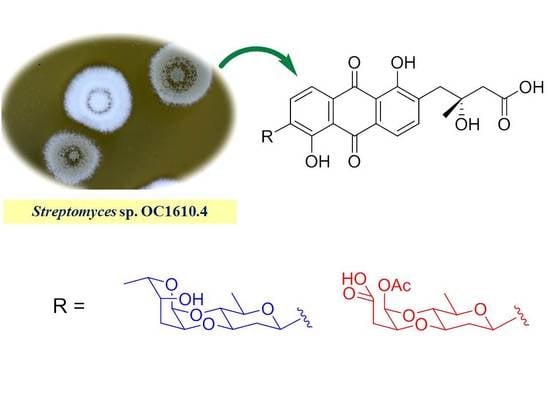
Cytotoxic, Anti-Migration, and Anti-Invasion Activities on Breast Cancer Cells of Angucycline Glycosides Isolated from a Marine-Derived Streptomyces sp.
Abstract
1. Introduction
2. Results and Discussion
3. Materials and Methods
3.1. General Experimental Procedures
3.2. Actinomycetes Strain
3.3. Fermentation, Extraction, and Isolation
3.4. Cell Culture
3.5. MTT Assay
3.6. Wound-Healing Assay
3.7. Transwell Assay
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Rohr, J.; Thiericke, R. Angucycline group antibiotics. Nat. Prod. Rep. 1992, 9, 103–137. [Google Scholar] [CrossRef] [PubMed]
- Kharel, M.K.; Pahari, P.; Shepherd, M.D.; Tibrewal, N.; Nybo, S.E.; Shaaban, K.A.; Rohr, J. Angucyclines: Biosynthesis, mode-of-action, new natural products, and synthesis. Nat. Prod. Rep. 2012, 29, 264–325. [Google Scholar] [CrossRef]
- He, H.Y.; Ding, W.D.; Bernan, V.S.; Richardson, A.D.; Ireland, C.M.; Greenstein, M.; Ellestad, G.A.; Carter, G.T. Lomaiviticins A and B, potent antitumor antibiotics from Micromonospora lomaivitiensis. J. Am. Chem. Soc. 2001, 123, 5362–5363. [Google Scholar] [CrossRef]
- Zhang, W.J.; Liu, Z.; Li, S.M.; Lu, Y.Z.; Chen, Y.C.; Zhang, H.B.; Zhang, G.T.; Zhu, Y.G.; Zhang, G.Y.; Zhang, W.M.; et al. Fluostatins I–K from the south China sea-derived Micromonospora rosaria SCSIO N160. J. Nat. Prod. 2012, 75, 1937–1943. [Google Scholar] [CrossRef]
- Wu, C.S.; van der Heul, H.U.; Melnik, A.V.; Lubben, J.; Dorrestein, P.C.; Minnaard, A.J.; Choi, Y.H.; van Wezel, G.P. Lugdunomycin, an angucycline-derived molecule with unprecedented chemical architecture. Angew. Chem. Int. Edit. 2019, 58, 2809–2814. [Google Scholar] [CrossRef] [PubMed]
- Shaaban, K.A.; Ahmed, T.A.; Leggas, M.; Rohr, J. Saquayamycins G-K, cytotoxic angucyclines from Streptomyces sp. including two analogues bearing the aminosugar rednose. J. Nat. Prod. 2012, 75, 1383–1392. [Google Scholar] [CrossRef]
