Aflatoxin B1 Contamination in Chicken Livers and Gizzards from Industrial and Small Abattoirs, Measured by ELISA Technique in Maputo, Mozambique
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling
2.2. Morphological Evaluation of Liver Samples
2.3. Analytical Procedures
2.3.1. Extraction of Aflatoxin B1
2.3.2. Analysis of AflatoxinB1 in Samples
2.4. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Food Agricultural Organization. Poultry Sector Review: Mozambique. In FAO Animal Production and Health Livestock Country Reviews; Thieme, O., Ed.; Food Agricultural Organization: Rome, Italy, 2013; Volume 5, pp. 1–41. [Google Scholar]
- Lizárraga-Paulín, E.G.; Moreno-Martínez, E.; Miranda-Castro, S.P. Aflatoxins and their impact on human and animal health: An emerging problem. In Aflatoxins Biochemistry and Molecular Biology; Guevara-Gonzalez, R.G., Ed.; InTech Press: Rijeka, Croatia, 2011; pp. 255–282. [Google Scholar]
- Wu, F.; Narrod, C.; Tiongco, M.; Liu, Y. The Health Economics of Aflatoxin: Global Burden of Disease; Working Paper No.4; International Food Policy Research Institute: Washington, DC, USA, 2011; pp. 1–17. [Google Scholar]
- Darsanaki, R.K.; Alikhani, F.; Mohammadi, M.; Aliabadi, M.A. Biological Control of Aflatoxins. Eur. J. Exp. Biol. 2013, 3, 162–166. [Google Scholar]
- El-Desouky, T.A.; Mohamed, S.R.; Abou-Arab, A.A.K.; Salim, A.B. Occurrence of aflatoxin B1 and M1 in some Egyptian chicken organs and their affected by ozonated water. Open Sci. J. Mod. Phys. 2014, 1, 24–30. [Google Scholar]
- Freire, F.C.O.; Vieira, I.G.P.; Guedes, M.I.F.; Mendes, F.N.P. Micotoxinas: Importância na Alimentação e na Saúde Humana e Animal, 110th ed.; Embrapa Agroindústria Tropical: Fortaleza, Brazil, 2007; pp. 1–48. [Google Scholar]
- Iqbal, S.Z.; Nisar, S.; Asi, M.R.; Jinap, S. Natural incidence of aflatoxins, ochratoxin A and zearalenone in chicken meat and eggs. Food Control 2014, 43, 98–103. [Google Scholar] [CrossRef]
- Wild, C.P.; Gong, Y.Y. Mycotoxins and human disease: A largely ignored global health issue. Carcinogenesis 2010, 31, 71–82. [Google Scholar] [CrossRef] [PubMed]
- Wu, F. Global impacts of aflatoxin in maize: Trade and human health. World Mycotoxin J. 2015, 8, 137–142. [Google Scholar] [CrossRef]
- Bbosa, G.S.; Kitya, D.; Odda, J.; Ogwal-Okeng, J. Aflatoxins metabolism, effects on epigenetic mechanisms and their role in carcinogenesis. Health 2013, 5, 14–34. [Google Scholar] [CrossRef]
- IARC. Overall evaluations of carcinogenicity: An updating of IARC monographs. IARC Monogr. Eval. Carcinog. Risks Hum. 2012, 100, 51–72. [Google Scholar]
- Oliveira, C.A.F.; Germano, P.M.L. Aflatoxins in foodstuffs: Current concepts on mechanisms of toxicity and its involvement in the etiology of hepatocellular carcinoma. Rev. Saúde Públ. 1997, 31, 417–424. [Google Scholar] [CrossRef]
