Biorefining and the Functional Properties of Proteins from Lipid and Pigment Extract Residue of Chlorella pyrenoidosa
Abstract
:1. Introduction
2. Results and Discussion
2.1. Optimization of Protein Extraction Conditions by Single-Factor Experiments
2.2. Optimization of Protein Extraction Conditions by RSM
2.2.1. Model Fitting and Adequacy Checking
7.25X1X3 + 19.70X1X4 − 41.57X2X3 + 0.85X2X4 + 6.88X3X4 − 25.37X12 −
27.48X22 − 25.28X32 − 6.17X42
2.2.2. Response Surface Analysis
2.3. Molecular Weight Distribution of MRPI
2.4. Amino Acid Profile of MRPI
2.5. Emulsifying Ability and Stability of MRPI
2.6. Antioxidant Activity of MRPI
3. Materials and Methods
3.1. Microalgal Residue
3.2. Optimization of Protein Extraction Conditions
3.3. Determination of Protein Yield
3.4. Purification of MRPI
3.5. Determination of the Molecular Weight of MRPI
3.6. Determination of the Amino Acid Composition of MRPI
3.7. Determination of the Emulsifying Properties of MRPI
3.8. Determination of the Antioxidant Activites of MRPI
3.9. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Pulz, O.; Gross, W. Valuable products from biotechnology of microalgae. Appl. Microbiol. Biotechnol. 2004, 65, 635–648. [Google Scholar] [CrossRef] [PubMed]
- Barkia, I.; Saari, N.; Manning, S.R. Microalgae for high-value products towards human health and nutrition. Mar. Drugs 2019, 17, 304. [Google Scholar] [CrossRef] [PubMed]
- D’Alessandro, E.B.; Antoniosi, N.R. Concepts and studies on lipid and pigments of microalgae: A review. Renew. Sustain. Energy Rev. 2016, 58, 832–841. [Google Scholar] [CrossRef]
- Koller, M.; Muhr, A.; Braunegg, G. Microalgae as versatile cellular factories for valued products. Algal Res. 2014, 6, 52–63. [Google Scholar] [CrossRef]
- Camacho, F.; Macedo, A.; Malcata, F. Potential industrial applications and commercialization of microalgae in the functional food and feed industries: A short review. Mar. Drugs 2019, 17, 312. [Google Scholar] [CrossRef]
