Effect of a 12-Week Online Walking Intervention on Health and Quality of Life in Cancer Survivors: A Quasi-Randomized Controlled Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants and Setting
2.2. Randomization and Blinding
2.3. Ethical Approval
2.4. Protocol
2.5. Intervention
2.5.1. STRIDE Website
2.5.2. Step Goals
2.6. Outcome Measures
2.6.1. Physical Activity
2.6.2. Anthropometry
2.6.3. Physiological Measures
2.6.4. Physical Fitness
2.6.5. Functional Status and Quality of Life (QOL)
2.7. Demographics
2.8. Data Analysis
3. Results
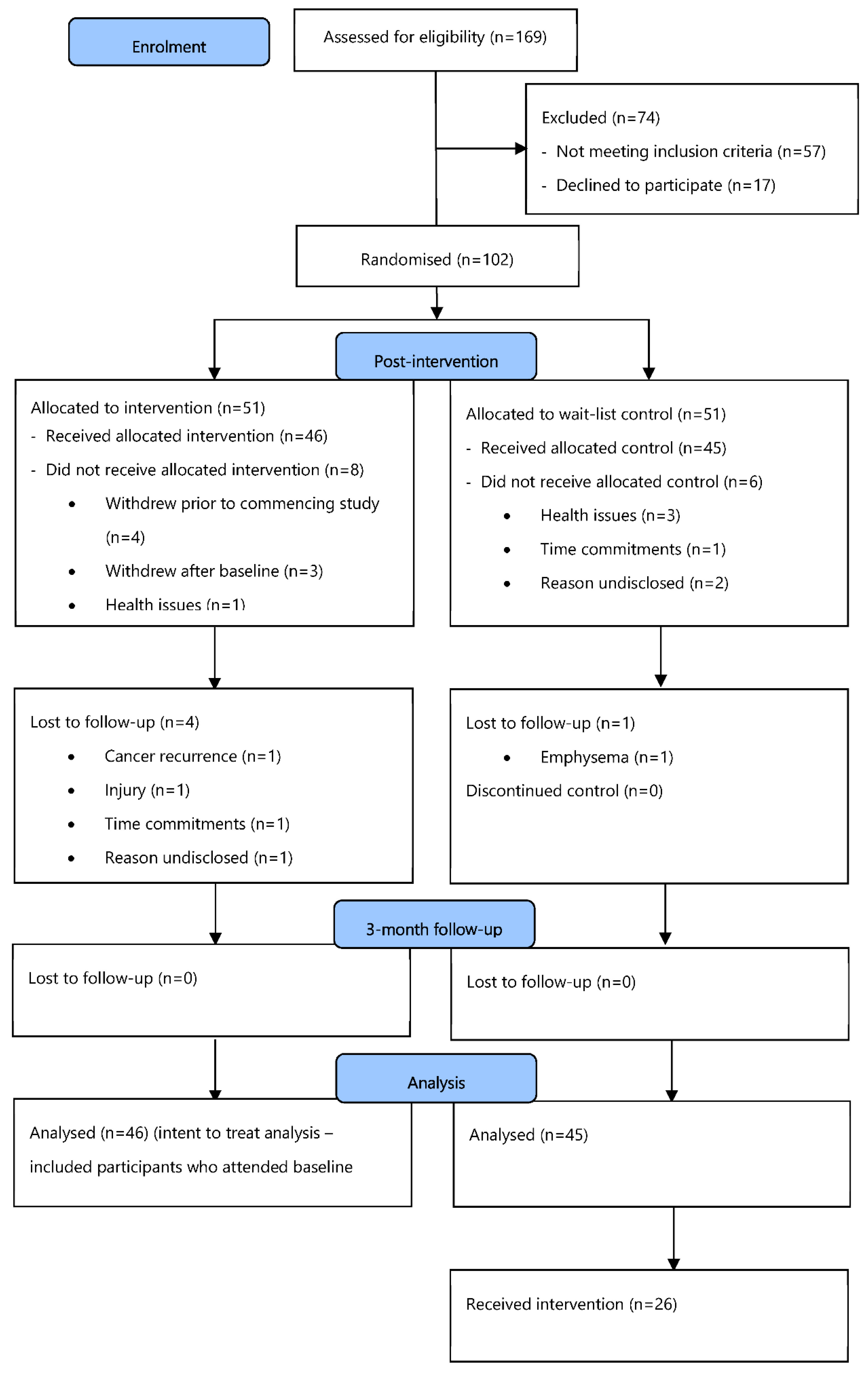
3.1. Participants
3.2. Reliability of Constructs Derived from SF-36v2
3.3. Visits to STRIDE Website
3.4. Effect of the Intervention on Outcome Variables
3.4.1. Health Variables
3.4.2. Physical Fitness (6MWT)
3.4.3. SF-36 Scores
3.4.4. Physiological Variables
- Blood pressure
- BMI
- Waist girth
3.4.5. Quality of Life (QOL)
4. Discussion
4.1. QOL
4.2. Physical Fitness (6MWT)
4.3. Blood Pressure
4.4. BMI
4.5. Waist Girth
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- National Cancer Institute. Surveillance, epidemiology and End Results Program. Available online: http://seer.cancer.gov/statfacts/html/all.html (accessed on 21 September 2018).