- Vicente, J.; Stewart, A.K.; van Wagoner, R.M.; Elliott, E.; Bourdelais, A.J.; Wright, J.L.C. Monacyclinones, new angucyclinone metabolites isolated from Streptomyces sp. M7_15 associated with the Puerto Rican sponge Scopalina ruetzleri. Mar. Drugs 2015, 13, 4682–4700. [Google Scholar] [CrossRef]
- Zhu, X.C.; Duan, Y.W.; Cui, Z.M.; Wang, Z.; Li, Z.X.; Zhang, Y.; Ju, J.H.; Huang, H.B. Cytotoxic rearranged angucycline glycosides from deep sea-derived Streptomyces lusitanus SCSIO LR32. J. Antibiot. 2017, 70, 819–822. [Google Scholar] [CrossRef] [PubMed]
- Korynevska, A.; Heffeter, P.; Matselyukh, B.; Elbling, L.; Micksche, M.; Stoika, R.; Berger, W. Mechanisms underlying the anticancer activities of the angucycline landomycin E. Biochem Pharmacol. 2007, 74, 1713–1726. [Google Scholar] [CrossRef] [PubMed]
- Panchuk, R.R.; Lehka, L.V.; Terenzi, A.; Matselyukh, B.P.; Rohr, J.; Jha, A.K.; Downey, T.; Kril, I.J.; Herbacek, I.; van Schoonhoven, S.; et al. Rapid generation of hydrogen peroxide contributes to the complex cell death induction by the angucycline antibiotic landomycin E. Free Radical Bio. Med. 2017, 106, 134–147. [Google Scholar] [CrossRef] [PubMed]
- Hall, S.R.; Blundon, H.L.; Ladda, M.A.; Robertson, A.W.; Martinez-Farina, C.F.; Jakeman, D.L.; Goralski, K.B. Jadomycin breast cancer cytotoxicity is mediated by a copper-dependent, reactive oxygen species-inducing mechanism. Pharmacol. Res. Perspect. 2015, 3, e00110. [Google Scholar] [CrossRef]
- Hall, S.R.; Toulany, J.; Bennett, L.G.; Martinez-Farina, C.F.; Robertson, A.W.; Jakeman, D.L.; Goralski, K.B. Jadomycins inhibit type II topoisomerases and promote DNA damage and apoptosis in multidrug-resistant triple-negative breast cancer cells. J. Pharmacol. Exp. Ther. 2017, 363, 196–210. [Google Scholar] [CrossRef]
- Woo, C.M.; Beizer, N.E.; Janso, J.E.; Herzon, S.B. The cytotoxicity of (-)-lomaiviticin A arises from induction of double-strand breaks in DNA. Nat. Chem. 2014, 6, 504–510. [Google Scholar]