- Wu, F.; Khlangwiset, P. Health economic impacts and cost-effectiveness of aflatoxin reduction strategies in Africa: Case studies in biocontrol and postharvest interventions. Food Addit. Contam. A 2010, 27, 496–509. [Google Scholar] [CrossRef] [PubMed]
- Feddern, V.; Dors, G.C.; Tavernari, F.C.; Mazzuco, H.; Cunha, A., Jr.; Krabbe, E.L.; Scheuermann, G.N. Aflatoxins importance on animal nutrition. In Aflatoxins: Recent Advances and Future Prospects; InTech Press: Rijeka, Croatia, 2013; pp. 171–195. [Google Scholar]
- Newberne, P.M.; Butler, W.H. Acute and chronic effects of aflatoxin on the liver of domestic and laboratory animals: A review. Cancer Res. 1969, 29, 236–250. [Google Scholar] [PubMed]
- Dos Anjos, F.R.; Ledoux, D.R.; Rottinghaus, G.E.; Chimonyo, M. Efficacy of Mozambican bentonite and diatomaceous earth in reducing the toxic effects of aflatoxins in chicks. World Mycotoxin J. 2016, 9, 63–72. [Google Scholar] [CrossRef]
- Diaz, G.J.; Murcia, H.W. Biotransformation of aflatoxin B1 and its relationship with the differential toxicological response to aflatoxin in commercial poultry species. In Aflatoxins Biochemistry and Molecular Biology; Guevara-Gonzalez, R.G., Ed.; InTech Press: Rijeka, Croatia, 2011; pp. 3–20. [Google Scholar]
- Dhanasekaran, D.; Shanmugapriya, S.; Thajuddin, N.; Panneerselvam, A. Aflatoxins and Aflatoxicosis in Human and Animals. In Aflatoxins Biochemistry and Molecular Biology; Guevara-Gonzalez, R.G., Ed.; InTech Press: Rijeka, Croatia, 2011; pp. 221–254. [Google Scholar]
- Herzallah, S.; Al-Ameiri, N.; Al-Dmoor, H.; Masoud, S.; Shawabkeh, K. Meat and organs quality of broiler chickens fed diet contaminated with B1 aflatoxin. Glob. Vet. 2014, 12, 376–380. [Google Scholar] [CrossRef]
- Yunus, A.W.; Razzazi-Fazeli, E.; Bohm, J. Aflatoxin B1 in affecting broiler’s performance, immunity, and gastrointestinal tract: A review of history and contemporary issues. Toxins 2011, 3, 566–590. [Google Scholar] [CrossRef] [PubMed]
- Mabee, M.S.; Chipley, J.R. Tissue distribution and metabolism of aflatoxin B1-14C in broiler chickens. Appl. Microbiol. 1973, 25, 763–769. [Google Scholar] [PubMed]
- Cardwell, K.F. Mycotoxin contamination of foods in Africa: Anti-nutritional factors. Food Nutr. Bull. 2001, 21, 488–492. [Google Scholar] [CrossRef]
- Turner, P.C.; Collinson, A.C.; Cheung, Y.B.; Gong, Y.Y.; Hall, A.J.; Prentice, A.M.; Wild, C.P. Aflatoxin exposure in utero causes growth faltering in Gambian infants. Int. J. Epidemiol. 2007, 36, 1119–1125. [Google Scholar] [CrossRef] [PubMed]
- Atici, C. Food Safety Regulations and Export Responses of Developing Countries: The Case of Turkey’s Fig and Hazelnut Exports; Research Working Paper No. 39; Food and Agriculture Organization of the United Nations: Rome, Italy, 2013; pp. 1–14. [Google Scholar]
- Rastogi, S.; Dwivedi, P.D.; Khanna, S.K.; Das, M. Detection of Aflatoxin M1 contamination in milk and infant milk products from Indian markets by ELISA. Food Control 2004, 15, 287–290. [Google Scholar] [CrossRef]