- Huang, J.J.H.; Lin, S.L.; Xu, W.W.; Cheung, P.C.K. Occurrence and biosynthesis of carotenoids in phytoplankton. Biotechnol. Adv. 2017, 35, 597–618. [Google Scholar] [CrossRef]
- Barsanti, L.; Gualtieri, P. Is exploitation of microalgae economically and energetically sustainable? Algal Res. 2018, 31, 107–115. [Google Scholar] [CrossRef]
- Ehimen, E.A.; Connaughton, S.; Sun, Z.F.; Carrington, G.C. Energy recovery from lipid extracted, transesterified and glycerol codigested microalgae biomass. Gcb Bioenergy 2009, 1, 371–381. [Google Scholar] [CrossRef]
- Norzagaray-Valenzuela, C.D.; Valdez-Ortiz, A.; Shelton, L.M.; Jimenez-Edeza, M.; Rivera-Lopez, J.; Valdez-Flores, M.A.; German-Baez, L.J. Residual biomasses and protein hydrolysates of three green microalgae species exhibit antioxidant and anti-aging activity. J. Appl. Phycol. 2017, 29, 189–198. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, X. Separation, antitumor activities, and encapsulation of polypeptide from Chlorella pyrenoidosa. Biotechnol. Prog. 2013, 29, 681–687. [Google Scholar] [CrossRef]
- Soetrisno, U.S.S.; Holmes, Z. Protein yields and characteristics from acid and salt coagulations of yellow pea (Pisum Sativum L. Miranda) flour extractions. J. Agric. Food Chem. 1992, 40, 970–974. [Google Scholar] [CrossRef]
- Shen, L.; Wang, X.; Wang, Z.; Wu, Y.; Chen, J. Studies on tea protein extraction using alkaline and enzyme methods. Food Chem. 2008, 107, 929–938. [Google Scholar] [CrossRef]
- Phongthai, S.; Lim, S.-T.; Rawdkuen, S. Ultrasonic-assisted extraction of rice bran protein using response surface methodology. J. Food Biochem. 2017, 41, e12314. [Google Scholar] [CrossRef]
- Horax, R.; Hettiarachchy, N.; Kannan, A.; Chen, P. Protein extraction optimisation, characterisation, and functionalities of protein isolate from bitter melon (Momordica charantia) seed. Food Chem. 2011, 124, 545–550. [Google Scholar] [CrossRef]
