longitudinal, and Structural Equation Models, 1st ed.; Chapman and Hall: New York, NY, USA, 2004. [Google Scholar]
- Levy, J.I.; Fabian, M.P.; Peters, J.L. Meta-analytic approaches for multi-stressor dose-response function development: Strengths, limitations, and case studies. Risk Anal. 2015, 36, 1040–1049. [Google Scholar] [CrossRef] [PubMed]
- Peters, J.L.; Fabian, M.P.; Levy, J.I. Combined impact of lead, cadmium, polychlorinated biphenyls and non-chemical risk factors on blood pressure in NHANES. Environ. Res. 2014, 132, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Sagiv, S.K.; Thurston, S.W.; Bellinger, D.C.; Tolbert, P.E.; Altshul, L.M.; Korrick, S.A. Prenatal organochlorine exposure and behaviors associated with attention deficit hyperactivity disorder in school-aged children. Am. J. Epidemiol. 2010, 171, 593–601. [Google Scholar] [CrossRef] [PubMed]
- Tallarida, R.J. Revisiting the isobole and related quantitative methods for assessing drug synergism. J. Pharmacol. Exp. Ther. 2012, 342, 2–8. [Google Scholar] [CrossRef] [PubMed]
- Ganter, B.; Wille, R. Formal Concept Analysis: Mathematical Foundations; Springer: New York, NY, USA, 1998. [Google Scholar]
- National Institute of Environmental Health Sciences. 2012–2017 Strategic Plan Advancing Science, Improving Health: A Plan for Environmental Health Research; NIH Publication No. 12-7935; U.S. Department of Health and Human Services: Washington, DC, USA, 2012.
- Risk Assessment Forum. Guidelines for the Health Risk Assessment of Chemical Mixtures; U.S. Environmental Protection Agency: Washington, DC, USA, 1986.
- Evans, G.W.; Marcynyszyn, L.A. Environmental justice, cumulative environmental risk, and health among low- and middle-income children in upstate New York. Am. J. Public Health 2004, 94, 1942–1944. [Google Scholar] [CrossRef]
- Gray, S.C.; Edwards, S.E.; Schultz, B.D.; Miranda, M.L. Assessing the impact of race, social factors and air pollution on birth outcomes: A population-based study. Environ. Health 2014, 13, 4. [Google Scholar] [CrossRef] [PubMed]
- Can, A.; Dao, D.T.; Arad, M.; Terrillion, C.E.; Piantadosi, S.C.; Gould, T.D. The mouse forced swim test. J. Vis. Exp. 2012, 59, 3638. [Google Scholar] [CrossRef] [PubMed]
- Stone, E.A.; Lin, Y. Open-space forced swim model of depression for mice. Curr. Protoc. Neurosci. 2011, 54, 9–36. [Google Scholar] [CrossRef]
- Konarska, M.; Stewart, R.E.; McCarty, R. Predictability of chronic intermittent stress: Effects on sympathetic-adrenal medullary responses of laboratory rats. Behav. Neural. Biol. 1990, 53, 231–243. [Google Scholar] [CrossRef]
- Clougherty, J.E.; Levy, J.I.; Hynes, H.P.; Spengler, J.D. A longitudinal analysis of the efficacy of environmental interventions on asthma-related quality of life and symptoms among children in urban public housing. J. Asthma 2006, 43, 335–343. [Google Scholar] [CrossRef]
- Clougherty, J.E.; Levy, J.I.; Kubzansky, L.D.; Ryan, P.B.; Suglia, S.F.; Canner, M.J.; Wright, R.J. Synergistic effects of traffic-related air pollution and exposure to violence on urban asthma etiology. Environ. Health Perspect. 2007, 115, 1140–1146. [Google Scholar] [CrossRef] [PubMed]
- Clougherty, J.E.; Rossi, C.A.; Lawrence, J.; Long, M.S.; Diaz, E.A.; Lim, R.H.; McEwen, B.; Koutrakis, P.; Godleski, J.J. Chronic social stress and susceptibility to concentrated ambient fine particles in rats. Environ. Health Perspect. 2010, 118, 769–775. [Google Scholar] [CrossRef] [PubMed]
- Taylor, K.W.; Joubert, B.R.; Braun, J.M.; Dilworth, C.; Gennings, C.; Hauser, R.; Heindel, J.J.; Rider, C.V.; Webster, T.F.; Carlin, D.J. Statistical Approaches for Assessing Health Effects of Environmental Chemical Mixtures in Epidemiology: Lessons from an Innovative Workshop. Environ. Health Perspect. 2016, 124, A227–A229. [Google Scholar] [CrossRef] [PubMed]
| Short Title | Full Title | Institution(s) | Link to Grant Abstract and Annual Reports | Exposures | Outcomes | |
|---|---|---|---|---|---|---|
