Nature, Nurture and Evolution of Intra-Species Variation in Mosquito Arbovirus Transmission Competence
Abstract
:1. Introduction
2. The Vector Competence Phenotype
3. Determinants of Vector Competence
4. Mosquito Genetic Factors Influence Mosquito Competence for Arboviruses
5. Environmental Factors Influence Mosquito Competence for Arboviruses
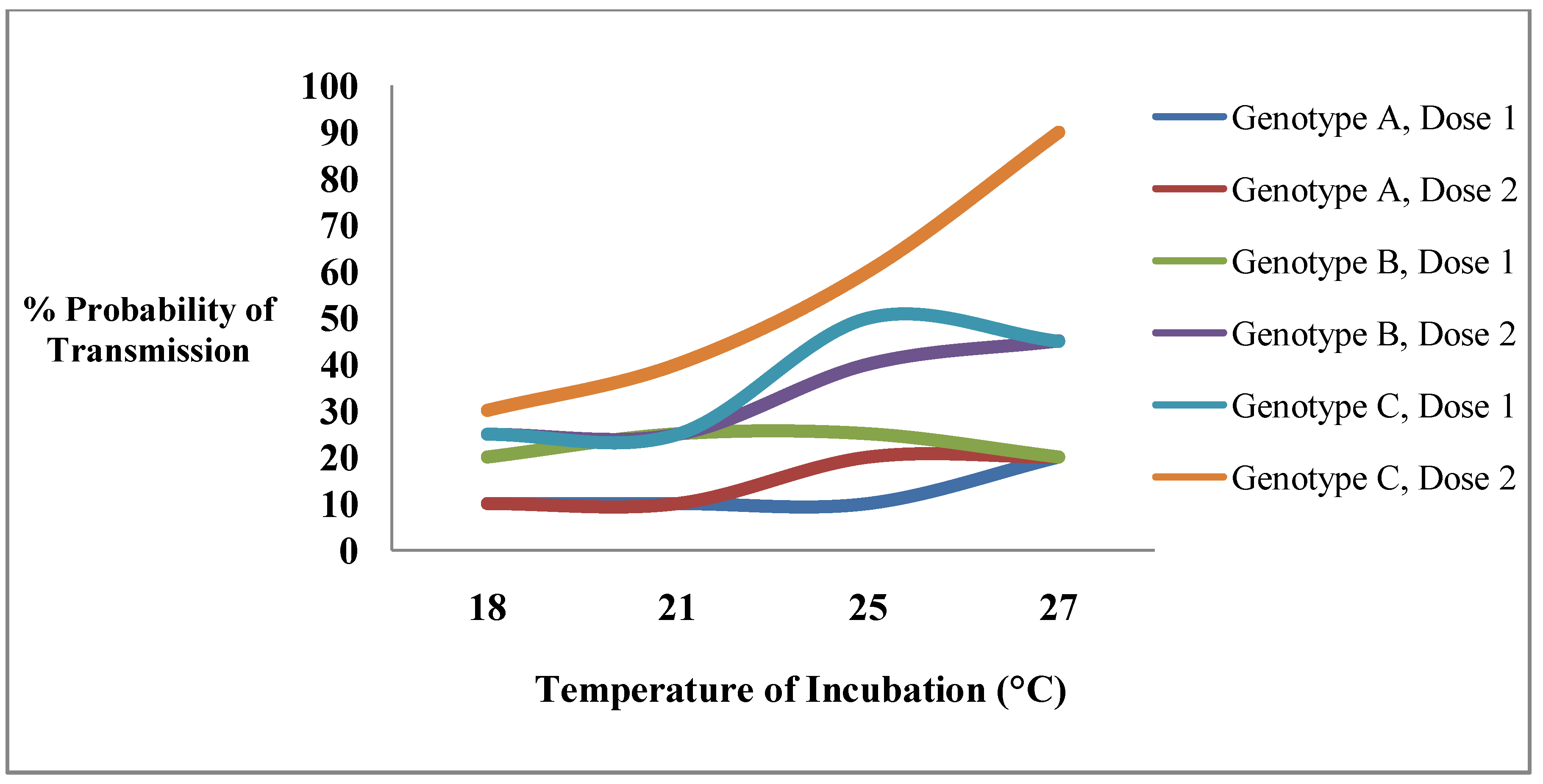
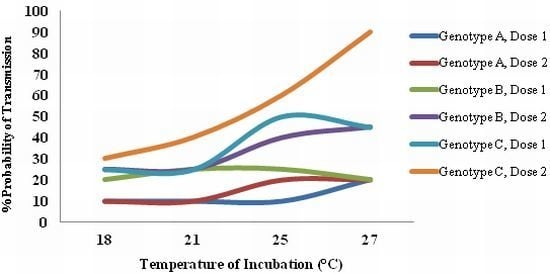
6. The Importance of Vector Competence Variation in Arbovirus Epidemiology
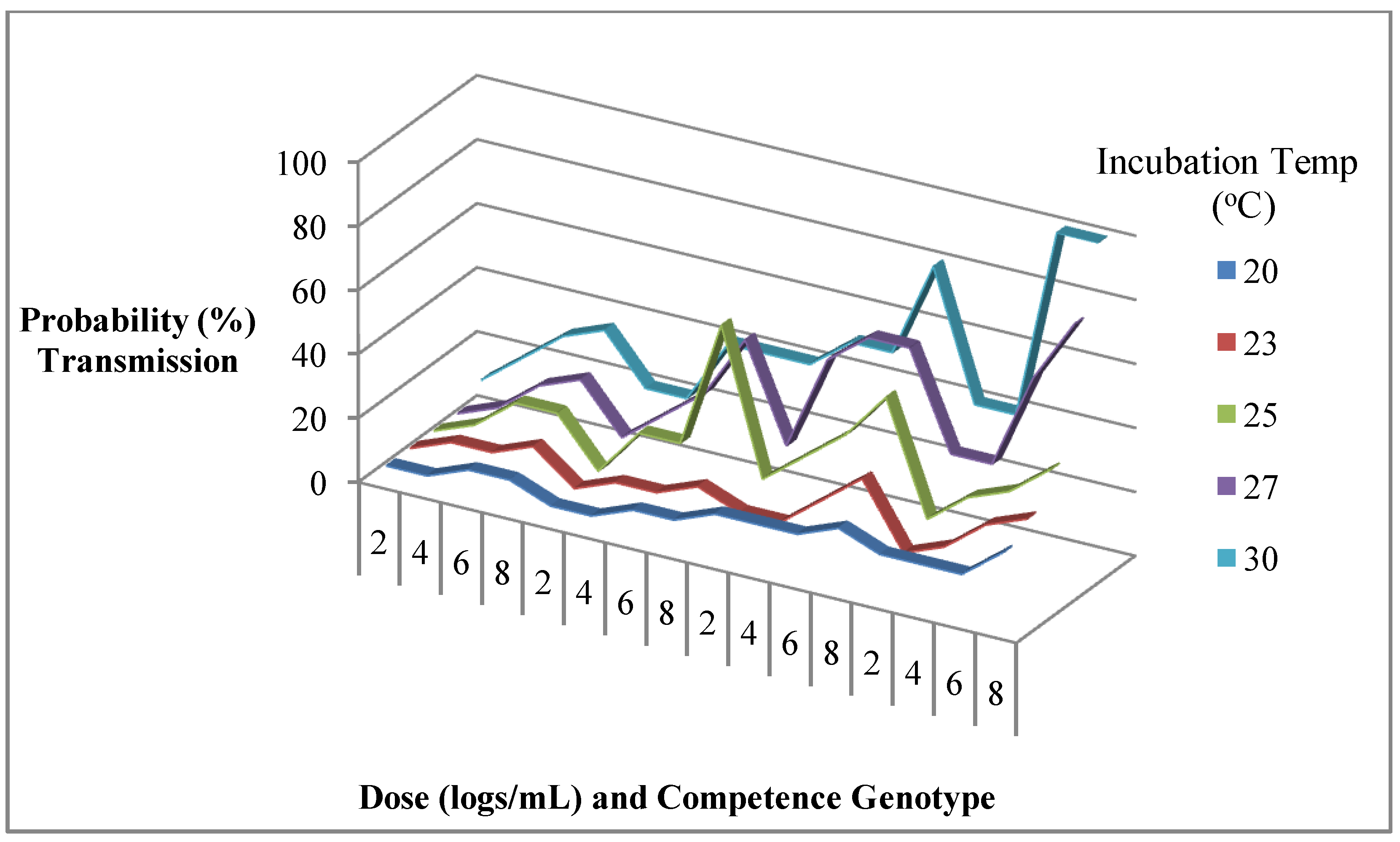
7. Challenges in Characterizing Causes of Variation in Mosquito Competence for Arboviruses
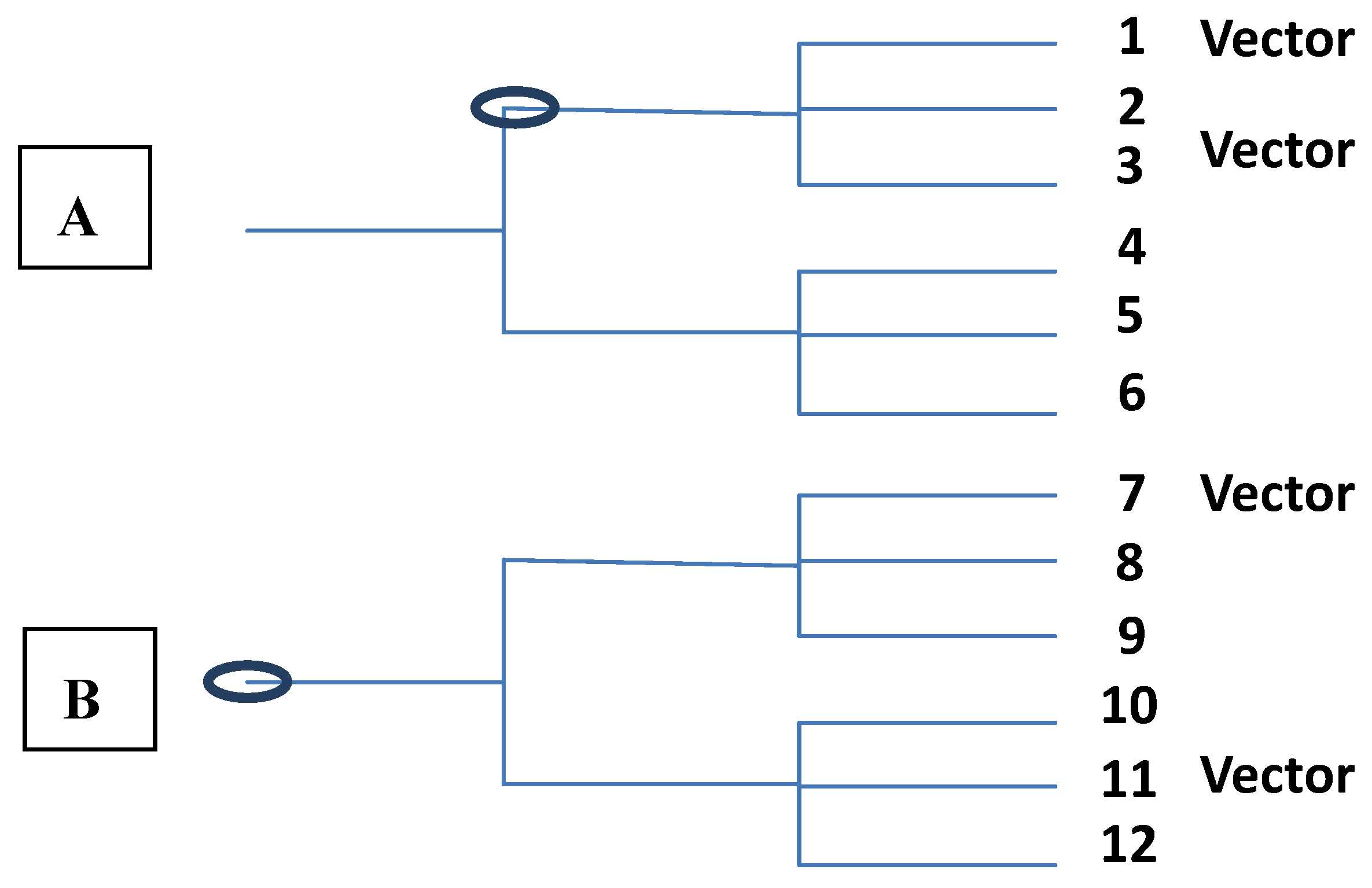
8. Conclusions
Acknowledgments
Conflict of Interest
References
- Hardy, J.L.; Houk, E.J.; Kramer, L.D.; Reeves, W.C. Intrinsic factors affecting vector competence of mosquitoes for arboviruses. Annu. Rev. Entomol. 1983, 28, 229–262. [Google Scholar] [CrossRef]
- Hardy, J.L. Susceptibility and resistance of vector mosquitoes. In The Arboviruses: Epidemiology and Ecology; Monath, T.P., Ed.; CRC Press Inc.: Boca Raton, FL, USA, 1988; Volume 1, pp. 87–126. [Google Scholar]
- Rodhain, F. Functioning of virus-vector systems. Ann. Soc. Belg. Med. Trop. 1991, 71, 189–199. [Google Scholar]
- Beerntsen, B.T.; James, A.A.; Christensen, B.M. Genetics of mosquito vector competence. Microbiol. Mol. Biol. Rev. 2000, 64, 115–137. [Google Scholar] [CrossRef]
- Mellor, P.S. Replication of arboviruses in insect vectors. J. Comp. Pathol. 2000, 123, 231–247. [Google Scholar] [CrossRef]
- Kramer, L.D.; Ebel, G.D. Dynamics of flavivirus infection in mosquitoes. Adv. Virus Res. 2003, 60, 187–232. [Google Scholar] [CrossRef]
- Kuno, G.; Chang, G.-J. Biological transmission and arboviruses: Reexamination of and new insight into components, mechanisms, and unique traits as well as their evolutionary trends. Clin. Microbiol. Rev. 2005, 18, 608–637. [Google Scholar] [CrossRef]
- Black, W.C., IV; Severson, D.W. Genetics of vector competence. In Biology of Disease Vectors, 2nd; Marquardt, W.C., Ed.; Elsevier Academic Press: Amsterdam, NL, USA, 2005; pp. 415–448. [Google Scholar]
- Barillas-Mury, C.; Kumar, S. Plasmodium-mosquito interactions: A tale of dangerous liaisons. Cell Microbiol. 2005, 7, 1539–1545. [Google Scholar] [CrossRef]
- Osta, M.A.; Christophides, G.K.; Kafatos, F.C. Effects of mosquito genes on Plasmodium development. Science 2004, 303, 2030–2032. [Google Scholar]
- Koutsos, A.C.; Blass, C.; Meister, S.; Schmidt, S.; MacCallum, R.M.; Soares, M.B.; Collins, F.H.; Benes, V.; Zdobnov, E.; Kafatos, F.C.; Christophides, G.K. Life cycle transcriptome of the malaria mosquito Anopheles gambiae and comparison with the fruitfly Drosophila melanogaster. Proc. Natl. Acad. Sci. USA 2007, 104, 11304–11309. [Google Scholar]
- Riehle, M.M.; Markianos, K.; Niare, O.; Xu, J.; Li, J.; Toure, A.M.; Podiougou, B.; Oduol, F.; Diawara, S.; Diallo, M.; et al. Natural malaria infection in Anopheles gambiae is regulated by a single genomic control region. Science 2006, 312, 577–579. [Google Scholar]
- Harris, C.; Lambrechts, L.; Rousset, F.; Abate, L.; Nsango, S.E.; Fontenille, D.; Morlais, I.; Cohuet, A. Polymorphisms in Anopheles gambiae immune genes associated with natural resistance with natural Plasmodium falciparum infection. PLoS ONE 2010. [Google Scholar] [CrossRef]
- Horton, A.A.; Lee, Y.; Coulibaly, C.A.; Rashbrook, V.K.; Cornel, A.J.; Lanzaro, G.C.; Luckhart, S. Identification of three single nucleotide polymorphisms in Anopheles gambiae immune signaling genes that are associated with Plasmodium falciparum infection. Malaria J. 2010. [Google Scholar] [CrossRef]
- Rottshaefer, S.M.; Riehle, M.M.; Coulibaly, B.; Sacko, M.; Niare, O.; Marlais, I.; Traore, S.F.; Vernick, K.D.; Lazzaro, B.P. Exceptional diversity, maintenance of polymorphism, and recent directional selection on the APL1 malaria resistance genes of Anopheles gambiae. PLoS Biol. 2011. [Google Scholar] [CrossRef]
- Mitri, C.; Vernick, K.D. Anopheles gambiae pathogen susceptibility: The intersection of genetic, immunity and ecology. Curr. Opin. Microbiol. 2012, 15, 285–291. [Google Scholar]
- Gubler, D.J.; Rosen, L. Variation among geographic strains of Aedes albopictus in susceptibility to infection with dengue viruses. Am. J. Trop. Med. Hyg. 1976, 25, 318–325. [Google Scholar]
- Tesh, R.B.; Gubler, D.J.; Rosen, L. Variation among geographic strains of Aedes albopictus in susceptibility to infection with chikungunya virus. Am. J. Trop. Med. Hyg. 1976, 25, 326–335. [Google Scholar]
- Grimstad, P.R.; Craig, G.B., Jr.; Ross, Q.E.; Yuill, T.M. Aedes triseriatus and La Crosse virus: Geographic variation in vector susceptibility and ability to transmit. Am. J. Trop. Med. Hyg. 1977, 26, 990–996. [Google Scholar]
- Takahashi, M. Variation in susceptibility among colony strains of Culex tritaeniorhynchus to Japanese encephalitis virus infection. Jpn.J. Med. Sci. Biol. 1980, 33, 321–329. [Google Scholar]
- Mitchell, C.J.; Gubler, D.J.; Monath, T.P. Variation in infectivity of St. Louis encephalitis virus strains for Culex pipiens quinquefasciatus (Diptera: Culicidae). J. Med. Entomol. 1983, 20, 526–533. [Google Scholar]
- Kay, B.H.; Fanning, I.D.; Carley, J.G. The vector competence of Australian Culex annulirostris with Murray Valley encephalitis and Kunjin viruses. Aust. J. Exp. Biol. Med. Sci. 1984, 62, 641–650. [Google Scholar] [CrossRef]
- Tabachnick, W.J.; Wallis, G.P.; Aitken, T.H.; Miller, B.R.; Amato, G.D.; Lorenz, L.; Powell, J.R.; Beaty, B.R. Oral infection of Aedes aegypti with yellow fever virus: Geographic variation and genetic considerations. Am. J. Trop. Med. Hyg. 1985, 34, 1219–1224. [Google Scholar]
- Boromisa, R.D.; Rai, K.S.; Grimstad, P.R. Variation in the vector competence of geographic strains of Aedes albopictus for dengue 1 virus. J. Am. Mosq. Contr. Assoc. 1987, 3, 378–386. [Google Scholar]
