Jellyfish Bioprospecting in the Mediterranean Sea: Antioxidant and Lysozyme-Like Activities from Aurelia coerulea (Cnidaria, Scyphozoa) Extracts
Abstract
1. Introduction
2. Results
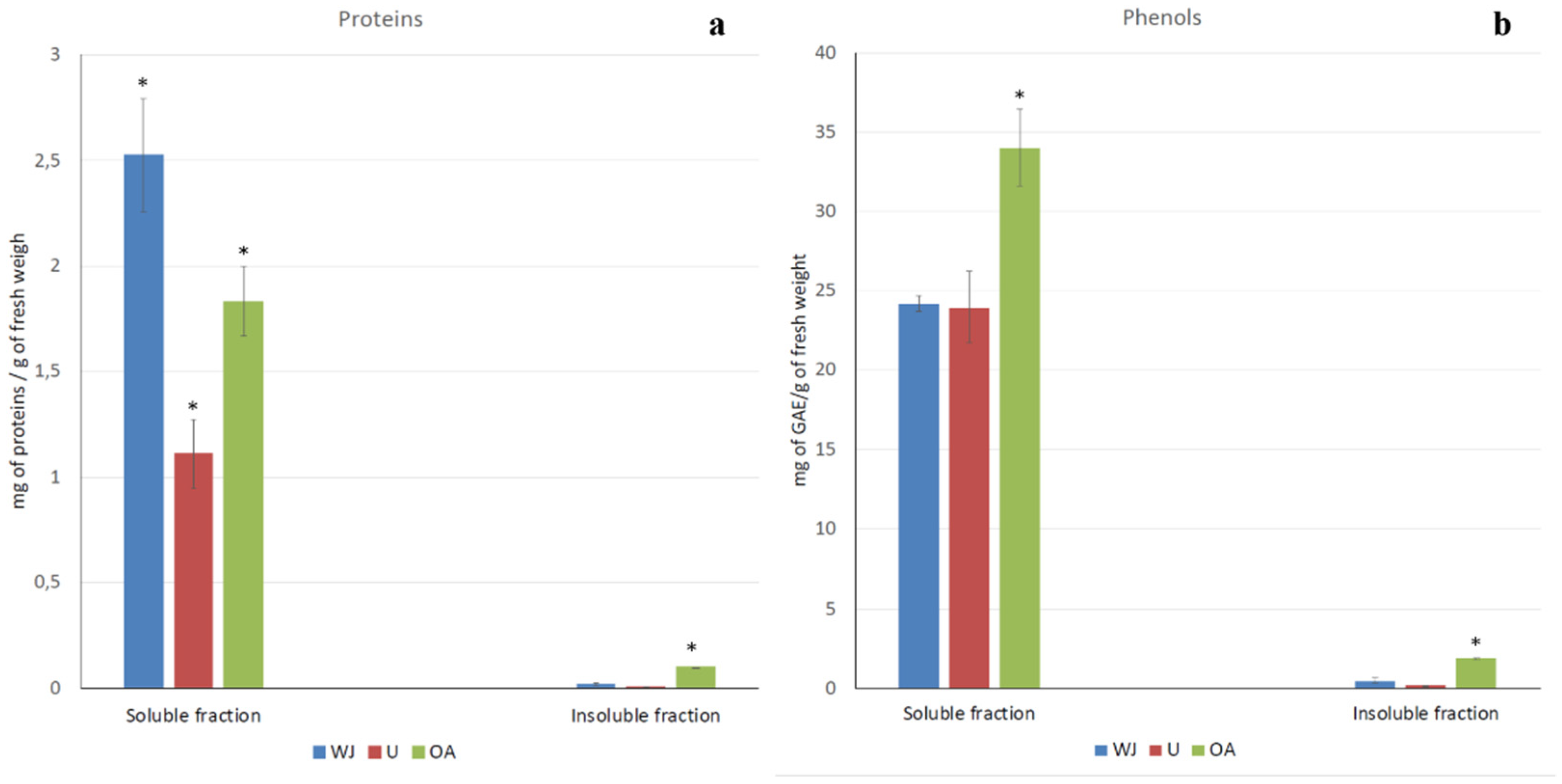
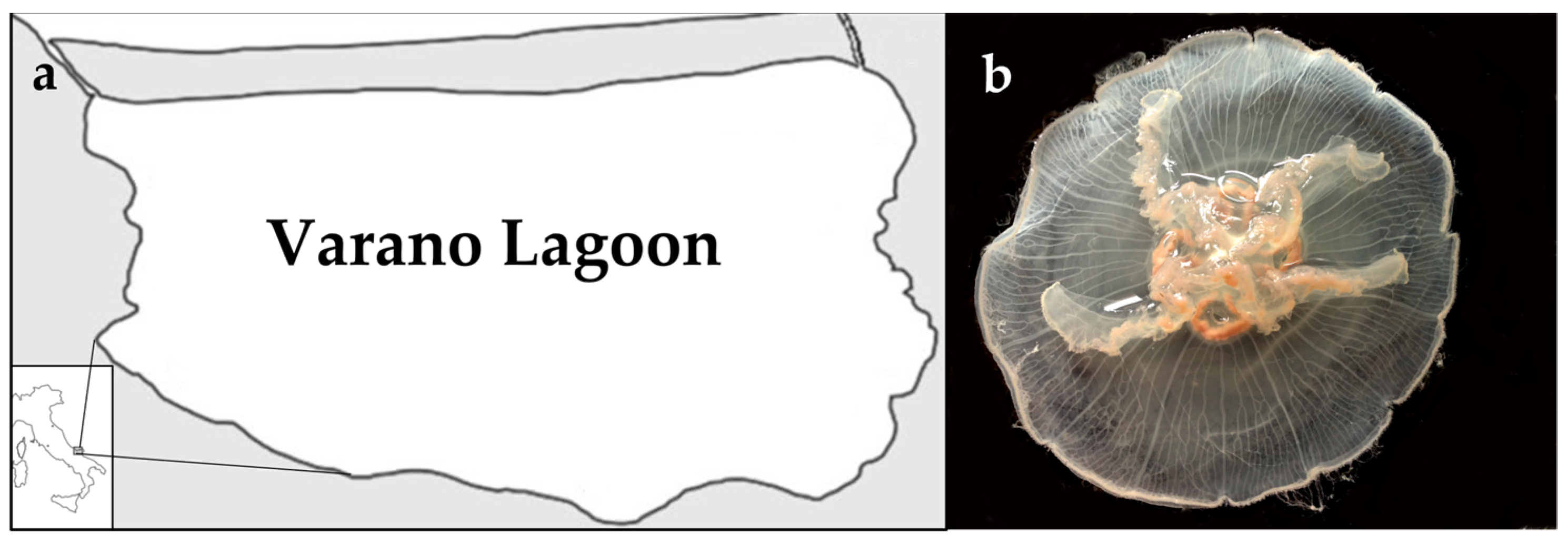
2.1. Jellyfish Sample Characterization
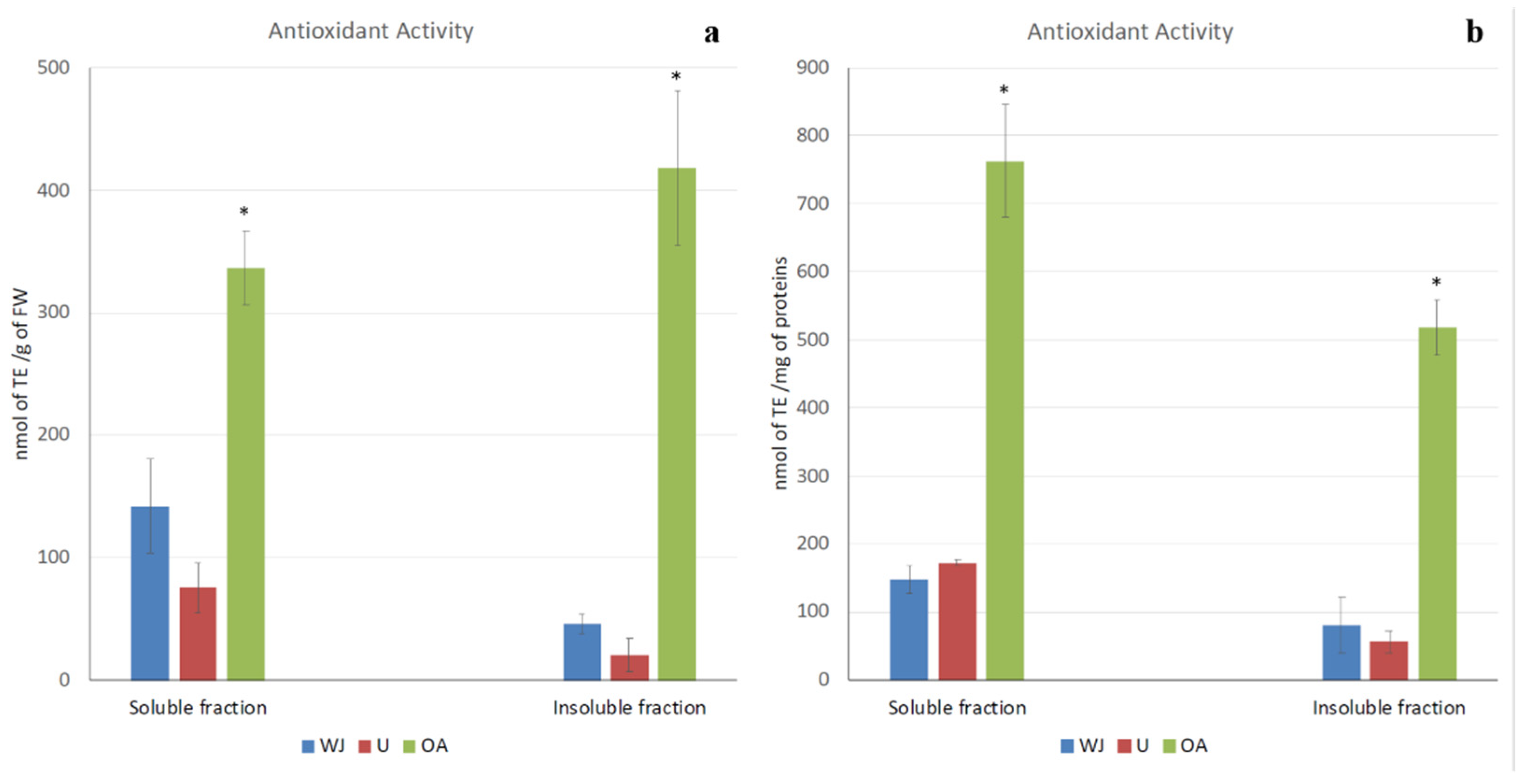
2.2. Antioxidant Activity
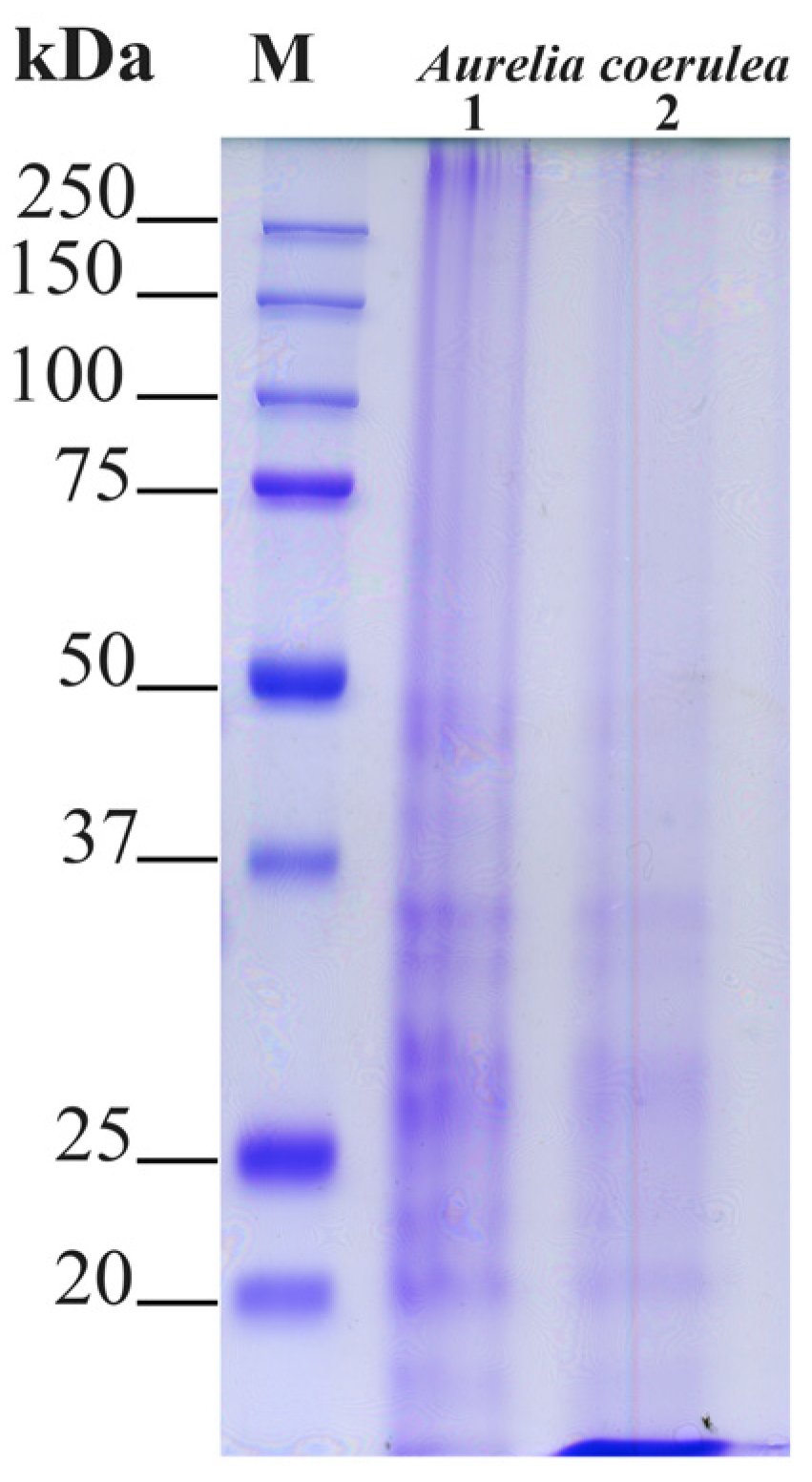
SDS-PAGE Separation of Jellyfish Proteins
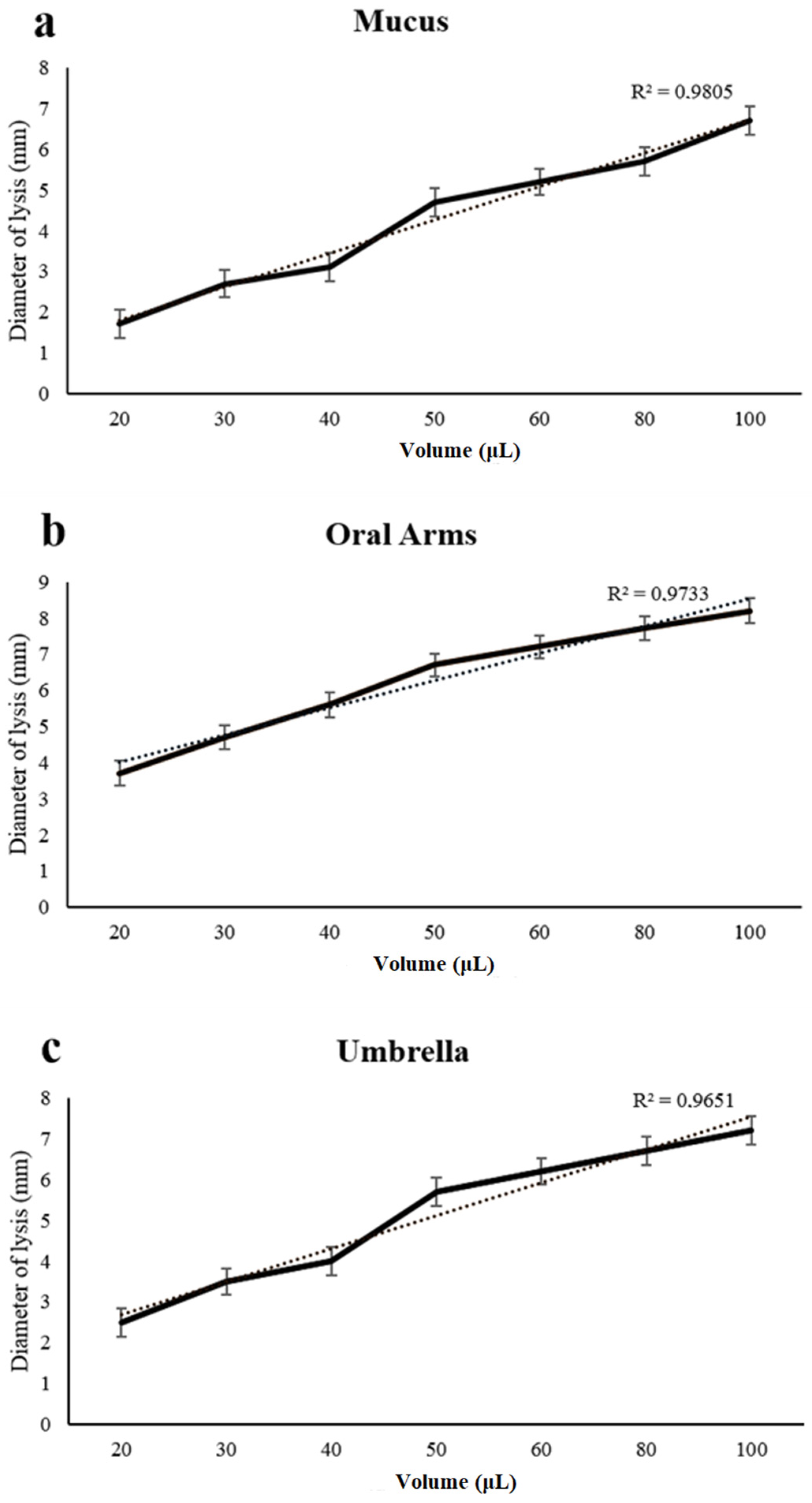
2.3. Lysozyme-Like Activity
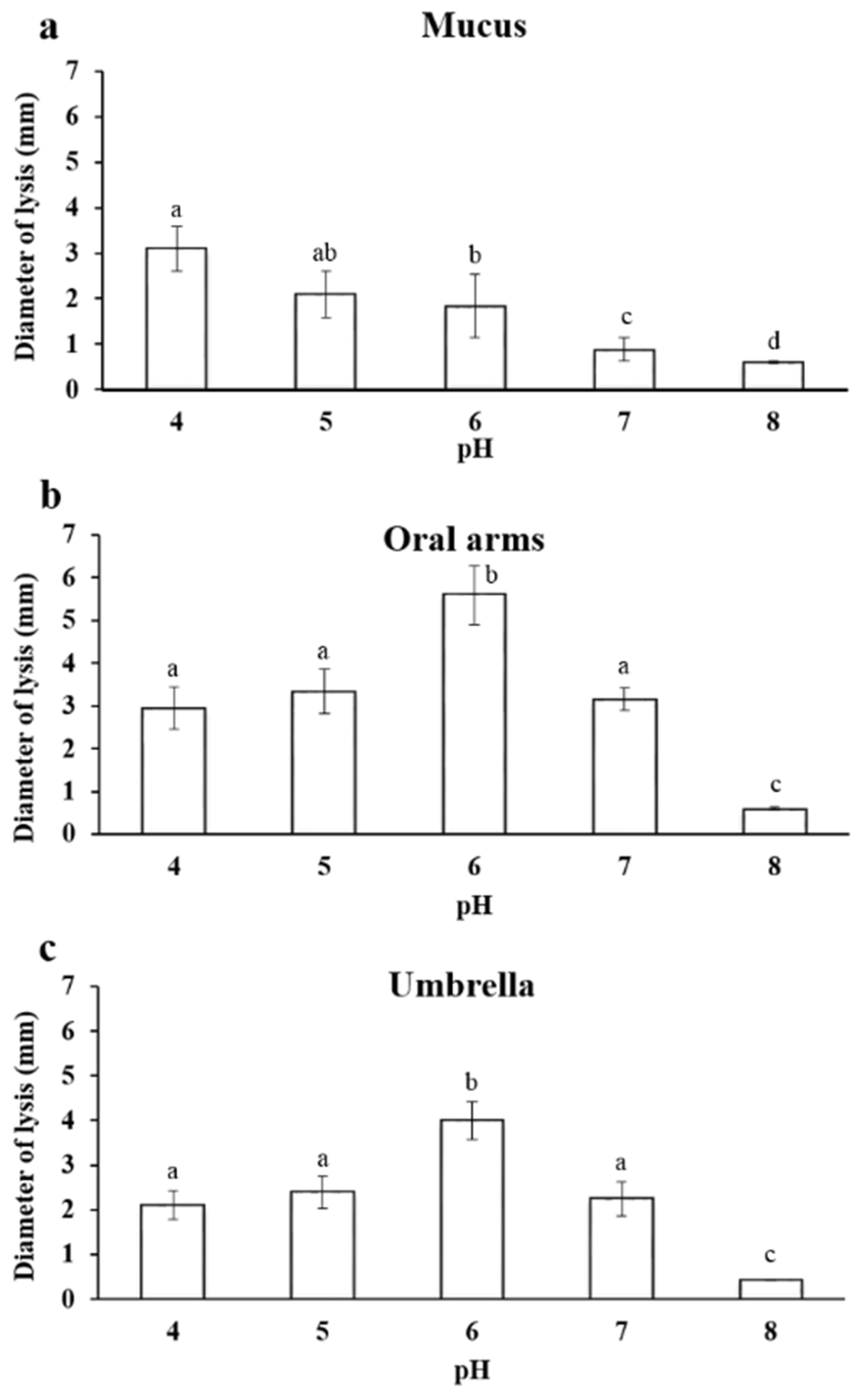
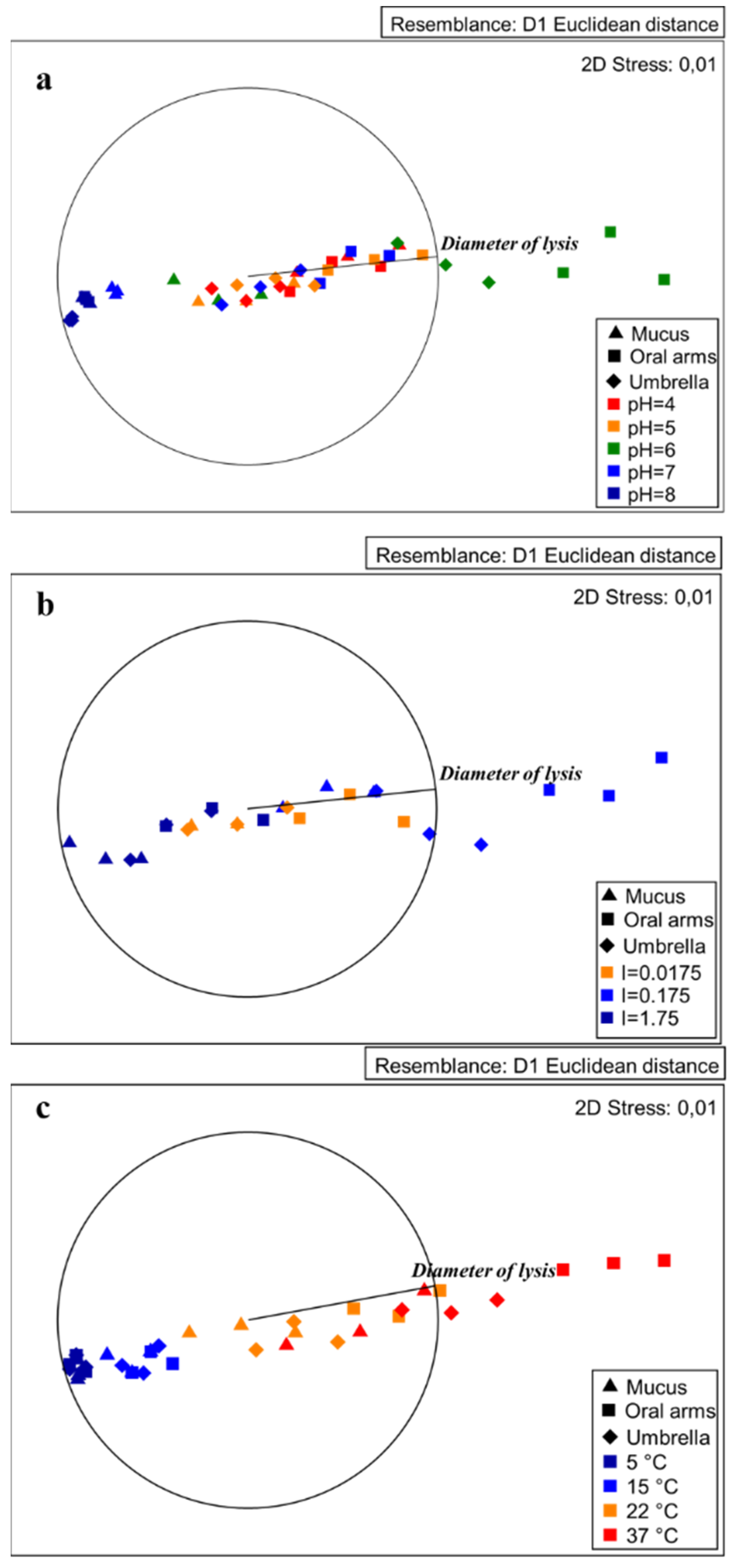
2.3.1. Effect of pH on Lysozyme-Like Activity
2.3.2. Effect of Ionic Strength on Lysozyme–Like Activity
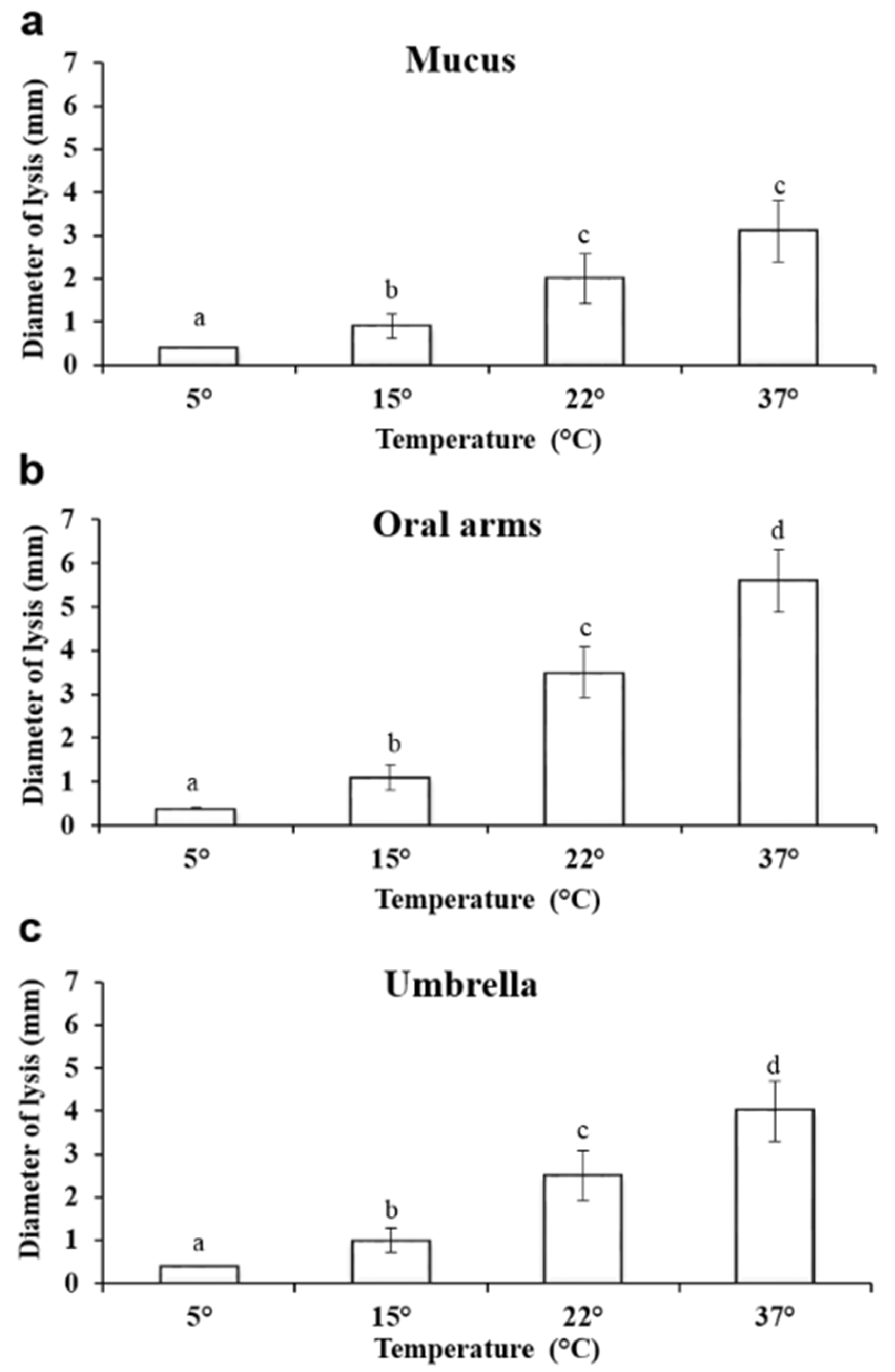
2.3.3. Effect of Temperature on Lysozyme–Like Activity
3. Discussion
4. Materials and Methods
4.1. Sample Collection and Preparation
4.2. Protein, Phenolic and Antioxidant Analyses
4.2.1. Protein Content
4.2.2. Phenol Content
4.2.3. Antioxidant Activity
4.2.4. SDS-PAGE Analysis
4.3. Lysozyme-Like Activity
4.4. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Aneiros, A.; Garateix, A. Bioactive peptides from marine sources: Pharmacological properties and isolation procedures. J. Chromatogr. B 2004, 803, 41–53. [Google Scholar] [CrossRef]
- Parisi, M.G.; Parrinello, D.; Stabili, L.; Cammarata, M. Cnidarian immunity and the repertoire of defense mechanisms in anthozoans. Biology 2020, 9, 283. [Google Scholar] [CrossRef]
- Watters, M.R. Tropical Marine Neurotoxins: Venoms to Drugs. Semin. Neurol. 2005, 25, 278–289. [Google Scholar] [CrossRef]
- Marino, A.; Valveri, V.; Muià, C.; Crupi, R.; Rizzo, G.; Musci, G.; La Spada, G. Cytotoxicity of the nematocyst venom from the sea anemone Aiptasia mutabilis. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2004, 139, 295–301. [Google Scholar] [CrossRef] [PubMed]
- Radwan, F.F.Y.; Burnett, J.W.; Bloom, D.A.; Coliano, T.; Eldefrawi, M.E.; Erderly, H.; Aurelian, L.; Torres, M.; Heimer-de la Cotera, E.P. A comparison of the toxicological characteristics of two Cassiopea and Aurelia species. Toxicon 2001, 39, 245–257. [Google Scholar] [CrossRef]
- Bayazit, V. Cytotoxic effects of some animal and vegetable extracts and some chemicals on liver and colon carcinoma and myosarcoma. Saudi Med. J. 2004, 25, 156–163. [Google Scholar]
- Lotan, A.; Fishman, L.; Zlotkin, E. Toxin compartmentation and delivery in the Cnidaria: The nematocyst’s tubule as a multiheaded poisonous arrow. J. Exp. Zool. 1996, 275, 444–451. [Google Scholar] [CrossRef]
- Li, J.; Li, Q.; Li, J.; Zhou, B. Peptides derived from Rhopilema esculentum hydrolysate exhibit angiotensin converting enzyme (ACE) inhibitory and antioxidant abilities. Molecules 2014, 19, 13587–13602. [Google Scholar] [CrossRef]
