Dietary Supplementation with the Red Seaweed Porphyra umbilicalis Protects against DNA Damage and Pre-Malignant Dysplastic Skin Lesions in HPV-Transgenic Mice
Abstract
:1. Introduction
2. Results
2.1. General Findings
2.2. HPV-Induced Lesions and Hepatic Histology
2.3. Serum Biochemical Parameters
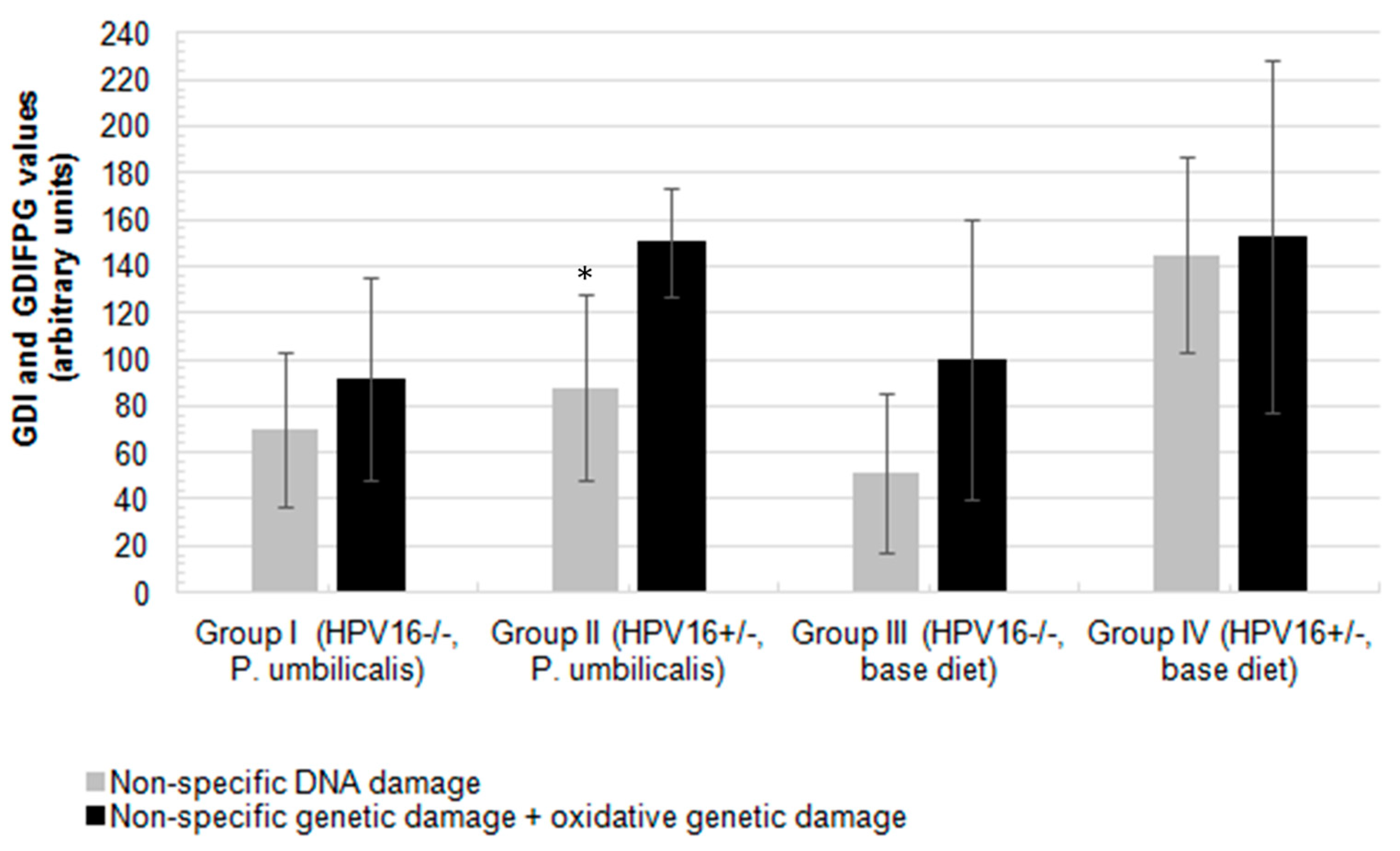
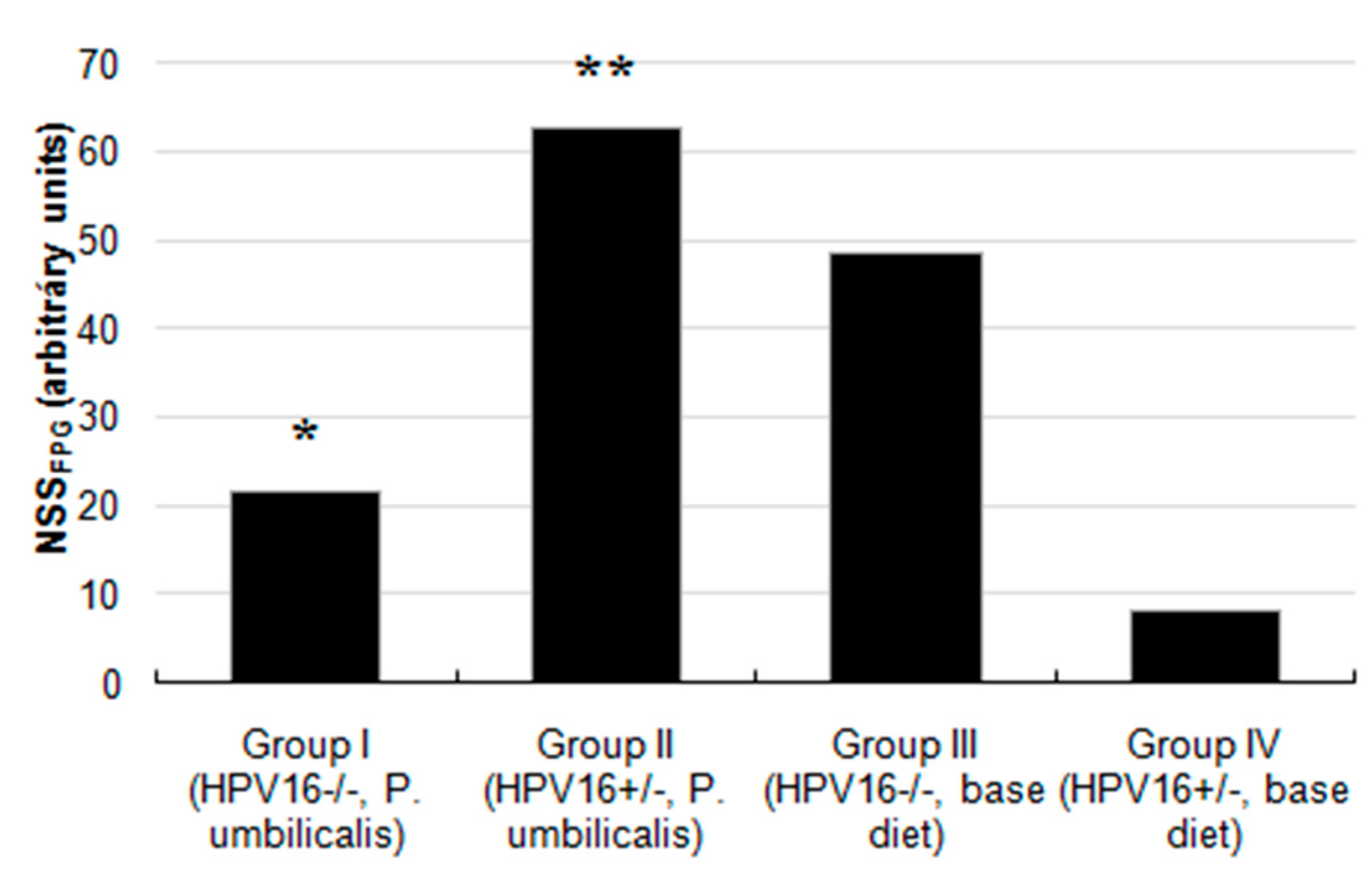
2.4. Comet Assay
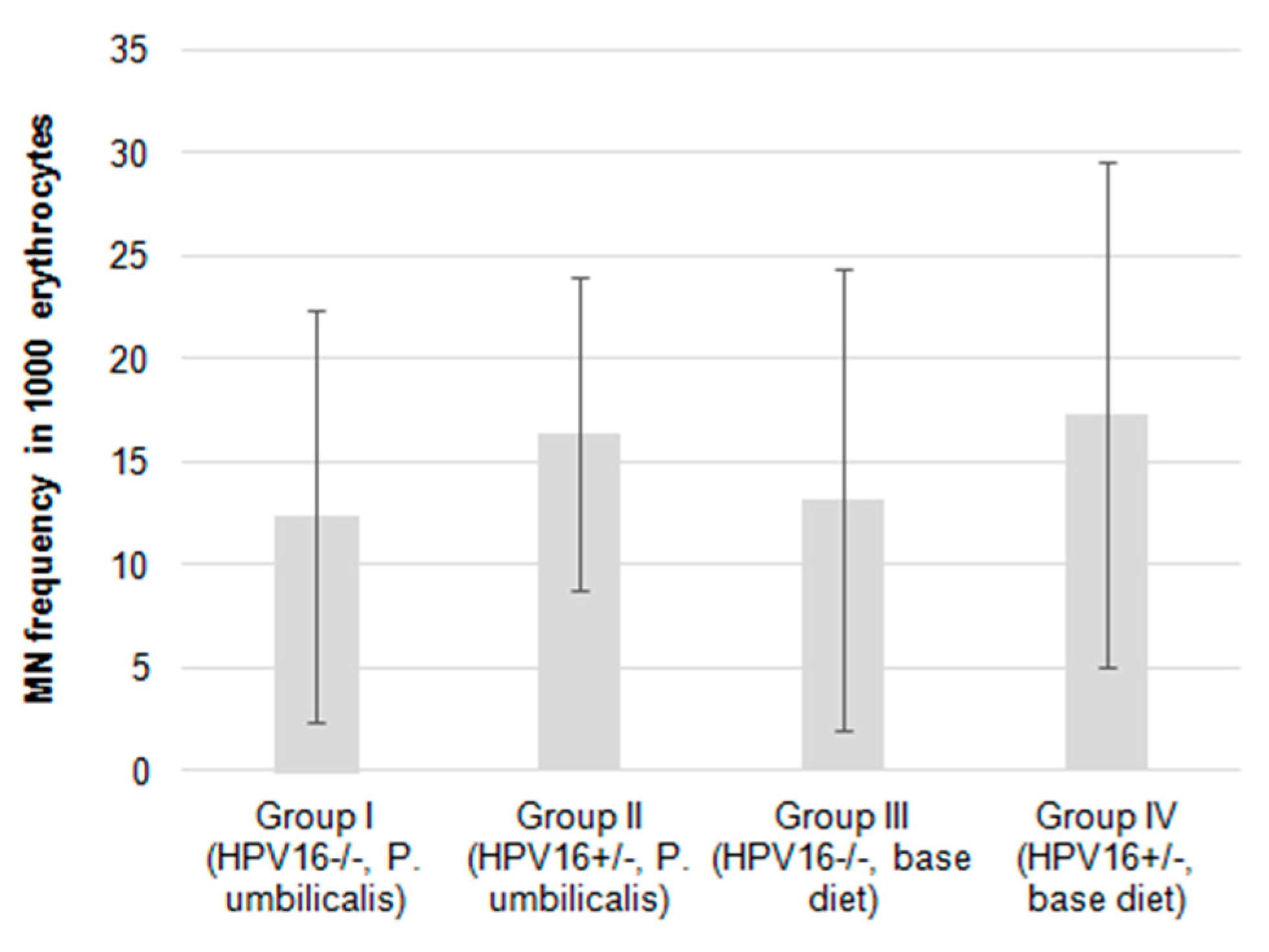
2.5. Micronucleus Test
3. Discussion
4. Material and Methods
4.1. Animals
4.2. Diet Preparation
4.3. Experimental Conditions
4.4. Experimental Design
4.5. Histological Analysis
4.6. Biochemical Analysis of Serum
4.7. Genotoxicity Assays
4.7.1. Comet Assay
4.7.2. Micronucleus Test
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Shalaby, E.A. Algae as promising organisms for environment and health. Plant Signal. Behav. 2011, 6, 1338–1350. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cofrades, S.; Benedí, J.; Garcimartin, A.; Sánchez-muniz, F.J.; Jimenez-colmenero, F. A comprehensive approach to formulation of seaweed-enriched meat products: From technological development to assessment of healthy properties. Food Res. Int. 2017, 99, 1084–1094. [Google Scholar] [CrossRef] [PubMed]
- Brownlee, I.; Fairclough, A.; Hall, A.; Paxman, J. The potential health benefits of seaweed and seaweed extract. In Seaweed: Ecology, Nutrient Composition and Medicinal Uses; Pomin, V.H., Ed.; Nova Science: Hauppauge, NY, USA, 2012; pp. 119–136. [Google Scholar]
- Brown, E.M.; Allsopp, P.J.; Magee, P.J.; Gill, C.I.; Nitecki, S.; Strain, C.R.; Mcsorley, E.M. Seaweed and human health. Nutr. Rev. 2014, 72, 205–216. [Google Scholar] [CrossRef] [PubMed]
- MacArtain, P.; Gill, C.I.R.; Brooks, M.; Campbell, R.; Rowland, I.R. Nutritional value of edible seaweeds. Nutr. Rev. 2007, 65, 535–543. [Google Scholar] [CrossRef] [PubMed]
- Pereira, L. A review of the nutrient composition of selected edible seaweeds. In Seaweed: Ecology, Nutrient Composition and Medicinal Uses, 1st ed.; Pomin, V.H., Ed.; Nova Science: Hauppauge, NY, USA, 2011; pp. 15–47. [Google Scholar]
