Bioactivity of Spongian Diterpenoid Scaffolds from the Antarctic Sponge Dendrilla antarctica
Abstract
1. Introduction
2. Results and Discussion
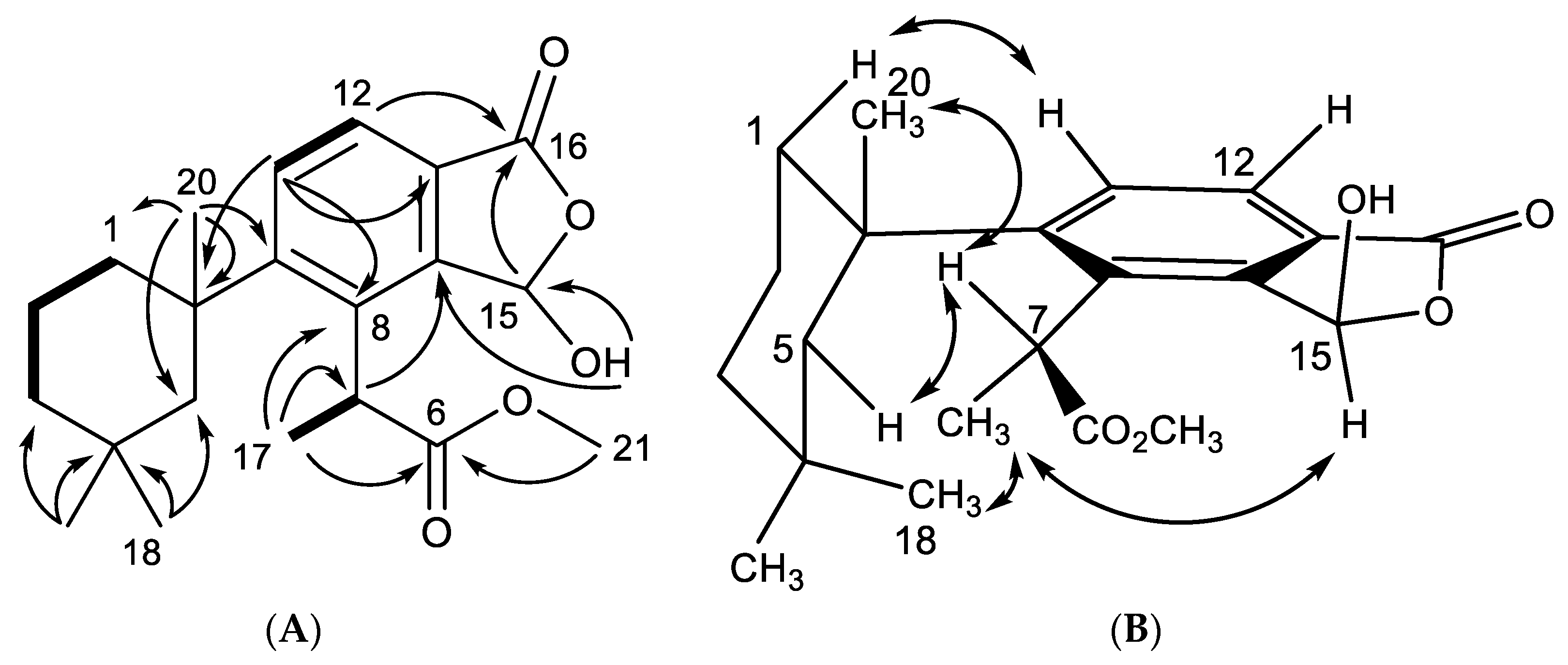
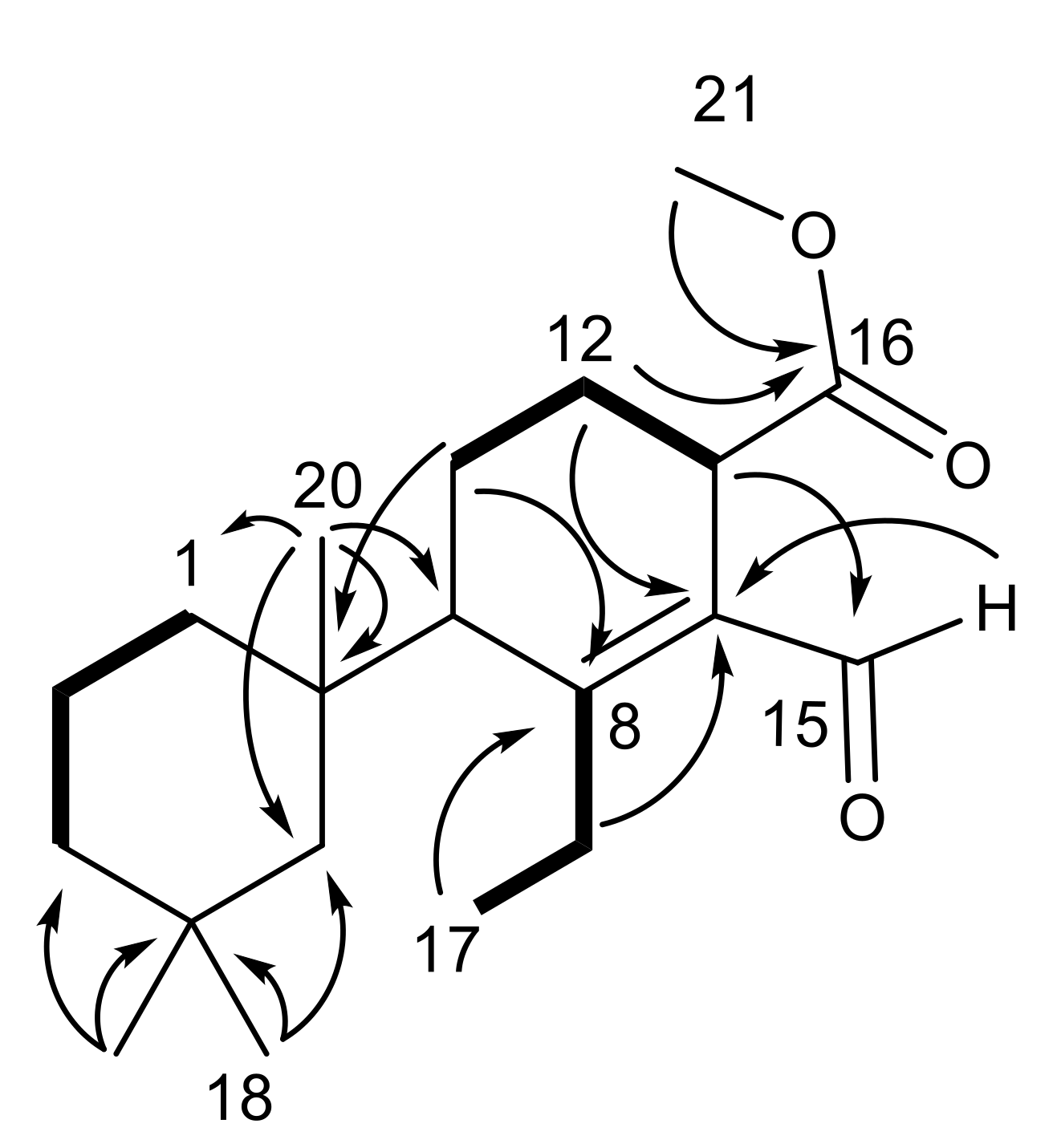
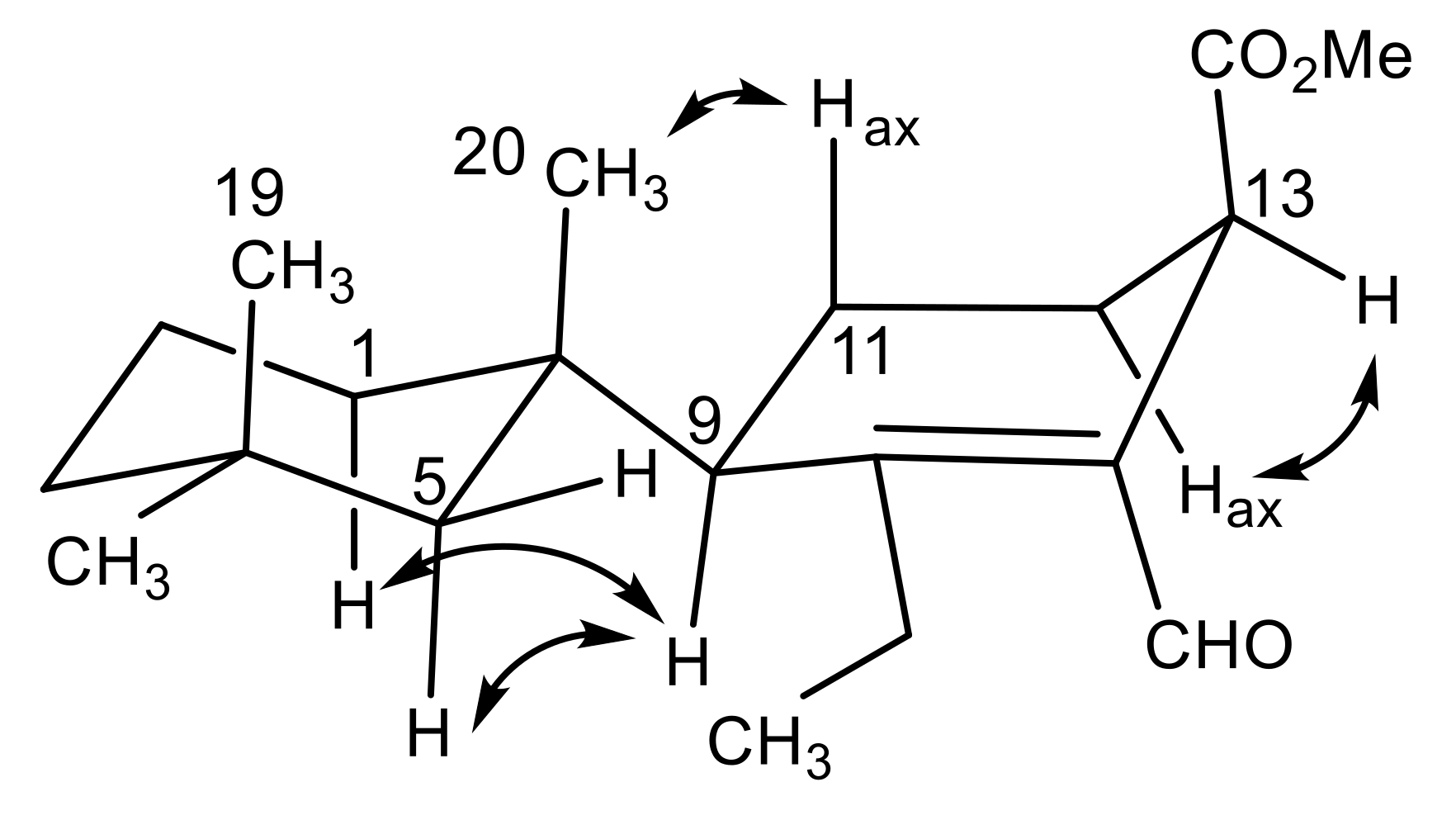
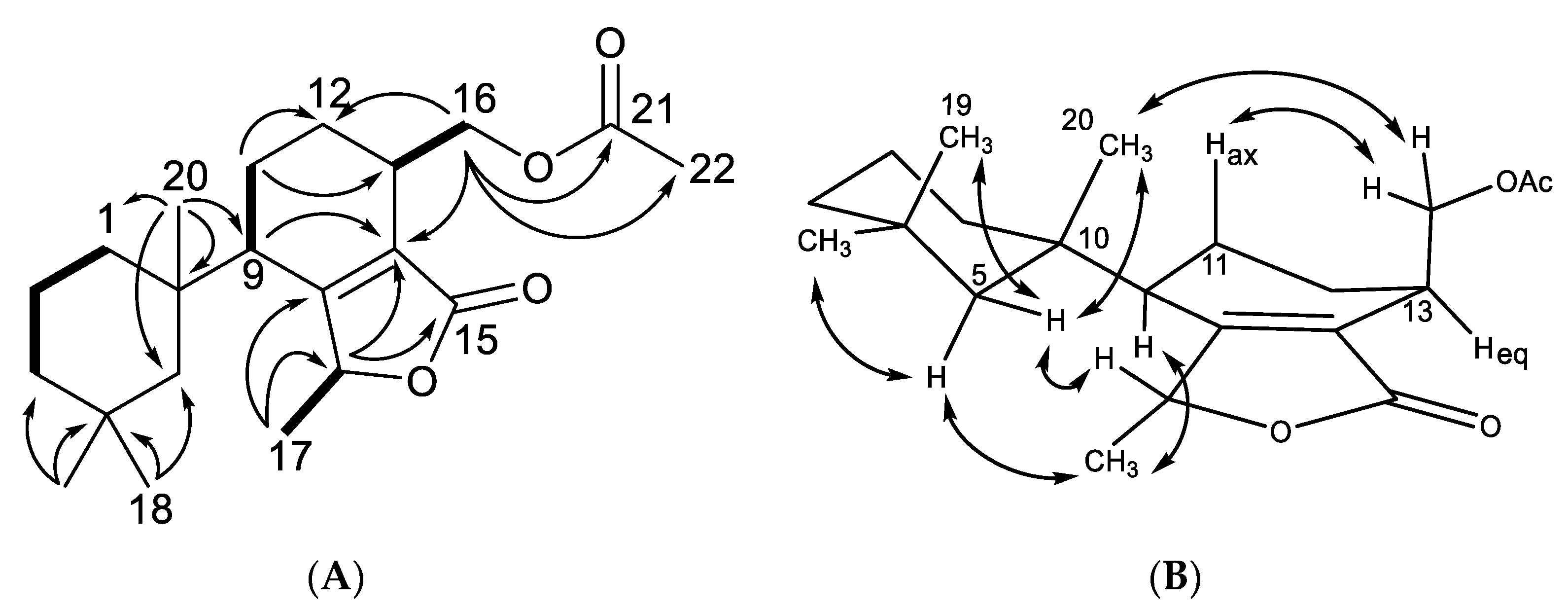
2.1. Chemical Analysis of Dendrilla antarctica
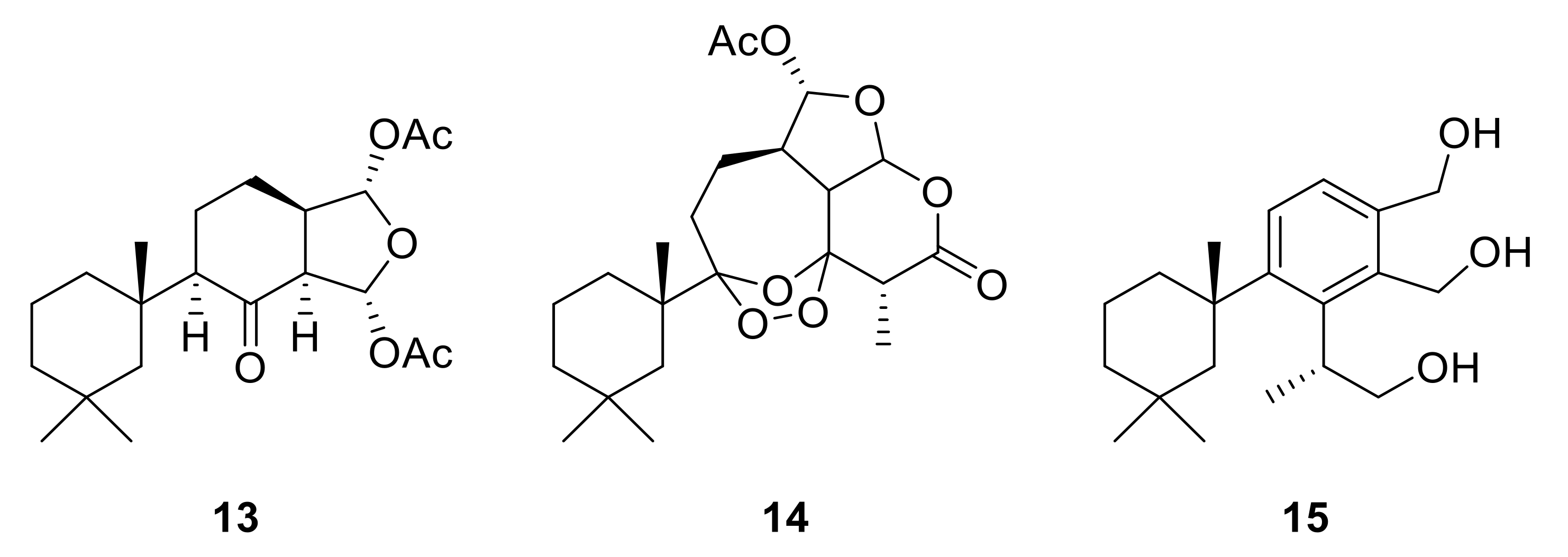
2.2. Semisynthetic Studies