- Yu, H.; Liu, X.; Xing, R.; Liu, S.; Li, C.; Li, P. Radical scavenging activity of protein from tentacles of jellyfish Rhopilema esculentum. Bioorg. Med. Chem. Lett. 2005, 15, 2659–2664. [Google Scholar] [CrossRef]
- Leone, A.; Lecci, R.M.; Durante, M.; Meli, F.; Piraino, S. The Bright Side of Gelatinous Blooms: Nutraceutical Value and Antioxidant Properties of Three Mediterranean Jellyfish (Scyphozoa). Mar. Drugs 2015, 13, 4654–4681. [Google Scholar] [CrossRef] [PubMed]
- De Domenico, S.; De Rinaldis, G.; Paulmery, M.; Piraino, S.; Leone, A. Barrel Jellyfish (Rhizostoma pulmo) as Source of Antioxidant Peptides. Mar. Drugs 2019, 17, 134. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, Y.; Hou, H.; Zhao, X.; Zhang, Z.; Li, B. Effects of collagen and collagen hydrolysate from jellyfish (Rhopilema esculentum) on mice skin photoaging induced by UV Irradiation. J. Food Sci. 2009, 74, H187–H188. [Google Scholar] [CrossRef]
- Zhuang, Y.; Sun, L.; Zhao, X.; Wang, J.; Hou, H.; Li, B. Antioxidant and melanogenesis-inhibitory activities of collagen peptide from jellyfish (Rhopilema esculentum). J. Sci. Food Agric. 2009, 89, 1722–1727. [Google Scholar] [CrossRef]
- Koch, E. Structural forms and possible roles of aligned cytoskeletal biopolymers in hagfish (slime eel) mucus*1. J. Struct. Biol. 1991, 106, 205–210. [Google Scholar] [CrossRef]
- Davies, J.M.; Viney, C. Water–mucin phases: Conditions for mucus liquid crystallinity. Thermochim. Acta 1998, 315, 39–49. [Google Scholar] [CrossRef]
- Brusca, R.C.; Brusca, G.J. Invertebrates. xviii, 922p. Sunderland, Mass: Ainauer Associates, 1990. J. Mar. Biol. Assoc. UK 1991, 71, 245. [Google Scholar] [CrossRef]
- Stabili, L.; Schirosi, R.; Parisi, M.G.; Piraino, S.; Cammarata, M. The Mucus of Actinia equina (Anthozoa, Cnidaria): An Unexplored Resource for Potential Applicative Purposes. Mar. Drugs 2015, 13, 5276–5296. [Google Scholar] [CrossRef]
- Tidball, J.G. Cnidaria: Secreted Surface. In Biology of the Integument; Bereiter-Hahn, J., Matoltsy, A.G., Richards, K.S., Eds.; Springer: Berlin/Heidelberg, Germany, 1984. [Google Scholar] [CrossRef]
- Zasloff, M. Antimicrobial peptides of multicellular organisms. Nature 2002, 415, 389–395. [Google Scholar] [CrossRef]
- Ovchinnikova, T.V.; Balandin, S.V.; Aleshina, G.M.; Tagaev, A.A.; Leonova, Y.F.; Krasnodembsky, E.D.; Menshenin, A.V.; Kokryakov, V.N. Aurelin, a novel antimicrobial peptide from jellyfish Aurelia aurita with structural features of defensins and channel-blocking toxins. Biochem. Biophys. Res. Commun. 2006, 348, 514–523. [Google Scholar] [CrossRef] [PubMed]
- Rocha, J.; Peixe, L.; Gomes, N.C.M.; Calado, R. Cnidarians as a source of new marine bioactive compounds—An overview of the last decade and future steps for bioprospecting. Mar. Drugs 2011, 9, 1860–1886. [Google Scholar] [CrossRef] [PubMed]