- Hardy, J.L.; Reeves, W.C. Experimental studies on infection in vectors. In Epidemiology and Control of Mosquito-Borne Arboviruses in California; Reeves, W.C., Ed.; California Mosquito and Vector Control Association: Sacramento, CA, USA, 1990; pp. 145–253. [Google Scholar]
- Tardieux, I.; Poupel, O.; Lapchin, L.; Rodhain, F. Variation among strains of Aedes aegypti in susceptibility to oral infection with dengue type 2. Am. J. Trop. Med. Hyg. 1990, 43, 308–313. [Google Scholar]
- Failloux, A.B.; Raymond, R.M.; Ung, A.; Glaziou, P.; Martin, P.M.; Pasteur, N. Variation in the vector competence of Aedes polynesiensis for Wuchereria bancrofti. Parasitology 1995, 111, 19–29. [Google Scholar] [CrossRef]
- Vazeille-Falcoz, M.; Mousson, L.; Rodhain, F.; Chungue, E.; Failloux, A.B. Variation in oral susceptibility to dengue type 2 virus of populations of Aedes aegypti from the islands of Tahiti and Moorea, French Polynesia. Am. J. Trop. Med. Hyg. 1999, 60, 292–299. [Google Scholar]
- Bennett, K.E.; Olson, K.E.; Muñoz Mde, L.; Fernandez-Salas, I.; Farfan-Ale, J.A.; Higgs, S.; Black, W.C., IV; Beaty, B.J. Variation in vector competence for dengue 2 virus among 24 collections of Aedes aegypti from Mexico and the United States. Am. J. Trop. Med. Hyg. 2002, 67, 85–92. [Google Scholar]
- Johnson, B.W.; Chambers, T.V.; Crabtree, M.B.; Filippis, A.M.B.; Vilarinhos, P.T.R.; Resende, M.C.; Macoris, M.L.G.; Miller, B.R. Vector competence of Brazilian Aedes aegypti and Ae. albopictus for a Brazilian yellow fever virus isolate. Trans. Roy. Soc. Trop. Med. Hyg. 2002, 96, 611–613. [Google Scholar] [CrossRef]
- Paupy, C.N.; Girod, R.; Salvan, M.; Rodhain, F.; Failloux, A.B. Population structure of Aedes albopictus from La Reunion Island (Indian Ocean) with respect to susceptibility to a dengue virus. Heredity 2001, 87, 273–283. [Google Scholar] [CrossRef]
- Paupy, C.; Chantha, N.; Vazeille, M.; Reynes, J.M.; Rodhain, F.; A.B. Failloux, A.B. Variation over space and time of Aedes aegypti in Phnom Penh (Cambodia): Genetic structure and oral susceptibility to a dengue virus. Genet. Res. 2003, 82, 171–182. [Google Scholar] [CrossRef]
- Huber, K.; Le Loan, L.; Hoang, T.H.; Tien, T.K.; Rodhain, F.; Failloux, A.B. Aedes aegypti in south Vietnam: Ecology, genetic structure, vectorial competence and resistance to insecticides. Southeast Asian J. Trop. Med. Public Health 2003, 34, 81–86. [Google Scholar]
- Knox, T.B.; Kay, B.H.; Hall, R.A.; Ryan, P.A. Enhanced vector competence of Aedes aegypti (Diptera: Culicidae) from the Torres Strait compared with mainland Australia for dengue 2 and 4 viruses. J. Med. Entomol. 2003, 40, 950–956. [Google Scholar] [CrossRef]
- Vaidyanathan, R.; Scott, T.W. Geographic variation in vector competence for West Nile virus in the Culex pipiens (Diptera: Culicidae) complex in California. Vector-Borne Zoonotic Dis. 2007, 7, 193–198. [Google Scholar] [CrossRef]
- Diallo, M.; Ba, Y.; Faye, O.; Soumare, M.L.; Dia, I.; Sall, A.A. Vector competence of Aedes aegypti populations from Senegal for sylvatic and epidemic dengue 2 virus isolated in West Africa. Trans. Roy. Soc. Trop. Med. Hyg. 2008, 102, 493–498. [Google Scholar] [CrossRef]
- Van den Hurk, A.F.; Hall-Mendelin, S.; Pyke, A.T.; Smith, G.A.; Mackenzie, J.S. Vector competence of Australian mosquitoes for chikungunya virus. Vector-Borne Zoonotic Dis. 2010, 10, 489–495. [Google Scholar]
- Reisen, W.K.; Meyer, R.P.; Presser, S.B.; Hardy, J.L. Effect of temperature on the transmission of western equine encephalomyelitis and St. Louis encephalitis viruses by Culex tarsalis (Diptera: Culicidae). J. Med. Entomol. 1993, 30, 151–160. [Google Scholar]
- Reisen, W.K.; Hardy, J.L.; Presser, S.B.; Chiles, R.E. Seasonal variation in the vector competence of Culex tarsalis (Diptera: Culicidae) from the Coachella Valley of California for western equine encephalomyelitis and St. Louis encephalitis viruses. J. Med. Entomol. 1996, 33, 433–437. [Google Scholar]
- Dohm, D.J.; O’Guinn, M.L.; Turell, M.J. Effect of environmental temperature on the ability of Culex pipiens (Diptera: Culicidae) to transmit West Nile virus. J. Med. Entomol. 2002, 39, 221–225. [Google Scholar] [CrossRef]
- Mahmood, F.; Chiles, R.E.; Fang, Y.; Green, E.N.; Reisen, W.K. Effects of time after infection, mosquito genotype, and infectious viral dose on the dynamics of Culex tarsalis vector competence for western equine encephalomyelitis virus. J. Am. Mosq. Contr. Assoc. 2006, 22, 272–281. [Google Scholar] [CrossRef]
- Alto, B.W.; Lounibos, L.P.; Mores, C.N.; Reiskind, M.H. Larval competition alters susceptibility of adult Aedes mosquitoes to dengue infection. Proc. Biol. Sci. 2008, 275, 463–471. [Google Scholar] [CrossRef]
- Richards, S.L.; Anderson, S.L.; Lord, C.C.; Smartt, C.T.; Tabachnick, W.J. Relationships between infection, dissemination, and transmission of West Nile virus RNA in Culex pipiens quinquefasciatus (Diptera: Culicidae). J. Med. Entomol. 2012, 49, 132–142. [Google Scholar] [CrossRef]
- Tabachnick, W.J. Genetics of insect vector competence for arboviruses. Adv. Dis. Vector Res. 1994, 10, 93–108. [Google Scholar] [CrossRef]
- Tabachnick, W.J.; Black, W.C., IV. Making a case for molecular population genetic studies of arthropod vectors. Parasitol. Today 1994, 11, 27–30. [Google Scholar] [CrossRef]
- Tabachnick, W.J.; Black, W.C., IV. Population genetics in vector biology. In The Biology of Disease Vectors, 1st; Beaty, B.J., Marquardt, W.C., Eds.; University Press of Colorado: Niwot, CO, USA, 1996; pp. 417–437. [Google Scholar]
- Black, W.C., IV; Tabachnick, W.J. Population genetics of disease vectors. In Biology of Disease Vectors, 2nd; Marquardt, W.C., Ed.; Elsevier Academic Press: Amsterdam, NL, USA, 2005; pp. 465–504. [Google Scholar]
