Phenanthrene Mitigates Cadmium Toxicity in Earthworms Eisenia fetida (Epigeic Specie) and Aporrectodea caliginosa (Endogeic Specie) in Soil
Abstract
1. Introduction
2. Materials and Methods
2.1. Soil Properties
2.2. Test Soil Preparation
2.3. Test Organism
2.4. Acute Toxicity Test
2.5. Toxicity Test with Chemicals Mixture
2.6. Experimental Monitoring
2.7. Kinetics Parameters
2.8. Chemical Analysis
2.8.1. Cadmium
2.8.2. Phenanthrene Determination
2.9. Statistical Analysis
3. Results
3.1. Response of Earthworms to Individually Chemicals
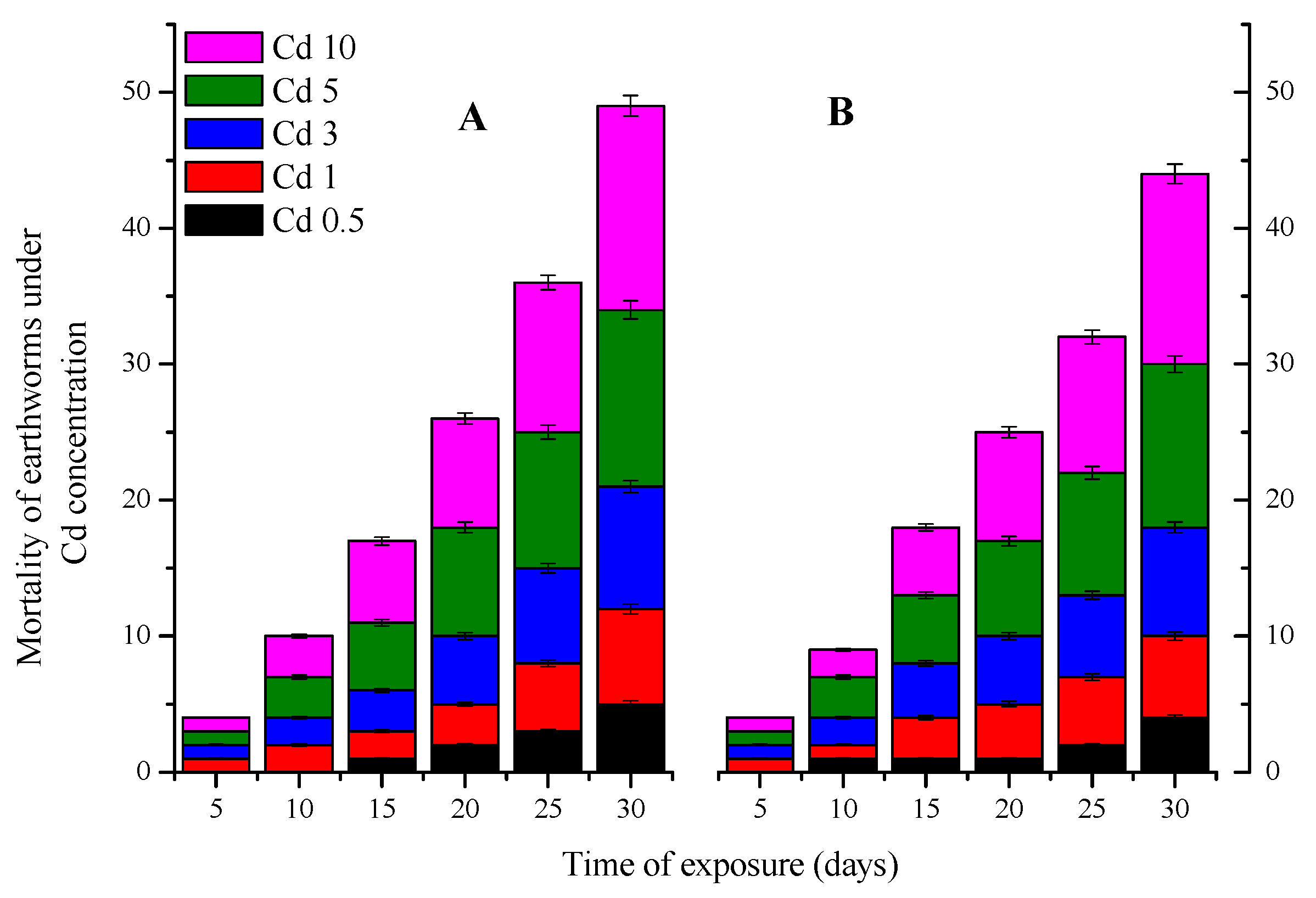
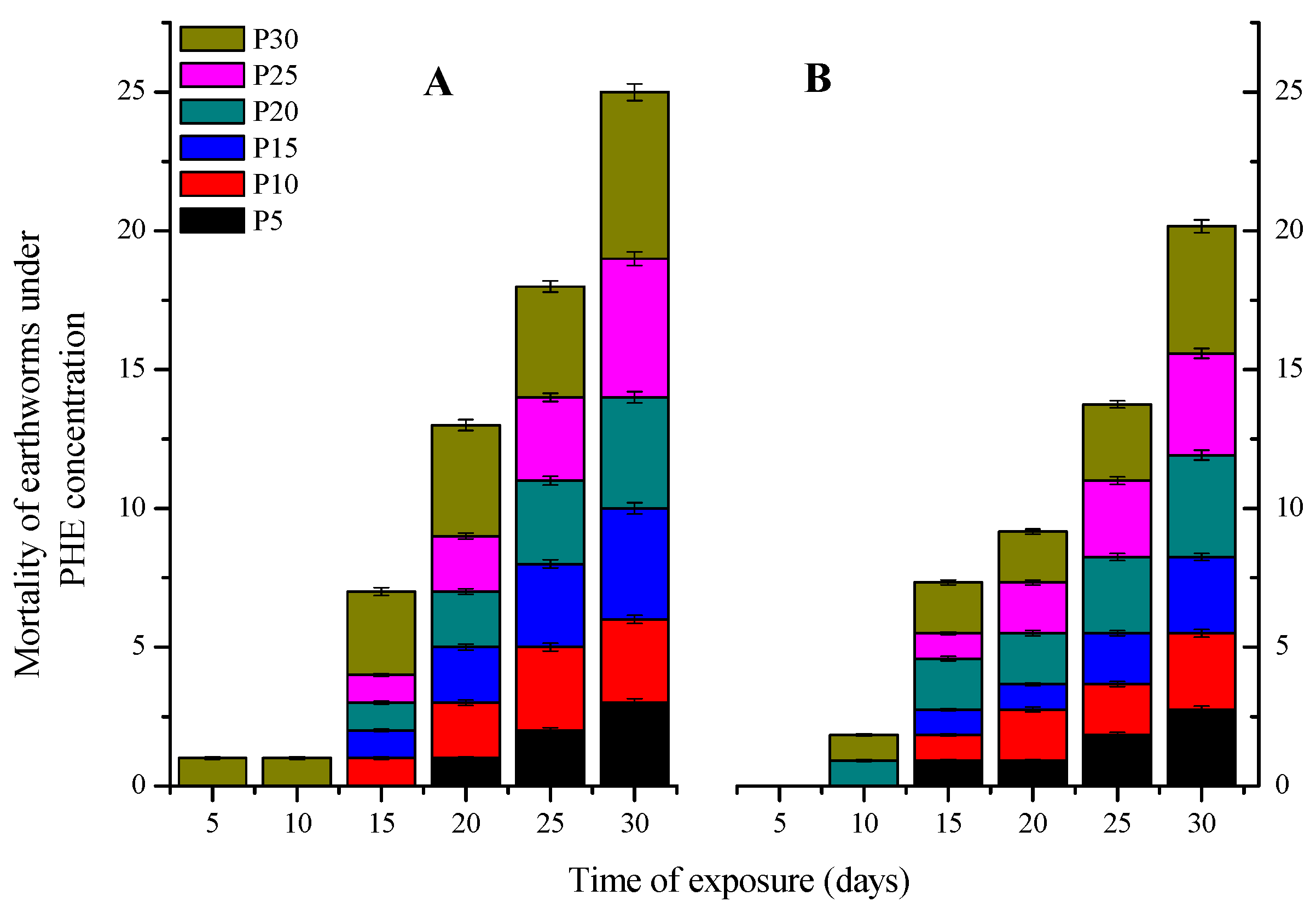
3.1.1. Mortality
3.1.2. Body Weight Variation
3.2. Responses of Earthworms to Chemical Mixture
3.2.1. Mortality
3.2.2. Body Mass Variation
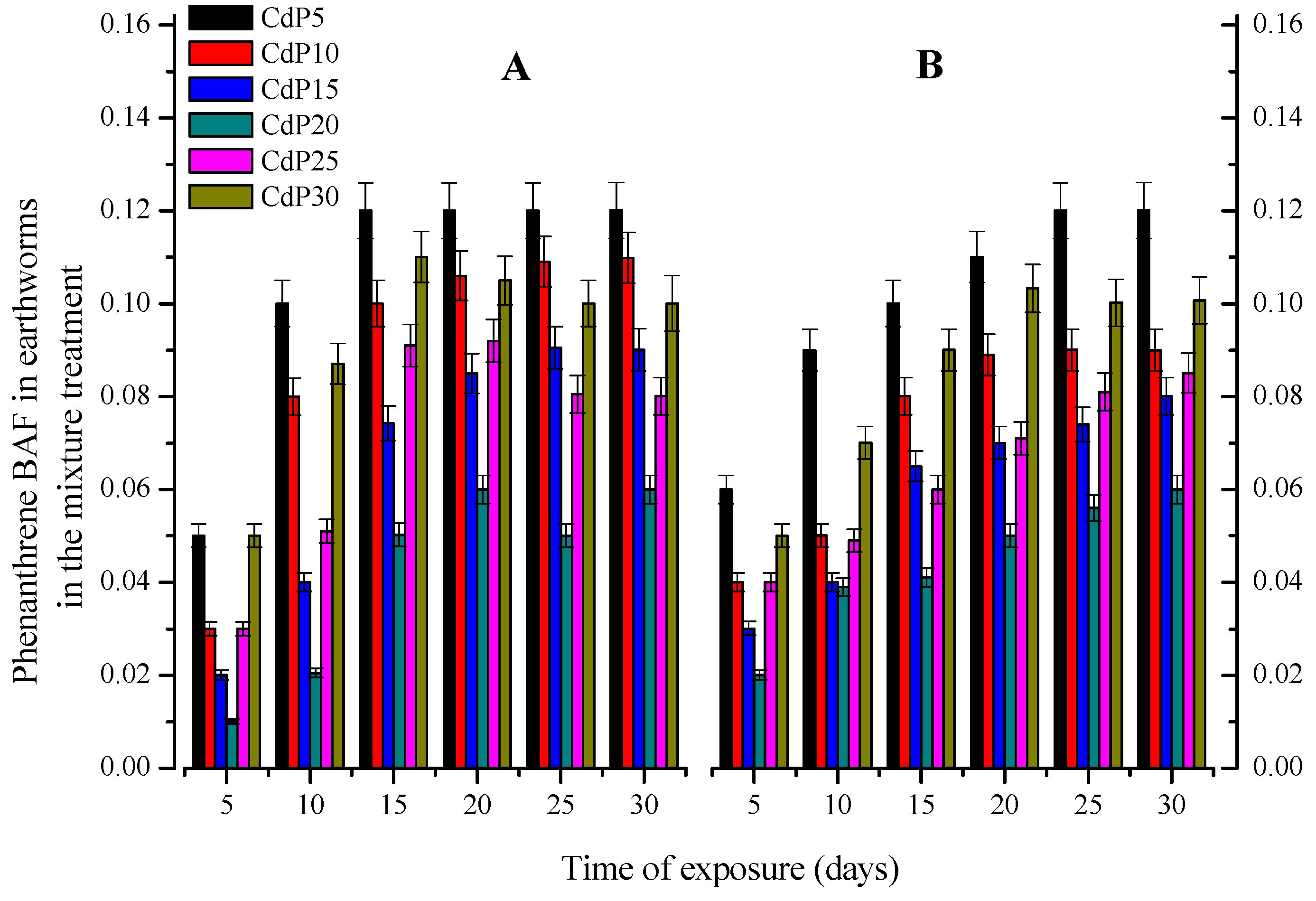
3.3. Chemicals Accumulation in Earthworms
3.3.1. Single Exposure Treatments
Cadmium
Phenanthrene
3.3.2. Combined Dose Treatments
4. Discussion
4.1. Mortality in Earthworms
4.2. Bioaccumulation and Earthworms Mortality
4.3. Body Weight Variation
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chen, M.; Xu, P.; Zeng, G.; Yang, C.; Huang, D.; Zhang, J. Bioremediation of soils contaminated with polycyclic aromatic hydrocarbons, petroleum, pesticides, chlorophenols and heavy metals by composting: Applications, microbes and future research needs. Biotechnol. Adv. 2015, 33, 745–755. [Google Scholar] [CrossRef] [PubMed]
- Monaco, D.; Chianese, E.; Riccio, A.; Delgado-Sanchez, A.; Lacorte, S. Spatial distribution of heavy hydrocarbons, PAHs and metals in polluted areas. The case of “Galicia”, Spain. Mar. Pollut. Bull. 2017, 121, 230–237. [Google Scholar] [CrossRef] [PubMed]
- Kremer, R.J. Environmental implications of herbicide resistance: Soil biology and ecology. Weed Sci. 2014, 62, 415–426. [Google Scholar] [CrossRef]
- Ding, J.; Jiang, X.; Guan, D.; Zhao, B.; Ma, M.; Zhou, B.; Cao, F.; Yang, X.; Li, L.; Li, J. Influence of inorganic fertilizer and organic manure application on fungal communities in a long-term field experiment of Chinese Mollisols. Appl. Soil Ecol. 2017, 111, 114–122. [Google Scholar] [CrossRef]
- De los Reyes, J.A. Mining shareholder value: Institutional shareholders, transnational corporations and the geography of gold mining. Geoforum 2017, 84, 251–264. [Google Scholar] [CrossRef]
- Ignatowicz, K. The impact of sewage sludge treatment on the content of selected heavy metals and their fractions. Environ. Res. 2017, 156, 19–22. [Google Scholar] [CrossRef] [PubMed]
