Coarse and Fine Culturable Fungal Air Concentrations in Urban and Rural Homes in Egypt
Abstract
:1. Introduction
2. Experimental Section
2.1. Sampling Strategy and Sites
| Characteristic | Urban (%) | Rural (%) | |
|---|---|---|---|
| Age of building | 1–5 years | 7 | 0 |
| 5–10 years | 22 | 6 | |
| 10–15 years | 15 | 6 | |
| >15 years | 56 | 88 | |
| Number of occupants | 1–2 person | 11 | 19 |
| 3 person | 4 | 0 | |
| 4 person | 41 | 13 | |
| ≥5 person | 44 | 69 | |
| Type of building | Single house | 0 | 19 |
| Small complex (2–3 apartments) | 0 | 81 | |
| Big complex (>5 apartments) | 100 | 0 | |
| Pets | Yes | 4 | 50 |
| No | 96 | 50 |
2.2. Sampling of Fungi
2.3. Statistical Analysis
3. Results
3.1. Overall Concentrations
| Coarse (> 8µm) | Fine (< 8µm) | |||||
|---|---|---|---|---|---|---|
| Indoor | Outdoor | Indoor | Outdoor | Indoor | ||
| Urban (n = 26) | ||||||
| Range | 1–641 | 8–742 | 43–4,108 | 32–3,076 | 65–4,523 | 67–3,697 |
| Mean | 129 | 174 | 765 | 810 | 894 | 984 |
| Median | 101 | 128 | 495 | 541 | 608 | 675 |
| I/O ratio a | 0.789 | 0.91 | 0.908 | |||
| Rural (n = 17) | ||||||
| Range | 40–2,744 | 1–4,604 | 67–32,040 | 120–14,264 | 111–34,784 | 360–16,492 |
| Mean | 572 | 613 | 3,273 | 2,854 | 3845 | 3467 |
| Median | 404 | 353 | 1,537 | 1,441 | 1,932 | 1,872 |
| I/O ratio a | 1.14 | 1.07 | 1.03 | |||
3.2. Seasonal Concentrations
| Winter | Spring | Summer | Autumn | |||||
|---|---|---|---|---|---|---|---|---|
| Outdoor | Outdoor | Outdoor | Outdoor | |||||
| Urban (n = 26) | ||||||||
| Range | 317–2,868 | 283–3,697 | 67–2,600 | 194–3,236 | ||||
| Mean | 887 | 829 | 706 | 1,500 | ||||
| Median | 763 | 516 | 511 | 1,225 | ||||
| I/O ratio a | 0.81 | 0.88 | 1.06 | 0.746 | ||||
| Rural (n = 17) | ||||||||
| Range | 449–6,323 | 3,052–16,492 | 360–3,200 | 704–2,712 | ||||
| Median | 2,028 | 7,636 | 988 | 1,509 | ||||
| Mean | 20,730 | 21,073 | 9,782 | 17,143 | ||||
| I/O ratio a | 0.84 | 1.13 | 1.49 | 1.21 | ||||
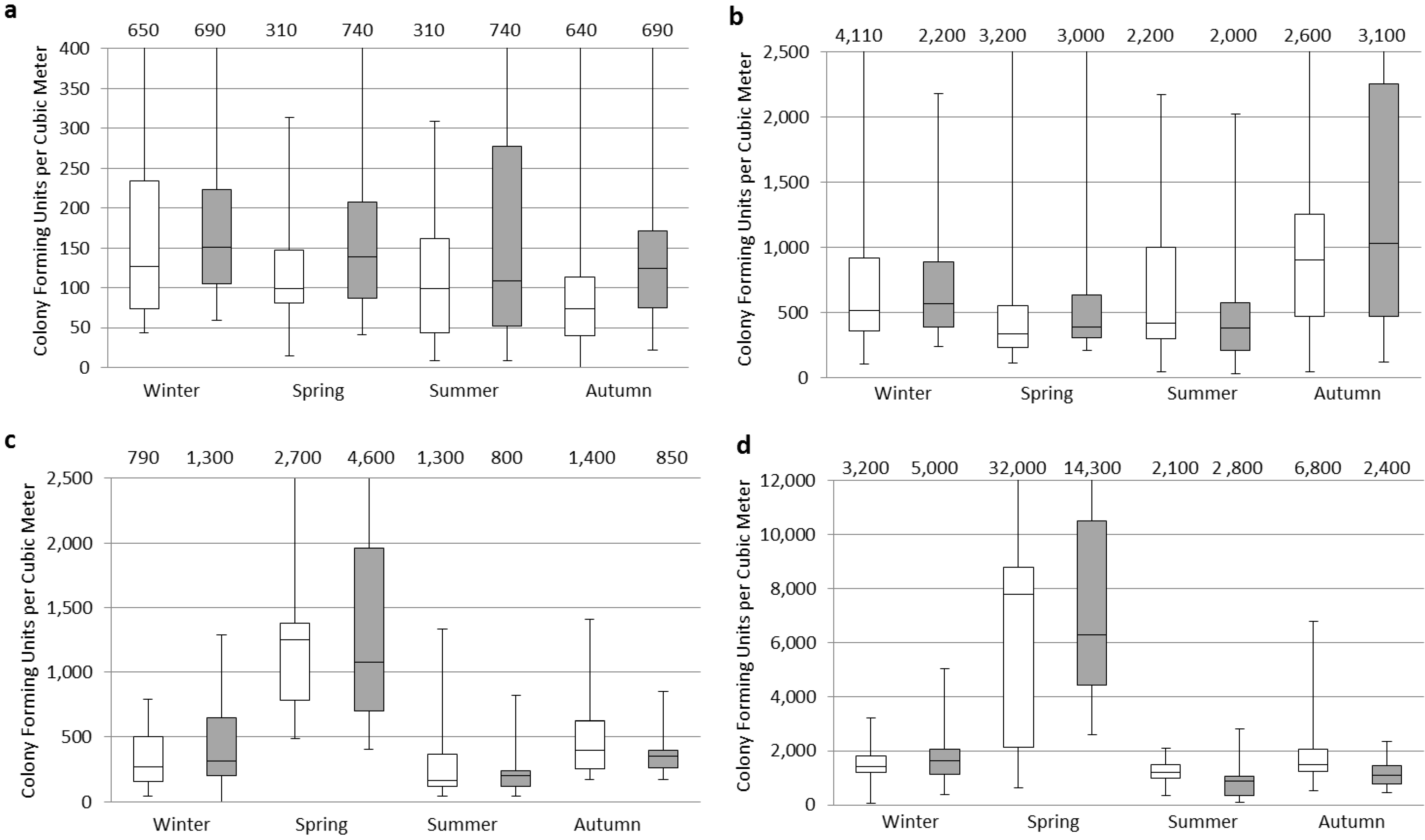
3.3. Identification of Fungi
| Urban | |||||
|---|---|---|---|---|---|
| Indoor | Outdoor | Indoor | |||
| % | % | % | |||
| Alternaria | 1.4 | 2.2 | 2.4 | ||
| Aspergillus spp. | 33.9 | 27.8 | 7.9 | ||
| Cladosporium | 28.3 | 36.3 | 67.2 | ||
| Fusarium | 1.2 | 1.0 | 2.3 | ||
