Marine Natural Products: A Source of Novel Anticancer Drugs
Abstract
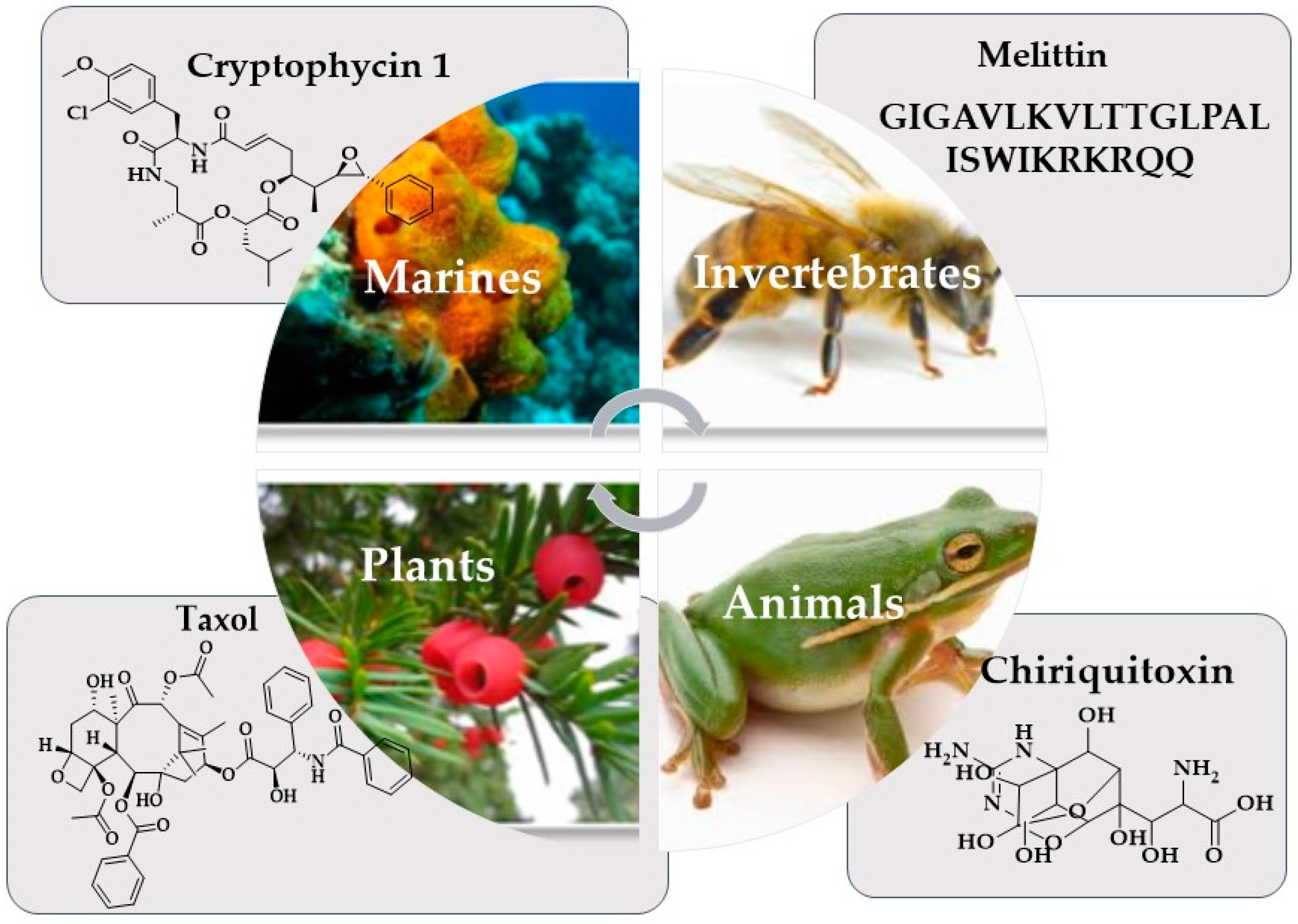
1. Introduction
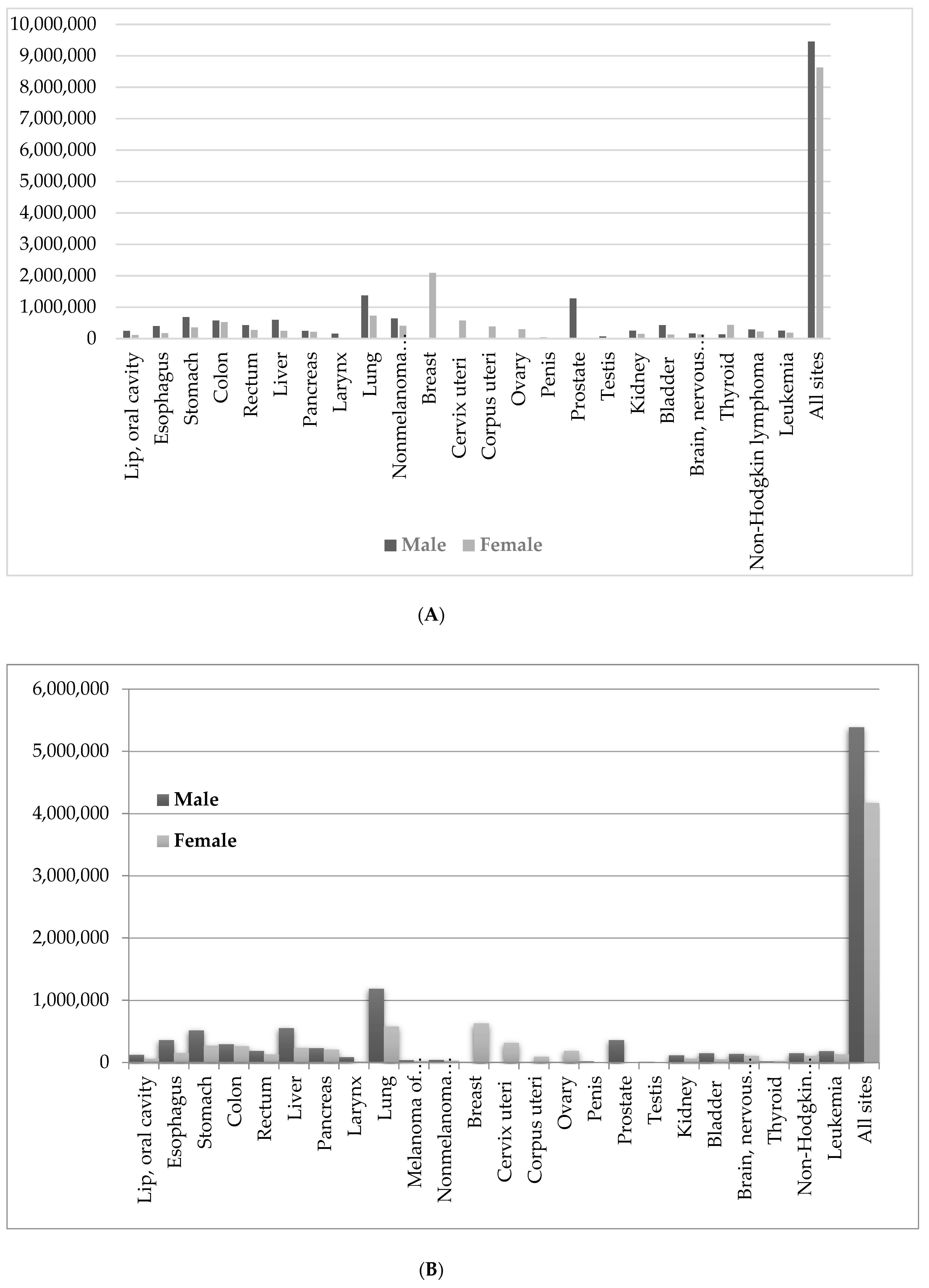
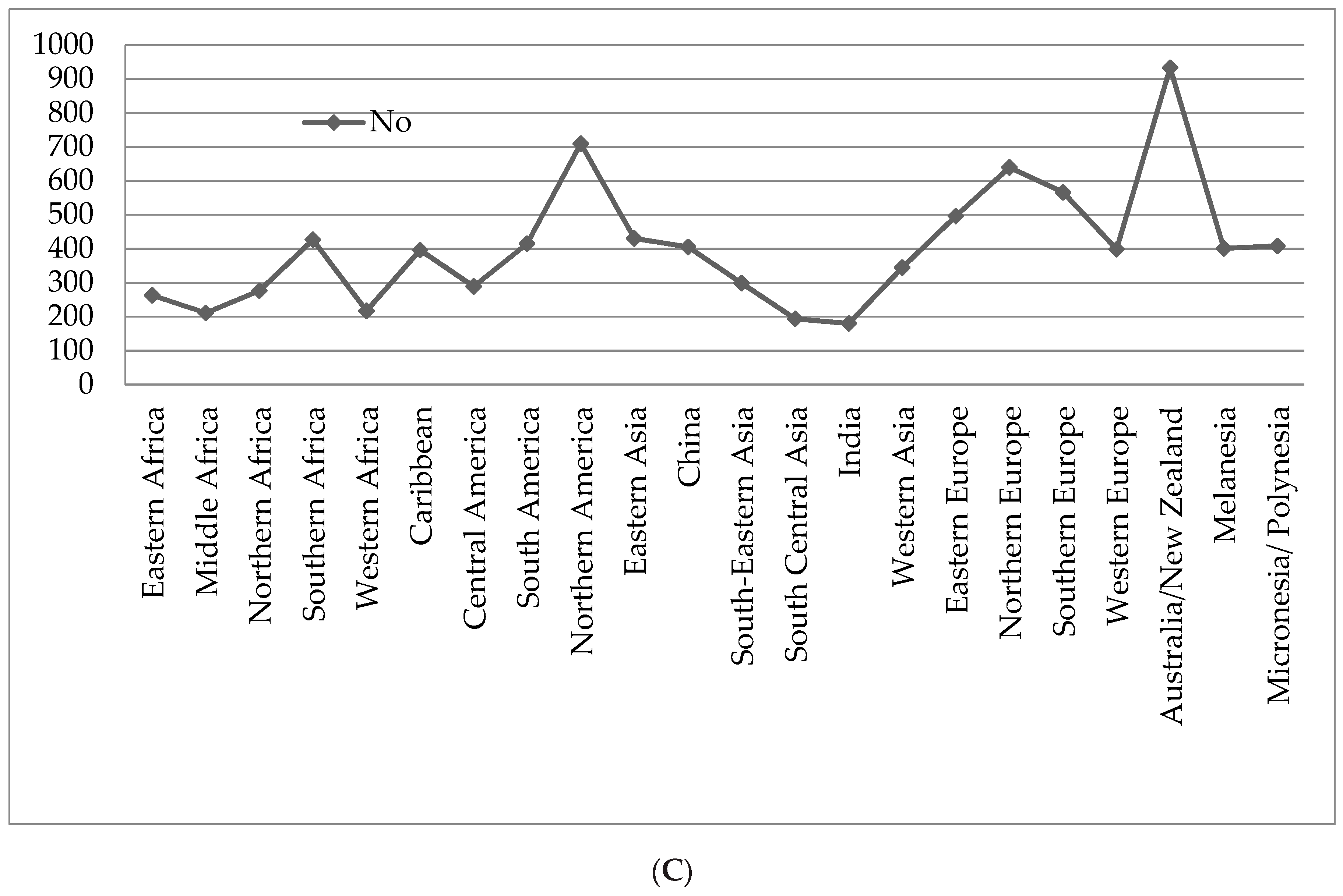
2. Nature and Cancer Chemotherapy
3. Marine Organisms and Cancer Chemotherapy
4. Characterization of Marine Metabolites
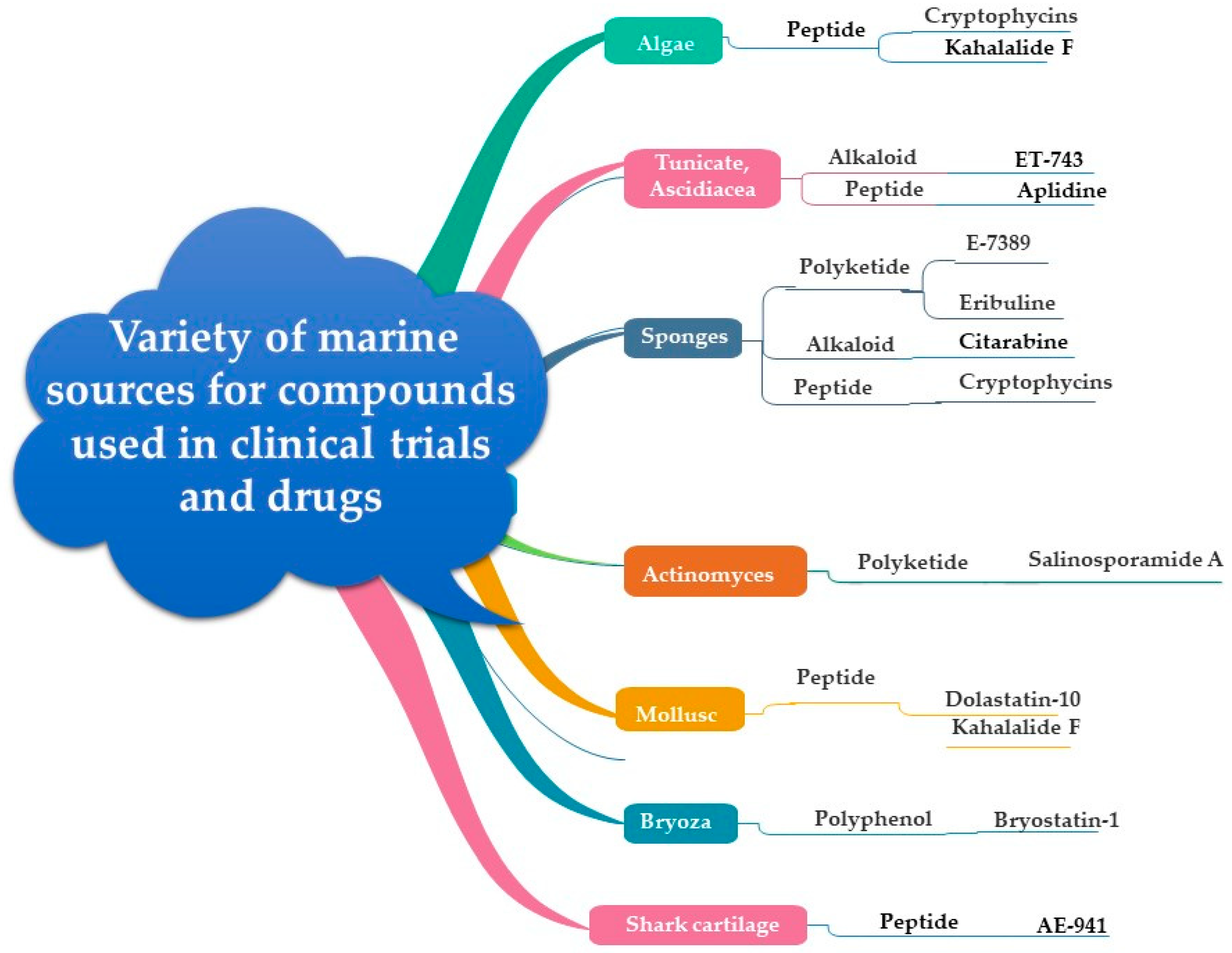
5. From Marine Organisms to Anticancer Drugs
5.1. Marine Plants
5.1.1. Macroalgae (Seaweed)
5.1.2. Microalgae
5.2. Marine Bacteria
5.3. Marine Actinomycetes
5.4. Marine Fungi
5.5. Marine Sponges
5.6. Soft Corals
6. Bioactive Constituents of Marine Organisms
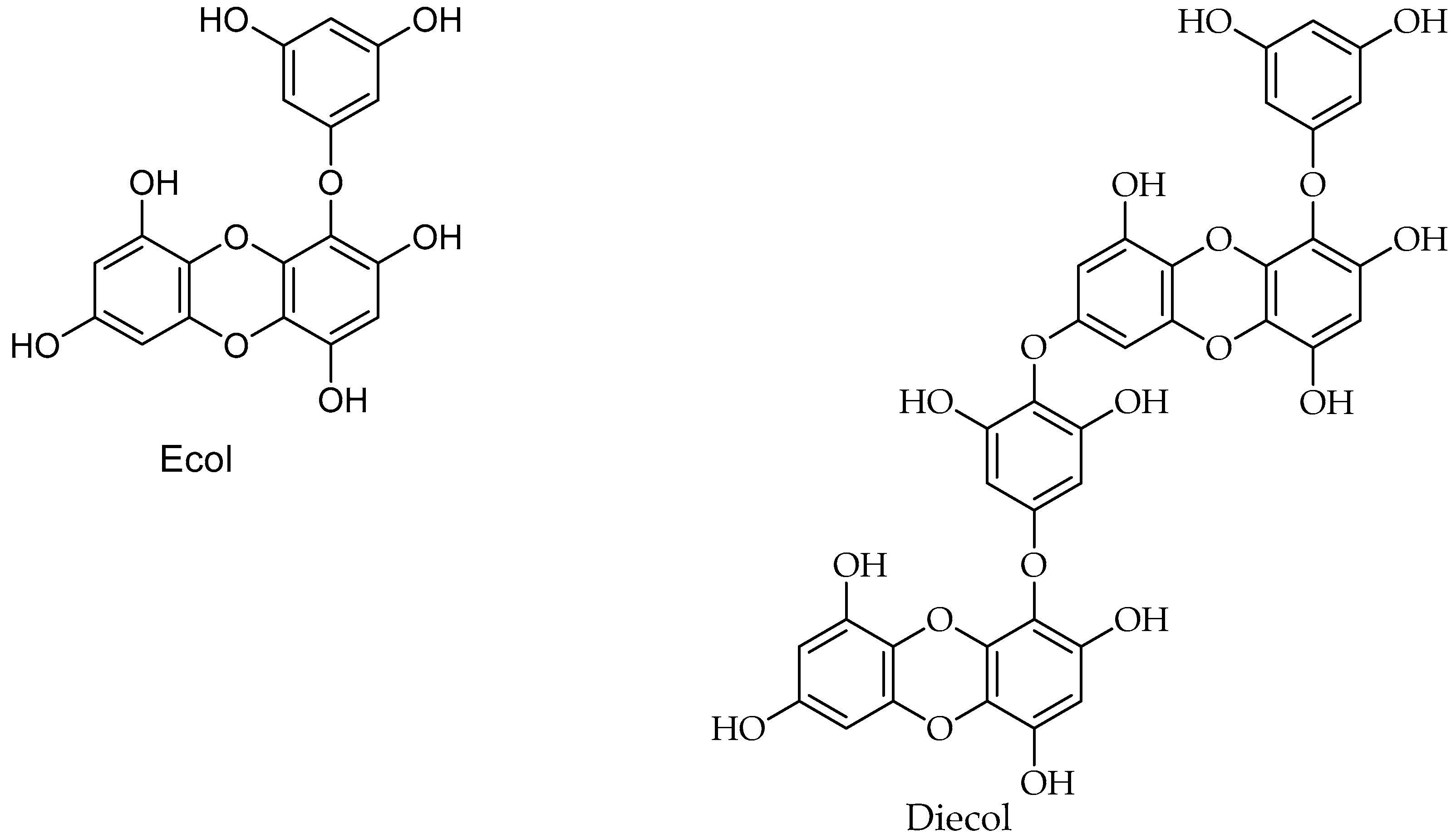
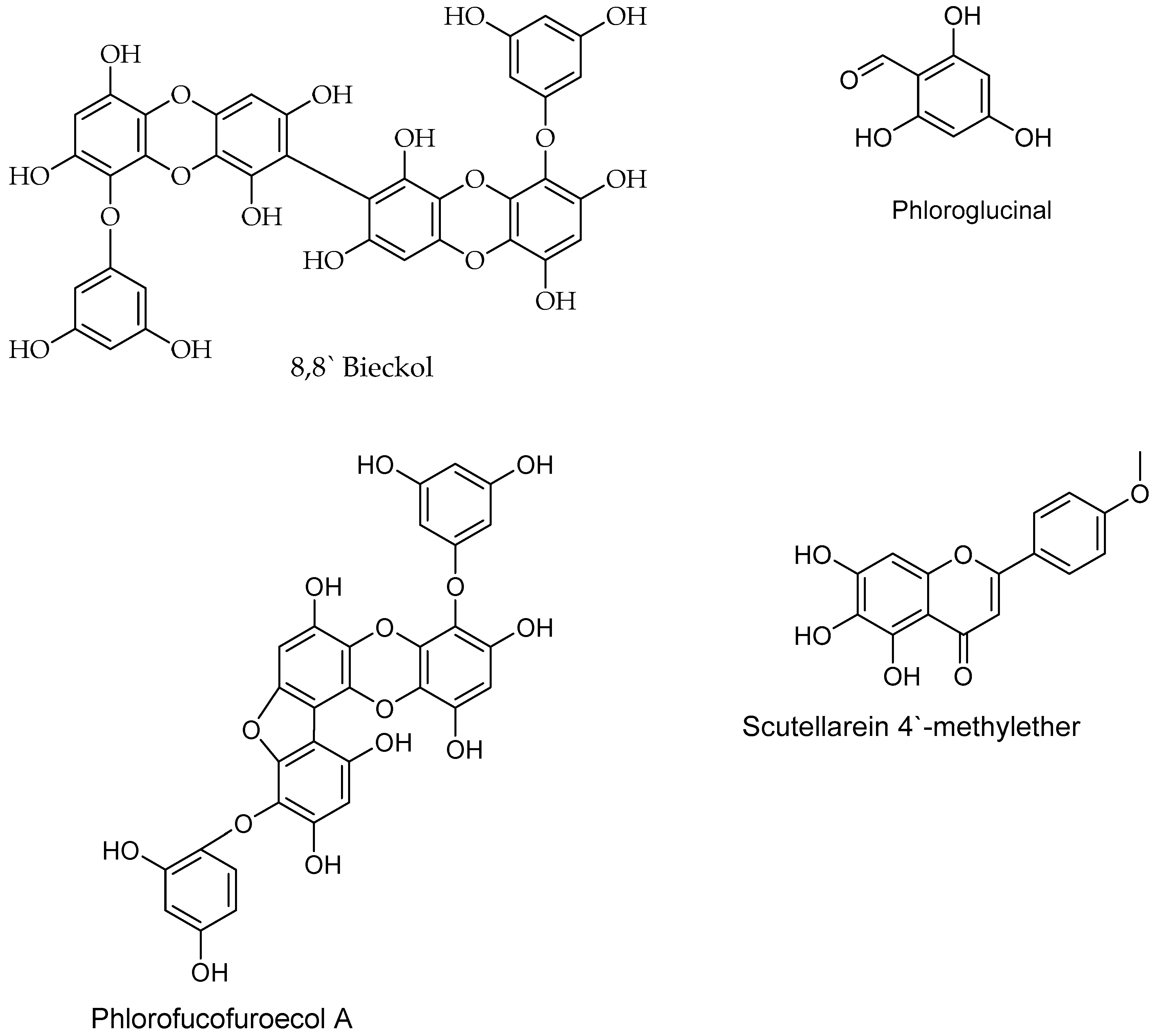
6.1. Polyphenols
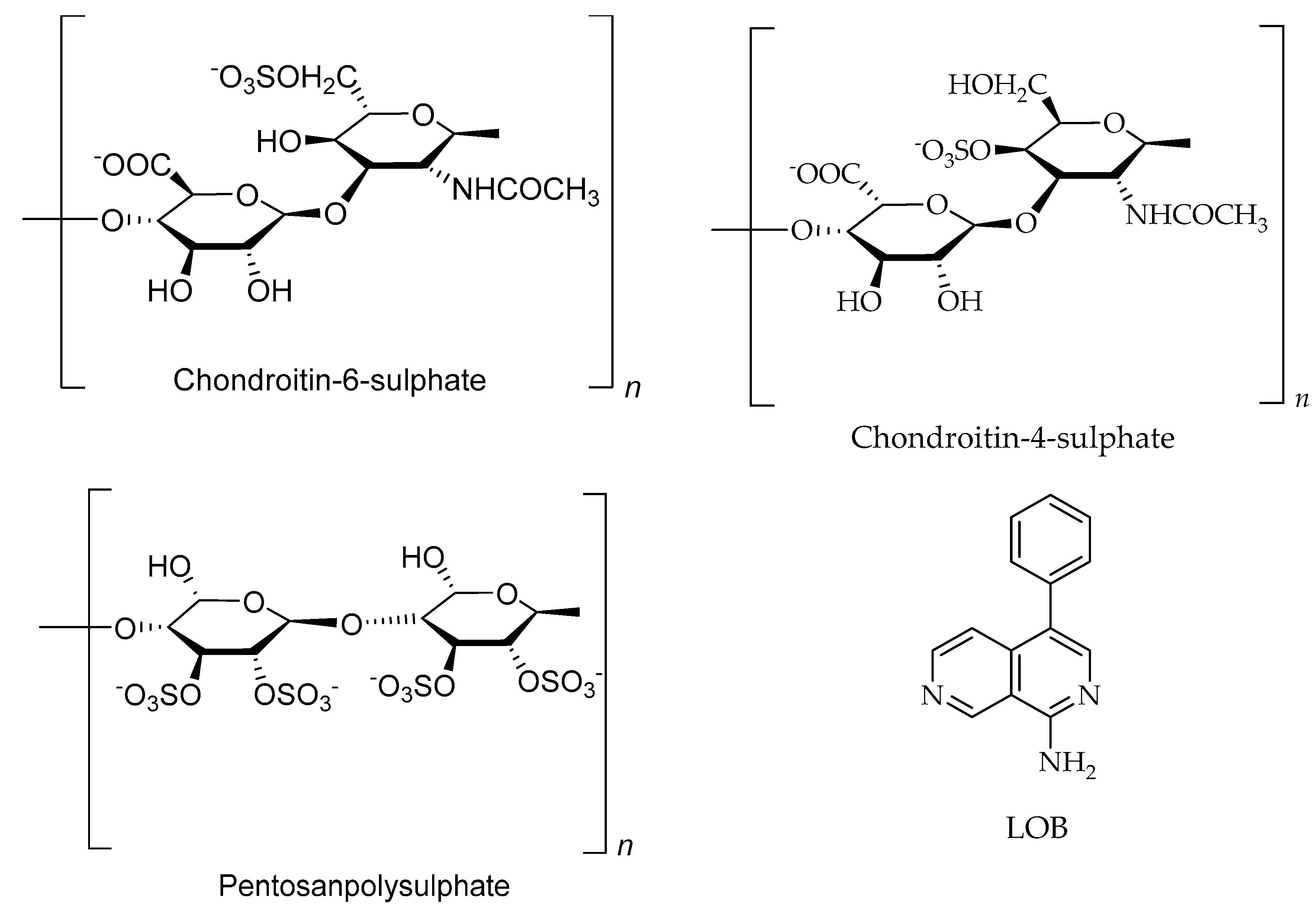
6.2. Polysaccharides
6.3. Alkaloids
6.4. Peptides
7. Anticancer Bioactive Antibiotics Derived from Marine Sources
8. Chemical Property Space
9. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA A Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- White, M.C.; Holman, D.M.; Boehm, J.E.; Peipins, L.A.; Grossman, M.; Henley, S.J. Age and cancer risk: A potentially modifiable relationship. Am. J. Prev. Med. 2014, 46, S7–S15. [Google Scholar] [CrossRef] [PubMed]
- Gurib-Fakim, A. Medicinal plants: Traditions of yesterday and drugs of tomorrow. Mol. Asp. Med. 2006, 27, 93. [Google Scholar] [CrossRef] [PubMed]
- Ramsay, R.R.; Popovic-Nikolic, M.R.; Nikolic, K.; Uliassi, E.; Bolognesi, M.L. A perspective on multi-target drug discovery and design for complex diseases. Clin. Transl. Med. 2018, 7, 3. [Google Scholar] [CrossRef] [PubMed]
- Garattini, S.; La Vecchia, C. Perspectives in cancer chemotherapy. Eur. J. Cancer 2001, 37, 128–147. [Google Scholar] [CrossRef]
- Ang, K.K.H.; Holmes, M.J.; Higa, T.; Hamann, M.T.; Kara, U.A.K. In vivo antimalarial activity of the beta-carboline alkaloid manzamine A. Antimicrob. Agents Chemother. 2000, 44, 1645–1649. [Google Scholar] [CrossRef] [PubMed]
- Ang, K.; Holmes, M.; Kara, U. Immune-mediated parasite clearance in mice infected with Plasmodium berghei following treatment with manzamine A. Parasitol. Res. 2001, 87, 715–721. [Google Scholar] [CrossRef] [PubMed]
