Effects of Ingesting Fucoidan Derived from Cladosiphon okamuranus Tokida on Human NK Cells: A Randomized, Double-Blind, Parallel-Group, Placebo-Controlled Pilot Study
Abstract
1. Introduction
2. Results
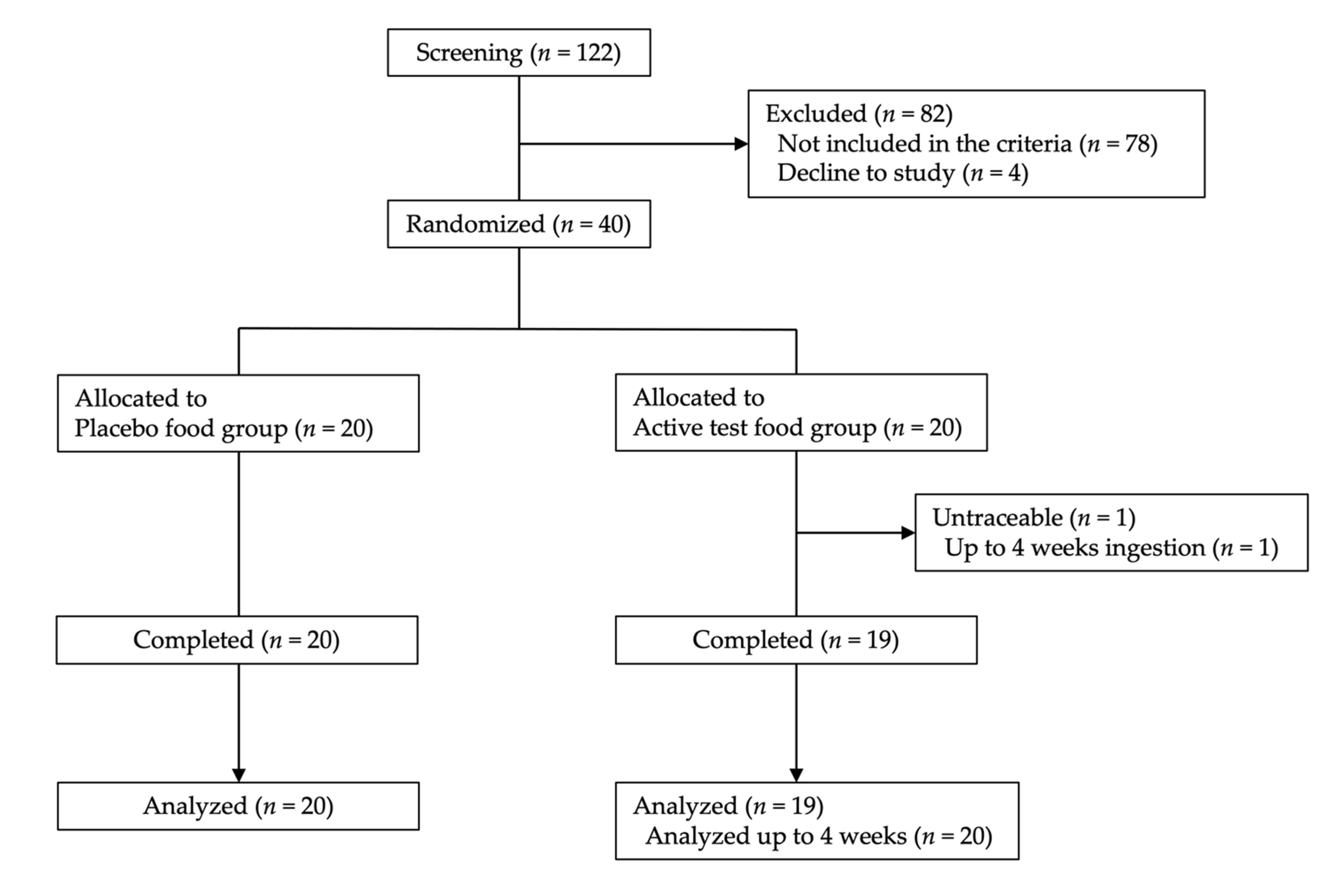
2.1. Subject Background and Test Food Ingestion Rate
2.2. Immnologycal Assessment
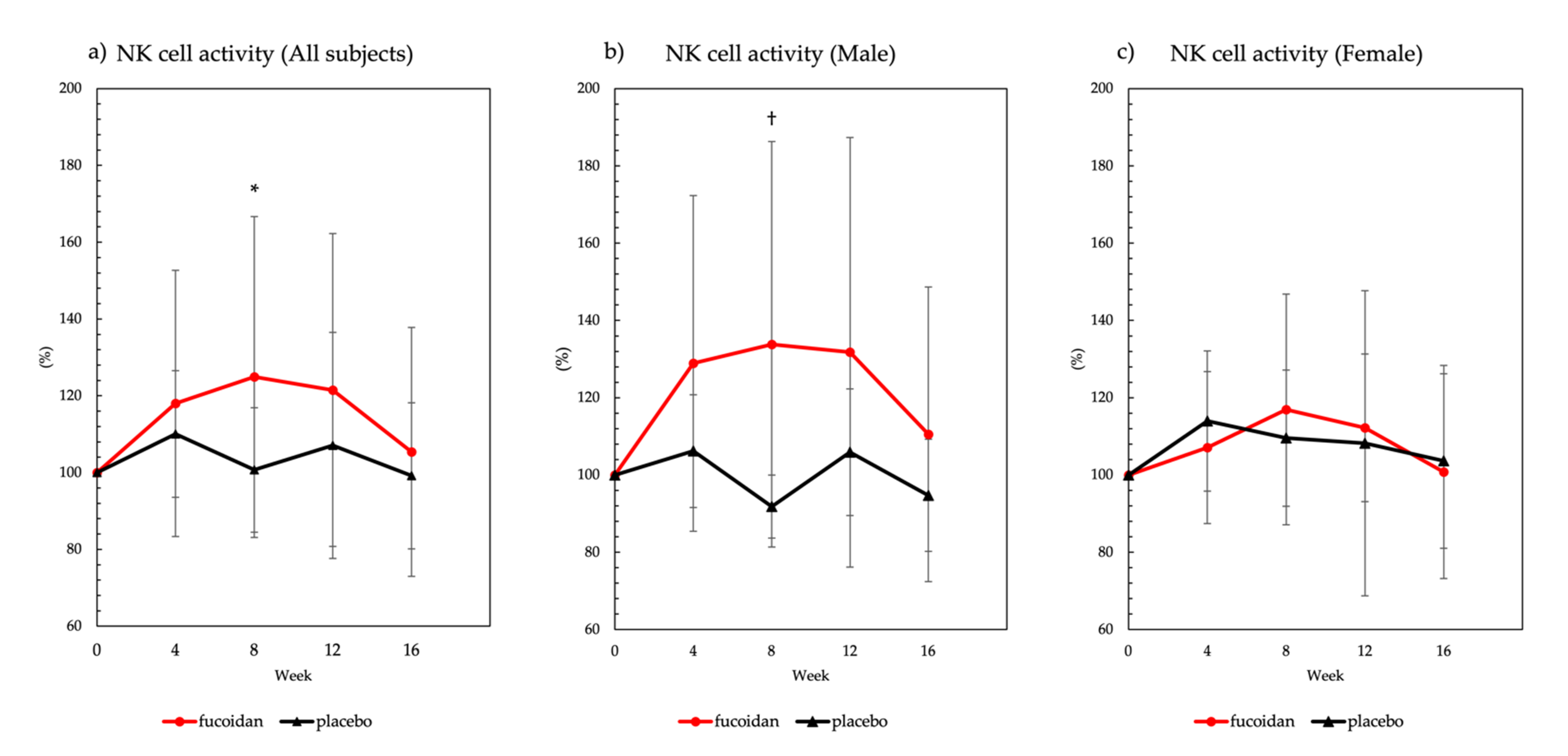
2.2.1. NK Cell Activity
2.2.2. Interferon-γ (IFN-γ) and Interleukin-2 (IL-2) Concentrations in the Blood
2.3. Safty Assessment
2.3.1. Blood Tests
2.3.2. Biochemical Tests
2.3.3. Evaluation of Adverse Events
3. Discussion
3.1. NK Cell Activity
3.2. Safety Assessment
4. Materials and Methods
4.1. Materials
4.2. Subjects
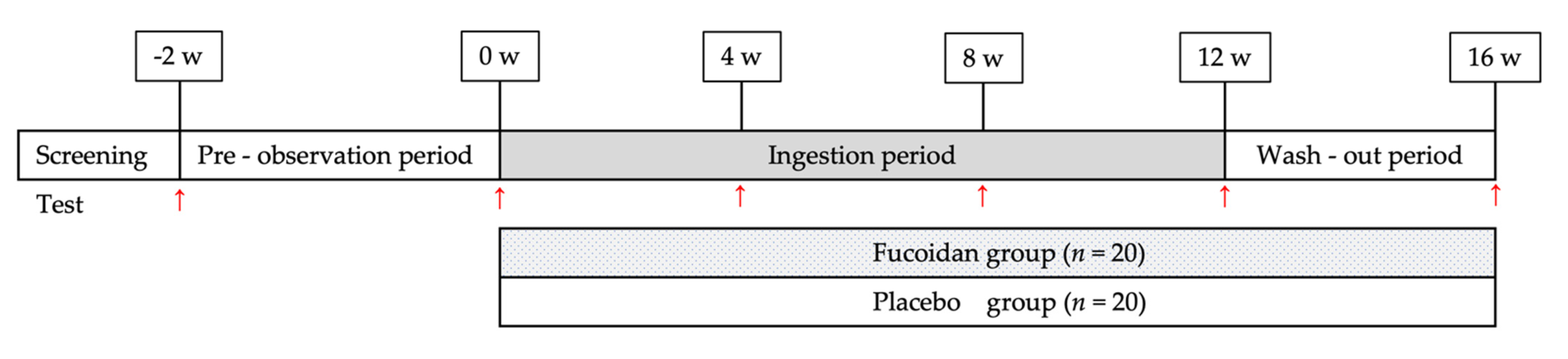
4.3. Study Design
4.4. Examination
4.4.1. NK Cell Activity
4.4.2. INF-γ and IL-2 Concentrations in the Blood
4.4.3. Blood Tests, Biochemical Tests, and Safety Assessment
4.5. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Li, B.; Lu, F.; Wei, X.; Zhao, R. Fucoidan: Structure and bioactivity. Molecules 2008, 13, 1671–1695. [Google Scholar] [CrossRef] [PubMed]
- Cumashi, A.; Ushakova, N.A.; Preobrazhenskaya, M.E.; D′Incecco, A.; Piccoli, A.; Totani, L.; Tinari, N.; Morozevich, G.E.; Berman, A.E.; Bilan, M.I.; et al. A comparative study of the anti-inflammatory, anticoagulant, antiangiogenic, antiadhesive activities of nine different fucoidans from brown seaweeds. Glycobiology 2007, 17, 541–552. [Google Scholar] [CrossRef] [PubMed]
- Prokofjeva, M.M.; Imbs, T.I.; Shevchenko, N.M.; Spirin, P.V.; Horn, S.; Fehse, B.; Zvyagintseva, T.N.; Prassolov, V.S. Fucoidans as potential inhibitors of HIV-1. Mar. Drugs 2013, 11, 3000–3014. [Google Scholar] [CrossRef] [PubMed]
- Thuy, T.T.T.; Ly, B.M.; Van, T.T.T.; Van Quang, N.; Tu, H.C.; Zheng, Y.; Seguin-Devaux, C.; Mi, B.; Ai, U. Anti-HIV activity of fucoidans from three brown seaweed species. Carbohyd. Polym. 2015, 115, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Hidari, K.I.; Takahashi, N.; Arihara, M.; Nagaoka, M.; Morita, K.; Suzuki, T. Structure and anti-dengue virus activity of sulfated polysaccharide from a marine alga. Biochem. Bioph. Res. Commun. 2008, 376, 91–95. [Google Scholar] [CrossRef]
