Partial Characterization of Protease Inhibitors of Ulva ohnoi and Their Effect on Digestive Proteases of Marine Fish
Abstract
1. Introduction
2. Results
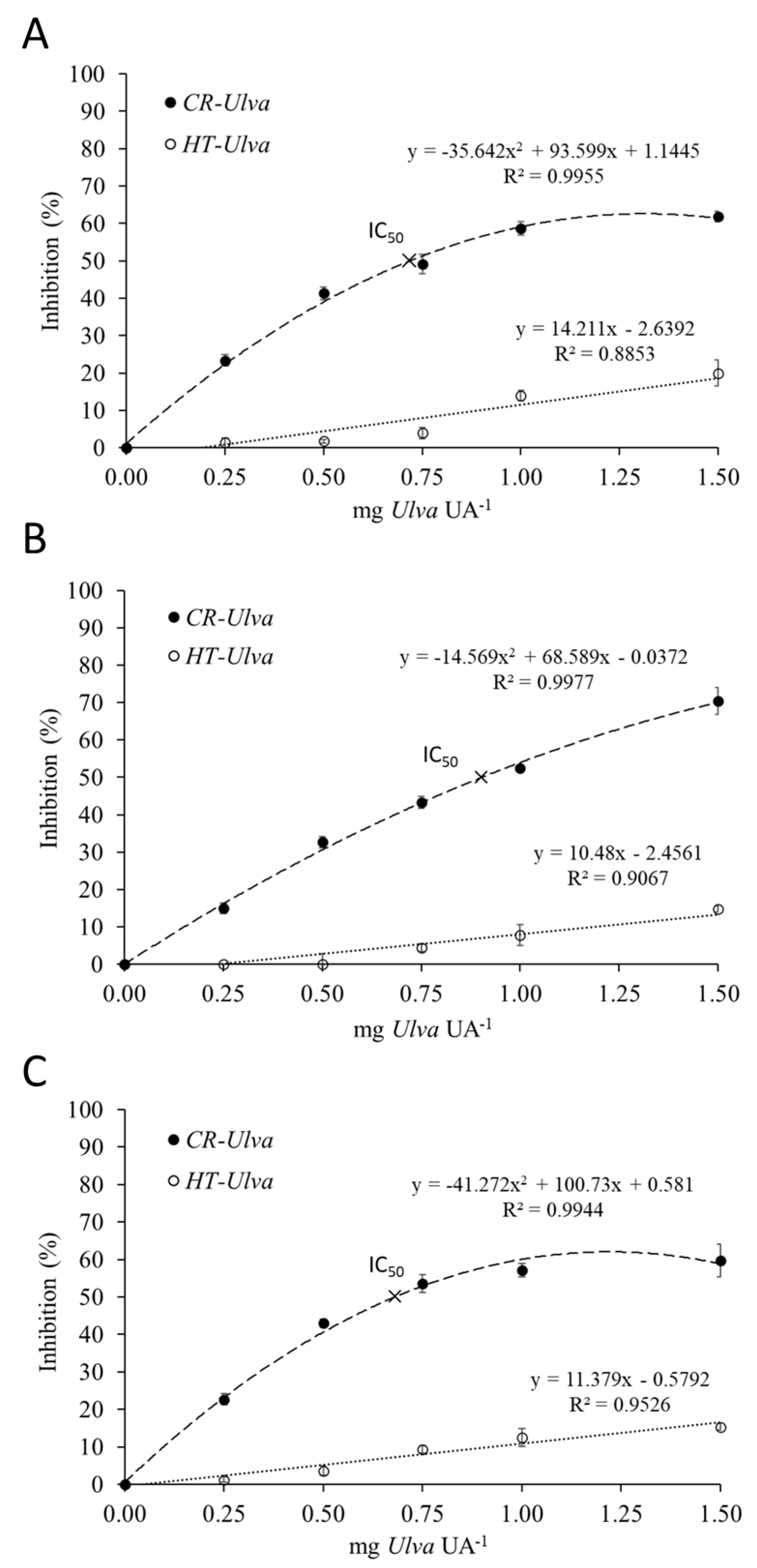
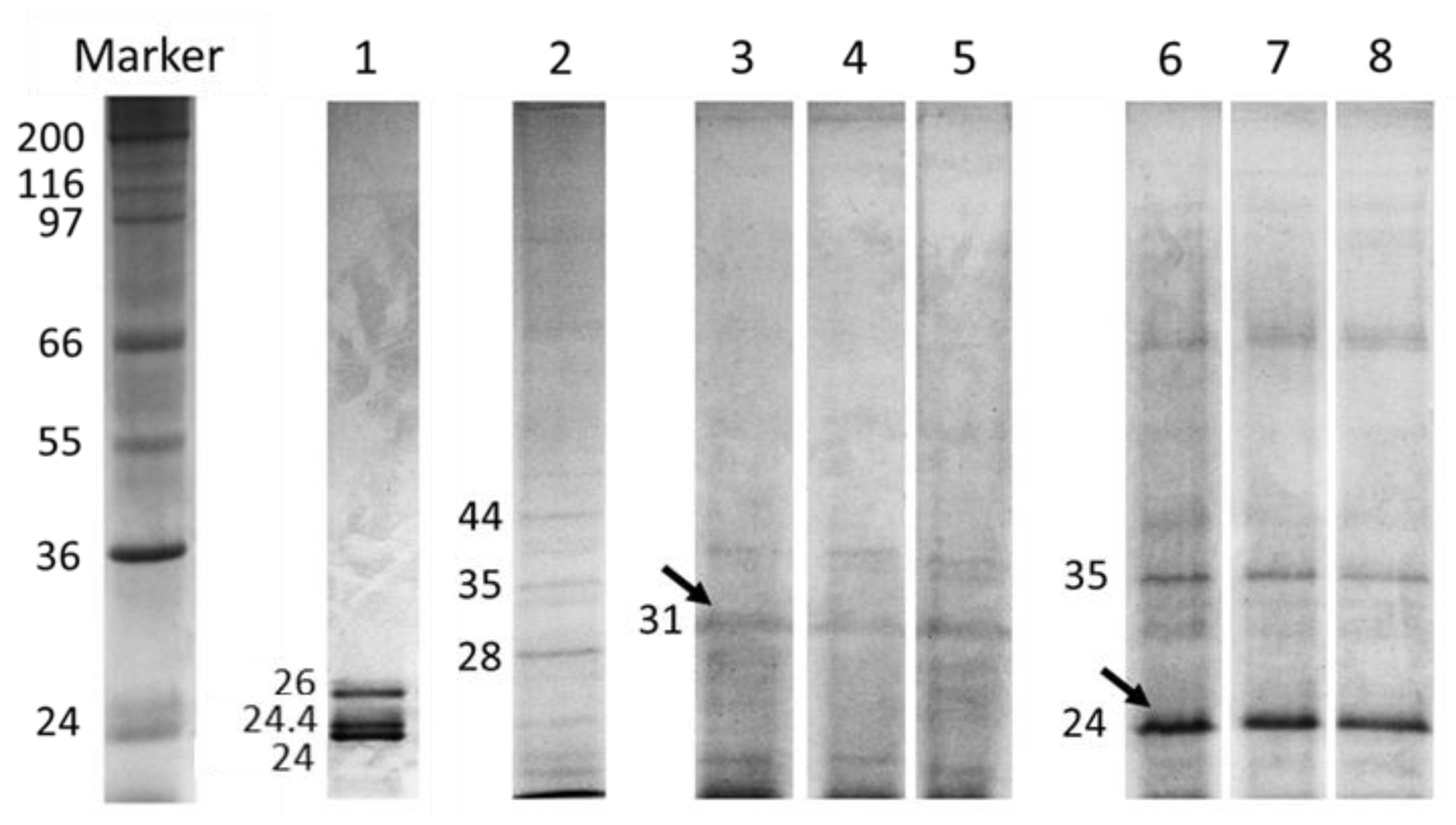
2.1. Inhibition Assay
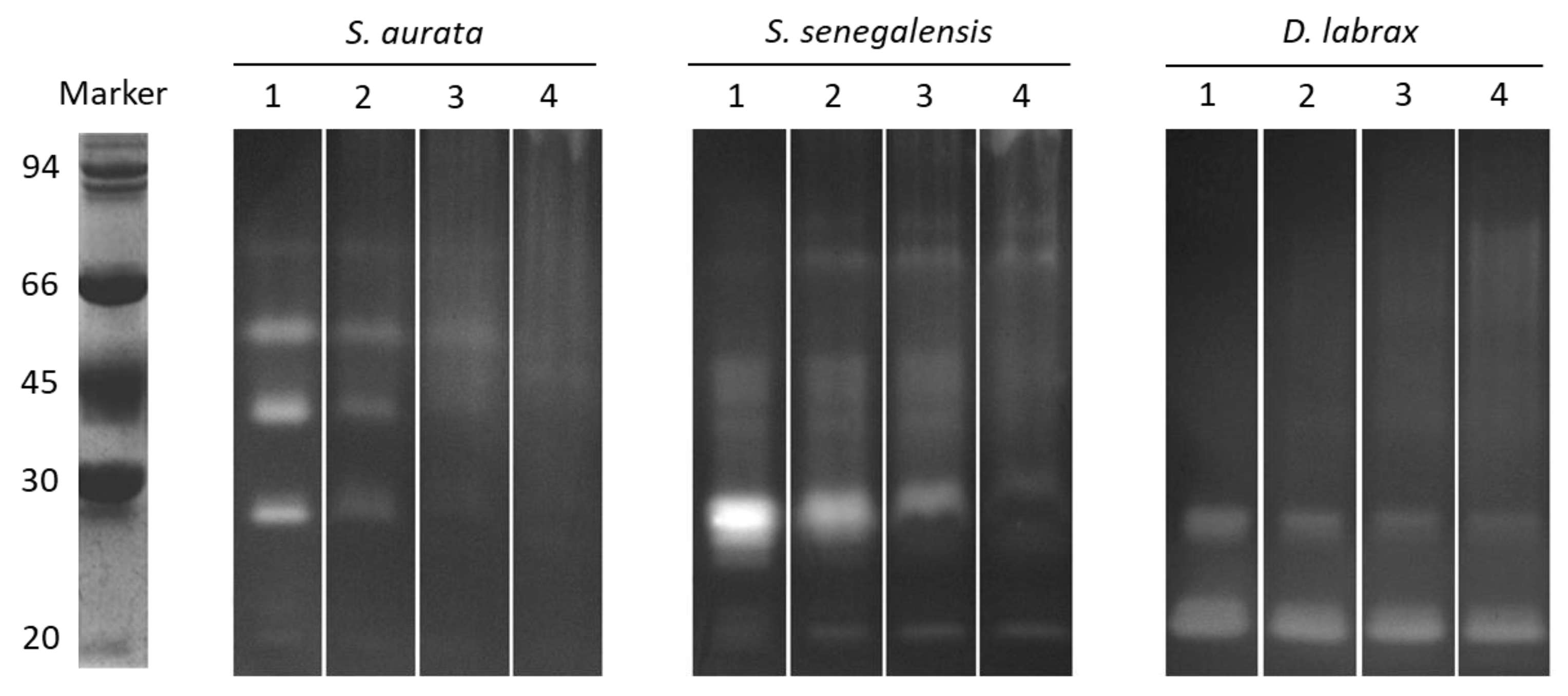
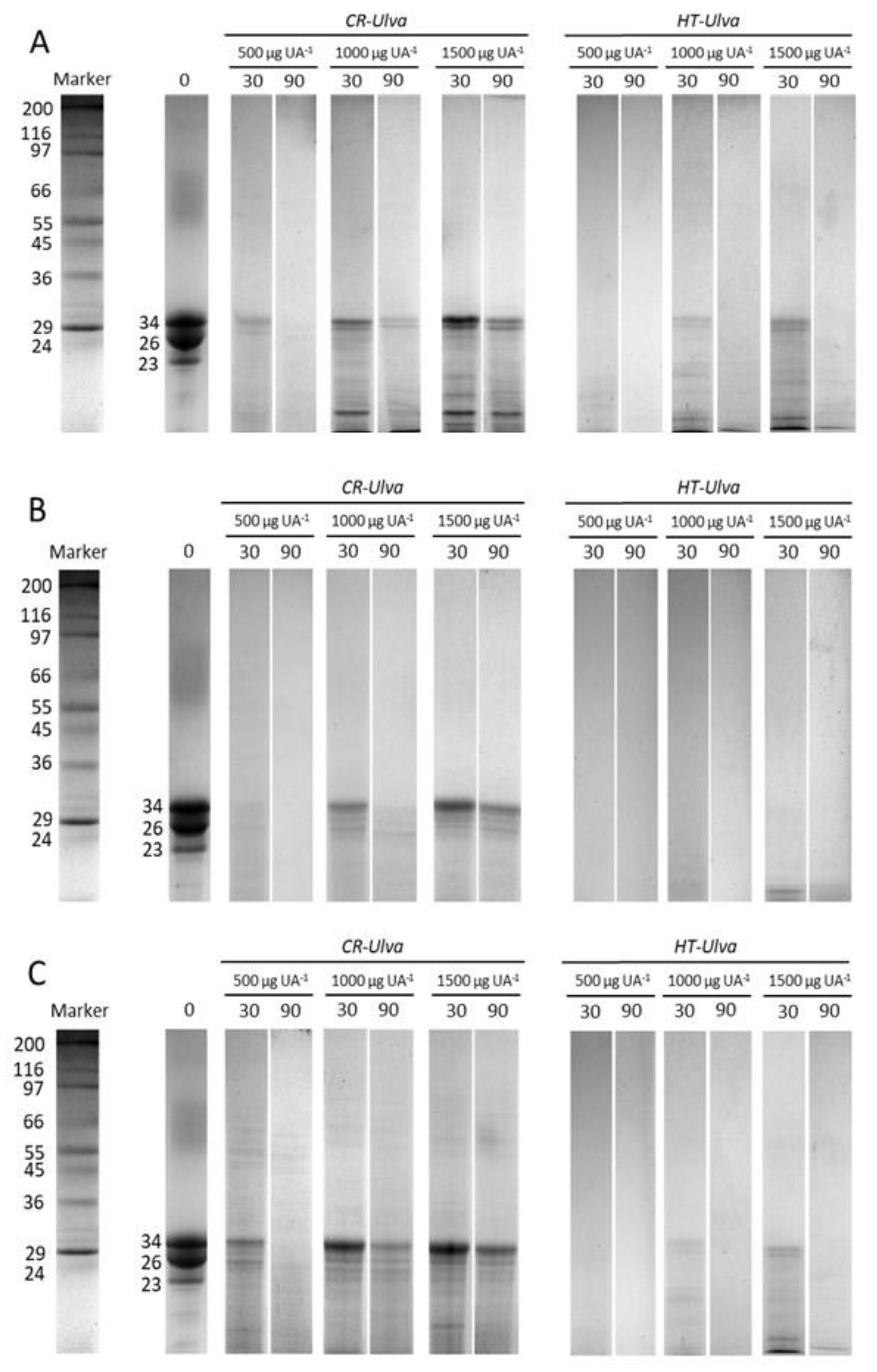
2.2. In Vitro Casein Hydrolysis
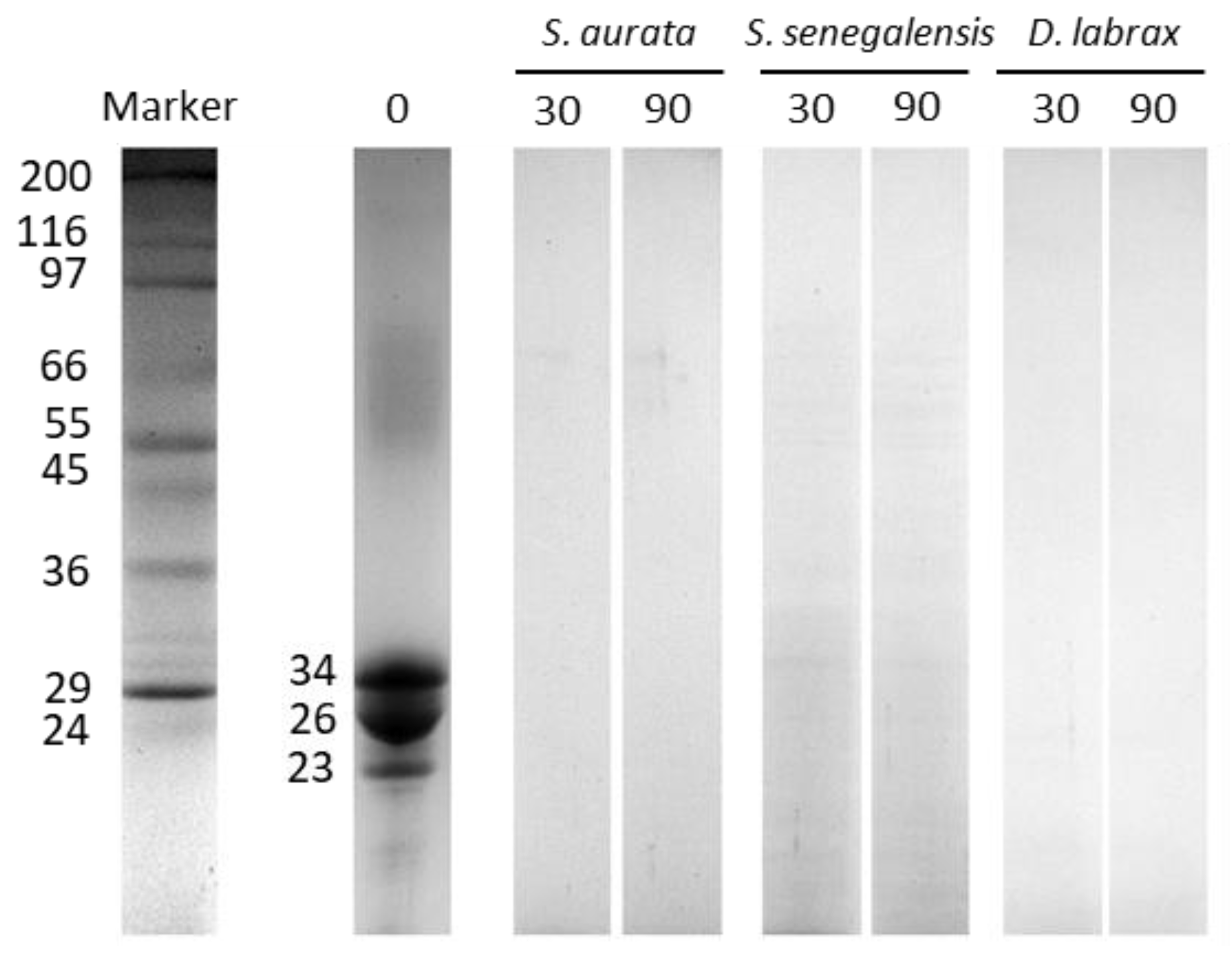
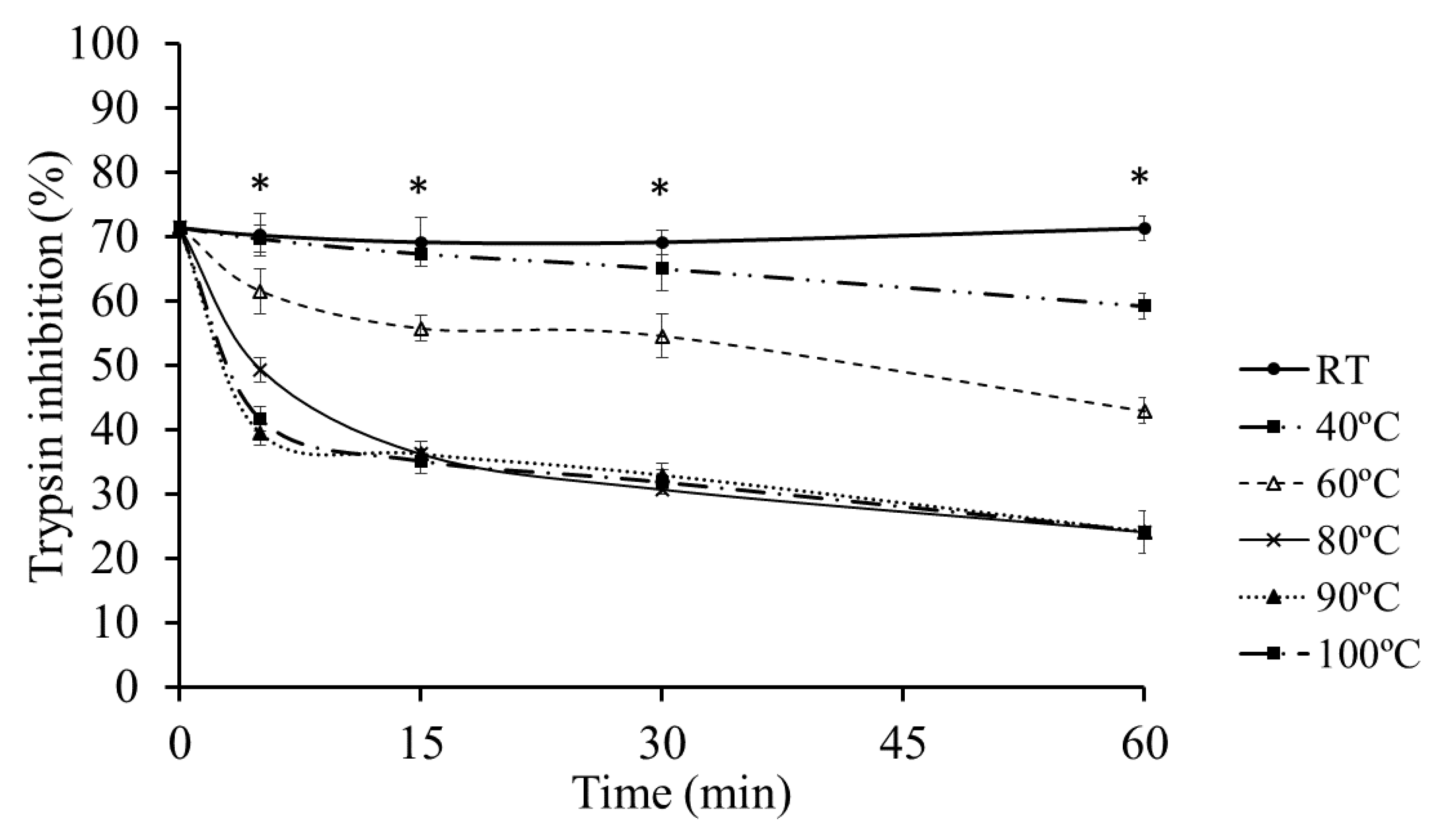
2.3. Thermal Stability of Protease Inhibitors
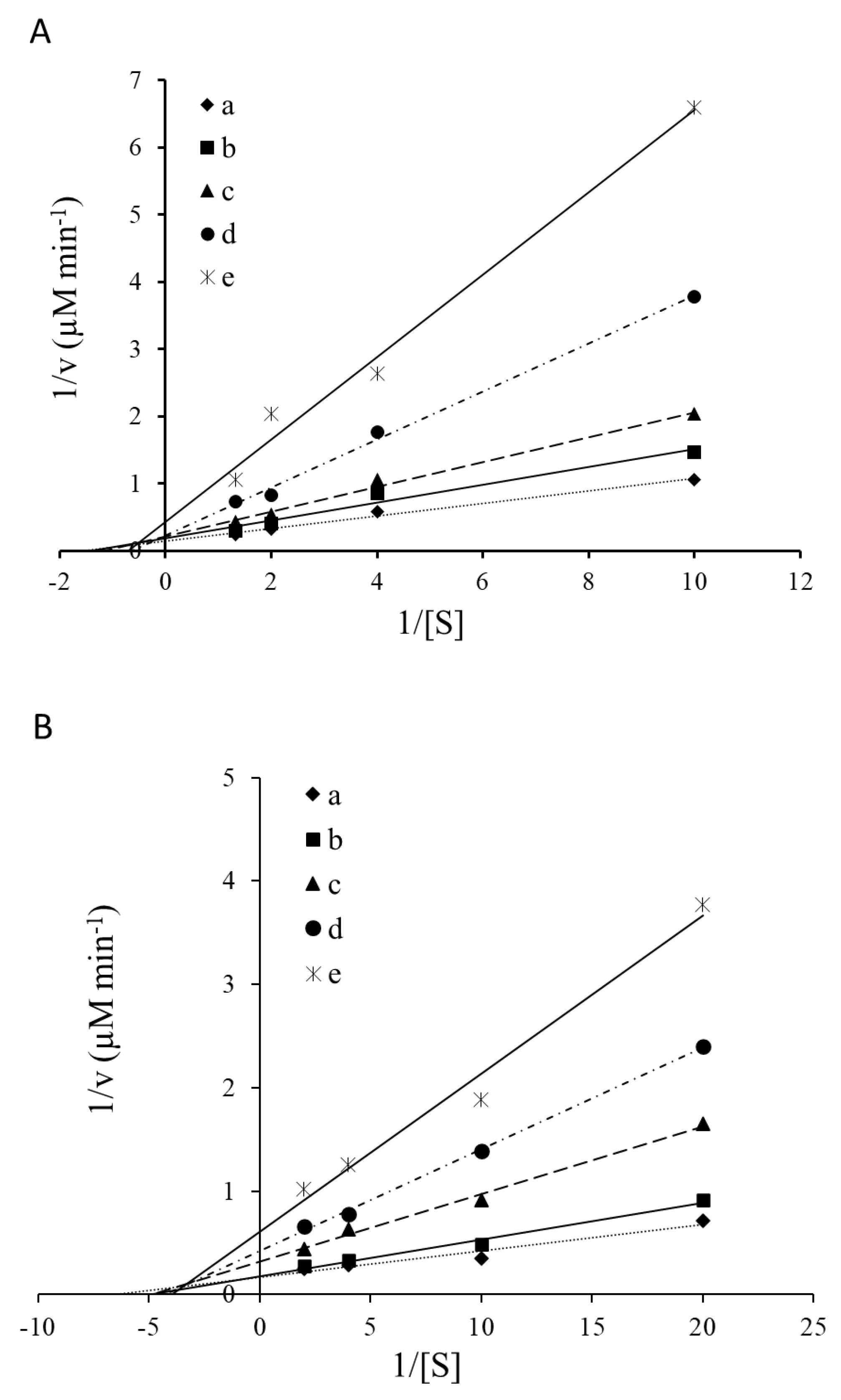
2.4. Kinetic Parameter’s
3. Discussion
4. Materials and Methods
4.1. Ulva Biomass
4.2. Fish Enzyme Extracts
4.3. Testing the Presence of Protease Inhibitors in Ulva
4.4. Effect of Ulva on Fish Digestive Proteases
4.5. Partial Characterization of Ulva Protease Inhibitors
4.5.1. Effect of Temperature on Protease Inhibitors
4.5.2. Trypsin and Chymotrypsin Inhibition Kinetics
4.6. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Prabu, E.; Rajagopalsamy, C.B.T.; Ahilan, B.; Santhakumar, R.; Jeevagan, I.J.M.A.; Renuhadevi, M. An overview of anti-nutritional factors in fish feed ingredients and their effects in fish. J. Aquac. Trop. 2017, 32, 149. [Google Scholar]
- Thakur, A.; Sharma, V.; Thakur, A. An overview of anti-nutritional factors in food. Int. J. Chem. Stud. 2019, 7, 2472–2479. [Google Scholar]
- Glencross, B.D.; Baily, J.; Berntssen, M.H.; Hardy, R.; MacKenzie, S.; Tocher, D.R. Risk assessment of the use of alternative animal and plant raw material resources in aquaculture feeds. Rev. Aquac. 2019, 1–56. [Google Scholar] [CrossRef]
- Chakraborty, P.; Mallik, A.; Sarang, N.; Lingam, S.S. A review on alternative plant protein sources available for future sustainable aqua feed production. Int. J. Chem. Stud. 2019, 7, 1399–1404. [Google Scholar]
