Blue Mussel-Derived Peptides PIISVYWK and FSVVPSPK Trigger Wnt/β-Catenin Signaling-Mediated Osteogenesis in Human Bone Marrow Mesenchymal Stem Cells
Abstract
1. Introduction
2. Results
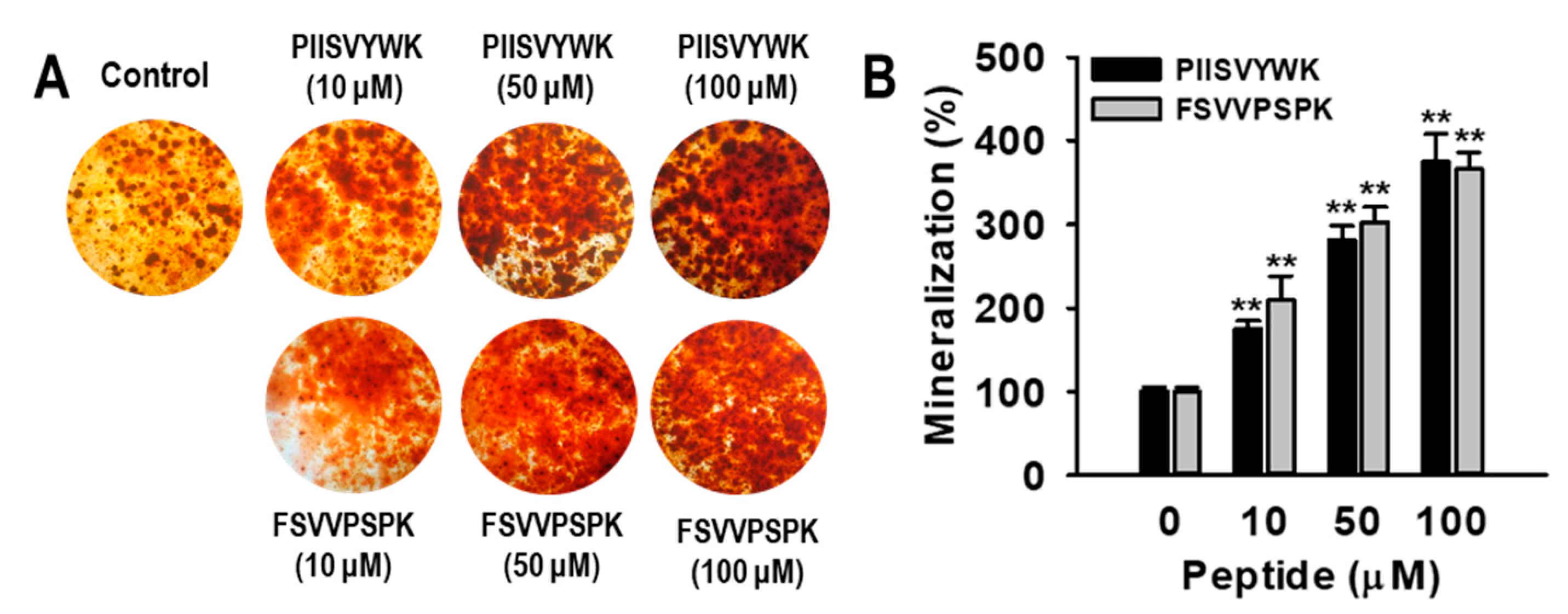
2.1. Blue Mussel-Derived Peptides PIISVYWK and FSVVPSPK Increase Osteoblast Differentiation in Human Bone Marrow-Derived Mesenchymal Stem Cells (hBMMSCs)
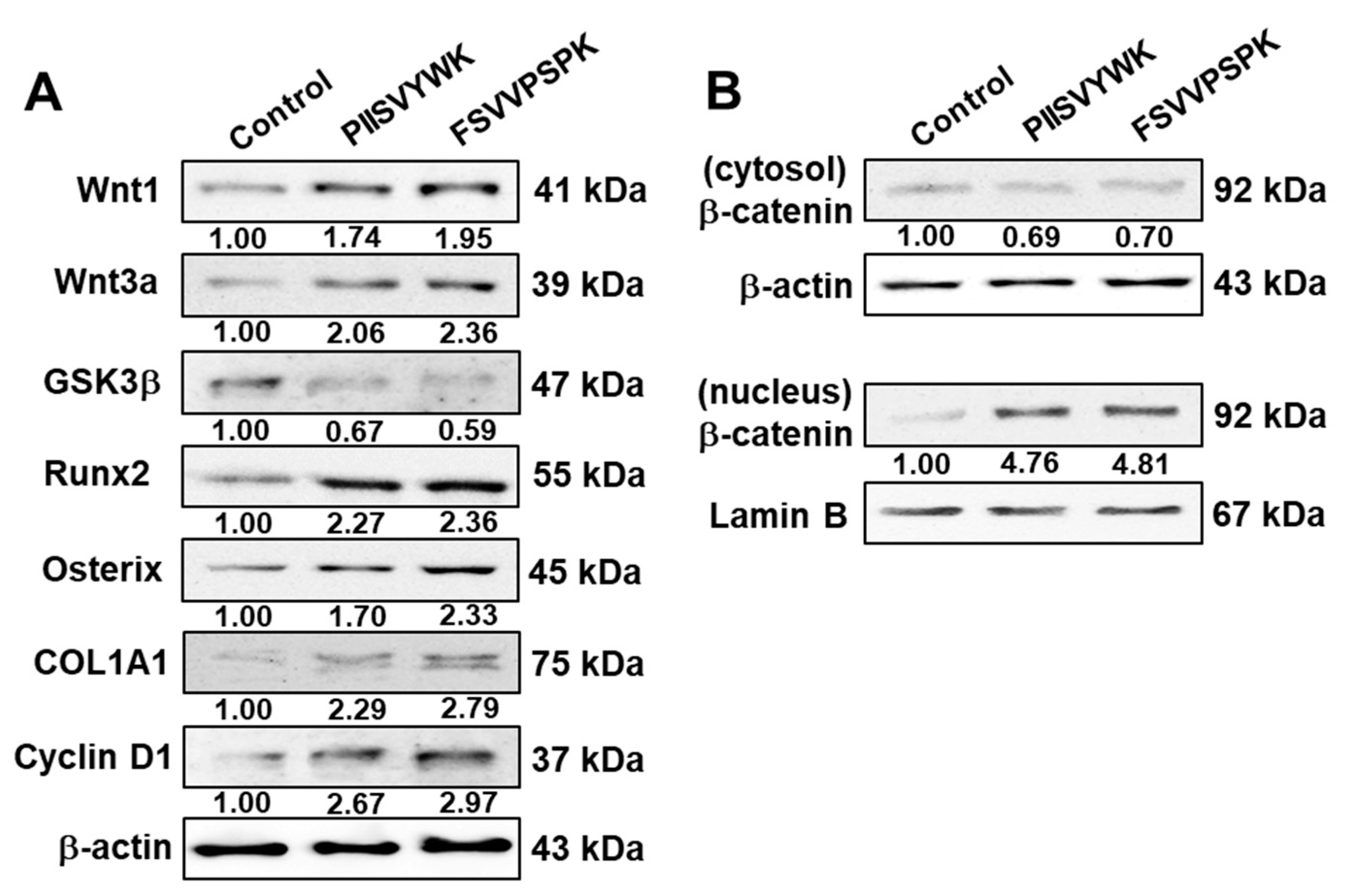
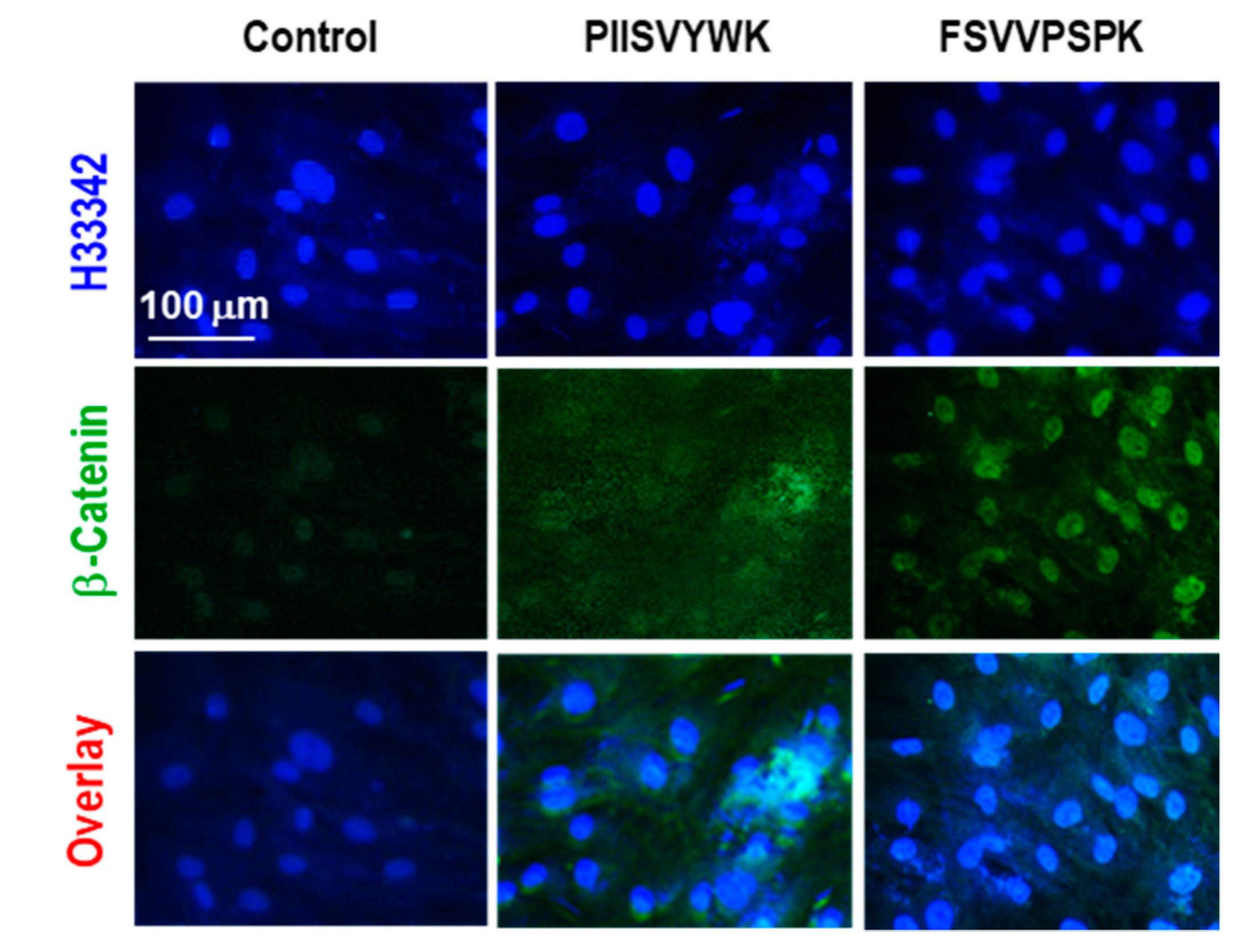
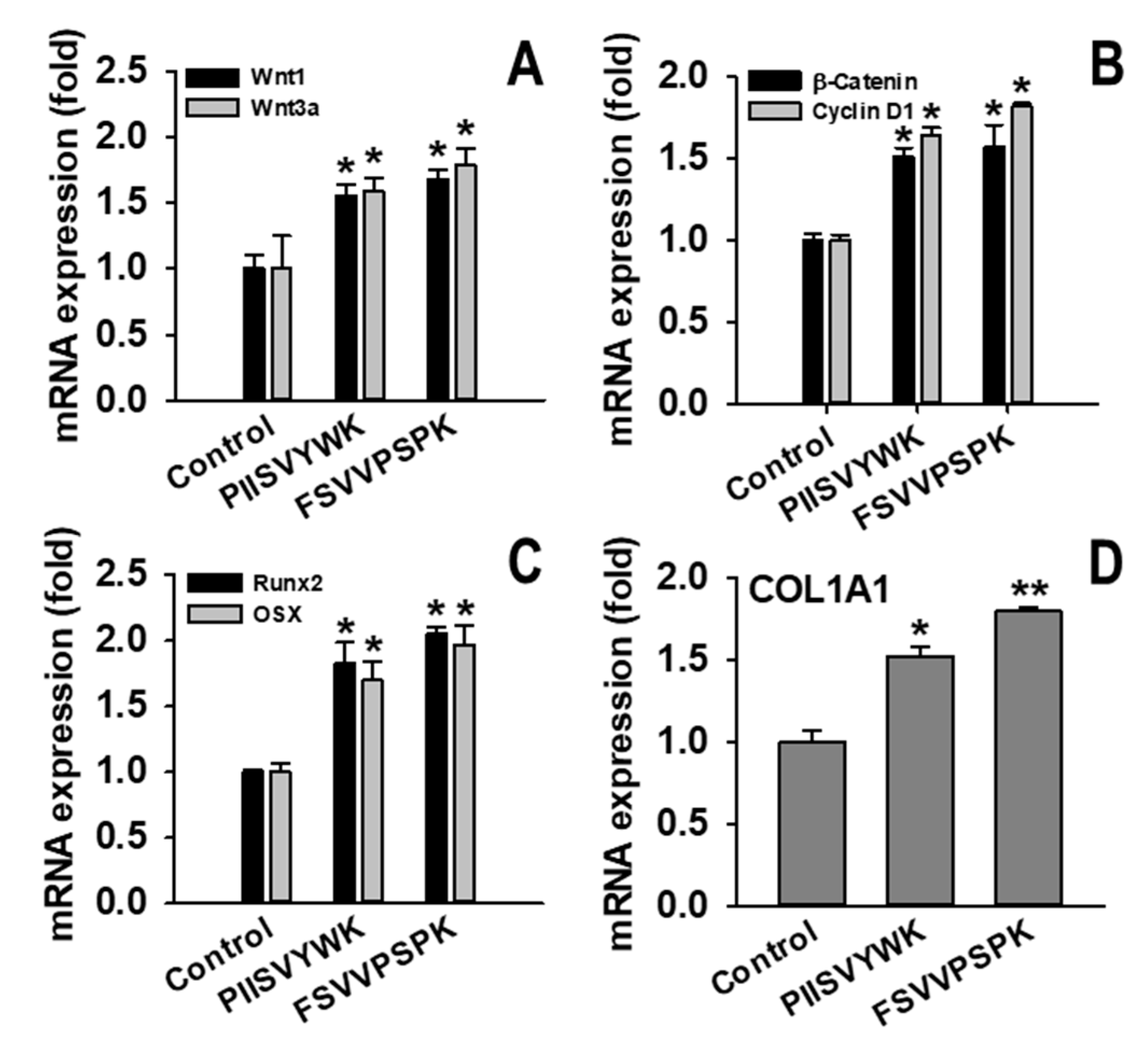
2.2. PIISVYWK and FSVVPSPK Activates Wnt/Β-Catenin Signaling
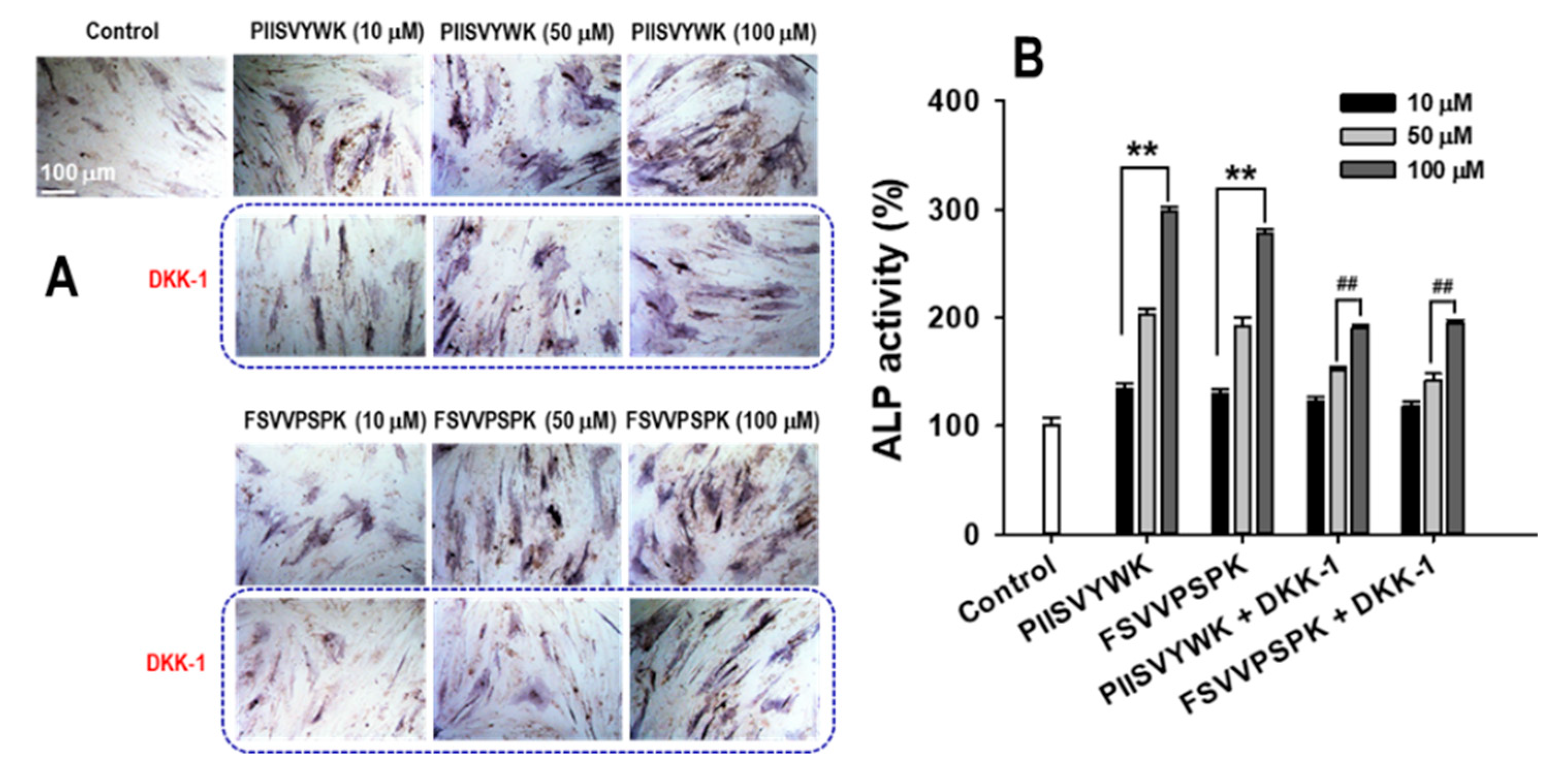
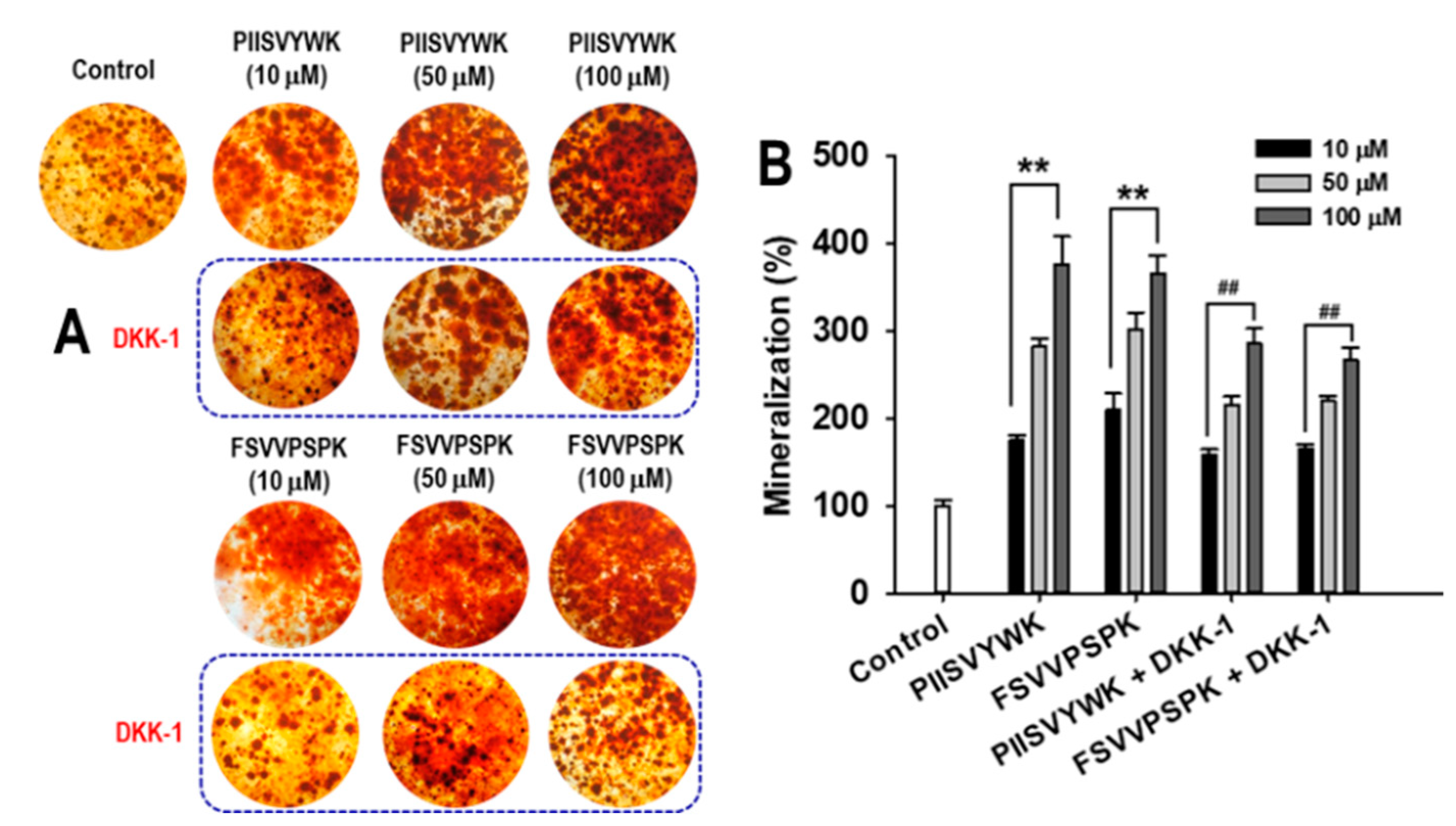
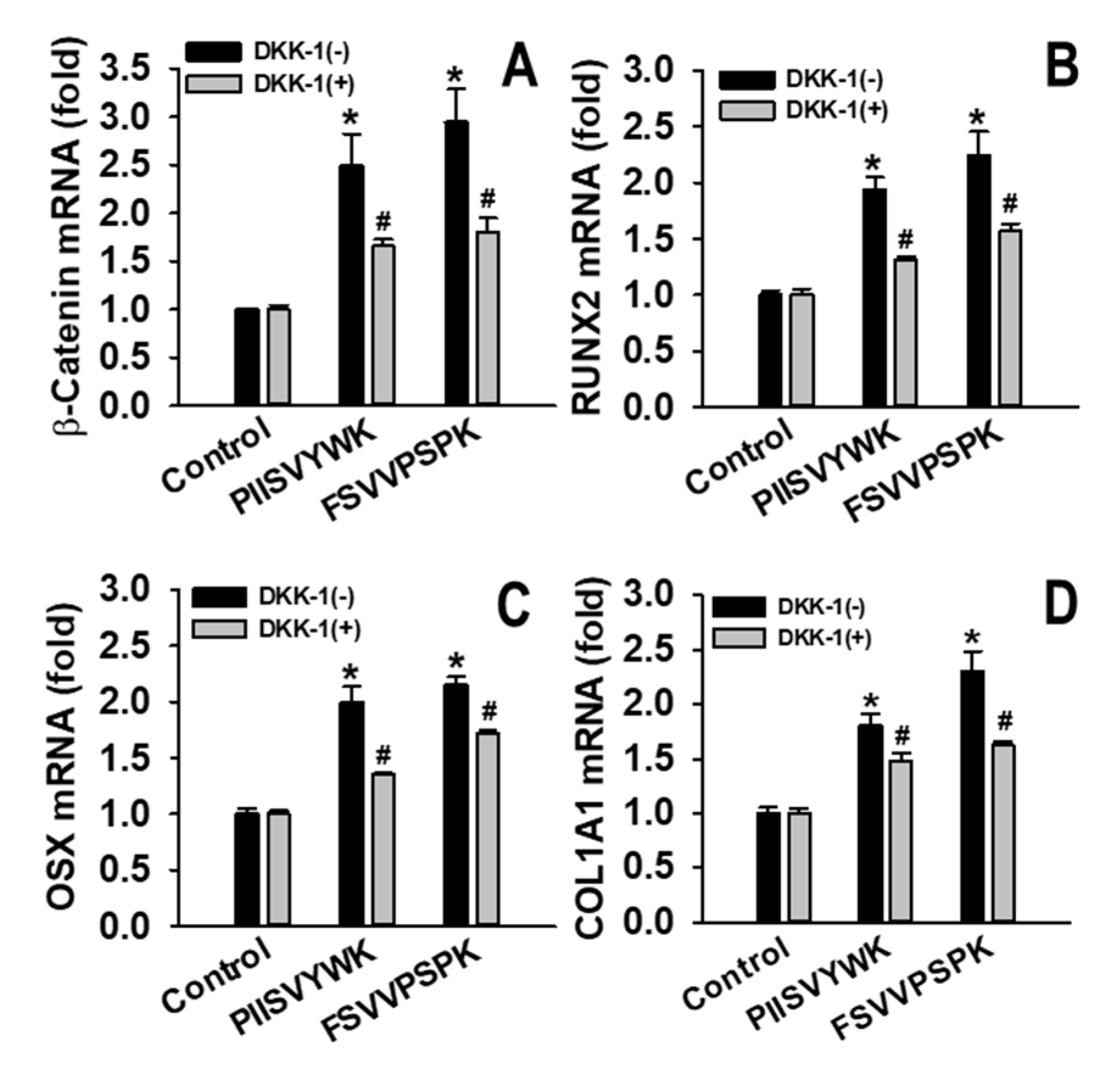
2.3. Dickkopf-1 (DKK-1) Reversed PIISVYWK and FSVVPSPK-Enhanced Osteoblast Differentiation
3. Discussion
4. Materials and Methods
4.1. Peptide Synthesis
4.2. Osteogenic Differentiation and Treatment of Peptides
4.3. MTT Assay
4.4. ALP Activity
4.5. Mineralization Assay
4.6. Western Blot Analysis
4.7. Real-Time Quantitative Polymerase Chain Reaction (RT-qPCR)
4.8. Immunofluorescence Staining for β-Catenin Nuclear Translocation
4.9. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Xia, G.; Wang, S.; He, M.; Zhou, X.; Zhao, Y.; Wang, J.; Xue, C. Anti-osteoporotic activity of sialoglycoproteins isolated from the eggs of Carassius auratus by promoting osteogenesis and increasing OPG/RANKL ratio. J. Funct. Foods 2015, 15, 137–150. [Google Scholar] [CrossRef]