- Tabachnick, W.J. The yellow fever mosquito: Evolutionary genetics and arthropod-borne disease. Am. Entomol. 1991, 37, 14–24. [Google Scholar]
- Mousson, L.; Vazeille, M.; Chawprom, S.; Prajakwong, S.; Rodhain, F.; Failloux, A.B. Genetic structure of Aedes aegypti populations in Chiang Mai (Thailand) and relation with dengue transmission. Trop. Med. Int. Health. 2002, 7, 865–872. [Google Scholar] [CrossRef]
- Gorrochotegui-Escalante, N.; Gomez-Machorro, C.; Lozano-Fuentes, S.; Muñoz, I.; Farfan-Ale, J.A.; Garcia-Rejon, J.; Beaty, B.J.; Black, W.C., IV. The breeding structure of Aedes aegypti in Mexico varies by region. Am. J. Trop. Med. Hyg. 2002, 66, 213–222. [Google Scholar]
- Huber, K.; LeLoan, L.; Hoang, T.H.; Ravel, S.; Rodhain, F.; Failloux, A.B. Genetic differentiation of the dengue vector, Aedes aegypti (Ho Chi Minh City, Vietnam) using microsatellite marker. Mol. Ecol. 2002, 11, 1629–1635. [Google Scholar] [CrossRef]
- Huber, K.; Ba, Y.; Dia, I.; Mathiot, C.; Sall, A.A.; Diallo, M. Aedes aegypti in Senegal: Genetic diversity and genetic structure of domestic and sylvatic populations. Am. J. Trop. Med. Hyg. 2008, 79, 218–229. [Google Scholar]
- Sylla, M.; Bosio, C.; Urdaneta-Marquez, L.; Ndiaye, M.; Black IV, W.C. Gene flow, subspecies composition, and dengue virus-2 susceptibility among Aedes aegypti collections in Senegal. PLoS Negl. Trop. Dis. 2009. [Google Scholar] [CrossRef]
- Bracco, J.E.; Capurro, M.L.; Lourenço-de-Oliveira, R.; Sallum, M. Genetic variability of Aedes aegypti in the Americas using a mitochondrial gene: Evidence of multiple introductions. Mem. Inst. Oswaldo. Cruz. 2007, 102, 573–580. [Google Scholar] [CrossRef]
- Urdaneta-Marquez, L.; Failloux, A.B. Population genetic structure of Aedes aegypti,the principal vector of dengue viruses. Infect. Genet. Evol. 2011, 11, 253–261. [Google Scholar] [CrossRef]
- Urdaneta-Marquez, L.; Bosio, C.; Herrera, F.; Rubio-Palis, Y.; Salasek, M.; Black, W.C., IV. Genetic relationships among Aedes aegypti collections in Venezuela as determined by mitochondrial DNA variation and nuclear single nucleotide polymorphisms. Am. J. Trop. Med. Hyg. 2008, 78, 479–491. [Google Scholar]
- Brown, J.E.; McBride, C.S.; Johnson, P.; Ritchie, S.; Paupy, C.; Bossin, H.; Lutomiah, J.; Fernandez-Salas, I.; Ponlawat, A.; Cornel, A.J.; et al. Worldwide patterns of genetic differentiation imply multiple “domestications” of Aedes aegypti, a major vector of human diseases. Proc. Biol. Sci. 2011, 278, 2446–2454. [Google Scholar] [CrossRef]
- Kambhampati, S.; Black, W.C., IV; Rai, K.S. Geographic origin of the US and Brazilian Aedes albopictus inferred from allozyme analysis. Heredity 1991, 67, 85–93. [Google Scholar] [CrossRef]
- Urbanelli, S.; Bellini, R.; Carrieri, M.; Sallicandro, P.; Celli, G. Population structure of Aedes albopictus (Skuse): The mosquito which is colonizing Mediterranean countries. Heredity 2000, 84, 331–337. [Google Scholar] [CrossRef]
- Ayres, C.F.; Melo-Santos, M.A.; Solé-Cava, A.M.; Furtado, A.F. Genetic differentiation of Aedesaegypti (Diptera: Culicidae), the major dengue vector in Brazil. J. Med. Entomol. 2003, 40, 430–435. [Google Scholar] [CrossRef]
- Usmani-Brown, S.; Cohnstaedt, L.; Munstermann, L.E. Population genetics of Aedes albopictus (Diptera: Culicidae) invading populations, using mitochondrial nicotinamide adenine dinucleotide dehydrogenase subunit 5 sequences. Ann. Entomol. Soc. Am. 2009, 102, 144–150. [Google Scholar] [CrossRef]
- Kamgang, B.; Brengues, C.; Fontenille, D.; Njiokou, F.; Simard, F.; Paupy, C. Genetic structure of the tiger mosquito, Aedes albopictus, in Cameroon (Central Africa). PLoS ONE 2011. [Google Scholar] [CrossRef]
- Cui, F.; Qiao, C.L.; Shen, B.C.; Marquine, M.; Weill, M.; Raymond, R.M. Genetic differentiation of Culexpipiens (Diptera: Culicidae) in China. Bull. Entomol. Res. 2007, 97, 291–297. [Google Scholar] [CrossRef]
- Huang, S.; Molaei, G.; Andreadis, T.G. Genetic insights into the population structure of Culexpipiens (Diptera: Culicidae) in the Northeastern United States by using microsatellite analysis. Am. J. Trop. Med. Hyg. 2007, 79, 518–527. [Google Scholar]
- Edillo, F.; Kiszewski, A.; Manjourides, J.; Pagano, M.; Hutchinson, M.; Kyle, A.; Arias, J.; Gaines, D.; Lampman, R.; Novak, R.; et al. Effects of latitude and longitude on the population structure of Culexpipiens s.l., vectors of West Nile virus in North America. Am. J. Trop. Med. Hyg. 2009, 81, 842–428. [Google Scholar] [CrossRef]
- Fonseca, D.M.; Smith, J.L.; Kim, H.C.; Mogi, M. Population genetics of the mosquito Culex pipiens pallens reveals sex-linked asymmetric introgression by Culex quinquefasciatus. Infect. Genet. Evol. 2009, 9, 1197–1203. [Google Scholar] [CrossRef]
- Sharma, A.K.; Mendki, M.J.; Tikar, S.N.; Chandel, K.; Sukumaran, D.; Parashar, B.D.; Veer, V.; Agarwal, O.P.; Prakash, S. Genetic variability in geographical populations of Culex quinquefasciatus Say (Diptera: Culicidae) from India based on random amplified polymorphic DNA analysis. Acta Trop. 2009, 112, 71–76. [Google Scholar] [CrossRef]
- Weitzel, T.; Collado, A.; Jöst, A.; Pietsch, K.; Storch, V.; Becker, N. Genetic differentiation of populations within the Culex pipiens complex and phylogeny of related species. J. Am. Mosq. Contr. Assoc. 2009, 25, 6–17. [Google Scholar] [CrossRef]
- Failloux, A.B.; Vazeille-Falcoz, M.; Mousson, L.; Rodhain, F. Genetic control of vectorial competence in Aedes mosquitoes. Bull. Soc. Pathol. Exot. 1999, 92, 266–273. [Google Scholar]
- Severson, D.W.; Behura, S.K. Mosquito genomics: Progress and challenges. Annu. Rev. Entomol. 2012, 57, 143–166. [Google Scholar] [CrossRef]
