The Neglected Marine Fungi, Sensu stricto, and Their Isolation for Natural Products’ Discovery
Abstract
1. Introduction
2. Sources of Marine Fungi
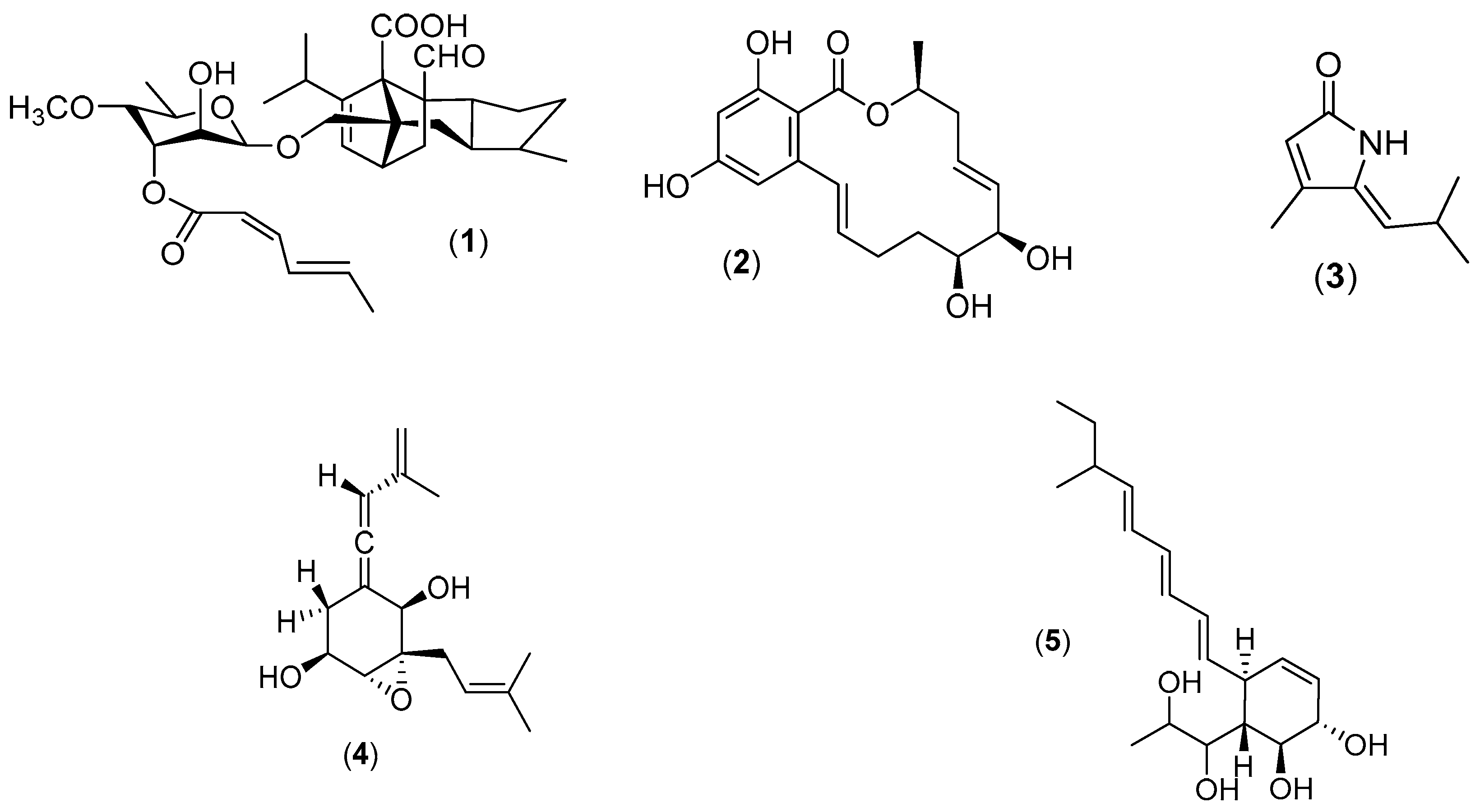
3. Natural Products from Marine Fungi (Sensu stricto)
4. Isolation Techniques
4.1. Sample Collection
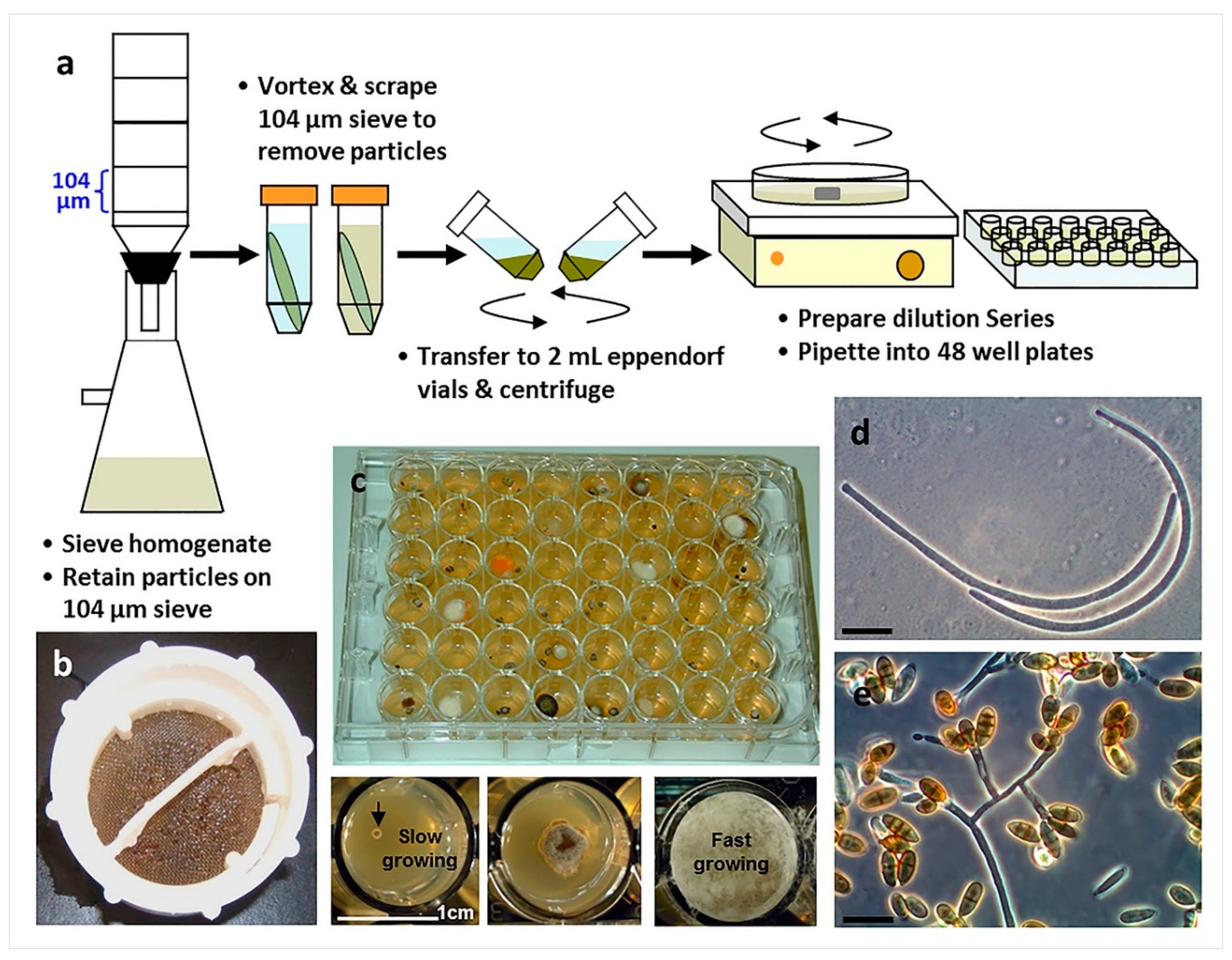
4.2. Direct Plating vs. Particle Filtration and Dilution to Extinction Plating
4.3. Damp Chambers
4.4. Baiting Stations
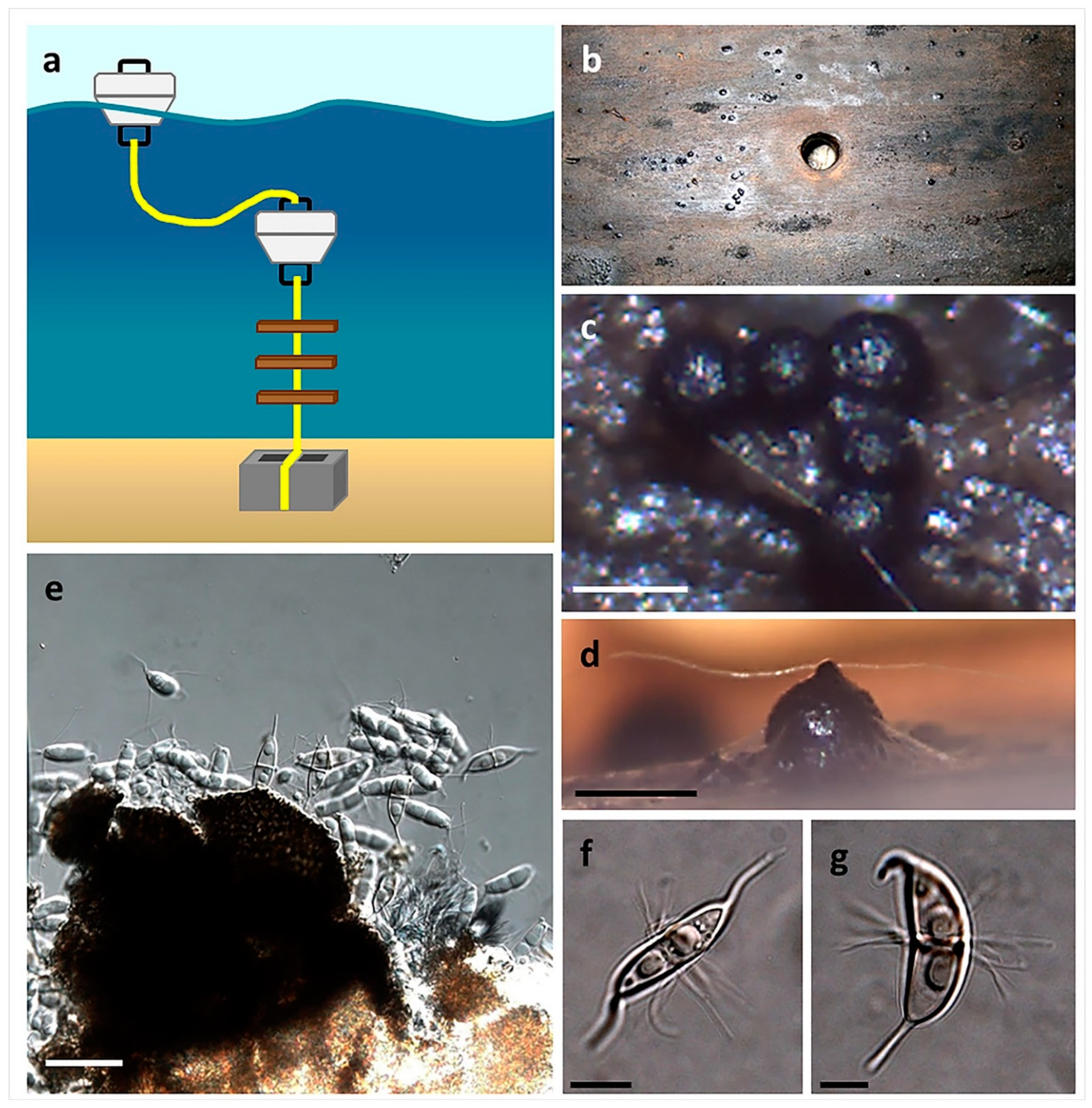
5. In Situ Culturing
6. Isolation and Examination of Samples
7. Preservation
8. Concluding Remarks
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hover, B.M.; Kim, S.-H.; Katz, M.; Charlop-Powers, Z.; Owen, J.G.; Ternei, M.A.; Maniko, J.; Estrela, A.B.; Molina, H.; Park, S. Culture-independent discovery of the malacidins as calcium-dependent antibiotics with activity against multidrug-resistant Gram-positive pathogen. Nat. Microbiol. 2018, 3, 415–422. [Google Scholar] [CrossRef] [PubMed]
- Overy, D.P.; Bayman, P.; Kerr, R.G.; Bills, G.F. An assessment of natural product discovery from marine (sensu strictu) and marine-derived fungi. Mycology 2014, 5, 145–167. [Google Scholar] [CrossRef]
- Bugni, T.S.; Ireland, C.M. Marine-derived fungi: A chemically and biologically diverse group of microorganisms. Nat. Prod. Rep. 2004, 21, 143–163. [Google Scholar] [CrossRef] [PubMed]
- Rateb, M.E.; Ebel, R. Secondary metabolites of fungi from marine habitats. Nat. Prod. Rep. 2011, 28, 290–344. [Google Scholar] [CrossRef] [PubMed]
- Tasdemir, D. Marine fungi in the spotlight: Opportunities and challenges for marine fungal natural product discovery and biotechnology. Fungal Biol. Biotechnol. 2017, 4, 5. [Google Scholar] [CrossRef]
- Karwehl, S.; Stadler, M. Exploitation of fungal biodiversity for discovery of novel antibiotics. In How to Overcome the Antibiotic Crisis; Stadler, M., Dersch, P., Eds.; Springer International Publishing AG: Cham, Switzerland, 2016; pp. 303–338. [Google Scholar]
- Jones, E.B.G. Fifty years of marine mycology. Fungal Divers. 2011, 50, 73–112. [Google Scholar] [CrossRef]
- Zettler, E.R.; Mincer, T.J.; Amaral-Zettler, L.A. Life in the “Plastisphere”: Microbial communities on plastic marine debris. Environ. Sci. Technol. 2013, 47, 7137–7146. [Google Scholar] [CrossRef]