- Cardoso, S.M.; Pereira, O.R.; Seca, A.M.L.; Pinto, D.C.G.A.; Silva, A.M.S. Seaweeds as preventive agents for cardiovascular diseases: From nutrients to functional foods. Mar. Drugs 2015, 13, 6838–6865. [Google Scholar] [CrossRef] [PubMed]
- Déléris, P.; Nazih, H.; Bard, J.M. Seaweeds in Human Health. In Seaweed in Health and Disease Prevention; Academic Press, Elsevier: Cambridge, MA, USA, 2016; pp. 319–367. [Google Scholar]
- Gutiérrez-Rodríguez, A.G.; Juárez-Portilla, C.; Olivares-Bañuelos, T.; Zepeda, R.C. Anticancer activity of seaweeds. Drug Discov. Today 2018, 23, 434–447. [Google Scholar] [CrossRef] [PubMed]
- Rocha, D.H.A.; Seca, A.M.L.; Pinto, D.C.G.A. Seaweed Secondary Metabolites In Vitro and In Vivo Anticancer Activity. Mar. Drugs 2018, 16, 410. [Google Scholar] [CrossRef]
- Brawley, S.H.; Blouin, N.A.; Ficko-Blean, E.; Wheeler, G.L.; Lohr, M.; Goodson, H.V.; Jenkins, J.W.; Blaby-Haas, C.E.; Helliwell, K.E.; Chan, C.X.; et al. Insights into the red algae and eukaryotic evolution from the genome of Porphyra umbilicalis (Bangiophyceae, Rhodophyta). Proc. Natl. Acad. Sci. USA 2017, 114, E6361–E6370. [Google Scholar] [CrossRef]
- Cofrades, S.; López-Lopez, I.; Bravo, L.; Ruiz-Capillas, C.; Bastida, S.; Larrea, M.T.; Jiménez-Colmenero, F. Nutritional and antioxidant properties of different brown and red Spanish edible seaweeds. Food Sci. Technol. Int. 2010, 16, 361–370. [Google Scholar] [CrossRef]
- López-López, I.; Bastida, S.; Ruiz-Capillas, C.; Bravo, L.; Larrea, M.T.; Sánchez-Muniz, F.; Cofrades, S.; Jiménez-Colmenero, F. Composition and antioxidant capacity of low-salt meat emulsion model systems containing edible seaweeds. Meat Sci. 2009, 83, 492–498. [Google Scholar] [CrossRef]
- Gaglia, M.M.; Munger, K. More than just oncogenes: Mechanisms of tumorigenesis by human viruses. Curr. Opin. Virol. 2018, 32, 48–59. [Google Scholar] [CrossRef] [PubMed]