- Allemani, C.; Matsuda, T.; Di Carlo, V.; Harewood, R.; Matz, M.; Nikšić, M.; Bonaventure, A.; Valkov, M.; Christopher, J.; Johnson, C.J.; et al. Global surveillance of trends in cancer survival 2000–2014 (CONCORD-3): Analysisof individual records for 37513025 patients diagnosed with one of 18 cancers from 322 population-based registries in 71 countries. Lancet 2018, 391, 1023–1075. [Google Scholar] [CrossRef]
- Australian Institute of Health and Welfare. Cancers in Australia 2017. Available online: https://www.aihw.gov.au/reports/cancer/cancer-in-australia-2017/contents/summary (accessed on 21 September 2018).
- Glare, P.A.; Davies, P.S.; Finlay, E.; Gulati, A.; Lemanne, D.; Moryl, N.; Oeffinger, K.C.; Paice, J.A.; Stubblefield, M.D.; Syrjala, K.L. Pain in cancer survivors. J. Clin. Oncol. 2014, 32, 1739–1747. [Google Scholar] [CrossRef] [PubMed]
- Corbett, T.; Groarke, A.; Walsh, J.C.; McGuire, B.E. Cancer-related fatigue in post-treatment cancer survivors: Application of the common sense model of illness representations. BMC Cancer 2017, 3, e8. [Google Scholar] [CrossRef] [PubMed]
- Fidler, M.M.; Reulen, R.C.; Winter, D.L.; Kelly, J.; Jenkinson, H.C.; Skinner, R.; Frobisher, C.; Hawkins, M.M. Long term cause specific mortality among 34489 five year survivors of childhood cancer in Great Britain: Population based cohort study. BMJ 2016, 354. [Google Scholar] [CrossRef]
- Deimling, G.T.; Kahana, B.; Bowman, K.F.; Schaefer, M.L. Cancer survivorship and psychological distress in later life. Psychooncology 2002, 11, 479–494. [Google Scholar] [CrossRef] [PubMed]
- Arndt, V.; Koch-Gallenkamp, L.; Jansen, L.; Bertram, H.; Eberle, A.; Holleczek, B.; Schmid-Höpfner, S.; Waldmann, A.; Zeissig, S.R.; Brenner, H. Quality of life in long-term and very long-term cancer survivors versus population controls in Germany. Acta Oncol. 2017, 56, 190–197. [Google Scholar] [CrossRef] [PubMed]
- Gunnell, A.S.; Joyce, S.; Tomlin, S.; Taaffe, D.R.; Prue, C.; Newton, R.U.; Joseph, D.; Spry, N.; Einarsdóttir, K.; Galvão, D.A. Physical activity and survival among long-term cancer survivor and non-cancer cohorts. Front. Public Health 2017. [Google Scholar] [CrossRef] [PubMed]
- Friedenreich, C.M.; Neilson, H.K.; Farris, M.S.; Courneya, K.S. Physical activity and cancer outcomes: A precision medicine approach. Clin. Cancer Res. 2016, 22, 4766–4775. [Google Scholar] [CrossRef] [PubMed]
- Burke, S.; Wurz, A.; Bradshaw, A.; Saunders, S.; West, M.A.; Brunet, J. Physical Activity and Quality of Life in Cancer Survivors: A Meta-Synthesis of Qualitative Research. Cancers 2017, 9, 53. [Google Scholar] [CrossRef] [PubMed]
- Gjerset, G.M.; Fosså, S.D.; Courneya, K.S.; Skovlund, E.; Thorsen, L. Exercise behavior in cancer survivors and associated factors. J. Cancer Surviv. 2011, 5, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Pinto, B.M.; Ciccolo, J.T. Physical activity motivation and cancer survivorship. Recent Results Cancer Res. 2011, 186, 367–387. [Google Scholar] [PubMed]
- Jong, K.E.; Smith, D.P.; Yu, X.Q.; O’Connell, D.L.; Goldstein, D.; Armstrong, B.K. Remoteness of residence and survival from cancer in New South Wales. Med. J. Aust. 2004, 180, 618–622. [Google Scholar] [PubMed]
