Comprehensive Assessment of Herbicide Toxicity on Navicula sp. Algae: Effects on Growth, Chlorophyll Content, Antioxidant System, and Lipid Metabolism
Abstract
1. Introduction
2. Results
2.1. Effect of Herbicides on the Growth Density of Navicula
2.2. Effect of Herbicides on the Chlorophyll Content of Navicula
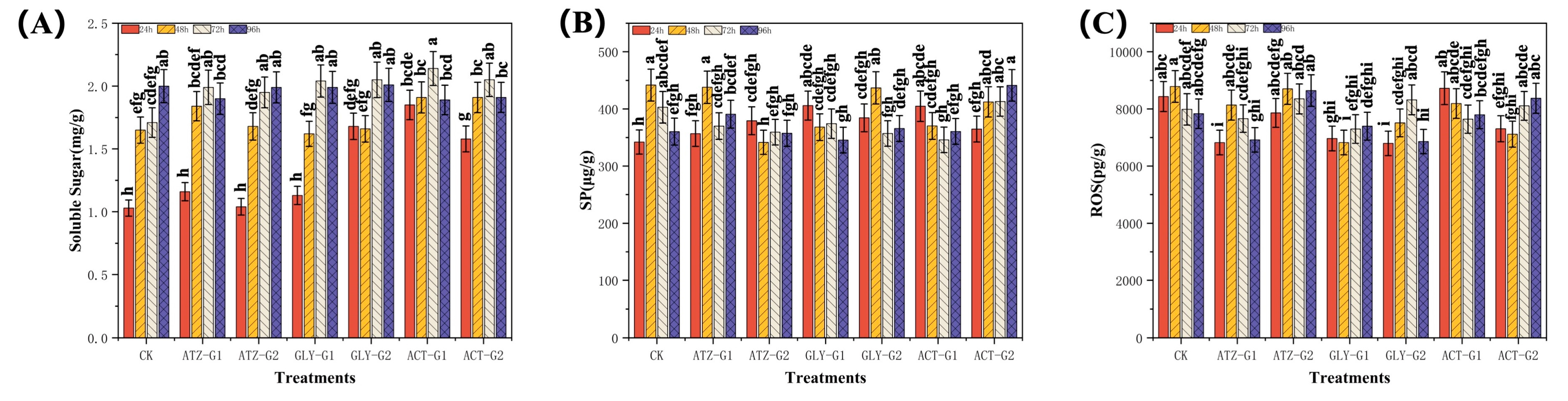
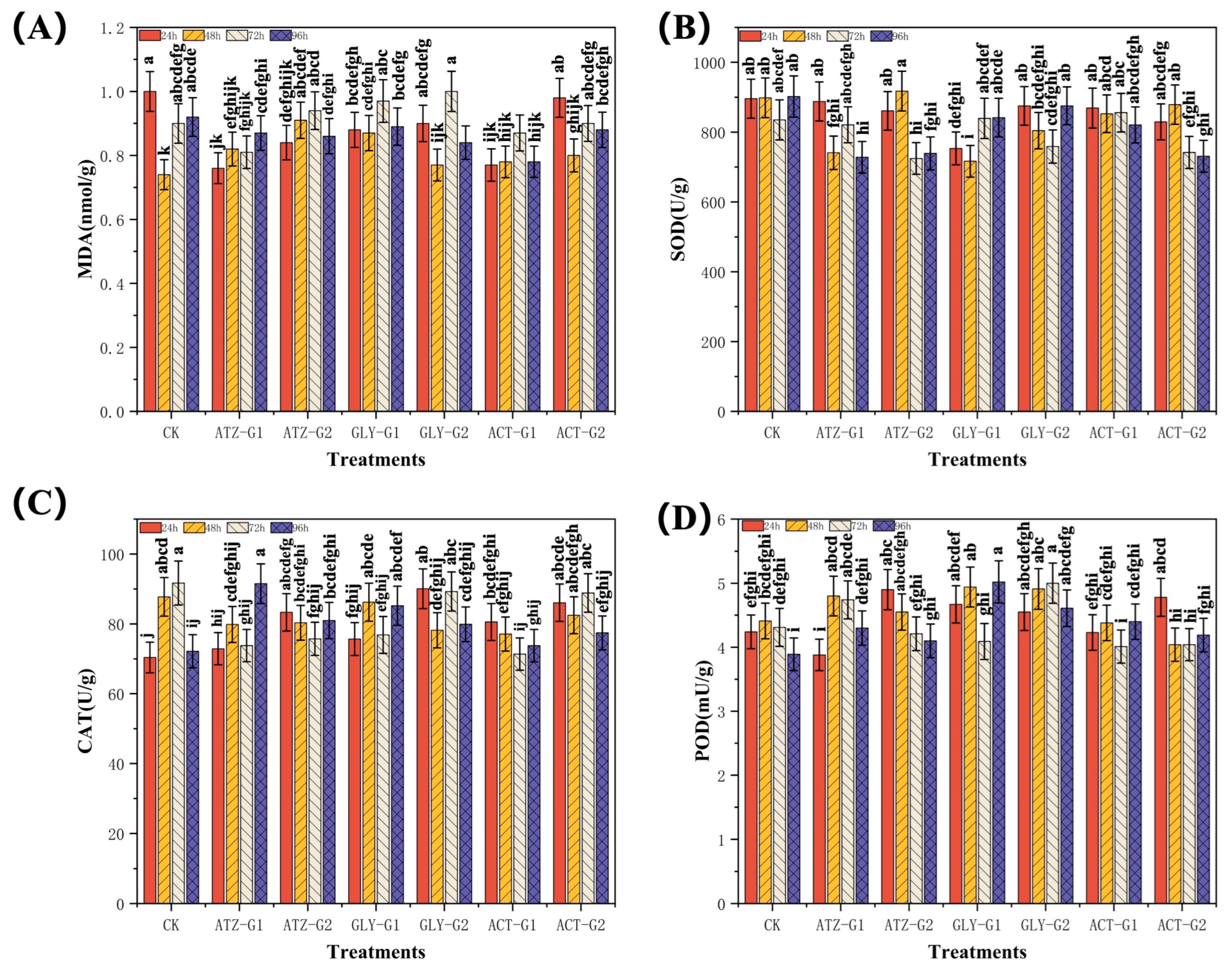
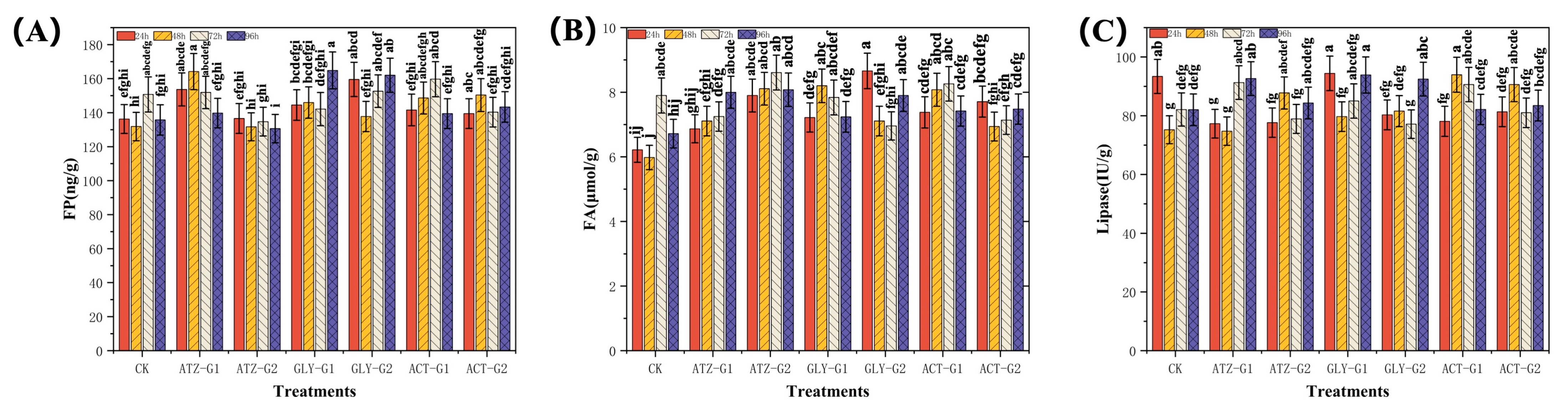
2.3. Effect of Herbicides on the Antioxidant Activities of Navicula
2.4. Effect of Herbicides on the Lipid Composition of Navicula
3. Discussion
4. Materials and Methods
4.1. Chemicals and Reagents
4.2. Algae Growth and Cultivation
4.3. Algal Toxicity
4.4. Measurement of Cell Density
4.5. Quantification of Pigment Concentration
4.6. Assessment of the Antioxidative Activities
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| Serial Number | Abbreviation | Details |
| 1 | ATZ | atrazine |
| 2 | ACT | acetochlor |
| 3 | Gly | glyphosate |
| 4 | EPA | Environmental Protection Agency |
| 5 | EC | European Commission |
| 6 | Chlla | chlorophyll a |
| 7 | Chllb | chlorophyll b |
| 8 | SS | soluble sugar |
| 9 | SP | soluble protein |
| 10 | ROS | reactive oxygen species |
| 11 | MDA | malondialdehyde |
| 12 | SOD | superoxide dismutase |
| 13 | CAT | catalase |
| 14 | POD | peroxidase |
| 15 | FP | free proline |
| 16 | FA | fatty acid |
| 17 | SFAs | saturated fatty acids |
| 18 | MUFAs | monounsaturated fatty acids |
| 19 | PUFAs | polyunsaturated fatty acids |
References
- Chen, X.W.; Chen, H.; Zhao, H.L.; Li, D.W.; Ou, L.J. Triazine Herbicide Reduced the Toxicity of the Harmful Dinoflagellate Karenia Mikimotoi by Impairing Its Photosynthetic Systems. Ecotoxicol. Environ. Saf. 2024, 269, 115740. [Google Scholar] [CrossRef]
- DeLorenzo, M.E.; Leatherbury, M.; Weiner, J.A.; Lewitus, A.J.; Fulton, M.H. Physiological Factors Contributing to the Species-Specific Sensitivity of Four Estuarine Microalgal Species Exposed to the Herbicide Atrazine. Aquat. Ecosyst. Health Manag. 2004, 7, 137–146. [Google Scholar] [CrossRef]
- Majewska, M.; Harshkova, D.; Pokora, W.; Baścik-Remisiewicz, A.; Tułodziecki, S.; Aksmann, A. Does Diclofenac Act like a Photosynthetic Herbicide on Green Algae? Chlamydomonas Reinhardtii Synchronous Culture-Based Study with Atrazine as Reference. Ecotoxicol. Environ. Saf. 2021, 208, 111630. [Google Scholar] [CrossRef] [PubMed]
- Pinheiro, R.A.; Duque, T.S.; Barroso, G.M.; Soares, M.A.; Cabral, C.M.; Zanuncio, J.C.; dos Santos, J.B. Herbicides May Threaten Advances in Biological Control of Diseases and Pests. Environ. Sci. Pollut. Res. 2023, 30, 111850–111870. [Google Scholar] [CrossRef] [PubMed]