- Phillips, D.C. The three-dimensional structure of an enzyme molecule. Sci. Am. 1966, 215, 78–90. [Google Scholar] [CrossRef]
- Sava, G. Pharmacological aspects and therapeutic applications of lysozymes. Exs 1996, 75, 433–449. [Google Scholar] [PubMed]
- Stabili, L.; Rizzo, L.; Fanizzi, F.P.; Angilè, F.; Del Coco, L.; Girelli, C.R.; Lomartire, S.; Piraino, S.; Basso, L. The Jellyfish Rhizostoma pulmo (Cnidaria): Biochemical Composition of Ovaries and Antibacterial Lysozyme-like Activity of the Oocyte Lysate. Mar. Drugs 2019, 17, 17. [Google Scholar] [CrossRef] [PubMed]
- Stabili, L.; Rizzo, L.; Basso, L.; Marzano, M.; Fosso, B.; Pesole, G.; Piraino, S. The Microbial Community Associated with Rhizostoma pulmo: Ecological Significance and Potential Consequences for Marine Organisms and Human Health. Mar. Drugs 2020, 18, 437. [Google Scholar] [CrossRef] [PubMed]
- Basso, L.; Rizzo, L.; Marzano, M.; Intranuovo, M.; Fosso, B.; Pesole, G.; Piraino, S.; Stabili, L. Jellyfish summer outbreaks as bacterial vectors and potential hazards for marine animals and humans health? The case of Rhizostoma pulmo (Scyphozoa, Cnidaria). Sci. Total Environ. 2019, 692, 305–318. [Google Scholar] [CrossRef]
- Scorrano, S.; Aglieri, G.; Boero, F.; Dawson, M.N.; Piraino, S. Unmasking Aurelia species in the Mediterranean Sea: An integrative morphometric and molecular approach. Zool. J. Linn. Soc. 2017. [Google Scholar] [CrossRef]
- Manzari, C.; Fosso, B.; Marzano, M.; Annese, A.; Caprioli, R.; D’Erchia, A.M.; Gissi, C.; Intranuovo, M.; Picardi, E.; Santamaria, M.; et al. The influence of invasive jellyfish blooms on the aquatic microbiome in a coastal lagoon (Varano, SE Italy) detected by an Illumina-based deep sequencing strategy. Biol. Invasions 2015, 17, 923–940. [Google Scholar] [CrossRef]
- Hamner, W.M.; Dawson, M.N. A review and synthesis on the systematics and evolution of jellyfish blooms: Advantageous aggregations and adaptive assemblages. Hydrobiologia 2009, 616, 161–191. [Google Scholar] [CrossRef]
- Falkenhaug, T. Review of Jellyfish Blooms in the Mediterranean and Black Sea. Mar. Biol. Res. 2014, 10, 1038–1039. [Google Scholar] [CrossRef]
- Rossi, S.; Isla, E.; Bosch-Belmar, M.; Galli, G.; Gori, A.; Gristina, M.; Ingrosso, G.; Milisenda, G.; Piraino, S.; Rizzo, L.; et al. Changes of energy fluxes in marine animal forests of the Anthropocene: Factors shaping the future seascape. ICES J. Mar. Sci. 2019, 76, 2008–2019. [Google Scholar] [CrossRef]
- Boero, F.; Brotz, L.; Gibbons, M.J.; Piraino, S.; Zampardi, S. 3.10 Impacts and effects of ocean warming on jellyfish. In Explaining Ocean Warming: Causes, Scale, Effects and Consequences; IUCN: Gland, Switzerland, 2016. [Google Scholar]