2.3. Biological Profiling
3. Conclusions
4. Materials and Methods
4.1. General Procedures
4.2. Collection of Dendrilla antarctica
4.3. Extraction and Isolation of Natural Products
4.4. Leishmania Donovani and J774A.1-Cell Cytotoxicity Assay
4.5. Liver-Stage Plasmodium Falciparum Assay
4.6. Minimum MRSA Biofilm Eradication Concentration Assay
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Projan, S.J. Why is big Pharma getting out of antibacterial drug discovery? Curr. Opin. Microbiol. 2003, 6, 427–430. [Google Scholar] [CrossRef] [PubMed]
- Luepke, K.H.; Mohr, J.F., III. The antibiotic pipeline: Reviving research and development and speeding drugs to market. Expert Rev. Anti-Infect. Ther. 2017, 15, 425–433. [Google Scholar] [CrossRef] [PubMed]
- CDC. Antibiotic Resistance Threats in the United States; U.S. Department of Health and Human Services, Centers for Disease Control: Atlanta, GA, USA, 2019. [Google Scholar]
- De Kraker, M.E.A.; Stewardson, A.J.; Harbarth, S. Will 10 million people die a year due to antimicrobial resistance by 2050? PLoS Med. 2016, 13, e1002184. [Google Scholar] [CrossRef]
- Parasites. Available online: https://www.cdc.gov/parasites/index.html (accessed on 11 June 2020).