- Ma, T.; Wang, Q.; Wu, H. Optimization of extraction conditions for improving solubility of peanut protein concentrates by response surface methodology. LWT Food Sci. Technol. 2010, 43, 1450–1455. [Google Scholar] [CrossRef]
- Cui, Q.; Ni, X.; Zeng, L.; Tu, Z.; Li, J.; Sun, K.; Chen, X.; Li, X. Optimization of protein extraction and decoloration conditions for tea residues. Hortic. Plant J. 2017, 3, 172–176. [Google Scholar] [CrossRef]
- Ayim, I.; Ma, H.L.; Alenyorege, E.A. Optimizing and predicting degree of hydrolysis of ultrasound assisted sodium hydroxide extraction of protein from tea (Camellia sinensis L.) residue using response surface methodology. J. Food Sci. Technol. 2018, 55, 5166–5174. [Google Scholar] [CrossRef]
- Hemwimon, S.; Pavasant, P.; Shotipruk, A. Microwave-assisted extraction of antioxidative anthraquinones from roots of Morinda citrifolia. Sep. Purif. Technol. 2007, 54, 44–50. [Google Scholar] [CrossRef]
- Zhang, C.; Sanders, J.P.M.; Bruins, M.E. Critical parameters in cost-effective alkaline extraction for high protein yield from leaves. Biomass Bioenergy 2014, 67, 466–472. [Google Scholar] [CrossRef]
- Gerde, J.A.; Wang, T.; Yao, L.; Jung, S.; Johnson, L.A.; Lamsal, B. Optimizing protein isolation from defatted and non-defatted Nannochloropsis microalgae biomass. Algal Res. 2013, 2, 145–153. [Google Scholar] [CrossRef]
- Feyzi, S.; Milani, E.; Golimovahhed, Q.A. Grass pea (Lathyrus sativus L.) protein isolate: The effect of extraction optimization and drying methods on the structure and functional properties. Food Hydrocolloid. 2018, 74, 187–196. [Google Scholar] [CrossRef]
- Al-Farsi, M.A.; Lee, C.Y. Optimization of phenolics and dietary fibre extraction from date seeds. Food Chem. 2008, 108, 977–985. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, S.; Nikolov, Z. Process for selective extraction of pigments and functional proteins from Chlorella vulgaris. Algal Res. 2018, 35, 185–193. [Google Scholar] [CrossRef]