- Fingler, S.; Mendaš, G.; Dvoršćak, M.; Stipičević, S.; Vasilić; Drevenkar, V. Herbicide Micropollutants in Surface, Ground and Drinking Waters within and near the Area of Zagreb, Croatia. Environ. Sci. Pollut. Res. 2017, 24, 11017–11030. [Google Scholar] [CrossRef] [PubMed]
- Rumschlag, S.L.; Casamatta, D.A.; Mahon, M.B.; Hoverman, J.T.; Raffel, T.R.; Carrick, H.J.; Hudson, P.J.; Rohr, J.R. Pesticides Alter Ecosystem Respiration via Phytoplankton Abundance and Community Structure: Effects on the Carbon Cycle? Glob. Chang. Biol. 2022, 28, 1091–1102. [Google Scholar] [CrossRef]
- Liu, H.; Wang, X.; Ou, Y.; Cheng, L.; Hou, X.; Yan, L.; Tian, L. Characterization of Acetochlor Degradation and Role of Microbial Communities in Biofilters with Varied Substrate Types. Chem. Eng. J. 2023, 467, 143417. [Google Scholar] [CrossRef]
- Kapsi, M.; Tsoutsi, C.; Paschalidou, A.; Albanis, T. Environmental Monitoring and Risk Assessment of Pesticide Residues in Surface Waters of the Louros River (N.W. Greece). Sci. Total Environ. 2019, 650, 2188–2198. [Google Scholar] [CrossRef]
- Rohr, J.R. The Atrazine Saga and Its Importance to the Future of Toxicology, Science, and Environmental and Human Health. Environ. Toxicol. Chem. 2021, 40, 1544–1558. [Google Scholar] [CrossRef]
- de Souza, V.V.; Souza, T.d.S.; de Campos, J.M.S.; de Oliveira, L.A.; Ribeiro, Y.M.; Hoyos, D.C.d.M.; Xavier, R.M.P.; Charlie-Silva, I.; Lacerda, S.M.d.S.N. Ecogenotoxicity of Environmentally Relevant Atrazine Concentrations: A Threat to Aquatic Bioindicators. Pestic. Biochem. Physiol. 2023, 189, 105297. [Google Scholar] [CrossRef]
- Das, S.; Sakr, H.; Al-Huseini, I.; Jetti, R.; Al-Qasmi, S.; Sugavasi, R.; Sirasanagandla, S.R. Atrazine Toxicity: The Possible Role of Natural Products for Effective Treatment. Plants 2023, 12, 2278. [Google Scholar] [CrossRef] [PubMed]
- Graymore, M.; Stagnitti, F.; Allinson, G. Impacts of Atrazine in Aquatic Ecosystems. Environ. Int. 2001, 26, 483–495. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.; Xie, F.; Guo, Q.N.; Yang, H. Environmental Disappearance of Acetochlor and Its Bioavailability to Weed: A General Prototype for Reduced Herbicide Application Instruction. Chemosphere 2021, 265, 129108. [Google Scholar] [CrossRef]
- Li, H.; Feng, Y.; Li, X.; Zeng, D. Analytical Confirmation of Various Herbicides in Drinking Water Resources in Sugarcane Production Regions of Guangxi, China. Bull. Environ. Contam. Toxicol. 2018, 100, 815–820. [Google Scholar] [CrossRef]