- Hajra, A.; Mazumder, A.; Verma, A.; Ganguly, D.P.; Mohanty, B.P.; Sharma, A.P. Antinutritional factors in plant origin fish feed ingredients: The problems and probable remedies. Adv. Fish Res. 2013, 5, 193–202. [Google Scholar]
- Mæhre, H.K. Seaweed Proteins—How to Get to Them? Effects of Processing on Nutritional Value, Bioaccessibility and Extractability. Ph.D. Thesis, The Arctic University of Norway, Tromsø, Norway, 2015. [Google Scholar]
- Diken, G.; Demir, O.; Naz, M. The inhibitory effects of different diets on the protease activities of Argyrosomus regius (Pisces, Scianidae) larvae as a potential candidate species. J. Appl. Anim. Res. 2016, 46, 1–6. [Google Scholar] [CrossRef]
- Vizcaíno, A.J.; Fumanal, M.; Sáez, M.I.; Martínez, T.F.; Moriñigo, M.A.; Fernández-Díaz, C.; Anguís, V.; Balebona, M.C.; Alarcón, F.J. Evaluation of Ulva ohnoi as functional dietary ingredient in juvenile Senegalese sole (Solea senegalensis): Effects on the structure and functionality of the intestinal mucosa. Algal Res. 2019, 42, 101608. [Google Scholar] [CrossRef]
- Guerreiro, I.; Magalhães, R.; Coutinho, F.; Couto, A.; Sousa, S.; Delerue-Matos, C.; Domingues, V.F.; Oliva-Teles, A.; Peres, H. Evaluation of the seaweeds Chondrus crispus and Ulva lactuca as functional ingredients in gilthead seabream (Sparus aurata). J. Appl. Phycol. 2019, 31, 2115–2124. [Google Scholar] [CrossRef]
- Pereira, V.; Marques, A.; Gaivão, I.; Rego, A.; Abreu, H.; Pereira, R.; Santos, M.A.; Guilherme, S.; Pacheco, M. Marine macroalgae as a dietary source of genoprotection in gilthead seabream (Sparus aurata) against endogenous and exogenous challenges. Comp. Biochem. Phys. C 2019, 219, 12–24. [Google Scholar] [CrossRef]