- Schwenzfeier, A.; Wierenga, P.A.; Gruppen, H. Isolation and characterization of soluble protein from the green microalgae Tetraselmis sp. Bioresour. Technol. 2011, 102, 9121–9127. [Google Scholar] [CrossRef] [PubMed]
- Kadam, S.U.; Alvarez, C.; Tiwari, B.K.; O’Donnell, C.P. Extraction and characterization of protein from Irish brown seaweed Ascophyllum nodosum. Food Res. Int. 2017, 99, 1021–1027. [Google Scholar] [CrossRef] [PubMed]
- Jambrak, A.R.; Mason, T.J.; Lelas, V.; Paniwnyk, L.; Herceg, Z. Effect of ultrasound treatment on particle size and molecular weight of whey proteins. J. Food Eng. 2014, 121, 15–23. [Google Scholar] [CrossRef]
- Becker, E.W. Micro-algae as a source of protein. Biotechnol. Adv. 2007, 25, 207–210. [Google Scholar] [CrossRef] [PubMed]
- Ursu, A.V.; Marcati, A.; Sayd, T.; Sante-Lhoutellier, V.; Djelveh, G.; Michaud, P. Extraction, fractionation and functional properties of proteins from the microalgae Chlorella vulgaris. Bioresour. Technol. 2014, 157, 134–139. [Google Scholar] [CrossRef] [PubMed]
- Joint WHO/FAO/UNU Expert Consultation. World Health Organization Technical Report Series. In Protein and Amino Acid Requirements in Human Nutrition; WHO Press: Geneva, Switzerland, 2007; Volume 935, p. 150. [Google Scholar]
- Sui, Y.X.; Muys, M.; Van de Waal, D.B.; D’Adamo, S.; Vermeir, P.; Fernandes, T.V.; Vlaeminck, S.E. Enhancement of co-production of nutritional protein and carotenoids in Dunaliella salina using a two-phase cultivation assisted by nitrogen level and light intensity. Bioresour. Technol. 2019, 287, 121398. [Google Scholar] [CrossRef] [PubMed]
- Kent, M.; Welladsen, H.M.; Mangott, A.; Li, Y. Nutritional evaluation of Australian microalgae as potential human health supplements. PLoS ONE 2015, 10, e0118985. [Google Scholar] [CrossRef] [PubMed]
- Moure, A.; Sineiro, J.; Domínguez, H.; Parajó, J.C. Functionality of oilseed protein products: A review. Food Res. Int. 2006, 39, 945–963. [Google Scholar] [CrossRef]