- Kohlmeyer, J.; Kohlmeyer, E. Marine Mycology: The Higher Fungi; Academic Press: New York, NY, USA, 1979. [Google Scholar]
- Jones, E.B.G. Ultrastructure and taxonomy of the aquatic ascomycetous order Halosphaeriales. Can. J. Bot. 1995, 73, 790–801. [Google Scholar] [CrossRef]
- Overy, D.P.; Berrue, F.; Correa, H.; Hanif, N.; Hay, K.; Lanteigne, M.; Mquilian, K.; Duffy, S.; Boland, P.; Jagannathan, R.; et al. Sea foam as a source of fungal inoculum for the isolation of biologically active natural products. Mycology 2014, 5, 130–144. [Google Scholar] [CrossRef]
- Zuccaro, A.; Mitchell, J. Fungal communities of seaweeds. In The fungal Community: Its Organization and Role in the Ecosystem; Deighton, J., White, J., Oudemans, P., Eds.; CRC Press: New York, NY, USA, 2005; pp. 553–580. [Google Scholar]
- Gessner, R.V.; Goos, R. Fungi from Spartina alterniflora in sRhode Island. Mycologia 1973, 65, 1296–1301. [Google Scholar] [CrossRef]
- Newell, S.Y.; Porter, D.; Lingle, W.L. Lignocellulolysis by ascomycetes (fungi) of a saltmarsh grass (smooth cordgrass). Microsc. Res. Tech. 1996, 33, 32–46. [Google Scholar] [CrossRef]
- Kohlmeyer, J.; Volkmann-Kohlmeyer, B. The biodiversity of fungi on Juncus roemerianus. Mycol. Res. 2001, 105, 1411–1412. [Google Scholar] [CrossRef]
- Walker, A.K.; Campbell, J. Marine fungal diversity: A comparison of natural and created salt marshes of the north-central Gulf of Mexico. Mycologia 2010, 102, 513–521. [Google Scholar] [CrossRef] [PubMed]
- Elmer, W.H.; Marra, R.E. New species of Fusarium associated with dieback of Spartina alterniflora in Atlantic salt marshes. Mycologia 2011, 103, 806–819. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Wang, N.-F.; Zhang, Y.-Q.; Liu, H.-Y.; Yu, L.-Y. Diversity and distribution of aquatic fungal communities in the Ny-Ålesund region, Svalbard (high arctic). Microb. Ecol. 2016, 71, 543–554. [Google Scholar] [CrossRef] [PubMed]
- Hassett, B.T.; Ducluzeau, A.-L.L.; Collins, R.E.; Gradinger, R. Spatial distribution of aquatic marine fungi across the western Arctic and sub-arctic. Environ. Microbiol. 2017, 19, 475–484. [Google Scholar] [CrossRef]
