Exploring the Antiangiogenic Potential of Solomonamide A Bioactive Precursors: In Vitro and In Vivo Evidences of the Inhibitory Activity of Solo F-OH During Angiogenesis
Abstract
1. Introduction
2. Results
2.1. Effects of Solomonamide A Analogues in Tubular-Like Structures’ Formation of Endothelial Cells
2.2. Solo F–OH Does Not Produce Disruption of Endothelial Tubular-Like Structures Formed on Matrigel
2.3. Solo F–OH Decreases the Migratory Potential of Endothelial Cells
2.4. Solo F–OH Inhibits the Invasive Capability of Endothelial Cells
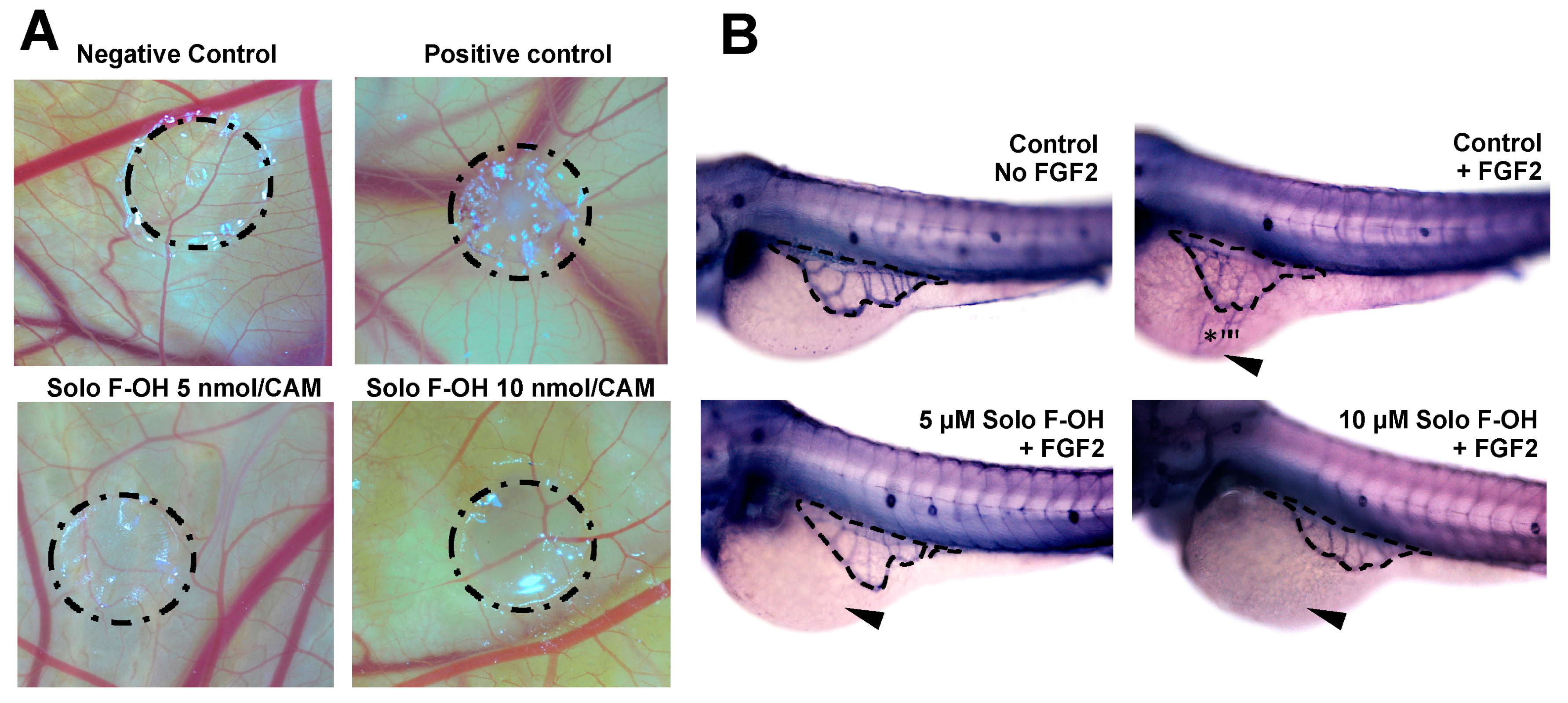
2.5. Solo F–OH Inhibits Angiogenesis In Vivo
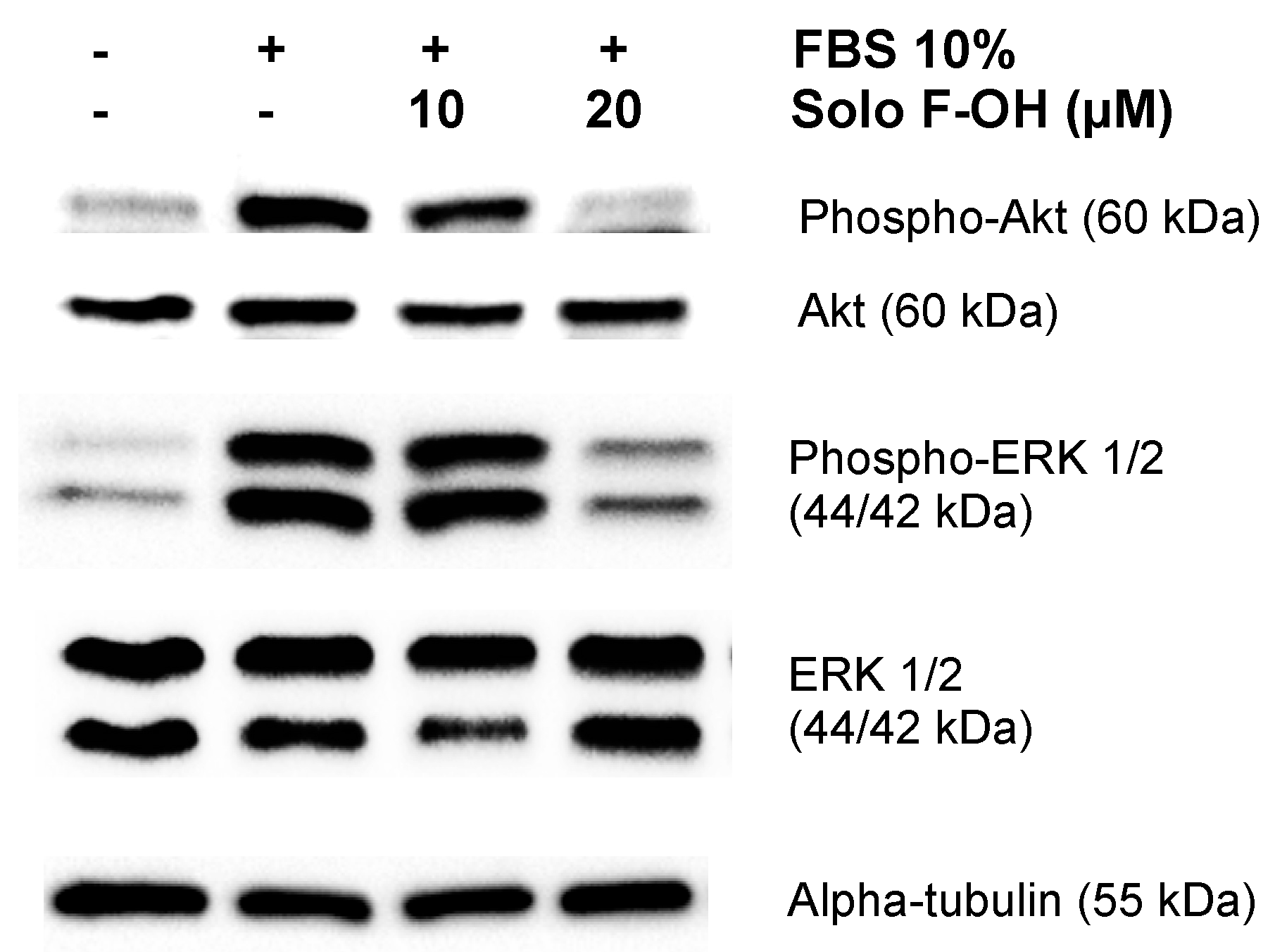
2.6. Solo F–OH Interferes with the Activation of ERK1/2 and Akt Pathways
2.7. Solo F–OH Does Not Inhibit the Tyrosine Kinase Activity of VEGFR2
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Cell Cultures
4.3. Tubular-Like Structures Formation on Matrigel
4.4. Wound Healing Assay
4.5. Cell Invasion Assay
4.6. Zymographic Assays for MMP-2 and MMP-9 Detection
4.7. Chick Chorioallantoic Membrane (CAM) Assay
4.8. FGF-2 Induced Angiogenesis Zebrafish Yolk Membrane (ZFYM) Assay
4.9. Western Blot Analysis
4.10. In Vitro Measure of VEGFR2 TK Activity
4.11. Ethical Statement
4.12. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ercolano, G.; De Cicco, P.; Ianaro, A. New Drugs from the Sea: Pro-Apoptotic Activity of Sponges and Algae Derived Compounds. Mar. Drugs 2019, 17, 31. [Google Scholar] [CrossRef]
- Zhang, H.; Dong, M.; Chen, J.; Wang, H.; Tenney, K.; Crews, P. Bioactive Secondary Metabolites from the Marine Sponge Genus Agelas. Mar. Drugs 2017, 15, 351. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Poveda, B.; Quesada, A.R.; Medina, M.Á. Pleiotropic Role of Puupehenones in Biomedical Research. Mar. Drugs 2017, 5, 325. [Google Scholar] [CrossRef]
- Calcabrini, C.; Catanzaro, E.; Bishayee, A.; Turrini, E.; Fimognari, C. Marine Sponge Natural Products with Anticancer Potential: An Updated Review. Mar. Drugs. 2017, 15, 310. [Google Scholar] [CrossRef] [PubMed]
- Ruocco, N.; Costantini, S.; Palumbo, F.; Costantini, M. Marine Sponges and Bacteria as Challenging Sources of Enzyme Inhibitors for Pharmacological Applications. Mar. Drugs 2017, 15, 173. [Google Scholar] [CrossRef]