| Penicillium | 27.9 | 25.6 | 12.0 | ||
| Yeasts | 2.7 | 2.3 | 2.4 | ||
| Other | 4.5 | 4.8 | 5.7 | ||
| Sum of all Samples | 100 | 100 | 100 | ||
3.4. Frequency of the Occurrence of Fungal Genera by Season
4. Discussion
| Urban | Rural | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Winter | Spring | Summer | Autumn | Winter | Spring | Summer | Autumn | |||||||||
| Out | Out | Out | Out | Out | Out | Out | Out | |||||||||
| Alternaria | 135 | 959 | 975 | 30 | 819 | 6,380 | 880 | 291 | ||||||||
| Aspergillus spp. | 6,804 | 3,860 | 7,989 | 8,136 | 3,896 | 800 | 5,104 | 3,779 | ||||||||
| Cladosporium | 65 | 7,145 | 4,150 | 19,086 | 31,689 | 277,714 | 4,148 | 11,117 | ||||||||
| Fusarium | 104 | 1,560 | 288 | 0 | 599 | 1,560 | 2,924 | 639 | ||||||||
| Penicillium | 8,340 | 6,021 | 1,750 | 8,568 | 4,805 | 45,428 | 3,176 | 4,421 | ||||||||
| Yeasts | 194 | 1,123 | 884 | 37 | 555 | 4,144 | 1,160 | 316 | ||||||||
| Other | 1,154 | 8,181 | 5,318 | 20,394 | 35,863 | 278,914 | 5,358 | 14,161 | ||||||||
| Total | 16,731 | 21,704 | 17,204 | 37,165 | 46,537 | 337,226 | 18,602 | 23,607 | ||||||||
| Winter | Spring | Summer | Autumn | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Urban | Rural | Urban | Rural | Urban | Rural | Urban | Rural | |||||||||
| Out | Out | Out | Out | Out | Out | Out | Out | |||||||||
| Alternaria | 30 | 58.8 | 65 | 82 | 61.5 | 53 | 3.8 | 29 | ||||||||
| Aspergillus spp. | 100 | 82.4 | 100 | 64.7 | 92.3 | 88.2 | 88.5 | 100 | ||||||||
| Cladosporium | 88.4 | 100 | 53.8 | 100 | 42 | 53 | 88 | 100 | ||||||||
| Fusarium | 15 | 53 | 53 | 76 | 38 | 88 | 0 | 64 | ||||||||
| Penicillium | 100 | 100 | 84 | 94 | 53 | 88 | 88 | 100 | ||||||||
| Yeasts | 15 | 47 | 69 | 100 | 50 | 64 | 2 | 41 | ||||||||
| Other | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | ||||||||
5. Conclusions
Acknowledgment
References
- Ozkara, A.; Ocak, I.; Korcan, S.; Konuk, M. Determination of fungal air spora in Afyonkarahisar, Turkey. Mycotaxon 2007, 102, 199–202. [Google Scholar]
- Mota, L.; Gibbs, S.; Green, C.; Flores, C.; Tarwater, P.; Ortiz, M. Seasonal fine and coarse culturable fungal constituents and concentrations from indoor and outdoor air samples taken from an arid environment. J. Occup. Environ. Hyg. 2008, 5, 511–518. [Google Scholar] [CrossRef]