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer 2015, 136, E359–E386. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Global Status Report on Noncommunicable Diseases 2014; World Health Organization: Geneva, Switzerland, 2014. [Google Scholar]
- Sawadogo, W.; Boly, R.; Cerella, C.; Teiten, M.; Dicato, M.; Diederich, M. A survey of marine natural compounds and their derivatives with anti-cancer activity reported in 2012. Molecules 2015, 20, 7097–7142. [Google Scholar] [CrossRef]
- Butler, M.S. The role of natural product chemistry in drug discovery. J. Nat. Prod. 2004, 67, 2141–2153. [Google Scholar] [CrossRef]
- Newman, D.J.; Cragg, G.M.; Snader, K.M. Natural products as sources of new drugs over the period 1981–2002. J. Nat. Prod. 2003, 66, 1022–1037. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs over the 30 years from 1981 to 2010. J. Nat. Prod. 2012, 75, 311–335. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs from 1981 to 2014. J. Nat. Prod. 2016, 79, 629–661. [Google Scholar] [CrossRef] [PubMed]
- White, J. Drug Addiction: From Basic Research to Therapy. Drug Alcohol Rev. 2009, 28, 455. [Google Scholar] [CrossRef]
- Nobili, S.; Lippi, D.; Witort, E.; Donnini, M.; Bausi, L.; Mini, E.; Capaccioli, S. Natural compounds for cancer treatment and prevention. Pharmacol. Res. 2009, 59, 365–378. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.; Cragg, G. Marine-sourced anti-cancer and cancer pain control agents in clinical and late preclinical development. Mar. Drugs 2014, 12, 255–278. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Gordon, M.C. Drugs and drug candidates from marine sources: An assessment of the current “state of play”. Planta Med. 2016, 82, 775–789. [Google Scholar] [CrossRef] [PubMed]
- Bhanot, A.; Sharma, R.; Noolvi, M.N. Natural sources as potential anti-cancer agents: A review. Int. J. Phytomedicine 2011, 3, 9–26. [Google Scholar]
- Dyshlovoy, S.A.; Honecker, F. Marine compounds and cancer: 2017 updates. Mar. Drugs 2018, 16, 41. [Google Scholar] [CrossRef] [PubMed]
- Drug Bank. Available online: http://www.drugbank.ca (accessed on 17 August 2019).