- Haneji, K.; Matsuda, T.; Tomita, M.; Kawakami, H.; Ohshiro, K.; Uchihara, J.N.; Masuda, M.; Takasu, N.; Tanaka, Y.; Ohta, T.; et al. Fucoidan extracted from Cladosiphon okamuranus Tokida induces apoptosis of human T-cell leukemia virus type 1-infected T-cell lines and primary adult T-cell leukemia cells. Nutr. Cancer 2005, 52, 189–201. [Google Scholar] [CrossRef]
- Araya, N.; Takahashi, K.; Sato, T.; Nakamura, T.; Sawa, C.; Hasegawa, D.; Ando, H.; Aratani, S.; Yagishita, N.; Fujii, R.; et al. Fucoidan therapy decreases the proviral load in patients with human T-lymphotropic virus type-1-assoiciated neurological disease. Antivir. Ther. 2011, 16, 89–98. [Google Scholar] [CrossRef]
- Atashrazm, F.; Lowenthal, R.M.; Woods, G.M.; Holloway, A.F.; Dickinson, J.L. Fucoidan and cancer: A multifunctional molecule with anti-tumor potential. Mar. Drugs 2015, 13, 2327–2346. [Google Scholar] [CrossRef]
- Lin, Y.; Qi, X.; Liu, H.; Xue, K.; Xu, S.; Tian, Z. The anti-cancer effects of fucoidan: A review of both in vivo and in vitro investigations. Cancer Cell Int. 2020, 20, 154. [Google Scholar] [CrossRef]
- Mori, N.; Nakasone, K.; Tomimori, K.; Ishikawa, C. Beneficial effects of fucoidan in patients with chronic hepatitis C virus infection. World J. Gastroenterol. 2012, 18, 2225–2230. [Google Scholar] [CrossRef]
- Choi, J.I.; Raghavendran, H.R.B.; Sung, N.Y.; Kim, J.H.; Chun, B.S.; Ahn, D.H.; Choi, H.S.; Kang, K.W.; Lee, J.W. Effect of fucoidan on aspirin-induced stomach ulceration in rats. Chem. Biol. Interact. 2010, 183, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Nagaoka, M.; Shibata, H.; Kimura-Takagi, I.; Hashimoto, S.; Kimura, K.; Makino, T.; Aiyama, R.; Ueyama, S.; Yokokura, T. Structural study of fucoidan from Cladosiphon okamuranus Tokida. Glycoconj. J. 1999, 16, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Tokita, Y.; Nakajima, K.; Mochida, H.; Iha, M.; Nagamine, T. Development of a fucoidan-specific antibody and measurement of fucoidan in serum and urine by sandwich ELISA. Biosci. Biotech. Biochem. 2010, 74, 350–357. [Google Scholar] [CrossRef] [PubMed]