- Hassett, B.T.; Gradinger, R. Chytrids dominate arctic marine fungal communities. Environ. Microbiol. 2016, 18, 2001–2009. [Google Scholar] [CrossRef]
- Loque, C.P.; Medeiros, A.O.; Pellizzari, F.M.; Oliveira, E.C.; Rosa, C.A.; Rosa, L.H. Fungal community associated with marine macroalgae from Antarctica. Polar Biol. 2010, 33, 641–648. [Google Scholar] [CrossRef]
- Rämä, T.; Hassett, B.T.; Bubnova, E. Arctic marine fungi from filaments and flagella to operational taxonomic units and beyond. Bot. Mar. 2017, 60, 433–452. [Google Scholar] [CrossRef]
- Arvas, M.; Kivioja, T.; Mitchell, A.; Saloheimo, M.; Ussery, D.; Penttila, M.; Oliver, S. Comparison of protein coding gene contents of the fungal phyla Pezizomycotina and Saccharomycotina. BMC Genom. 2007, 8, 325. [Google Scholar] [CrossRef]
- Lackner, G.; Misiek, M.; Braesel, J.; Hoffmeister, D. Genome mining reveals the evolutionary origin and biosynthetic potential of basidiomycete polyketide synthases. Fungal Genet. Biol. 2012, 49, 996–1003. [Google Scholar] [CrossRef] [PubMed]
- Ohm, R.A.; Feau, N.; Henrissat, B.; Schoch, C.L.; Horwitz, B.A.; Barry, K.W.; Condon, B.J.; Copeland, A.C.; Dhillon, B.; Glaser, F.; et al. Diverse lifestyles and strategies of plant pathogenesis encoded in the genomes of eighteen Dothideomycetes fungi. PLoS Pathog. 2012, 8, e1003037. [Google Scholar] [CrossRef] [PubMed]
- Isaka, M.; Suyarnsestakorn, C.; Tanticharoen, M.; Kongsaeree, P.; Thebtaranonth, Y. Aigialomycins A-E, new resorcylic macrolides from the marine mangrove fungus Aigialus parvus. J. Org. Chem. 2002, 67, 1561–1566. [Google Scholar] [CrossRef] [PubMed]
- Isaka, M.; Yangchum, A.; Intamas, S.; Kocharin, K.; Jones, E.G.; Kongsaeree, P.; Prabpai, S. Aigialomycins and related polyketide metabolites from the mangrove fungus Aigialus parvus BCC 5311. Tetrahedron 2009, 65, 4396–4403. [Google Scholar] [CrossRef]
- Vongvilai, P.; Isaka, M.; Kittakoop, P.; Srikitikulchai, P.; Kongsaeree, P.; Thebtaranonth, Y. Ketene acetal and spiroacetal constituents of the marine fungus Aigialus parvus BCC 5311. J. Nat. Prod. 2004, 67, 457–460. [Google Scholar] [CrossRef] [PubMed]
- Kohlmeyer, J.; Schatz, S. Aigialus gen. novo (Ascomycetes) with two marine species from mangroves. Trans. Br. Mycol. Soc. 1985, 85, 699–707. [Google Scholar] [CrossRef]