- Walsh, V.; Goodman, J. Cancer chemotherapy, biodiversity, public and private property: The case of the anti-cancer drug Taxol. Soc. Sci. Med. 1999, 49, 1215–1225. [Google Scholar] [CrossRef]
- Mugera, G.M. Useful Drugs and Cancer Causing Chemicals in Kenya Medical and Toxic Plants; Department of Veterinary Pathology and Microbiology: Nairobi, Kenya, 1977. [Google Scholar]
- Yousaf, M.; Hammond, N.L.; Peng, J.; Wahyuono, S.; McIntosh, K.A.; Charman, W.N.; Mayer, A.M.S.; Hamann, M.T. New manzamine alkaloids from an Indo-Pacific sponge. Pharmacokinetics, oral availability, and the significant activity of several manzamines against HIV-I, AIDS opportunistic infections, and inflammatory diseases. J. Med. Chem. 2004, 47, 3512–3517. [Google Scholar] [CrossRef] [PubMed]
- Choudhary, A.; Naughton, L.; Montánchez, I.; Dobson, A.; Rai, D. Current status and future prospects of marine natural products (MNPs) as antimicrobials. Mar. Drugs 2017, 15, 272. [Google Scholar] [CrossRef] [PubMed]
- Pereira, F.; Aires-de-Sousa, J. Computational methodologies in the exploration of marine natural product leads. Mar. Drugs 2018, 16, 236. [Google Scholar] [CrossRef] [PubMed]
- Malakoff, D. Extinction on the high seas. Science 1997, 227, 486–488. [Google Scholar] [CrossRef]
- Boeuf, G. Marine biodiversity characteristics. Comptes Rendus Biol. 2011, 334, 435–440. [Google Scholar] [CrossRef] [PubMed]
- Whitman, W.B. The unseen majority. Proc. Natl. Acad. Sci. USA 1998, 74, 5088–5090. [Google Scholar]
- Rocha-Martin, J.; Harrington, C.; Dobson, A.; O’Gara, F. Emerging strategies and integrated systems microbiology technologies for biodiscovery of marine bioactive compounds. Mar. Drugs 2014, 12, 3516–3559. [Google Scholar] [CrossRef]
- Sithranga Boopathy, N.; Kathiresan, K. Anticancer drugs from marine flora: An overview. J. Oncol. 2010, 2010, 18. [Google Scholar] [CrossRef]
- Ramirez-Llodra, E.; Brandt, A.; Danovaro, R.; De Mol, B.; Escobar, E.; German, C.; Levin, L.; Arbizu, P.; Menot, L.; Buhl-Mortensen, P. Deep, diverse and definitely different: Unique attributes of the world’s largest ecosystem. Biogeosciences 2010, 7, 2851–2899. [Google Scholar] [CrossRef]
- Bergmann, W.; Feeney, R.J. The isolation of a new thymine pentoside from sponges1. J. Am. Chem. Soc. 1950, 72, 2809–2810. [Google Scholar] [CrossRef]
- Cragg, G.M.; Newman, D.J.; Weiss, R.B. Coral reefs, forests, and thermal vents: The worldwide exploration of nature for novel antitumor agents. Semin. Oncol. 1997, 24, 156–163. [Google Scholar] [PubMed]
- Bergmann, W.; Feeney, R.J. Nucleosides of sponges: Discovery of the arabinosebased nucleotides—Tethya crypta. J. Org. Chem 1951, 16, 981–987. [Google Scholar] [CrossRef]
- Bergmann, W.; Feeney, R.J. Contributions to the study of marine products. XXXIX. The nucleosides of sponges. III. 1 Spongothymidine and spongouridine2. J. Org. Chem. 1955, 20, 1501–1507. [Google Scholar] [CrossRef]
- Rinehart, K.L. Antitumor compounds from tunicates. Med. Res. Rev. 2000, 20, 27. [Google Scholar] [CrossRef]
- Schwartsmann, G.; Brondani, A.; Berlinck, R.G.S.; Jimeno, J. Marine organisms and other novel natural sources of new cancer drugs. Ann. Oncol. 2000, 11, 235–243. [Google Scholar] [CrossRef] [PubMed]
- Fsulkner, D.J. Highlights of marine natural products chemistry (1972–1999). Nat. Prod. Rep. 2000, 17, 1–6. [Google Scholar] [CrossRef]
- Faulkner, D.J. Marine pharmacology. Antonie Van Leeuwenhoek 2000, 77, 135–145. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Fernandez, L.F.; Reyes, F.; Sanchez-Puelles, J.M. The marine pharmacy: New antitumoral compounds from the sea. Pharm. News 2002, 9, 495–502. [Google Scholar]
- Haefner, B. Drugs from the deep: Marine natural products as drug candidates. Drug Discov. Today 2003, 8, 536–544. [Google Scholar] [CrossRef]
- Laatsch, H. AntiBase 2011: The Natural Compound Identifier; Wiley-Vch: Weinheim, Germany, 2011; Volume 313, ISBN 3527338411. [Google Scholar]
- Newman, D.J.; Cragg, G.M. Advanced preclinical and clinical trials of natural products and related compounds from marine sources. Curr. Med. Chem. 2004, 11, 1693–1713. [Google Scholar] [CrossRef]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs over the last 25 years. J. Nat. Prod. 2007, 70, 461–477. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.; Kim, Y.; Kim, J.; Nam, B.-H.; Kim, D.-G.; An, C.; Lee, J.; Kim, P.; Lee, H.; Oh, J.-S. Liquid chromatography-mass spectrometry-based rapid secondary-metabolite profiling of marine Pseudoalteromonas sp. M2. Mar. Drugs 2016, 14, 24. [Google Scholar] [CrossRef] [PubMed]
- Fries, E.; Dekiff, J.H.; Willmeyer, J.; Nuelle, M.-T.; Ebert, M.; Remy, D. Identification of polymer types and additives in marine microplastic particles using pyrolysis-GC/MS and scanning electron microscopy. Environ. Sci. Process. Impacts 2013, 15, 1949–1956. [Google Scholar] [CrossRef] [PubMed]
- Krock, B.; Busch, J.; Tillmann, U.; García-Camacho, F.; Sánchez-Mirón, A.; Gallardo-Rodríguez, J.; López-Rosales, L.; Andree, K.; Fernández-Tejedor, M.; Witt, M. LC-MS/MS detection of karlotoxins reveals new variants in strains of the marine dinoflagellate Karlodinium veneficum from the Ebro Delta (NW Mediterranean). Mar. Drugs 2017, 15, 391. [Google Scholar] [CrossRef] [PubMed]
- Bose, U.; Hewavitharana, A.; Ng, Y.; Shaw, P.; Fuerst, J.; Hodson, M. LC-MS-Based metabolomics study of marine bacterial secondary metabolite and antibiotic production in Salinispora arenicola. Mar. Drugs 2015, 13, 249–266. [Google Scholar] [CrossRef] [PubMed]
- Panagiotopoulos, C.; Repeta, D.J.; Mathieu, L.; Rontani, J.-F.; Sempere, R. Molecular level characterization of methyl sugars in marine high molecular weight dissolved organic matter. Mar. Chem. 2013, 154, 34–45. [Google Scholar] [CrossRef]
- Freitas, S.; Martins, R.; Costa, M.; Leão, P.; Vitorino, R.; Vasconcelos, V.; Urbatzka, R. Hierridin B isolated from a marine cyanobacterium alters VDAC1, mitochondrial activity, and cell cycle genes on HT-29 colon adenocarcinoma cells. Mar. Drugs 2016, 14, 158. [Google Scholar] [CrossRef] [PubMed]
- Garson, M.; Hehre, W.; Pierens, G. Revision of the structure of acremine P from a marine-derived strain of Acremonium persicinum. Molecules 2017, 22, 521. [Google Scholar] [CrossRef] [PubMed]
- Macintyre, L.; Zhang, T.; Viegelmann, C.; Martinez, I.; Cheng, C.; Dowdells, C.; Abdelmohsen, U.; Gernert, C.; Hentschel, U.; Edrada-Ebel, R. Metabolomic tools for secondary metabolite discovery from marine microbial symbionts. Mar. Drugs 2014, 12, 3416–3448. [Google Scholar] [CrossRef]
- Pérez-Victoria, I.; Martín, J.; Reyes, F. Combined LC/UV/MS and NMR strategies for the dereplication of marine natural products. Planta Med. 2016, 82, 857–871. [Google Scholar] [CrossRef]
- Viegelmann, C.; Margassery, L.; Kennedy, J.; Zhang, T.; O’Brien, C.; O’Gara, F.; Morrissey, J.; Dobson, A.; Edrada-Ebel, R. Metabolomic profiling and genomic study of a marine sponge-associated Streptomyces sp. Mar. Drugs 2014, 12, 3323–3351. [Google Scholar] [CrossRef] [PubMed]
- Morvan, D. Functional metabolomics uncovers metabolic alterations associated to severe oxidative stress in MCF7 breast cancer cells exposed to ascididemin. Mar. Drugs 2013, 11, 3846–3860. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Leung, R.K.-K.; Guo, M.; Tuo, L.; Guo, L.; Yew, W.W.; Lou, I.; Lee, S.M.Y.; Sun, C. Genome-guided investigation of antibiotic substances produced by Allosalinactinospora lopnorensis CA15-2 T from Lop Nor region, China. Sci. Rep. 2016, 6, 20667. [Google Scholar] [CrossRef] [PubMed]
- Bourhill, T.; Narendran, A.; Johnston, R.N. Enzastaurin: A lesson in drug development. Crit. Rev. Oncol. Hematol. 2017, 112, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.-Q.; Miao, Z.-H. Marine-derived angiogenesis inhibitors for cancer therapy. Mar. Drugs 2013, 11, 903–933. [Google Scholar] [CrossRef]
- Ritchie, J.W.A.; Williams, R.J. Cancer research UK centre for drug development: Translating 21st-century science into the cancer medicines of tomorrow. Drug Discov. Today 2015, 20, 995–1003. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, I.; Al-Katib, A.M.; Beck, F.W.J.; Mohammad, R.M. Sequential treatment of a resistant chronic lymphocytic leukemia patient with bryostatin 1 followed by 2-chlorodeoxyadenosine: Case report. Clin. Cancer Res. 2000, 6, 1328–1332. [Google Scholar]
- Garcia-carbonero, R.; Harmon, D.; Seiden, M.; Jimeno, J.; Merriam, P.; Waxman, A.; Supko, J.; Quigley, M.T.; Ryan, D. Ecteinascidin-743 (et-743) induces objective responses and disease control in patients with advanced non-osseous sarcomas: Results from phase Ii trials. Ann. Oncol. 2000, 11, 126. [Google Scholar]
- Erba, E.; Bergamaschi, D.; Bassano, L.; Damia, G.; Ronzoni, S.; Faircloth, G.T.; d’Incalci, M. Ecteinascidin-743 (ET-743), a natural marine compound, with a unique mechanism of action. Eur. J. Cancer 2001, 37, 97–105. [Google Scholar] [CrossRef]
- Poncet, J. The dolastatins, a family of promising antineoplastic agents. Curr. Pharm. Des. 1999, 5, 139–162. [Google Scholar]
- Pathak, S.; Multani, A.S.; Ozen, M.; Richardson, M.A.; Newman, R.A. Dolastatin-10 induces polyploidy, telomeric associations and apoptosis in a murine melanoma cell line. Oncol. Rep. 1998, 5, 373–379. [Google Scholar] [CrossRef] [PubMed]
- Bai, R.; Petit, G.R.; Hamel, E. Dolastatin 10, a powerful cytostatic peptide derived from a marine animal: Inhibition of tubulin polymerization mediated through the vinca alkaloid binding domain. Biochem. Pharmacol. 1990, 39, 1941–1949. [Google Scholar] [CrossRef]
- Dring, M.J.; Dring, M.J.; Dring, M.H. The Biology of Marine Plants; Cambridge University Press: Cambridge, UK, 1991; ISBN 0521427657. [Google Scholar]
- Orlikova, B.; Legrand, N.; Panning, J.; Dicato, M.; Diederich, M. Anti-inflammatory and anticancer drugs from nature. In Advances in Nutrition and Cancer; Springer: Berlin/Heidelberg, Germany, 2014; pp. 123–143. [Google Scholar]
- Bhosale, S.H.; Nagle, V.L.; Jagtap, T.G. Antifouling potential of some marine organisms from India against species of Bacillus and Pseudomonas. Mar. Biotechnol. 2002, 4, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Alghazwi, M.; Kan, Y.Q.; Zhang, W.; Gai, W.P.; Garson, M.J.; Smid, S. Neuroprotective activities of natural products from marine macroalgae during 1999–2015. J. Appl. Phycol. 2016, 28, 3599–3616. [Google Scholar] [CrossRef]