- Heathcote, K.; Armstrong, B. Disparities in cancer outcomes in regional and rural Australia. Cancer Forum 2007, 31, 70–73. [Google Scholar]
- Jong, K.E.; Vale, P.J.; Armstrong, B.K. Rural inequalities in cancer care and outcome. Med. J. Aust. 2005, 182, 13–14. [Google Scholar] [PubMed]
- Noyes, K.; Holub, D.; Rizvi, I.; Swanger, A.; Reisdorf, C.; Tomaszewski, K.; Constine, L.S. Cancer survivorship care in rural community: Patient perspective. J. Clin. Oncol. 2017, 35. [Google Scholar] [CrossRef]
- Milton, K.; Bull, F.C.; Bauman, A. Reliability and validity testing of a single-item physical activity measure. Br. J. Sports Med. 2011, 45, 203–208. [Google Scholar] [CrossRef] [PubMed]
- Sports Medicine Australia. Sports Medicine Australia (SMA) Pre-Exercise Screening System 2005; Sports Medicine Australia: Canberra, Australia, 2005. [Google Scholar]
- Frensham, L.; Zarnowiecki, D.; Parfitt, G.; Stanley, R.; Dollman, J. Steps toward improving diet and exercise for cancer survivors (STRIDE): A quasi-randomised controlled trial protocol. BioMed. Cent. Cancer 2014, 14, 428. [Google Scholar] [CrossRef] [PubMed]
- Verhagen, A.; de Vet, H.; de Bie, R.; Kessels, A.; Boers, M.; Bouter, L.; Knipschild, P. The Delphi list: A criteria list for quality assessment of randomized clinical trials for conducting systematic reviews developed by Delphi consensus. J. Clin. Epidemiol. 1998, 51, 1235–1241. [Google Scholar] [CrossRef]
- American Thoracic Society. ATS Statement: Guidelines for the six minute walk test. Am. J. Respir. Crit. Care Med. 2002, 166, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Schneider, P.; Crouter, S.E.; Lukajic, O.; Bassett, D.R.J. Accuracy and reliability of pedometers for measuring steps over a 400-m walk. Med. Sci. Sports Exerc. 2003, 35, 1779–1784. [Google Scholar] [CrossRef] [PubMed]
- National Health and Medical Research Council. Australian Dietary Guidelines: Providing the Scientific Evidence for Healthier Australian Diets; National Health and Medical Research Council: Canberra, Australia, 2013.
- Borg, G. Borg’s Perceived Exertion and Pain Scales; Human Kinetics: Champaign, IL, USA, 1998. [Google Scholar]
- Borg, G. Perceived exertion as an indicator of somatic stress. Scand. J. Rehabil. Med. 1970, 2, 92–98. [Google Scholar] [PubMed]
- Horowitz, M.B.; Mahler, D.A. Dyspnea ratings for prescription of cross-modal exercise in patients with COPD. Chest 1998, 113, 60–64. [Google Scholar] [CrossRef] [PubMed]
- Bryan, A.; Hutchison, K.E.; Seals, D.R.; Allen, D.L. A transdisciplinary modelintegrating genetic, physiological, and psychological correlates of voluntary exercise. Health Psychol. 2007, 26, 30–39. [Google Scholar] [CrossRef] [PubMed]
- Hardy, C.; Rejeski, J. Not what, but how one feels: The measurement of affect during exercise. J. Sport Exerc. Psychol. 1989, 11, 304–317. [Google Scholar] [CrossRef]
- Ekkekakis, P.; Parfitt, G.; Petruzzello, S.J. The pleasure and displeasure people feel when they exercise at different intensities: Decennial update and progress towards a tripartite rationale for exercise intensity prescription. Sports Med. 2011, 41, 641–671. [Google Scholar] [CrossRef] [PubMed]
- Drenowatz, C.; Eisenmann, J.; Pfeiffer, K.; Welk, G.; Heelan, K.; Gentile, D.; Walsh, D. Influence of socio-economic status on habitual physical activity and sedentary behavior in 8- to 11-year old children. BMC Public Health 2010, 10, 214. [Google Scholar] [CrossRef] [PubMed]
