Chondracanthus teedei var. lusitanicus: The Nutraceutical Potential of an Unexploited Marine Resource
Abstract
:1. Introduction
2. Results
2.1. Macro- and Micro-Element Profile
2.2. Nutritional Evaluation
2.3. Polyssacharides from C. teedei var. lusitanicus
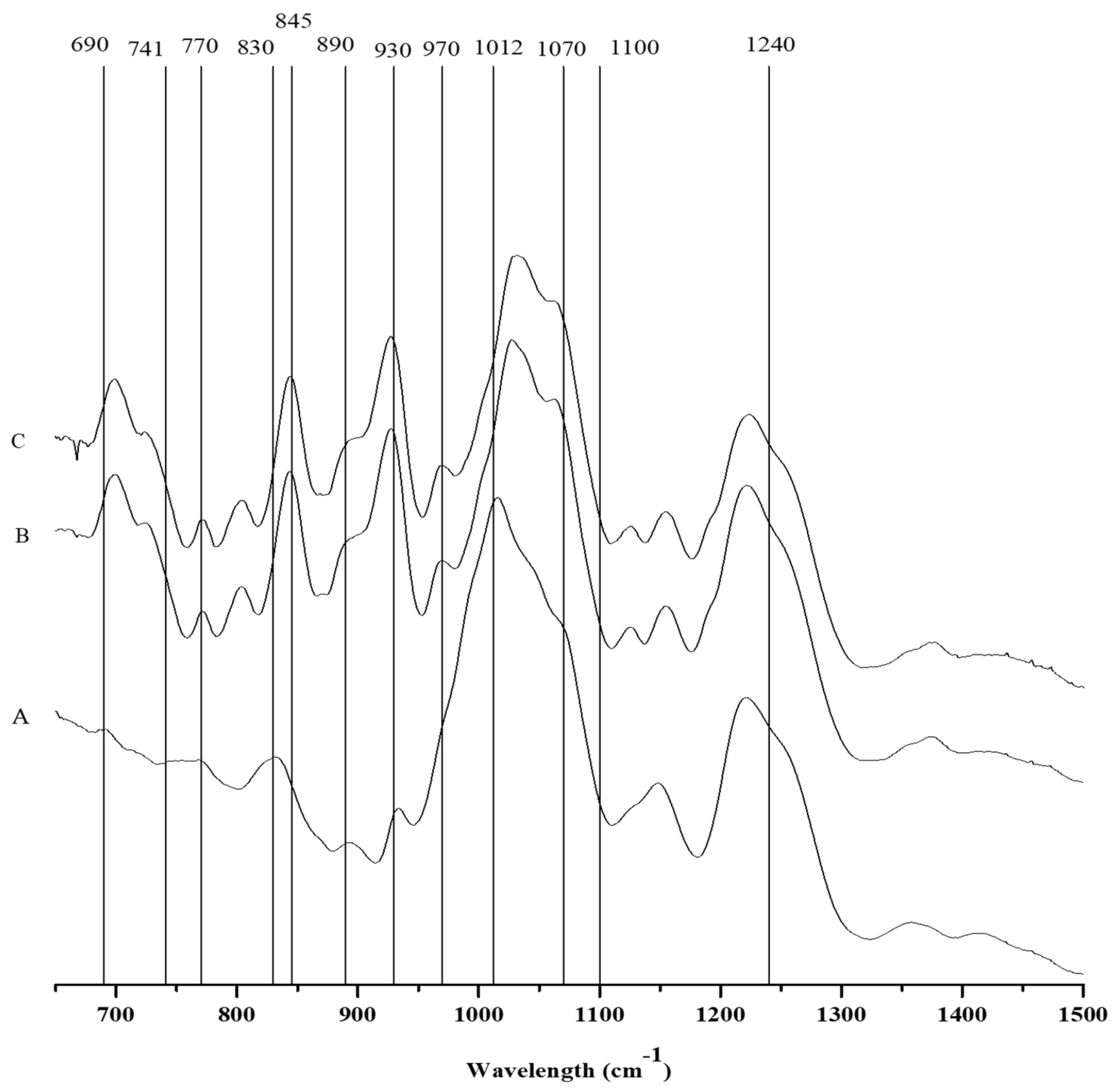
2.3.1. FTIR-ATR
2.3.2. 1H-RMN
2.3.3. 13C-RMN
3. Discussion
4. Materials and Methods
4.1. Seaweed Harvesting and Preparation
4.2. Mineral and Trace Element Characterization
4.3. Nutritional Profile
4.3.1. Moisture and Ashes Content
4.3.2. Crude Lipids
4.3.3. Total Nitrogen/Protein
4.3.4. Crude Fiber and Total Carbohydrates/Nitrogen-Free Extractives
4.4. Polyssacharides Characterization
4.4.1. Carrageenan Extraction
4.4.2. FTIR-ATR Characterization
4.4.3. 1H-NMR Characterization
4.4.4. 13C-NMR Characterization
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Leandro, A.; Pacheco, D.; Cotas, J.; Marques, J.C.; Pereira, L.; Gonçalves, A.M.M. Seaweed’s Bioactive Candidate Compounds to Food Industry and Global Food Security. Life 2020, 10, 140. [Google Scholar] [CrossRef] [PubMed]
- García-Poza, S.; Leandro, A.; Cotas, C.; Cotas, J.; Marques, J.C.; Pereira, L.; Gonçalves, A.M.M. The Evolution Road of Seaweed Aquaculture: Cultivation Technologies and the Industry 4.0. Int. J. Environ. Res. Public Health 2020, 17, 6528. [Google Scholar] [CrossRef] [PubMed]
- Denis, C.; Morançais, M.; Li, M.; Deniaud, E.; Gaudin, P.; Wielgosz-Collin, G.; Barnathan, G.; Jaouen, P.; Fleurence, J. Study of the chemical composition of edible red macroalgae Grateloupia turuturu from Brittany (France). Food Chem. 2010, 119, 913–917. [Google Scholar] [CrossRef]
- Milinovic, J.; Mata, P.; Diniz, M.; Noronha, J.P. Umami taste in edible seaweeds: The current comprehension and perception. Int. J. Gastron. Food Sci. 2021, 23, 100301. [Google Scholar] [CrossRef]
- Kılınç, B.; Cirik, S.; Turan, G.; Klnc, B.; Cirik, S.; Turan, G.; Tekogul, H.; Koru, E.; Kılınç, B.; Cirik, S.; et al. Seaweeds for Food and Industrial Applications. In Food Industry; Intechopen: London, UK, 2013; pp. 735–748. [Google Scholar] [CrossRef] [Green Version]
- Fleurence, J. Seaweeds as Food. In Seaweed in Health and Disease Prevention; Elsevier: Amsterdam, The Netherlands, 2016; pp. 149–167. ISBN 9780128027936. [Google Scholar]
- Love, R. Optimisation of the culture of the red algae Chondrachantus teedei. In Effects of Irradiance, Temperature, Salinity and Nutrient Enrichment; Facultad de ciencias del mar y ambientales, Universidad de Cádiz: Cadiz, Spain, 2018. [Google Scholar]
- Fleurence, J. Seaweed proteins: Biochemical, nutritional aspects and potential uses. Trends Food Sci. Technol. 1999, 10, 25–28. [Google Scholar] [CrossRef]