- Reid, I.R. Anti-resorptive therapies for osteoporosis. Semin. Cell Dev. Biol. 2008, 19, 473–478. [Google Scholar] [CrossRef]
- Sanna, G.; Zampino, M.; Pelosi, G.; Nole, F.; Goldhirsch, A. Jaw avascular bone necrosis associated with long-term use of biphosphonates. Ann. Oncol. 2005, 16, 1207–1208. [Google Scholar] [CrossRef]
- Tao, K.; Xiao, D.; Weng, J.; Xiong, A.; Kang, B.; Zeng, H. Berberine promotes bone marrow-derived mesenchymal stem cells osteogenic differentiation via canonical Wnt/β-catenin signaling pathway. Toxicol. Lett. 2016, 240, 68–80. [Google Scholar] [CrossRef]
- Lee, H.W.; Suh, J.H.; Kim, H.N.; Kim, A.Y.; Park, S.Y.; Shin, C.S.; Choi, J.Y.; Kim, J.B. Berberine promotes osteoblast differentiation by Runx2 activation with p38 MAPK. J. Bone Miner. Res. 2008, 23, 1227–1237. [Google Scholar] [CrossRef]
- Yun, H.M.; Park, K.R.; Quang, T.H.; Oh, H.; Hong, J.T.; Kim, Y.C.; Kim, E.C. 2,4,5-Trimethoxyldalbergiquinol promotes osteoblastic differentiation and mineralization via the BMP and Wnt/beta-catenin pathway. Cell Death Dis. 2015, 6, e1819. [Google Scholar] [CrossRef]
- Zhang, J.-F.; Li, G.; Chan, C.-Y.; Meng, C.-L.; Lin, M.C.-M.; Chen, Y.-C.; He, M.-L.; Leung, P.-C.; Kung, H.-F. Flavonoids of Herba Epimedii regulate osteogenesis of human mesenchymal stem cells through BMP and Wnt/β-catenin signaling pathway. Mol. Cell. Endocrinol. 2010, 314, 70–74. [Google Scholar] [CrossRef]