- Wang, H.; Meng, Z.; Zhou, L.; Cao, Z.; Liao, X.; Ye, R.; Lu, H. Effects of Acetochlor on Neurogenesis and Behaviour in Zebrafish at Early Developmental Stages. Chemosphere 2019, 220, 954–964. [Google Scholar] [CrossRef]
- Pollegioni, L.; Schonbrunn, E.; Siehl, D. Molecular Basis of Glyphosate Resistance—Different Approaches through Protein Engineering. FEBS J. 2011, 278, 2753–2766. [Google Scholar] [CrossRef] [PubMed]
- Duke, S.O.; Powles, S.B. Glyphosate: A Once-in-a-Century Herbicide. Pest Manag. Sci. 2008, 66, 270–276. [Google Scholar] [CrossRef]
- Lu, T.; Xu, N.; Zhang, Q.; Zhang, Z.; Debognies, A.; Zhou, Z.; Sun, L.; Qian, H. Understanding the Influence of Glyphosate on the Structure and Function of Freshwater Microbial Community in a Microcosm. Environ. Pollut. 2020, 260, 114012. [Google Scholar] [CrossRef]
- Lupi, L.; Miglioranza, K.S.B.; Aparicio, V.C.; Marino, D.; Bedmar, F.; Wunderlin, D.A. Occurrence of Glyphosate and AMPA in an Agricultural Watershed from the Southeastern Region of Argentina. Sci. Total Environ. 2015, 536, 687–694. [Google Scholar] [CrossRef]
- Falciatore, A.; Jaubert, M.; Bouly, J.P.; Bailleul, B.; Mock, T. Diatom Molecular Research Comes of Age: Model Species for Studying Phytoplankton Biology and Diversity. Plant Cell 2020, 32, 547–572. [Google Scholar] [CrossRef]
- Lopez, P.J.; Desclés, J.; Allen, A.E.; Bowler, C. Prospects in Diatom Research. Curr. Opin. Biotechnol. 2005, 16, 180–186. [Google Scholar] [CrossRef] [PubMed]
- Dalu, T.; Froneman, P.W. Diatom-Based Water Quality Monitoring in Southern Africa: Challenges and Future Prospects. Water SA 2016, 42, 551. [Google Scholar] [CrossRef]
- Whitton, B.A.; Ellwood, N.T.W.; Kawecka, B. Biology of the Freshwater Diatom Didymosphenia. Hydrobiologia 2009, 630, 1–37. [Google Scholar] [CrossRef]
- Poulin, M.; Cardinal, A. Sea Ice Diatoms from Manitounuk Sound, Southeastern Hudson Bay (Quebec, Canada). II. Naviculaceae, Genus Navicula. Can. J. Bot. 1982, 60, 2825–2845. [Google Scholar] [CrossRef]
- Ding, T.; Wang, S.; Yang, B.; Li, J. Biological Removal of Pharmaceuticals by Navicula sp. and Biotransformation of Bezafibrate. Chemosphere 2020, 240, 124949. [Google Scholar] [CrossRef]
- Corrales, N.; Meerhoff, M.; Antoniades, D. Glyphosate-Based Herbicide Exposure Affects Diatom Community Development in Natural Biofilms. Environ. Pollut. 2021, 284, 117354. [Google Scholar] [CrossRef]
- Zhang, Z.; Chen, Q.; Chen, B.; Dong, T.; Chen, M. Toxic Effects of Pesticides on the Marine Microalga Skeletonema Costatum and Their Biological Degradation. Sci. China Earth Sci. 2023, 66, 663–674. [Google Scholar] [CrossRef]