- Nagamine, T.; Nakazato, K.; Tomioka, S.; Iha, M.; Nakajima, K. Intestinal absorption of fucoidan extracted from the brown seaweed, Cladosiphon okamuranus. Mar. Drugs 2015, 13, 48–64. [Google Scholar] [CrossRef] [PubMed]
- Kadena, K.; Tomori, M.; Iha, M.; Nagamine, T. Absorption study of mozuku fucoidan in Japanese volunteers. Mar. Drugs 2018, 16, 254. [Google Scholar] [CrossRef]
- Tomori, M.; Nagamine, T.; Iha, M. Are Helicobacter pylori infection and fucoidan consumption associated with fucoidan absorption? Mar. Drugs 2020, 18, 235. [Google Scholar] [CrossRef]
- Tomori, M.; Nagamine, T.; Miyamoto, T.; Iha, M. Evaluation of the immunomodulatory effects of fucoidan derived from Cladosiphon okamuranus Tokida in mice. Mar. Drugs 2019, 17, 547. [Google Scholar] [CrossRef]
- Nagamine, T.; Kadena, K.; Tomori, M.; Nakajima, K.; Iha, M. Activation of NK cells in male cancer survivors by fucoidan extracted from Cladosiphon okamuranus. Mol. Clin. Oncol. 2020, 12, 81–88. [Google Scholar] [CrossRef]
- Maruyama, H.; Tamauchi, H.; Hashimoto, M.; Nakano, T. Suppression of Th2 immune responses by mekabu fucoidan from Undaria pinnatifida sporophylls. Int. Arch. Allergy Immunol. 2005, 137, 289–294. [Google Scholar] [CrossRef]
- Negishi, H.; Mori, M.; Mori, H.; Yamori, Y. Supplementation of elderly Japanese men and women with fucoidan from seaweed increases immune responses to seasonal influenza vaccination. J. Nutr. 2013, 143, 1794–1798. [Google Scholar] [CrossRef]
- Takahashi, H.; Kawaguchi, M.; Kitamura, K.; Narumiya, S.; Kawamura, M.; Tengan, I.; Nishimoto, S.; Hanamure, Y.; Majima, Y.; Tsubura, S.; et al. An exploratory study on the anti-inflammatory effects of fucoidan in relation to quality of life in advanced cancer patients. Integr. Cancer Ther. 2018, 17, 282–291. [Google Scholar] [CrossRef] [PubMed]
- Ohnogi, H.; Naito, Y.; Higashimura, Y.; Uno, K.; Yoshikawa, T. Immune efficacy and safety of fucoidan extracted from Gagome kombu (Kjellmaniella crassifolia) in healthy Japanese subjects. Jpn. J. Complement. Altern. Med. 2015, 12, 87–93. [Google Scholar]