- Majumdar, M.K.; Thiede, M.A.; Mosca, J.D.; Moorman, M.; Gerson, S.L. Phenotypic and functional comparison of cultures of marrow-derived mesenchymal stem cells (MSCs) and stromal cells. J. Cell. Physiol. 1998, 176, 57–66. [Google Scholar] [CrossRef]
- Lin, G.L.; Hankenson, K.D. Integration of BMP, Wnt, and notch signaling pathways in osteoblast differentiation. J. Cell. Biochem. 2011, 112, 3491–3501. [Google Scholar] [CrossRef]
- Peng, S.; Zhou, G.; Luk, K.D.; Cheung, K.M.; Li, Z.; Lam, W.M.; Zhou, Z.; Lu, W.W. Strontium promotes osteogenic differentiation of mesenchymal stem cells through the Ras/MAPK signaling pathway. Cell. Physiol. Biochem. 2009, 23, 165–174. [Google Scholar] [CrossRef]
- Majidinia, M.; Sadeghpour, A.; Yousefi, B. The roles of signaling pathways in bone repair and regeneration. J. Cell. Physiol. 2018, 233, 2937–2948. [Google Scholar] [CrossRef] [PubMed]
- Karner, C.M.; Long, F. Wnt signaling and cellular metabolism in osteoblasts. Cell. Mol. Life Sci. 2017, 74, 1649–1657. [Google Scholar] [CrossRef] [PubMed]
- Ngo, D.-H.; Vo, T.-S.; Ngo, D.-N.; Wijesekara, I.; Kim, S.-K. Biological activities and potential health benefits of bioactive peptides derived from marine organisms. Int. J. Biol. Macromol. 2012, 51, 378–383. [Google Scholar] [CrossRef] [PubMed]