- Ganesan, P.; Kumar, C.S.; Bhaskar, N. Antioxidant properties of methanol extract and its solvent fractions obtained from selected Indian red seaweeds. Bioresour. Technol. 2008, 99, 2717–2723. [Google Scholar] [CrossRef] [PubMed]
- Carmichael, W.W. Cyanobacteria secondary metabolites—the cyanotoxins. J. Appl. Bacteriol. 1992, 72, 445–459. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, E.A.; Aly, H.F.; Baker, D.A.; Mahmoud, K.; El-Baz, F.K. Marine algal sterol hydrocarbon with anti-inflammatory, anticancer and anti-oxidant properties. Int. J. Pharma Bio Sci. 2016, 7, 392–398. [Google Scholar]
- Bandaranayake, W.M. Bioactivities, bioactive compounds and chemical constituents of mangrove plants. Wetl. Ecol. Manag. 2002, 10, 421–452. [Google Scholar] [CrossRef]
- Mans, D.R.A.; Da Rocha, A.B.; Schwartsmann, G. Anti-cancer drug discovery and development in Brazil: Targeted plant collection as a rational strategy to acquire candidate anti-cancer compounds. Oncologist 2000, 5, 185–198. [Google Scholar] [CrossRef]
- Lavakumar, V.; Ahamed, K.F.H.; Ravichandran, V. Anticancer and antioxidant effect of Acanthophora spicifera against EAC induced carcinoma in mice. J. Pharm. Res. 2012, 5, 1503–1507. [Google Scholar]
- Zhuang, C.; Itoh, H.; Mizuno, T.; Ito, H. Antitumor active fucoidan from the brown seaweed, umitoranoo (Sargassum thunbergii). Biosci. Biotechnol. Biochem. 1995, 59, 563–567. [Google Scholar] [CrossRef] [PubMed]
- Vischer, P.; Buddecke, E. Different action of heparin and fucoidan on arterial smooth muscle cell proliferation and thrombospondin and fibronectin metabolism. Eur. J. Cell Biol. 1991, 56, 407–414. [Google Scholar] [PubMed]
- Barbier, P.; Guise, S.; Huitorel, P.; Amade, P.; Pesando, D.; Briand, C.; Peyrot, V. Caulerpenyne from Caulerpa taxifolia has an antiproliferative activity on tumor cell line SK-N-SH and modifies the microtubule network. Life Sci. 2001, 70, 415–429. [Google Scholar] [CrossRef]
- Palermo, J.A.; Flower, P.B.; Seldes, A.M. Chondriamides A and B, new indolic metabolites from the red alga Chondria sp. Tetrahedron Lett. 1992, 33, 3097–3100. [Google Scholar] [CrossRef]
- Parent-Massin, D. Evaluation of the toxicological risk to humans of caulerpenyne using human hematopoietic progenitors, melanocytes, and keratinocytes in culture. J. Toxicol. Environ. Heal. Part A 1996, 47, 47–59. [Google Scholar] [CrossRef] [PubMed]
- Urones, J.G.; Araujo, M.E.M.; Palma, F.M.S.B.; Basabe, P.; Marcos, I.S.; Moro, R.F.; Lithgow, A.M.; Pineda, J. Meroterpenes from Cystoseira usneoides II. Phytochemistry 1992, 31, 2105–2109. [Google Scholar] [CrossRef]
- Fischel, J.L.; Lemee, R.; Formento, P.; Caldani, C.; Moll, J.L.; Pesando, D.; Meinesz, A.; Grelier, P.; Pietra, P.; Guerriero, A. Cell growth inhibitory effects of caulerpenyne, a sesquiterpenoid from the marine algae Caulerpa taxifolia. Anticancer Res. 1995, 15, 2155–2160. [Google Scholar] [PubMed]
- Athukorala, Y.; Jung, W.-K.; Vasanthan, T.; Jeon, Y.-J. An anticoagulative polysaccharide from an enzymatic hydrolysate of Ecklonia cava. Carbohydr. Polym. 2006, 66, 184–191. [Google Scholar] [CrossRef]
- Gerwick, W.H.; Fenical, W. Ichthyotoxic and cytotoxic metabolites of the tropical brown alga Stypopodium zonale (Lamouroux) Papenfuss. J. Org. Chem. 1981, 46, 22–27. [Google Scholar] [CrossRef]
- Encarnação, T.; Pais, A.A.; Campos, M.G.; Burrows, H.D. Cyanobacteria and microalgae: A renewable source of bioactive compounds and other chemicals. Sci. Prog. 2015, 98, 145–168. [Google Scholar] [CrossRef]
- Bachvaroff, T.R.; Adolf, J.E.; Squier, A.H.; Harvey, H.R.; Place, A.R. Characterization and quantification of karlotoxins by liquid chromatography–mass spectrometry. Harmful Algae 2008, 7, 473–484. [Google Scholar] [CrossRef]
- Tan, L.T. Bioactive natural products from marine cyanobacteria for drug discovery. Phytochemistry 2007, 68, 954–979. [Google Scholar] [CrossRef] [PubMed]
- Rickards, R.W.; Rothschild, J.M.; Willis, A.C.; de Chazal, N.M.; Kirk, J.; Kirk, K.; Saliba, K.J.; Smith, G.D. Calothrixins A and B, novel pentacyclic metabolites from Calothrix cyanobacteria with potent activity against malaria parasites and human cancer cells. Tetrahedron 1999, 55, 13513–13520. [Google Scholar] [CrossRef]
- Schmidt, E.W.; Nelson, J.T.; Rasko, D.A.; Sudek, S.; Eisen, J.A.; Haygood, M.G.; Ravel, J. Patellamide A and C biosynthesis by a microcin-like pathway in Prochloron didemni, the cyanobacterial symbiont of Lissoclinum patella. Proc. Natl. Acad. Sci. USA 2005, 102, 7315–7320. [Google Scholar] [CrossRef] [PubMed]
- Donia, M.S.; Hathaway, B.J.; Sudek, S.; Haygood, M.G.; Rosovitz, M.J.; Ravel, J.; Schmidt, E.W. Natural combinatorial peptide libraries in cyanobacterial symbionts of marine ascidians. Nat. Chem. Biol. 2006, 2, 729. [Google Scholar] [CrossRef]
- Donia, M.S.; Ravel, J.; Schmidt, E.W. A global assembly line for cyanobactins. Nat. Chem. Biol. 2008, 4, 341. [Google Scholar] [CrossRef] [PubMed]
- Williams, D.E.; Moore, R.E.; Paul, V.J. The structure of ulithiacyclamide B. Antitumor evaluation of cyclic peptides and macrolides from Lissoclinum patella. J. Nat. Prod. 1989, 52, 732–739. [Google Scholar] [CrossRef] [PubMed]
- Banker, R.; Carmeli, S. Tenuecyclamides A–D, Cyclic Hexapeptides from the Cyanobacterium Nostoc s pongiaeforme var. t enue. J. Nat. Prod. 1998, 61, 1248–1251. [Google Scholar] [CrossRef]
- Davidson, B.S. New dimensions in natural products research: Cultured marine microorganisms. Curr. Opin. Biotechnol. 1995, 6, 284–291. [Google Scholar] [CrossRef]
- Moore, R.E. Cyclic peptides and depsipeptides from cyanobacteria: A review. J. Ind. Microbiol. 1996, 16, 134–143. [Google Scholar] [CrossRef]
- Trimurtulu, G.; Ohtani, I.; Patterson, G.M.L.; Moore, R.E.; Corbett, T.H.; Valeriote, F.A.; Demchik, L. Total structures of cryptophycins, potent antitumor depsipeptides from the blue-green alga Nostoc sp. strain GSV 224. J. Am. Chem. Soc. 1994, 116, 4729–4737. [Google Scholar] [CrossRef]
- Taori, K.; Paul, V.J.; Luesch, H. Structure and activity of largazole, a potent antiproliferative agent from the Floridian marine cyanobacterium Symploca sp. J. Am. Chem. Soc. 2008, 130, 1806–1807. [Google Scholar] [CrossRef]
- Luesch, H.; Moore, R.E.; Paul, V.J.; Mooberry, S.L.; Corbett, T.H. Isolation of dolastatin 10 from the marine cyanobacterium Symploca species VP642 and total stereochemistry and biological evaluation of its analogue symplostatin 1. J. Nat. Prod. 2001, 64, 907–910. [Google Scholar] [CrossRef]
- Medina, R.A.; Goeger, D.E.; Hills, P.; Mooberry, S.L.; Huang, N.; Romero, L.I.; Ortega-Barría, E.; Gerwick, W.H.; McPhail, K.L. Coibamide A, a potent antiproliferative cyclic depsipeptide from the Panamanian marine cyanobacterium Leptolyngbya sp. J. Am. Chem. Soc. 2008, 130, 6324–6325. [Google Scholar] [CrossRef]
- Bhatnagar, I.; Kim, S.-K. Immense essence of excellence: Marine microbial bioactive compounds. Mar. Drugs 2010, 8, 2673–2701. [Google Scholar] [CrossRef]
- Stevenson, C.S.; Capper, E.A.; Roshak, A.K.; Marquez, B.; Grace, K.; Gerwick, W.H.; Jacobs, R.S.; Marshall, L.A. Scytonemin-a marine natural product inhibitor of kinases key in hyperproliferative inflammatory diseases. Inflamm. Res. 2002, 51, 112–114. [Google Scholar] [CrossRef]
- Stevenson, C.S.; Capper, E.A.; Roshak, A.K.; Marquez, B.; Eichman, C.; Jackson, J.R.; Mattern, M.; Gerwick, W.H.; Jacobs, R.S.; Marshall, L.A. The identification and characterization of the marine natural product scytonemin as a novel antiproliferative pharmacophore. J. Pharmacol. Exp. Ther. 2002, 303, 858–866. [Google Scholar] [CrossRef]
- Carte, B.K. Biomedical potential of marine natural products. Bioscience 1996, 46, 271–286. [Google Scholar]
- Gerwick, W.H.; Proteau, P.J.; Nagle, D.G.; Hamel, E.; Blokhin, A.; Slate, D.L. Structure of curacin A, a novel antimitotic, antiproliferative and brine shrimp toxic natural product from the marine cyanobacterium Lyngbya majuscula. J. Org. Chem. 1994, 59, 1243–1245. [Google Scholar] [CrossRef]
- Javed, F.; Qadir, M.I.; Janbaz, K.H.; Ali, M. Novel drugs from marine microorganisms. Crit. Rev. Microbiol. 2011, 37, 245–249. [Google Scholar] [CrossRef]
- Peng, J.; Place, A.R.; Yoshida, W.; Anklin, C.; Hamann, M.T. Structure and absolute configuration of karlotoxin-2, an ichthyotoxin from the marine dinoflagellate Karlodinium veneficum. J. Am. Chem. Soc. 2010, 132, 3277–3279. [Google Scholar] [CrossRef] [PubMed]
- Romanenko, L.A.; Uchino, M.; Kalinovskaya, N.I.; Mikhailov, V. V Isolation, phylogenetic analysis and screening of marine mollusc-associated bacteria for antimicrobial, hemolytic and surface activities. Microbiol. Res. 2008, 163, 633–644. [Google Scholar] [CrossRef] [PubMed]
- Isnansetyo, A.; Kamei, Y. Bioactive substances produced by marine isolates of Pseudomonas. J. Ind. Microbiol. Biotechnol. 2009, 36, 1239–1248. [Google Scholar] [CrossRef] [PubMed]
- Malaker, A.; Ahmad, S.A.I. Therapeutic potency of anticancer peptides derived from marine organism. Int. J. Eng. 2013, 2, 2305–8269. [Google Scholar]
- Goldin, B.; Gorbach, S.L. Alterations in fecal microflora enzymes related to diet, age, lactobacillus supplements, and dimethylhydrazine. Cancer 1977, 40, 2421–2426. [Google Scholar] [CrossRef]
- Mitall, B.K.; Garg, S.K. Anticarcinogenic, hypocholesterolemic, and antagonistic activities of Lactobacillus acidophilus. Crit. Rev. Microbiol. 1995, 21, 175–214. [Google Scholar] [CrossRef]
- Wollowski, I.; Rechkemmer, G.; Pool-Zobel, B.L. Protective role of probiotics and prebiotics in colon cancer. Am. J. Clin. Nutr. 2001, 73, 451s–455s. [Google Scholar] [CrossRef]
- Bitzer, J.; Große, T.; Wang, L.; Lang, S.; Beil, W.; Zeeck, A. New aminophenoxazinones from a marine Halomonas sp.: Fermentation, structure elucidation, and biological activity. J. Antibiot. (Tokyo) 2006, 59, 86. [Google Scholar] [CrossRef]
- Sagar, S.; Esau, L.; Holtermann, K.; Hikmawan, T.; Zhang, G.; Stingl, U.; Bajic, V.B.; Kaur, M. Induction of apoptosis in cancer cell lines by the Red Sea brine pool bacterial extracts. BMC Complement. Altern. Med. 2013, 13, 344. [Google Scholar] [CrossRef]