- Slivinski, N. The resilient parasite. Scientist 2019, 33, 24–31. [Google Scholar]
- Burza, S.; Croft, S.L.; Boelaert, M. Leishmaniasis. Lancet 2018, 392, 951–970. [Google Scholar] [CrossRef]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs over the nearly four decades from 01/1981 to 09/2019. J. Nat. Prod. 2020, 83, 770–803. [Google Scholar] [CrossRef]
- Carroll, A.R.; Copp, B.R.; Davis, R.A.; Keyzers, R.A.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2020, 37, 175–223. [Google Scholar] [CrossRef]
- Mehbub, M.F.; Lei, J.; Franco, C.; Zhang, W. Marine sponge derived natural products between 2001 and 2010: Trends and opportunities for discovery of bioactives. Mar. Drugs 2014, 12, 4539–4577. [Google Scholar] [CrossRef]
- Soldatou, S.; Baker, B.J. Cold-water marine natural products, 2006 to 2016. Nat. Prod. Rep. 2017, 34, 585–626. [Google Scholar] [CrossRef]
- Lebar, M.D.; Heimbegner, J.L.; Baker, B.J. Cold-water marine natural products. Nat. Prod. Rep. 2007, 24, 774–797. [Google Scholar] [CrossRef]
- Abbas, S.; Kelly, M.; Bowling, J.; Sims, J.; Waters, A.; Hamann, M. Advancement into the Arctic region for bioactive sponge secondary metabolites. Mar. Drugs 2011, 9, 2423–2437. [Google Scholar] [CrossRef]
- Von Salm, J.L.; Wilson, N.G.; Vesely, B.A.; Kyle, D.E.; Cuce, J.; Baker, B.J. Shagenes A and B, new tricyclic sesquiterpenes produced by an undescribed Antarctic octocoral. Org. Lett. 2014, 16, 2630–2633. [Google Scholar] [CrossRef] [PubMed]
- Shilling, A.J.; Witowski, C.G.; Maschek, J.A.; Azhari, A.; Vesely, B.; Kyle, D.E.; Amsler, C.D.; McClintock, J.B.; Baker, B.J. Spongian diterpenoids derived from the Antarctic sponge Dendrilla antarctica are potent inhibitors of the Leishmania parasite. J. Nat. Prod. 2020, 83, 1553–1562. [Google Scholar] [CrossRef] [PubMed]
- Knestrick, M.A.; Wilson, N.G.; Roth, A.; Adams, J.H.; Baker, B.J. Friomaramide, a highly modified linear hexapeptide from an Antarctic sponge, inhibits Plasmodium falciparum liver-stage development. J. Nat. Prod. 2019, 82, 2354–2358. [Google Scholar] [CrossRef] [PubMed]
- Von Salm, J.L.; Witowski, C.G.; Fleeman, R.M.; McClintock, J.B.; Amsler, C.D.; Shaw, L.N.; Baker, B.J. Darwinolide, a new diterpene scaffold thati inhibits methicillin-resistant Staphylococcus aureus biofilm from the Antarctic sponge Dendrilla membranosa. Org. Lett. 2016, 18, 2596–2599. [Google Scholar] [CrossRef]
- Ankisetty, S.; Amsler, C.D.; McClintock, J.B.; Baker, B.J. Further membranolide diterpenes from the antarctic sponge Dendrilla membranosa. J. Nat. Prod. 2004, 67, 1172–1174. [Google Scholar] [CrossRef]
- Molinski, T.F.; Faulkner, D.J. Metabolites of the antarctic sponge Dendrilla membranosa. J. Org. Chem. 1987, 52, 296–298. [Google Scholar] [CrossRef]