- Pereira, L. A review of the nutrient composition of selected edible seaweeds. In Seaweed: Ecology, Nutrient Composition and Medicinal Uses; Nova Science Publishers: Hauppauge, NY, USA, 2011; ISBN 9781614708780. [Google Scholar]
- Davis, G.D.J.; Vasanthi, A.H.R. Seaweed metabolite database (SWMD): A database of natural compounds from marine algae. Bioinformation 2011, 5, 361–364. [Google Scholar] [CrossRef] [Green Version]
- Nunraksa, N.; Rattanasansri, S.; Praiboon, J.; Chirapart, A. Proximate composition and the production of fermentable sugars, levulinic acid, and HMF from Gracilaria fisheri and Gracilaria tenuistipitata cultivated in earthen ponds. J. Appl. Phycol. 2019, 31, 683–690. [Google Scholar] [CrossRef]
- Ganesan, A.R.; Tiwari, U.; Rajauria, G. Seaweed nutraceuticals and their therapeutic role in disease prevention. Food Sci. Hum. Wellness 2019, 8, 252–263. [Google Scholar] [CrossRef]
- Hannan, M.A.; Dash, R.; Haque, M.N.; Mohibbullah, M.; Sohag, A.A.M.; Rahman, M.A.; Uddin, M.J.; Alam, M.; Moon, I.S. Neuroprotective Potentials of Marine Algae and Their Bioactive Metabolites: Pharmacological Insights and Therapeutic Advances. Mar. Drugs 2020, 18, 347. [Google Scholar] [CrossRef]
- Cherry, P.; O’Hara, C.; Magee, P.J.; McSorley, E.M.; Allsopp, P.J. Risks and benefits of consuming edible seaweeds. Nutr. Rev. 2019, 77, 307–329. [Google Scholar] [CrossRef] [Green Version]
- Mišurcová, L.; Machů, L.; Orsavová, J. Seaweed Minerals as Nutraceuticals. In Advances in Food and Nutrition Research; Kim, S.-K., Ed.; Academic Press: Cambridge, MA, USA, 2011; pp. 371–390. [Google Scholar]
- National Research Council (U.S.). Committee on Diet and Health. Diet and Health: Implications for Reducing Chronic Disease Risk/Committee on Diet and Health, Food and Nutrition Board, Commission on Life Sciences, National Research Council; National Academy Press: Washington, DC, USA, 1989; ISBN 0-309-03994-0. [Google Scholar]
- Lamb, M.W.; Harden, M.L. Mineral Elements in Human Nutrition. In The Meaning of Human Nutrition; Lamb, M.W., Harden, M.L., Eds.; Elsevier: Amsterdam, The Netherlands, 1973; pp. 193–210. [Google Scholar]
- Doley, J. Vitamins and Minerals in Older Adults. In Nutrition and Functional Foods for Healthy Aging; Watson, R.R., Ed.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 125–137. ISBN 978-0-12-805376-8. [Google Scholar]
- British Nutrition Foundation. Average Daily Intakes of Vitamins from Food Sources Only: % Below the Lower Reference Nutrient Intake (LRNI), by Age and Sex. Available online: https://www.nutrition.org.uk/attachments/article/255/LRNIvitaminsandminerals.pdf (accessed on 14 September 2021).