- Máximo, P.; Ferreira, L.M.; Branco, P.; Lima, P.; Lourenço, A. The Role of Spongia sp. in the Discovery of Marine Lead Compounds. Mar. Drugs 2016, 14, 139. [Google Scholar] [CrossRef]
- García-Vilas, J.A.; Martínez-Poveda, B.; Quesada, A.R.; Medina, M.Á. Aeroplysinin-1, a Sponge-Derived Multi-Targeted Bioactive Marine Drug. Mar. Drugs 2015, 14, 1. [Google Scholar] [CrossRef] [PubMed]
- Festa, C.; De Marino, S.; Sepe, V.; Monti, M.; Luciano, P.; D’Auria, M.V.; Débitus, C.; Bucci, M.; Vellecco, V.; Zampella, A. Perthamides C and D, two new potent anti-inflammatory cyclopeptides from a Solomon Lithistid sponge Theonella swinhoei. Tetrahedron 2009, 65, 10424–10429. [Google Scholar] [CrossRef]
- Festa, C.; De Marino, S.; D’Auria, M.V.; Monti, M.; Bucci, M.; Velleco, V.; Débitus, C.; Zampella, A. Anti-inflammatory cyclopeptides from the marine sponge Theonella swinhoei. Tetrahedron 2012, 68, 2851–2857. [Google Scholar] [CrossRef]
- Carmely, S.; Kashman, Y. Structure of swinholide-A, a new macrolide from the marine sponge Theonella swinhoei. Tetrahedron Lett. 1985, 26, 511–514. [Google Scholar] [CrossRef]
- De Marino, S.; Festa, C.; D’Auria, M.V.; Cresteil, T.; Débitus, C.; Zampella, A. Swinholide J, a potent cytotoxin from the marine sponge Theonella swinhoei. Mar. Drugs. 2011, 9, 1133–1141. [Google Scholar] [CrossRef]
- Festa, C.; De Marino, S.; Sepe, V.; D’Auria, M.V.; Bifulco, G.; Débitus, C.; Bucci, M.; Vellecco, V.; Zampella, A. Solomonamides A and B, new anti-inflammatory peptides from Theonella swinhoei. Org. Lett. 2011, 13, 1532–1535. [Google Scholar] [CrossRef] [PubMed]
- Jachak, G.R.; Athawale, P.R.; Agarwal, H.; Barthwal, M.K.; Lauro, G.; Bifulco, G.; Reddy, D.S. Total synthesis of the potent anti-inflammatory natural product solomonamide A along with structural revision and biological activity evaluation. Org. Biomol. Chem. 2018, 16, 9138–9142. [Google Scholar] [CrossRef]
- Thapa, P.; Espiritu, M.J.; Cabalteja, C.; Bingham, J.P. The emergence of cyclic peptides: The potential of bioengineered peptide drugs. Int. J. Pept. Res. Ther. 2014, 20, 545–551. [Google Scholar] [CrossRef]