- Baker, B.J.; Kopitzke, R.W.; Yoshida, W.Y.; McClintock, J.B. Chemical and ecological studies of the Antarctic sponge Dendrilla membranosa. J. Nat. Prod. 1995, 58, 1459–1462. [Google Scholar] [CrossRef]
- Karuso, P.; Skelton, B.W.; Taylor, W.C.; White, A.H. The constituents of marine sponges. I. The isolation from Aplysilla sulphurea (Dendroceratida) of (lR*,l′S*,l″R*,3R*)-1-acetoxy-4-ethyl-5-(1,3,3-trimethylcyclohexy1)-1,3-dihydroisobenzofuran-1′(4),3-carbolactone and the determination of its crystal structure. Aust. J. Chem. 1984, 37, 1081–1093. [Google Scholar]
- Buckleton, J.S.; Bergquist, P.R.; Cambie, R.C.; Clark, G.R.; Karuso, P.; Rickard, C.E.F. Structure of tetrahydroaplysulphurin-1. Acta Crystallogr. Sec. C 1987, 43, 2430–2432. [Google Scholar] [CrossRef]
- Tischler, M.; Andersen, R.J. Glaciolide, a degraded diterpenoid with a new carbon skeleton from the nudibranch Cadlina luteomarginata and the sponge Aplysilla glacialis. Tetrahedron Lett. 1989, 30, 5717–5720. [Google Scholar] [CrossRef]
- Mayol, L.; Piccialli, V.; Sica, D. New degraded diterpenes from the sponge Spongionella gracilis. Gazzetta Chim. Ital. 1988, 118, 559–563. [Google Scholar]
- Keyzers, R.A.; Northcote, P.T.; Zubkov, O.A. Novel anti-inflammatory spongian diterpenes from the New Zealand marine sponge Chelonaplysilla violacea. Eur. J. Org. Chem. 2004, 2004, 419–425. [Google Scholar] [CrossRef]
- Graham, S.K.; Garson, M.J.; Bernhardt, P.V. The absolute structure of (+)-aplysulfurin. J. Chem. Crystallogr. 2010, 40, 468–471. [Google Scholar] [CrossRef]
- Diaz-Marrero, A.R.; Dorta, E.; Cueto, M.; San-Martin, A.; Darias, J. Conformational analysis and absolute stereochemistry of ′spongian′-related metabolites. Tetrahedron 2004, 60, 1073–1078. [Google Scholar] [CrossRef]
- Puliti, R.; Fontana, A.; Cimino, G.; Mattia, C.A.; Mazzarella, L. Structure of a keto derivative of 9,11-dihydrogracilin A. Acta Chem. Scan. C. Cryst. Struct. Comm. 1993, C49, 1373–1376. [Google Scholar] [CrossRef]
- Phyo, A.P.; Jittamala, P.; Nosten, F.H.; Pukrittayakamee, S.; Imwong, M.; White, N.J.; Duparc, S.; Macintyre, F.; Baker, M.; Möhrle, J.J. Antimalarial activity of artefenomel (OZ439), a novel synthetic antimalarial endoperoxide, in patients with Plasmodium falciparum and Plasmodium vivax malaria: An open-label phase 2 trial. Lancet Infect. Dis. 2016, 16, 61–69. [Google Scholar] [CrossRef]
- Tang, Y.; Dong, Y.; Vennerstrom, J.L. Synthetic peroxides as antimalarials. Med. Res. Rev. 2004, 24, 425–448. [Google Scholar] [CrossRef]
- Roth, A.; Maher, S.P.; Conway, A.J.; Ubalee, R.; Chaumeau, V.; Andolina, C.; Kaba, S.A.; Vantaux, A.; Bakowski, M.A.; Luque, R.T. A comprehensive model for assessment of liver stage therapies targeting Plasmodium vivax and Plasmodium falciparum. Nat. Commun. 2018, 9, 1837. [Google Scholar] [CrossRef]