- Alloway, B.J. Sources of heavy metals and metalloids in soils. In Heavy Metals in Soils; Springer: Berlin, Germany, 2013; pp. 11–50. [Google Scholar]
- Tang, W.-W.; Zeng, G.-M.; Gong, J.-L.; Liang, J.; Xu, P.; Zhang, C.; Huang, B.-B. Impact of humic/fulvic acid on the removal of heavy metals from aqueous solutions using nanomaterials: A review. Sci. Total Environ. 2014, 468, 1014–1027. [Google Scholar] [CrossRef] [PubMed]
- Dziubanek, G.; Baranowska, R.; Ćwieląg-Drabek, M.; Spychała, A.; Piekut, A.; Rusin, M.; Hajok, I. Cadmium in edible plants from Silesia, Poland, and its implications for health risk in populations. Ecotoxicol. Environ. Saf. 2017, 142, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Ci, D.; Jiang, D.; Dai, T.; Jing, Q.; Cao, W. Effects of cadmium on plant growth and physiological traits in contrast wheat recombinant inbred lines differing in cadmium tolerance. Chemosphere 2009, 77, 1620–1625. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Wang, L.; Zhu, L.; Wang, J. Individual and combined effects of enrofloxacin and cadmium on soil microbial biomass and the ammonia-oxidizing functional gene. Sci. Total Environ. 2018, 624, 900–907. [Google Scholar] [CrossRef] [PubMed]
- Árvay, J.; Tomáš, J.; Hauptvogl, M.; Massányi, P.; Harangozo, Ľ.; Tóth, T.; Stanovič, R.; Bryndzová, Š.; Bumbalová, M. Human exposure to heavy metals and possible public health risks via consumption of wild edible mushrooms from Slovak Paradise National Park, Slovakia. J. Environ. Sci. Health Part B 2015, 50, 833–843. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.-H.; Zeng, G.-M.; Niu, Q.-Y.; Liu, Y.; Zhou, L.; Jiang, L.-H.; Tan, X.-F.; Xu, P.; Zhang, C.; Cheng, M. Bioremediation mechanisms of combined pollution of PAHs and heavy metals by bacteria and fungi: A mini review. Bioresour. Technol. 2017, 224, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Waigi, M.G.; Kang, F.; Goikavi, C.; Ling, W.; Gao, Y. Phenanthrene biodegradation by sphingomonads and its application in the contaminated soils and sediments: A review. Int. Biodeterior. Biodegrad. 2015, 104, 333–349. [Google Scholar] [CrossRef]
- Maliszewska-Kordybach, B.; Klimkowicz-Pawlas, A.; Smreczak, B.; Janusauskaite, D. Ecotoxic effect of phenanthrene on nitrifying bacteria in soils of different properties. J. Environ. Qual. 2007, 36, 1635–1645. [Google Scholar] [CrossRef] [PubMed]