- Ruiz-Ruiz, C.; Srivastava, G.K.; Carranza, D.; Mata, J.A.; Llamas, I.; Santamaría, M.; Quesada, E.; Molina, I.J. An exopolysaccharide produced by the novel halophilic bacterium Halomonas stenophila strain B100 selectively induces apoptosis in human T leukaemia cells. Appl. Microbiol. Biotechnol. 2011, 89, 345–355. [Google Scholar] [CrossRef]
- Erba, E.; Bergamaschi, D.; Ronzoni, S.; Faretta, M.; Taverna, S.; Bonfanti, M.; Catapano, C. V; Faircloth, G.; Jimeno, J.; D’incalci, M. Mode of action of thiocoraline, a natural marine compound with anti-tumor activity. Br. J. Cancer 1999, 80, 971. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Groβe, T.; Stevens, H.; Brinkhoff, T.; Simon, M.; Liang, L.; Bitzer, J.; Bach, G.; Zeeck, A.; Tokuda, H. Bioactive hydroxyphenylpyrrole-dicarboxylic acids from a new marine Halomonas sp.: Production and structure elucidation. Appl. Microbiol. Biotechnol. 2006, 72, 816–822. [Google Scholar] [CrossRef] [PubMed]
- Heald, S.C.; Brandão, P.F.B.; Hardicre, R.; Bull, A.T. Physiology, biochemistry and taxonomy of deep-sea nitrile metabolising Rhodococcus strains. Antonie Van Leeuwenhoek 2001, 80, 169–183. [Google Scholar] [CrossRef] [PubMed]
- Moran, M.A.; Rutherford, L.T.; Hodson, R.E. Evidence for indigenous Streptomyces populations in a marine environment determined with a 16S rRNA probe. Appl. Environ. Microbiol. 1995, 61, 3695–3700. [Google Scholar] [PubMed]
- Jensen, P.R.; Mincer, T.J.; Williams, P.G.; Fenical, W. Marine actinomycete diversity and natural product discovery. Antonie Van Leeuwenhoek 2005, 87, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Maldonado, L.A.; Fenical, W.; Jensen, P.R.; Kauffman, C.A.; Mincer, T.J.; Ward, A.C.; Bull, A.T.; Goodfellow, M. Salinispora arenicola gen. nov., sp. nov. and Salinispora tropica sp. nov., obligate marine actinomycetes belonging to the family Micromonosporaceae. Int. J. Syst. Evol. Microbiol. 2005, 55, 1759–1766. [Google Scholar] [CrossRef] [PubMed]
- Mincer, T.J.; Fenical, W.; Jensen, P.R. Culture-dependent and culture-independent diversity within the obligate marine actinomycete genus Salinispora. Appl. Environ. Microbiol. 2005, 71, 7019–7028. [Google Scholar] [CrossRef]
- Genilloud, O. Actinomycetes: Still a source of novel antibiotics. Nat. Prod. Rep. 2017, 34, 1203–1232. [Google Scholar] [CrossRef]
- Olano, C.; Méndez, C.; Salas, J. Antitumor compounds from marine actinomycetes. Mar. Drugs 2009, 7, 210–248. [Google Scholar] [CrossRef]
- Berdy, J. Bioactive microbial metabolites. J. Antibiot. (Tokyo) 2005, 58, 1. [Google Scholar] [CrossRef]
- Hong, K.; Gao, A.-H.; Xie, Q.-Y.; Gao, H.G.; Zhuang, L.; Lin, H.-P.; Yu, H.-P.; Li, J.; Yao, X.-S.; Goodfellow, M. Actinomycetes for marine drug discovery isolated from mangrove soils and plants in China. Mar. Drugs 2009, 7, 24–44. [Google Scholar] [CrossRef] [PubMed]
- Lam, K.S. Discovery of novel metabolites from marine actinomycetes. Curr. Opin. Microbiol. 2006, 9, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Cho, J.Y.; Williams, P.G.; Kwon, H.C.; Jensen, P.R.; Fenical, W. Lucentamycins A–D, cytotoxic peptides from the marine-derived actinomycete Nocardiopsis lucentensis. J. Nat. Prod. 2007, 70, 1321–1328. [Google Scholar] [CrossRef] [PubMed]
- Maskey, R.P.; Helmke, E.; Kayser, O.; Fiebig, H.H.; Maier, A.; Busche, A.; Laatsch, H. Anti-cancer and antibacterial trioxacarcins with high anti-malaria activity from a marine streptomycete and their absolute stereochemistry. J. Antibiot. (Tokyo) 2004, 57, 771–779. [Google Scholar] [CrossRef]
- Hawas, U.W.; Shaaban, M.; Shaaban, K.A.; Speitling, M.; Maier, A.; Kelter, G.; Fiebig, H.H.; Meiners, M.; Helmke, E.; Laatsch, H. Mansouramycins A–D, cytotoxic isoquinolinequinones from a marine Streptomycete. J. Nat. Prod. 2009, 72, 2120–2124. [Google Scholar] [CrossRef]
- Pérez, M.; Crespo, C.; Schleissner, C.; Rodríguez, P.; Zúñiga, P.; Reyes, F. Tartrolon D, a cytotoxic macrodiolide from the marine-derived actinomycete Streptomyces sp. MDG-04-17-069. J. Nat. Prod. 2009, 72, 2192–2194. [Google Scholar] [CrossRef]
- Chauhan, D.; Catley, L.; Li, G.; Podar, K.; Hideshima, T.; Velankar, M.; Mitsiades, C.; Mitsiades, N.; Yasui, H.; Letai, A. A novel orally active proteasome inhibitor induces apoptosis in multiple myeloma cells with mechanisms distinct from Bortezomib. Cancer Cell 2005, 8, 407–419. [Google Scholar] [CrossRef]
- Fenical, W.; Jensen, P.R.; Palladino, M.A.; Lam, K.S.; Lloyd, G.K.; Potts, B.C. Discovery and development of the anticancer agent salinosporamide A (NPI-0052). Bioorg. Med. Chem. 2009, 17, 2175–2180. [Google Scholar] [CrossRef]
- Newman, D.J.; Hill, R.T. New drugs from marine microbes: The tide is turning. J. Ind. Microbiol. Biotechnol. 2006, 33, 539–544. [Google Scholar] [CrossRef]
- Bhadury, P.; Mohammad, B.T.; Wright, P.C. The current status of natural products from marine fungi and their potential as anti-infective agents. J. Ind. Microbiol. Biotechnol. 2006, 33, 325. [Google Scholar] [CrossRef]
- Guerriero, A.; D’Ambrosio, M.; Cuomo, V.; Pietra, F. A novel, degraded polyketidic lactone, leptosphaerolide, and its likely diketone precursor, leptosphaerodione. Isolation from cultures of the marine ascomycete Leptosphaeria oraemaris (Linder). Helv. Chim. Acta 1991, 74, 1445–1450. [Google Scholar] [CrossRef]
- Pallenberg, A.J.; White, J.D. The synthesis and absolute configuration of (+)-leptosphaerin. Tetrahedron Lett. 1986, 27, 5591–5594. [Google Scholar] [CrossRef]
- Schiehser, G.A.; White, J.D.; Matsumoto, G.; Pezzanite, J.O.; Clardy, J. The structure of leptosphaerin. Tetrahedron Lett. 1986, 27, 5587–5590. [Google Scholar] [CrossRef]
- Abdel-Lateff, A.; König, G.M.; Fisch, K.M.; Höller, U.; Jones, P.G.; Wright, A.D. New antioxidant hydroquinone derivatives from the algicolous marine fungus Acremonium sp. J. Nat. Prod. 2002, 65, 1605–1611. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Lateff, A.; Klemke, C.; König, G.M.; Wright, A.D. Two new xanthone derivatives from the algicolous marine fungus Wardomyces anomalus. J. Nat. Prod. 2003, 66, 706–708. [Google Scholar] [CrossRef] [PubMed]
- Suja, M.; Vasuki, S.; Sajitha, N. Anticancer activity of compounds isolated from marine endophytic fungus Aspergillus terreus. World J. Pharm. Pharm. Sci. 2014, 3, 661–672. [Google Scholar]
- Tan, D.S.P.; Marchio, C.; Reis-Filho, J.S. Hereditary breast cancer: From molecular pathology to tailored therapies. J. Clin. Pathol. 2008, 61, 1073–1082. [Google Scholar] [CrossRef] [PubMed]
- Du, L.; Zhu, T.; Fang, Y.; Liu, H.; Gu, Q.; Zhu, W. Aspergiolide A, a novel anthraquinone derivative with naphtho [1, 2, 3-de] chromene-2, 7-dione skeleton isolated from a marine-derived fungus Aspergillus glaucus. Tetrahedron 2007, 63, 1085–1088. [Google Scholar] [CrossRef]
- Du, L.; Feng, T.; Zhao, B.; Li, D.; Cai, S.; Zhu, T.; Wang, F.; Xiao, X.; Gu, Q. Alkaloids from a deep ocean sediment-derived fungus Penicillium sp. and their antitumor activities. J. Antibiot. (Tokyo) 2010, 63, 165. [Google Scholar] [CrossRef]
- Zhang, J.; Tao, L.; Liang, Y.; Chen, L.; Mi, Y.; Zheng, L.; Wang, F.; She, Z.; Lin, Y.; To, K.K.W. Anthracenedione derivatives as anticancer agents isolated from secondary metabolites of the mangrove endophytic fungi. Mar. Drugs 2010, 8, 1469–1481. [Google Scholar] [CrossRef]
- Han, B.-N.; Hong, L.-L.; Gu, B.-B.; Sun, Y.-T.; Wang, J.; Liu, J.-T.; Lin, H.-W. Natural Products from Sponges. In Symbiotic Microbiomes of Coral Reefs Sponges and Corals; Springer: Dordrecht, The Netherlands, 2019; pp. 329–463. [Google Scholar]
- Newman, D.J.; Cragg, G.M.; Snader, K.M. The influence of natural products upon drug discovery. Nat. Prod. Rep. 2000, 17, 215–234. [Google Scholar] [CrossRef] [PubMed]
- Hirata, Y.; Uemura, D. Halichondrins-antitumor polyether macrolides from a marine sponge. Pure Appl. Chem. 1986, 58, 701–710. [Google Scholar] [CrossRef]
- Zovko, A.; Novak, M.; Hååg, P.; Kovalerchick, D.; Holmlund, T.; Färnegårdh, K.; Ilan, M.; Carmeli, S.; Lewensohn, R.; Viktorsson, K. Compounds from the marine sponge Cribrochalina vasculum offer a way to target IGF-1R mediated signaling in tumor cells. Oncotarget 2016, 7, 50258. [Google Scholar] [CrossRef] [PubMed]
- Cortes, J.; O’Shaughnessy, J.; Loesch, D.; Blum, J.L.; Vahdat, L.T.; Petrakova, K.; Chollet, P.; Manikas, A.; Diéras, V.; Delozier, T. Eribulin monotherapy versus treatment of physician’s choice in patients with metastatic breast cancer (EMBRACE): A phase 3 open-label randomised study. Lancet 2011, 377, 914–923. [Google Scholar] [CrossRef]
- Ei-Seedi, H.R.; Gomaa, M.; Salem, M.M.; Benchoula, K.; Keshk, H.M.; Yosri, N.; Ayesh, A.; Asker, A.M.; Soliman, K.; Hamza, Z. Cytotoxic effects of the Red Sea soft coral Sarcophyton Trocheliophorum. Acta Pol. Pharm. 2016, 73, 1587–1592. [Google Scholar]
- Gomaa, M.N.; Soliman, K.; Ayesh, A.; Abd El-Wahed, A.; Hamza, Z.; Mansour, H.M.; Khalifa, S.A.M.; Mohd Ali, H.B.; El-Seedi, H.R. Antibacterial effect of the red sea soft coral Sarcophyton trocheliophorum. Nat. Prod. Res. 2016, 30, 729–734. [Google Scholar] [CrossRef]
- Gross, H.; Kehraus, S.; Nett, M.; König, G.M.; Beil, W.; Wright, A.D. New cytotoxic cembrane based diterpenes from the soft corals Sarcophyton cherbonnieri and Nephthea sp. Org. Biomol. Chem. 2003, 1, 944–949. [Google Scholar] [CrossRef]
- Chao, C.-H.; Wen, Z.-H.; Wu, Y.-C.; Yeh, H.-C.; Sheu, J.-H. Cytotoxic and anti-inflammatory cembranoids from the soft coral Lobophytum crassum. J. Nat. Prod. 2008, 71, 1819–1824. [Google Scholar] [CrossRef]
- Spada, P.D.S.; de Souza, G.G.N.; Bortolini, G.V.; Henriques, J.A.P.; Salvador, M. Antioxidant, mutagenic, and antimutagenic activity of frozen fruits. J. Med. Food 2008, 11, 144–151. [Google Scholar] [CrossRef]
- Mohsen, S.M.; Ammar, A.S.M. Total phenolic contents and antioxidant activity of corn tassel extracts. Food Chem. 2009, 112, 595–598. [Google Scholar] [CrossRef]
- Shan, X.U.; Li, L.; Li-Qun, Z.; Zhuo, L.; Li-Li, Q.; Qi, C.; Chang-Fen, X. Reversal effect of 4’-methylether-scutellarein on multidrug resistance of human choriocarcinoma JAR/VP16 cell line. Prog. Biochem. Biophys. 2006, 33, 1061–1073. [Google Scholar]
- Gawron, A.; Kruk, I. Cytotoxic effect of xanthotoxol (8-hydroxypsoralen) on TCTC cells in vitro. Pol. J. Pharmacol. Pharm. 1992, 44, 51–57. [Google Scholar] [PubMed]