- Tapia-Paniagua, S.T.; Fumanal, M.; Anguis, V.; Fernandez-Diaz, C.; Alarcón, F.J.; Moriñigo, M.A.; Balebona, M.C. Modulation of intestinal microbiota in Solea senegalensis fed low dietary level of Ulva ohnoi. Front. Microbiol. 2019, 10, 171. [Google Scholar] [CrossRef]
- Peixoto, M.J.; Magnoni, L.; Gonçalves, J.F.; Twijnstra, R.H.; Kijjoa, A.; Pereira, R.; Palstra, A.P.; Ozório, R.O. Effects of dietary supplementation of Gracilaria sp. extracts on fillet quality, oxidative stress, and immune responses in European seabass (Dicentrarchus labrax). J. Appl. Phycol. 2019, 31, 761–770. [Google Scholar] [CrossRef]
- Silva, D.M.; Valente, L.M.P.; Sousa-Pinto, I.; Pereira, R.; Pires, M.A.; Seixas, F.; Rema, P. Evaluation of IMTA-produced seaweeds (Gracilaria, Porphyra, and Ulva) as dietary ingredients in Nile tilapia, Oreochromis niloticus L., juveniles. Effects on growth performance and gut histology. J. Appl. Phycol. 2015, 27, 1671–1680. [Google Scholar] [CrossRef]
- Oliveira, M.N.; Ponte-Freitas, A.L.; Urano-Carvalho, A.F.; Taveres-Sampaio, T.M.; Farias, D.F.; Alves-Teixera, D.I.; Gouveia, S.T.; Gomes-Pereira, J.; Castro-Catanho de Sena, M.M. Nutritive and non-nutritive attributes of washed-up seaweeds from the coast of Ceará. Food Chem. 2009, 11, 254–259. [Google Scholar] [CrossRef]
- Francis, G.; Makkar, H.P.; Becker, K. Antinutritional factors present in plant-derived alternate fish feed ingredients and their effects in fish. Aquaculture 2001, 199, 197–227. [Google Scholar] [CrossRef]
- Le Bourvellec, C.; Renard, C.M. Interactions between polyphenols and macromolecules: Quantification methods and mechanisms. Crit. Rev. Food Sci. Nutr. 2012, 52, 213–248. [Google Scholar] [CrossRef] [PubMed]