- Zhang, W.; Okimura, T.; Oda, T.; Jin, O. Ascophyllan induces activation of natural killer cells in mice in vivo and in vitro. Mar. Drugs 2019, 17, 197. [Google Scholar] [CrossRef] [PubMed]
- Murayama, H.; Tamauchi, H.; Iizuka, M.; Nakano, T. The role of NK cells in antitumor activity of dietary fucoidan from Undaria pinnatifida sporophylls (Mekabu). Planta Med. 2006, 72, 1415–1417. [Google Scholar] [CrossRef]
- Doi, T.; Higashino, K.I.; Kurihara, Y.; Wada, Y.; Miyazaki, T.; Nakamura, H.; Uesugi, S.; Imanishi, T.; Kawabe, Y.; Itakura, H. Charged collagen structure mediate the recognition of negatively charged macromolecules by macrophage scavenger receptors. J. Biol. Chem. 1993, 268, 2126–2133. [Google Scholar] [CrossRef]
- Lin, Z.; Tan, X.; Zhang, Y.; Li, F.; Luo, P.; Liu, H. Molecular targets and related biologic activities of fucoidan: A review. Mar. Drugs 2020, 18, 376. [Google Scholar] [CrossRef]
- Miyazaki, Y.; Iwaihara, Y.; Bak, J.; Nakano, H.; Takeuchi, S.; Takeuchi, H.; Matsui, T.; Tachikawa, D. The cooperative induction of macrophage activation by fucoidan derived Cladosiphon okamuranus and β-glucan derived from Saccharomyces cerevisiae. Biochem. Biophys. Res. Commun. 2019, 516, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Klein, S.L.; Flanagan, K.L. Sex differences in immune responses. Nat. Rev. Immunol. 2016, 16, 626–638. [Google Scholar] [CrossRef] [PubMed]
- Pisitkun, P.; Deane, J.A.; Difilippantonio, M.J.; Tarasenko, T.; Satterthwaite, A.B.; Bolland, S. Autoreactive B cell responses to RNA-related antigens due to TLR7 gene duplication. Science 2006, 312, 1669–1672. [Google Scholar] [CrossRef]
- Ngo, S.T.; Steyn, F.J.; McCombe, P.A. Gender differences in autoimmune disease. Front. Neuroendocrinol. 2014, 35, 347–369. [Google Scholar] [PubMed]
- Namkoong, S.; Kim, J.Y.; Kim, T.; Sohn, H.E. Immunomodulatory effects of fucoidan on NK cells in ovariectomized rats. Korean J. Plant Res. 2012, 25, 317–322. [Google Scholar] [CrossRef]
- Abe, S.; Hiramatsu, K.; Ichikawa, O.; Kawamoto, H.; Kasagi, T.; Miki, Y.; Kimura, T.; Ikeda, T. Safety evaluation of excessive ingestion of mozuku fucoidan in human. J. Food Sci. 2013, 78, T648–T651. [Google Scholar] [CrossRef] [PubMed]