- De Martel, C.; Plummer, M.; Vignat, J.; Franceschi, S. Worldwide burden of cancer attributable to HPV by site, country and HPV type. Int. J. Cancer 2017, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.X.; Zhang, X.S.; Guan, H.S.; Wang, W. Potential anti-HPV and related cancer agents from marine resources: An overview. Mar. Drugs 2014, 12, 2019–2035. [Google Scholar] [CrossRef] [PubMed]
- Pereira, L. Biological and therapeutic properties of the seaweed polysaccharides. Int. Biol. Rev. 2018, 2, 1–50. [Google Scholar]
- Blaszczak, W.; Lach, M.S.; Barczak, W.; Suchorska, W.M. Fucoidan Exerts Anticancer Effects Against Head and Neck Squamous Cell Carcinoma In Vitro. Molecules 2018, 23, 3302. [Google Scholar] [CrossRef]
- Coussens, L.M.; Hanahan, D.; Arbeit, J.M. Genetic predisposition and parameters of malignant progression in K14- HPV16 transgenic mice. Am. J. Pathol. 1996, 149, 1899–1917. [Google Scholar]
- Medeiros-Fonseca, B.; Mestre, V.F.; Colaço, B.; Pires, M.J.; Martins, T.; Gil da Costa, R.M.; Neuparth, M.J.; Medeiros, R.; Moutinho, M.S.S.; Dias, M.I.; et al. Laurus nobilis (laurel) aqueous leaf extract’s toxicological and anti-tumor activities in HPV16-transgenic mice. Food Funct. 2018, 9, 4419–4428. [Google Scholar] [CrossRef]
- Santos, J.M.O.; Fernandes, M.; Araújo, R.; Sousa, H.; Ribeiro, J.; Bastos, M.M.S.M.; Oliveira, P.A.; Carmo, D.; Casaca, F.; Silva, S.; et al. Dysregulated expression of microRNA-150 in human papillomavirus-induced lesions of K14-HPV16 transgenic mice. Life Sci. 2017, 175, 31–36. [Google Scholar] [CrossRef]
- Gil da Costa, R.M.; Aragão, S.; Moutinho, M.; Alvarado, A.; Carmo, D.; Casaca, F.; Silva, S.; Ribeiro, J.; Sousa, H.; Ferreira, R.; et al. HPV16 induces a wasting syndrome in transgenic mice: Amelioration by dietary polyphenols via NF-κB inhibition. Life Sci. 2017, 169, 11–19. [Google Scholar] [CrossRef]
- Minkiewicz, P.; Turło, M.; Iwaniak, A. Free Accessible Databases as a Source of Information about Food Components and Other Compounds with Anticancer Activity—Brief Review. Molecules 2019, 24, 789. [Google Scholar] [CrossRef]