| Psychosocial/Nonchemical Stressors Evaluated | Pollutant/Chemical Stressors Evaluated | Health Outcomes Evaluated | ||||
| The BU/Chelsea STAR Project | New Methods for Analysis of Cumulative Risk in Urban Populations | Boston University; The Chelsea Collaborative | https://cfpub.epa.gov/ncer_abstracts/index.cfm/fuseaction/display.abstractDetail/abstract/9278/report/0 | Crisis In Family Systems (CRISYS) survey items: financial, legal and home issue domains; Neighborhood/Block Conditions, IV Environmental Assessments, Exposure to Violence Assessment, Inner City Asthma Study, The Multigroup Ethnic Identity Measure, Reactions to Race; food accessibility, use of parks, perceptions of noise and odor. | High traffic roadways, toxic release inventories, land uses. | Self-rated health; diagnosed diabetes, heart attack, heart disease, asthma, emphysema/respiratory disease, overweight, arthritis, hypertension, psoriasis, vitiligo, cancer, depression/mental health and chronic disease; symptoms |
| The BU/NorthStar STAR Project | Effects-Based Cumulative Risk Assessment in a Low-Income Urban Community near a Superfund Site | Boston University; NorthStar Learning Centers | https://cfpub.epa.gov/ncer_abstracts/index.cfm/fuseaction/display.abstractDetail/abstract/9144/report/0 | Sociodemographic proxies (education, race/ethnicity, income), maternal smoking during pregnancy, maternal stress during pregnancy (characterized in part by community violence), diet, access to health care. | ADHD-like behavior: Blood Pb (cord blood and at various ages), serum polychlorinated biphenyls (PCBs), serum DDE, hair mercury, ETS exposure approximated by questionnaire data and modelsBlood pressure: Blood Pb, blood Hg, blood Cd, ETS | ADHD-like behavior, blood pressure |
| The University of Pittsburgh/WE ACT STAR Project | Community Stressors and Susceptibility to Air Pollution in Urban Asthma | University of Pittsburgh; West Harlem Environmental Action (WE ACT) | https://cfpub.epa.gov/ncer_abstracts/index.cfm/fuseaction/display.abstractDetail/abstract/9279/report/0 | Community stressors were identified in citywide focus groups. GIS indicators were collected from administrative data (yrs 2008–2010) on crime and violence, mental and general health, built environment, healthcare access, noise disruption, child-specific stressors (e.g., schools conditions), socio-economic position. Citywide surveys measured perceived stress, anxiety, depression, caregiver stress, urban life events, unfair treatment, social support, and perceived neighborhood disorder, social capital, and violence. | DOHMH New York City Community Air Survey (NYCCAS) city-wide pollution (PM2.5, NO2, EC, SO2, O3), 2008–2010. Environmental Protection Agency (EPA) AQS regulatory daily. | Child asthma exacerbation and morbidity |
| The Rutgers/Ironbound STAR Project | Effects of Stress and Traffic Pollutants on Childhood Asthma in an Urban Community | Rutgers, The State University of New Jersey; Ironbound Community Corporation (ICC) | https://cfpub.epa.gov/ncer_abstracts/index.cfm/fuseaction/display.abstractDetail/abstract/9277/report/0 | UCLA interview, focus groups, stress reactivity, glucocorticoid and b2-adrenergic receptor levels. | Personal real-time black carbon, 24-h NO2 by passive sampler, EPA central site priority pollutant | Emergency department data on asthma exacerbation measured as symptoms, medication use, exhaled nitric oxide, spirometry |
| The Rochester STAR Project | Combined Effects of Metals and Stress on Central Nervous System Function | University of Rochester School of Medicine and Dentistry | https://cfpub.epa.gov/ncer_abstracts/index.cfm/fuseaction/display.abstractDetail/abstract/9275/report/0 | In animal models, the stressors are directly imposed and measures of its efficacy can include any changes in function of the hypothalamic-pituitary-adrenal (HPA) axis or changes in behavior or brain function. | Blood lead and brain measures of metals by atomic absorption spectrometry | Cognitive function, IQ, attention deficit |