- Kavitha, N.; Chandrasekhar, S. Scalable synthesis of the unusual amino acid segment (ADMOA unit) of marine anti-inflammatory peptide: Solomonamide A. Org. Biomol. Chem. 2015, 13, 6242–6248. [Google Scholar] [CrossRef]
- Newman, D.J. Developing natural product drugs: Supply problems and how they have been overcome. Pharmacol. Ther. 2016, 162, 1–9. [Google Scholar] [CrossRef]
- Cheng-Sánchez, I.; Carrillo, P.; Sánchez-Ruiz, A.; Martínez-Poveda, B.; Quesada, A.R.; Medina, M.Á.; López-Romero, J.M.; Sarabia, F. Exploring the Ring-Closing Metathesis for the Construction of the Solomonamide Macrocyclic Core: Identification of Bioactive Precursors. J. Org. Chem. 2018, 83, 5365–5383. [Google Scholar] [CrossRef]
- Potente, M.; Gerhardt, H.; Carmeliet, P. Basic and therapeutic aspects of angiogenesis. Cell 2011, 146, 873–887. [Google Scholar] [CrossRef]
- Quesada, A.R.; Medina, M.Á.; Muñoz-Chápuli, R.; Ponce, Á.L. Do not say ever never more: The ins and outs of antiangiogenic therapies. Curr. Pharm. Des. 2010, 16, 3932–3957. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef]
- Folkman, J. Tumor angiogenesis: Therapeutic implications. N. Engl. J. Med. 1971, 285, 1182–1186. [Google Scholar] [CrossRef]
- Folkman, J. Angiogenesis: An organizing principle for drug discovery? Nat. Rev. Drug Discov. 2007, 6, 273–286. [Google Scholar] [CrossRef]
- Carmeliet, P. Angiogenesis in life, disease and medicine. Nature 2005, 438, 932–936. [Google Scholar] [CrossRef]
- López-Jiménez, A.; Gallardo, E.; Espartero, J.L.; Madrona, A.; Quesada, A.R.; Medina, M.Á. Comparison of the anti-angiogenic potential of hydroxytyrosol and five derivatives. Food Funct. 2018, 9, 4310–4316. [Google Scholar] [CrossRef]
- García-Vilas, J.A.; Pino-Ángeles, A.; Martínez-Poveda, B.; Quesada, A.R.; Medina, M.Á. The noni anthraquinone damnacanthal is a multi-kinase inhibitor with potent anti-angiogenic effects. Cancer lett. 2017, 385, 1–11. [Google Scholar] [CrossRef]
- García-Caballero, M.; Cañedo, L.; Fernández-Medarde, A.; Medina, M.Á.; Quesada, A.R. The marine fungal metabolite, AD0157, inhibits angiogenesis by targeting the Akt signaling pathway. Mar. Drugs 2014, 12, 279–299. [Google Scholar] [CrossRef]