- Calder, P.C.; Grimble, R.F. Polyunsaturated fatty acids, inflammation and immunity. Eur. J. Clin. Nutr. 2002, 56, S14–S19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Paiva, I.; Gil da Costa, R.M.; Ribeiro, J.; Sousa, H.; Bastos, M.; Faustino-Rocha, A.; Oliveira, P.A.; Medeiros, R. A role for MicroRNA-155 expression in microenvironment associated to HPV-induced carcinogenesis in K14-HPV16 transgenic mice. PLoS ONE 2015, 10, e0116868. [Google Scholar] [CrossRef] [PubMed]
- Shirnekhi, H.K.; Kelley, E.P.; Deluca, J.G.; Herman, J.A.; Bloom, K.S. Spindle assembly checkpoint signaling and sister chromatid cohesion are disrupted by HPV E6-mediated transformation. Mol. Biol. Cell 2017, 28, 2035–2041. [Google Scholar] [CrossRef] [PubMed]
- Reshmi, G.; Pillai, M.R. Interplay Between HPV Oncoproteins and MicroRNAs in Cervical Cancer. IntechOpen 2012, 347–360. [Google Scholar]
- Santos, C.; Neto, T.; Ferreirinha, P.; Sousa, H.; Ribeiro, J.; Bastos, M.M.S.M.; Faustino-Rocha, A.I.; Oliveira, P.A.; Medeiros, R.; Vilanova, M.; et al. Celecoxib promotes degranulation of CD8 + T cells in HPV-induced lesions of K14-HPV16 transgenic mice. Life Sci. 2016, 157, 67–73. [Google Scholar] [CrossRef]
- Arbeit, J.M.; Munger, K.; Howley, P.M.; Hanahan, D. Progressive squamous epithelial neoplasia in K14-human papillomavirus type 16 transgenic mice. J. Virol. 1994, 68, 4358–4368. [Google Scholar] [PubMed]
- Smith-McCune, K.; Zhu, Y.-H.; Hanhan, D.; Arbeit, A. Cross-species comarison of angiogenesis during the premalignant stages of squamous carcinogenesis in the human cervix and K14-HPV16 transgenic mice. Cancer Res. 1997, 57, 1294–1300. [Google Scholar]
- Hussain, T.; Tan, B.; Yin, Y.; Blachier, F.; Tossou, M.C.B.; Rahu, N. Oxidative Stress and Inflammation: What Polyphenols Can Do for Us? Oxidative Med. Cell. Longev. 2016, 2016, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Działo, M.; Mierziak, J.; Korzun, U.; Preisner, M.; Szopa, J.; Kulma, A. The Potential of Plant Phenolics in Prevention and Therapy of Skin Disorders. Int. J. Mol. Sci. 2016, 17, 160. [Google Scholar] [CrossRef]