- Oh, Y.; Ahn, C.-B.; Nam, K.H.; Kim, Y.K.; Yoon, N.Y.; Je, J.-Y. Amino Acid Composition, Antioxidant, and Cytoprotective Effect of Blue Mussel (Mytilus edulis) Hydrolysate through the Inhibition of Caspase-3 Activation in Oxidative Stress-Mediated Endothelial Cell Injury. Mar. Drugs 2019, 17, 135. [Google Scholar] [CrossRef]
- Oh, Y.; Shim, K.-B.; Ahn, C.-B.; Kim, S.S.; Je, J.-Y. Sea Squirt (Halocynthia roretzi) Hydrolysates Induce Apoptosis in Human Colon Cancer HT-29 Cells through Activation of Reactive Oxygen Species. Nutr. Cancer 2019, 71, 118–127. [Google Scholar] [CrossRef]
- Oh, Y.; Ahn, C.-B.; Yoon, N.Y.; Nam, K.H.; Kim, Y.-K.; Je, J.-Y. Protective effect of enzymatic hydrolysates from seahorse (Hippocampus abdominalis) against H2O2-mediated human umbilical vein endothelial cell injury. Biomed. Pharmacother. 2018, 108, 103–110. [Google Scholar] [CrossRef]
- Park, S.Y.; Je, J.-Y.; Ahn, C.-B. Protein Hydrolysates and Ultrafiltration Fractions Obtained from Krill (Euphausia superba): Nutritional, Functional, Antioxidant, and ACE-Inhibitory Characterization. J. Aquat. Food Prod. Technol. 2016, 25, 1266–1277. [Google Scholar] [CrossRef]
- Pérez, M.J.; Falqué, E.; Domínguez, H. Antimicrobial action of compounds from marine seaweed. Mar. Drugs 2016, 14, 52. [Google Scholar] [CrossRef]
- Kim, S.S.; Ahn, C.-B.; Moon, S.W.; Je, J.-Y. Purification and antioxidant activities of peptides from sea squirt (Halocynthia roretzi) protein hydrolysates using pepsin hydrolysis. Food Biosci. 2018, 25, 128–133. [Google Scholar] [CrossRef]