- Zaghini, A.G.; Martelli, G.; Roncada, P.; Simoli, M.; Rizzi, L. Mamanoligosaccharides and aflatoxin B1 and M1 residues in eggs and aflatoxin B1 levels in liver. Poult. Sci. 2005, 84, 825–832. [Google Scholar] [CrossRef] [PubMed]
- Herzallah, S.M. Determination of aflatoxins in eggs, milk, meat and meat products using HPLC fluorescent and UV detectors. Food Chem. 2009, 114, 1141–1146. [Google Scholar] [CrossRef]
- Markov, K.; Pleadin, J.; Bevardi, M.; Vahcic, N.; Sokolic-Mihalak, D.; Frece, J. Natural occurrence of aflatoxin B1, ochratoxin A and citrinin in Croatian fermented meat products. Food Control 2013, 34, 312–317. [Google Scholar] [CrossRef]
- Saeed, A.; Afzal, S.; Hussain, M.W.; Bokhari, S.Y.A.; Shahzad, M.S.; Qayyum, A.; Raza, M.H. Effect of aflatoxin B1 on different body tissues of Gallus domesticus. J. Anim. Vet. Adv. 2003, 2, 76–78. [Google Scholar]
- Bintvihok, A.; Davitiyananda, D. Aflatoxins and their metabolites residues in chicken tissues from 5 parts (10 provinces) of Thailand. Thail. J. Health Res. 2002, 16, 37–50. [Google Scholar]
- Pourelmi, M.R.; Palizdar, M.H.; Shirali, S.; Barami, A.R. Aflatoxin B1 contamination in local and industrial eggs measured by ELISA technique in Mazandaran. Eur. J. Zool. Res. 2013, 2, 89–92. [Google Scholar]
- Mondlane, I.A.P.; Capece, B.P.S.; Parruque, A.F. Relação Entre a Ocorrência de Fungos e a Presença de Aflatoxinas B1 em Rações para Aves Fabricadas em Maputo; Boletim do Instituto de Investigação Agrária de Moçambique (IIAM) No. 3; Instituto de Investigação Agrária de Moçambique: Maputo, Mozambique, 2005; pp. 1–12. [Google Scholar]
- Warth, B.; Parich, A.; Atehnkeng, J.; Bandyopadhyay, R.; Schuhmacher, R.; Sulyok, M.; Krska, R. Quantitation of mycotoxins in food and feed from Burkina Faso and Mozambique using a modern LC-MS/MS multitoxin method. J. Agric. Food Chem. 2012, 60, 9352–9363. [Google Scholar] [CrossRef] [PubMed]
- Augusto, J.; Atehnkeng, J.; Akello, J.; Cotty, P.; Bandyopadhyay, R. Prevalence and distribution of Aspergillus section Flavi in maize and groundnut fields and aflatoxin contamination in Mozambique. In Proceedings of the APS-CPS Joint Meeting in Minneapolis, Phytopathology, Minneapolis, MN, USA, 9–13 August 2014; Volume 104. [Google Scholar] [CrossRef]
- Van Egmond, H.P.; Jonker, M.A. Worldwide regulations on aflatoxins: The situation in 2002. J. Toxicol. Toxin Rev. 2004, 23, 273–293. [Google Scholar] [CrossRef]
- Van Egmond, H.P.; Schothorst, R.C.; Jonker, M.A. Regulations relating to mycotoxins in food: Perspectives in a global and European context. Anal. Bioanal. Chem. 2007, 389, 147–157. [Google Scholar] [CrossRef] [PubMed]
- USDA. Giblets and Food Safety; Food Safety and Inspection Service, United States Department of Agriculture: Philadelphia, PA, USA, 2008; pp. 1–2.