- Tan, T.K.; Teng, C.L.; Jones, E.B.G. Substrate type and microbial interactions as factors affecting ascocarp formation by mangrove fungi. Hydrobiologia 1995, 295, 127–134. [Google Scholar] [CrossRef]
- Alias, S.; Kuthubutheen, A.; Jones, E.G. Frequency of occurrence of fungi on wood in Malaysian mangroves. In Asia-Pacific Symposium on Mangrove Ecosystems, Developments in Hydrobiology; Alias, S.A., Kuthubutheen, A.J., Jones, E.B.G., Eds.; Springer: Dordrecht, The Netherlands, 1995; Volume 106, pp. 97–106. [Google Scholar]
- Abbanat, D.; Leighton, M.; Maiese, W.; Jones, E.; Pearce, C.; Greenstein, M. Cell wall active antifungal compounds produced by the marine fungus Hypoxylon oceanicum LL-15G256. J. Antibiot. 1998, 51, 296–302. [Google Scholar] [CrossRef]
- Ogita, J.; Hayashi, A.; Sato, S.; Furutani, W. Antibiotic Zopfimarin. Japan Patent 62-40292, 1987. [Google Scholar]
- Kondo, M.; Takayama, T.; Furuya, K.; Okudaira, M.; Hayashi, T.; Kinoshita, M. A nuclear magnetic resonance study of Zopfinol isolated from Zopfiella marina. Annu. Rep. Sankyo Res. Lab. 1987, 39, 45–53. [Google Scholar]
- Barluenga, S.; Dakas, P.Y.; Ferandin, Y.; Meijer, L.; Winssinger, N. Modular asymmetric synthesis of aigialomycin D, a kinase-inhibitory scaffold. Angew. Chem. Int. Ed. 2006, 45, 3951–3954. [Google Scholar] [CrossRef]
- Alvi, K.A.; Casey, A.; Nair, B.G. Pulchellalactam: A CD45 protein tyrosine phosphatase inhibitor from the marine fungus Corollospora pulchella. J. Antibiot. 1998, 51, 515–517. [Google Scholar] [CrossRef]
- Elsebai, M.F.; Kehraus, S.; Gütschow, M.; Koenig, G.M. New polyketides from the marine-derived fungus Phaeosphaeria spartinae. Nat. Prod. Commun. 2009, 4, 1463–1468. [Google Scholar] [PubMed]
- Elsebai, M.F.; Kehraus, S.; Gütschow, M.; Koenig, G.M. Spartinoxide, a new enantiomer of A82775C with inhibitory activity toward HLE from the marine-derived fungus Phaeosphaeria spartinae. Nat. Prod. Commun. 2010, 5, 1071–1076. [Google Scholar] [PubMed]
- Hyde, K.D.; Jones, E.B.G.; Leaño, E.; Pointing, S.B.; Poonyth, A.D.; Vrijmoed, L.L.P. Role of fungi in marine ecosystems. Biodivers. Conserv. 1998, 7, 1147–1161. [Google Scholar] [CrossRef]