- Milisenda, G.; Martinez-Quintana, A.; Fuentes, V.L.; Bosch-Belmar, M.; Aglieri, G.; Boero, F.; Piraino, S. Reproductive and bloom patterns of Pelagia noctiluca in the Strait of Messina, Italy. Estuar. Coast. Shelf Sci. 2018, 201, 29–39. [Google Scholar] [CrossRef]
- Angilè, F.; Del Coco, L.; Girelli, C.R.; Basso, L.; Rizzo, L.; Piraino, S.; Stabili, L.; Fanizzi, F.P. 1H NMR Metabolic Profile of Scyphomedusa Rhizostoma pulmo (Scyphozoa, Cnidaria) in Female Gonads and Somatic Tissues: Preliminary Results. Molecules 2020, 25, 806. [Google Scholar] [CrossRef]
- Basso, L.; Papadia, P.; Rizzo, L.; Migoni, D.; Fanizzi, F.P.; Piraino, S. Trace Metals Do Not Accumulate Over Time in The Edible Mediterranean Jellyfish Rhizostoma pulmo (Cnidaria, Scyphozoa) from Urban Coastal Waters. Water 2021, 13, 1410. [Google Scholar] [CrossRef]
- Stabili, L.; Fraschetti, S.; Acquaviva, M.I.; Cavallo, R.A.; De Pascali, S.A.; Fanizzi, F.P.; Gerardi, C.; Narracci, M.; Rizzo, L. The Potential Exploitation of the Mediterranean Invasive Alga Caulerpa cylindracea: Can the Invasion Be Transformed into a Gain? Mar. Drugs 2016, 14, 210. [Google Scholar] [CrossRef] [PubMed]
- Pham-Huy, L.A.; He, H.; Pham-Huy, C. Free radicals, antioxidants in disease and health. Int. J. Biomed. Sci. IJBS 2008, 4, 89. [Google Scholar] [PubMed]
- Leone, A.; Lecci, R.M.; Milisenda, G.; Piraino, S. Mediterranean jellyfish as novel food: Effects of thermal processing on antioxidant, phenolic and protein contents. Eur. Food Res. Technol. 2019. [Google Scholar] [CrossRef]
- Leone, A.; Lecci, R.M.; Durante, M.; Piraino, S. Extract from the zooxanthellate jellyfish Cotylorhiza tuberculata modulates gap junction intercellular communication in human cell cultures. Mar. Drugs 2013, 11, 1728–1762. [Google Scholar] [CrossRef] [PubMed]
- De Rinaldis, G.; Leone, A.; De Domenico, S.; Bosch-Belmar, M.; Slizyte, R.; Milisenda, G.; Santucci, A.; Albano, C.; Piraino, S. Biochemical Characterization of Cassiopea andromeda (Forsskål, 1775), Another Red Sea Jellyfish in the Western Mediterranean Sea. Mar. Drugs 2021, 19, 498. [Google Scholar] [CrossRef]
- Li, A.N.; Li, S.; Zhang, Y.J.; Xu, X.R.; Chen, Y.M.; Li, H.B. Resources and biological activities of natural polyphenols. Nutrients 2014, 6, 6020–6047. [Google Scholar] [CrossRef]
- Weichselbaum, E.; Buttriss, J.L. Polyphenols in the diet. Nutr. Bull. 2010, 35, 157–164. [Google Scholar] [CrossRef]
- Tsao, R. Chemistry and biochemistry of dietary polyphenols. Nutrients 2010, 2, 1231–1246. [Google Scholar] [CrossRef]
- Najafian, L.; Babji, A.S. A review of fish-derived antioxidant and antimicrobial peptides: Their production, assessment, and applications. Peptides 2012, 33, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Dhainaut, A.; Scaps, P. Immune defense and biological responses induced by toxics in Annelida. Can. J. Zool. 2001, 79, 233–253. [Google Scholar] [CrossRef]