- Andreasson, U.; Perret-Liaudet, A.; van Doorn, L.J.C.; Blennow, K.; Chiasserini, D.; Engelborghs, S.; Fladby, T.; Genc, S.; Kruse, N.; Kuiperij, H.B.; et al. A practical guide to immunoassay method validation. Front. Neurol. 2015, 6, 1–8. [Google Scholar] [CrossRef]
- Ravindran, V. Animal feed safety. In Poultry Feed Availability and Nutrition in Developing Countries; Poultry Development Review; Food Agricultural Organization: Rome, Italy, 2013; pp. 1–3. [Google Scholar]
- Salle, C.T.P.; Lorenzini, G.; Sfoggia, M.; Cé, M.C.; Guahyba, A.S.; Moraes, H.L.S.; Nascimento, V.P.; Salle, F.O. The presence of aflatoxins in field broiler livers. Arquit. Facul. Vet. UFRGS 2001, 29, 101–106. [Google Scholar] [CrossRef]
- Nicolau, Q.C.; Borges, A.C.G.; Souza, J.G. Cutting poultry production chain from Mozambique: Characterization and competitiveness. Revista de Ciências Agrárias 2011, 1, 182–198. [Google Scholar]
- Rodríguez-Amaya, D.B.; Sabino, M. Mycotoxin research in Brazil: The last decade in review. Braz. J. Microbiol. 2002, 33, 1–11. [Google Scholar] [CrossRef]
- Stamford, T.L.M.; Vilar, E.A.; Bastos, S.T.G.; Silva, C.G.M. Pesquisa micotoxicológica de produtos avícolas “in natura” e processados. Bol. CEPPA Curitiba 2005, 23, 135–160. [Google Scholar] [CrossRef]
- Li, P.; Zhang, Q.; Zhang, D.; Guan, G.; Xiaoxia; Liu, D.X.; Fang, S.; Wang, X.; Zhang, W. Aflatoxin Measurement and Analysis. In Aflatoxins-Detection, Measurement and Control; Torres-Pacheco, I., Ed.; InTech Press: Rijeka, Croatia, 2011; pp. 183–208. [Google Scholar]
- Zheng, M.Z.; Richard, J.L.; Binder, J. A review of rapid methods for the analysis of mycotoxins. Mycopathologia 2006, 161, 261–273. [Google Scholar] [CrossRef] [PubMed]