- D’Arcy, N.; Canales, M; Spratt, D.; Lai, K. Healthy schools: Standardization of culturing methods for seeking airborne pathogens in bioaerosols emitted from human sources. Aerobiologia 2012, 28, 413–422. [Google Scholar] [CrossRef]
- Horner, W.; Worthan, A.; Morey, P. Air and dustborne mycoflora in houses free of water damage and fungal growth. Appl. Environ. Microbiol. 2004, 70, 6394–6400. [Google Scholar] [CrossRef]
- Koskinen, O.; Husman, T.; Hyvärinen, A.; Reponen, T.; Nevalainen, A. Respiratory symptoms and infections among children in a day care centre with mold problems. Indoor Air 1995, 5, 3–9. [Google Scholar]
- Meklin, T.; Husman, T.; Vepsäläinen, A.; Vahteristo, M.; Koivisto, J.; Halla-Aho, J.; Hyvärinen, A.; Moschandreas, D.; Nevalainen, A. Indoor air microbes and respiratory symptoms of children in moisture damaged and reference schools. Indoor Air 2002, 12, 175–183. [Google Scholar] [CrossRef]
- Codina, R.; Fox, R.; Lockey, R.; DeMarco, P.; Bagg, A. Typical levels of airborne fungal spores in houses without obvious moisture problems during a rainy season in Florida, USA. J. Investig. Allergol. Clin. Immunol. 2008, 18, 156–162. [Google Scholar]
- Sessa, R.; di Pietro, M.; Sehiavoni, G.; Santino, I.; Altieri, A.; Pinelli, S.; del Piano, M. Microbiological indoor air quality in healthy buildings. New Microbiol. 2002, 25, 51–56. [Google Scholar]
- Nevalainen, A.; Hyvarinen, A.; Pasanen, A.L.; Reponen, T. Fungi and bacteria in normal and mouldy dwellings. In Health Implications of Fungi in Indoor Environments; Samson, R.A., Flannigan, B., Flannigan, M.E., Verhoeff, A.P., Adan, O.C.G., Hoekstra, E.S., Eds.; Elsevier: Amsterdam, The Netherlands, 1994; Volume 2, pp. 163–168. [Google Scholar]
- Pepeljnak, S.; Segvic, M. Occurrence of fungi in air and on plants investigation of different climatic regions in Croatia. Aerobiologia 2003, 19, 11–19. [Google Scholar] [CrossRef]
- Su, H.; Wu, P.; Lin, C. Fungal exposure of children at homes and schools: A health perspective. Arch. Environ. Health 2001, 56, 144–149. [Google Scholar] [CrossRef]
- Reponen, T.; Nevalainen, A.; Jantunen, M.; Pellikka, M.; Kalliokoski, P. Normal range for indoor air bacteria and fungal spores in a subarctic climate. Indoor Air 1992, 2, 26–31. [Google Scholar]