- Rumschlag, S.L.; Mahon, M.B.; Hoverman, J.T.; Raffel, T.R.; Carrick, H.J.; Hudson, P.J.; Rohr, J.R. Consistent Effects of Pesticides on Community Structure and Ecosystem Function in Freshwater Systems. Nat. Commun. 2020, 11, 6333. [Google Scholar] [CrossRef] [PubMed]
- Valiente Moro, C.; Bricheux, G.; Portelli, C.; Bohatier, J. Comparative Effects of the Herbicides Chlortoluron and Mesotrione on Freshwater Microalgae. Environ. Toxicol. Chem. 2012, 31, 778–786. [Google Scholar] [CrossRef]
- Gómez-Martínez, D.; Bengtson, J.; Nilsson, A.K.; Clarke, A.K.; Nilsson, R.H.; Kristiansson, E.; Corcoll, N. Phenotypic and Transcriptomic Acclimation of the Green Microalga Raphidocelis Subcapitata to High Environmental Levels of the Herbicide Diflufenican. Sci. Total Environ. 2023, 875, 162604. [Google Scholar] [CrossRef]
- Nunes, B.W.; Hedlund, K.F.; Oliveira, M.A.; Carissimi, E. Ecotoxicological Analysis of Acrylamide Using a Microalga as an Indicator Organism. Water Environ. Res. 2018, 90, 442–451. [Google Scholar] [CrossRef] [PubMed]
- Duttagupta, S.; Mukherjee, A.; Bhattacharya, A.; Bhattacharya, J. Wide Exposure of Persistent Organic Pollutants (PoPs) in Natural Waters and Sediments of the Densely Populated Western Bengal Basin, India. Sci. Total Environ. 2020, 717, 137187. [Google Scholar] [CrossRef] [PubMed]
- Ferreira Mendes, K.; Paula Justiniano Régo, A.; Takeshita, V.; Luiz Tornisielo, V. Water Resource Pollution by Herbicide Residues. In Biochemical Toxicology—Heavy Metals and Nanomaterials; Intech Open: London, UK, 2020. [Google Scholar] [CrossRef]
- Thomas, M.C.; Flores, F.; Kaserzon, S.; Fisher, R.; Negri, A.P. Toxicity of Ten Herbicides to the Tropical Marine Microalgae Rhodomonas Salina. Sci. Rep. 2020, 10, 7612. [Google Scholar] [CrossRef]
- Roubeix, V.; Mazzella, N.; Schouler, L.; Fauvelle, V.; Morin, S.; Coste, M.; Delmas, F.; Margoum, C. Variations of Periphytic Diatom Sensitivity to the Herbicide Diuron and Relation to Species Distribution in a Contamination Gradient: Implications for Biomonitoring. J. Environ. Monit. 2011, 3, 1768–1774. [Google Scholar] [CrossRef]
- Dupraz, V.; Coquillé, N.; Ménard, D.; Sussarellu, R.; Haugarreau, L.; Stachowski-Haberkorn, S. Microalgal Sensitivity Varies between a Diuron-Resistant Strain and Two Wild Strains When Exposed to Diuron and Irgarol, Alone and in Mixtures. Chemosphere 2016, 151, 241–252. [Google Scholar] [CrossRef] [PubMed]