| Dendrillin B (10) | Dendrillin C (11) | Dendrillin D (12) | ||||
|---|---|---|---|---|---|---|
| Pos. | δC b | δH c | δC d | δH e | δC b | δH c |
| 1α | 41.1 | 2.35 (1H, d, 13.6) | 37.7 | 1.35 (1H, ov, m) | 36.7 | 1.15 (1H, td, 12.8, 3.7) |
| β | 1.50 (1H, m) | 1.42 (1H, ov, m) | 1.53 (1H, m) | |||
| 2α | 19.9 | 1.70 (1H, m) | 19.6 | 1.54 (2H, m) | 19.1 | 1.54 (1H, m) |
| β | 1.79 (1H, m) | 1.60 (1H, m) | ||||
| 3α | 39.3 | 1.32 (2H, m) | 39.1 | 1.11 (1H, m) | 39.0 | 1.05 (1H, td, 12.6, 3.4) |
| β | 1.35 (1H, ov, m) | 1.44 (1H, m) | ||||
| 4 | 31.8 | 31.3 | 31.2 | |||
| 5α | 51.1 | 1.53 (1H, d, 14.3) | 52.6 | 1.35 (1H, ov d, 13.5) | 51.9 | 1.11 (1H, d, 13.2) |
| β | 2.03 (1H, d, 14.3) | 1.46 (1H, ov d, 13.5) | 1.41 (1H, d, 12.6) | |||
| 6 | 176.0 | |||||
| 7a | 40.6 | 4.78 (1H, q, 7.3) | 26.7 | 2.43 (1H, dq, 13.8, 7.3) | 80.5 | 5.21 (1H, q, 6.5) |
| b | 3.20 (1H, dq, 13.8, 7.3) | |||||
| 8 | 136.6 | 167.9 | 169.3 | |||
| 9 | 154.4 | 50.1 | 2.29 (1H, br t, 4.3) | 47.9 | 2.18 (1H, t, 7.0) | |
| 10 | 40.1 | 39.5 | 37.7 | |||
| 11α | 124.0 | 7.78 (1H, d, 8.3) | 24.5 | 1.40 (1H, ov m) | 20.9 | 1.74 (2H, m) |
| β | 2.01 (1H, dm, 14.0) | |||||
| 12α | 130.4 | 7.76 (1H, d, 8.3) | 23.5 | 1.82 (1H, m) | 23.7 | 1.50 (1H, m) |
| β | 1.91 (1H, m) | 1.85 (1H, m) | ||||
| 13 | 125.9 | 39.4 | 3.60 (1H, t, 7.9) | 30.7 | 2.83 (1H, m) | |
| 14 | 145.5 | 134.2 | 128.0 | |||
| 15 | 97.3 | 6.70 (1H, d, 9.2) | 190.5 | 10.18 (1H, s) | 172.5 | |
| 16 | 168.1 | 174.8 | 64.1 | 4.21 (2H, m) | ||
| 17 | 14.8 | 1.75 (3H, d, 7.5) | 16.6 | 1.13 (3H, br t, 7.5) | 19.7 | 1.45 (3H, d, 6.5) |
| 18 | 27.6 | 0.36 (3H, s) | 34.6 | 0.98 (3H, s) | 35.5 | 0.99 (3H, s) |
| 19 | 32.8 | 0.93 (3H, s) | 28.9 | 0.92 (3H, s) | 27.4 | 0.92 (3H, s) |
| 20 | 32.2 | 1.40 (3H, s) | 24.6 | 1.03 (3H, s) | 21.2 | 0.94 (3H, s) |
| 21 | 52.7 | 3.67 (3H, s) | 51.8 | 3.65 (3H, s) | 171.0 | |
| 22 | 21.0 | 2.06 (3H, s) | ||||
| OH | 4.28 (1H, d, 9.2) | |||||
| Compound | Leishmania donovani IC50 (μM) a | Plasmodium falciparum Inhibition d at 5 μg/mL | MRSA Biofilm Inhibition f | ||||
|---|---|---|---|---|---|---|---|
| Infected Macrophage b | Uninfected Macrophage | Infected PHHs e | Uninfected PHHs | 100 μg/mL | 50 μg/mL | 25 μg/mL | |
| Positive control | Miltefosine | Primaquine | None | ||||
| 2.9 | >120 | 97.8 (5.3) | 0 (2.2) | ND | ND | ND | |
| Aplysulphurin (2) | 3.1 c | 12 c | 100 (8.4) | 7.6 (4.8) | 21.1 (15.0) | 29.4 (14.9) | 6.5 (5.7) |
| 9,11-Dihydrogracilin A (3) | 9.1 c | 23 c | 0 | 1.1 (1.0) | 12.7 (14.3) | 29.1 (9.7) | 0 |