- Mercurio, D.G.; Wagemaker, T.A.L.; Alves, V.M.; Benevenuto, C.G.; Gaspar, L.R.; Campos, P.M.B.G.M. In vivo photoprotective effects of cosmetic formulations containing UV filters, vitamins, Ginkgo biloba and red algae extracts. J. Photochem. Photobiol. B 2015, 153, 121–126. [Google Scholar] [CrossRef]
- Fernandes, J.V.; Fernandes, T.A.A.D.M.; de Azevedo, J.C.V.; Cobucci, R.N.O.; de Carvalho, M.G.F.; Andrade, V.S.; De Araújo, J.M.G. Link between chronic inflammation and human papillomavirus-induced carcinogenesis (Review). Oncol. Lett. 2015, 9, 1015–1026. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferreira, T.; Campos, S.; Silva, M.G.; Ribeiro, R.; Santos, S.; Almeida, J.; Pires, M.J.; Gil da Costa, R.M.; Córdova, C.; Nogueira, A.; et al. The Cyclooxigenase-2 Inhibitor Parecoxib Prevents Epidermal Dysplasia in HPV16-Transgenic Mice: Efficacy and Safety Observations. Int. J. Mol. Sci. 2019, 20, 3902. [Google Scholar] [CrossRef] [PubMed]
- Santos, J.M.O.; Moreira-Pais, A.; Neto, T.; Peixoto da Silva, S.; Oliveira, P.A.; Ferreira, R.; Mendes, J.; Bastos, M.M.S.M.; Lopes, C.; Casaca, F.; et al. Dimethylaminoparthenolide reduces the incidence of dysplasia and ameliorates a wasting syndrome in HPV16-transgenic mice. Drug Dev. Res. 2019, 80, 824–830. [Google Scholar] [CrossRef] [PubMed]
- Peirone, C.; Mestre, V.F.; Medeiros-Fonseca, B.; Colaço, B.; Pires, M.J.; Martins, T.; Gil da Costa, R.M.; Neuparth, M.J.; Medeiros, R.; Bastos, M.M.S.M.; et al. Ozone therapy prevents the onset of dysplasia in HPV16-transgenic mice—A preclinical efficacy and safety study. Biomed. Pharmacother. 2018, 104, 275–279. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.H.; Kwon, J.Y.; Lee, J.K.; Seo, Y.R. Recent Advances in In Vivo Genotoxicity Testing: Prediction of Carcinogenic Potential Using Comet and Micronucleus Assay in Animal Models. J. Cancer Prev. 2013, 18, 277–288. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cotelle, S.; Fe, J.F. Comet Assay in Genetic Ecotoxicology: A Review. Environ. Mol. Mutagen. 1999, 255, 246–255. [Google Scholar] [CrossRef]