- Frensham, L.J.; Parfitt, G.; Dollman, J. Predicting engagement with online walking promotion among metropolitan and rural cancer survivors. Cancer Nurs. 2018. [Google Scholar]
- Stewar, A.; Marfell-Jones, M.; International Society for the Advancement of Kinanthropometry. International Standards for Anthropometric Assessment; The International Society for the Advancement of Kinanthropometry: Lower Hutt, New Zealand, 2011. [Google Scholar]
- Solway, S.; Brooks, D.; Lacasse, Y.; Thomas, S. A qualitative systematic overview of the measurement properties of functional walk tests used in the cardiorespiratory domain. Chest 2001, 119, 256–270. [Google Scholar] [CrossRef] [PubMed]
- Sanson-Fisher, R.W.; Perkins, J. Adaptation and validation of the SF-36 health survey for use in Australia. J. Clin. Epidemiol. 1998, 5, 961–967. [Google Scholar] [CrossRef]
- Ware, J.E. User’s Manual for the SF-36v2 Health Survey, 2nd ed.; Quality Health Metric Incorporated: Lincoln, RI, USA, 2007. [Google Scholar]
- Andersen, M.R.; Bowen, D.J.; Morea, J.; Stein, K.D.; Baker, F. Involvement in decision-making and breast cancer survivor quality of life. Health Psychol. 2009, 28, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Ware, J.E.; Sherboume, C.D. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med. Care 1992, 30, 473–483. [Google Scholar] [CrossRef] [PubMed]
- RAND Health. Medical Outcomes Study: 36-Item Short Form Survey Scoring Instructions. Available online: http://www.rand.org/health/surveys_tools/mos/mos_core_36item_scoring.html (accessed on 15 June 2016).
- Nunnally, J.C. Psychometric Theory, 2nd ed.; McGraw Hill: New York, NY, USA, 1978. [Google Scholar]
- Prochaska, J.O.; DiClemente, C.C. The transtheoretical appoach. In Handbook of Eclectic Psychotherapy; Norcross, J., Ed.; Brunner/Mazel: New York, NY, USA, 1986. [Google Scholar]
- Blanchard, C.; Corneya, K.; Rogers, W.; Murnaghan, D. Determinants of exercise intention and behavior in survivors of breast and prostate cancer: An application of the theory of planned behavior. Cancer Nurs. 2002, 25, 88–95. [Google Scholar] [CrossRef] [PubMed]
- Mikkelsen, K.; Stojanovska, L.; Polenakovic, M.; Bosevski, M.; Apostolopoulos, V. Exercise and mental health. Maturitas 2017, 106, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Frensham, L.J. Promoting Regular Walking among Adult Cancer Survivors Using an Online Interactive Resource: An Evaluation of Project Stride; University of South Australia: Adelaide, Australia, 2015. [Google Scholar]
- Pinto, B.; Frierson, G.M.; Rabin, C.; Trunzo, J.J.; Marcus, B.H. Home-based physical activity intervention for breast cancer patients. J. Clin. Oncol. 2005, 23, 3577–3587. [Google Scholar] [CrossRef] [PubMed]
- Vallance, J.K.H.; Courneya, K.S.; Plontikoff, C.; Yasui, Y.; Mackey, J.R. Randomized controlled trial of the effects of print materials and step pedometers on physical activity and quality of life in breast cancer survivors. J. Clin. Oncol. 2007, 25, 2352–2359. [Google Scholar] [CrossRef] [PubMed]