- Hardy, J.L.; Apperson, G.; Asman, S.M.; Reeves, W.C. Selection of a strain of Culex tarsalis highly resistant to infection following ingestion of western equine encephalomyelitis virus. Am. J. Trop. Med. Hyg. 1978, 27, 313–331. [Google Scholar]
- Wallis, G.P.; Aitken, T.H.G.; Beaty, B.J.; Lorenz, L.; Amato, G.D.; Tabachnick, W.J. Selection for susceptibility and refractoriness of Aedes aegypti to oral infection with yellow fever virus. Am. J. Trop. Med. Hyg. 1985, 34, 1225–1231. [Google Scholar]
- Miller, B.R.; Mitchell, C.J. Genetic selection of a flavivirus-refractory strain of the yellow fever mosquito Aedes aegypti. Am. J. Trop. Med. Hyg. 1991, 45, 399–407. [Google Scholar]
- Tardieux, I.; Poupel, O.; Lapchin, L.; Rodhain, F. Analysis of inheritance of oral susceptibility of Aedes aegypti (Diptera: Culicidae) dengue-2 virus using isofemale lines. J. Med. Entomol. 1991, 28, 518–521. [Google Scholar]
- Mourya, D.T.; Gokhale, M.D.; Malunjkar, A.S.; Bhat, H.R.; Banerjee, K. Inheritance of oral susceptibility of Aedes aegypti to chikungunya virus. Am. J. Trop. Med. Hyg. 1994, 51, 295–300. [Google Scholar]
- Lambrechts, L.; Chevillon, C.; Albright, R.G.; Thaisomboonsuk, B.; Richardson, J.H.; Jarman, R.G.; Scott, T.W. Genetic specificity and potential for local adaptation between dengue viruses and mosquito vectors. BMC Evolut. Biol. 2009. [Google Scholar] [CrossRef]
- Tabachnick, W.J. Genetic control of oral susceptibility to infection of Culicoides variipennis for bluetongue virus. Am. J. Trop. Med. Hyg. 1991, 45, 666–671. [Google Scholar]
- Black, W.C., IV; Bennett, K.E.; Gorrochótegui-Escalante, N.; Barillas-Mury, C.V.; Fernández-Salas, I.; de Lourdes Muñoz, M.; Farfán-Alé, J.A.; Olson, K.E.; Beaty, B.J. Flavivirus susceptibility in Aedes aegypti. Arch. Med. Res. 2002, 33, 379–388. [Google Scholar] [CrossRef]
- Bosio, C.F.; Fulton, R.E.; Salasek, M.L.; Beaty, B.J.; Black, W.C., IV. Quantitative trait loci that control vector competence for dengue-2 virus in the mosquito Aedes aegypti. Genetics 2000, 156, 687–698. [Google Scholar]
- Bennett, K.E.; Flick, D.; Fleming, K.H.; Beaty, B.J.; Black, W.C., IV. Quantitative trait loci that control Dengue-2 virus dissemination in the mosquito Aedes aegypti. Genetics 2005, 170, 185–194. [Google Scholar] [CrossRef]
- Bennett, K.E.; Beaty, B.J.; Black, W.C., IV. Selection of D2S3, an Aedes aegypti (Diptera: Culicidae) strain with high oral susceptibility to dengue 2 virus and D2MEB, a strain with a midgut barrier to dengue escape. J. Med. Entomol. 2005, 42, 110–119. [Google Scholar] [CrossRef]
- Griffiths, A.J.F.; Wessler, S.R.; Carroll, S.B.; Doebley, J. An Introduction to Genetic Analysis, 7th ed; W.H. Freeman: San Francisco, CA, USA, 2010; p. 800. [Google Scholar]
- Chen, X.G.; Mathur, G.; James, A.A. Gene expression studies in mosquitoes. Adv. Genet. 2008, 64, 19–50. [Google Scholar] [CrossRef]
- Bartholomay, L.C.; Waterhouse, R.M.; Mayhew, G.F.; Campbell, C.L.; Michel, K.; Zou, Z.; Ramirez, J.L.; Das, S.; Alvarez, K.; Arensburger, P.; et al. Pathogenomics of Culex quinquefasciatus and meta-analysis of infection response to diverse pathogens. Science 2010, 330, 88–90. [Google Scholar]
- Girard, Y.A.; Mayhew, G.F.; Fuchs, J.F.; Schneider, B.S.; McGee, C.E.; Rocheleau, T.A.; Helmy, H.; Christensen, B.M.; Higgs, S.; Bartholomay, L.C. Transcriptome changes in Culex quinquefasciatus (Diptera; Culicidae) salivary glands during West Nile infection. J. Med. Entomol. 2010, 4, 421–435. [Google Scholar]
- Tchankouo-Nguetcheu, S.; Khun, H.; Pincet, L.; Roux, P.; Bahut, M.; Huerre, M.; Guette, C.; Choumet, V. Differential protein modulation in midguts of Aedes aegypti infected with chikungunya and dengue 2 viruses. PLoS ONE 2010. [Google Scholar] [CrossRef]
- Behura, S.K.; Gomez-Machorro, C.; Harker, B.W.; de Bruyn, B.; Lovin, D.D.; Hemme, R.R.; Mori, A.; Romero-Severson, J.; Severson, D.W. Global cross-talk of genes of the mosquito Aedes aegypti in response to dengue virus infection. PLoS Negl. Trop. Dis. 2011. [Google Scholar] [CrossRef]
- Colpitts, T.M.; Cox, J.; Vanlandingham, D.L.; Feitosa, F.M.; Chang, G.; Kurscheid, S.; Wang, P.; Krisham, M.N.; Higgs, S.; Fikrig, E. Alterations in the Aedes aegypti transcriptome during infection with West Nile, dengue and yellow fever viruses. PLoS Pathog. 2011. [Google Scholar] [CrossRef]
- Bonizzoni, M.; Dunn, W.A.; Campbell, C.L.; Olson, K.E.; Marinotti, O.; James, A.A. Strain variation in the Transcriptome of the dengue fever vector, Aedes aegypti. G3: Genes|Genomes|Genetics 2012, 2, 103–114. [Google Scholar]
- Christophides, G.K.; Vlachou, D.; Kafatos, F.C. Comparative and functional genomics of the innate immune system in the malaria vector Anopheles gambiae. Immunol. Rev. 2004, 198, 127–148. [Google Scholar] [CrossRef]
- Sanders, H.R.; Evans, A.M.; Ross, L.S.; Gill, S.S. Blood meal induces global changes in midgut gene expression in the disease vector, Aedes aegypti. Insect. Biochem. Mol. Biol. 2003, 33, 1105–1122. [Google Scholar] [CrossRef]
- Sanders, H.R.; Foy, B.D.; Evans, A.M.; Ross, L.S.; Beaty, B.J.; Olson, K.E.; Gill, S.S. Sindbis virus induces transport processes and alters expression of innate immunity pathway genes in the midgut of the disease vector, Aedes aegypti. Insect Biochem. Mol. Biol. 2005, 35, 1293–1307. [Google Scholar] [CrossRef]
- Xi, Z.; Ramirez, J.L.; Dimopoulos, G. The Aedes aegypti toll pathway controls dengue virus infection. PLoS Pathog. 2008. [Google Scholar] [CrossRef]
- Fragkoudis, R.; Attarzadeh-Yazdi, G.; Nash, A.A.; Fazakerley, J.K.; Kohl, A. Advances in dissecting mosquito innate immune responses to arbovirus infection. J. Gen. Virol. 2009, 90, 2061–2072. [Google Scholar] [CrossRef]
- Souza-Neto, J.A.; Sim, S.; Dimopoulos, G. An evolutionary conserved function of the JAK-STAT pathway in anti-dengue defense. Proc. Natl. Acad. Sci. USA 2009, 106, 17841–17846. [Google Scholar] [CrossRef]