- Bermejo, R.; Macías, M.; Cara, C.L.; Sánchez-García, J.; Hernández, I. Culture of Chondracanthus teedei and Gracilariopsis longissima in a traditional salina from southern Spain. J. Appl. Phycol. 2019, 31, 561–573. [Google Scholar] [CrossRef]
- Bermejo, R.; Cara, C.L.; Macías, M.; Sánchez-García, J.; Hernández, I. Growth rates of Gracilariopsis longissima, Gracilaria bursa-pastoris and Chondracanthus teedei (Rhodophyta) cultured in ropes: Implication for N biomitigation in Cadiz Bay (Southern Spain). J. Appl. Phycol. 2020, 32, 1879–1891. [Google Scholar] [CrossRef]
- Pereira, L.; Silva, P. A concise review of the red macroalgae Chondracanthus teedei (Mertens ex Roth) Kützing and Chondracanthus teedei var. lusitanicus (J.E. De Mesquita Rodrigues) Bárbara & Cremades. J. Appl. Phycol. 2021, 33, 111–131. [Google Scholar] [CrossRef]
- Guiry, M.D.; Guiry, G.M. AlgaeBase. World-Wide Electronic Publication. Available online: https://www.algaebase.org (accessed on 23 July 2021).
- Soares, F.; Fernandes, C.; Silva, P.; Pereira, L.; Gonçalves, T. Antifungal activity of carrageenan extracts from the red alga Chondracanthus teedei var. lusitanicus. J. Appl. Phycol. 2016, 28, 2991–2998. [Google Scholar] [CrossRef]
- Pereira, L. (Ed.) Edible Seaweeds of the World; CRC Press: Boca Raton, FL, USA, 2016; ISBN 9780429154041. [Google Scholar]
- Soares, F. Antifungal, Antibacterial and Antiviral Activity of Chondracanthus teedei var. Lusitanicus (Gigartinaceae, Rhodophyta). Master’s Thesis, University of Coimbra, Coimbra, Portugal, 2015. [Google Scholar]
- Cotas, J.; Pacheco, D.; Araujo, G.S.; Valado, A.; Critchley, A.T.; Pereira, L. On the Health Benefits vs. Risks of Seaweeds and Their Constituents: The Curious Case of the Polymer Paradigm. Mar. Drugs 2021, 19, 164. [Google Scholar] [CrossRef] [PubMed]
- Pereira, L.; Mesquita, J.F. Carrageenophytes of occidental Portuguese coast: 1-spectroscopic analysis in eight carrageenophytes from Buarcos bay. Biomol. Eng. 2003, 20, 217–222. [Google Scholar] [CrossRef] [Green Version]
- Pereira, L.; Amado, A.M.; Critchley, A.T.; van de Velde, F.; Ribeiro-Claro, P.J.A. Identification of selected seaweed polysaccharides (phycocolloids) by vibrational spectroscopy (FTIR-ATR and FT-Raman). Food Hydrocoll. 2009, 23, 1903–1909. [Google Scholar] [CrossRef] [Green Version]
- Pereira, L.; Gheda, S.F.; Ribeiro-Claro, P.J.A. Analysis by Vibrational Spectroscopy of Seaweed Polysaccharides with Potential Use in Food, Pharmaceutical, and Cosmetic Industries. Int. J. Carbohydr. Chem. 2013, 2013, 537202. [Google Scholar] [CrossRef]
- Pereira, L.; Critchley, A.T.; Amado, A.M.; Ribeiro-Claro, P.J.A. A comparative analysis of phycocolloids produced by underutilized versus industrially utilized carrageenophytes (Gigartinales, Rhodophyta). J. Appl. Phycol. 2009, 21, 599–605. [Google Scholar] [CrossRef]
- Pereira, L.; Mesquita, J.F. Population studies and carrageenan properties of Chondracanthus teedei var. lusitanicus (Gigartinaceae, Rhodophyta). J. Appl. Phycol. 2004, 16, 369–383. [Google Scholar] [CrossRef]
- Knutsen, S.H.; Grasdalen, H. Characterization of Water-extractable Polysaccharides from Norwegian Furcellaria lumbricalis (Huds.) Lamour. (Gigartinales, Rhodophyceae) by IR and NMR Spectroscopy. Bot. Mar. 1987, 30, 497–505. [Google Scholar] [CrossRef]
- Van de Velde, F.; Knutsen, S.H.; Usov, A.I.; Rollema, H.S.; Cerezo, A.S. 1H and 13C high resolution NMR spectroscopy of carrageenans: Application in research and industry. Trends Food Sci. Technol. 2002, 13, 73–92. [Google Scholar] [CrossRef]
- Van de Velde, F.; Rollema, H.S.; Grinberg, N.V.; Burova, T.V.; Grinberg, V.Y.; Tromp, R.H. Coil-helix transition of?-carrageenan as a function of chain regularity. Biopolymers 2002, 65, 299–312. [Google Scholar] [CrossRef] [PubMed]
- Usov, A.I.; Yarotsky, S.V.; Shashkov, A.S. 13C-NMR spectroscopy of red algal galactans. Biopolymers 1980, 19, 977–990. [Google Scholar] [CrossRef]
- Falshaw, R.; Furneaux, R.H. Carrageenan from the tetrasporic stage of Gigartina decipiens (Gigartinaceae, Rhodophyta). Carbohydr. Res. 1994, 252, 171–182. [Google Scholar] [CrossRef]