- Tomori, M.; Hirata, T.; Nagamine, T.; Iha, M. Safety evaluation of long-term and excessive ingestion of Cladosiphon okamuranus fucoidan in healthy subjects. A randomized, double-blind, placebo-controlled, parallel study. Jpn. Pharmacol. Ther. 2017, 45, 447–461. (In Japanese) [Google Scholar]
- Tomori, M.; Matsuda, K.; Nakamura, Y.; Kadena, K.; Shimoji, S.; Nagamine, T.; Iha, M. Effect of Cladosiphon okamuranus fucoidan containing drink on improvement of defecation in healthy subjects with mild constipation—A randomized, doble-blind, placebo-controlled, crossover study. Jpn. Pharmacol. Ther. 2016, 44, 1621–1626. (In Japanese) [Google Scholar]
| Parameter | Placebo | Fucoidan | p-Value |
|---|---|---|---|
| Subjects (n) | 20 | 20 | 1.00 |
| Male (n) | 10 | 10 | 1.00 |
| Female (n) | 10 | 10 | 1.00 |
| Age (years) | 45.5 ± 7.8 | 47.0 ± 7.6 | 0.55 |
| Body weight (kg) | 59.1 ± 9.5 | 57.6 ± 9.8 | 0.63 |
| Body mass index (kg/m2) | 21.7 ± 2.3 | 21.0 ± 2.6 | 0.39 |
| Group | 0 w | 4 w | 8 w | 12 w | 16 w |
|---|---|---|---|---|---|
| Fucoidan | 4 | 4 | 4 | 6 | 3 |
| Placebo | 5 | 6 | 3 | 4 | 3 |
| Parameter | Group | 0 w | 4 w | 8 w | 12 w | 16 w |
|---|---|---|---|---|---|---|
| WBCs (μL) | Fucoidan | 5720 ± 1233 | 5080 ± 1307 | 5179± 1280 | 5168 ± 1433 | 5589 ± 1610 |
| Placebo | 5950 ± 1674 | 5535 ± 911 | 5660 ± 985 | 5520 ± 1282 | 5685 ± 1132 | |
| RBCs (×104/μL) | Fucoidan | 453.6 ± 41.4 | 458.2 ± 37.5 | 453.3 ± 36.4 | 456.8 ± 40.2 | 463.6 ± 37.4 |
| Placebo | 459.8 ± 28.3 | 462.7 ± 30.3 | 462.7 ± 28.9 | 472.1 ± 33.0 | 465.3 ± 36.2 | |
| Hb (g/dL) | Fucoidan | 13.7 ± 1.6 | 13.8 ± 1.5 | 13.7 ± 1.7 | 13.6 ± 1.7 | 13.8 ± 1.7 |
| Placebo | 14.2 ± 0.9 | 14.2 ± 1.0 | 14.2 ± 0.8 | 14.5 ± 1.1 | 14.2 ± 1.2 | |
| Ht (%) | Fucoidan | 42.7 ± 4.7 | 42.9 ± 4.2 | 42.3 ± 4.2 | 42.5 ± 4.4 | 43.2 ± 4.7 |
| Placebo | 43.1 ± 2.2 | 43.7 ± 3.0 | 43.6 ± 2.1 | 44.7 ± 2.8 | 43.8 ± 3.2 | |
| Plt (×104/μL) | Fucoidan | 26.2 ± 5.4 | 26.5 ± 5.9 | 26.1 ± 4.6 | 28.4 ± 9.4 | 27.4 ± 4.8 |
| Placebo | 27.3 ± 3.5 | 27.4 ± 4.0 | 27.5 ± 4.2 | 27.9 ± 4.4 | 28.3 ± 4.3 |
| Parameter | Group | 0 w | 4 w | 8 w | 12 w | 16 w |