- El Nasri, N.A.; El Tinay, A.H. Functional properties of fenugreek (Trigonella foenum graecum) protein concentrate. Food Chem. 2007, 103, 582–589. [Google Scholar] [CrossRef]
- Wasswa, J.; Tang, J.; Gu, X.H. Functional properties of Grass carp (Ctenopharyngodon idella), Nile perch (Lates niloticus) and Nile tilapia (Oreochromis niloticus) skin hydrolysates. Int. J. Food Prop. 2008, 11, 339–350. [Google Scholar] [CrossRef]
- Sheih, I.C.; Wu, T.K.; Fang, T.J. Antioxidant properties of a new antioxidative peptide from algae protein waste hydrolysate in different oxidation systems. Bioresour. Technol. 2009, 100, 3419–3425. [Google Scholar] [CrossRef] [PubMed]
- Cacciuttolo, M.A.; Trinh, L.; Lumpkin, J.A.; Rao, G. Hyperoxia Induces DNA Damage in Mammalian-Cells. Free Radic. Biol. Med. 1993, 14, 267–276. [Google Scholar] [CrossRef]
- Zhang, M.; Mu, T.H.; Wang, Y.B.; Sun, M.J. Evaluation of free radical-scavenging activities of sweet potato protein and its hydrolysates as affected by single and combination of enzyme systems. Int. J. Food Sci. Technol. 2012, 47, 696–702. [Google Scholar] [CrossRef]
- Pan, M.; Jiang, T.S.; Pan, J.L. Antioxidant activities of rapeseed protein hydrolysates. Food Bioproc. Technol. 2011, 4, 1144–1152. [Google Scholar] [CrossRef]
- Ben Slama-Ben Salem, R.; Bkhairia, I.; Abdelhedi, O.; Nasri, M. Octopus vulgaris protein hydrolysates: characterization, antioxidant and functional properties. J. Food Sci. Technol 2017, 54, 1442. [Google Scholar] [CrossRef]
- Nasri, R.; Bougatef, A.; Ben Khaled, H.; Nedjar-Arroume, N.; Chaabouni, M.K.; Dhulster, P.; Nasri, M. Antioxidant and free radical-scavenging activities of goby (Zosterisessor ophiocephalus) muscle protein hydrolysates obtained by enzymatic treatment. Food Biotechnol. 2012, 26, 266–279. [Google Scholar] [CrossRef]
- Gong, M.; Li, X.; Bassi, A. Investigation of simultaneous lutein and lipid extraction from wet microalgae using Nile Red as solvatochromic shift probe. J. Appl. Phycol. 2018, 30, 1617–1627. [Google Scholar] [CrossRef]