- Knols, R.; de Bruin, E.D.; Uebelhart, D.; Aufdemkampe, G.; Schanz, U.; Stenner-Liewen, F.; Hitz, F.; Taverna, C.; Aaronson, N.K. Effects of an outpatient physical exercise program on hematopoietic stem-cell transplantation recipients: A randomized clinical trial. Bone Marrow Traspl. 2011, 46, 1245–1255. [Google Scholar] [CrossRef] [PubMed]
- Shelton, M.L.; Lee, J.Q.; Morris, G.S.; Massey, P.R.; Kendall, D.G.; Munsell, M.F.; Anderson, K.O.; Simmonds, M.J.; Giralt, S.A. A randomized control trial of a supervised versus a self-directed exercise program for allogeneic stem cell transplant patients. Psychooncology 2009, 18, 353–359. [Google Scholar] [CrossRef] [PubMed]
- Tudor-Locke, C.; Craig, C.L.; Aoyagi, Y.; Bell, R.; Croteau, K.A.; De Bourdeaudhuij, I.; Ewald, B.; Gardner, A.W.; Hatano, Y.; Lutes, L.D.; et al. How many steps/day are enough? For older adults and special populations. BMC 2011, 8, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Miller, R.; Brown, W.J.; Tudor-Locke, C. But what about swimming and cycling? How to “count” non-ambulatory activity when using pedometers to assess physical activity. J. Phys. Act. Health 2006, 3, 257–266. [Google Scholar] [CrossRef] [PubMed]
- Rivera-Brown, A.M.; Frontera, W.R. Principles of exercise physiology: Responses to acute exercise and long-term adaptations to training. PM R 2012, 4, 797–804. [Google Scholar] [CrossRef] [PubMed]
- Andersson, M.; Moberg, L.; Svantesson, U.; Sundbom, A.; Johansson, H.; Emtner, M. Measuring walking speed in COPD: Test-retest reliability of the 30-metre walk test and comparison with the 6-minute walk test. Prim. Care Respir. J. 2011, 20, 434–440. [Google Scholar] [CrossRef] [PubMed]
- Skough, K.; Broman, L.; Borg, K. Test-retest reliability of the 6-min walk test in patients with postpolio syndrome. Int. J. Rehabil. Res. 2013, 36, 140–145. [Google Scholar] [CrossRef] [PubMed]
- Whelton, S.P.; Chin, A.; Xin, X.; He, J. Effect of aerobic exercise on blood pressure: A meta-analysis of randomized controlled trials. Ann. Intern. Med. 2002, 136, 493–503. [Google Scholar] [CrossRef] [PubMed]
- Murphy, M.; Nevill, A.M.; Murtagh, E.M.; Holder, R.L. The effect of walking on fitness, fatness, and resting blood pressure: A meta-analysis of randomised controlled trials. Prev. Med. 2007, 44, 377–385. [Google Scholar] [CrossRef] [PubMed]
- Lewington, S.; Clarke, R.; Qizilbash, N.; Peto, R.; Collins, R.; Prospective Studies Collaboration. Age-specific relevance of usual blood pressure to vascular mortality: A meta-analysis of individual data for one million adults in 61 prospective studies. Lancet 2002, 360, 1903–1913. [Google Scholar] [PubMed]
- Beevers, G.G.; Lip, G.Y.; O’Brien, E. ABC of hypertension. Blood pressure measurement. Part I-sphygmomanometry: Factors common to all techniques. BMJ 2001, 21, 981–985. [Google Scholar] [CrossRef]
- Bravata, D.; Smith-Spangler, C.; Sundaram, V.; Gienger, A.; Lin, N.; Lewis, R.; Stave, C.; Olkin, I.; Sirard, J. Using pedometers to increase physical activity and improve health: A systematic review. JAMA 2007, 298, 2296–2304. [Google Scholar] [CrossRef] [PubMed]
- Rothman, K.J. BMI-related errors in the measurement of obesity. Int. J. Obes. 2008, 3, S56–S59. [Google Scholar] [CrossRef] [PubMed]