- Bremer, G. Isolation and culture of thraustochytrids. In Marine Mycology—A Practical Approach; Hyde, K.D., Pointing, S.B., Eds.; Fungal Diversity Press: Hong Kong, China, 2000; pp. 49–61. [Google Scholar]
- Vrijmoed, L.L.P. Isolation and culture of higher filamentous fungi. In Marine Mycology—A Practical Approach; Hyde, K.D., Pointing, S.B., Eds.; Fungal Diversity Press: Hong Kong, China, 2000; pp. 1–20. [Google Scholar]
- Walker, A.K. Marine Fungi of U.S. Gulf of Mexico Barrier Island Beaches: Biodiversity and Sampling Strategy. Ph.D. Thesis, University of Southern Missisippi, Hattiesburg, MS, USA, December 2012. [Google Scholar]
- Koehn, R.D. Fungi isolated from sea foam collected at North Padre island beaches. Southwest. Nat. 1982, 27, 17–21. [Google Scholar] [CrossRef]
- Kirk, P.W. Direct enumeration of marine arenicolous fungi. Mycologia 1983, 75, 670–682. [Google Scholar] [CrossRef]
- Hyde, K.D.; Jones, E.G. Introduction to fungal succession. Bot. J. Linn. Soc. 1989, 100, 237–254. [Google Scholar] [CrossRef]
- Rämä, T.; Nordén, J.; Davey, M.L.; Mathiassen, G.H.; Spatafora, J.W.; Kauserud, H. Fungi ahoy! Diversity on marine wooden substrata in the high North. Fungal Ecol. 2014, 8, 46–58. [Google Scholar] [CrossRef]
- Bills, G.F.; Christensen, M.; Powell, M.; Thorn, G. Saprobic soil fungi. In Biodiversity of Fungi, Inventory and Monitoring Methods; Mueller, G., Bills, G.F., Foster, M., Eds.; Elsevier Academic Press: San Diego, CA, USA, 2004; pp. 271–302. [Google Scholar]
- Collado, J.; Platas, G.; Paulus, B.; Bills, G.F. High-throughput culturing of fungi from plant litter by a dilution-to-extinction technique. FEMS Microbiol. Ecol. 2007, 60, 521–533. [Google Scholar] [CrossRef]
- Unterseher, M.; Schnittler, M. Dilution-to-extinction cultivation of leaf-inhabiting endophytic fungi in beech (Fagus sylvatica L.)—Different cultivation techniques influence fungal biodiversity assessment. Mycol. Res. 2009, 113, 645–654. [Google Scholar] [CrossRef]
- Shrestha, P.; Szaro, T.M.; Bruns, T.D.; Taylor, J.W. Systematic search for cultivatable fungi that best deconstruct cell walls of Miscanthus and sugarcane in the field. Appl. Environ. Microbiol. 2011, 77, 5490–5504. [Google Scholar] [CrossRef] [PubMed]