- Sáez, M.I.; Martinez, T.F.; Alarcón, F.J. Effect of the dietary of seaweeds on intestinal proteolytic activity of juvenile sea bream Sparus Aurata. Int. Aquafeed 2013, 16, 38–40. [Google Scholar]
- Deivasigamani, B.; Subamanian, V. Applications of immunostimulants in aquaculture: A review. Int. J. Curr. Microbiol. App. Sci. 2016, 5, 447–453. [Google Scholar] [CrossRef][Green Version]
- Moutinho, S.; Linares, F.; Rodriguez, J.L.; Sousa, V.; Valente, L.M.P. Inclusion of 100% seaweed meal in diets for juvenile and on-growing life stages of Senegaleses sole (Solea senegalensis). J. Appl. Phycol. 2018, 30, 3589–3601. [Google Scholar] [CrossRef]
- Haard, N.F.; Dimes, L.E.; Arndt, R.E.; Dong, F.M. Estimation of protein digestibility. IV. Digestive proteinases from the pyloric caeca of coho salmo (Oncorhynchus kisutch) fed diets containing soybean meal. Comp. Biochem. Physiol. 1996, 115, 533–540. [Google Scholar] [CrossRef]
- Krogdahl, Å.; Penn, M.; Thorsen, J.; Refstie, S.; Bakke, A.M. Important antinutrients in plant feedstuffs for aquaculture: An update on recent findings regarding responses in salmonids. Aquac. Res. 2010, 41, 333–344. [Google Scholar] [CrossRef]
- Magnoni, L.J.; Martos-Sitcha, J.A.; Queiroz, A.; Calduch-Giner, J.A.; Gonçalves, J.F.M.; Rocha, C.M.R.; Abreu, H.T.; Schrama, J.W.; Ozorio, R.O.A.; Pérez-Sánchez, J. Dietary supplementation of heat-treated Gracilaria and Ulva seaweeds enhanced acute hypoxia tolerance in gilthead sea bream (Sparus aurata). Biol. Open 2017, 6, 897–908. [Google Scholar] [CrossRef] [PubMed]
- Bandara, T. Alternative feed ingredients in aquaculture: Opportunities and challenges. J. Entomol. Zool. Stud. 2018, 6, 3087–3094. [Google Scholar]
- Vizcaíno, A.J.; Sáez, M.I.; Martínez, T.F.; Acién, F.G.; Alarcón, F.J. Differential hydrolysis of proteins of four microalgae by the digestive enzymes of gilthead sea bream and Senegalese sole. Algal Res. 2019, 37, 145–153. [Google Scholar] [CrossRef]