- Falshaw, R.; Furneaux, R.H. Carrageenans from the tetrasporic stages of Gigartina clavifera and Gigartina alveata (Gigartinaceae, Rhodophyta). Carbohydr. Res. 1995, 276, 155–165. [Google Scholar] [CrossRef]
- Torres, P.; Nagai, A.; Teixeira, D.I.A.; Marinho-Soriano, E.; Chow, F.; dos Santos, D.Y.A.C. Brazilian native species of Gracilaria (Gracilariales, Rhodophyta) as a source of valuable compounds and as nutritional supplements. J. Appl. Phycol. 2019, 31, 3163–3173. [Google Scholar] [CrossRef]
- Alwaleed, E.A. Biochemical Composition and Nutraceutical Perspectives Red Sea Seaweeds. Am. J. Appl. Sci. 2019, 16, 346–354. [Google Scholar] [CrossRef]
- Palmieri, N.; Forleo, M.B. The potential of edible seaweed within the western diet. A segmentation of Italian consumers. Int. J. Gastron. Food Sci. 2020, 20, 100202. [Google Scholar] [CrossRef]
- Menezes, B.S.; Coelho, M.S.; Meza, S.L.R.; Salas-Mellado, M.; Souza, M.R.A.Z. Macroalgal biomass as an additional ingredient of bread. Int. Food Res. J. 2015, 22, 812–817. [Google Scholar]
- Zertuche-González, J.A.; Sandoval-Gil, J.M.; Rangel-Mendoza, L.K.; Gálvez-Palazuelos, A.I.; Guzmán-Calderón, J.M.; Yarish, C. Seasonal and interannual production of sea lettuce (Ulva sp.) in outdoor cultures based on commercial size ponds. J. World Aquac. Soc. 2021, 2021, 1–12. [Google Scholar] [CrossRef]
- Stévant, P.; Ólafsdóttir, A.; Déléris, P.; Dumay, J.; Fleurence, J.; Ingadóttir, B.; Jónsdóttir, R.; Ragueneau, É.; Rebours, C.; Rustad, T. Data on the sensory characteristics and chemical composition of the edible red seaweed dulse (Palmaria palmata) after dry and semi-dry storage. Data Brief 2020, 33, 106343. [Google Scholar] [CrossRef]
- Britton, D.; Schmid, M.; Revill, A.T.; Virtue, P.; Nichols, P.D.; Hurd, C.L.; Mundy, C.N. Seasonal and site-specific variation in the nutritional quality of temperate seaweed assemblages: Implications for grazing invertebrates and the commercial exploitation of seaweeds. J. Appl. Phycol. 2021, 33, 603–616. [Google Scholar] [CrossRef]
- Osman, M.E.H.; Abo-Shady, A.M.; Elshobary, M.E.; Abd El-Ghafar, M.O.; Abomohra, A.E.-F. Screening of seaweeds for sustainable biofuel recovery through sequential biodiesel and bioethanol production. Environ. Sci. Pollut. Res. 2020, 27, 32481–32493. [Google Scholar] [CrossRef] [PubMed]
- Roleda, M.Y.; Hurd, C.L. Seaweed nutrient physiology: Application of concepts to aquaculture and bioremediation. Phycologia 2019, 58, 552–562. [Google Scholar] [CrossRef] [Green Version]
- Afonso, C.; Correia, A.P.; Freitas, M.V.; Baptista, T.; Neves, M.; Mouga, T. Seasonal Changes in the Nutritional Composition of Agarophyton vermiculophyllum (Rhodophyta, Gracilariales) from the Center of Portugal. Foods 2021, 10, 1145. [Google Scholar] [CrossRef] [PubMed]
- Milinovic, J.; Campos, B.; Mata, P.; Diniz, M.; Noronha, J.P. Umami free amino acids in edible green, red, and brown seaweeds from the Portuguese seashore. J. Appl. Phycol. 2020, 32, 3331–3339. [Google Scholar] [CrossRef]
- Hamid, S.S.; Wakayama, M.; Soga, T.; Tomita, M. Drying and extraction effects on three edible brown seaweeds for metabolomics. J. Appl. Phycol. 2018, 30, 3335–3350. [Google Scholar] [CrossRef]
- Madden, M.; Mitra, M.; Ruby, D.; Schwarz, J. Seasonality of selected nutritional constituents of edible Delmarva seaweeds. J. Phycol. 2012, 48, 1289–1298. [Google Scholar] [CrossRef]
- Rocha, C.P.; Pacheco, D.; Cotas, J.; Marques, J.C.; Pereira, L.; Gonçalves, A.M.M. Seaweeds as Valuable Sources of Essential Fatty Acids for Human Nutrition. Int. J. Environ. Res. Public Health 2021, 18, 4968. [Google Scholar] [CrossRef]
- Francavilla, M.; Franchi, M.; Monteleone, M.; Caroppo, C. The red seaweed Gracilaria gracilis as a multi products source. Mar. Drugs 2013, 11, 3754–3776. [Google Scholar] [CrossRef] [Green Version]
- Institute of Medicine. Dietary Reference Intakes: The Essential Guide to Nutrient Requirements; Meyers, L.D., Hellwig, J.P., Otten, J.J., Eds.; National Academies Press: Washington, DC, USA, 2006. [Google Scholar]
- The European Parliament of the European Union Regulation (EU) 1169/2011 on the provision of food information to consumers. Off. J. Eur. Union 2011, 304, 18–63.