- Bai, X.; Sun, C.; Xie, J.; Song, H.; Zhu, Q.; Su, Y.; Qian, H.; Fu, Z. Effects of Atrazine on Photosynthesis and Defense Response and the Underlying Mechanisms in Phaeodactylum Tricornutum. Environ. Sci. Pollut. Res. 2015, 22, 17499–17507. [Google Scholar] [CrossRef]
- Kabra, A.N.; Ji, M.K.; Choi, J.; Kim, J.R.; Govindwar, S.P.; Jeon, B.H. Toxicity of Atrazine and Its Bioaccumulation and Biodegradation in a Green Microalga, Chlamydomonas Mexicana. Environ. Sci. Pollut. Res. 2014, 21, 12270–12278. [Google Scholar] [CrossRef]
- Annett, R.; Habibi, H.R.; Hontela, A. Impact of Glyphosate and Glyphosate-Based Herbicides on the Freshwater Environment. J. Appl. Toxicol. 2014, 34, 458–479. [Google Scholar] [CrossRef]
- Schaffer, J.D.; Sebetich, M.J. Effects of Aquatic Herbicides on Primary Productivity of Phytoplankton in the Laboratory. Bull. Environ. Contam. Toxicol. 2004, 72, 1032–1037. [Google Scholar] [CrossRef]
- Vera, M.S.; Lagomarsino, L.; Sylvester, M.; Pérez, G.L.; Rodríguez, P.; Mugni, H.; Sinistro, R.; Ferraro, M.; Bonetto, C.; Zagarese, H.; et al. New Evidences of Roundup® (Glyphosate Formulation) Impact on the Periphyton Community and the Water Quality of Freshwater Ecosystems. Ecotoxicology 2010, 19, 710–721. [Google Scholar] [CrossRef]
- Romero, D.M.; Ríos de Molina, M.C.; Juárez, Á.B. Oxidative Stress Induced by a Commercial Glyphosate Formulation in a Tolerant Strain of Chlorella Kessleri. Ecotoxicol. Environ. Saf. 2011, 74, 741–747. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Xu, E.G.; Ren, Y.; Jin, S.; Zhang, T.; Liu, J.; Li, Z. Mixture Toxicity of Bensulfuron-Methyl and Acetochlor to Red Swamp Crayfish (Procambarus Clarkii): Behavioral, Morphological and Histological Effects. Int. J. Environ. Res. Public Health 2017, 14, 1466. [Google Scholar] [CrossRef]
- Hennig, T.B.; Bandeira, F.O.; Puerari, R.C.; Fraceto, L.F.; Matias, W.G. A Systematic Review of the Toxic Effects of a Nanopesticide on Non-Target Organisms: Estimation of Protective Concentrations Using a Species Sensitivity Distribution (SSD) Approach—The Case of Atrazine. Sci. Total Environ. 2023, 871, 162094. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Calabrese, E.J.; Zhang, J.; Gao, D.; Qin, M.; Lin, Z. A Trigger Mechanism of Herbicides to Phytoplankton Blooms: From the Standpoint of Hormesis Involving Cytochrome B559, Reactive Oxygen Species and Nitric Oxide. Water Res. 2020, 173, 115584. [Google Scholar] [CrossRef] [PubMed]
- Pereira, A.G.; Otero, P.; Echave, J.; Carreira-Casais, A.; Chamorro, F.; Collazo, N.; Jaboui, A.; Lourenço-Lopes, C.; Simal-Gandara, J.; Prieto, M.A. Xanthophylls from the Sea: Algae as Source of Bioactive Carotenoids. Mar. Drugs 2021, 19, 188. [Google Scholar] [CrossRef]