- Fan, G.J.; Han, B.H.; Kang, Y.H.; Park, M.K. Evaluation of inhibitory potentials of chinese medicinal plants on platelet-activating factor (PAF) receptor binding. Nat. Prod. Sci. 2001, 7, 33–37. [Google Scholar]
- Shahidi, F.; Janitha, P.K.; Wanasundara, P.D. Phenolic antioxidants. Crit. Rev. Food Sci. Nutr. 1992, 32, 67–103. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Moreno, C.; Larrauri, J.A.; Saura-Calixto, F. Free radical scavenging capacity and inhibition of lipid oxidation of wines, grape juices and related polyphenolic constituents. Food Res. Int. 1999, 32, 407–412. [Google Scholar] [CrossRef]
- Yuan, Y. V; Carrington, M.F.; Walsh, N.A. Extracts from dulse (Palmaria palmata) are effective antioxidants and inhibitors of cell proliferation in vitro. Food Chem. Toxicol. 2005, 43, 1073–1081. [Google Scholar] [CrossRef]
- Zhao, M.; Yang, B.; Wang, J.; Liu, Y.; Yu, L.; Jiang, Y. Immunomodulatory and anticancer activities of flavonoids extracted from litchi (Litchi chinensis Sonn.) pericarp. Int. Immunopharmacol. 2007, 7, 162–166. [Google Scholar] [CrossRef]
- Gorelik, E. Augmentation of the antimetastatic effect of anticoagulant drugs by immunostimulation in mice. Cancer Res. 1987, 47, 809–815. [Google Scholar]
- Yim, J.H.; Son, E.; Pyo, S.; Lee, H.K. Novel sulfated polysaccharide derived from red-tide microalga Gyrodinium impudicum strain KG03 with immunostimulating activity in vivo. Mar. Biotechnol. 2005, 7, 331–338. [Google Scholar] [CrossRef]
- Zhou, G.; Xin, H.; Sheng, W.; Sun, Y.; Li, Z.; Xu, Z. In vivo growth-inhibition of S180 tumor by mixture of 5-Fu and low molecular λ-carrageenan from Chondrus ocellatus. Pharmacol. Res. 2005, 51, 153–157. [Google Scholar] [CrossRef]
- Gorelik, E.; Bere, W.W.; Herberman, R.B. Role of NK cells in the antimetastatic effect of anticoagulant drugs. Int. J. Cancer 1984, 33, 87–94. [Google Scholar] [CrossRef] [PubMed]
- Dziarski, R. Synergistic enhancement of T cell responses and interleukin-1 receptor expression by interleukin-1 and heparin or dextran sulfate. Cell. Immunol. 1992, 145, 100–110. [Google Scholar] [CrossRef]
- Dziarski, R. Enhancement of mixed leukocyte reaction and cytotoxic antitumor responses by heparin. J. Immunol. 1989, 143, 356–365. [Google Scholar] [PubMed]
- O’Sullivan, G.M.; Boswell, C.M.; Halliday, G.M. Langerhans cell migration is modulated by N-sulfated glucosamine moieties in heparin. Exp. Dermatol. 2000, 9, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, M.; Yamori, T.; Naitoh, M.; Okutani, K. Structural revision of sulfated polysaccharide B-1 isolated from a marine Pseudomonas species and its cytotoxic activity against human cancer cell lines. Mar. Biotechnol. 2003, 5, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Joyce, J.A.; Freeman, C.; Meyer-Morse, N.; Parish, C.R.; Hanahan, D. A functional heparan sulfate mimetic implicates both heparanase and heparan sulfate in tumor angiogenesis and invasion in a mouse model of multistage cancer. Oncogene 2005, 24, 4037. [Google Scholar] [CrossRef] [PubMed]
- Berry, D.; Lynn, D.M.; Sasisekharan, R.; Langer, R. Poly (β-amino ester) s promote cellular uptake of heparin and cancer cell death. Chem. Biol. 2004, 11, 487–498. [Google Scholar] [CrossRef] [PubMed]
- Itoh, H.; Noda, H.; Amano, H.; Ito, H. Immunological analysis of inhibition of lung metastases by fucoidan (GIV-A) prepared from brown seaweed Sargassum thunbergii. Anticancer Res. 1995, 15, 1937–1947. [Google Scholar]
- Berteau, O.; Mulloy, B. Sulfated fucans, fresh perspectives: Structures, functions, and biological properties of sulfated fucans and an overview of enzymes active toward this class of polysaccharide. Glycobiology 2003, 13, 29R–40R. [Google Scholar] [CrossRef]
- Itoh, H.; Noda, H.; Amano, H.; Zhuaug, C.; Mizuno, T.; Ito, H. Antitumor activity and immunological properties of marine algal polysaccharides, especially fucoidan, prepared from Sargassum thunbergii of Phaeophyceae. Anticancer Res. 1993, 13, 2045–2052. [Google Scholar]
- Boisson-Vidal, C.; Zemani, F.; Caligiuri, G.; Galy-Fauroux, I.; Colliec-Jouault, S.; Helley, D.; Fischer, A.-M. Neoangiogenesis induced by progenitor endothelial cells: Effect of fucoidan from marine algae. Cardiovasc. Hematol. Agents Med. Chem. (Former. Curr. Med. Chem. Hematol. Agents) 2007, 5, 67–77. [Google Scholar] [CrossRef]
- Aisa, Y.; Miyakawa, Y.; Nakazato, T.; Shibata, H.; Saito, K.; Ikeda, Y.; Kizaki, M. Fucoidan induces apoptosis of human HS-sultan cells accompanied by activation of caspase-3 and down-regulation of ERK pathways. Am. J. Hematol. 2005, 78, 7–14. [Google Scholar] [CrossRef]
- Gross, H.; Goeger, D.E.; Hills, P.; Mooberry, S.L.; Ballantine, D.L.; Murray, T.F.; Valeriote, F.A.; Gerwick, W.H. Lophocladines, bioactive alkaloids from the red alga Lophocladia sp. J. Nat. Prod. 2006, 69, 640–644. [Google Scholar] [CrossRef] [PubMed]
- Ma, D.; Zou, B.; Cai, G.; Hu, X.; Liu, J.O. Total synthesis of the cyclodepsipeptide apratoxin A and its analogues and assessment of their biological activities. Chem. Eur. J. 2006, 12, 7615–7626. [Google Scholar] [CrossRef] [PubMed]
- Marquez, B.L.; Watts, K.S.; Yokochi, A.; Roberts, M.A.; Verdier-Pinard, P.; Jimenez, J.I.; Hamel, E.; Scheuer, P.J.; Gerwick, W.H. Structure and absolute stereochemistry of hectochlorin, a potent stimulator of actin assembly. J. Nat. Prod. 2002, 65, 866–871. [Google Scholar] [CrossRef]
- Mooberry, S.L.; Leal, R.M.; Tinley, T.L.; Luesch, H.; Moore, R.E.; Corbett, T.H. The molecular pharmacology of symplostatin 1: A new antimitotic dolastatin 10 analog. Int. J. Cancer 2003, 104, 512–521. [Google Scholar] [CrossRef]
- Kalemkerian, G.P.; Ou, X.; Adil, M.R.; Rosati, R.; Khoulani, M.M.; Madan, S.K.; Pettit, G.R. Activity of dolastatin 10 against small-cell lung cancer in vitro and in vivo: Induction of apoptosis and bcl-2 modification. Cancer Chemother. Pharmacol. 1999, 43, 507–515. [Google Scholar] [CrossRef]
- Costa, M.; Costa-Rodrigues, J.; Fernandes, M.H.; Barros, P.; Vasconcelos, V.; Martins, R. Marine cyanobacteria compounds with anticancer properties: A review on the implication of apoptosis. Mar. Drugs 2012, 10, 2181–2207. [Google Scholar] [CrossRef]
- Yu, Z.; Lang, G.; Kajahn, I.; Schmaljohann, R.; Imhoff, J.F. Scopularides A and B, cyclodepsipeptides from a marine sponge-derived fungus, Scopulariopsis brevicaulis. J. Nat. Prod. 2008, 71, 1052–1054. [Google Scholar] [CrossRef]
- Vasko, R.C.; Rodriguez, R.A.; Cunningham, C.N.; Ardi, V.C.; Agard, D.A.; McAlpine, S.R. Mechanistic studies of Sansalvamide A-amide: An allosteric modulator of Hsp90. ACS Med. Chem. Lett. 2010, 1, 4–8. [Google Scholar] [CrossRef]
- Sabina, H.; Aliya, R. Seaweed as a new source of flavone, scutellarein 4′-methyl-ether. Pak. J. Bot 2009, 41, 1927–1930. [Google Scholar]
- Devi, S.K.; Velmurugan, D. Molecular modeling, quantitative structure activity relationship and pharmacophore studies on antiǧviral, antiǧmalarial and antiǧinflammatory bioactive compounds from marine sources. Asian J. Pharm. Clin. Res. 2015, 8, 36–43. [Google Scholar]
- Lopes-Costa, E.; Abreu, M.; Gargiulo, D.; Rocha, E.; Ramos, A.A. Anticancer effects of seaweed compounds fucoxanthin and phloroglucinol, alone and in combination with 5-fluorouracil in colon cells. J. Toxicol. Environ. Heal. Part A 2017, 80, 776–787. [Google Scholar] [CrossRef] [PubMed]
- Magalhaes, K.D.; Costa, L.S.; Fidelis, G.P.; Oliveira, R.M.; Nobre, L.T.D.B.; Dantas-Santos, N.; Camara, R.B.G.; Albuquerque, I.R.L.; Cordeiro, S.L.; Sabry, D.A. Anticoagulant, antioxidant and antitumor activities of heterofucans from the seaweed Dictyopteris delicatula. Int. J. Mol. Sci. 2011, 12, 3352–3365. [Google Scholar] [CrossRef] [PubMed]
- Kale, V.; Freysdottir, J.; Paulsen, B.S.; Friðjónsson, Ó.H.; Hreggviðsson, G.O.; Omarsdottir, S. Sulphated polysaccharide from the sea cucumber Cucumaria frondosa affect maturation of human dendritic cells and their activation of allogeneic CD4 (+) T cells in vitro. Bioact. Carbohydr. Diet. Fibre 2013, 2, 108–117. [Google Scholar] [CrossRef]
- Clinical Trial.gov. Available online: https://clinicaltrials.gov/NCT00005838 (accessed on 17 August 2019).
- Drug Bank. Available online: http://www.drugbank.ca/DB05387 (accessed on 17 August 2019).
- Zhang, X.; Ye, X.; Chai, W.; Lian, X.-Y.; Zhang, Z. New metabolites and bioactive actinomycins from marine-derived Streptomyces sp. ZZ338. Mar. Drugs 2016, 14, 181. [Google Scholar] [CrossRef]
- Clinical Trial.gov. Available online: https://clinicaltrials.gov/NCT00491946 (accessed on 17 August 2019).
- Drug Bank. Available online: http://www.drugbank.ca/DB00970 (accessed on 17 August 2019).
- Da Rocha, A.B.; Lopes, R.M.; Schwartsmann, G. Natural products in anticancer therapy. Curr. Opin. Pharmacol. 2001, 1, 364–369. [Google Scholar] [CrossRef]
- Drug Bank. Available online: http://www.drugbank.ca/DB04977 (accessed on 17 August 2019).
- Clinical Trial.gov. Available online: https://clinicaltrials.gov/NCT00884286 (accessed on 17 August 2019).
- Clinical Trial.gov. Available online: https://clinicaltrials.gov/NCT00112476 (accessed on 17 August 2019).
- Drug Bank. Available online: http://www.drugbank.ca/DB00987 (accessed on 17 August 2019).
- Clinical Trial.gov. Available online: https://clinicaltrials.gov/NCT00003677 (accessed on 17 August 2019).
- Drug Bank. Available online: http://www.drugbank.ca/DB05109 (accessed on 17 August 2019).
- Clinical Trials.gov. Available online: https://clinicaltrials.gov/NCT00050427 (accessed on 17 August 2019).
- Drug Bank. Available online: http://www.drugbank.ca/DB04940 (accessed on 17 August 2019).
- Clinical Trials.gov. Available online: https://clinicaltrials.gov/NCT00965523 (accessed on 17 August 2019).
- Drug Bank. Available online: http://www.drugbank.ca/DB08871 (accessed on 17 August 2019).
- Clinical Trials.gov. Available online: https://https://clinicaltrials.gov/NCT01669252 (accessed on 17 August 2019).
- Drug Bank. Available online: http://www.drugbank.ca/DB05158 (accessed on 17 August 2019).
- Clinical Trial.gov. Available online: https://clinicaltrials.gov/NCT00884845 (accessed on 17 August 2019).
- Clinical Trial.gov. Available online: https://clinicaltrials.gov/NCT00629473 (accessed on 17 August 2019).