|---|---|---|---|---|---|---|
| AST (U/L) | Fucoidan | 22.4 ± 11.2 | 23.9 ± 17.6 | 21.2 ± 11.2 | 21.2 ± 8.8 | 22.7 ± 8.9 |
| Placebo | 20.2 ± 5.9 | 21.3 ± 6.0 | 20.9 ± 7.1 | 21.8 ± 6.7 | 22.2 ± 4.8 | |
| ALT (U/L) | Fucoidan | 17.9 ± 10.7 | 19.6 ± 17.1 | 16.9 ± 10.8 | 16.5 ± 10.0 | 18.7 ± 10.9 |
| Placebo | 17.7 ± 9.6 | 18.3 ± 10.2 | 17.7 ± 7.9 | 20.9 ± 11.9 | 19.5 ± 9.3 | |
| LDH (U/L) | Fucoidan | 177.5 ± 25.5 | 180.5 ± 28.0 | 176.6 ± 25.4 | 178.8 ± 28.6 | 177.3 ± 20.9 |
| Placebo | 171.1 ± 21.6 | 176.0 ± 28.5 | 179.8 ± 29.8 | 173.0 ± 24.9 | 170.8 ± 25.1 | |
| T-bil (mg/dL) | Fucoidan | 0.9 ± 0.3 | 0.9 ± 0.3 | 0.7 ± 0.2 | 0.8 ± 0.3 | 0.8 ± 0.3 |
| Placebo | 0.9 ± 0.4 | 0.8 ± 0.4 | 0.8 ± 0.3 | 0.8 ± 0.3 | 0.7 ± 0.2 | |
| ALP (U/L) | Fucoidan | 183.6 ± 51.5 | 180.7 ± 46.8 | 190.1 ± 53.4 | 189.9 ± 53.5 | 198.1 ± 55.1 |
| Placebo | 172.4 ± 49.3 | 174.1 ± 47.1 | 175.6 ± 47.0 | 179.0 ± 52.5 | 182.6 ± 61.8 | |
| γ-GTP (U/L) | Fucoidan | 41.2 ± 57.4 | 37.7 ± 46.6 | 34.4 ± 37.1 | 34.6 ± 31.9 | 32.5 ± 29.7 |
| Placebo | 22.6 ± 15.5 | 23.4 ± 16.7 | 25.1 ± 17.3 | 27.1 ± 24.8 | 25.2 ± 18.1 | |
| CK (U/L) | Fucoidan | 119.3 ± 67.2 | 133.9 ± 109.8 | 114.2 ± 47.5 | 122.3 ± 74.6 | 141.8 ± 112.0 |
| Placebo | 122.7 ± 66.0 | 118.6 ± 66.5 | 136.3 ± 92.0 | 126.9 ± 88.1 | 114.7 ± 57.3 | |
| FBS (mg/dL) | Fucoidan | 83.1 ± 8.3 | 82.4 ± 9.7 | 81.3 ± 7.4 | 79.1 ± 8.2 | 82.6 ± 12.5 |
| Placebo | 82.8 ± 8.0 | 82.5 ± 7.0 | 82.7 ± 7.2 | 82.3 ± 7.8 | 81.0 ± 6.1 | |
| HbA1c (%) | Fucoidan | 5.4 ± 0.3 | 5.5 ± 0.2 | 5.4 ± 0.3 | 5.4 ± 0.3 | 5.4 ± 0.2 |
| Placebo | 5.4 ± 0.3 | 5.5 ± 0.3 | 5.4 ± 0.3 | 5.4 ± 0.2 | 5.4 ± 0.3 | |
| TC (mg/dL) | Fucoidan | 200.7 ± 32.0 | 203.2 ± 29.7 | 201.7 ± 26.7 | 202.6 ± 26.6 | 213.9 ± 33.1 |
| Placebo | 203.2 ± 29.8 | 205.3 ± 30.1 | 212.3 ± 33.9 | 215.7 ± 37.1 | 213.1 ± 30.8 | |
| LDL-C (mg/dL) | Fucoidan | 111.1 ± 32.0 | 111.1 ± 31.8 | 109.8 ± 31.9 | 106.8 ± 32.4 | 115.5 ± 34.5 |
| Placebo | 117.6 ± 26.7 | 119.2 ± 26.0 | 125.6 ± 29.9 | 126.9 ± 33.7 | 124.6 ± 31.6 | |
| HDL-C (mg/dL) | Fucoidan | 69.8 ± 16.2 | 72.1 ± 17.2 | 68.8 ± 16.1 | 72.0 ± 20.1 | 76.4 ± 21.5 |