- Guilherme, S.; Gaivão, I.; Santos, M.A.; Pacheco, M. European eel (Anguilla anguilla) genotoxic and pro-oxidant responses following short-term exposure to Roundup®—A glyphosate-based herbicide. Mutagenesis 2010, 25, 523–530. [Google Scholar] [CrossRef]
- Llana-Ruiz-Cabello, M.; Puerto, M.; Maisanaba, S.; Guzmán-Guillén, R.; Pichardo, S.; Cameán, A.M. Use of micronucleus and comet assay to evaluate evaluate the genotoxicity of oregano essential oil (Origanum vulgare L. Virens) in rats orally exposed for 90 days. J. Toxicol. Environ. Heal. 2018, 81, 525–533. [Google Scholar]
- Droge, W. Free Radicals in the Physiological Control of Cell Function. Siol. Rev. 2002, 82, 47–95. [Google Scholar] [CrossRef]
- Marques, A.; Ferreira, J.; Abreu, H.; Pereira, R.; Rego, A.; Serôdio, J.; Christa, G.; Gaivão, I.; Pacheco, M. Searching for antigenotoxic properties of marine macroalgae dietary supplementation against endogenous and exogenous challenges. J. Toxicol. Environ. Heal. 2018, 81, 939–956. [Google Scholar] [CrossRef]
- Ferreira, J.; Marques, A.; Abreu, H.; Pereira, R.; Rego, A.; Gaivão, I. Red seaweeds Porphyra umbilicalis and Grateloupia turuturu display antigenotoxic and longevity-promoting potential in Drosophila melanogaster. Eur. J. Phycol. 2019. [Google Scholar] [CrossRef]
- Forbes, D.; Blom, H.; Kostomitsopulos, N.; Moore, G.; Perretta, G. Euroguide: On the Accommodation and Care of Animals Used for Experimental and Other Scientific Purposes; FELASA: London, UK, 2007. [Google Scholar]
- Shaposhnikov, S.; Azqueta, A.; Henriksson, S.; Meier, S.; Gaivão, I.; Huskisson, N.H.; Smart, A.; Brunborg, G.; Nilsson, M.; Collins, A.R. Twelve-gel slide format optimised for comet assay and fluorescent in situ hybridisation. Toxicol. Lett. 2010, 195, 31–34. [Google Scholar] [CrossRef] [PubMed]
- Guilherme, S.; Santos, M.A.; Gaivão, I.; Pacheco, M. DNA and chromosomal damage induced in fish (Anguilla anguilla L.) by aminomethylphosphonic acid (AMPA)—The major environmental breakdown product of glyphosate. Environ Sci Pollut Res. 2014, 21, 8730–8739. [Google Scholar] [CrossRef] [PubMed]