- García-Caballero, M.; Marí-Beffa, M.; Cañedo, L.; Medina, M.Á.; Quesada, A.R. Toluquinol, a marine fungus metabolite, is a new angiosuppresor that interferes with the Akt pathway. Biochem. Pharmacol. 2013, 85, 1727–1740. [Google Scholar] [CrossRef]
- García-Vilas, J.A.; Quesada, A.R.; Medina, M.Á. 4-methylumbelliferone inhibits angiogenesis in vitro and in vivo. J. Agric. Food Chem. 2013, 61, 4063–4071. [Google Scholar] [CrossRef]
- Muñoz-Cápuli, R.; Quesada, A.R.; Medina, M.Á. Angiogenesis and signal transduction in endothelial cells. Cell. Mol. Life Sci. 2004, 61, 2224–2243. [Google Scholar] [CrossRef]
- García-Caballero, M.; Quesada, A.R.; Medina, M.Á.; Marí-Beffa, M. Fishing anti(lymph)angiogenic drugs with zebrafish. Drug Discov. Today 2018, 23, 366–374. [Google Scholar] [CrossRef]
- Mendel, D.B.; Laird, A.D.; Xin, X.; Louie, S.G.; Christensen, J.G.; Li, G.; Schreck, R.E.; Abrams, T.J.; Ngai, T.J.; Lee, L.B.; et al. In vivo antitumor activity of SU11248, a novel tyrosine kinase inhibitor targeting vascular endothelial growth factor and platelet-derived growth factor receptors: Determination of a pharmacokinetic/pharmacodynamics relationship. Clin. Cancer Res. 2003, 9, 327–337. [Google Scholar] [PubMed]
- Sun, L.; Liang, C.; Shirazian, S.; Zhou, Y.; Miller, T.; Cui, J.; Fukuda, J.Y.; Chu, J.Y.; Nematalla, A.; Wang, X.; et al. Discovery of 5-[fluoro-2-oxo-1,2-dihydroindol-(3Z)-ylidenemethyl]-2,4-dimethil-1H -pyrrole-3-carboxylic acid (2-diethylaminoethyl)amide, a novel tyrosine kinase inhibitor targeting vascular endothelial and plateled-derived growth factor receptor tyrosine kinase. J. Med. Chem. 2003, 46, 1116–1119. [Google Scholar]
- Keyzers, R.A.; Davies-Coleman, M.T. Anti-inflammatory metabolites from marine sponges. Chem. Soc. Rev. 2005, 34, 355–365. [Google Scholar] [CrossRef]
- Laport, M.S.; Santos, O.C.; Muricy, G. Marine sponges: Potential sources of new antimicrobial drugs. Curr. Pharm. Biotechnol. 2009, 10, 86–105. [Google Scholar] [CrossRef]