- Gaspar, M.; Cabello, M.; Cazau, M.; Pollero, R. Effect of phenanthrene and Rhodotorula glutinis on arbuscular mycorrhizal fungus colonization of maize roots. Mycorrhiza 2002, 12, 55–59. [Google Scholar] [CrossRef] [PubMed]
- Aksmann, A.; Tukaj, Z. The effect of anthracene and phenanthrene on the growth, photosynthesis, and SOD activity of the green alga Scenedesmus armatus depends on the PAR irradiance and CO2 level. Arch. Environ. Contam. Toxicol. 2004, 47, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Muratova, A.; Golubev, S.; Wittenmayer, L.; Dmitrieva, T.; Bondarenkova, A.; Hirche, F.; Merbach, W.; Turkovskaya, O. Effect of the polycyclic aromatic hydrocarbon phenanthrene on root exudation of Sorghum bicolor (L.) Moench. Environ. Exp. Bot. 2009, 66, 514–521. [Google Scholar] [CrossRef]
- Cassee, F.R.; Groten, J.P.; Van Bladeren, P.J.; Feron, V.J. Toxicological evaluation and risk assessment of chemical mixtures. Crit. Rev. Toxicol. 1998, 28, 73–101. [Google Scholar] [CrossRef] [PubMed]
- Uwizeyimana, H.; Wang, M.; Chena, W.; Khan, K. The eco-toxic effects of pesticide and heavy metal mixtures towards earthworms in soil. Environ. Toxicol. Pharmacol. 2017, 55, 20–29. [Google Scholar] [CrossRef] [PubMed]
- Lu, M.; Zhang, Z.-Z.; Wang, J.-X.; Zhang, M.; Xu, Y.-X.; Wu, X.-J. Interaction of heavy metals and pyrene on their fates in soil and tall fescue (Festuca arundinacea). Environ. Sci. Technol. 2014, 48, 1158–1165. [Google Scholar] [CrossRef] [PubMed]
- Gauthier, P.T.; Norwood, W.P.; Prepas, E.E.; Pyle, G.G. Metal-PAH mixtures in the aquatic environment: A review of co-toxic mechanisms leading to more-than-additive outcomes. Aquat. Toxicol. 2014, 154, 253–269. [Google Scholar] [CrossRef] [PubMed]
- Gust, K.A.; Fleeger, J.W. Exposure-related effects on Cd bioaccumulation explain toxicity of Cd-phenanthrene mixtures in Hyalella azteca. Environ. Toxicol. Chem. 2005, 24, 2918–2926. [Google Scholar] [CrossRef] [PubMed]
- Gust, K.A.; Fleeger, J.W. Exposure to cadmium-phenanthrene mixtures elicits complex toxic responses in the freshwater tubificid oligochaete, Ilyodrilus templetoni. Arch. Environ. Contam. Toxicol. 2006, 51, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Lu, Y.; Chen, W. Single and joint toxic effects of cadmium and phenanthrene on enchytraeid Fridericia bulbosa. Eur. J. Soil Biol. 2008, 44, 260–265. [Google Scholar] [CrossRef]