- Paul, V.J.; Puglisi, M.P. Chemical mediation of interactions among marine organisms. Nat. Prod. Rep. 2004, 21, 189. [Google Scholar] [CrossRef]
- Dobretsov, S.; Dahms, H.U.; Qian, P.Y. Antibacterial and anti-diatom activity of Hong Kong sponges. Aquat. Microb. Ecol. 2005, 38, 191–201. [Google Scholar] [CrossRef][Green Version]
- Kelly, S.R.; Garo, E.; Jensen, P.R.; Fenical, W.; Pawlik, J.R. Effects of Caribbean sponge secondary metabolites on bacterial surface colonization. Aquat. Microb. Ecol. 2005, 40, 191–203. [Google Scholar] [CrossRef]
- Stabili, L.; Licciano, M.; Pagliara, P. Evidence of antibacterial and lysozyme-like activity in different planktonic larval stages of Paracentrotus lividus. Mar. Biol. 1994, 119, 501–505. [Google Scholar] [CrossRef]
- Stabili, L.; Miglietta, A.M.; Belmonte, G. Lysozyme-like and trypsin-like activities in the cysts of Artemia franciscana Kellog, 1906. J. Exp. Mar. Bio. Ecol. 1999, 237, 291–303. [Google Scholar] [CrossRef]
- Suganthi, K.; Bragadeeswaran, S. Antimicrobial and immunomodulatory activities of jellyfish (Chrysaora quinquecirrha) venom. In Prospects in Bioscience: Addressing the Issues; Springer: Berlin, Germany, 2012; pp. 283–292. [Google Scholar]
- Radwan, F.F.Y.; Gershwin, L.; Burnett, J.W. Toxinological studies on the nematocyst venom of Chrysaora achlyos. Toxicon 2000, 38, 1581–1591. [Google Scholar] [CrossRef]
- Stabili, L.; Canicattì, C. Antibacterial activity of the seminal plasma of Paracentrotus lividus. Can. J. Zool. 1994, 72, 1211–1216. [Google Scholar] [CrossRef]
- Marcano, L.; Nusetti, O.; Rodríguez-Grau, J.; Briceño, J.; Vilas, J. Coelomic Fluid Lysozyme Activity Induction in the Polychaete Eurythoe complanata as a Biomarker of Heavy Metal Toxicity. Bull. Environ. Contam. Toxicol. 1997, 59, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Zachary, D.; Hoffmann, D. Lysozyme is stored in the granules of certain haemocyte types in Locusta. J. Insect Physiol. 1984, 30, 405–411. [Google Scholar] [CrossRef]
- Olsen, A.M.; Nilsen, I.W.; Sletten, K.; Myrnes, B. Multiple invertebrate lysozymes in blue mussel (Mytilus edulis). Comp. Biochem. Physiol. 2003, B136, 107–115. [Google Scholar] [CrossRef]
- Xue, Q.-G.; Schey, K.L.; Volety, A.K.; Chu, F.-L.E.; La Peyre, J.F. Purification and characterization of lysozyme from plasma of the eastern oyster (Crassostrea virginica). Comp. Biochem. Physiol. Part B Biochem. Mol. Biol. 2004, 139, 11–25. [Google Scholar] [CrossRef]
- Zhang, L.; He, Q.; Wang, Q.; Zhang, B.; Wang, B.; Xu, F.; Wang, T.; Xiao, L.; Zhang, L. Intracellular Ca2+ Overload Induced by Extracellular Ca2+ Entry Plays an Important Role in Acute Heart Dysfunction by Tentacle Extract from the Jellyfish Cyanea capillata. Cardiovasc. Toxicol. 2014, 14, 260–274. [Google Scholar] [CrossRef] [PubMed]