- Huxley, R.; Mendis, S.; Zheleznyakov, E.; Reddy, S.; Chan, J. Body mass index, waist circumference and waist:hip ratio as predictors of cardiovascular risk–a review of the literature. Eur. J. Clin. Nutr. 2010, 64, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Wilson, D.B.; Porter, J.S.; Parker, G.; Kilpatrick, J. Anthropometric changes using a walking intervention in African American breast cancer survivors: A pilot study. Prev. Chronic Dis. 2005, 2, A16. [Google Scholar] [PubMed]
- Ryan, R.M.; Deci, E.L. Intrinsic and extrinsic motivations: Classic definitions and new directions. Contemp. Educ. Psychol. 2000, 25, 54–67. [Google Scholar] [CrossRef] [PubMed]
- Ryan, R.M.; Patrick, H.; Deci, E.L.; Williams, G.C. Facilitating health behaviour change and its maintenance: Interventions based on Self-Determination Theory. Eur. Health Psychol. 2008, 10, 2–5. [Google Scholar]


| Characteristics | Intervention (n = 46) | Control (n = 45) | ||
|---|---|---|---|---|
| n | (%) | n | (%) | |
| Age, y | ||||
| Mean | 65.2 | 66.1 | ||
| SD | 9.3 | 9.4 | ||
| Range | 29–78 | 44–86 | ||
| Sex | ||||
| Male | 21 | (45.7) | 23 | (51.1) |
| Female | 25 | (54.3) | 22 | (48.9) |
| Location | ||||
| Metropolitan | 24 | (52.2) | 24 | (53.3) |
| Rural | 22 | (47.8) | 21 | (46.7) |
| Marital status | ||||
| Married/living | 37 | (80.4) | 35 | (77.8) |
| with partner | ||||
| Not married | 9 | (19.6) | 10 | (22.2) |
| Race/ethnicity | ||||
| Caucasian | 45 | (97.8) | 42 | (93.3) |
| Asian | 0 | (0) | 2 | (4.4) |
| ATSI | 1 | (2.2) | 1 | (2.2) |
| Education | ||||
| <High school | 0 | (0) | 2 | (4.4) |
| High school or diploma | 31 | (67.4) | 35 | (77.8) |
| University | 15 | (32.6) | 7 | (15.6) |
| SES (SEIFA) | ||||
| Low | 18 | (39.1) | 18 | (40.0) |
| Middle | 18 | (39.1) | 14 | (31.1) |
| High | 10 | (21.7) | 12 | (26.7) |
| BMI, kg/m2 | ||||
| Normal (<25) | 6 | (13.0) | 9 | (20.0) |
| Overweight (25–29) | 15 | (32.6) | 16 | (35.6) |
| Obese (30–39) | 23 | (50.0) | 18 | (40.0) |
| Morbidly obese (40+) | 2 | (4.3) | 2 | (4.4) |
| Smoking status | ||||
| Smoker | 0 | (0) | 4 | (8.9) |
| Ex-smoker | 11 | (23.9) | 19 | (42.2) |
| Non-smoker | 35 | (76.1) | 22 | (48.9) |
| Length of time using Internet, y | ||||
| First time | 1 | (2.2) | 4 | (8.9) |
| <6–12 months | 0 | (0) | 6 | (13.3) |
| 1–3 | 6 | (13.0) | 1 | (2.2) |
| 4–6 | 5 | (10.9) | 5 | (11.1) |
| ≥7 | 34 | (73.9) | 29 | (64.4) |
| Characteristics | Intervention (n = 46) | Control (n = 45) | ||
|---|---|---|---|---|
| n | (%) | n | (%) | |
| Cancer type | ||||
| Breast | 21 | (45.7) | 17 | (37.8) |
| Prostate | 9 | (19.6) | 11 | (24.4) |
| Colorectal | 5 | (10.9) | 4 | (8.9) |
| Head and neck | 3 | (6.5) | 3 | (6.7) |
| Gynaecologic * | 1 | (2.2) | 2 | (4.4) |
| Lung | 1 | (2.2) | 0 | (0) |
| Other | 6 | (13.0) | 8 | (17.8) |
| No. of cancers | ||||
| 1 | 39 | (84.8) | 39 | (86.7) |
| >1 | 7 | (15.2) | 6 | (13.3) |
| Time since first cancer diagnosis, y | ||||
| <2 | 3 | (6.5) | 1 | (2.2) |
| 2–5 | 25 | (54.3) | 26 | (57.8) |
| 6–9 | 11 | (23.9) | 6 | (13.3) |
| ≥10 | 6 | (13.0) | 9 | (20.0) |
| Comorbidity | ||||
| Diabetes II | 4 | (8.7) | 3 | (6.5) |
| Arthritis | 5 | (10.9) | 5 | (11.1) |
| Hypertension | 12 | (26.1) | 13 | (28.3) |
| High cholesterol | 10 | (21.8) | 9 | (20.0) |
| Dimensions | Cronbach’s α |
|---|---|
| Physical functioning | 0.865 |
| Role physical | 0.926 |
| Bodily pain | 0.724 |
| General health | 0.809 |
| Vitality | 0.805 |