- Zhu, J.; Zhao, Z.-Y.; Lu, Y.-T. Evaluation of genotoxicity of combined soil pollution by cadmium and phenanthrene on earthworm. J. Environ. Sci. 2006, 18, 1210–1215. [Google Scholar] [CrossRef]
- Suthar, S.; Singh, S.; Dhawan, S. Earthworms as bioindicator of metals (Zn, Fe, Mn, Cu, Pb and Cd) in soils: Is metal bioaccumulation affected by their ecological category? Ecol. Eng. 2008, 32, 99–107. [Google Scholar] [CrossRef]
- Heydari, M.; Poorbabaei, H.; Bazgir, M.; Salehi, A.; Eshaghirad, J. Earthworms as indicators for different forest management types and human disturbance in Ilam oak forest, Iran. Folia For. Pol. 2014, 56, 121–134. [Google Scholar] [CrossRef]
- Cortet, J.; Gomot-De Vauflery, A.; Poinsot-Balaguer, N.; Gomot, L.; Texier, C.; Cluzeau, D. The use of invertebrate soil fauna in monitoring pollutant effects. Eur. J. Soil Biol. 1999, 35, 115–134. [Google Scholar] [CrossRef]
- Piron, D.; Boizard, H.; Heddadj, D.; Pérès, G.; Hallaire, V.; Cluzeau, D. Indicators of earthworm bioturbation to improve visual assessment of soil structure. Soil Tillage Res. 2017, 173, 53–63. [Google Scholar] [CrossRef]
- Lim, S.L.; Wu, T.Y.; Sim, E.Y.S.; Lim, P.N.; Clarke, C. Biotransformation of rice husk into organic fertilizer through vermicomposting. Ecol. Eng. 2012, 41, 60–64. [Google Scholar] [CrossRef]
- Dittbrenner, N.; Moser, I.; Triebskorn, R.; Capowiez, Y. Assessment of short and long-term effects of imidacloprid on the burrowing behaviour of two earthworm species (Aporrectodea caliginosa and Lumbricus terrestris) by using 2D and 3D post-exposure techniques. Chemosphere 2011, 84, 1349–1355. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Zhou, D.; Wang, P.; Peijnenburg, W.J. Kinetics of cadmium uptake and subcellular partitioning in the earthworm Eisenia fetida exposed to cadmium-contaminated soil. Arch. Environ. Contam. Toxicol. 2009, 57, 718–724. [Google Scholar] [CrossRef] [PubMed]
- Spurgeon, D.; Hopkin, S. Comparisons of metal accumulation and excretion kinetics in earthworms (Eisenia fetida) exposed to contaminated field and laboratory soils. Appl. Soil Ecol. 1999, 11, 227–243. [Google Scholar] [CrossRef]
- Peijnenburg, W.J.; Baerselman, R.; de Groot, A.C.; Jager, T.; Posthuma, L.; Van Veen, R.P. Relating environmental availability to bioavailability: Soil-type-dependent metal accumulation in the oligochaete Eisenia andrei. Ecotoxicol. Environ. Saf. 1999, 44, 294–310. [Google Scholar] [CrossRef] [PubMed]