- Krikśtaponis, A. Diversity of Fungi Species in Occupational and Residential Environments and their Biological Peculiarities (Toxicity, Pathogenicity, Proteolytic, Lipolytic and Cellulolytic Activity). Ph.D. Thesis, Institute of Botany, Vilnius University, Vilnius, Lithuania, 2000. [Google Scholar]
- Pastuszka, J.; Paw, T.; Lis, O.; Wlazlo, A.; Ulfig, K. Bacterial and fungal bioaerosol in indoor environment in Upper Silesia, Poland. Atmos. Environ. 2000, 34, 3833–3842. [Google Scholar] [CrossRef]
- Seltzer, J.M. Biologic contamination. Occup. Med. State Art Rev. 1995, 10, 1–25. [Google Scholar]
- Abdel Hameed, A.A.; Gibbs, S.G.; Tarwater, P.M.; Casillas, M.E.; Green, C.F. Seasonal evaluation of fine and coarse culturable bacterial aerosols from residences within a rural and an urban city in Egypt. Int. J. Environ. Health 2012. [Google Scholar] [CrossRef]
- Mullins, J.; Seaton, A. Fungal spores in lung and sputum. Clin. Allergy 1978, 8, 525–533. [Google Scholar] [CrossRef]
- Andersen, A. New sampler for the collection, sizing and enumeration of viable airborne particles. J. Bacteriol. 1958, 76, 471–484. [Google Scholar]
- Raper, K.B.; Fennell, D.I. The Genus Aspergillus; Krieger: New York, NY, USA, 1973. [Google Scholar]
- Pitt, J.I. The Genus Penicillium and Its Teleomorphic States Eupenicillium and Talaromyces; Academic Press: London, UK, 1979. [Google Scholar]
- Carmichael, J.W.; Kendrick, W.B.; Conners, I.L.; Sigler, L. Genera of Hyphomycetes; The University of Alberta: Edmonton, Canada, 1980. [Google Scholar]
- Barnett, H.L.; Hunter, B.B. Illustrated Genera of Imperfect Fungi, 4th ed; The American Phytopathological Society (APS): St. Paul, MN, USA, 1999; p. 218. [Google Scholar]
- Levetin, E.; Shaughnessy, R.; Fisher, E.; Ligman, B.; Harrison, J.; Brennan, T. Indoor air quality in schools; exposure to fungal allergens. Aerobiologia 1995, 11, 27–34. [Google Scholar] [CrossRef]
- Jaffal, A.; Banat, I.; El Mogheth, A. Residential indoor airborne microbial populations in the United Arab Emirates. Environ. Int. 1997, 23, 529–533. [Google Scholar] [CrossRef]
- Sen, B.; Asan, A. Fungal flora in indoor and outdoor air of different residential homes in Tekirdag City (Turkey): Seasonal distribution and relationship with climatic factors. Environ. Monit. Assess. 2009, 15, 209–219. [Google Scholar]