- Jones, E.B.G. Marine fungi: Some factors influencing biodiversity. Fungal Divers. 2000, 4, 53–73. [Google Scholar]
- Pang, K.-L.; Chow, R.; Chan, C.; Vrijmoed, L. Diversity and physiology of marine lignicolous fungi in Arctic waters: A preliminary account. Polar Res. 2011, 30, 5859–5863. [Google Scholar] [CrossRef]
- Hyde, K.D.; Jones, E.B.G. Introduction to fungal succession. Fungal Divers. 2002, 10, 1–4. [Google Scholar]
- Shearer, C.A. Fungi of the Chesapeake bay and its tributaries. III. The distribution of wood-inhabiting ascomycetes and fungi imperfecti of the Patuxent river. Am. J. Bot. 1972, 59, 961–969. [Google Scholar] [CrossRef]
- Lamore, B.J.; Goos, R.D. Wood-inhabiting fungi of a freshwater stream in Rhode Island. Mycologia 1978, 70, 1025–1034. [Google Scholar] [CrossRef]
- Rappé, M.S.; Giovannoni, S.J. The uncultured microbial majority. Annu. Rev. Microbiol. 2003, 57, 369–394. [Google Scholar] [CrossRef] [PubMed]
- Nichols, D.; Cahoon, N.; Trakhtenberg, E.M.; Pham, L.; Mehta, A.; Belanger, A.; Kanigan, T.; Lewis, K.; Epstein, S.S. Use of Ichip for high-throughput in situ cultivation of “uncultivable” microbial species. Appl. Environ. Microbiol. 2010, 76, 2445–2450. [Google Scholar] [CrossRef] [PubMed]
- Ling, L.L.; Schneider, T.; Peoples, A.J.; Spoering, A.L.; Engels, I.; Conlon, B.P.; Mueller, A.; Schäberle, T.F.; Hughes, D.E.; Epstein, S.; et al. A new antibiotic kills pathogens without detectable resistance. Nature 2015, 517, 455–459. [Google Scholar] [CrossRef] [PubMed]
- Gavrish, E.; Bollmann, A.; Epstein, S.; Lewis, K. A trap for in situ cultivation of filamentous actinobacteria. J. Microbiol. Methods 2008, 72, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Epstein, S.S.; Lewis, K.; Nichols, D.; Gavrish, E. New approaches to microbial isolation. In Manual of Industrial Microbiology and Biotechnology; Baltz, R.H., Demain, A.L., Davies, J.E., Bull, A.T., Junker, B., Katz, L., Lynd, L.R., Masurekar, P., Reeves, C.D., Zhao, H., Eds.; ASM Press: Washington, DC, USA, 2010; pp. 3–12. [Google Scholar]
- Mueller, G.M.; Bills, G.F.; Foster, M.S. Biodiversity of Fungi: Inventory and Monitoring Methods; Elsevier Academic Press: San Diego, CA, USA, 2004. [Google Scholar]
- Kirk, P.W. Isolation and culture of lignicolous marine fungi. Mycologia 1969, 61, 174–177. [Google Scholar] [CrossRef]