- Sierra, L.M.; Gaivão, I. Genotoxicity and DNA Repair: A Practical Approach. In Humana Press; Springer: Oviedo, Spain; Vila Real, Portugal, 2014; pp. 103–113. [Google Scholar]
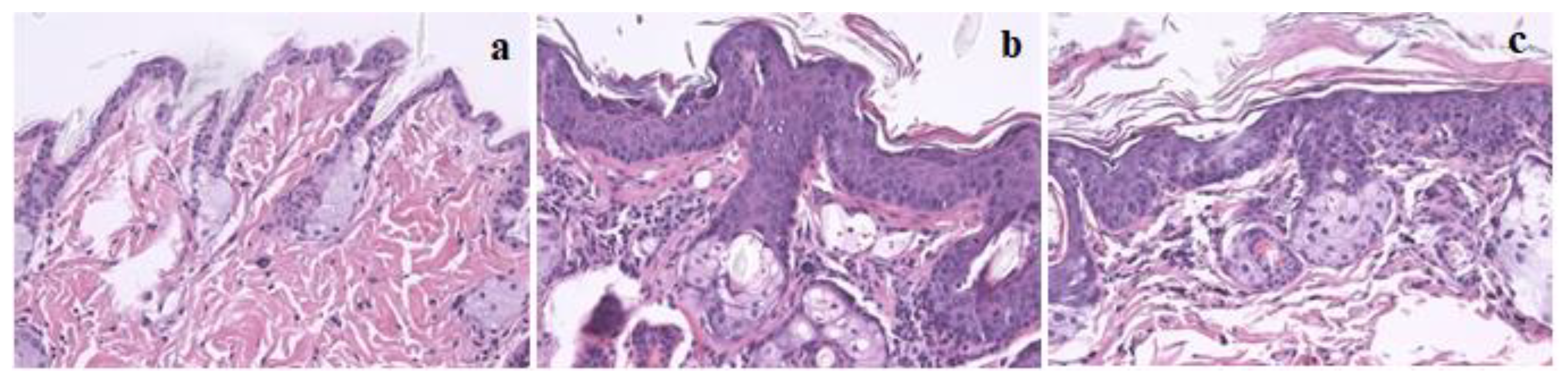
| Liver | Right Kidney | Left Kidney | Spleen | Lung | Heart | Bladder | Thymus | |
|---|---|---|---|---|---|---|---|---|
| Group I (HPV16−/−, Porphyra umbilicalis) | 0.0541 ± 0.0049 | 0.0060 ± 0.0002 | 0.0058 ± 0.0003 | 0.0040 ± 0.0002 | 0.0052 ± 0.0002 1 | 0.0037 ± 0.0002 | 0.0006 ± 0.0001 | 0.0010 ± 0.0001 |
| Group II (HPV16+/−, P. umbilicalis) | 0.0670 ± 0.0015 | 0.0067 ± 0.0002 | 0.0065 ± 0.0001 | 0.0063 ± 0.0007 | 0.0063 ± 0.0003 | 0.0046 ± 0.0002 | 0.0010 ± 0.0001 | 0.0011 ± 0.0001 |
| Group III (HPV16−/−, base diet) | 0.0574 ± 0.0012 | 0.0057 ± 0.0002 | 0.0062 ± 0.0002 | 0.0047 ± 0.0002 | 0.0063 ± 0.0003 | 0.0042 ± 0.0002 | 0.0003 ± 0.0002 | 0.0012 ± 0.0002 |
| Group IV (HPV16+/−, base diet) | 0.0717 ± 0.0019 | 0.0069 ± 0.0002 | 0.0068 ± 0.0002 | 0.0083 ± 0.0010 | 0.0071 ± 0.0002 | 0.0051 ± 0.0002 | 0.0008 ± 0.0001 | 0.0014 ± 0.0001 |
| Skin Chest Incidence/n (%) | Ear Incidence/n (%) | |||||
|---|---|---|---|---|---|---|
| Normal | Epidermal Hyperplasia | Epidermal Dysplasia | Normal | Epidermal Hyperplasia | Epidermal Dysplasia | |
| Group I (HPV16−/−, P. umbilicalis) | 11/11 (100.0%) | 0/11 (0%) | 0/11 (0%) | 11/11 (100.0%) | 0/11 (0%) | 0/11 (0%) |
| Group II (HPV16+/−, P. umbilicalis) | 0/11 (0%) | 11/11 (100.0%) | 0/11 (0%) | 0/11 (0%) | 7/9 (77.8%) | 2/9 (22.2%) |
| Group III (HPV16−/−, base diet) | 11/11 (100.0%) | 0/11 (0%) | 0/11 (0%) | 11/11 (100.0%) | 0/11 (0%) | 0/11 (0%) |
| Group IV (HPV16+/−, base diet) | 0/11 (0%) | 4/11 1 (36.4%) | 7/11 (63.6%) | 0/11 (0%) | 4/11 (36.4%) | 7/11 (63.6%) |
| Group I (HPV16−/−, P. umbilicalis) | Group II (HPV16+/−, P. umbilicalis) | Group III (HPV16−/− Base Diet) | Group IV (HPV16+/− Base Diet) | |
|---|---|---|---|---|
| Albumin (g/L) | 28.65 ± 1.37 | 30.43 ± 0.93 | 29.78 ± 1.71 | 30.37 ± 0.96 |
| Total proteins (g/L) | 45.95 ± 1.72 | 50.32 ± 2.24 | 51.34 ± 4.07 | 49.62 ± 1.12 |