- Yasumatsu, K.; Sawada, K.; Moritaka, S.; Misaki, M.; Toda, J.; Wada, T.; Ishii, K. Whipping and Emulsifying Properties of Soybean Products. Agric. Biol. Chem. 1972, 36, 719–727. [Google Scholar] [CrossRef]
- Qin, Y.; Xiong, L.; Li, M.; Liu, J.; Wu, H.; Qiu, H.W.; Mu, H.Y.; Xu, X.F.; Sun, Q.J. Preparation of bioactive polysaccharide nanoparticles with enhanced radical scavenging activity and antimicrobial activity. J. Agric. Food Chem. 2018, 66, 4373–4383. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.R.; Qiu, Y.T.; Zhao, Y.Q.; Chi, C.F.; Wang, B. Purification and characterization of antioxidant peptides derived from protein hydrolysate of the marine bivalve mollusk Tergillarca granosa. Mar. Drugs 2019, 17, 251. [Google Scholar] [CrossRef] [PubMed]
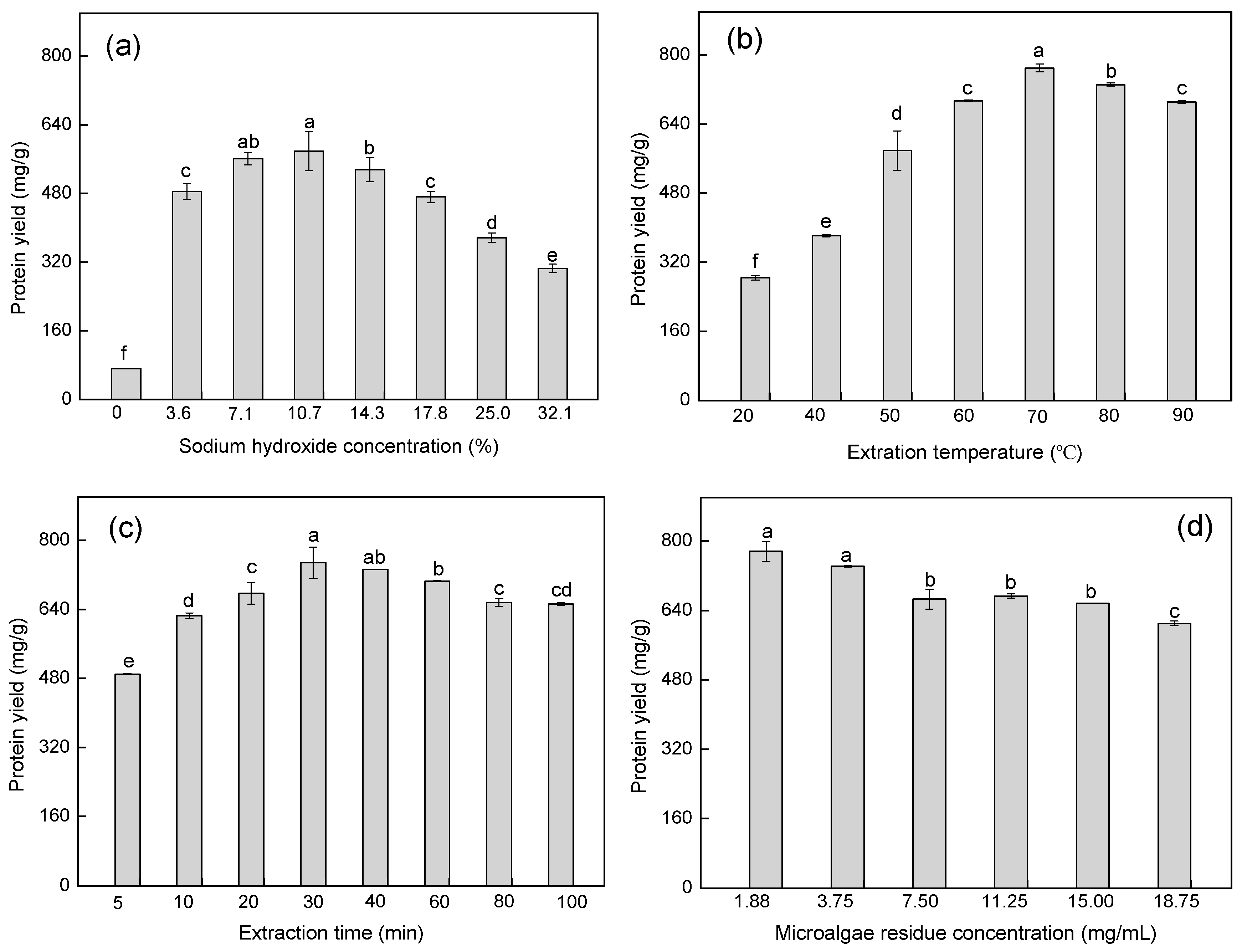
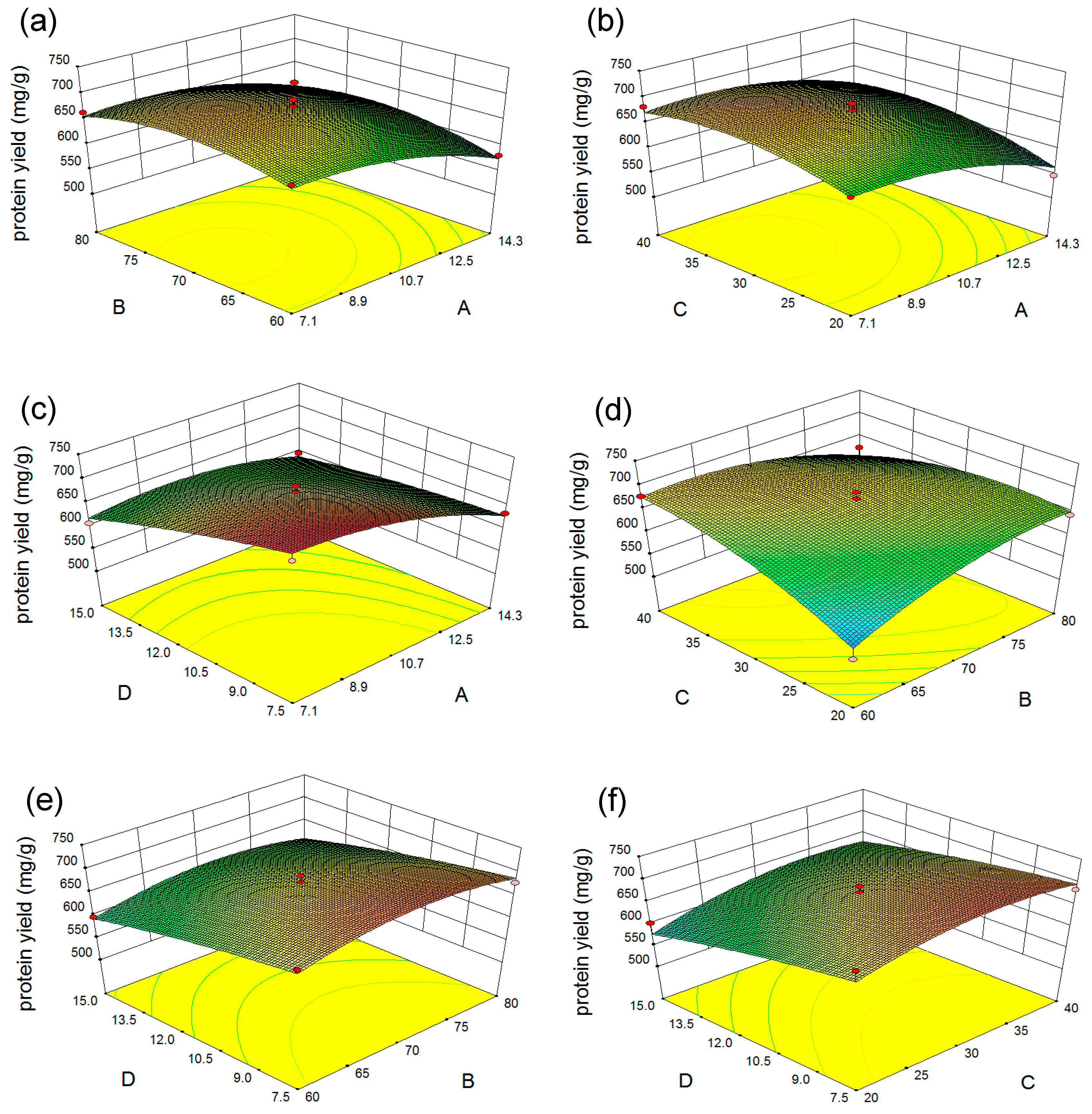
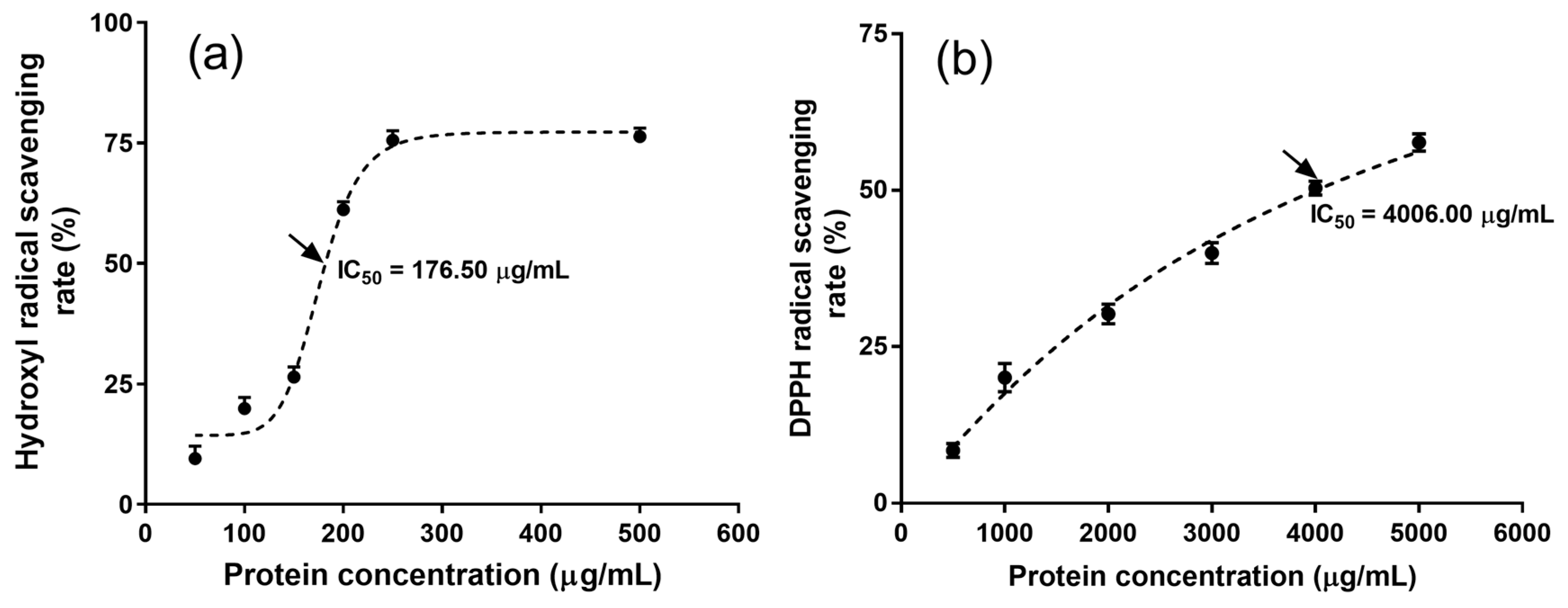
| Runs | Sodium Hydroxide Concentration (%) (X1) | Extraction Temperature (°C) (X2) | Extraction Time (min) (X3) | Microalgal Residue Concentration (mg/mL) (X4) | Protein Yield (mg/g) (Y) |
|---|---|---|---|---|---|
| 1 | −1(7.1) | −1(60) | 0(30) | 0(11.25) | 661.99 |
| 2 | 1(14.3) | −1(60) | 0(30) | 0(11.25) | 580.09 |
| 3 | −1(7.1) | 1(80) | 0(30) | 0(11.25) | 663.72 |
| 4 | 1(14.3) | 1(80) | 0(30) | 0(11.25) | 609.07 |
| 5 | 0(10.7) | 0(70) | −1(20) | −1(7.50) | 693.98 |
| 6 | 0(10.7) | 0(70) | 1(40) | −1(7.50) | 683.62 |
| 7 | 0(10.7) | 0(70) | −1(20) | 1(15.00) | 602.86 |
| 8 | 0(10.7) | 0(70) | 1(40) | 1(15.00) | 620.02 |
| 9 | −1(7.1) | 0(70) | 0(30) | −1(7.50) | 722.48 |
| 10 | 1(14.3) | 0(70) | 0(30) | −1(7.50) | 633.35 |
| 11 | −1(7.1) | 0(70) | 0(30) | 1(15.00) | 607.84 |
| 12 | 1(14.3) | 0(70) | 0(30) | 1(15.00) | 597.47 |
| 13 | 0(10.7) | −1(60) | −1(20) | 0(11.25) | 530.00 |
| 14 | 0(10.7) | 1(80) | −1(20) | 0(11.25) | 642.97 |
| 15 | 0(10.7) | −1(60) | 1(40) | 0(11.25) | 678.30 |
| 16 | 0(10.7) | 1(80) | 1(40) | 0(11.25) | 624.99. |
| 17 | −1(7.1) | 0(70) | −1(20) | 0(11.25) | 646.43 |