- Colis, L.C.; Hegan, D.C.; Kaneko, M.; Glazer, P.M.; Herzon, S.B. Mechanism of action studies of lomaiviticin A and the monomeric lomaiviticin aglycon. Selective and potent activity toward DNA double-strand break repair-deficient cell lines. J. Am. Chem. Soc. 2015, 137, 5741–5747. [Google Scholar] [CrossRef]
- Herzon, S.B. The mechanism of action of (-)-lomaiviticin A. Accounts Chem. Res. 2017, 50, 2577–2588. [Google Scholar] [CrossRef]
- Guo, L.; Xie, Z.P.; Yang, Q.; Feng, L.L.; Zhang, L.; Zhang, Y.Z.; Li, X.N.; Pescitelli, G.; Zhang, S.M. Kiamycins B and C, unusual bridged angucyclinones from a marine sediment-derived Streptomyces sp. Tetrahedron Lett. 2018, 59, 2176–2180. [Google Scholar] [CrossRef]
- Jin, J.; Yang, X.Y.; Liu, T.; Xiao, H.; Wang, G.Y.; Zhou, M.J.; Liu, F.W.; Zhang, Y.T.; Liu, D.; Chen, M.H.; et al. Fluostatins M-Q featuring a 6-5-6-6 ring skeleton and high oxidized A-rings from marine Streptomyces sp. PKU-MA00045. Mar. Drugs 2018, 16, 87. [Google Scholar] [CrossRef]
- Hu, Z.J.; Qin, L.L.; Wang, Q.Q.; Ding, W.J.; Chen, Z.; Ma, Z.J. Angucycline antibiotics and its derivatives from marine-derived actinomycete Streptomyces sp. A6H. Nat. Prod. Res. 2016, 30, 2551–2558. [Google Scholar] [CrossRef]
- Yang, L.; Hou, L.K.; Li, H.Y.; Li, W.L. Antibiotic angucycline derivatives from the deepsea-derived Streptomyces lusitanus. Nat. Prod. Res. 2019, 1–7. [Google Scholar] [CrossRef]
- Gui, C.; Liu, Y.N.; Zhou, Z.B.; Zhang, S.W.; Hu, Y.F.; Gu, Y.C.; Huang, H.B.; Ju, J.H. Angucycline glycosides from mangrove-derived Streptomyces diastaticus subsp. SCSIO GJ056. Mar. Drugs 2018, 16, 185. [Google Scholar] [CrossRef]
- Peng, A.H.; Qu, X.Y.; Liu, F.Y.; Li, X.; Li, E.W.; Xie, W.D. Angucycline glycosides from an intertidal sediments strain Streptomyces sp. and their cytotoxic activity against hepatoma carcinoma cells. Mar. Drugs 2018, 16, 470. [Google Scholar] [CrossRef]