- Lazcano-Pérez, F.; Arellano, R.O.; Garay, E.; Arreguín-Espinosa, R.; Sánchez-Rodríguez, J. Electrophysiological activity of a neurotoxic fraction from the venom of box jellyfish Carybdea marsupialis. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2017, 191, 177–182. [Google Scholar] [CrossRef]
- Sánchez-Rodríguez, J.; Torrens, E.; Segura-Puertas, L. Partial purification and characterization of a novel neurotoxin and three cytolysins from box jellyfish (Carybdea marsupialis) nematocyst venom. Arch. Toxicol. 2006, 80, 163–168. [Google Scholar] [CrossRef] [PubMed]
- Stabili, L.; Schirosi, R.; Licciano, M.; Giangrande, A. The mucus of Sabella spallanzanii (Annelida, Polychaeta): Its involvement in chemical defence and fertilization success. J. Exp. Mar. Bio. Ecol. 2009, 374, 144–149. [Google Scholar] [CrossRef]
- Calow, P. Why Some Metazoan Mucus Secretions are More Susceptible to Microbial Attack than Others. Am. Nat. 1979, 114, 149–152. [Google Scholar] [CrossRef]
- Coffroth, M.A. Mucous sheet formation on poritid corals: An evaluation of coral mucus as a nutrient source on reefs. Mar. Biol. 1990, 105, 39–49. [Google Scholar] [CrossRef]
- Flatt, P.M.; Gautschi, J.T.; Thacker, R.W.; Musafija-Girt, M.; Crews, P.; Gerwick, W.H. Identification of the cellular site of polychlorinated peptide biosynthesis in the marine sponge Dysidea (Lamellodysidea) herbacea and symbiotic cyanobacterium Oscillatoria spongeliae by CARD-FISH analysis. Mar. Biol. 2005, 147, 761–774. [Google Scholar] [CrossRef]
- Lopanik, N.; Lindquist, N.; Targett, N. Potent cytotoxins produced by a microbial symbiont protect host larvae from predation. Oecologia 2004, 139, 131–139. [Google Scholar] [CrossRef]
- Ridley, C.P.; Bergquist, P.R.; Harper, M.K.; Faulkner, D.J.; Hooper, J.N.A.; Haygood, M.G. Speciation and Biosynthetic Variation in Four Dictyoceratid Sponges and Their Cyanobacterial Symbiont, Oscillatoria spongeliae. Chem. Biol. 2005, 12, 397–406. [Google Scholar] [CrossRef] [PubMed]
- Reshef, L.; Koren, O.; Loya, Y.; Zilber-Rosenberg, I.; Rosenberg, E. The Coral Probiotic Hypothesis. Environ. Microbiol. 2006, 8, 2068–2073. [Google Scholar] [CrossRef]
- Ritchie, K.B.; Smith, G.W. Microbial Communities of Coral Surface Mucopolysaccharide Layers. In Coral Health and Disease; Springer: Berlin/Heidelberg, Germany, 2004; pp. 259–264. [Google Scholar]
- Rohwer, F.; Kelley, S. Culture-Independent Analyses of Coral-Associated Microbes. In Coral Health and Disease; Springer: Berlin/Heidelberg, Germany, 2004; pp. 265–277. [Google Scholar]
- Derby, C.D. Escape by Inking and Secreting: Marine Molluscs Avoid Predators Through a Rich Array of Chemicals and Mechanisms. Biol. Bull. 2007, 213, 274–289. [Google Scholar] [CrossRef]