- Nakagiri, A.; Jones, E.G. Long-term maintenance of cultures. In Marine Mycology—A Practical Approach; Hyde, K.D., Pointing, S.B., Eds.; Fungal Diversity Press: Hong Kong, China, 2000; pp. 62–68. [Google Scholar]
- Hyde, K.D.; Sarma, V.V. Pictorial key to higher marine fungi. In Marine Mycology—A Practical Approach; Hyde, K.D., Pointing, S.B., Eds.; Fungal Diversity Press: Hong Kong, China, 2000; pp. 205–270. [Google Scholar]
- Jones, E.B.G.; Sakayaroj, J.; Suetrong, S.; Somrithipol, S.; Pang, K.-L. Classification of marine Ascomycota, anamorphic taxa and Basidiomycota. Fungal Divers. 2009, 35, 1–187. [Google Scholar]
- Kohlmeyer, J.; Volkmann-Kohlmeyer, B. Illustrated key to the filamentous higher marine fungi. Bot. Mar. 1991, 34, 1–61. [Google Scholar] [CrossRef]
- Volkmann-Kohlmeyer, B.; Kohlmeyer, J. How to prepare truly permanent microscope slides. Mycologist 1996, 10, 107–108. [Google Scholar] [CrossRef]
- Schoch, C.L.; Seifert, K.A.; Huhndorf, S.; Robert, V.; Spouge, J.L.; Levesque, C.A.; Chen, W.; Consortium, F.B. Nuclear ribosomal internal transcribed spacer (ITS) region as a universal DNA barcode marker for Fungi. Proc. Natl. Acad. Sci. USA 2012, 109, 6241–6246. [Google Scholar] [CrossRef] [PubMed]
- Fell, J.W. Yeasts in marine environments. In Marine Fungi and Fungal-like Organisms; Jones, E.B.G., Pang, K.-L., Eds.; Walter de Gruyter: Berlin, Germany, 2012; pp. 91–102. [Google Scholar]
- Overy, D.P.; Correa, H.; Roullier, C.; Chi, W.-C.; Pang, K.-L.; Rateb, M.; Ebel, R.; Shang, Z.; Capon, R.; Bills, G.F.; et al. Does osmotic stress affect natural product expression in fungi? Mar. Drugs 2017, 15. [Google Scholar] [CrossRef] [PubMed]
- Campbell, J.; Shearer, C.A.; Mitchell, J.I.; Eaton, R.A. Corollospora revisited: A molecular approach. In Marine Mycology—A Practical Approach; Hyde, K.D., Pointing, S.B., Eds.; Fungal Diversity Press: Hong Kong, China, 2000; pp. 15–33. [Google Scholar]
- Schoch, C.L.; Sung, G.-H.; Volkmann-Kohlmeyer, B.; Kohlmeyer, J.; Spatafora, J.W. Marine fungal lineages in the Hypocreomycetidae. Mycol. Res. 2007, 111, 154–162. [Google Scholar] [CrossRef] [PubMed]