- González-Alcaraz, M.N.; Loureiro, S.; van Gestel, C.A. Toxicokinetics of Zn and Cd in the earthworm Eisenia andrei exposed to metal-contaminated soils under different combinations of air temperature and soil moisture content. Chemosphere 2018, 197, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Palaniappan, P.R.; Karthikeyan, S. Bioaccumulation and depuration of chromium in the selected organs and whole body tissues of freshwater fish Cirrhinus mrigala individually and in binary solutions with nickel. J. Environ. Sci. 2009, 21, 229–236. [Google Scholar] [CrossRef]
- Song, Y.; Jing, X.; Fleischmann, S.; Wilke, B.-M. Comparative study of extraction methods for the determination of PAHs from contaminated soils and sediments. Chemosphere 2002, 48, 993–1001. [Google Scholar] [CrossRef]
- Shi, Z.-M.; Xu, L.; Hu, F. A hierarchic method for studying the distribution of phenanthrene in Eisenia fetida. Pedosphere 2014, 24, 743–752. [Google Scholar] [CrossRef]
- Contreras-Ramos, S.M.; Alvarez-Bernal, D.; Dendooven, L. Removal of polycyclic aromatic hydrocarbons from soil amended with biosolid or vermicompost in the presence of earthworms (Eisenia fetida). Soil Biol. Biochem. 2008, 40, 1954–1959. [Google Scholar] [CrossRef]
- Finney, D.J. Statistical Method in Biological Assay; Charles Griffin: London, UK, 1952. [Google Scholar]
- Schon, N.; Mackay, A.; Gray, R.; Van Koten, C.; Dodd, M. Influence of earthworm abundance and diversity on soil structure and the implications for soil services throughout the season. Pedobiologia 2017, 62, 41–47. [Google Scholar] [CrossRef]
- Anyanwu, I.N.; Semple, K.T. Effects of phenanthrene and its nitrogen-heterocyclic analogues aged in soil on the earthworm Eisenia fetida. Appl. Soil Ecol. 2016, 105, 151–159. [Google Scholar] [CrossRef]
- Wu, S.; Wu, E.; Qiu, L.; Zhong, W.; Chen, J. Effects of phenanthrene on the mortality, growth, and anti-oxidant system of earthworms (Eisenia fetida) under laboratory conditions. Chemosphere 2011, 83, 429–434. [Google Scholar] [CrossRef] [PubMed]
- Moreau, C.; Klerks, P.; Haas, C. Interaction between phenanthrene and zinc in their toxicity to the sheepshead minnow (Cyprinodon variegatus). Arch. Environ. Contam. Toxicol. 1999, 37, 251–257. [Google Scholar] [CrossRef] [PubMed]
- Gorria, M.; Tekpli, X.; Sergent, O.; Huc, L.; Gaboriau, F.; Rissel, M.; Chevanne, M.; Dimanche-Boitrel, M.T.; Lagadic-Gossmann, D. Membrane Fluidity Changes Are Associated with Benzo [a] Pyrene-Induced Apoptosis in F258 Cells. Ann. N. Y. Acad. Sci. 2006, 1090, 108–112. [Google Scholar] [CrossRef] [PubMed]