| Tetrahydroaplysulphurin-1 (4) | 3.5 c | >130 c | 66.7 (10.4) | 3.0 (2.4) | 20.8 (15.3) | 31.1 (11.6) | 35.0 (10.2) |
| Membranolide (5) | 9.7 c | 77 c | 59.7 (19.1) | 0 | 100.0 (0.0) | 99.9 (0.1) | 99.7 (0.1) |
| Glaciolide (6) | 8.8 | >170 | 0 | 4.5 (3.9) | 32.2 (10.5) | 20.8 (8.2) | 20.0 (20.7) |
| Compound 7 | 14 | 18 | 39.3 (32.7) | 0.7 (1.0) | 2.9 (5.2) | 28.7 (15.0) | 3.9 (6.1) |
| Cadlinolide C (8) | 16 | >160 | 0 | 0 | 85.4 (5.6) | 35.0 (20.5) | 2.6 (3.9) |
| Dendrillin A (9) | 6 | 9 | 4.3 (3.8) | 2.9 (0.7) | 0 | 0 | 0 |
| Dendrillin B (10) | 3.5 | >140 | 0 | 0 | 90.7 (1.2) | 19.4 (8.4) | 37.0 (12.5) |
| Dendrillin C (11) | >31 | >160 | 1.3 (3.2) | 3.4 (4.8) | 99.2 (0.1) | 97.8 (0.4) | 84.0 (1.0) |
| 8-Ketodihydrogracilin (13) | 4.5 | 22 | 0 | 2.3 (1.9) | 99.7 (0.2) | 97.7 (1.1) | 5.7 (9.0) |
| Ozonide (14) | 9.9 | 17 | 61.7 (24.7) | 18.6 (3.4) | 99.9 (0.0) | 40.6 (5.3) | 17.7 (12.7) |
| Triol (15) | 4.2 | >160 | 81.0 (23.1) | 2.2 (1.4) | 9.3 (7.8) | 12.3 (9.0) | 2.6 (3.6) |
 |  |  |  |  | ||
| Tetrahydro- aplysulphurum-1 (4) | Dendrillin B (10) | 8-Keto- Dihydrogracilin (13) | Membranolide (5) | Dencrillin C (11) | Triol (15) | |
| Leishmania donovani | Leishmania donovani | Leishmania donovani | MRSA Biofilm | MRSA Biofilm | Plasmodium falciparum | |
| Pathogen Activity | 3.5 a | 3.5 | 4.5 | 58 | 78 | ≤16 |
| J774 Cytotoxiciy | >130 | >140 | 22 | 77 | >160 | >160 |
| Selectivity Index (SI) | 37 | 40 | 5 | 1 | 2.0 | >10 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bory, A.; Shilling, A.J.; Allen, J.; Azhari, A.; Roth, A.; Shaw, L.N.; Kyle, D.E.; Adams, J.H.; Amsler, C.D.; McClintock, J.B.; et al. Bioactivity of Spongian Diterpenoid Scaffolds from the Antarctic Sponge Dendrilla antarctica. Mar. Drugs 2020, 18, 327. https://doi.org/10.3390/md18060327
Bory A, Shilling AJ, Allen J, Azhari A, Roth A, Shaw LN, Kyle DE, Adams JH, Amsler CD, McClintock JB, et al. Bioactivity of Spongian Diterpenoid Scaffolds from the Antarctic Sponge Dendrilla antarctica. Marine Drugs. 2020; 18(6):327. https://doi.org/10.3390/md18060327
Chicago/Turabian StyleBory, Alexandre, Andrew J. Shilling, Jessie Allen, Ala Azhari, Alison Roth, Lindsey N. Shaw, Dennis E. Kyle, John H. Adams, Charles D. Amsler, James B. McClintock, and et al. 2020. "Bioactivity of Spongian Diterpenoid Scaffolds from the Antarctic Sponge Dendrilla antarctica" Marine Drugs 18, no. 6: 327. https://doi.org/10.3390/md18060327
APA StyleBory, A., Shilling, A. J., Allen, J., Azhari, A., Roth, A., Shaw, L. N., Kyle, D. E., Adams, J. H., Amsler, C. D., McClintock, J. B., & Baker, B. J. (2020). Bioactivity of Spongian Diterpenoid Scaffolds from the Antarctic Sponge Dendrilla antarctica. Marine Drugs, 18(6), 327. https://doi.org/10.3390/md18060327