- He, H.; Li, X.; Kong, X.; Hua, Y.; Chen, Y. Heat-induced inactivation mechanism of soybean Bowman-Birk inhibitors. Food Chem 2017, 232, 712–720. [Google Scholar] [CrossRef] [PubMed]
- Vagadia, B.H.; Vanga, S.K.; Raghavan, V. Inactivation methods of soybean trypsin inhibitor—A review. Trends Food Sci. Technol. 2017, 64, 115–125. [Google Scholar] [CrossRef]
- Maehre, H.K.; Edvinsen, G.K.; Eilertsen, K.E.; Elvevoll, E.O. Heat treatment increases the protein bioaccessibility in the red seaweed dulse (Palmaria palmata), but not in the brown seaweed winged kelp (Alaria esculenta). J. Appl. Phycol. 2016, 28, 581–590. [Google Scholar] [CrossRef]
- Bijina, B.; Chellappan, S.; Krishna, J.G.; Basheer, S.M.; Elyas, K.K.; Bahkali, A.H.; Chandrasekaran, M. Protease inhibitor from Moringa oleifera with potential for use as therapeutic drug and as seafood preservative. Saudi J. Biol. Sci. 2011, 18, 273–281. [Google Scholar] [CrossRef]
- Chang, T.S. An updated review of tyrosinase inhibitors. Int. J. Mol. Sci. 2009, 10, 2440–2475. [Google Scholar] [CrossRef]
- Sharma, R. Enzyme inhibition: Mechanisms and scope. In Enzyme inhibition and bioapplications; InTech: Rijeka, Croatia, 2012; pp. 1–36. [Google Scholar]
- Dantzger, M.; Vasconcelos, I.M.; Scorsato, V.; Aparicio, R.; Marangoni, S.; Macedo, M.L.R. Bowman—Birk proteinase inhibitor from Clitoria fairchildiana seeds: Isolation, biochemical properties and insecticidal potential. Phytochemistry 2015, 118, 224–235. [Google Scholar] [CrossRef]
- Macedo, M.L.R.; Freire, M.G.M.; Cabrini, E.C.; Toyama, M.H.; Novello, J.C.; Marangoni, S. A trypsin inhibitor from Peltophorum dubium seeds active against pest proteases and its effect on the survival of Anagasta kuehniella (Lepidoptera:Pyralidae). Biochim. Biophys. Gen. Subj. 2003, 1621, 170–182. [Google Scholar] [CrossRef]