- Podbielska, M.; Szpyrka, E. Microplastics—An Emerging Contaminants for Algae. Critical Review and Perspectives. Sci. Total Environ. 2023, 885, 163842. [Google Scholar] [CrossRef]
- Warne, M.S.J.; Neelamraju, C.; Strauss, J.; Turner, R.D.R.; Smith, R.A.; Mann, R.M. Estimating the Aquatic Risk from Exposure to up to Twenty-Two Pesticide Active Ingredients in Waterways Discharging to the Great Barrier Reef. Sci. Total Environ. 2023, 892, 164632. [Google Scholar] [CrossRef]
- Singh, M.; Ramoeta, P. A Bond Energy Theory to Analysis the Melting Temperature and Debye Temperature of Nanoscale Solids. Int. J. Eng. Technol. 2018, 7, 3014–3017. [Google Scholar]
- Yu, H.; Peng, J.; Cao, X.; Wang, Y.; Zhang, Z.; Xu, Y.; Qi, W. Effects of Microplastics and Glyphosate on Growth Rate, Morphological Plasticity, Photosynthesis, and Oxidative Stress in the Aquatic Species Salvinia Cucullata. Environ. Pollut. 2021, 279, 116900. [Google Scholar] [CrossRef]
- Kaeoboon, S.; Suksungworn, R.; Sanevas, N. Toxicity Response of Chlorella Microalgae to Glyphosate Herbicide Exposure Based on Biomass, Pigment Contents and Photosynthetic Efficiency. Plant Sci. Today 2021, 8, 293–300. [Google Scholar] [CrossRef]
- Xiong, Q.; Liu, Y.S.; Hu, L.X.; Shi, Z.Q.; Ying, G.G. Levofloxacin and Sulfamethoxazole Induced Alterations of Biomolecules in Pseudokirchneriella Subcapitata. Chemosphere 2020, 253, 126722. [Google Scholar] [CrossRef] [PubMed]
- Rezayian, M.; Niknam, V.; Ebrahimzadeh, H. Oxidative Damage and Antioxidative System in Algae. Toxicol. Rep. 2019, 6, 1309–1313. [Google Scholar] [CrossRef]
- Morris, J.J.; Rose, A.L.; Lu, Z. Reactive Oxygen Species in the World Ocean and Their Impacts on Marine Ecosystems. Redox Biol. 2022, 52, 102285. [Google Scholar] [CrossRef]
- Wei, L.; Li, H.; Lu, J. Algae-Induced Photodegradation of Antibiotics: A Review. Environ. Pollut. 2021, 272, 115589. [Google Scholar] [CrossRef] [PubMed]
- Tajvidi, E.; Nahavandizadeh, N.; Pournaderi, M.; Pourrashid, A.Z.; Bossaghzadeh, F.; Khoshnood, Z. Study the Antioxidant Effects of Blue-Green Algae Spirulina Extract on ROS and MDA Production in Human Lung Cancer Cells. Biochem. Biophys. Rep. 2021, 28, 101139. [Google Scholar] [CrossRef] [PubMed]
- Gu, P.; Li, Q.; Zhang, W.; Gao, Y.; Sun, K.; Zhou, L.; Zheng, Z. Biological Toxicity of Fresh and Rotten Algae on Freshwater Fish: LC50, Organ Damage and Antioxidant Response. J. Hazard. Mater. 2021, 407, 124620. [Google Scholar] [CrossRef]