- Ramirez, J.L.; Dimopoulos, G. The toll immune signaling pathway control conserved anti-dengue defenses across diverse Ae. aegypti strains and against multiple dengue virus serotypes. Dev. Comp. Immunol. 2010, 34, 625–6259. [Google Scholar] [CrossRef]
- Sanchez-Vargas, I.; Scott, J.C.; Smith, B.K.; Franz, A.W.; Barbosa-Solomieu, V.; Wilusz, J.; Olson, K.E.; Blair, C.D. Dengue virus type 2 infections of Aedes aegypti are modulated by the mosquitoes RNA interference pathway. PLoS Pathog. 2009. [Google Scholar] [CrossRef]
- Khoo, C.; Piper, J.; Sanchez-Vargas, I.; Olson, K.; Franz, A. The RNA interference pathway affects midgut infection- and escape barriers for Sindbis virus in Aedes aegypti. BMC Microbiol. 2010. [Google Scholar] [CrossRef]
- Blair, C.D. Mosquito RNAi is the major innate immune pathway controlling arbovirus infection and transmission. Future Microbiol. 2011, 6, 265–277. [Google Scholar] [CrossRef]
- Hess, A.M.; Prasad, A.N.; Pittsyn, A.G.; Ebel, G.D.; Olson, K.E.; Barbacioru, C.; Monighetti, C.; Campbell, C.L. Small RNA profiling of dengue virus-mosquito interactions implicate the PIWI RNA pathway in anti-viral defense. BMC Microbiol. 2011. [Google Scholar] [CrossRef]
- Bernhardt, S.A.; Simmons, M.P.; Olson, K.E.; Beaty, B.J.; Blair, C.D.; Black, W.C. Rapid intraspecific evolution of miRNA and siRNA genes in the mosquito Aedes aegypti. PLoS ONE 2012. [Google Scholar] [CrossRef]
- Molina-Cruz, A.; Gupta, L.; Richardson, J.; Bennett, K.; Black, W.C., IV.; Barillas-Mury, C. Effect of mosquito midgut trypsin activity on dengue-2 virus infection and dissemination in Aedes aegypti. Am. J. Trop. Med. Hyg. 2005, 72, 631–637. [Google Scholar]
- Brackney, D.E.; Foy, B.D.; Olson, K.E. The effects of midgut serine proteases on dengue virus type 2 infectivity of Aedes aegypti. Am. J. Trop. Med. Hyg. 2008, 79, 267–274. [Google Scholar]
- Houk, E.J.; Arcus, Y.M.; Hardy, J.L.; Kramer, L.D. Binding of western equine encephalomyelitis virus to brush border fragments isolated from mesenteronal epithelial cells of mosquitoes. Virus Res. 1990, 17, 105–117. [Google Scholar] [CrossRef]
- Ludwig, G.V.; Kondig, J.P.; Smith, J.F. A putative receptor for Venezuelan equine encephalitis virus in mosquito from mosquito cells. J. Virol. 1996, 70, 5592–5599. [Google Scholar]
- Mourya, D.T.; Ranadive, S.N.; Gokhale, M.D.; Barde, P.V.; Padbidri, V.S.; Banerjee, K. Putative chikungunya virus-specific receptor proteins on the midgut brush border of Aedes aegypti mosquito. IndianJ. Med. Res. 1998, 107, 10–14. [Google Scholar]
- Yazi, M.M.; Salas-Benito, J.S.; Lanz-Mandoza, H.; Hernandez-Martinez, S.; del Angel, R.M. A putative receptor for dengue virus in mosquito tissues: Localization of a 45-kDa glycoprotein. Am. J. Trop. Med. Hyg. 2002, 67, 76–84. [Google Scholar]
- Salas-Benito, J.; Reyes-Del Valle, J.; Salas-Benito, M.; Ceballos-Olvera, I.; Mosso, C.; del Angel, R.M. Evidence that the 45-kD glycoprotein, part of a putative dengue virus receptor complex in the mosquito cell line C6/36, is a heat-shock related protein. Am. J. Trop. Med. Hyg. 2007, 77, 283–290. [Google Scholar]
- Mercado-Curiel, R.F.; Esquinca-Aviles, H.F.; Tovar, R.; Diaz-Badillo, A.; Camacho-Nuez, M.; Munoz, M.L. The four serotypes of dengue recognize the same putative receptor molecules in Aedes aegypti midgut and Ae. albopictus cells. BMC Microbiol. 2006. [Google Scholar] [CrossRef]
- Mercado-Curiel, R.F.; Black, W.C., IV.; Muñoz Mde, L. A dengue receptor as possible genetic marker of vector competence in Aedes aegypti. BMC Microbiol. 2008. [Google Scholar] [CrossRef]
- Cao-Lormeau, V. Dengue viruses binding proteins from Aedes aegypti and Aedes polynesiensis salivary glands. Virol. J. 2009. [Google Scholar] [CrossRef]
- Hidari, K.I.; Suzuki, T. Dengue virus receptor. Trop. Med. Health 2011, 39, 37–43. [Google Scholar] [CrossRef]
- Smith, D.R. An update on mosquito cell expressed dengue virus receptor proteins. Insect Mol. Biol. 2012, 21, 1–7. [Google Scholar] [CrossRef]
- Chauhan, C.; Behura, S.K.; Debruyn, B.; Lovin, D.D.; Harker, B.W.; Gomez-Machorro, C.; Mori, A.; Romero-Severson, J.; Severson, D.W. Comparative expression profiles of midgut genes in dengue virus refractory and susceptible Aedes aegypti across critical period for virus infection. PLoS ONE 2012. [Google Scholar] [CrossRef]
- Smith, D.R.; Adams, A.P.; Kenney, J.L.; Wang, E.; Weaver, S.C. Venezuelan equine encephalitis virus in the mosquito vector Aedes taeniorhynchus: Infection initiated by a small number of susceptible epithelial cells and a population bottleneck. Virology 2008, 372, 176–186. [Google Scholar] [CrossRef]
- Cox, J.; Brown, H.E.; Rico-Hesse, R. Variation in vector competence for dengue viruses does not depend on mosquito midgut binding affinity. PLoS Negl. Trop. Dis. 2011. [Google Scholar] [CrossRef]
- Behura, S.K.; Severson, D.W. Intrinsic features of Aedes aegypti genes affect transcriptional responsiveness of mosquito genes to dengue infection. Infect. Genet. Evol. 2012, 12, 1413–1418. [Google Scholar] [CrossRef]
- Hurlburt, H.S. The effect of environmental temperature upon the transmission of St. Louis encephalitis virus by Culex pipiens quinquefasciatus. J. Med. Entomol. 1973, 10, 1–12. [Google Scholar]
- Watts, D.M.; Burke, D.S.; Harrison, B.A.; Whitmire, R.E.; Nisalak, A. Effect of temperature on the vector efficiency of Aedes aegypti for dengue 2 virus. Am. J. Trop. Med. Hyg. 1987, 36, 143–52. [Google Scholar]
- Turell, M.J.; Lundström, J.O. Effect of environmental temperature on the vector competence of Aedes aegypti and Ae. taeniorhynchus for Ockelbo virus. Am. J. Trop. Med. Hyg. 1990, 43, 543–550. [Google Scholar]
- Turell, M.J. Effect of environmental temperature on the vector competence of Aedes taeniorhynchus for Rift Valley fever and Venezuelan equine encephalitis viruses. Am. J. Trop. Med. Hyg. 1993, 49, 672–676. [Google Scholar]