- Bahobail, A.A.S.; Hassan, S.A.; El-Deeb, B.A. Microbial quality and content aflatoxins of commercially available eggs in Taif, Saudi Arabia. Afr. J. Microbiol. Res. 2012, 6, 3337–3342. [Google Scholar] [CrossRef]
- Kumar, R.; Balachandran, C. Histopathological changes in broiler chickens fed aflatoxin and cyclopiazonic acid. Vet. Arch. 2009, 79, 31–40. [Google Scholar]
- Ito, T.; Kobayashi, Y.; Morita, T.; Horimoto, T.; Kawaoka, Y. Virulent influenza A viruses induce apoptosis in chickens. Virus Res. 2002, 84, 27–35. [Google Scholar] [CrossRef]
- Vilar, E.A.; Oliveira, M.C.M.; Stamford, T.L.M. Pesquisa micotoxicológica em fígado de aves produzidas e comercializadas em Pernambuco. Bol. CEPPA Curitiba 2002, 20, 335–346. [Google Scholar] [CrossRef]
- Bryden, W.L.; Cumming, R.B. Observations on the liver of the chicken following aflatoxin B1 ingestion. Avian Pathol. 1980, 9, 551–556. [Google Scholar] [CrossRef] [PubMed]
- Trampel, D.W.; Sell, J.L.; Ahn, D.U.; Sebranek, J.G. Preharvest feed withdrawal affects liver lipid and liver color in broiler chickens. Poult. Sci. 2005, 84, 137–142. [Google Scholar] [CrossRef] [PubMed]
- Aengwanich, W.; Simaraks, S. Pathology of heart, lung, liver and kidney in broilers under chronic heat stress. SongklanakarinJ. Sci. Technol. 2004, 26, 417–424. [Google Scholar]
- Hussain, Z.; Khan, M.Z.; Khan, A.; Javed, I.; Saleemi, M.K.; Mahmood, S.; Asi, M.R. Residues of aflatoxin B1 in broiler meat: Effect of age and dietary aflatoxin B1 levels. Food Chem. Toxicol. 2010, 48, 3304–3307. [Google Scholar] [CrossRef] [PubMed]
- Sawhney, D.S.; Vadehra, D.V.; Baker, R.C. The metabolism of 14C aflatoxins in laying hens. Poult. Sci. 1973, 52, 1302–1309. [Google Scholar] [CrossRef] [PubMed]