- Milinovic, J.; Rodrigues, C.; Diniz, M.; Noronha, J.P. Determination of total iodine content in edible seaweeds: Application of inductively coupled plasma-atomic emission spectroscopy. Algal Res. 2021, 53, 102149. [Google Scholar] [CrossRef]
- Cundy, T.; Grey, A.; Reid, I.R. Calcium, phosphate and magnesium. In Clinical Biochemistry: Metabolic and Clinical Aspects; Marshall, W.J., Day, A.P., Lapsley, M., Ayling, R.M., Eds.; Elsevier: Amsterdam, The Netherlands, 2014; pp. 93–123. [Google Scholar]
- Bhatia, V.; Kulkarni, A.; Nair, V.V. Disorders of Mineral and Bone Metabolism. In Practical Pediatric Endocrinology in a Limited Resource Setting; Zacharin, M., Ed.; Elsevier: Amsterdam, The Netherlands, 2013; pp. 159–186. [Google Scholar]
- Penney, M.D. Sodium, water and potassium. In Clinical Biochemistry: Metabolic and Clinical Aspects; Marshall, W.J., Day, P.D., Lapsley, M., Ayling, R.M., Eds.; Elsevier: Amsterdam, The Netherlands, 2014; pp. 27–64. [Google Scholar]
- Rao, M.K.; Anderson, S. The Kidney in Aging. In Primer on Kidney Diseases; Greenberg, A., Coffman, T.M., Cheung, A.K., Jennette, J.C., Falk, R.J., Eds.; Elsevier: Amsterdam, The Netherlands, 2009; pp. 413–419. [Google Scholar]
- Rude, R.K. Magnesium. In Encyclopedia of Dietary Supplements; Coates, P.M., Betz, J.M., Blackman, M.R., Cragg, G.M., Levine, M., Moss, J., White, J.D., Eds.; Informa Healthcare; Rude RK: New York, NY, USA, 2010; pp. 527–537. [Google Scholar]
- Rude, R.K. Magnesium. In Modern Nutrition in Health and Disease; Ross, A.C., Caballero, B., Cousins, R.J., Tucker, K.L., Ziegler, T.R., Eds.; Lippincott Williams & Wilkins: Baltimore, MD, USA, 2012; pp. 159–175. [Google Scholar]
- Whitbread, D. Top 10 Vegetables Highest in Magnesium. Available online: https://www.myfooddata.com/articles/high-magnesium-vegetables.php (accessed on 1 August 2021).
- Stone, M.; Martyn, L.; Weaver, C. Potassium Intake, Bioavailability, Hypertension, and Glucose Control. Nutrients 2016, 8, 444. [Google Scholar] [CrossRef] [Green Version]
- US Department of Health and Human Services. Potassium Fact Sheet for Health Professionals. Available online: https://ods.od.nih.gov/factsheets/Potassium-HealthProfessional/ (accessed on 18 August 2021).
- Whitbread, D. 20 Vegetables High in Potassium—A Ranking from Highest to Lowest. Available online: https://www.myfooddata.com/articles/high-potassium-vegetables.php (accessed on 18 August 2021).
- Nutrition Information for Raw Vegetables|FDA. Available online: https://www.fda.gov/food/food-labeling-nutrition/nutrition-information-raw-vegetables (accessed on 23 July 2021).
- Yates, A.A.; Schlicker, S.A.; Suitor, C.W. Dietary Reference Intakes. J. Am. Diet. Assoc. 1998, 98, 699–706. [Google Scholar] [CrossRef]
- Allen, L.; de Benoist, B.; Dary, O.; Hurrel, R. Guidelines on Food Fortification with Micronutrients; WHO: Geneva, Switzerland, 2006. [Google Scholar]
- Whitbread, D. Top 10 Foods Highest in Manganese. Available online: https://www.myfooddata.com/articles/foods-high-in-manganese.php (accessed on 2 July 2021).
- National Institute of Health Manganese—Health Professional Fact Sheet. Available online: https://ods.od.nih.gov/factsheets/Manganese-HealthProfessional/ (accessed on 2 July 2021).
- Marriott, B.P.; Birt, D.F.; Stallings, V.A.; Yates, A. Present Knowledge in Nutrition: Basic Nutrition and Metabolism; Academic Press: Cambridge, MA, USA, 2020; Volume 1. [Google Scholar]
- Li, L.; Yang, X. The Essential Element Manganese, Oxidative Stress, and Metabolic Diseases: Links and Interactions. Oxidative Med. Cell. Longev. 2018, 2018, 7580707. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aschner, J.L.; Aschner, M. Nutritional aspects of manganese homeostasis. Mol. Asp. Med. 2005, 26, 353–362. [Google Scholar] [CrossRef]
- Chen, P. Manganese metabolism in humans. Front. Biosci. 2018, 23, 4665. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Whitbread, D. Top 10 Foods Highest in Zinc. Available online: https://www.myfooddata.com/articles/high-zinc-foods.php (accessed on 2 July 2021).