| Glucose (mg/dL) | 222.29 ± 11.16 | 197.65 ± 15.97 | 195.70 ± 15.99 | 198.07 ± 13.36 |
| Aspartate aminotransferase (U/L) | 35.63 ± 4.03 | 40.89 ± 5.65 | 37.28 ± 4.70 | 38.85 ± 3.54 |
| Alanine aminotransferase (U/L) | 59.34 ± 5.17 | 65.66 ± 7.16 | 44.74 ± 3.76 | 51.82 ± 3.70 |
| Gamma glutamyltransferase (U/L) | 31.75 ± 3.17 | 40.07 ± 7.67 | 48.61 ± 6.11 | 60.78 ± 8.35 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Santos, S.; Ferreira, T.; Almeida, J.; Pires, M.J.; Colaço, A.; Lemos, S.; Gil da Costa, R.M.; Medeiros, R.; Bastos, M.M.S.M.; Neuparth, M.J.; et al. Dietary Supplementation with the Red Seaweed Porphyra umbilicalis Protects against DNA Damage and Pre-Malignant Dysplastic Skin Lesions in HPV-Transgenic Mice. Mar. Drugs 2019, 17, 615. https://doi.org/10.3390/md17110615
Santos S, Ferreira T, Almeida J, Pires MJ, Colaço A, Lemos S, Gil da Costa RM, Medeiros R, Bastos MMSM, Neuparth MJ, et al. Dietary Supplementation with the Red Seaweed Porphyra umbilicalis Protects against DNA Damage and Pre-Malignant Dysplastic Skin Lesions in HPV-Transgenic Mice. Marine Drugs. 2019; 17(11):615. https://doi.org/10.3390/md17110615
Chicago/Turabian StyleSantos, Susana, Tiago Ferreira, José Almeida, Maria J. Pires, Aura Colaço, Sílvia Lemos, Rui M. Gil da Costa, Rui Medeiros, Margarida M. S. M. Bastos, Maria J. Neuparth, and et al. 2019. "Dietary Supplementation with the Red Seaweed Porphyra umbilicalis Protects against DNA Damage and Pre-Malignant Dysplastic Skin Lesions in HPV-Transgenic Mice" Marine Drugs 17, no. 11: 615. https://doi.org/10.3390/md17110615
APA StyleSantos, S., Ferreira, T., Almeida, J., Pires, M. J., Colaço, A., Lemos, S., Gil da Costa, R. M., Medeiros, R., Bastos, M. M. S. M., Neuparth, M. J., Abreu, H., Pereira, R., Pacheco, M., Gaivão, I., Rosa, E., & Oliveira, P. A. (2019). Dietary Supplementation with the Red Seaweed Porphyra umbilicalis Protects against DNA Damage and Pre-Malignant Dysplastic Skin Lesions in HPV-Transgenic Mice. Marine Drugs, 17(11), 615. https://doi.org/10.3390/md17110615