| Placebo | 64.7 ± 13.1 | 64.7 ± 15.6 | 67.4 ± 13.5 | 66.4 ± 15.4 | 66.0 ± 15.7 | |
| TG (mg/dL) | Fucoidan | 81.9 ± 32.0 | 81.3 ± 48.8 | 93.8 ± 48.8 | 95.2 ± 92.4 | 97.0 ± 105.7 |
| Placebo | 98.0 ± 92.3 | 86.4 ± 47.7 | 84.8 ± 39.9 | 88.8 ± 39.8 | 96.8 ± 44.2 | |
| TP (g/dL) | Fucoidan | 7.2 ± 0.4 | 7.2 ± 0.3 | 7.1 ± 0.4 | 7.3 ± 0.3 | 7.3 ± 0.3 |
| Placebo | 7.3 ± 0.4 | 7.4 ± 0.4 | 7.3 ± 0.4 | 7.4 ± 0.4 | 7.4 ± 0.3 | |
| Alb (g/dL) | Fucoidan | 4.5 ± 0.3 | 4.5 ± 0.2 | 4.4 ± 0.2 | 4.5 ± 0.2 | 4.5 ± 0.2 |
| Placebo | 4.5 ± 0.3 | 4.5 ± 0.2 | 4.5 ± 0.2 | 4.5 ± 0.2 | 4.5 ± 0.2 | |
| UN (mg/dL) | Fucoidan | 12.4 ± 4.4 | 12.2 ± 3.7 | 12.2 ± 2.9 | 13.1 ± 3.9 | 13.6 ± 3.1 |
| Placebo | 13.1 ± 3.5 | 13.0 ± 3.9 | 12.2 ± 3.7 | 12.2 ± 3.7 | 14.3 ± 4.7 | |
| CRE (mg/dL) | Fucoidan | 0.7 ± 0.1 | 0.7 ± 0.1 | 0.7 ± 0.1 | 0.7 ± 0.1 | 0.7 ± 0.1 |
| Placebo | 0.8 ± 0.1 | 0.8 ± 0.1 | 0.7 ± 0.1 | 0.7 ± 0.1 | 0.7 ± 0.1 | |
| UA (mg/dL) | Fucoidan | 5.3 ± 1.8 | 5.3 ± 1.6 | 5.1 ± 1.6 | 5.2 ± 1.6 | 5.2 ± 1.6 |
| Placebo | 5.1 ± 1.3 | 5.2 ± 1.3 | 5.2 ± 1.3 | 5.3 ± 1.2 | 5.2 ± 1.6 | |
| Na (mEq/L) | Fucoidan | 140.1 ± 1.8 | 140.0 ± 1.6 | 140.4 ± 2.1 | 140.8 ± 1.8 | 140.2 ± 1.1 |
| Placebo | 140.4 ± 1.1 | 140.3 ± 1.5 | 141.0 ± 1.5 | 141.0 ± 1.8 | 140.7 ± 1.1 | |
| Cl (mEq/L) | Fucoidan | 103.0 ± 1.6 | 103.1 ± 2.2 | 103.9 ± 1.9 | 103.7 ± 2.1 | 103.3 ± 1.9 |
| Placebo | 103.3 ± 1.3 | 103.7 ± 1.6 | 103.9 ± 1.6 | 103.6 ± 2.0 | 104.1 ± 1.8 | |
| K (mEq/L) | Fucoidan | 4.3 ± 0.3 | 4.2 ± 0.3 | 4.4 ± 0.2 | 4.3 ± 0.3 | 4.4 ± 0.2 |
| Placebo | 4.2 ± 0.2 | 4.3 ± 0.4 | 4.3 ± 0.3 | 4.2 ± 0.3 | 4.3 ± 0.2 | |
| Ca (mg/dL) | Fucoidan | 9.6 ± 0.3 | 9.6 ± 0.3 | 9.4 ± 0.3 | 9.6 ± 0.4 | 9.6 ± 0.3 |
| Placebo | 9.7 ± 0.3 | 9.7 ± 0.3 | 9.5 ± 0.3 | 9.6 ± 0.3 | 9.6 ± 0.3 | |
| IP (mg/dL) | Fucoidan | 3.6 ± 0.5 | 3.6 ± 0.5 | 3.6 ± 0.4 | 3.8 ± 0.5 | 3.6 ± 0.5 |
| Placebo | 3.7 ± 0.4 | 3.7 ± 0.5 | 3.6 ± 0.5 | 3.6 ± 0.4 | 3.6 ± 0.6 | |
| Mg (mg/dL) | Fucoidan | 2.1 ± 0.1 | 2.2 ± 0.2 | 2.1 ± 0.1 | 2.2 ± 0.1 † | 2.1 ± 0.1 † |
| Placebo | 2.2 ± 0.1 | 2.2 ± 0.2 | 2.1 ± 0.1 | 2.1 ± 0.1 † | 2.2 ± 0.1 † | |
| Fe (μg/dL) | Fucoidan | 117.1 ± 49.0 | 104.7 ± 35.0 | 112.9 ± 37.7 † | 108.7 ± 51.6 | 98.9 ± 52.6 |