- Biswas, B.; Sarkar, B.; Mandal, A.; Naidu, R. Heavy metal-immobilizing organoclay facilitates polycyclic aromatic hydrocarbon biodegradation in mixed-contaminated soil. J. Hazard. Mater. 2015, 298, 129–137. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Yuan, X.; Lin, X.; Sun, B.; Zhao, Z. Low-molecular-weight organic acids enhance the release of bound PAH residues in soils. Soil Tillage Res. 2015, 145, 103–110. [Google Scholar] [CrossRef]
- Kitturmath, M.; Giraddi, R.; Viraktamath, S.; Sattigi, H. Evaluation of different feeding additives for bio-degradation of groundnut shell and rice husk using earthworm, Eudrilus eugeniae (Kingberg). Karnataka J. Agric. Sci. 2010, 17, 52–56. [Google Scholar]
- Wu, B.; Liu, Z.; Xu, Y.; Li, D.; Li, M. Combined toxicity of cadmium and lead on the earthworm Eisenia fetida (Annelida, Oligochaeta). Ecotoxicol. Environ. Saf. 2012, 81, 122–126. [Google Scholar] [CrossRef] [PubMed]
- Maleri, R.; Reinecke, A.; Reinecke, S. Metal uptake of two ecophysiologically different earthworms (Eisenia fetida and Aporrectodea caliginosa) exposed to ultramafic soils. Appl. Soil Ecol. 2008, 38, 42–50. [Google Scholar] [CrossRef]
- Liang, S.-H.; Chen, S.-C.; Chen, C.-Y.; Kao, C.-M.; Yang, J.-I.; Shieh, B.-S.; Chen, J.-H.; Chen, C.-C. Cadmium-induced earthworm metallothionein-2 is associated with metal accumulation and counteracts oxidative stress. Pedobiologia 2011, 54, 333–340. [Google Scholar] [CrossRef]
- Rodriguez-Campos, J.; Dendooven, L.; Alvarez-Bernal, D.; Contreras-Ramos, S.M. Potential of earthworms to accelerate removal of organic contaminants from soil: A review. Appl. Soil Ecol. 2014, 79, 10–25. [Google Scholar] [CrossRef]
- García-Segura, D.; Castillo-Murrieta, I.M.; Martínez-Rabelo, F.; Gomez-Anaya, A.; Rodríguez-Campos, J.; Hernández-Castellanos, B.; Contreras-Ramos, S.M.; Barois, I. Macrofauna and mesofauna from soil contaminated by oil extraction. Geoderma 2017, 332, 180–189. [Google Scholar] [CrossRef]
- Wu, S.; Xu, X.; Zhao, S.; Shen, F.; Chen, J. Evaluation of phenanthrene toxicity on earthworm (Eisenia fetida): An ecotoxicoproteomics approach. Chemosphere 2013, 93, 963–971. [Google Scholar] [CrossRef] [PubMed]


| Time | d-0 | d-5 | d-10 | d-15 | d-20 | d-25 | d-30 |
|---|---|---|---|---|---|---|---|
| E. fetida | 1.84 ± 0.5 a | 1.83 ± 0.6 a | 1.72 ± 0.5 b | 1.71 ± 0.6 b | 1.67 ± 0.4 c | 1.65 ± 0.4 c | 1.64 ± 0.7 c |