| The UT/TCCAP STAR Project | Analytical Strategies for Assessing Cumulative Effects of Chemical and Nonchemical Stressors | University of Texas; Texas City Community Advisory Panel (TCCAP) | https://cfpub.epa.gov/ncer_abstracts/index.cfm/fuseaction/display.abstractDetail/abstract/9276/report/0 | Neighborhood stressors (crime, infrastructure, income distribution, segregation) Social stressors (poverty, employment, discrimination, access to care, educational attainment) Psychosocial stress (perceived stress, perception of environmental risk, self-reported coping, social support. | Chronic: EPA National Air Toxics Assessment 2005 ambient concentrations of air toxics (cumulative cancer risk). Acute: Upset event releases of toxics, monitoring data, ozone warning days, PM alerts, high allergen counts, self-reported exposures. | Allostatic load, SR-36, glucose and lipid markers, antibody titers, inflammatory markers, current conditions, current meds |
| The UTHealth/Mano a Mano STAR Project | Hypertension in Mexican-Americans: Assessing Disparities in Air Pollutant Risks | University of Texas Health Science Center at Houston (UTHealth) School of Public Health; MD Anderson Cancer Center; National Chiao-Tung University | https://cfpub.epa.gov/ncer_abstracts/index.cfm/fuseaction/display.abstractDetail/abstract/9147/report/0 | Individual-, family- and neighborhood-level stressors identified through community focus groups:—Individual level (anxiety/depression, aches and pains, trouble sleeping, worrying about health/money/time for oneself)—Family level (family illness/separation, domestic violence, trouble with children)—Neighborhood level (noise, traffic, litter, safety, violence at children‘s school)—Other social stressors (coming into contact with authorities, feeling discriminated against due to race/ethnicity or immigration status)—Stressors at work (worrying about working too hard or harmful substance exposures) | Publicly available data sets from the Texas Commission on Environmental Quality (TCEQ); concentrations of particulate matter ≤2.5 micrometers in aerodynamic diameter (PM2.5) and ozone (O3). | Self-reported hypertension |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Payne-Sturges, D.C.; Scammell, M.K.; Levy, J.I.; Cory-Slechta, D.A.; Symanski, E.; Carr Shmool, J.L.; Laumbach, R.; Linder, S.; Clougherty, J.E. Methods for Evaluating the Combined Effects of Chemical and Nonchemical Exposures for Cumulative Environmental Health Risk Assessment. Int. J. Environ. Res. Public Health 2018, 15, 2797. https://doi.org/10.3390/ijerph15122797
Payne-Sturges DC, Scammell MK, Levy JI, Cory-Slechta DA, Symanski E, Carr Shmool JL, Laumbach R, Linder S, Clougherty JE. Methods for Evaluating the Combined Effects of Chemical and Nonchemical Exposures for Cumulative Environmental Health Risk Assessment. International Journal of Environmental Research and Public Health. 2018; 15(12):2797. https://doi.org/10.3390/ijerph15122797
Chicago/Turabian StylePayne-Sturges, Devon C., Madeleine K. Scammell, Jonathan I. Levy, Deborah A. Cory-Slechta, Elaine Symanski, Jessie L. Carr Shmool, Robert Laumbach, Stephen Linder, and Jane E. Clougherty. 2018. "Methods for Evaluating the Combined Effects of Chemical and Nonchemical Exposures for Cumulative Environmental Health Risk Assessment" International Journal of Environmental Research and Public Health 15, no. 12: 2797. https://doi.org/10.3390/ijerph15122797
APA StylePayne-Sturges, D. C., Scammell, M. K., Levy, J. I., Cory-Slechta, D. A., Symanski, E., Carr Shmool, J. L., Laumbach, R., Linder, S., & Clougherty, J. E. (2018). Methods for Evaluating the Combined Effects of Chemical and Nonchemical Exposures for Cumulative Environmental Health Risk Assessment. International Journal of Environmental Research and Public Health, 15(12), 2797. https://doi.org/10.3390/ijerph15122797