- Shelton, G.; Kirkland, H.; Dana Flanders, W.; Morris, K. Profiles of airborne fungi in buildings and outdoor environments in the United States. Appl. Environ. Microbiol. 2002, 68, 1743–1753. [Google Scholar] [CrossRef]
- Lee, T.; Grinshpun, S.; Martuzevicius, D.; Adhikari, A.; Crawford, C.; Reponen, T. Culturability in six single family homes. Atmos. Environ. 2006, 40, 2902–2910. [Google Scholar] [CrossRef]
- Garrett, M.; Hooper, B.; Cole, F.; Hooper, M. Airborne fungal spores in 80 homes in the Latrobe valley, Australia: Levels, seasonality and indoor-outdoor relationship. Aerobiologia 1997, 13, 121–126. [Google Scholar] [CrossRef]
- Gots, R.; Layton, N.; Pirages, S. Indoor health: Background levels of fungi. AIHA J. 2003, 64, 427–438. [Google Scholar] [CrossRef]
- Ponce-Caballero, C.; Ceron-Palma, I.; Lopez-Pacheco, M.; Gamboa-Marrufo, M.; Quintal-Franco, C. Indoor-outdoor fungal aerosols ratios of domestic homes in Merida, Mexico. Ingenierìa 2010, 14, 169–175. [Google Scholar]
- Abdulla, H.; Morshedy, H.; Dewedar, A. Characterization of actinomycetes isolated from the indoor air of the church of Saint Katherine Monastery, Egypt. Aerobiologia 2008, 24, 35–41. [Google Scholar] [CrossRef]
- El Sayed, M. Preliminary survey of indoor and outdoor airborne microfungi at coastal buildings in Egypt. Aerobiologia 2006, 22, 197–210. [Google Scholar] [CrossRef]
- Dutkiewicz, J.; Gôrny, R. Biological factors hazardous to human health: Classification and criteria of exposure assessment (in Polish). Med. Pr. 2002, 53, 29–39. [Google Scholar]
- Rao, C.; Burge, H.; Chang, J. Review of quantitative standards and guidelines for fungi in indoor air. J. Air Waste Manag. Assoc. 1996, 46, 899–908. [Google Scholar] [CrossRef]
- WHO Regional Publications European Series No 31: Indoor Air Quality: Biological Contaminants; World Health Organization: Copenhagen, Denmark, 1988.
- Singapore-Ministry of Environment. Guidelines for Good Indoor Air Quality in Office Premises. 1996. Available online: http://www.nea.gov.sg/cms/qed/indoor_air.pdf (accessed on 10 February 2012).
- Guidelines for the Assessment about Aerosols in the Indoor Environment; American Conference of Governmental Industrial Hygienists: OH, USA, 1989.