- Wolzak, A.; Pearson, A.M.; Coleman, T.H. Aflatoxin carryover and clearance from tissues of laying hens. Food Chem. Toxicol. 1986, 24, 37–41. [Google Scholar] [CrossRef]
- Hassan, Z.U.; Khan, M.Z.; Khan, A.; Javed, I.; Hussain, Z. Effects of individual and combined administration of ochratoxin A and aflatoxin B1 in tissues and eggs of White Leghorn breeder hens. J. Sci. Food Agric. 2012, 92, 1540–1544. [Google Scholar] [CrossRef] [PubMed]
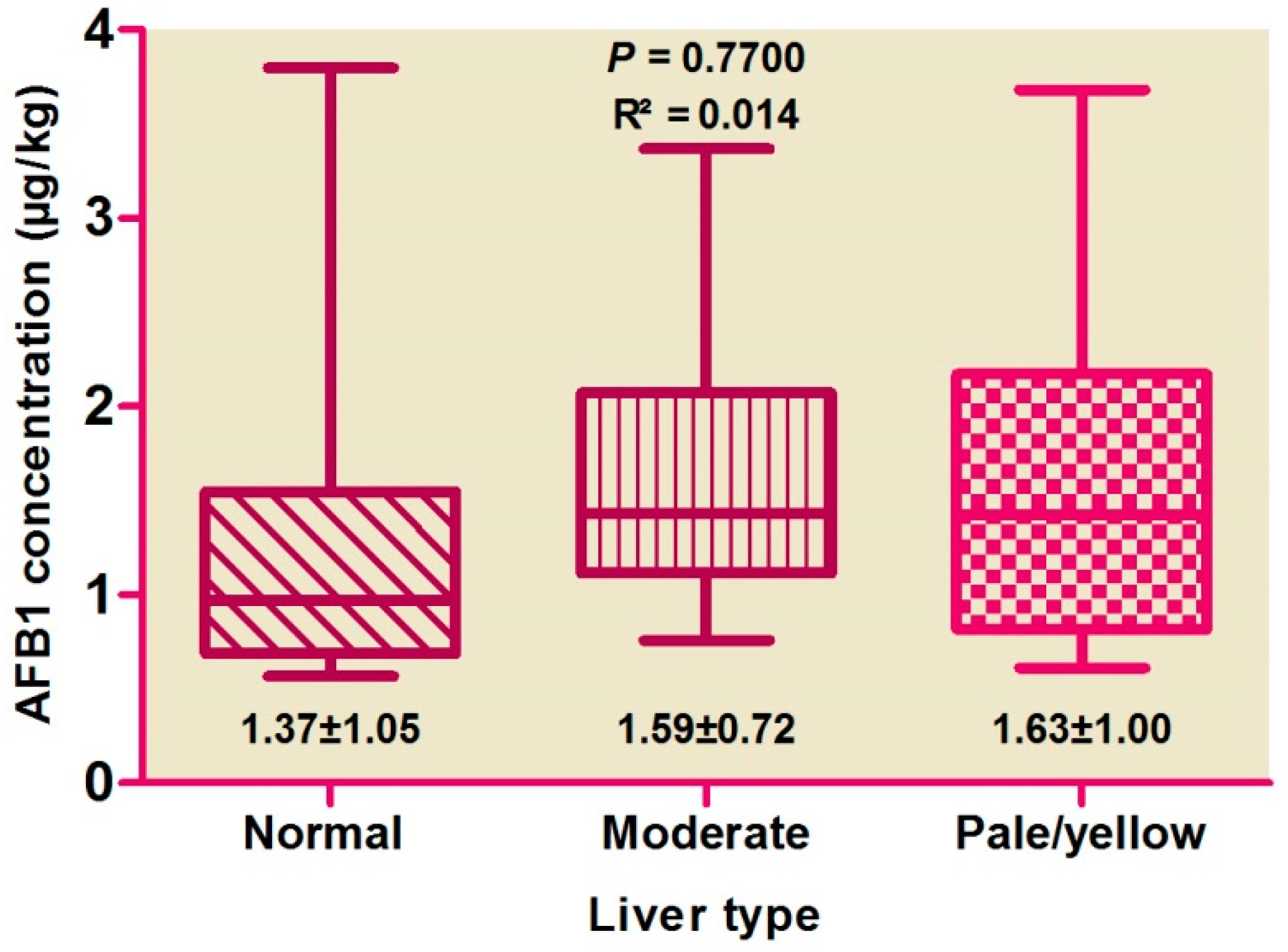
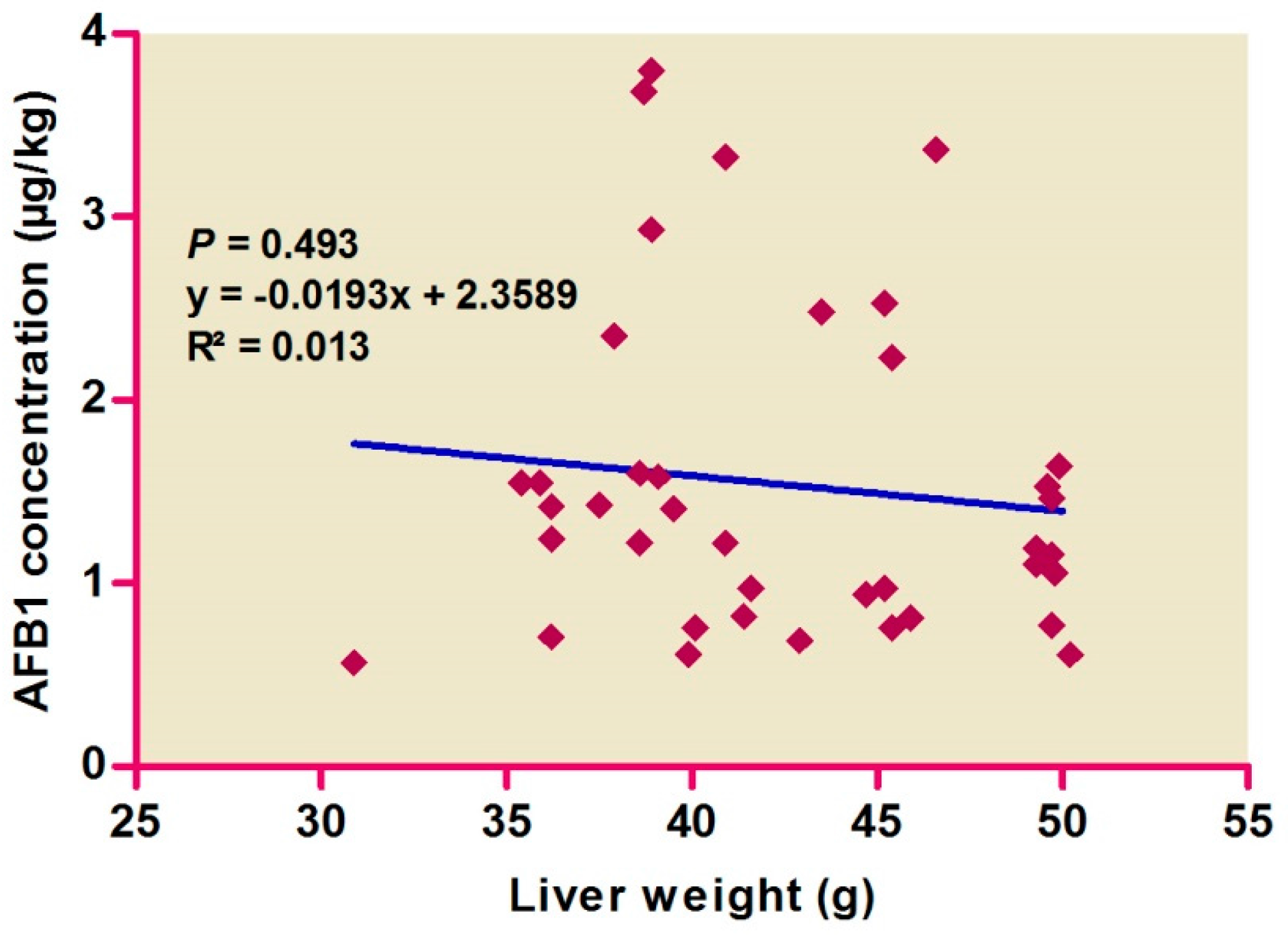
| Liver Type | IS Livers | LS Livers | ||||||
|---|---|---|---|---|---|---|---|---|
| Samples | Weight (g) | Samples | Weight (g) | |||||
| Range | Mean ± SD | 95% CI | Range | Mean ± SD | 95% CI | |||
| Normal | 35 (50.0) | 23.80–46.80 | 35.47 ± 0.79 a | 33.85–37.10 | 12 (40.0) | 30.20–45.60 | 39.24 ± 1.40 a | 36.16–42.32 |
| Moderate | 28 (40.0) | 30.20–45.40 | 39.28 ± 0.78 a | 37.66–40.89 | 12 (40.0) | 40.80–49.70 | 46.03 ± 0.90 a | 44.03–48.02 |
| Pale/yellow | 7 (10.0) | 37.90–50.20 | 45.59 ± 2.06 a | 40.54–50.63 | 6 (20.0) | 36.20–49.70 | 41.67 ± 2.23 a | 35.93–47.31 |
| Total | 70 (70.0) | 23.80–50.20 | 38.00 ± 5.48 b | 36.70–39.30 | 30 (30.0) | 30.20–49.70 | 42.40 ± 5.24 b | 40.50–44.40 |
| AFB1 Spiked Level (µg/kg) | AFB1 Recovery * (µg/kg) | R (%) | |||||
|---|---|---|---|---|---|---|---|
| Assay 1 | Assay 2 | Assay 3 | Assay 4 | Assay 5 | Mean ± SD | ||
| 5.0 | 4.67 | 4.64 | 4.34 | 4.54 | 4.49 | 4.54 ± 0.13 | 90.20 |
| 10.0 | 9.23 | 9.54 | 9.17 | 9.34 | 9.29 | 9.31 ± 0.14 | 93.14 |
| Sample Type | Samples | AFB1 Content (µg/kg) | Samples with Level | |||
|---|---|---|---|---|---|---|
| Analyzed | Positive | Range | Mean ± SD | <10 * µg/kg | >10 µg/kg | |