- Cornel, A.J.; Jupp, P.G.; Blackburn, N.K. Environmental temperature on the vector competence of Culex univittatus (Diptera: Culicidae) for West Nile virus. J. Med. Entomol. 1993, 30, 449–456. [Google Scholar]
- Thu, H.M.; Aye, K.M.; Thein, S. The effect of temperature and humidity on dengue virus propagation in Aedes aegypti mosquitos. Southeast Asian J. Trop. Med. Public Health 1998, 29, 280–284. [Google Scholar]
- Brubaker, J.F.; Turell, M.J. Effect of environmental temperature on the susceptibility of Culex pipiens (Diptera: Culicidae) to Rift Valley fever virus. J. Med. Entomol. 1998, 35, 918–921. [Google Scholar]
- Kramer, L.D.; Hardy, J.L.; Presser, S.B. Characterization of modulation of western equine encephalomyelitis virus by Culex tarsalis (Diptera: Culicidae) maintained at 32 °C following parenteral infection. J. Med. Entomol. 1998, 35, 289–295. [Google Scholar]
- Kay, B.H.; Jennings, C.D. Enhancement or modulation of the vector competence of Ochlerotatus vigilax (Diptera: Culicidae) for Ross River virus by temperature. J. Med. Entomol. 2002, 39, 99–105. [Google Scholar] [CrossRef]
- Mourya, D.T.; Yadav, P.; Mishra, A.C. Effect of temperature stress on immature stages and susceptibility of Aedes aegypti mosquitoes to chikungunya virus. Am. J. Trop. Med. Hyg. 2004, 70, 346–350. [Google Scholar]
- Reiskind, M.H.; Pesko, K.; Westbrook, C.J.; Mores, C.N. Susceptibility of Florida mosquitoes to infection with chikungunya virus. Am. J. Trop. Med. Hyg. 2008, 78, 422–425. [Google Scholar]
- Reisen, W.K.; Fang, Y.; Martinez, V.M. Effects of temperature on the transmission of West Nile virus by Culex tarsalis (Diptera: Culicidae). J. Med. Entomol. 2006, 43, 309–317. [Google Scholar] [CrossRef]
- Rohani, A.; Wong, Y.C.; Zamre, I.; Lee, H.L.; Zurainee, M.N. The effect of extrinsic incubation temperature on development of dengue serotype 2 and 4 viruses in Aedes aegypti (L.). Southeast Asian J. Trop. Med. Public Health 2009, 40, 942–950. [Google Scholar]
- Kilpatrick, A.M.; Fonseca, D.M.; Ebel, G.D.; Reddy, M.R.; Kramer, L.D. Spatial and temporal variation in vector competence of Culex pipiens and Cx. restuans mosquitoes for West Nile virus. Am. J. Trop. Med. Hyg. 2010, 83, 607–613. [Google Scholar] [CrossRef]
- Lambrechts, L.; Paaijmans, K.P.; Fansiri, T.; Carrington, L.B.; Kramer, L.D.; Thomas, M.B.; Scott, T.W. Impact of daily temperature fluctuations on dengue virus transmission by Aedes aegypti. Proc. Natl. Acad. Sci. USA 2011, 108, 7460–7465. [Google Scholar]
- Kramer, L.D.; Hardy, J.L.; Presser, S.B. Effect of temperature of extrinsic incubation on the vector competence of Culex tarsalis for western equine encephalomyelitis virus. Am. J. Trop. Med. Hyg. 1983, 32, 1130–1139. [Google Scholar]
- Grimstad, P.R.; Haramis, L.D. Aedes triseriatus (Diptera: Culicidae) and La Crosse virus. III. Enhanced oral transmission by nutrition-deprived mosquitoes. J. Med. Entomol. 1984, 21, 249–256. [Google Scholar]
- Kay, B.H.; Fanning, I.D.; Mottram, P. Rearing temperature influences flavivirus vector competence of mosquitoes. Med. Vet. Entomol. 1989, 3, 415–422. [Google Scholar] [CrossRef]
- Westbrook, C.J.; Reiskind, M.H.; Pesko, K.N.; Greene, K.E.; Lounibos, L.P. Larval environmental temperature and the susceptibility of Aedes albopictus Skuse (Diptera: Culicidae) to chikungunya virus. Vector-Borne Zoonotic Dis. 2010, 10, 241–247. [Google Scholar] [CrossRef]
- Muturi, E.J.; Alto, B.W. Larval environmental temperature and insecticide exposure alter Aedes aegypti competence for arboviruses. Vector-Borne Zoonotic Dis. 2011, 11, 1157–1163. [Google Scholar] [CrossRef]
- Muturi, E.J.; Costanzo, K.; Kesavaraju, B.; Alto, B.W. Can pesticides and larval competition alter susceptibility of Aedes mosquitoes (Diptera: Culicidae) to arbovirus infection? J. Med. Entomol. 2011, 48, 429–436. [Google Scholar] [CrossRef]
- Yadav, P.; Barde, P.V.; Gokhale, M.D.; Vipat, V.; Mishra, A.C.; Pal, J.K.; Mourya, D.T. Effect of temperature and insecticide stress on Aedes aegypti larvae and their influence on the susceptibility of mosquitoes to dengue-2 virus. Southeast Asian J. Trop. Med. Public Health 2005, 36, 1139–1144. [Google Scholar]
- Muturi, E.J.; Kim, C-H.; Alto, B.W.; Schuler, M.A.; Berenbaum, M.R. Larval environmental stress alters adult mosquito fitness and competence for arboviruses. Trop. Med. Internat. Health 2011, 16, 955–964. [Google Scholar] [CrossRef]
- Gubler, D.J.; Nalim, S.; Tan, H.; Saipan, S.; Saroso, J. Variation in susceptibility to oral infection with dengue viruses among geographic strains of Aedes aegypti. Am. J. Trop. Med. Hyg. 1979, 28, 1045–1052. [Google Scholar]
- Miller, B.R.; Mitchell, C.J. Passage of yellow fever virus: Its effect on infection and transmission rates in Aedes aegypti. Am. J. Trop. Med. Hyg. 1986, 35, 1302–1309. [Google Scholar]
- Mitchell, C.J.; Miller, B.R.; Gubler, D.J. Vector competence of Aedes albopictus from Houston, Texas, for dengue serotypes 1 to 4, yellow fever and Ross River viruses. J. Am. Mosq. Contr. Assoc. 1987, 3, 460–465. [Google Scholar]
- Turell, M.J.; Ludwig, G.V.; Beaman, J.R. Transmission of Venezuelan equine encephalomyelitis virus by Aedes sollicitans and Aedes taeniorhynchus (Diptera: Culicidae). J. Med. Entomol. 1992, 29, 62–65. [Google Scholar]
- Mecham, J.O.; Nunamaker, R.A. Complex interactions between vectors and pathogens: Culicoides variipennis sonorensis (Diptera: Ceratopogonidae) infection rates with bluetongue viruses. J. Med. Entomol. 1994, 31, 903–907. [Google Scholar]
- Armstrong, P.M.; Rico-Hesse, R. Differential susceptibility of Aedes aegypti to infection by the American and Southeast Asia genotypes of dengue type 2 virus. Vector-Borne Zoonotic Dis. 2001, 1, 159–168. [Google Scholar] [CrossRef]