| A. caliginosa | 3.03 ± 0.5 a | 3.01 ± 0.5 a | 2.98 ± 0.4 b | 2.94 ± 0.5 bc | 2.94 ± 0.6 bc | 2.91 ± 0.6 bc | 2.82 ± 0.5 c |
| Eisenia fetida Weight (g) | |||||||
| Time | d-0 | d-5 | d-10 | d-15 | d-20 | d-25 | d-30 |
| CdP5 | 1.84 ± 0.5 a | 1.82 ± 0.6 a | 1.74 ± 0.4 b | 1.71 ± 0.6 b | 1.67 ± 0.5 c | 1.66 ± 0.4 c | 1.65 ± 0.8 c |
| CdP10 | 1.83 ± 0.4 a | 1.81 ± 0.5 a | 1.72 ± 0.6 b | 1.73 ± 0.7 b | 1.68 ± 0.6 c | 1.66 ± 0.5 c | 1.65 ± 0.6 c |
| CdP15 | 1.84 ± 0.5 a | 1.82 ± 0.7 a | 1.74 ± 0.4 b | 1.72 ± 0.5 b | 1.67 ± 0.5 c | 1.67 ± 0.6 c | 1.65 ± 0.5 c |
| CdP20 | 1.83 ± 0.6 a | 1.83 ± 0.6 a | 1.73 ± 0.6 b | 1.72 ± 0.8 b | 1.68 ± 0.6 c | 1.66 ± 0.8 c | 1.65 ± 0.4 c |
| CdP25 | 1.84 ± 0.8 a | 1.82 ± 0.5 a | 1.74 ± 0.5 b | 1.71 ± 0.5 b | 1.67 ± 0.5 c | 1.65 ± 0.5 c | 1.65 ± 0.6 c |
| CdP30 | 1.84 ± 0.6 a | 1.82 ± 0.5 a | 1.72 ± 0.6 b | 1.70 ± 0.6 b | 1.67 ± 0.5 c | 1.65 ± 0.5 c | 1.64 ± 0.5 c |
| Aporrectodea caliginosa Weight (g) | |||||||
| CdP5 | 3.03 ± 0.5 a | 3.01 ± 0.6 a | 2.98 ± 0.5 a | 2.94 ± 0.8 b | 2.94 ± 0.5 b | 2.91 ± 0.6 bc | 2.83 ± 0.5 c |
| CdP10 | 3.02 ± 0.4 a | 3.01 ± 0.5 a | 2.98 ± 0.6 a | 2.95 ± 0.5 b | 2.95 ± 0.5 b | 2.92 ± 0.5 bc | 2.83 ± 0.6 c |
| CdP15 | 3.02 ± 0.6 a | 3.02 ± 0.6 a | 2.99 ± 0.5 a | 2.95 ± 0.6 b | 2.95 ± 0.6 b | 2.92 ± 0.5 bc | 2.83 ± 0.6 c |
| CdP20 | 3.03 ± 0.6 a | 3.02 ± 0.5 a | 2.99 ± 0.5 a | 2.95 ± 0.4 b | 2.95 ± 0.6 b | 2.92 ± 0.5 bc | 2.84 ± 0.7 c |
| CdP25 | 3.02 ± 0.7 a | 3.02 ± 0.7 a | 2.99 ± 0.6 a | 2.96 ± 0.6 b | 2.96 ± 0.5 b | 2.93 ± 0.4 bc | 2.84 ± 0.5 c |
| CdP30 | 3.03 ± 0.3 a | 3.02 ± 0.5 a | 2.99 ± 0.7 a | 2.95 ± 0.4 b | 2.97 ± 0.8 b | 2.92 ± 0.5 bc | 2.84 ± 0.6 c |
| Treatment | Eisenia fetida | Aporrectodea caliginosa | ||||
|---|---|---|---|---|---|---|
| BAF | k1 (d−1) | k2 (d−1) | BAF | k1 (d−1) | k2 (d−1) | |
| Cd0.5 | 1.31 ± 0.3 a* | 0.024 ± 0.018 a† | 0.018 ± 0.06 d‡ | 1.72 ± 0.4 a* | 0.022 ± 0.02 a† | 0.013 ± 0.04 d† |
| Cd1 | 1.20 ± 0.5 a* | 0.05 ± 0.020 a† | 0.041 ± 0.04 c† | 1.58 ± 0.2 a* | 0.030 ± 0.02 a† | 0.019 ± 0.01 c‡ |
| Cd3 | 0.39 ± 0.3 b* | 0.02 ± 0.021 a† | 0.048 ± 0.07 c† | 0.49 ± 0.1 b* | 0.010 ± 0.10 ab† | 0.021 ± 1.02 bc† |
| Cd5 | 0.28 ± 0.2 c* | 0.021 ± 0.02 a‡ | 0.073 ± 0.01 b† | 0.31 ± 0.1 c* | 0.012 ± 0.10 ab‡ | 0.041 ± 1.01 b† |
| Cd10 | 0.22 ± 0.3 d* | 0.018 ± 0.06 ab‡ | 0.084 ± 0.02 a† | 0.27 ± 0.2 d* | 0.017 ± 0.24 a‡ | 0.063 ± 1.22 a† |
| Time | d-5 | d-10 | d-15 | d-20 | d-25 | d-30 |
|---|---|---|---|---|---|---|