| Social functioning | 0.607 |
| Role emotional | 0.913 |
| Mental health | 0.827 |
| Health Indicators | Intervention | Control | ||||
|---|---|---|---|---|---|---|
| Baseline | Week 12 | Week 24 | Baseline | Week 12 | Week 24 | |
| Physical fitness (6MWT) (m) a** | 530.4 | 553.9 | 565.3 | 515.2 | 521.3 | 528.9 |
| (66.8) | (72.4) | (81.3) | (88.7) | (101.9) | (102.6) | |
| Systolic blood pressure [mmHg] a** | 138.7 | 134.4 | 131.83 | 139.7 | 133.2 | 130.6 |
| (15.5) | (16.8) | (15.5) | (20.2) | (19.3) | (16.6) | |
| Diastolic blood pressure [mmHg] a** | 81.3 | 77.2 | 75.7 | 79.8 | 79.4 | 76.5 |
| (9.6) | (9.4) | (10.2) | (10.8) | (12.1) | (11.0) | |
| Waist girth (cm) a** | 99.7 | 98.6 | 99.8 | 97.6 | 96.8 | 97.8 |
| (13.1) | (13.3) | (13.5) | (13.3) | (13.4) | (13.1) | |
| BMI (kg/m2) | 30.4 | 30.2 | 30.1 | 28.6 | 28.6 | 28.6 |
| (4.9) | (4.9) | (4.9) | (4.5) | (4.4) | (4.4) | |
| SF-36v2 Subscales | Intervention | Control | ||||
|---|---|---|---|---|---|---|
| Baseline | Week 12 | Week 24 | Baseline | Week 12 | Week 24 | |
| Physical functioning | 79.5 | 80.8 | 80.9 | 74.5 | 74.3 | 75.1 |
| (15.8) | (16.3) | (17.5) | (20.5) | (20.6) | (17.5) | |
| Role physical | 71.1 | 72.9 | 72.9 | 71.0 | 68.4 | 75.1 |
| (26.3) | (19.0) | (22.3) | (25.3) | (21.2) | (19.4) | |
| Bodily pain a** | 40.5 | 63.3 | 65.7 | 35.5 | 61.6 | 66.3 |
| (20.8) | (18.5) | (18.2) | (16.1) | (20.3) | (21.5) | |
| General health a** | 53.2 | 69.5 | 72.7 | 50.9 | 67.4 | 68.3 |
| (11.9) | (16.5) | (19.7) | (9.8) | (15.7) | (16.6) | |
| Vitality | 58.7 | 57.1 | 55.6 | 61.1 | 55.2 | 55.7 |
| (10.0) | (15.9) | (16.0) | (9.3) | (13.8) | (12.8) | |
| Social functioning a** | 71.5 | 83.4 | 83.9 | 62.3 | 76.3 | 81.4 |
| (15.4) | (20.5) | (16.9) | (16.3) | (23.8) | (21.1) | |
| Role emotional bc* | 86.8 | 86.3 | 81.2 | 74.2 | 74.5 | 78.5 |
| (18.5) | (16.7) | (22.9) | (22.0) | (21.4) | (18.1) | |
| Mental health a* | 68.5 | 70.8 | 67.8 | 64.3 | 71.7 | 70.3 |
| (8.1) | (13.1) | (15.6) | (6.1) | (11.6) | (14.1) | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Frensham, L.J.; Parfitt, G.; Dollman, J. Effect of a 12-Week Online Walking Intervention on Health and Quality of Life in Cancer Survivors: A Quasi-Randomized Controlled Trial. Int. J. Environ. Res. Public Health 2018, 15, 2081. https://doi.org/10.3390/ijerph15102081
Frensham LJ, Parfitt G, Dollman J. Effect of a 12-Week Online Walking Intervention on Health and Quality of Life in Cancer Survivors: A Quasi-Randomized Controlled Trial. International Journal of Environmental Research and Public Health. 2018; 15(10):2081. https://doi.org/10.3390/ijerph15102081
Chicago/Turabian StyleFrensham, Lauren J., Gaynor Parfitt, and James Dollman. 2018. "Effect of a 12-Week Online Walking Intervention on Health and Quality of Life in Cancer Survivors: A Quasi-Randomized Controlled Trial" International Journal of Environmental Research and Public Health 15, no. 10: 2081. https://doi.org/10.3390/ijerph15102081
APA StyleFrensham, L. J., Parfitt, G., & Dollman, J. (2018). Effect of a 12-Week Online Walking Intervention on Health and Quality of Life in Cancer Survivors: A Quasi-Randomized Controlled Trial. International Journal of Environmental Research and Public Health, 15(10), 2081. https://doi.org/10.3390/ijerph15102081