- Jiao, Q.; Mu, Y.; Deng, J.; Yao, X.; Zhao, X.; Liu, X.; Li, X.; Jiang, X.; Zhang, F. Direct Toxicity of the Herbicide Florasulam against Chlorella Vulgaris: An Integrated Physiological and Metabolomic Analysis. Ecotoxicol. Environ. Saf. 2022, 246, 114135. [Google Scholar] [CrossRef] [PubMed]
- Marella, T.K.; Parine, N.R.; Tiwari, A. Potential of Diatom Consortium Developed by Nutrient Enrichment for Biodiesel Production and Simultaneous Nutrient Removal from Waste Water. Saudi J. Biol. Sci. 2018, 25, 704–709. [Google Scholar] [CrossRef]
- Maeda, Y.; Nojima, D.; Yoshino, T.; Tanaka, T. Structure and Properties of Oil Bodies in Diatoms. Philos. Trans. R. Soc. B Biol. Sci. 2017, 372, 20160408. [Google Scholar] [CrossRef]
- Smirnoff, N.; Cumbes, Q.J. Hydroxyl Radical Scavenging Activity of Compatible Solutes. Phytochemistry 1989, 28, 1057–1060. [Google Scholar] [CrossRef]
- Ashraf, M.; Foolad, M.R. Roles of Glycine Betaine and Proline in Improving Plant Abiotic Stress Resistance. Environ. Exp. Bot. 2007, 9, 206–216. [Google Scholar] [CrossRef]
- Sajjadi, B.; Chen, W.Y.; Raman, A.A.A.; Ibrahim, S. Microalgae Lipid and Biomass for Biofuel Production: A Comprehensive Review on Lipid Enhancement Strategies and Their Effects on Fatty Acid Composition. Renew. Sustain. Energy Rev. 2018, 97, 200–232. [Google Scholar] [CrossRef]
- Burdge, G.C.; Calder, P.C. Introduction to Fatty Acids and Lipids. World Rev. Nutr. Diet. 2015, 112, 1–16. [Google Scholar]
- Maltsev, Y.; Maltseva, K. Fatty Acids of Microalgae: Diversity and Applications. Rev. Environ. Sci. Biotechnol. 2021, 20, 515–547. [Google Scholar] [CrossRef]
- Li-Beisson, Y.; Thelen, J.J.; Fedosejevs, E.; Harwood, J.L. The Lipid Biochemistry of Eukaryotic Algae. Prog. Lipid Res. 2019, 74, 31–68. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.J.; Wu, K.C.; Chan, S.C.Y.; Yau, Y.H.; Chan, K.K.; Lee, F.W.F. Investigation of Growth, Lipid Productivity, and Fatty Acid Profiles in Marine Bloom-Forming Dinoflagellates as Potential Feedstock for Biodiesel. J. Mar. Sci. Eng. 2020, 8, 381. [Google Scholar] [CrossRef]
- Ben Mordechay, E.; Mordehay, V.; Tarchitzky, J.; Chefetz, B. Pharmaceuticals in Edible Crops Irrigated with Reclaimed Wastewater: Evidence from a Large Survey in Israel. J. Hazard. Mater. 2021, 416, 126184. [Google Scholar] [CrossRef] [PubMed]
- Du, J.; Izquierdo, D.; Xu, H.F.; Beisner, B.; Lavaud, J.; Ohlund, L.; Sleno, L.; Juneau, P. Responses to Herbicides of Arctic and Temperate Microalgae Grown under Different Light Intensities. Environ. Pollut. 2023, 333, 121985. [Google Scholar] [CrossRef]