- Xu, X.; Liu, J.; Wang, Y.; Si, Y.; Wang, X.; Wang, Z.; Zhang, Q.; Yu, H.; Wang, X. Kunitz-type serine protease inhibitor is a novel participator in anti-bacterial and anti-inflammatory responses in Japanese flounder (Paralichthys olivaceus). Fish Shellfish Immunol. 2018, 80, 22–30. [Google Scholar] [CrossRef]
- Harry, J.B.; Steiner, R.F. A soybean proteinase inhibitor: Thermodynamic and kinetic parameters of association with enzymes. Eur. J. Biochem. 1970, 16, 174–179. [Google Scholar] [CrossRef] [PubMed]
- Avilés-Gaxiola, S.; Chuck-Hernández, C.; del Refugio Rocha-Pizaña, M.; García-Lara, S.; López-Castillo, L.M.; Serna-Saldívar, S.O. Effect of thermal processing and reducing agents on trypsin inhibitor activity and functional properties of soybean and chickpea protein concentrates. LWT Food Sci. Technol. 2018, 98, 629–634. [Google Scholar] [CrossRef]
- Bradford, M. A rapid and sensitive method for the quantitation of microgramquantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Alarcón, F.J.; Díaz, M.; Moyano, F.J.; Abellán, E. Characterization and functional properties of digestive proteases in two sparids; gilthead sea bream (Sparus aurata) and common dentex (Dentex dentex). Fish Physiol. Biochem. 1998, 19, 257–267. [Google Scholar] [CrossRef]
- Alarcón, F.J.; García-Carreño, F.L.; Navarrete del Toro, M.A. Effect of plant protease inhibitors on digestive proteases in two fish species, Lutjanus argentiventris and L. novemfasciatus. Fish Physiol. Biochem. 2001, 24, 179–189. [Google Scholar] [CrossRef]
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef]