- O’Connor, J.M.; Hannigan, B.M.; Strain, J.J.; Bonham, M. The immune system as a physiological indicator of marginal copper status? Br. J. Nutr. 2002, 87, 393–403. [Google Scholar] [CrossRef]
- Uriu-Adams, J.Y.; Keen, C.L. Copper, oxidative stress, and human health. Mol. Asp. Med. 2005, 26, 268–298. [Google Scholar] [CrossRef]
- Krishnaiah, D.; Sarbatly, R.; Prasad, D.M.R.; Bono, A. Mineral Content of Some Seaweeds from Sabah’s South China Sea. Asian J. Sci. Res. 2008, 1, 166–170. [Google Scholar] [CrossRef] [Green Version]
- Clark, M.J.; Slavin, J.L. The Effect of Fiber on Satiety and Food Intake: A Systematic Review. J. Am. Coll. Nutr. 2013, 32, 200–211. [Google Scholar] [CrossRef] [PubMed]
- Salehi, B.; Sharifi-Rad, J.; Seca, A.M.; Pinto, D.C.; Michalak, I.; Trincone, A.; Mishra, A.P.; Nigam, M.; Zam, W.; Martins, N. Current Trends on Seaweeds: Looking at Chemical Composition, Phytopharmacology, and Cosmetic Applications. Molecules 2019, 24, 4182. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lozano Muñoz, I.; Díaz, N.F. Minerals in edible seaweed: Health benefits and food safety issues. Crit. Rev. Food Sci. Nutr. 2020, 1–16. [Google Scholar] [CrossRef]
- Pereira, L. Estudos em Macroalgas Carragenófitas (Gigartinales, Rhodophyceae) da Costa Portuguesa—Aspectos Ecológicos, Bioquímicos e Citológicos. Ph.D. Thesis, University of Coimbra, Coimbra, Portugal, 2004. [Google Scholar]
- Chopin, T.; Kerin, B.F.; Mazerolle, R. Phycocolloid chemistry as a taxonomic indicator of phylogeny in the Gigartinales, Rhodophyceae: A review and current developments using Fourier transform infrared diffuse reflectance spectroscopy. Phycol. Res. 1999, 47, 167–188. [Google Scholar] [CrossRef]
- Zinoun, M.; Cosson, J.; Deslandes, E. Influence of Culture Conditions on Growth and Physicochemical Properties of Carrageenans in Gigartina teedii (Rhodophyceae—Gigartinales). Bot. Mar. 1993, 36, 131–136. [Google Scholar] [CrossRef]
- Scientific Opinion on Dietary Reference Values for carbohydrates and dietary fibre. EFSA J. 2010, 8, 1462. [CrossRef] [Green Version]
- Teas, J. Dietary Seaweed and Breast Cancer: A Randomized Trial. Available online: https://clinicaltrials.gov/ct2/show/NCT01663792 (accessed on 3 October 2021).
- Tuohy, K.M.; Probert, H.M.; Smejkal, C.W.; Gibson, G.R. Using probiotics and prebiotics to improve gut health. Drug Discov. Today 2003, 8, 692–700. [Google Scholar] [CrossRef]
- Rowland, I. Optimal nutrition: Fibre and phytochemicals. Proc. Nutr. Soc. 1999, 58, 415–419. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA). Scientific Opinion on the substantiation of health claims related to dietary fibre (ID 744, 745, 746, 748, 749, 753, 803, 810, 855, 1415, 1416, 4308, 4330) pursuant to Article 13(1) of Regulation (EC) No 1924/2006. EFSA J. 2010, 8, 1735. [Google Scholar] [CrossRef]
- Clark, M.J.; Slavin, J.L. Scientific Opinion on the substantiation of health claims related to the replacement of mixtures of saturated fatty acids (SFAs) as present in foods or diets with mixtures of monounsaturated fatty acids (MUFAs) and/or mixtures of polyunsaturated fatty aci. EFSA J. 2011, 9, 2069. [Google Scholar] [CrossRef]
- Gibson, G.R.; Hutkins, R.; Sanders, M.E.; Prescott, S.L.; Reimer, R.A.; Salminen, S.J.; Scott, K.; Stanton, C.; Swanson, K.S.; Cani, P.D.; et al. Expert consensus document: The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 491–502. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huebbe, P.; Nikolai, S.; Schloesser, A.; Herebian, D.; Campbell, G.; Glüer, C.-C.; Zeyner, A.; Demetrowitsch, T.; Schwarz, K.; Metges, C.C.; et al. An extract from the Atlantic brown algae Saccorhiza polyschides counteracts diet-induced obesity in mice via a gut related multi-factorial mechanisms. Oncotarget 2017, 8, 73501–73515. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, J.; Kandasamy, S.; Zhang, J.; Kirby, C.W.; Karakach, T.; Hafting, J.; Critchley, A.T.; Evans, F.; Prithiviraj, B. Prebiotic effects of diet supplemented with the cultivated red seaweed Chondrus crispus or with fructo-oligo-saccharide on host immunity, colonic microbiota and gut microbial metabolites. BMC Complement. Altern. Med. 2015, 15, 279. [Google Scholar] [CrossRef] [PubMed]