- Armstrong, P.M.; Rico-Hesse, R. Efficiency of dengue serotype 2 virus strains to infect and disseminate in Aedes aegypti. Am. J. Trop. Med. Hyg. 2003, 68, 539–544. [Google Scholar]
- Brault, A.C.; Powers, A.M.; Ortiz, D.; Estrada-Franco, J.G.; Navarro-Lopez, R.; Weaver, S.C. Venezuelan equine encephalitis emergence: enhanced vector infection from a single amino acid substitution in the envelope glycoprotein. Proc. Natl. Acad. Sci. USA 2004, 101, 11344–11349. [Google Scholar]
- Vanlandingham, D.L.; McGee, C.E.; Klingler, K.A.; Galbraith, S.E.; Barrett, A.D.; Higgs, S. Short report: Comparison of oral infectious dose of West Nile virus isolates representing three distinct genotypes in Culex quinquefasciatus. Am. J. Trop. Med. Hyg. 2008, 79, 951–954. [Google Scholar]
- McElroy, K.L.; Tsetsarkin, K.A.; Vanlandingham, D.L.; Higgs, S. Role of the yellow fever virus structural protein genes in viral dissemination from the Aedes aegypti mosquito midgut. J. Gen. Virol. 2006, 87, 2993–3001. [Google Scholar]
- Moudy, R.M.; Meola, M.M.; Morin, M.M.; Ebel, G.D.; Kramer, L.D. A newly emergent genotype of West Nile virus is transmitted earlier and more efficiently by Culex mosquitoes. Am. J. Trop. Med. Hyg. 2007, 77, 365–370. [Google Scholar]
- Rico-Hesse, R. Dengue virus virulence and transmission determinants. Curr. Top. Microbiol. Immunol. 2010, 338, 45–55. [Google Scholar] [CrossRef]
- Deardorff, E.R.; Weaver, S.C. Vector competence of Culex (Melanoconion) taeniopus for equine-virulent subtype IE strains of Venezuelan equine encephalitis virus. Am. J. Trop. Med. Hyg. 2010, 82, 1047–1052. [Google Scholar] [CrossRef]
- Christofferson, R.C.; Mores, C.N. Estimating the magnitude and direction of altered arbovirus transmission due to viral phenotype. PLoS ONE 2011. [Google Scholar] [CrossRef]
- Andrade, C.C.; Maharaj, P.D.; Reisen, W.K.; Brault, A.C. North American WestNile virus genotype isolates demonstrate differential replicative capacities in response to temperature. J. Gen. Virol. 2011, 92, 2523–2533. [Google Scholar] [CrossRef]
- Styer, L.M.; Bernard, K.A.; Kramer, L.D. Enhanced early West Nile virus infection in young chickens infected by mosquito bite: effect of viral dose. Am. J. Trop. Med. Hyg. 2006, 75, 337–345. [Google Scholar]
- Richards, S.L.; Anderson, S.L.; Lord, C.C.; Tabachnick, W.J. Impact of West Nile virus dose and incubation period on vector competence of Culex nigripalpus. Vector-Borne Zoonotic Dis. 2011, 11, 1487–1491. [Google Scholar] [CrossRef]
- Pesko, K.; Westbrook, C.J.; Mores, C.N.; Lounibos, L.P.; Reiskind, M.H. Effects of infectious virus dose and bloodmeal delivery method on susceptibility of Aedes aegypti and Aedes albopictus to chikungunya virus. J. Med. Entomol. 2009, 46, 395–399. [Google Scholar] [CrossRef]
- Anderson, S.L.; Richards, S.L.; Smartt, C.T.; Tabachnick, W.J. The effects of West Nile virus dose and extrinsic incubation temperature on the temporal progression of vector competence in Culex pipiens quinquefasciatus Say (Diptera: Culicidae). J. Am. Mosq. Contr. Assoc. 2010, 26, 103–107. [Google Scholar] [CrossRef]
- Pan, X.; Zhou, G.; Wu, J.; Bian, G.; Lu, P.; Raikhel, A.S.; Xi, Z. Wolbachia induces reactive e oxygen species (ROS)-dependent activation of the Toll pathway to control dengue virus in the mosquito Aedes aegypti. Proc. Natl. Acad. Sci. USA 2012, 109, E23–E31. [Google Scholar]
- Rances, E.; Ye, Y.H.; Woolfit, M.; McGraw, E.A.; O’Neill, S.L. The relative importance of innate immune priming in Wolbachia-mediated dengue interference. PLoS Pathog. 2012. [Google Scholar] [CrossRef]
- Richards, S.L.; Lord, C.C.; Pesko, K.; Tabachnick, W.J. Environmental and biological factors influencing Culex pipiens quinquefasciatus Say (Diptera: Culicidae) vector competence for St. Louis encephalitis virus. Am. J. Trop. Med. Hyg. 2009, 81, 264–272. [Google Scholar]
- Richards, S.L.; Lord, C.C.; Pesko, K.N.; Tabachnick, W.J. Environmental and biological factors influencing Culex pipiens quinquefasciatus Say (Diptera: Culicidae) vector competence for West Nile virus. Am. J. Trop. Med. Hyg. 2010, 83, 126–134. [Google Scholar] [CrossRef]
- Miller, B.R.; Monath, T.P.; Tabachnick, W.J.; Ezike, V.I. Epidemic yellow fever caused by an incompetent mosquito vector. Trop. Med. Parasitol. 1989, 40, 396–399. [Google Scholar]
- Smith, C.E. Factors influencing the transmission of western equine encephalomyelitis virus between its vertebrate maintenance and from them to humans. Am. J. Trop. Med. Hyg. 1987, 37, 33–39. [Google Scholar]
- Monath, T.P. The absence of yellow fever in Asia: hypotheses. A cause for concern? Virus Information Exchange Newsletter 1988, 6, 106–107. [Google Scholar]
- Amaku, M.; Coutinho, F.A.B.; Massad, E. Why dengue and yellow fever coexist in some areas of the world and not in others? Biosystems 2011, 106, 111–120. [Google Scholar] [CrossRef]
- Lewontin, R. The Triple Helix; Harvard University Press: Cambridge, MA, USA, 2000. [Google Scholar]
- Tabachnick, W.J. Reflections on the Anopheles gambiae genome sequence, transgenic mosquitoes and the prospect for controlling malaria and other vector borne diseases. J. Med. Entomol. 2003, 40, 597–606. [Google Scholar] [CrossRef]
- Tabachnick, W.J. Challenges in predicting climate and environmental effects on vector-borne disease episystems in a changing world. J. Exp. Biol. 2010, 213, 946–954. [Google Scholar] [CrossRef]
- Tabachnick, W.J.; Day, J.F. The impact of climate change on vector-borne arboviral episystems. In Viral Infections and Climate Change; Singh, S.K., Ed.; John Wiley & Sons/Blackwell Press: Hoboken, NJ, USA, 2012. [Google Scholar]
- Lambrechts, L.; Scott, T.W. Mode of transmission and the evolution of arbovirus virulence in mosquito vectors. Proc. Roy. Soc. B 2009, 276, 1369–1378. [Google Scholar] [CrossRef]