- Ruiz-Ruiz, F.; Mancera-Andrade, E.I.; Iqbal, H.M. Marine-Derived Bioactive Peptides for Biomedical Sectors: A Review. Protein Pept. Lett. 2017, 24, 109–117. [Google Scholar] [CrossRef]
- Ahn, C.B.; Je, J.Y. Bone health-promoting bioactive peptides. J. Food Biochem. 2019, 43, e12529. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.; Bai, L.; Zhu, L.; Yang, L.; Zhang, X. Marine Algae-Derived Bioactive Peptides for Human Nutrition and Health. J. Agric. Food Chem. 2014, 62, 9211–9222. [Google Scholar] [CrossRef] [PubMed]
- Elango, J.; Robinson, J.; Zhang, J.; Bao, B.; Ma, N.; de Val, J.E.M.S.; Wu, W. Collagen peptide upregulates osteoblastogenesis from bone marrow mesenchymal stem cells through MAPK-Runx2. Cells 2019, 8, 446. [Google Scholar] [CrossRef] [PubMed]
- Oh, Y.; Ahn, C.-B.; Je, J.-Y. Ark shell protein-derived bioactive peptides promote osteoblastic differentiation through upregulation of the canonical Wnt/β-catenin signaling in human bone marrow-derived mesenchymal stem cells. J. Food Biochem. 2020, e13440. [Google Scholar] [CrossRef]
- Hyung, J.-H.; Ahn, C.-B.; Je, J.-Y. Osteoblastogenic activity of ark shell protein hydrolysates with low molecular weight in mouse mesenchymal stem cells. RSC Adv. 2016, 6, 29365–29370. [Google Scholar] [CrossRef]