- Cherry, P.; Yadav, S.; Strain, C.R.; Allsopp, P.J.; McSorley, E.M.; Ross, R.P.; Stanton, C. Prebiotics from Seaweeds: An Ocean of Opportunity? Mar. Drugs 2019, 17, 327. [Google Scholar] [CrossRef] [Green Version]
- Beaumont, M.; Tran, R.; Vera, G.; Niedrist, D.; Rousset, A.; Pierre, R.; Shastri, V.P.; Forget, A. Hydrogel-Forming Algae Polysaccharides: From Seaweed to Biomedical Applications. Biomacromolecules 2021, 22, 1027–1052. [Google Scholar] [CrossRef]
- Morais, T.; Cotas, J.; Pacheco, D.; Pereira, L. Seaweeds Compounds: An Ecosustainable Source of Cosmetic Ingredients? Cosmetics 2021, 8, 8. [Google Scholar] [CrossRef]
- Pacheco-Quito, E.-M.; Ruiz-Caro, R.; Veiga, M.-D. Carrageenan: Drug Delivery Systems and Other Biomedical Applications. Mar. Drugs 2020, 18, 583. [Google Scholar] [CrossRef]
- Brown, E.M.; Allsopp, P.J.; Magee, P.J.; Gill, C.I.; Nitecki, S.; Strain, C.R.; McSorley, E.M. Seaweed and human health. Nutr. Rev. 2014, 72, 205–216. [Google Scholar] [CrossRef]
- Liu, L.; Heinrich, M.; Myers, S.; Dworjanyn, S.A. Towards a better understanding of medicinal uses of the brown seaweed Sargassum in Traditional Chinese Medicine: A phytochemical and pharmacological review. J. Ethnopharmacol. 2012, 142, 591–619. [Google Scholar] [CrossRef]
- Kumar, M.S.; Sharma, S.A. Toxicological effects of marine seaweeds: A cautious insight for human consumption. Crit. Rev. Food Sci. Nutr. 2021, 61, 500–521. [Google Scholar] [CrossRef]
- Pal, A.; Kamthania, M.C.; Kumar, A. Bioactive Compounds and Properties of Seaweeds—A Review. OALib 2014, 1, 1–17. [Google Scholar] [CrossRef]
- Ana, P.; Nathalie, B.; Gilles, B.; Daniel, R.; Tomás, M.-S.; Yolanda, F.-P. Anti-Herpes simplex virus (HSV-1) activity and antioxidant capacity of carrageenan-rich enzymatic extracts from Solieria filiformis (Gigartinales, Rhodophyta). Int. J. Biol. Macromol. 2021, 168, 322–330. [Google Scholar] [CrossRef] [PubMed]
- Grassauer, A.; Weinmuellner, R.; Meier, C.; Pretsch, A.; Prieschl-Grassauer, E.; Unger, H. Iota-Carrageenan is a potent inhibitor of rhinovirus infection. Virol. J. 2008, 5, 107. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moreira Leite, B.S. Novas Alternativas para o Uso de Macroalgas da Costa Portuguesa em Alimentação. Master’s Thesis, University of Lisbon, Lisbon, Portugal, 2017. [Google Scholar]
- Vaskoska, R.S. Raising a Need for a Risk Assessment of Bromoform Transferred from Feed to Food. Available online: https://foodlegal.com.au/inhouse/document/2440 (accessed on 3 July 2021).
- Kinley, R.D.; Martinez-Fernandez, G.; Matthews, M.K.; de Nys, R.; Magnusson, M.; Tomkins, N.W. Mitigating the carbon footprint and improving productivity of ruminant livestock agriculture using a red seaweed. J. Clean. Prod. 2020, 259, 120836. [Google Scholar] [CrossRef]
- Vucko, M.J.; Magnusson, M.; Kinley, R.D.; Villart, C.; de Nys, R. The effects of processing on the in vitro antimethanogenic capacity and concentration of secondary metabolites of Asparagopsis taxiformis. J. Appl. Phycol. 2017, 29, 1577–1586. [Google Scholar] [CrossRef]
- Vijn, S.; Compart, D.P.; Dutta, N.; Foukis, A.; Hess, M.; Hristov, A.N.; Kalscheur, K.F.; Kebreab, E.; Nuzhdin, S.V.; Price, N.N.; et al. Key Considerations for the Use of Seaweed to Reduce Enteric Methane Emissions from Cattle. Front. Vet. Sci. 2020, 7, 597430. [Google Scholar] [CrossRef] [PubMed]
- Chan, J.C.-C.; Cheung, P.C.-K.; Ang, P.O. Comparative Studies on the Effect of Three Drying Methods on the Nutritional Composition of Seaweed Sargassum hemiphyllum (Turn.) C. Ag. J. Agric. Food Chem. 1997, 45, 3056–3059. [Google Scholar] [CrossRef]
- Nitschke, U.; Stengel, D.B. Quantification of iodine loss in edible Irish seaweeds during processing. J. Appl. Phycol. 2016, 28, 3527–3533. [Google Scholar] [CrossRef]
- Lucas, M.D.; Sequeira, E.M. Pedologia Determinação do Cu, Zn, Mn, Fe, Ca, Mg, K, e Na Totais das Plantas por Espectrofotometria de Absorção Atómica e Fotometria de Chama. Pedologia 1976, 11, 163–169. [Google Scholar]