- Sharma, S.; Guru, S.K.; Manda, S.; Kumar, A.; Mintoo, M.J.; Prasad, V.D.; Sharma, P.R.; Mondhe, D.M.; Bharate, S.B.; Bhushan, S. A marine sponge alkaloid derivative 4-chloro fascaplysin inhibits tumor growth and VEGF mediated angiogenesis by disrupting PI3K/Akt/mTOR signaling cascade. Chem. Biol. Interact. 2017, 275, 47–60. [Google Scholar] [CrossRef]
- Senthilkumar, K.; Venkatesan, J.; Manivasagan, P.; Kim, S.K. Antiangiogenic effects of marine sponge derived compounds on cancer. Environ. Toxicol. Pharmacol. 2013, 36, 1097–1108. [Google Scholar] [CrossRef]
- Kashinath, K.; Vasudevan, N.; Reddy, D.S. Studies toward the synthesis of potent anti-inflammatory peptides solomonamides A and B: Synthesis of a macrocyclic skeleton and key fragment 4-amino-6-(2′-amino-4′-hydroxyphenyl)-3-hydroxy-2-methyl-6-oxohexanoic acid (AHMOA). Org. Lett. 2012, 14, 6222–6225. [Google Scholar] [CrossRef]
- Vasudevan, N.; Kashinath, K.; Reddy, D.S. Total synthesis of Deoxy-solomonamide B by mimicking biogenesis. Org. Lett. 2014, 16, 6148–6151. [Google Scholar] [CrossRef] [PubMed]
- Kashinath, K.; Dhara, S.; Reddy, D.S. Breaking and making of olefins simultaneously using ozonolysis: Application to the synthesis of useful building blocks and macrocyclic core of solomonamides. Org. Lett. 2015, 17, 2090–2093. [Google Scholar] [CrossRef]
- Kashinath, K.; Jachak, G.R.; Athawale, P.R.; Marelli, U.K.; Gonnade, R.G.; Reddy, D.S. Total Synthesis of the Marine Natural Product Solomonamide B Necessitates Stereochemical Revision. Org. Lett. 2016, 18, 3178–3181. [Google Scholar] [CrossRef]
- Cheng-Sánchez, I.; García-Ruiz, C.; Sarabia, F. An Olefin Metathesis Approach towards the Solomonamides. Tetrahedron Lett. 2016, 57, 3392–3395. [Google Scholar] [CrossRef]
- Tozer, G.M.; Kanthou, C.; Baguley, B.C. Disrupting tumour blood vessels. Nat. Rev. Cancer 2005, 5, 423–435. [Google Scholar] [CrossRef]
- Egeblad, M.; Werb, Z. New functions for the matrix metalloproteinases in cancer progression. Nat Rev. Cancer 2002, 2, 161–174. [Google Scholar] [CrossRef]
- Handsley, M.M.; Edwards, D.R. Metalloproteinases and their inhibitors in tumor angiogenesis. Int. J. Cancer 2005, 115, 849–860. [Google Scholar] [CrossRef]