- Hyung, J.-H.; Ahn, C.-B.; Je, J.-Y. Ark shell protein hydrolysates inhibit adipogenesis in mouse mesenchymal stem cells through the down-regulation of transcriptional factors. RSC Adv. 2017, 7, 6223–6228. [Google Scholar] [CrossRef]
- Hyung, J.-H.; Ahn, C.-B.; Je, J.-Y. Blue mussel (Mytilus edulis) protein hydrolysate promotes mouse mesenchymal stem cell differentiation into osteoblasts through up-regulation of bone morphogenetic protein. Food Chem. 2018, 242, 156–161. [Google Scholar] [CrossRef]
- Oh, Y.; Ahn, C.-B.; Hyung, J.-H.; Je, J.-Y. Two novel peptides from ark shell protein stimulate osteoblast differentiation and rescue ovariectomy-induced bone loss. Toxicol. Appl. Pharmacol. 2019, 385, 114779. [Google Scholar] [CrossRef]
- Park, S.Y.; Kim, Y.-S.; Ahn, C.-B.; Je, J.-Y. Partial purification and identification of three antioxidant peptides with hepatoprotective effects from blue mussel (Mytilus edulis) hydrolysate by peptic hydrolysis. J. Funct. Foods 2016, 20, 88–95. [Google Scholar] [CrossRef]
- Moorer, M.C.; Riddle, R.C. Regulation of osteoblast metabolism by Wnt signaling. Endocrinol. Metab. 2018, 33, 318–330. [Google Scholar] [CrossRef]
- Blair, H.C.; Larrouture, Q.C.; Li, Y.; Lin, H.; Beer-Stoltz, D.; Liu, L.; Tuan, R.S.; Robinson, L.J.; Schlesinger, P.H.; Nelson, D.J. Osteoblast Differentiation and Bone Matrix Formation In Vivo and In Vitro. Tissue Eng. Part B Rev. 2017, 23, 268–280. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Bian, Y.Q.; Tao, H.M.; Yang, X.F.; Mu, W.D. Simvastatin induces osteogenic differentiation of MSCs via Wnt/β-catenin pathway to promote fracture healing. Eur. Rev. Med Pharmacol. Sci. 2018, 22, 2896–2905. [Google Scholar] [PubMed]
- Day, T.F.; Guo, X.; Garrett-Beal, L.; Yang, Y. Wnt/beta-catenin signaling in mesenchymal progenitors controls osteoblast and chondrocyte differentiation during vertebrate skeletogenesis. Dev. Cell 2005, 8, 739–750. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, Y.; Maeda, K.; Takahashi, N. Roles of Wnt signaling in bone formation and resorption. Jpn. Dent. Sci. Rev. 2008, 44, 76–82. [Google Scholar] [CrossRef]
- Gaur, T.; Lengner, C.J.; Hovhannisyan, H.; Bhat, R.A.; Bodine, P.V.; Komm, B.S.; Javed, A.; van Wijnen, A.J.; Stein, J.L.; Stein, G.S.; et al. Canonical WNT signaling promotes osteogenesis by directly stimulating Runx2 gene expression. J. Biol. Chem. 2005, 280, 33132–33140. [Google Scholar] [CrossRef]
- Funck-Brentano, T.; Bouaziz, W.; Marty, C.; Geoffroy, V.; Hay, E.; Cohen-Solal, M. Dkk-1-mediated inhibition of Wnt signaling in bone ameliorates osteoarthritis in mice. Arthritis Rheumatol. 2014, 66, 3028–3039. [Google Scholar] [CrossRef]

| Genes | Forward Primer | Reverse Primer |
|---|---|---|
| Wnt1 | AACAGCGGCGTCTGATAC | GCGGAGGTGATAGCGAAG |
| Wnt3a | ATGAACCGCCACAACAAC | TTCTCCACCACCATCTCC |
| Runx2 | GGAGTGGACGAGGCAAGAGTTT | AGCTTCTGTCTGTGCCTTCTGG |