- Ribas, M.C.; Veiga, M.E.; Curto, A.; Oliveira, E.; Barbeitos, M.M.; Ferreira, M.; Pacheco, C.; Peralta, M.F.; Duarte, M.L. Métodos de Análise de Material Vegetal e Terras; MAPA—Laboratório Químico Agrícola Rebelo da Silva, Secção de Nutrição das Culturas: Lisboa, Portugal, 1988. [Google Scholar]
- Cunniff, P. Official Methods of Analysis of AOAC International; AOAC International: Gaithersburg, MD, USA, 1997. [Google Scholar]
- Angell, A.R.; Mata, L.; de Nys, R.; Paul, N.A. The protein content of seaweeds: A universal nitrogen-to-protein conversion factor of five. J. Appl. Phycol. 2016, 28, 511–524. [Google Scholar] [CrossRef]
- Pereira, L.; van de Velde, F. Portuguese carrageenophytes: Carrageenan composition and geographic distribution of eight species (Gigartinales, Rhodophyta). Carbohydr. Polym. 2011, 84, 614–623. [Google Scholar] [CrossRef] [Green Version]



| Macro- and Micro-Elements | g 100 g−1 | 7 g of C. teedei var. lusitanicus (g) | DRI (%) |
|---|---|---|---|
| Nitrogen | 2.13 ±0.01 | NA | NA |
| Phosphorus | 0.20 ± 0.01 | 1.4 × 10−2 | 2 |
| Calcium | 0.26 ± 0.03 | 1.82 × 10−2 | 2.27 |
| Magnesium | 0.86 ± 0.02 | 6.02 × 10−2 | 16.05 |
| Potassium | 2.29 ± 0.07 | 1.60 × 10−1 | 8.02 |
| Iron | 0.02 ± 0.03 | 1.28 × 10−3 | 9.15 |
| Copper | 3.0 × 10−4 ± 3.0 × 10−5 | 2.33 × 10−5 | 2.33 |
| Zinc | 2.4 × 10−3 ± 1.0 × 10−4 | 1.68 × 10−4 | 1.68 |
| Manganese | 1.2 × 10−3 ± 1.0 × 10−5 | 8.40 × 10−5 | 4.20 |
| C. teedei var. lusitanicus (g 100 g−1) | 7 g of C. teedei var. lusitanicus (g) | DRI (%) | |||
|---|---|---|---|---|---|
| FW | DW | DW:FW | |||
| Moisture | 86.52 ± 0.18 | NA | NA | NA | NA |
| Ash | 3.96 ± 0.07 | 29.35 ± 0.13 | 1:7.4 | 2.1 | NA |
| Total lipid | 0.19 ± 0.01 | 1.42 ± 0.01 | 1:7.4 | 0.10 | 0.14 |
| Fiber | 0.24 ± 0.01 | 1.78 ± 0.09 | 1:7.4 | 0.13 | 0.49 |
| Protein | 1.54 ± 0.01 | 11.42 ± 0.01 | 1:7.4 | 0.80 | 1.59 |
| Total carbohydrate | 7.55 ± 0.12 | 56.03 ± 0.05 | 1:7.4 | 3.98 | 1.50 |
| Energy (Kcal 100 g−1) | 38 ± 0.47 | 283 ± 0.23 | 1:7.4 | 19.81 * | 0.99 |
| Chondracanthus teedei var. lusitanicus Life Cycle Phase | DW (%) | 7 g of C. teedei var. lusitanicus (g) | DRI (%) |
|---|---|---|---|
| Female gametophyte | 40.9 ± 1.5 | 2.86 | 11.45 |
| Male gametophyte | 42.1 ± 4.5 | 2.95 | 11.79 |
| Tetrasporophyte | 28.1 ± 8.1 | 1.97 | 7.87 |
| Wave Number (cm−1) | Bound | Compound | CTGF | CTGM | CTT |
|---|---|---|---|---|---|
| 805 | C–O–SO3 on C2 of 3,6-anhydrogalactose | DA2S | + | + | - |
| 825–830 | C–O–SO3 on C2 of galactose | G/D2S | - | - | + |
| 845 | D-galactose-4-sulfate | G4S | + | + | - |
| 867 | C–O–SO3 on C6 of galactose | G/D6S | + | + | - |
| 890–900 | Unsulfated b-d-galactose | G/D | + | sh | sh |
| 905 | C–O–SO3 on C2 of 3,6-anhydrogalactose | DA2S | sh | sh | sh |
| 930 | C–O of 3,6-anhydrogalactose (agar/carrageenan) | (DA) | + | + | sh |
| 970–975 | Galactose | G/D | + | + | - |
| 1012 | Sulfated esters | S=O | + | + | + |
| 1070 | C–O of 3,6-anhydrogalactose | DA | + | + | sh |
| 1100 | Sulfated esters | S=O | + | + | + |
| 1240–1260 | Sulfated esters | S=O | + | + | + |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pacheco, D.; Cotas, J.; Domingues, A.; Ressurreição, S.; Bahcevandziev, K.; Pereira, L. Chondracanthus teedei var. lusitanicus: The Nutraceutical Potential of an Unexploited Marine Resource. Mar. Drugs 2021, 19, 570. https://doi.org/10.3390/md19100570
Pacheco D, Cotas J, Domingues A, Ressurreição S, Bahcevandziev K, Pereira L. Chondracanthus teedei var. lusitanicus: The Nutraceutical Potential of an Unexploited Marine Resource. Marine Drugs. 2021; 19(10):570. https://doi.org/10.3390/md19100570
Chicago/Turabian StylePacheco, Diana, João Cotas, Andreia Domingues, Sandrine Ressurreição, Kiril Bahcevandziev, and Leonel Pereira. 2021. "Chondracanthus teedei var. lusitanicus: The Nutraceutical Potential of an Unexploited Marine Resource" Marine Drugs 19, no. 10: 570. https://doi.org/10.3390/md19100570
APA StylePacheco, D., Cotas, J., Domingues, A., Ressurreição, S., Bahcevandziev, K., & Pereira, L. (2021). Chondracanthus teedei var. lusitanicus: The Nutraceutical Potential of an Unexploited Marine Resource. Marine Drugs, 19(10), 570. https://doi.org/10.3390/md19100570