- Hamdan, M.; Moyano, F.J.; Schuhardt, D. Optimization of a gastrointestinal model applicable to the evaluation of bioaccessibility in fish feeds. J. Sci. Food Agric. 2009, 89, 1195–1201. [Google Scholar] [CrossRef]
- Church, F.C.; Swaisgood, H.E.; Porter, D.H.; Catignani, G. Spectrophotometric assay using o-phthaldehyde for determination of proteolysis in milk proteins. J. Dairy Sci. 1983, 66, 1219–1227. [Google Scholar] [CrossRef]
- Erlanger, B.; Kokowsky, N.; Cohen, W. The preparation and properties of two new chromogenic substrates of trypsin. Arch. Biochem. Biophys 1961, 95, 271–278. [Google Scholar] [CrossRef]
- DelMar, E.G.; Largman, C.; Brodrick, J.W.; Geokas, M.C. A sensitive new substrate for chymotrypsin. Anal. Biochem. 1979, 99, 316–320. [Google Scholar] [CrossRef]
| Fish vs. Ulva Concentration | CPD (%) | Total Amino Acids Released (g 100 g protein−1) |
|---|---|---|
| Sparus aurata | ||
| Control | 91.6 ± 2.3 d | 22.1 ± 2.3 d |
| CR-Ulva 500 | 77.9 ± 2.0 c | 15.4 ± 0.1 c |
| CR-Ulva 1000 | 62.6 ± 4.1 b | 11.1 ± 0.7 b |
| CR-Ulva 1500 | 45.9 ± 2.0 a | 7.4 ± 2.4 a |
| HT-Ulva 500 | 91.4 ± 2.1 d | 20.7 ± 1.0 d |
| HT-Ulva 1000 | 90.6 ± 1.0 d | 20.8 ± 1.6 d |
| HT-Ulva 1500 | 89.2 ± 1.7 d | 20.7 ± 2.7 d |
| p | <0.001 | <0.001 |
| Solea senegalensis | ||
| Control | 91.9 ± 2.2 d | 22.1 ± 1.5 c |
| CR-Ulva 500 | 84.3 ± 0.4 c | 19.6 ± 0.8 c |
| CR-Ulva 1000 | 66.1 ± 1.5 b | 12.8 ± 0.3 b |
| CR-Ulva 1500 | 41.8 ± 4.4 a | 8.5 ± 1.0 a |
| HT-Ulva 500 | 89.9 ± 1.2 d | 21.9 ± 1.8 c |
| HT-Ulva 1000 | 89.1 ± 2.1 d | 21.5 ± 2.0 c |
| HT-Ulva 1500 | 88.2 ± 1.3 d | 20.8 ± 0.8 c |
| p | <0.001 | <0.001 |
| Dicentrarchus labrax | ||
| Control | 96.2 ± 1.2 c | 22.3 ± 2.5 d |
| CR-Ulva 500 | 91.2 ± 0.8 b | 16.3 ± 1.0 c |
| CR-Ulva 1000 | 69.9 ± 1.4 a | 11.8 ± 0.3 b |