- Adjei, A.A. Signal transduction pathway targets for anticancer drug discovery. Curr. Pharm. Des. 2000, 6, 361–378. [Google Scholar] [CrossRef]
- Lu, J.; Ma, Y.; Liang, J.; Xing, Y.; Xi, T.; Lu, Y. Aureolic acids from a marine-derived Streptomyces sp. WBF16. Microbiol. Res. 2012, 167, 590–595. [Google Scholar] [CrossRef]
- Tewey, K.M.; Chen, G.L.; Nelson, E.M.; Liu, L.-F. Intercalative antitumor drugs interfere with the breakage-reunion reaction of mammalian DNA topoisomerase II. J. Biol. Chem. 1984, 259, 9182–9187. [Google Scholar]
- Shi, J.-G.; Jia, Z.-J.; Cui, Y.-X. Novel tricyclic diterpenoids from Euphorbia micractina. J. Nat. Prod. 1995, 58, 51–56. [Google Scholar] [CrossRef]
- Wu, D.; Sorg, B.; Hecker, E. New myrsinol-related polyfunctional pentacyclic diterpene esters from roots of Euphorbia prolifera. J. Nat. Prod. 1995, 58, 408–413. [Google Scholar] [CrossRef] [PubMed]
- Albers, M.W.; Williams, R.T.; Brown, E.J.; Tanaka, A.; Hall, F.L.; Schreiber, S.L. FKBP-rapamycin inhibits a cyclin-dependent kinase activity and a cyclin D1-Cdk association in early G1 of an osteosarcoma cell line. J. Biol. Chem. 1993, 268, 22825–22829. [Google Scholar] [PubMed]
- Zunino, F.; Capranico, G. DNA topoisomerase II as the primary target of anti-tumor anthracyclines. Anti-cancer. Drug Des. 1990, 5, 307–317. [Google Scholar]
- Cardenas, M.E.; Sanfridson, A.; Cutler, N.S.; Heitman, J. Signal-transduction cascades as targets for therapeutic intervention by natural products. Trends Biotechnol. 1998, 16, 427–433. [Google Scholar] [CrossRef]
- Lin, H.-N.; Wang, K.-L.; Wu, Z.-H.; Tian, R.-M.; Liu, G.-Z.; Xu, Y. Biological and chemical diversity of bacteria associated with a marine flatworm. Mar. Drugs 2017, 15, 281. [Google Scholar] [CrossRef] [PubMed]
- Monk, B.J.; Herzog, T.J.; Kaye, S.B.; Krasner, C.N.; Vermorken, J.B.; Muggia, F.M.; Pujade-Lauraine, E.; Lisyanskaya, A.S.; Makhson, A.N.; Rolski, J. Trabectedin plus pegylated liposomal doxorubicin in recurrent ovarian cancer. J. Clin. Oncol. 2010, 28, 3107–3114. [Google Scholar] [CrossRef] [PubMed]
- Carter, N.J.; Keam, S.J. Trabectedin. Drugs 2007, 67, 2257–2276. [Google Scholar] [CrossRef] [PubMed]
- Kortmansky, J.; Schwartz, G.K. Bryostatin-1: A novel PKC inhibitor in clinical development. Cancer Investig. 2003, 21, 924–936. [Google Scholar] [CrossRef]
- Sekar, M.; Poomalai, S.; Gunasekaran, M.; Mani, P.; Krishnamurthy, A. Bioactive compounds from marine yeast inhibits lung cancer. J. Appl. Pharm. Sci. 2015, 5, 7–15. [Google Scholar]
- Wall, N.R.; Mohammad, R.M.; Reddy, K.B.; Al-Katib, A.M. Bryostatin 1 induces ubiquitination and proteasome degradation of Bcl-2 in the human acute lymphoblastic leukemia cell line, Reh. Int. J. Mol. Med. 2000, 5, 165–236. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.; Sharma, M.; Joshi, P.; Rawat, D.S. Clinical status of anti-cancer agents derived from marine sources. Anti-Cancer Agents Med. Chem. (Former. Curr. Med. Chem. Agents) 2008, 8, 603–617. [Google Scholar] [CrossRef]
- Dumez, H.; Gall, H.; Capdeville, R.; Dutreix, C.; van Oosterom, A.T.; Giaccone, G. A phase I and pharmacokinetic study of LAF389 administered to patients with advanced cancer. Anti-Cancer Drugs 2007, 18, 219–225. [Google Scholar] [CrossRef] [PubMed]
- Simmons, T.L.; Andrianasolo, E.; McPhail, K.; Flatt, P.; Gerwick, W.H. Marine natural products as anticancer drugs. Mol. Cancer Ther. 2005, 4, 333–342. [Google Scholar] [PubMed]
- Feling, R.H.; Buchanan, G.O.; Mincer, T.J.; Kauffman, C.A.; Jensen, P.R.; Fenical, W. Salinosporamide A: A highly cytotoxic proteasome inhibitor from a novel microbial source, a marine bacterium of the new genus Salinospora. Angew. Chem. Int. Ed. 2003, 42, 355–357. [Google Scholar] [CrossRef] [PubMed]
- Thakur, N.L.; Thakur, A.N. Marine Biotechnology: An overview. Indian J. Biotechnol. 2006, 5, 263–268. [Google Scholar]
- Schwartsmann, G.; da Rocha, A.B.; Berlinck, R.G.S.; Jimeno, J. Marine organisms as a source of new anticancer agents. Lancet Oncol. 2001, 2, 221–225. [Google Scholar] [CrossRef]
- Rosén, J.; Lövgren, A.; Kogej, T.; Muresan, S.; Gottfries, J.; Backlund, A. ChemGPS-NP Web: Chemical space navigation online. J. Comput. Aided Mol. Des. 2009, 23, 253–259. [Google Scholar] [CrossRef]
- Chemsketsh. Available online: www.acdlabs.com (accessed on 1 March 2019).
- Muigg, P.; Rosén, J.; Bohlin, L.; Backlund, A. In silico comparison of marine, terrestrial and synthetic compounds using ChemGPS-NP for navigating chemical space. Phytochem. Rev. 2013, 12, 449–457. [Google Scholar] [CrossRef]

| Compound Name/Class | Marine Source | Type of Cancer | Mechanism | References |
|---|---|---|---|---|
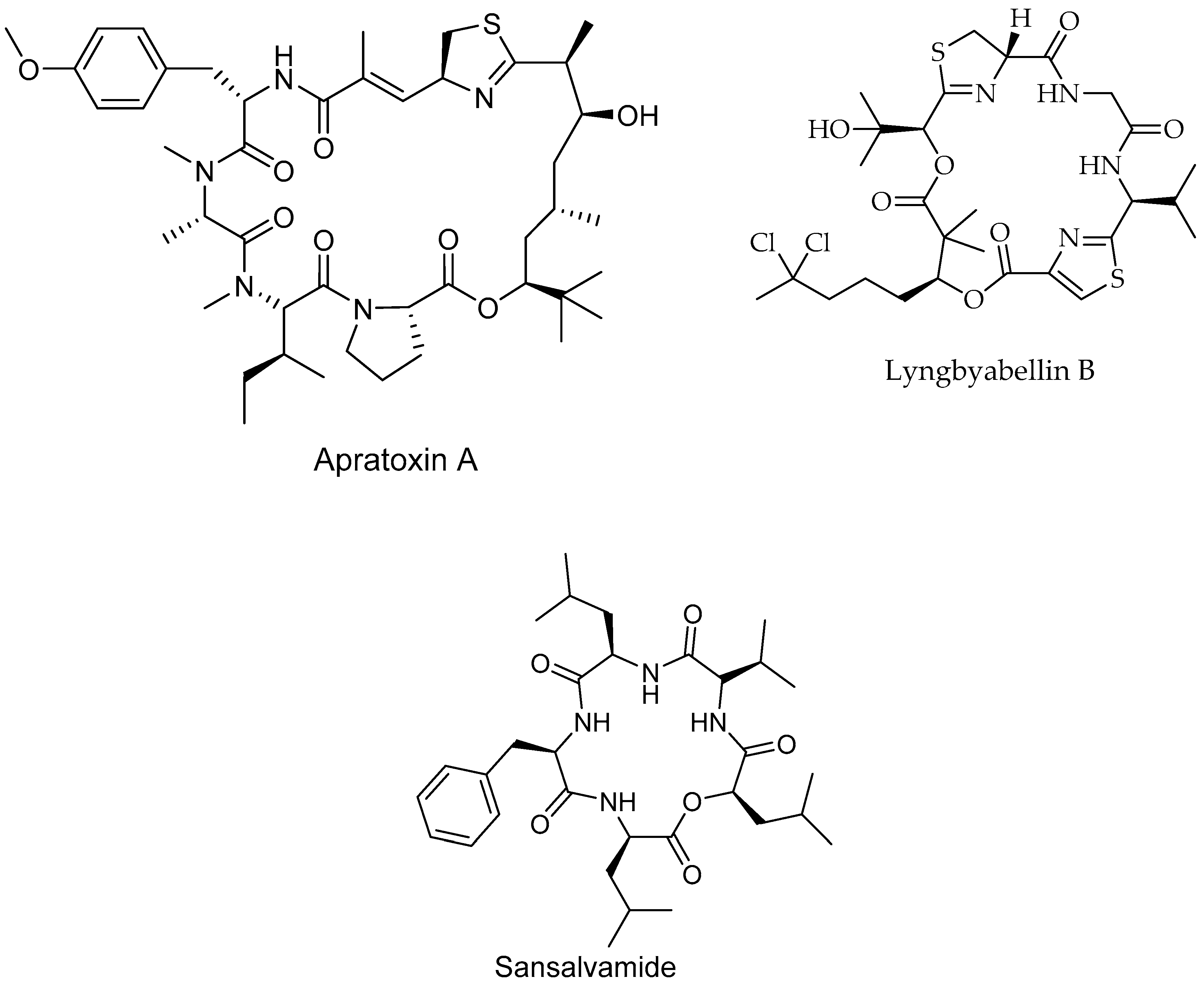
| Apratoxin A/Peptide | Lyngbya boulloni, bacteria | Cervical cancer | Cell cycle inhibition IC50 = 2.2 nM | [181] |
| Brugine/Alkaloid | Bruguiera sexangula, plant | Sarcoma 180 and Lewis | Not reported | [31] |
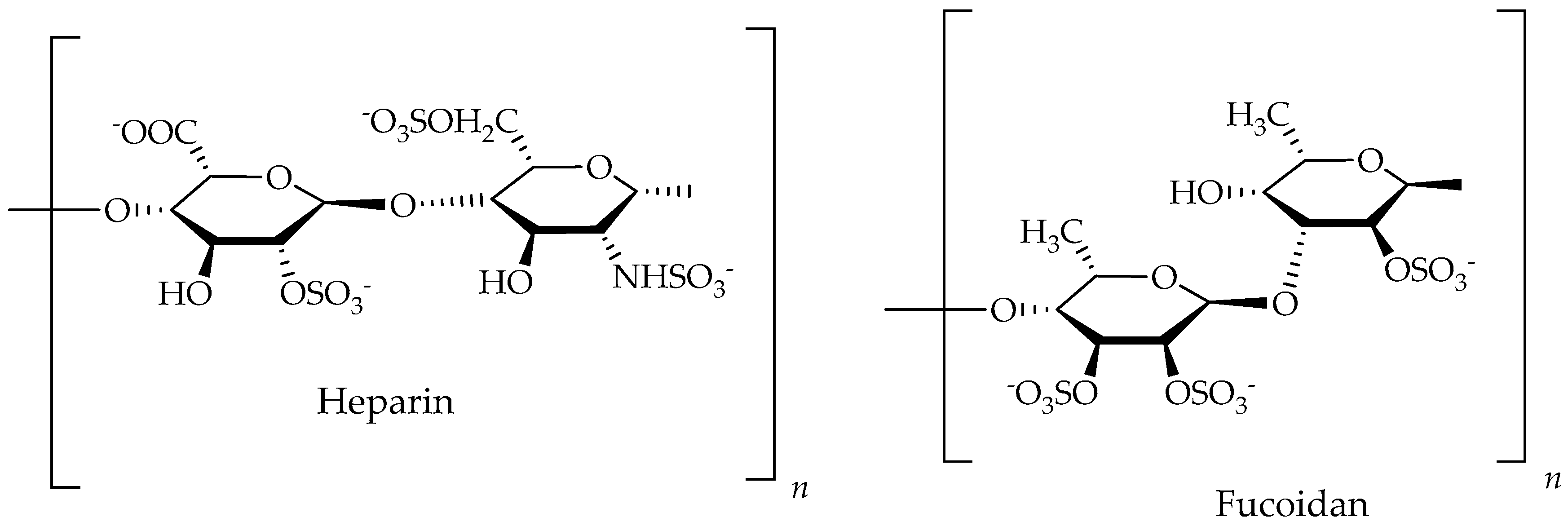
| Fucoidan/Polysaccharides | Ascophyllum nodosum, algea | Colon cancer | Inhibit the proliferation of arterial smooth muscle cells at conc. of 80 to 100 µg/mL | [78] |
| Lyngbyabellin B/p Peptide | Lyngbya majuscule, bacteria | Burkitt lymphoma cancer | Inhibit of cell growth IC50 = 0.02 µM | [182] |
| Sansalvamide A/Peptide | Marine fungi | Pancreatic, colon, breast and prostate cancers | Inhibits protein complex formation | [187] |