- Stabili, L.; Schirosi, R.; Licciano, M.; Giangrande, A. Role of Myxicola infundibulum (Polychaeta, Annelida) mucus: From bacterial control to nutritional home site. J. Exp. Mar. Biol. Ecol. 2014, 461, 344–349. [Google Scholar] [CrossRef]
- Brown, B.E.; Bythell, J.C. Perspectives on mucus secretion in reef corals. Mar. Ecol. Prog. Ser. 2005, 296, 291–309. [Google Scholar] [CrossRef]
- Wang, W.; Sun, J.; Liu, C.; Xue, Z. Application of immunostimulants in aquaculture: Current knowledge and future perspectives. Aquac. Res. 2017, 48, 1–23. [Google Scholar] [CrossRef]
- Hollis, A.; Ahmed, Z. The path of least resistance: Paying for antibiotics in non-human uses. Health Policy 2014, 118, 264–270. [Google Scholar] [CrossRef] [PubMed]
- Bao, P.; Sun, X.; Liu, Q.; Zhang, Y.; Liu, X. Synergistic effect of a combined live Vibrio anguillarum and Edwardsiella piscicida vaccine in turbot. Fish Shellfish Immun. 2019, 88, 84–90. [Google Scholar] [CrossRef]
- Ragland, S.A.; Criss, A.K. From bacterial killing to immune modulation: Recent insights into the functions of lysozyme. PLoS Pathog. 2017, 13, e1006512. [Google Scholar] [CrossRef] [PubMed]
- Labro, M.T. The prohost effect of antimicrobial agents as a predictor of clinical outcome. J. Chemother. 1997, 9, 100. [Google Scholar] [CrossRef] [PubMed]
- Kreuser, E.D.; Keppler, B.K.; Berdel, W.E.; Piest, A.; Thiel, E. Synergistic antitumor interactions between newly synthesized ruthenium complexes and cytokines in human colon carcinoma cell lines. Semin. Oncol. 1992, 19 (Suppl. 3), 73–81. [Google Scholar] [PubMed]
- Barresi, G.; Di Carlo, E.; Trapani, M.R.; Parisi, M.G.; Chille, C.; Mule, M.F.; Cammarata, M.; Palla, F. Marine organisms as source of bioactive molecules applied in restoration projects. Herit. Sci. 2015, 3. [Google Scholar] [CrossRef]
- Konstantinou, I.K.; Albanis, T.A. Worldwide occurrence and effects of antifouling paint booster biocides in the aquatic environment: A review. Environ. Int. 2004, 30, 235–248. [Google Scholar] [CrossRef]
- Ostroumov, S.A. On the concepts of biochemical ecology and hydrobiology: Ecological chemomediators. Contemp. Probl. Ecol. 2008, 1, 238–244. [Google Scholar] [CrossRef]
- Jelic-Mrcelic, G.; Sliskovic, M.; Antolic, B. Biofouling communities on test panels coated with TBT and TBT-free copper based antifouling paints. Biofouling 2006, 22, 293–302. [Google Scholar] [CrossRef]
- Turley, P.A.; Fenn, R.J.; Ritter, J.C.; Callow, M.E. Pyrithiones as antifoulants: Environmental fate and loss of toxicity. Biofouling 2005, 21, 31–40. [Google Scholar] [CrossRef]
- Longo, C.; Leo, L.; Leone, A. Carotenoids, fatty acid composition and heat stability of supercritical carbon dioxide-extracted-oleoresins. Int. J. Mol. Sci. 2012, 13, 4233–4254. [Google Scholar] [CrossRef]