- Li, Ch.; Kuo, Y. Microbiological indoor air quality in subtropical areas. Environ. Int. 1993, 19, 233–239. [Google Scholar] [CrossRef]
- Wu, P-C.; Su, H-J.; Lin, C-Y. Characteristic of indoor and outdoor airborne fungi at suburban and urban homes in two seasons. Sci. Total Environ. 2000, 253, 111–118. [Google Scholar] [CrossRef]
- Abdel Wahid, O.; Moustafa, A.; Moustafa, A. Fungal population in the atmosphere of Ismailia city. Aerobiologia 1996, 12, 249–255. [Google Scholar]
- Dacarro, C.; Picco, M.; Grisoli, P.; Rodolfi, M. Determination of aerial microbiological contamination in scholastic sports environments. J. Appl. Microbiol. 2003, 95, 904–912. [Google Scholar] [CrossRef]
- Abdel Hameed, A.; El Hawarry, S.; Kamel, M. Prevalence and distribution of airborne and waterborne fungi and actinomycetes in the Nile River. Aerobiologia 2008, 24, 231–240. [Google Scholar] [CrossRef]
- Chao, H.; Milton, D.; Schwartz, J.; Burge, H. Dustborne fungi in large office buildings. Mycopathologia 2001, 154, 93–106. [Google Scholar]
- Chakraborty, S.; Sen, S.; Bhattacharya, K. Indoor and outdoor aeromycological survey in Burdwan, West Bengal, India. Aerobiologia 2000, 16, 211–219. [Google Scholar] [CrossRef]
- Dubey, S.; Lanjewar, S.; Sahu, M.; Pandey, K.; Kutti, U. The monitoring of filamentous fungi in the indoor, air quality and health. J. Phytol. 2011, 3, 13–14. [Google Scholar]
- Hargreaves, M.; Parappukkaran, S.; Morawska, L.; Hitchins, J.; He, C.; Gilbert, D. Pilot investigation into associations between indoor airborne fungal and non biological particle concentrations in residential houses in Brisbane, Australia. Sci. Total Environ. 2003, 312, 89–101. [Google Scholar] [CrossRef]
- Lacey, J.; Dutkiewicz, J. Bioaerosols and occupational lung disease. J. AerosolSci. 1994, 25, 1371–1404. [Google Scholar] [CrossRef]
- Davis, P.J. Molds, Toxic Molds, and Indoor Air Quality. California Research Bureau (CRB), California State Library: Sacramento, CA, USA. Available online: http://www.library.ca.gov/crb/01/notes/v8n1.pdf (accessed on 10 February 2012).
- Abarca, M. Taxonomy and identification of the species involved in nosocomial aspergillosis. Rev. Iberoam. Micol. 2000, 17, 79–84. [Google Scholar]
- Lugauskas, A.; Krikstaponis, A.; Seskauskas, V. Species of conditionally pathogenic micromycetes in the air of dwellings and occupational premises. Indoor Built Environ. 2003, 12, 167–177. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Awad, A.H.A.; Gibbs, S.G.; Tarwater, P.M.; Green, C.F. Coarse and Fine Culturable Fungal Air Concentrations in Urban and Rural Homes in Egypt. Int. J. Environ. Res. Public Health 2013, 10, 936-949. https://doi.org/10.3390/ijerph10030936
Awad AHA, Gibbs SG, Tarwater PM, Green CF. Coarse and Fine Culturable Fungal Air Concentrations in Urban and Rural Homes in Egypt. International Journal of Environmental Research and Public Health. 2013; 10(3):936-949. https://doi.org/10.3390/ijerph10030936
Chicago/Turabian StyleAwad, Abdel Hameed A., Shawn G. Gibbs, Patrick M. Tarwater, and Christopher F. Green. 2013. "Coarse and Fine Culturable Fungal Air Concentrations in Urban and Rural Homes in Egypt" International Journal of Environmental Research and Public Health 10, no. 3: 936-949. https://doi.org/10.3390/ijerph10030936
APA StyleAwad, A. H. A., Gibbs, S. G., Tarwater, P. M., & Green, C. F. (2013). Coarse and Fine Culturable Fungal Air Concentrations in Urban and Rural Homes in Egypt. International Journal of Environmental Research and Public Health, 10(3), 936-949. https://doi.org/10.3390/ijerph10030936