| Placebo | 99.9 ± 43.5 | 95.9 ± 45.2 | 86.4 ± 32.5 † | 103.2 ± 48.4 | 116.6 ± 37.4 |
| Group | Adverse Event | Case | Treatment | Outcome | Causality | Study |
|---|---|---|---|---|---|---|
| Fucoidan | Cold | 7 | 3 | 7 | All unrelated | Continue all |
| Pharyngitis | 2 | 1 | 2 | Unrelated | Continue all | |
| Neck strain | 1 | 1 | 1 | Unrelated | Continue | |
| Tooth cavity | 1 | 1 | 1 | Unrelated | Continue | |
| Placebo | Cold | 10 | 9 | 10 | All unrelated | Continue all |
| Pharyngitis and cough | 1 | 0 | 1 | Unrelated | Continue | |
| Abdominal pain | 1 | 0 | 1 | Unrelated | Continue | |
| Automatic nerves disorder | 1 | 1 | 1 | Probably unrelated | Continue | |
| Sprain | 1 | 1 | 1 | Unrelated | Continue | |
| Back pain | 1 | 1 | 1 | Unrelated | Continue | |
| Fatigue | 1 | 1 | 1 | Unrelated | Continue | |
| Gum disease | 1 | 1 | 1 | Unrelated | Continue |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tomori, M.; Nagamine, T.; Miyamoto, T.; Iha, M. Effects of Ingesting Fucoidan Derived from Cladosiphon okamuranus Tokida on Human NK Cells: A Randomized, Double-Blind, Parallel-Group, Placebo-Controlled Pilot Study. Mar. Drugs 2021, 19, 340. https://doi.org/10.3390/md19060340
Tomori M, Nagamine T, Miyamoto T, Iha M. Effects of Ingesting Fucoidan Derived from Cladosiphon okamuranus Tokida on Human NK Cells: A Randomized, Double-Blind, Parallel-Group, Placebo-Controlled Pilot Study. Marine Drugs. 2021; 19(6):340. https://doi.org/10.3390/md19060340
Chicago/Turabian StyleTomori, Makoto, Takeaki Nagamine, Tomofumi Miyamoto, and Masahiko Iha. 2021. "Effects of Ingesting Fucoidan Derived from Cladosiphon okamuranus Tokida on Human NK Cells: A Randomized, Double-Blind, Parallel-Group, Placebo-Controlled Pilot Study" Marine Drugs 19, no. 6: 340. https://doi.org/10.3390/md19060340
APA StyleTomori, M., Nagamine, T., Miyamoto, T., & Iha, M. (2021). Effects of Ingesting Fucoidan Derived from Cladosiphon okamuranus Tokida on Human NK Cells: A Randomized, Double-Blind, Parallel-Group, Placebo-Controlled Pilot Study. Marine Drugs, 19(6), 340. https://doi.org/10.3390/md19060340