- Danishefsky, S.J.; Biing, J.U.; Quallich, G. Total synthesis of vineomycin-B2 aglycon. J. Am. Chem. Soc. 1984, 106, 2453–2455. [Google Scholar] [CrossRef]
- Maskey, R.P.; Helmke, E.; Laatsch, H. Himalomycin A and B: Isolation and structure elucidation of new fridamycin type antibiotics from a marine Streptomyces isolate. J. Antibiot. 2003, 56, 942–949. [Google Scholar] [CrossRef]
- Abdelfattah, M.S.; Kharel, M.K.; Hitron, J.A.; Baig, I.; Rohr, J. Moromycins A and B, isolation and structure elucidation of C-glycosylangucycline-type antibiotics from Streptomyces sp. KY002. J. Nat. Prod. 2008, 71, 1569–1573. [Google Scholar] [CrossRef]
- Uchida, T.; Imoto, M.; Watanabe, Y.; Miura, K.; Dobashi, T.; Matsuda, N.; Sawa, T.; Naganawa, H.; Hamada, M.; Takeuchi, T.; et al. Saquayamycins, new aquayamycin-group antibiotics. J. Antibiot. 1985, 38, 1171–1181. [Google Scholar] [CrossRef]
- Huang, H.B.; Yang, T.T.; Ren, X.M.; Liu, J.; Song, Y.X.; Sun, A.J.; Ma, J.Y.; Wang, B.; Zhang, Y.; Huang, C.G.; et al. Cytotoxic angucycline class glycosides from the deep sea actinomycete Streptomyces lusitanus SCSIO LR32. J. Nat. Prod. 2012, 75, 202–208. [Google Scholar] [CrossRef]
- Kawashima, A.; Yoshimura, Y.; Goto, J.; Nakaike, S.; Mizutani, T.; Hanada, K.; Omura, S. Pi-083, a new platelet-aggregation inhibitor. J. Antibiot. 1988, 41, 1913–1914. [Google Scholar] [CrossRef][Green Version]
- Lafourcade, A.; His, M.; Baglietto, L.; Boutron-Ruault, M.C.; Dossus, L.; Rondeau, V. Factors associated with breast cancer recurrences or mortality and dynamic prediction of death using history of cancer recurrences: The French E3N cohort. Bmc Cancer 2018, 18, 171. [Google Scholar] [CrossRef]
- Narod, S.A.; Sopik, V. Is invasion a necessary step for metastases in breast cancer? Breast Cancer Res. Tr. 2018, 169, 9–23. [Google Scholar] [CrossRef]
- Zheng, B.B.; Wu, L.H.; Ma, L.S.; Liu, S.S.; Li, L.; Xie, W.D.; Li, X. Telekin induces apoptosis associated with the mitochondria-mediated pathway in human hepatocellular carcinoma cells. Biol. Pharm. Bull. 2013, 36, 1118–1125. [Google Scholar] [CrossRef]