- Van Hinsbergh, V.W.; Engelse, M.A.; Quax, P.H. Pericellular proteases in angiogenesis and vasculogenesis. Arterioscler. Thromb. Vasc. Biol. 2006, 26, 716–728. [Google Scholar] [CrossRef]
- Elkin, M.; Miao, H.Q.; Nagler, A.; Aingorn, E.; Reich, R.; Hemo, I.; Dou, H.L.; Pines, M.; Vlodavsky, I. Halofuginone: A potent inhibitor of critical steps in angiogenesis progression. FASEB J. 2000, 14, 2477–2485. [Google Scholar] [CrossRef]
- Rodríguez-Nieto, S.; González-Iriarte, M.; Carmona, R.; Muñoz-Chápuli, R.; Medina, M.Á.; Quesada, A.R. Antiangiogenic activity of aeroplysinin-1, a brominated compound isolated from a marine sponge. FASEB J. 2002, 16, 261–263. [Google Scholar] [CrossRef]
- Philip, S.; Kundu, G.C. Osteopontin induces nuclear factor kappa B-mediated promatrix metalloproteinase-2 activation through I kappa B alpha /IKK signaling pathways, and curcumin (diferulolylmethane) down-regulates these pathways. J. Biol. Chem. 2003, 278, 14487–14497. [Google Scholar] [CrossRef]
- Castro, M.E.; González-Iriarte, M.; Barrero, A.F.; Salvador-Tormo, N.; Muñoz-Chápuli, R.; Medina, M.A.; Quesada, A.R. Study of puupehenone and related compounds as inhibitors of angiogenesis. Int. J. Cancer 2004, 110, 31–38. [Google Scholar] [CrossRef]
- Martínez-Poveda, B.; Quesada, A.R.; Medina, M.Á. Hyperforin, a bio-active compound of St. John’s wort, is a new inhibitor of angiogenesis targeting several key steps of the process. Int. J. Cancer 2005, 117, 775–780. [Google Scholar] [CrossRef]
- Cárdenas, C.; Quesada, A.R.; Medina, M.Á. Evaluation of the anti-angiogenic effect of aloe-emodin. Cell. Mol. Life Sci. 2006, 63, 3083–3089. [Google Scholar] [CrossRef]
- Cárdenas, C.; Quesada, A.R.; Medina, M.Á. Anti-angiogenic and anti-inflammatory properties of kahweol, a coffee diterpene. PLoS ONE 2011, 6, e23407. [Google Scholar] [CrossRef]
- Garcia-Caballero, M.; Mari-Beffa, M.; Medina, M.Á.; Quesada, A.R. Dimethylfumarate inhibits angiogenesis in vitro and in vivo: A possible role for its antipsoriatic effect? J. Investig. Dermatol. 2011, 131, 1347–1355. [Google Scholar] [CrossRef]
- Shiojima, I.; Walsh, K. Role of Akt signaling in vascular homeostasis and angiogenesis. Circ. Res. 2002, 90, 1243–1250. [Google Scholar] [CrossRef] [PubMed]