- Cohuet, A.; Harris, C.; Robert, V.; Fontenille, D. Evolutionary forces on Anopheles: What makes a malaria vector? Trends Parasitol. 2010, 26, 130–136. [Google Scholar] [CrossRef]
- Turell, M.J.; Reeves, W.C.; Hardy, J.L. Transovarial and trans-stadial transmission of California encephalitis virus in Aedes dorsalis and Aedes melanimon. Am. J. Trop. Med. Hyg. 1982, 31, 1021–1029. [Google Scholar]
- Grimstad, P.R.; Ross, Q.E.; Craig, G.B., Jr. Aedes triseriatus (Diptera: Culicidae) and La Crosse virus. II. Modification of mosquito feeding behavior by virus infection. J. Med. Entomol. 1980, 17, 1–7. [Google Scholar]
- Weaver, S.C.; Scott, T.W.; Lorenz, L.H.; Lerdthusnee, K.; Romoser, W.S. Togavirus-associated pathologic changes in the midgut of a natural mosquito vector. J. Virol. 1988, 62, 2083–2090. [Google Scholar]
- Hurd, H.; Hogg, J.C.; Renshaw, M. Interactions between bloodfeeding, fecundity and infection in mosquitoes. Parasitol. Today 1995, 11, 411–416. [Google Scholar] [CrossRef]
- Weaver, S.C.; Lorenz, L.H.; Scott, T.W. Pathologic changes in the midgut of Culex tarsalis following infection with Western equine encephalomyelitis virus. Am. J. Trop. Med. Hyg. 1992, 47, 691–701. [Google Scholar]
- Scott, T.W.; Lorenz, L.H. Reduction of Culiseta melanura fitness by eastern equine encephalomyelitis virus. Am. J. Trop. Med. Hyg. 1998, 59, 341–346. [Google Scholar]
- Moncayo, A.C.; Edman, J.D.; Turell, M.J. Effect of eastern equine encephalomyelitis virus on the survival of Aedes albopictus, Anopheles quadrimaculatus, and Coquillettidia perturbans (Diptera: Culicidae). J. Med. Entomol. 2000, 37, 701–706. [Google Scholar] [CrossRef]
- Mahmood, F.; Reisen, W.K.; Chiles, R.E.; Fang, Y. Western equine encephalomyelitis virus infection affects the life table characteristics of Culex tarsalis. J. Med. Entomol. 2004, 41, 982–986. [Google Scholar] [CrossRef]
- Gajanana, A.; Rajendran, R.; Samuel, P.P.; Thenmozhi, V.; Tsai, T.F.; Kimura-Kuroda, J.; Reuben, R. Japanese encephalitis in south Arcot district, Tamil Nadu, India: A three-year longitudinal study of vector abundance and infection frequency. J. Med. Entomol. 1997, 34, 651–659. [Google Scholar]
- Nasci, R.S.; White, D.J.; Stirling, H.; Oliver, J.; Daniels, T.J.; Falco, R.C.; Campbell, S.; Crans, W.J.; Savage, H.M.; Lanciotti, R.S.; Moore, C.G.; Godsey, M.S.; Gottfried, K.L.; Mitchell, C.J. West Nile virus isolates from mosquitoes in New York and New Jersey, 1999. Emerg. Infect. Dis. 2001, 7, 626–630. [Google Scholar]
- Jupp, P.G.; Kemp, A.; Grobbelaar, A.; Lema, P.; Burt, F.J.; Alahmed, A.M.; Al Mujalli, D.; Al Khamees, M.; Swanepoel, R. The 2000 epidemic of Rift Valley fever in Saudi Arabia: Mosquito vector studies. Med. Vet. Entomol. 2002, 16, 245–252. [Google Scholar] [CrossRef]
- Urdaneta, L.; Herrera, F.; Pernalete, M.; Zoghbi, N.; Rubio-Palis, Y.; Barrios, R.; Rivero, J.; Comach, G.; Jimenez, M.; Salcedo, M. Detection of dengue viruses in field-caught Aedes aegypti (Diptera: Culicidae) in Maracay, Aragua state, Venezuela by type-specific polymerase chain reaction. Infect. Genet. Evol. 2005, 5, 177–184. [Google Scholar] [CrossRef]
- Sang, R.C.; Ahmed, O.; Faye, O.; Kelly, C.L.; Yahaya, A.A.; Mmadi, I.; Toilibou, A.; Sergon, K.; Brown, J.; Agata, N.; et al. Entomologic investigations of a chikungunya virus epidemic in the Union of the Comoros, 2005. Am. J. Trop. Med. Hyg. 2008, 78, 77–82. [Google Scholar]
- Barrera, R.; MacKay, A.; Amador, M.; Smith, J.; Diaz, A.; Acevedo, V.; Caban, B.; Hunsperger, E.A. mosquito vectors of West Nile during an epizootic outbreak in Puerto Rico. J. Med. Entomol. 2010, 47, 1185–1195. [Google Scholar] [CrossRef]
- .Garcia-Rejon, J.E.; Lorono-Pino, M.A.; Farfan-Ale, J.A.; Flores-Flores, L.F.; Lopes-Uribe, M.P.; Najera-Vazquez Mdel, R.; Nunez-Ayala, G.; Beaty, B.J.; Eisen, L. Mosquito infestation and dengue virus infection in Aedes aegypti females in schools in Merida, Mexico. Am. J. Trop. Med. Hyg. 2011, 84, 489–496. [Google Scholar] [CrossRef]
- Williams, G.C. Adaptation and Natural Selection, 1st ed; Oxford University Press: Oxford, UK, 1966; p. 320. [Google Scholar]
- Gould, S.J.; Vrba, E.S. Exaptation—A missing term in the science of form. Paleobiology 1982, 8, 4–15. [Google Scholar]
- Lambrechts, L.; Knox, T.B.; Wong, J.; Liebman, K.A.; Albright, R.G.; Stoddard, S.T. Shifting priorities in vector biology to improve control of vector-borne disease. Trop. Med. Int. Health 2009, 14, 1505–1514. [Google Scholar] [CrossRef]
- Desenclos, J.-C. Transmission parameters of vector-borne infections. Med. Mal. Infect. 2011, 41, 588–593. [Google Scholar] [CrossRef]
- Dobzhansky, T. Nothing in biology makes sense except in the light of evolution. Am. Biol. Teacher 1973, 35, 125–129. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Tabachnick, W.J. Nature, Nurture and Evolution of Intra-Species Variation in Mosquito Arbovirus Transmission Competence. Int. J. Environ. Res. Public Health 2013, 10, 249-277. https://doi.org/10.3390/ijerph10010249
Tabachnick WJ. Nature, Nurture and Evolution of Intra-Species Variation in Mosquito Arbovirus Transmission Competence. International Journal of Environmental Research and Public Health. 2013; 10(1):249-277. https://doi.org/10.3390/ijerph10010249
Chicago/Turabian StyleTabachnick, Walter J. 2013. "Nature, Nurture and Evolution of Intra-Species Variation in Mosquito Arbovirus Transmission Competence" International Journal of Environmental Research and Public Health 10, no. 1: 249-277. https://doi.org/10.3390/ijerph10010249
APA StyleTabachnick, W. J. (2013). Nature, Nurture and Evolution of Intra-Species Variation in Mosquito Arbovirus Transmission Competence. International Journal of Environmental Research and Public Health, 10(1), 249-277. https://doi.org/10.3390/ijerph10010249