- Ma, J.H.; Qi, J.; Liu, FY.; Lin, S.Q.; Zhang, C.Y.; Xie, W.D.; Zhang, H.Y.; Li, X. Ivalin inhibits proliferation, migration and invasion by suppressing epithelial mesenchymal transition in breast cancer cells. Nutr. Cancer 2018, 70, 1330–1338. [Google Scholar] [CrossRef] [PubMed]

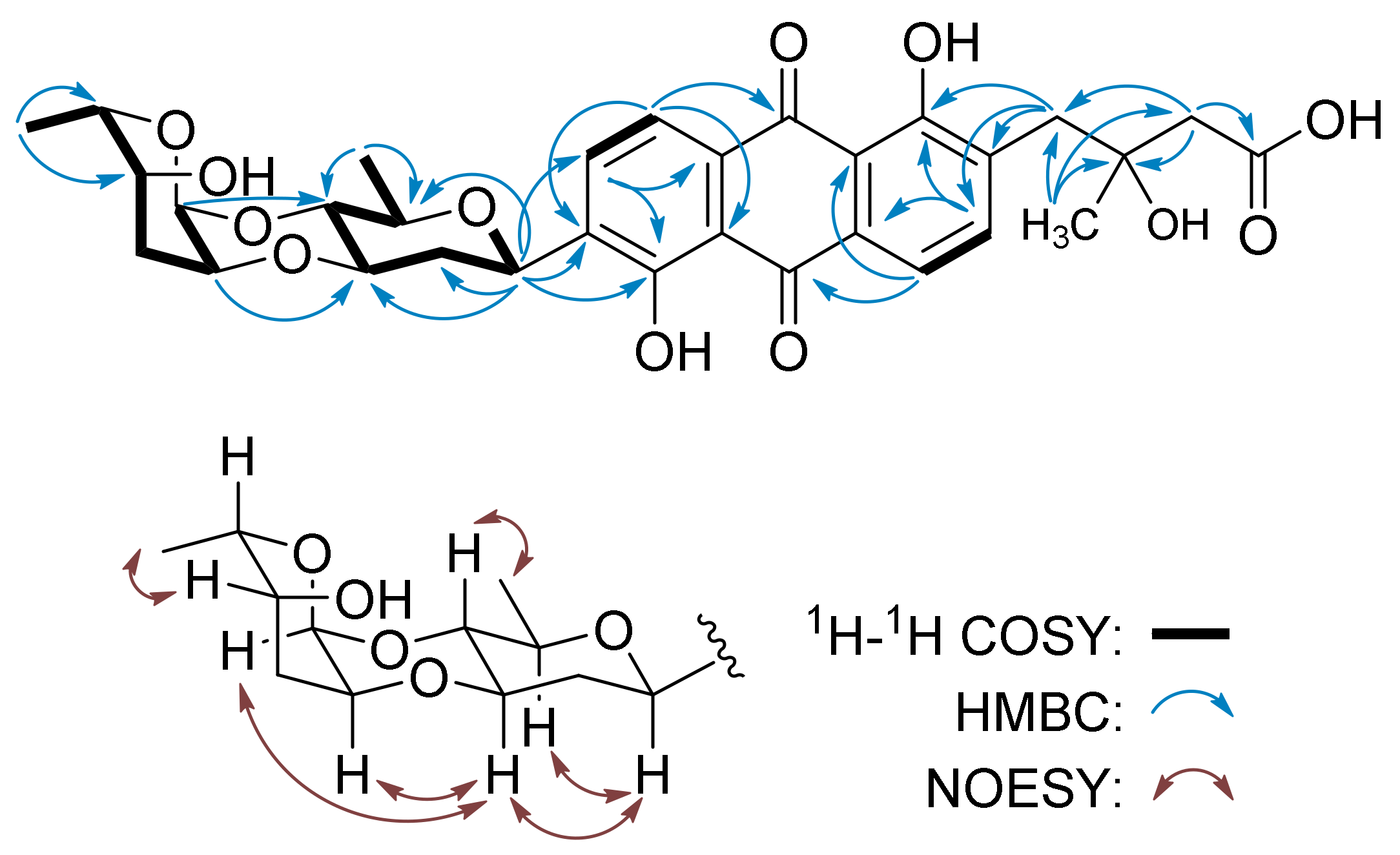
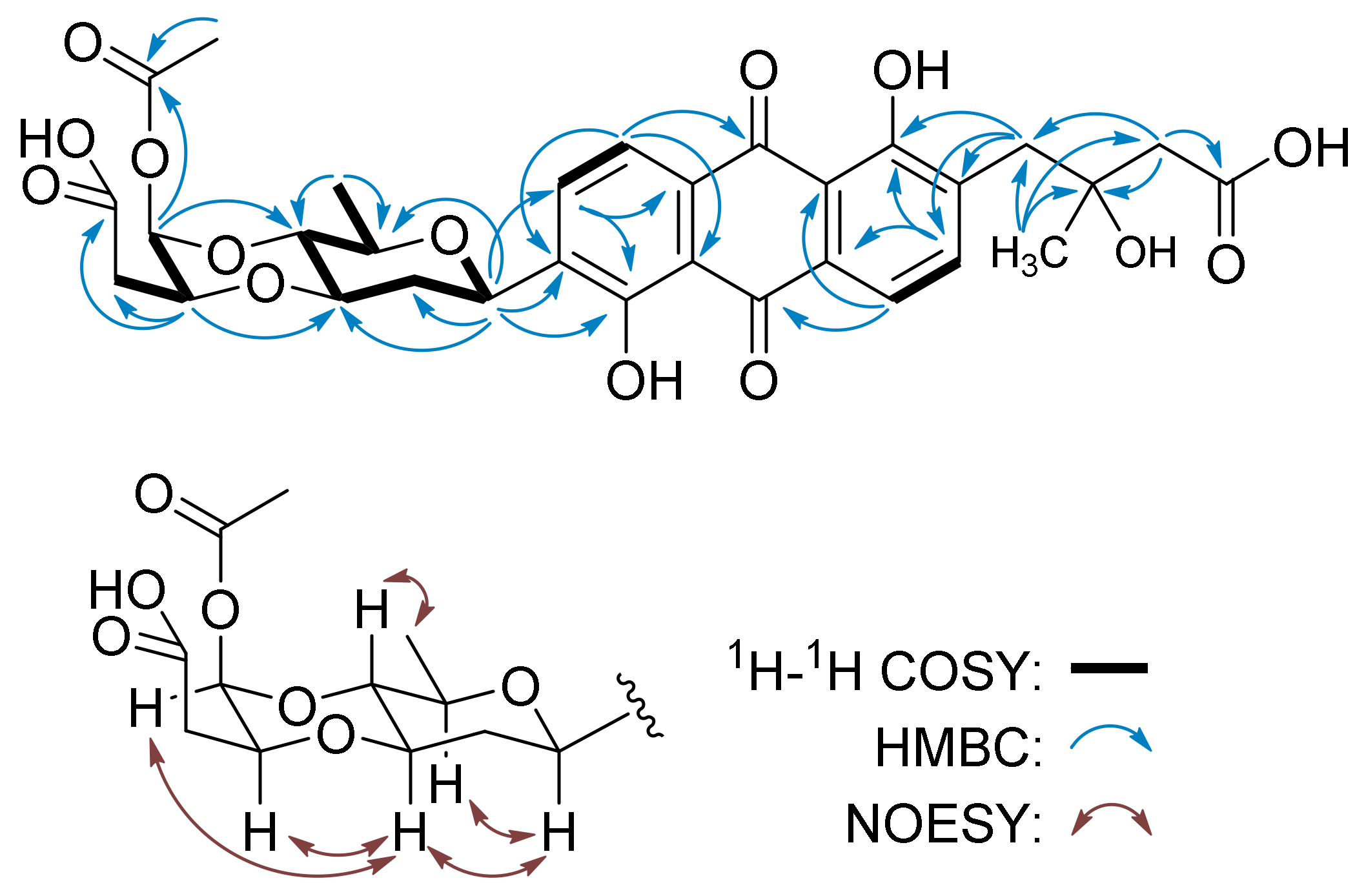

| No. | 1 b | 2 c | ||
|---|---|---|---|---|
| δC | δH | δC | δH | |
| 1 | 176.4 s | - | 173.8 s | - |
| 2 | 46.7 t | 2.48, d (15.5) 2.51, d (15.5) | 45.5 t | 2.56, d (15.2) 2.59, d (15.2) |
| 3 | 73.0 s | - | 72.1 s | - |
| 4 | 41.2 t | 3.01, d (13.1) 3.08, d (13.1) | 40.8 t | 3.10, brs |
| 4a | 136.3 s | - | 136.2 s | - |
| 5 | 140.8 d | 7.70, mc | 140.8 | 7.85, d (5.6) |
| 6 | 119.6 d | 7.68, mc | 119.3 d | 7.78, d (5.6) |
| 6a | 132.9 s | - | 132.5 s | - |
| 7 | 189.2 s | - | 189.1 s | - |
| 7a | 116.6 s | - | 116.3 s | - |
| 8 | 159.7 s | - | 159.6 s | - |
| 9 | 139.2 s | - | 138.8 s | - |
| 10 | 134.4 d | 7.84, d (7.3) | 134.2 d | 7.96, d (6.6) |
| 11 | 120.1 d | 7.72, d (7.3) | 119.8 d | 7.84, d (6.6) |
| 11a | 133.3 s | - | 133.0 s | - |
| 12 | 189.3 s | - | 189.2 s | - |
| 12a | 116.6 s | - | 116.4 s | - |
| 12b | 162.4 s | - | 162.3 s | - |
| 13 | 27.1 q | 1.27, s | 27.3 q | 1.31, s |
| Sugar A, β-d-olivose | ||||
| 1A | 72.7 d | 4.92, brd (11.4) | 72.2 d | 5.03, brd (9.1) |
| 2A | 37.9 t | 1.50, m2.38, m | 37.3 t | 1.56, m2.43, m |
| 3A | 78.0 d | 3.68, m | 77.5 d | 3.89, m |
| 4A | 75.1 d | 3.47, dd (9.3, 9.3) | 75.3 d | 3.46, dd (7.6, 7.8) |
| 5A | 75.6 d | 3.56, m | 74.6 d | 3.67, m |
| 6A | 17.9 q | 1.31, d (5.8) | 17.6 q | 1.26, d (5.6) |
| Sugar B | ||||
| 1B | 91.0 s | 4.97, brs | 90.5 d | 6.06, brs |
| 2B | 73.7 d | 3.88, m | 74.4 d | 4.34, m |
| 3B | 32.6 t | 1.92, m1.96, m | 35.9 d | 2.54, m |
| 4B | 64.1 d | 4.12, m | 171.4 s | - |
| 5B | 74.7 d | 4.24, m | 169.8 s | - |