- Somanath, P.R.; Razorenova, O.V.; Chen, J.; Byzova, T.V. Akt1 in endotelial cell and angiogenesis. Cell Cycle 2006, 5, 512–518. [Google Scholar] [CrossRef]
- Faivre, S.; Demetri, G.; Sargent, W.; Raymond, E. Molecular basis for sunitinib efficacy and future clinical development. Nat. Rev. Drug Discov. 2007, 6, 734–745. [Google Scholar] [CrossRef] [PubMed]
- Westerfield, M. The Zebrafish Book. A Guide for the Laboratory Use of Zebrafish (Danio rerio), 3rd ed.; University of Oregon Press: Eugene, OR, USA, 1995. [Google Scholar]
- Oikawa, T.; Shimamura, M.; Ashing, H.; Nakamura, O.; Kanayasu, T.; Morita, I.; Murota, S.I. Inhibition of angiogenesis by staurosporine, a potent protein kinase inhibitor. J. Antibiot. (Tokyo) 1992, 45, 1155–1160. [Google Scholar] [CrossRef] [PubMed]
- Vincent, L.; Kermani, P.; Young, L.M.; Cheng, J.; Zhang, F.; Shido, K.; Lam, G.; Bompais-Vincent, H.; Zhu, Z.; Hicklin, D.J.; et al. Combretastatin A4 phosphate induces rapid regression of tumor neovessels and growth through interference with vascular endothelial-cadherin signaling. J. Clin. Investig. 2005, 115, 2992–3006. [Google Scholar] [CrossRef]
- Fajardo, I.; Quesada, A.R.; Núñez de Castro, I.; Sánchez-Jiménez, F.; Medina, M.Á. A comparative study of the effects of genistein and 2-methoxyestradiol on the proteolytic balance and tumour cell proliferation. Br. J. Cancer 1999, 80, 17–24. [Google Scholar] [CrossRef][Green Version]
- Nicoli, S.; De Sena, G.; Presta, M. Fibroblast growth factor 2-induced angiogenesis in zebrafish: The zebrafish yolk membrane (ZFYM) angiogenesis assay. J. Cell. Mol. Med. 2009, 13, 2061–2068. [Google Scholar] [CrossRef]
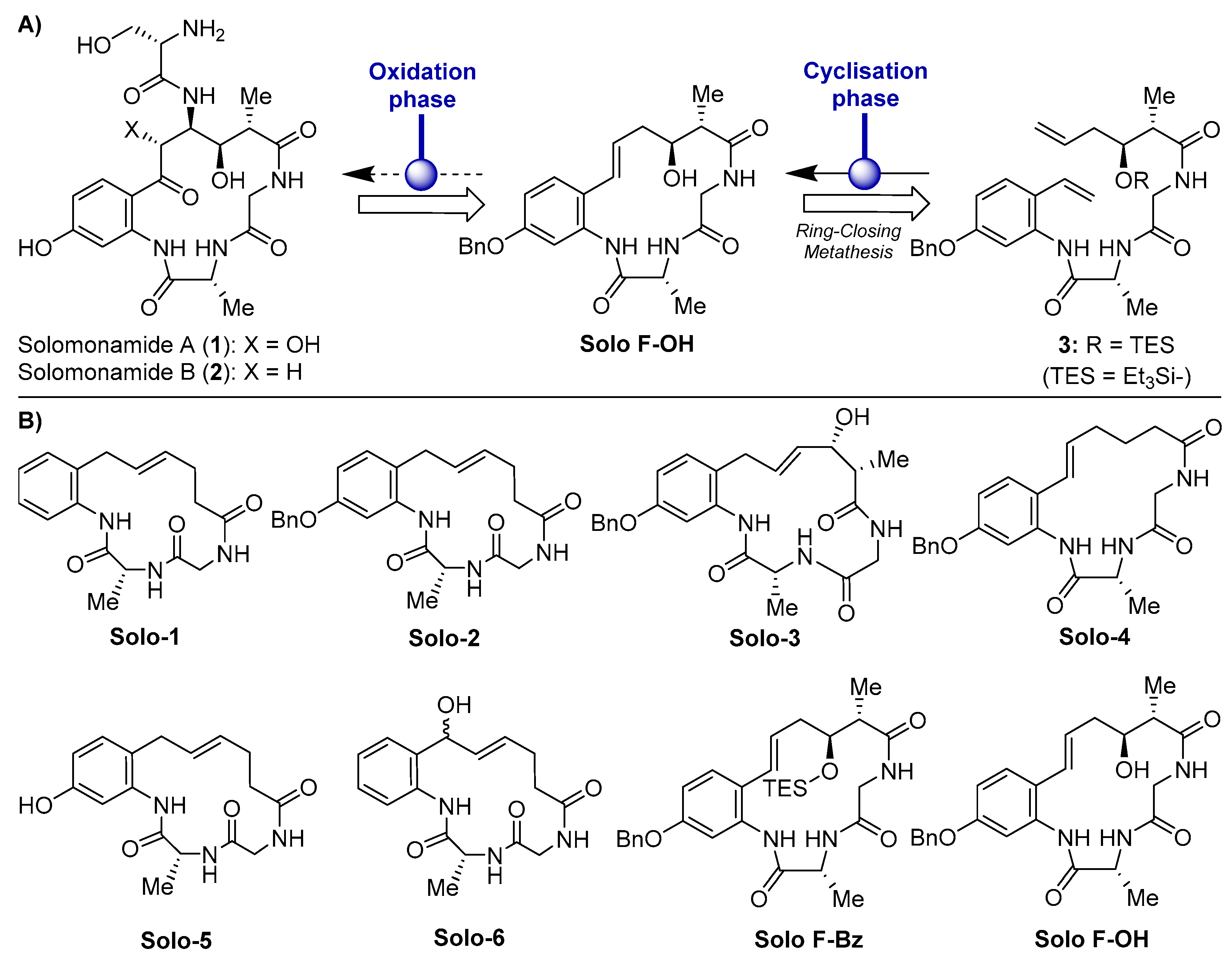
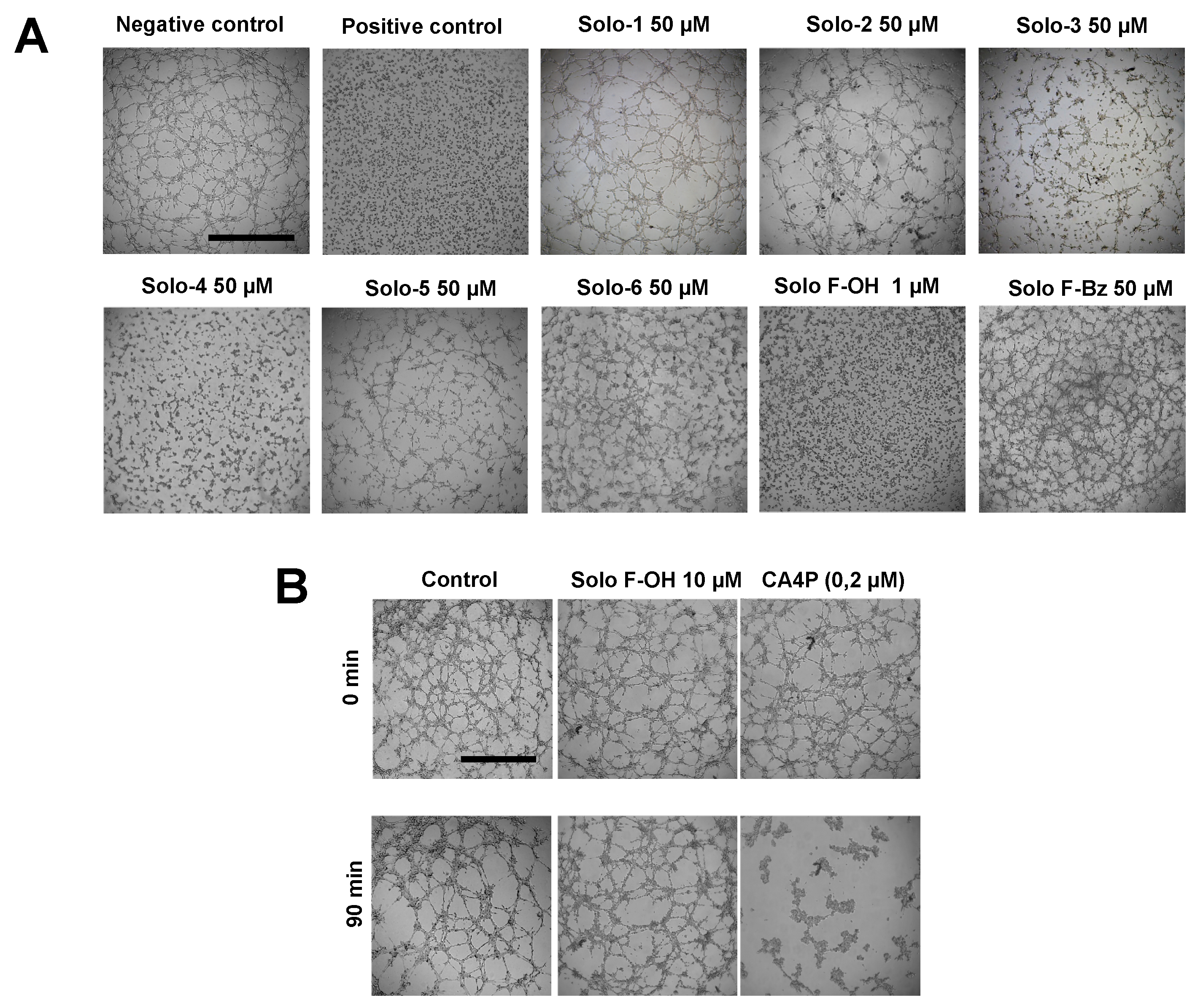
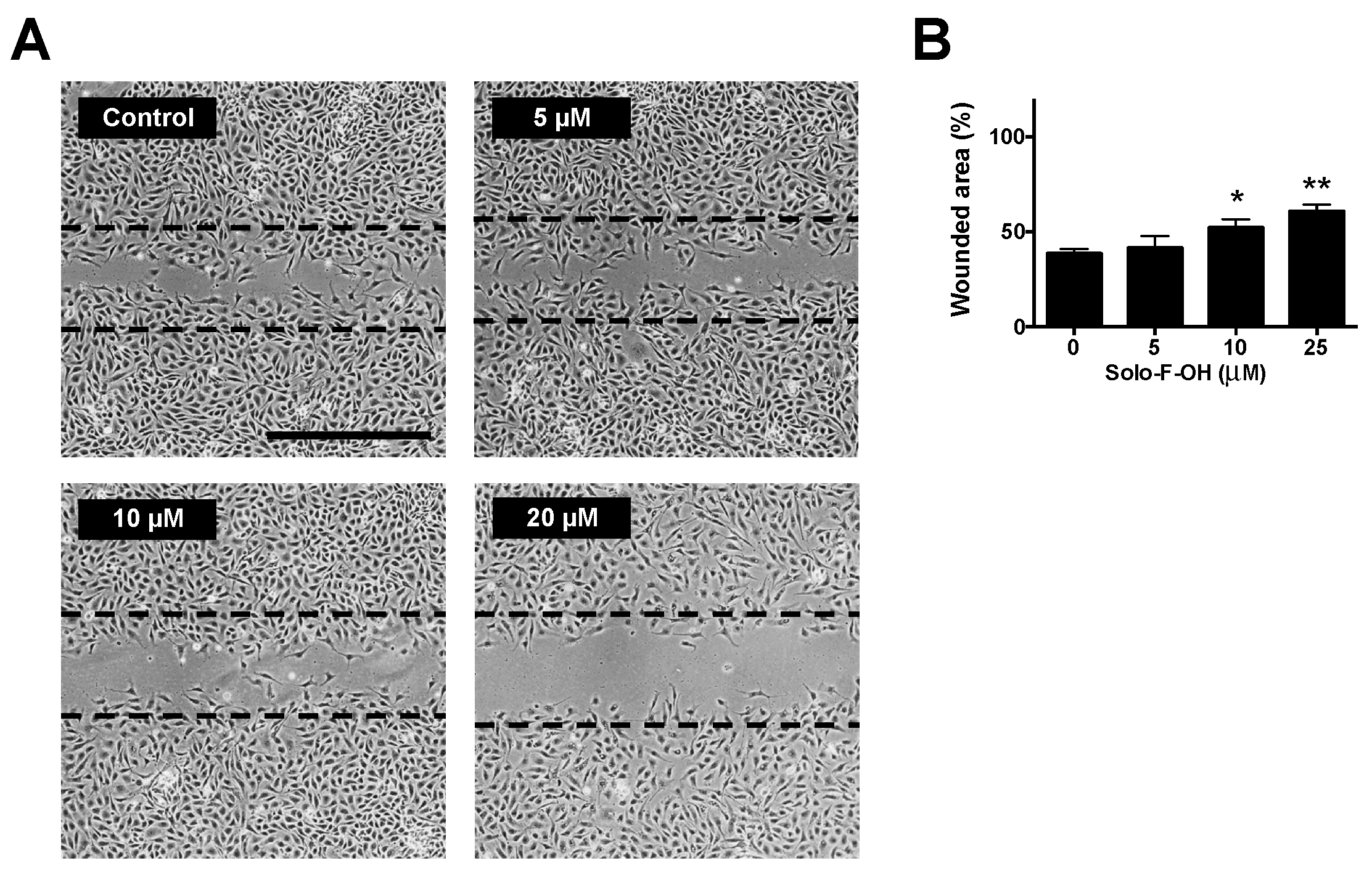
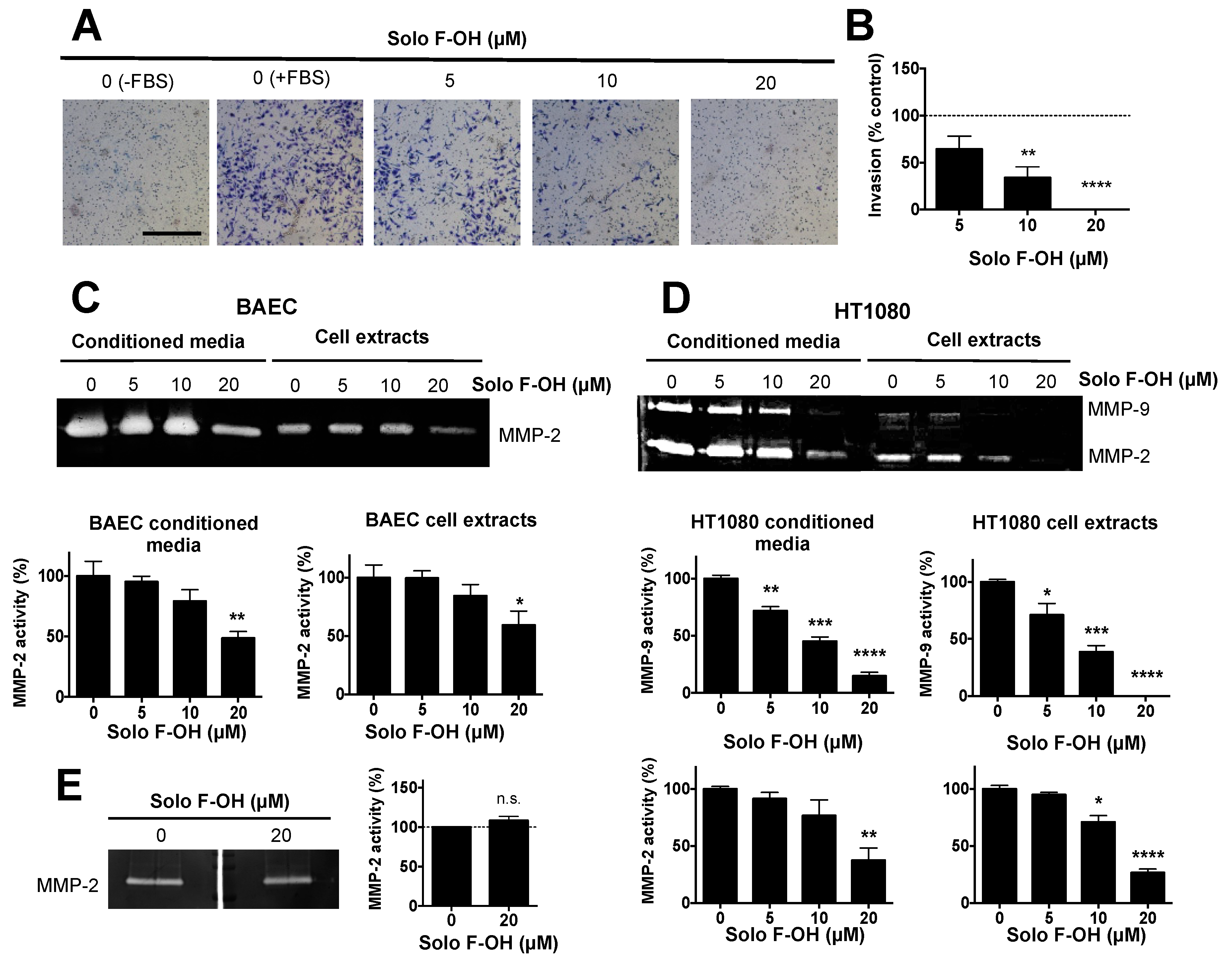
| Compound | IC50 1 (μM) in BAEC [17] | MIC Tubular Like-Structures Formation (μM) | MIC Wound-Healing Assay (μM) |
|---|---|---|---|
| Solo-1 | >100 | >50 | >50 |
| Solo-2 | 43.8 ± 1.2 | >50 | >50 |
| Solo-3 | >100 | >50 | >50 |
| Solo-4 | 69.6 ± 12.5 | 50 | >50 |
| Solo-5 | >100 | >50 | >50 |
| Solo-6 | >100 | >50 | >50 |
| Solo F–OH | 18.1 ± 2.2 | 1 | 10 |
| Solo F–Bz | >100 | >50 | >50 |