| IS livers | 70 | 19 (27.1) a | 0.61–2.48 | 1.35 ± 0.58 b | 70 | 0 |
| LS livers | 30 | 20 (66.7) a | 0.57–3.80 | 1.73 ± 1.09 b | 30 | 0 |
| Total | 100 | 39 (39.0) | 0.57–3.80 | 1.54 ± 0.89 b | 100 | 0 |
| IS gizzards | 50 | 2 (4.0) a | 0.81–1.34 | 1.07 ± 0.37 b | 50 | 0 |
| LS gizzards | 30 | 9 (30.0) a | 0.68–2.12 | 1.04 ± 0.44 b | 30 | 0 |
| Total | 80 | 11 (13.8) | 0.68–2.12 | 1.06 ± 0.42 b | 80 | 0 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sineque, A.R.; Macuamule, C.L.; Dos Anjos, F.R. Aflatoxin B1 Contamination in Chicken Livers and Gizzards from Industrial and Small Abattoirs, Measured by ELISA Technique in Maputo, Mozambique. Int. J. Environ. Res. Public Health 2017, 14, 951. https://doi.org/10.3390/ijerph14090951
Sineque AR, Macuamule CL, Dos Anjos FR. Aflatoxin B1 Contamination in Chicken Livers and Gizzards from Industrial and Small Abattoirs, Measured by ELISA Technique in Maputo, Mozambique. International Journal of Environmental Research and Public Health. 2017; 14(9):951. https://doi.org/10.3390/ijerph14090951
Chicago/Turabian StyleSineque, Alberto Romão, Custódia Lina Macuamule, and Filomena Rosa Dos Anjos. 2017. "Aflatoxin B1 Contamination in Chicken Livers and Gizzards from Industrial and Small Abattoirs, Measured by ELISA Technique in Maputo, Mozambique" International Journal of Environmental Research and Public Health 14, no. 9: 951. https://doi.org/10.3390/ijerph14090951
APA StyleSineque, A. R., Macuamule, C. L., & Dos Anjos, F. R. (2017). Aflatoxin B1 Contamination in Chicken Livers and Gizzards from Industrial and Small Abattoirs, Measured by ELISA Technique in Maputo, Mozambique. International Journal of Environmental Research and Public Health, 14(9), 951. https://doi.org/10.3390/ijerph14090951