| 6B | 11.5 q | 1.26, d (6.9) | 20.8 q | 2.17, s |
| Compounds | Cell lines | ||
|---|---|---|---|
| MCF-7 | MDA-MB-231 | BT-474 | |
| 1 | 6.07 ± 0.09 | 7.72 ± 0.76 | 4.27 ± 2.09 |
| 2 | >20 | >20 | >20 |
| 3 | >20 | >20 | >20 |
| 4 | >20 | >20 | >20 |
| 5 | 7.58 ± 1.19 | 8.01 ± 0.55 | 6.46 ± 1.92 |
| 6 | >20 | >20 | >20 |
| 7 | 0.42 ± 0.03 | 0.35 ± 0.03 | 0.67 ± 0.09 |
| 8 | 0.24 ± 0.01 | 0.16 ± 0.02 | 0.28 ± 0.09 |
| 9 | 0.40 ± 0.01 | 0.38 ± 0.04 | 0.41 ± 0.15 |
| 10 | >20 | >20 | >20 |
| 11 | >20 | >20 | >20 |
| Doxorubicin | 0.86 ± 0.64 | 1.30 ± 0.25 | 0.39 ± 0.06 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qu, X.-Y.; Ren, J.-W.; Peng, A.-H.; Lin, S.-Q.; Lu, D.-D.; Du, Q.-Q.; Liu, L.; Li, X.; Li, E.-W.; Xie, W.-D. Cytotoxic, Anti-Migration, and Anti-Invasion Activities on Breast Cancer Cells of Angucycline Glycosides Isolated from a Marine-Derived Streptomyces sp. Mar. Drugs 2019, 17, 277. https://doi.org/10.3390/md17050277
Qu X-Y, Ren J-W, Peng A-H, Lin S-Q, Lu D-D, Du Q-Q, Liu L, Li X, Li E-W, Xie W-D. Cytotoxic, Anti-Migration, and Anti-Invasion Activities on Breast Cancer Cells of Angucycline Glycosides Isolated from a Marine-Derived Streptomyces sp. Marine Drugs. 2019; 17(5):277. https://doi.org/10.3390/md17050277
Chicago/Turabian StyleQu, Xin-Ying, Jin-Wei Ren, Ai-Hong Peng, Shi-Qi Lin, Dan-Dan Lu, Qian-Qian Du, Ling Liu, Xia Li, Er-Wei Li, and Wei-Dong Xie. 2019. "Cytotoxic, Anti-Migration, and Anti-Invasion Activities on Breast Cancer Cells of Angucycline Glycosides Isolated from a Marine-Derived Streptomyces sp." Marine Drugs 17, no. 5: 277. https://doi.org/10.3390/md17050277
APA StyleQu, X.-Y., Ren, J.-W., Peng, A.-H., Lin, S.-Q., Lu, D.-D., Du, Q.-Q., Liu, L., Li, X., Li, E.-W., & Xie, W.-D. (2019). Cytotoxic, Anti-Migration, and Anti-Invasion Activities on Breast Cancer Cells of Angucycline Glycosides Isolated from a Marine-Derived Streptomyces sp. Marine Drugs, 17(5), 277. https://doi.org/10.3390/md17050277