| Osterix | CCTCTGCGGGACTCAACAAC | AGCCCATTAGTGCTTGTAAAGG |
| β-catenin | AGCTTCCAGACACGCTATCAT | ATGACCCTGTAGGCAGAAACC |
| COL1A1 | GACGTCCTGGTGAAGTTGGT | ACCAGGGAAGCCTCTCTCTC |
| Cyclin D1 | CCCTCGGTCTCCTACTTCA | GTTTTCTCCTCCGCCTCT |
| β-actin | CAATGTGGCCGAGGACTTT | CATTCTCCTTAGAGAGAAGTGG |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oh, Y.; Ahn, C.-B.; Je, J.-Y. Blue Mussel-Derived Peptides PIISVYWK and FSVVPSPK Trigger Wnt/β-Catenin Signaling-Mediated Osteogenesis in Human Bone Marrow Mesenchymal Stem Cells. Mar. Drugs 2020, 18, 510. https://doi.org/10.3390/md18100510
Oh Y, Ahn C-B, Je J-Y. Blue Mussel-Derived Peptides PIISVYWK and FSVVPSPK Trigger Wnt/β-Catenin Signaling-Mediated Osteogenesis in Human Bone Marrow Mesenchymal Stem Cells. Marine Drugs. 2020; 18(10):510. https://doi.org/10.3390/md18100510
Chicago/Turabian StyleOh, Yunok, Chang-Bum Ahn, and Jae-Young Je. 2020. "Blue Mussel-Derived Peptides PIISVYWK and FSVVPSPK Trigger Wnt/β-Catenin Signaling-Mediated Osteogenesis in Human Bone Marrow Mesenchymal Stem Cells" Marine Drugs 18, no. 10: 510. https://doi.org/10.3390/md18100510
APA StyleOh, Y., Ahn, C.-B., & Je, J.-Y. (2020). Blue Mussel-Derived Peptides PIISVYWK and FSVVPSPK Trigger Wnt/β-Catenin Signaling-Mediated Osteogenesis in Human Bone Marrow Mesenchymal Stem Cells. Marine Drugs, 18(10), 510. https://doi.org/10.3390/md18100510