| E. fetida | 0.30 ± 0.67 b | 0.36 ± 0.55 ab | 0.38 ± 0.47 a | 0.39 ± 0.48 a | 0.39 ± 0.43 a | 0.41 ± 0.43 a |
| A. caliginosa | 0.45 ± 0.81 b | 0.46 ± 0.51 ab | 0.47 ± 0.41 ab | 0.50 ± 0.36 a | 0.51 ± 0.39 a | 0.52 ± 0.58 a |
| Treatments | Eisenia fetida | Aporrectodea caliginosa | ||||
|---|---|---|---|---|---|---|
| BAF | k1 (d−1) | k2 (d−1) | BAF | k1 (d−1) | k2 (d−1) | |
| P5 | 0.42 ± 0.3 a* | 0.02 ± 0.06 c† | 0.05 ± 0.02 bc† | 0.47 ± 0.3 a* | 0.03 ± 0.06 ab† | 0.06 ± 0.02 d† |
| P10 | 0.23 ± 0.2 ab** | 0.016 ± 0.02 c‡ | 0.07 ± 0.01 bc† | 0.31 ± 0.2 ab* | 0.031 ± 0.06 ab‡ | 0.10 ± 0.03 c† |
| P15 | 0.22 ± 0.3 ab* | 0.02 ± 0.09 c‡ | 0.09 ± 0.03 b† | 0.27 ± 0.1 ab* | 0.03 ± 0.02 ab‡ | 0.11 ± 0.02 c† |
| P20 | 0.21 ± 0.3 ab* | 0.25 ± 0.06 b† | 0.12 ± 0.02 b† | 0.25 ± 0.3 ab* | 0.03 ± 0.09 ab‡ | 0.12 ± 0.03 c† |
| P25 | 0.22 ± 0.1 ab* | 0.05 ± 0.03 c‡ | 0.25 ± 0.03 a† | 0.25 ± 0.4 ab* | 0.07 ± 0.08 a‡ | 0.27 ± 0.02 b† |
| P30 | 0.22 ± 0.2 ab* | 0.6 ± 0.01 a† | 0.27 ± 0.05 a‡ | 0.24 ± 0.5 b* | 0.09 ± 0.02 a‡ | 0.39 ± 0.04 a† |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elyamine, A.M.; Afzal, J.; Rana, M.S.; Imran, M.; Cai, M.; Hu, C. Phenanthrene Mitigates Cadmium Toxicity in Earthworms Eisenia fetida (Epigeic Specie) and Aporrectodea caliginosa (Endogeic Specie) in Soil. Int. J. Environ. Res. Public Health 2018, 15, 2384. https://doi.org/10.3390/ijerph15112384
Elyamine AM, Afzal J, Rana MS, Imran M, Cai M, Hu C. Phenanthrene Mitigates Cadmium Toxicity in Earthworms Eisenia fetida (Epigeic Specie) and Aporrectodea caliginosa (Endogeic Specie) in Soil. International Journal of Environmental Research and Public Health. 2018; 15(11):2384. https://doi.org/10.3390/ijerph15112384
Chicago/Turabian StyleElyamine, Ali Mohamed, Javaria Afzal, Muhammad Shoaib Rana, Muhammad Imran, Miaomiao Cai, and Chengxiao Hu. 2018. "Phenanthrene Mitigates Cadmium Toxicity in Earthworms Eisenia fetida (Epigeic Specie) and Aporrectodea caliginosa (Endogeic Specie) in Soil" International Journal of Environmental Research and Public Health 15, no. 11: 2384. https://doi.org/10.3390/ijerph15112384
APA StyleElyamine, A. M., Afzal, J., Rana, M. S., Imran, M., Cai, M., & Hu, C. (2018). Phenanthrene Mitigates Cadmium Toxicity in Earthworms Eisenia fetida (Epigeic Specie) and Aporrectodea caliginosa (Endogeic Specie) in Soil. International Journal of Environmental Research and Public Health, 15(11), 2384. https://doi.org/10.3390/ijerph15112384