| CR-Ulva 1500 | 66.6 ± 2.5 a | 8.2 ± 0.3 a |
| HT-Ulva 500 | 95.1 ± 1.8 b,c | 22.0 ± 2.3 d |
| HT-Ulva 1000 | 94.1 ± 3.2 b,c | 21.3 ± 0.6 d |
| HT-Ulva 1500 | 93.4 ± 1.4 b | 20.1 ± 0.6 d |
| p | <0.001 | <0.001 |
| Ulva Concentration | Trypsin | Chymotrypsin | ||
|---|---|---|---|---|
| Km (mM) | Vmax (µMmol pNA min−1) | Km (mM) | Vmax (µMmol pNA min−1) | |
| Without Ulva | 0.65 | 7.03 | 0.15 | 5.92 |
| 200 µg Ulva per µg enzyme | 0.73 | 5.54 | 0.20 | 5.57 |
| 300 µg Ulva per µg enzyme | 0.87 | 4.71 | 0.21 | 3.15 |
| 400 µg Ulva per µg enzyme | 1.58 | 4.43 | 0.23 | 2.38 |
| 500 µg Ulva per µg enzyme | 2.53 | 4.12 | 0.25 | 1.65 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vizcaíno, A.J.; Galafat, A.; Sáez, M.I.; Martínez, T.F.; Alarcón, F.J. Partial Characterization of Protease Inhibitors of Ulva ohnoi and Their Effect on Digestive Proteases of Marine Fish. Mar. Drugs 2020, 18, 319. https://doi.org/10.3390/md18060319
Vizcaíno AJ, Galafat A, Sáez MI, Martínez TF, Alarcón FJ. Partial Characterization of Protease Inhibitors of Ulva ohnoi and Their Effect on Digestive Proteases of Marine Fish. Marine Drugs. 2020; 18(6):319. https://doi.org/10.3390/md18060319
Chicago/Turabian StyleVizcaíno, Antonio Jesús, Alba Galafat, María Isabel Sáez, Tomás Francisco Martínez, and Francisco Javier Alarcón. 2020. "Partial Characterization of Protease Inhibitors of Ulva ohnoi and Their Effect on Digestive Proteases of Marine Fish" Marine Drugs 18, no. 6: 319. https://doi.org/10.3390/md18060319
APA StyleVizcaíno, A. J., Galafat, A., Sáez, M. I., Martínez, T. F., & Alarcón, F. J. (2020). Partial Characterization of Protease Inhibitors of Ulva ohnoi and Their Effect on Digestive Proteases of Marine Fish. Marine Drugs, 18(6), 319. https://doi.org/10.3390/md18060319