- Lin, W.; Zhang, Z.; Chen, Y.; Zhang, Q.; Ke, M.; Lu, T.; Qian, H. The Mechanism of Different Cyanobacterial Responses to Glyphosate. J. Environ. Sci. 2023, 125, 258–265. [Google Scholar] [CrossRef]
- Yang, J.; Ahmed, W.; Mehmood, S.; Ou, W.; Li, J.; Xu, W.; Wang, L.; Mahmood, M.; Li, W. Evaluating the Combined Effects of Erythromycin and Levofloxacin on the Growth of Navicula sp. and Understanding the Underlying Mechanisms. Plants 2023, 12, 2547. [Google Scholar] [CrossRef]
- Zhang, S.; Wu, X.; Cui, J.; Zhang, F.; Wan, X.; Liu, Q.; Zhong, Y.; Lin, T. Physiological and Transcriptomic Analysis of Yellow Leaf Coloration in Populus Deltoides Marsh. PLoS ONE 2019, 14, e0216879. [Google Scholar] [CrossRef] [PubMed]


| Compound | Molecular Formula | Molecular Structure | Molecular Weight |
|---|---|---|---|
| Atrazine | C8H14CIN5 | 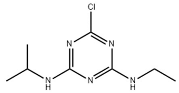 | 215.68 |
| Glyphosate | C3H8NO5P |  | 169.07 |
| Acetochlor | C14H20CINO2 |  | 269.77 |
| Serial Number | Treatments | Description |
|---|---|---|
| 1 | CK | Control |
| 2 | ATZ-G1 | Atrazine 100 µg L−1 |
| 3 | ATZ-G2 | Atrazine 300 µg L−1 |
| 4 | Gly-G1 | Glyphosate 100 µg L−1 |
| 5 | Gly-G2 | Glyphosate 300 µg L−1 |
| 6 | ACT-G1 | Acetochlor 100 µg L−1 |
| 7 | ACT-G2 | Acetochlor 300 µg L−1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zheng, C.; Yang, J.; Wang, Y.; Ahmed, W.; Khan, A.; Li, J.; Weng, J.; Mehmood, S.; Li, W. Comprehensive Assessment of Herbicide Toxicity on Navicula sp. Algae: Effects on Growth, Chlorophyll Content, Antioxidant System, and Lipid Metabolism. Mar. Drugs 2024, 22, 387. https://doi.org/10.3390/md22090387
Zheng C, Yang J, Wang Y, Ahmed W, Khan A, Li J, Weng J, Mehmood S, Li W. Comprehensive Assessment of Herbicide Toxicity on Navicula sp. Algae: Effects on Growth, Chlorophyll Content, Antioxidant System, and Lipid Metabolism. Marine Drugs. 2024; 22(9):387. https://doi.org/10.3390/md22090387
Chicago/Turabian StyleZheng, Chunyan, Jie Yang, Yunting Wang, Waqas Ahmed, Amir Khan, Jiannan Li, Jiechang Weng, Sajid Mehmood, and Weidong Li. 2024. "Comprehensive Assessment of Herbicide Toxicity on Navicula sp. Algae: Effects on Growth, Chlorophyll Content, Antioxidant System, and Lipid Metabolism" Marine Drugs 22, no. 9: 387. https://doi.org/10.3390/md22090387
APA StyleZheng, C., Yang, J., Wang, Y., Ahmed, W., Khan, A., Li, J., Weng, J., Mehmood, S., & Li, W. (2024). Comprehensive Assessment of Herbicide Toxicity on Navicula sp. Algae: Effects on Growth, Chlorophyll Content, Antioxidant System, and Lipid Metabolism. Marine Drugs, 22(9), 387. https://doi.org/10.3390/md22090387