| Scutellarein 4′-methylether/Polyphenol | Osmundea pinnatifida, algea | Choriocarcinoma cancer | Not reported | [158,188] |
| Phlorofucofuroecol A/Polyphenol | Brown seaweeds | Cancer | Not reported | [189] |
| Phloroglucinol/polyphenol | Brown seaweed | Colon cancer | Induce DNA damage, and cell death at 300 µM | [190] |
| Heparin/Heparan/Ppolysaccharides | Dictyopteris delicatula, Seaweed | Colon cancer | Inhibit the proliferation of arterial smooth muscle cells at 80 to 100 µg/mL | [78,191] |
| Chondroitin-4-sulphate/Polysaccharides | Cucumaria frondosa, sea cucumber | Not reported | [31,192] | |
| Chondroitin-6-sulphate/Polysaccharides | Cucumaria frondosa, sea cucumber | Not reported | [31,192] |
| Compound Name/Chemical Class | Marine Source | Type of Cancer | Mechanism | Clinical Status/Study Type | References |
|---|---|---|---|---|---|
| AE-941/Peptide | Shark cartilage | Renal, lung cancer | Inhibition of gelatinolytic and elastinolytic activities of MMP-2, MMP-9, and MMP-12. The MMP’s are often over expressed in tumors and play an important role in the degradation of the (extracellular matrix allowing tumor growth and invasion (metastasis) | Drug, phase 3, Investigationa, Interventional | [44,193,194] |
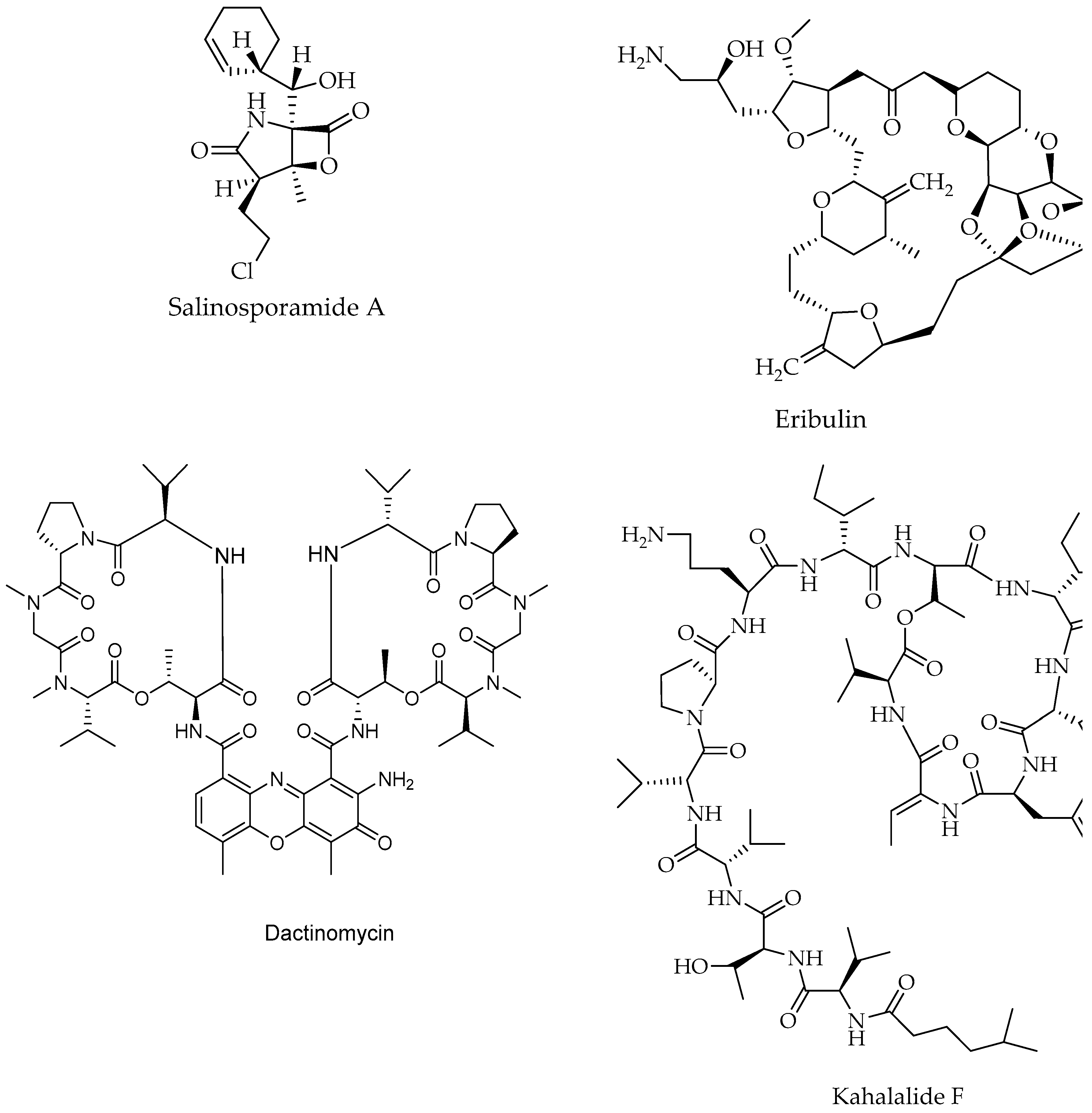
| Actinomycin/Peptide | Streptomyces parvullus, Streptomyces sp. ZZ338 Actinomyces | Childhood cancer, Wilms tumor | Inhibition of RNA polymerase | Drug, phase 4, Interventional | [195,196,197] |
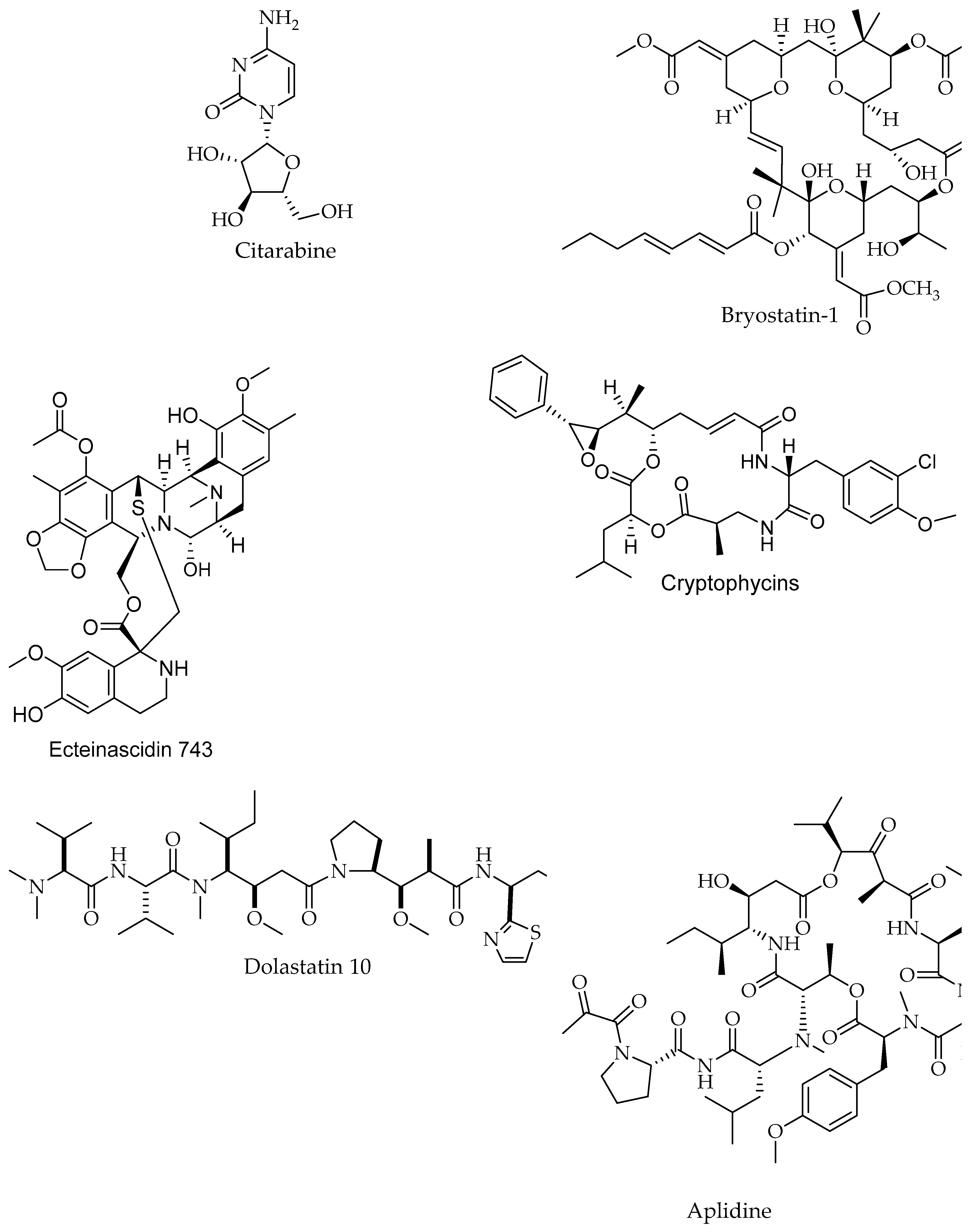
| Aplidine (Plitidepsin, Dehydrodide-mnin B)/Peptide | Aplidium albicans, Tunicate, Ascidiacea | Pancreatic, stomach, bladder, and prostate cancers | Activation of protein kinase C | Drug Investigational | [198,199] |
| Leukemia Non Hodgkin Lymphoma | Induce the apoptotic cascade | Drug phase 2, Interventional | [44,198,200] | ||
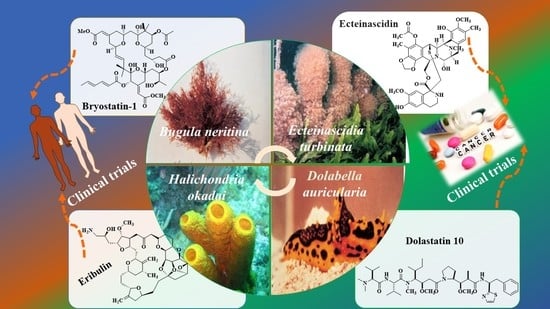
| Bryostatin-1/Polyketide | Bugula neritina, Bryozoa | Metastatic solid tumors | Inhibition of growth and alteration of differentiation | Drug phase 1, 2 Interventional | [44,198,201] |
| Citarabine/Alkaloid | Sponge | Leukemia (acute non-lymphoblastic) | Inhibition of DNA synthesis | Drug Approved, Investigational | [198,202] |
| Cryptophycins/Peptide | Nostoc sp., Macroalgae & Dysidea arenaria, Sponge | Not reported | Tubulin (inhibition of polymerization of microtuble) | Phase 1 | [44] |
| Dolastatin 10/Peptide | Dolabella auricularia, Mollusc | Pancreatic cancer | Inhibition of microtubules and pro-apoptotic effects | Drug phase 2, Interventional | [44,198,203] |
| ET-743 (Trabectedin, Ecteinascidin)/Alkaloid | Carribean tunicate Ecteinascidia turbinate Tunicate, Ascidiacea | Sarcomas and ovarian cancer | Binding to the minor groove of DNA interfering with cell division and genetic transcription processes and DNA repair machinery | Drug Approved, Investigational | [44,198,204] |
| Breast cancer | Alkylation ofguanine residues in the DNA minor groove | Drug phase 2, Interventional | [198,205] | ||
| Eribulin (E7389)/Polyketide | Lissodendoryx sp. Halichondria okadai., Sponge | Breast cancer | Activation of cellular apoptosis under anchorage-independent and -dependent cell culture conditions | Phase 1,2, Investigationa, Interventional | [44,206,207] |
| Advanced solid tumors, breast | Inhibition of growth phase of microtubules without affecting the shortening phase and sequesters tubulin into nonproductive aggregates | Drug, Approved, phase 2, Investigationa, Interventional | [208,209] | ||
| Kahalalide F/Peptide | Elysia rufescens, Mollusc/Bryopsis sp., Macroalgae | Prostate cancer | Induction of changes in lysosomal membrane | Phase 2 | [44] |
| PM02734/Peptide | Elysia rufescens, Mollusk | Breast, colon, pancreas, lung and prostate | Antiproliferative | Drug, phase 1, Investigation, Interventional | [210,211] |
| Salinosporamide A (Marizomib®) (NPI-0052)/Polyketide | Salinospora tropica, actinomyces | Prevention of proteins breakdown involved in signal transduction, which blocks the cancer cells growth and survival | Drug phase 1, Interventional | [17,44,212] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khalifa, S.A.M.; Elias, N.; Farag, M.A.; Chen, L.; Saeed, A.; Hegazy, M.-E.F.; Moustafa, M.S.; Abd El-Wahed, A.; Al-Mousawi, S.M.; Musharraf, S.G.; et al. Marine Natural Products: A Source of Novel Anticancer Drugs. Mar. Drugs 2019, 17, 491. https://doi.org/10.3390/md17090491
Khalifa SAM, Elias N, Farag MA, Chen L, Saeed A, Hegazy M-EF, Moustafa MS, Abd El-Wahed A, Al-Mousawi SM, Musharraf SG, et al. Marine Natural Products: A Source of Novel Anticancer Drugs. Marine Drugs. 2019; 17(9):491. https://doi.org/10.3390/md17090491
Chicago/Turabian StyleKhalifa, Shaden A. M., Nizar Elias, Mohamed A. Farag, Lei Chen, Aamer Saeed, Mohamed-Elamir F. Hegazy, Moustafa S. Moustafa, Aida Abd El-Wahed, Saleh M. Al-Mousawi, Syed G. Musharraf, and et al. 2019. "Marine Natural Products: A Source of Novel Anticancer Drugs" Marine Drugs 17, no. 9: 491. https://doi.org/10.3390/md17090491
APA StyleKhalifa, S. A. M., Elias, N., Farag, M. A., Chen, L., Saeed, A., Hegazy, M.-E. F., Moustafa, M. S., Abd El-Wahed, A., Al-Mousawi, S. M., Musharraf, S. G., Chang, F.-R., Iwasaki, A., Suenaga, K., Alajlani, M., Göransson, U., & El-Seedi, H. R. (2019). Marine Natural Products: A Source of Novel Anticancer Drugs. Marine Drugs, 17(9), 491. https://doi.org/10.3390/md17090491