- Canicatti, C.; Roch, P. Studies on Holothuria polii (Echinodermata) antibacterial proteins. I. Evidence for and activity of a coelomocyte lysozyme. Experientia 1989, 45, 756–759. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Core Team: Vienna, Austria, 2017. [Google Scholar]
- Canty, A.; Ripley, B. Boot: Bootstrap R (S-plus) Functions. Package Version 1.3–20. CRAN R Project. 2017. Available online: https://cran.r-project.org/web/packages/boot/index.html (accessed on 28 October 2021).
- Davison, A.C.; Hinkley, D.V. Bootstrap Methods and Their Application; Cambridge University Press: Cambridge, UK, 1997. [Google Scholar]
- Anderson, M.J. A new method for non-parametric multivariate analysis of variance. Austral Ecol. 2001, 26, 32–46. [Google Scholar] [CrossRef]
- Anderson, M.; Braak, C. Ter Permutation tests for multi-factorial analysis of variance. J. Stat. Comput. Simul. 2003, 73, 85–113. [Google Scholar] [CrossRef]
- Anderson, M.J.; Gorley, R.N.; Clarke, K.R. PERMANOVA+ for PRIMER: Guide to Software and Statistical Methods; PRIMER-E: Plymouth, UK, 2015. [Google Scholar]

| Source | df | MS | Pseudo-F | P(perm) |
|---|---|---|---|---|
| Co | 2 | 7.79 | 60.55 | |
| pH | 4 | 12.78 | 99.32 | |
| Co x pH | 8 | 2.24 | 17.45 | *** |
| Res | 30 | 0.13 | ||
| Total | 44 |
| Source | df | MS | Pseudo-F | P(perm) |
|---|---|---|---|---|
| Co | 2 | 3.40 | 24.91 | |
| T | 3 | 26.91 | 197.33 | |
| Co x T | 6 | 1.06 | 7.81 | *** |
| Res | 24 | 0.14 | ||
| Total | 35 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stabili, L.; Rizzo, L.; Caprioli, R.; Leone, A.; Piraino, S. Jellyfish Bioprospecting in the Mediterranean Sea: Antioxidant and Lysozyme-Like Activities from Aurelia coerulea (Cnidaria, Scyphozoa) Extracts. Mar. Drugs 2021, 19, 619. https://doi.org/10.3390/md19110619
Stabili L, Rizzo L, Caprioli R, Leone A, Piraino S. Jellyfish Bioprospecting in the Mediterranean Sea: Antioxidant and Lysozyme-Like Activities from Aurelia coerulea (Cnidaria, Scyphozoa) Extracts. Marine Drugs. 2021; 19(11):619. https://doi.org/10.3390/md19110619
Chicago/Turabian StyleStabili, Loredana, Lucia Rizzo, Rosa Caprioli, Antonella Leone, and Stefano Piraino. 2021. "Jellyfish Bioprospecting in the Mediterranean Sea: Antioxidant and Lysozyme-Like Activities from Aurelia coerulea (Cnidaria, Scyphozoa) Extracts" Marine Drugs 19, no. 11: 619. https://doi.org/10.3390/md19110619
APA StyleStabili, L., Rizzo, L., Caprioli, R., Leone, A., & Piraino, S. (2021). Jellyfish Bioprospecting in the Mediterranean Sea: Antioxidant and Lysozyme-Like Activities from Aurelia coerulea (Cnidaria, Scyphozoa) Extracts. Marine Drugs, 19(11), 619. https://doi.org/10.3390/md19110619