| CAM Assay | ||
|---|---|---|
| Solo F–OH (nmol/CAM) | Positive/Total | % Inhibition |
| 0 | 0/11 | 0 |
| 0.1 | 1/7 | 14 |
| 0.5 | 2/7 | 29 |
| 1 | 6/9 | 67 |
| 5 | 9/12 | 75 |
| 10 | 10/10 | 100 |
| ZFYM Assay | ||||
|---|---|---|---|---|
| FGF-2 Induction | Solo F–OH (µM) | Score (%) | ||
| – / Total(%) | + / Total(%) | ++ / Total(%) | ||
| None | 0 | 20/20 (100) | 0/20 (0) | 0/20 (0) |
| 2 ng | 0 | 5/21 (23.8) | 10/21 (47.6) | 6/21 (28.6) |
| 2 ng | 5 | 9/19 (47.4) | 7/19 (36.8) | 3/19 (15.8) |
| 2 ng | 10 | 13/23 (56.5) | 9/23 (39.1) | 1/23 (4.3) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carrillo, P.; Martínez-Poveda, B.; Cheng-Sánchez, I.; Guerra, J.; Tobia, C.; López-Romero, J.M.; Sarabia, F.; Medina, M.Á.; Quesada, A.R. Exploring the Antiangiogenic Potential of Solomonamide A Bioactive Precursors: In Vitro and In Vivo Evidences of the Inhibitory Activity of Solo F-OH During Angiogenesis. Mar. Drugs 2019, 17, 228. https://doi.org/10.3390/md17040228
Carrillo P, Martínez-Poveda B, Cheng-Sánchez I, Guerra J, Tobia C, López-Romero JM, Sarabia F, Medina MÁ, Quesada AR. Exploring the Antiangiogenic Potential of Solomonamide A Bioactive Precursors: In Vitro and In Vivo Evidences of the Inhibitory Activity of Solo F-OH During Angiogenesis. Marine Drugs. 2019; 17(4):228. https://doi.org/10.3390/md17040228
Chicago/Turabian StyleCarrillo, Paloma, Beatriz Martínez-Poveda, Iván Cheng-Sánchez, Jessica Guerra, Chiara Tobia, J. Manuel López-Romero, Francisco Sarabia, Miguel Ángel Medina, and Ana R. Quesada. 2019. "Exploring the Antiangiogenic Potential of Solomonamide A Bioactive Precursors: In Vitro and In Vivo Evidences of the Inhibitory Activity of Solo F-OH During Angiogenesis" Marine Drugs 17, no. 4: 228. https://doi.org/10.3390/md17040228
APA StyleCarrillo, P., Martínez-Poveda, B., Cheng-Sánchez, I., Guerra, J., Tobia, C., López-Romero, J. M., Sarabia, F., Medina, M. Á., & Quesada, A. R. (2019). Exploring the Antiangiogenic Potential of Solomonamide A Bioactive Precursors: In Vitro and In Vivo Evidences of the Inhibitory Activity of Solo F-OH During Angiogenesis. Marine Drugs, 17(4), 228. https://doi.org/10.3390/md17040228