| 18 | 1(14.3) | 0(70) | −1(20) | 0(11.25) | 546.15 |
| 19 | −1(7.1) | 0(70) | 1(40) | 0(11.25) | 681.98 |
| 20 | 1(14.3) | 0(70) | 1(40) | 0(11.25) | 610.80 |
| 21 | 0(10.7) | −1(60) | 0(30) | −1(7.50) | 682.49 |
| 22 | 0(10.7) | 1(80) | 0(30) | −1(7.50) | 677.32 |
| 23 | 0(10.7) | −1(60) | 0(30) | 1(15.00) | 597.66 |
| 24 | 0(10.7) | 1(80) | 0(30) | 1(15.00) | 595.86 |
| 25 | 0(10.7) | 0(70) | 0(30) | 0(11.25) | 688.89 |
| 26 | 0(10.7) | 0(70) | 0(30) | 0(11.25) | 675.59 |
| 27 | 0(10.7) | 0(70) | 0(30) | 0(11.25) | 667.18 |
| 28 | 0(10.7) | 0(70) | 0(30) | 0(11.25) | 669.89 |
| 29 | 0(10.7) | 0(70) | 0(30) | 0(11.25) | 674.09 |
| Source | Degrees of Freedom | Sum of Squares | Mean Square | F-Value | p-Value |
|---|---|---|---|---|---|
| Model | 14 | 56,420.89 | 4030.06 | 13.78 | <0.0001 ** |
| X1 | 1 | 13,824.44 | 13,824.44 | 47.25 | <0.0001 ** |
| X2 | 1 | 579.63 | 579.63 | 1.98 | 0.1811 |
| X3 | 1 | 4688.65 | 4688.65 | 16.03 | 0.0013 ** |
| X4 | 1 | 18,526.02 | 18,526.02 | 63.32 | <0.0001 ** |
| X1X2 | 1 | 186.32 | 186.32 | 0.64 | 0.4382 |
| X1X3 | 1 | 210.25 | 210.25 | 0.72 | 0.4108 |
| X1X4 | 1 | 1552.36 | 1552.36 | 5.31 | 0.0371 * |
| X2X3 | 1 | 6913.92 | 6913.92 | 23.63 | 0.0003 ** |
| X2X4 | 1 | 2.89 | 2.89 | 0.01 | 0.9222 |
| X3X4 | 1 | 189.06 | 189.06 | 0.65 | 0.4349 |
| X12 | 1 | 4174.94 | 4174.94 | 14.27 | 0.0020 ** |
| X22 | 1 | 4899.16 | 4899.16 | 16.75 | 0.0011 ** |
| X32 | 1 | 4146.19 | 4146.19 | 14.17 | 0.0021 ** |
| X42 | 1 | 246.93 | 246.93 | 0.84 | 0.3738 |
| Residual | 14 | 4095.80 | 292.56 | - | - |
| Lack of Fit | 10 | 3814.67 | 381.47 | 5.43 | 0.0587 |
| Pure Error | 4 | 281.13 | 70.28 | - | - |
| Total | 28 | 60,516.69 | - | - | - |
| R2 | 93.23% | ||||
| Adj R2 | 86.46% | ||||
| CV | 2.67% |
| Molecular Weight Distribution | Content (%) |
|---|---|
| >10.00 kDa | 1.45 |
| 5.00–10.00 kDa | 0.18 |
| 1.50–5.00 kDa | 18.02 |
| 0.50–1.50 kDa | 58.99 |
| 0.18–0.50 kDa | 20.81 |
| <0.18 kDa | 0.55 |
| Amino Acid Composition | MR | MRPI |
|---|---|---|
| Threonine * | 4.82 | 3.51 |
| Valine * | 5.68 | 6.93 |
| Methionine * | 1.59 | 1.65 |
| Isoleucine * | 4.09 | 4.69 |
| Leucine * | 9.19 | 10.99 |
| Phenylalanine * | 5.80 | 6.57 |
| Lysine b * | 5.50 | 5.28 |
| Histidine b | 1.77 | 0.63 |
| Arginine b | 5.62 | 5.85 |
| Aspartic acid a | 9.10 | 10.47 |
| Serine | 4.22 | 2.57 |
| Glutamic acid a | 11.63 | 14.40 |
| Proline | 4.69 | 5.00 |
| Glycine | 5.66 | 4.53 |
| Alanine | 7.69 | 7.94 |
| Cystine | 0.44 | 0.19 |
| Tyrosine | 3.53 | 3.65 |
| EAAI c | 1.43 | 1.49 |
| Protein Types | EA (%) | ES (%) | Emulsified Protein Content (%) |
|---|---|---|---|
| MRPI | 57 ± 1.2 | 55 ± 1.6 | 84 ± 2.7 |
| Soy protein isolate | 59 ± 1.8 | 58 ± 2.9 | 89 ± 3.2 |
| Na-caseinates | 56 ± 2.1 | 53 ± 3.1 | 72 ± 1.2 |
| Whey protein | 48 ± 0.9 | 48 ± 1.5 | 78 ± 2.5 |
| Egg white protein | 52 ± 2.3 | 48 ± 0.8 | 68 ± 1.9 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lu, K.; Zhao, X.; Ho, S.-H.; Ma, R.; Xie, Y.; Chen, J. Biorefining and the Functional Properties of Proteins from Lipid and Pigment Extract Residue of Chlorella pyrenoidosa. Mar. Drugs 2019, 17, 454. https://doi.org/10.3390/md17080454
Lu K, Zhao X, Ho S-H, Ma R, Xie Y, Chen J. Biorefining and the Functional Properties of Proteins from Lipid and Pigment Extract Residue of Chlorella pyrenoidosa. Marine Drugs. 2019; 17(8):454. https://doi.org/10.3390/md17080454
Chicago/Turabian StyleLu, Kongyong, Xurui Zhao, Shih-Hsin Ho, Ruijuan Ma, Youping Xie, and Jianfeng Chen. 2019. "Biorefining and the Functional Properties of Proteins from Lipid and Pigment Extract Residue of Chlorella pyrenoidosa" Marine Drugs 17, no. 8: 454. https://doi.org/10.3390/md17080454
APA StyleLu, K., Zhao, X., Ho, S.-H., Ma, R., Xie, Y., & Chen, J. (2019). Biorefining and the Functional Properties of Proteins from Lipid and Pigment Extract Residue of Chlorella pyrenoidosa. Marine Drugs, 17(8), 454. https://doi.org/10.3390/md17080454