- Schoch, C.L.; Sung, G.-H.; López-Giráldez, F.; Townsend, J.P.; Miadlikowska, J.; Hofstetter, V.; Robbertse, B.; Matheny, P.B.; Kauff, F.; Wang, Z.; et al. The Ascomycota tree of life: A phylum-wide phylogeny clarifies the origin and evolution of fundamental reproductive and ecological traits. Syst. Biol. 2009, 58, 224–239. [Google Scholar] [CrossRef] [PubMed]
- Zuccaro, A.; Schoch, C.L.; Spatafora, J.W.; Kohlmeyer, J.; Draeger, S.; Mitchell, J.I. Detection and identification of fungi intimately associated with the brown seaweed Fucus serratus. Appl. Environ. Microbiol. 2008, 74, 931–941. [Google Scholar] [CrossRef]
- Suetrong, S.; Schoch, C.L.; Spatafora, J.W.; Kohlmeyer, J.; Volkmann-Kohlmeyer, B.; Sakayaroj, J.; Phongpaichit, S.; Tanaka, K.; Hirayama, K.; Jones, E.B.G. Molecular systematics of the marine Dothideomycetes. Stud. Mycol. 2009, 64, 155–173. [Google Scholar] [CrossRef]
- Abraham, S.; Hoang, T.; Alam, M.; Jones, E.G. Chemistry of the cytotoxic principles of the marine fungus Lignincola laevis. Pure Appl. Chem. 1994, 66, 2391–2394. [Google Scholar] [CrossRef]
- Lin, Y.; Wu, X.; Deng, Z.; Wang, J.; Zhou, S.; Vrijmoed, L.; Jones, E.G. The metabolites of the mangrove fungus Verruculina enalia No. 2606 from a salt lake in the Bahamas. Phytochemistry 2002, 59, 469–471. [Google Scholar] [CrossRef]
- Poch, G.K.; Gloer, J.B. Obionin A: A new polyketide metabolite form the marine fungus Leptosphaeria obiones. Tetrahedron Lett. 1989, 30, 3483–3486. [Google Scholar] [CrossRef]
- Poch, G.K.; Gloer, J.B. Helicascolides A and B: New lactones from the marine fungus Helicascus kanaloanus. J. Nat. Prod. 1989, 52, 257–260. [Google Scholar] [CrossRef] [PubMed]





| Technique/Substrate | Substrate | References | Notes |
|---|---|---|---|
| Isolation from sea foam | Seafoam | 9,11,43–45 | Especially suitable for arenicolous species, can yield generalist fungi |
| Direct plating | Any | 41,46 | Important to employ multiple media to ensure growth of targeted fungus |
| Particle filtration and dilution to extinction plating | Any | 2,47–50 | Especially suited for the isolation of slower growing, unique fungi |
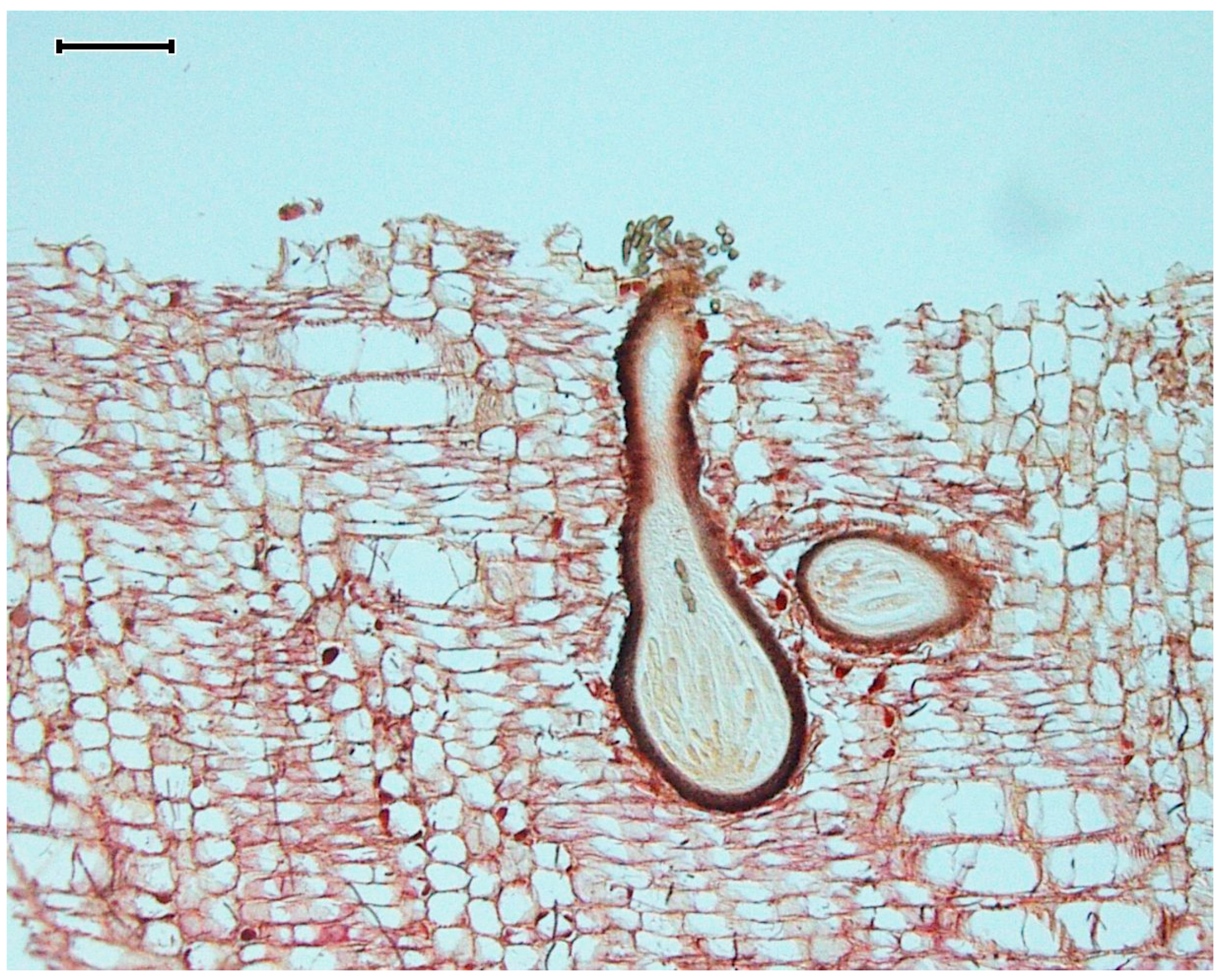
| Damp chambers | Driftwood, various of macroorganisms | 40–42,51–53 | Important to employ preventative measures to avoid mite contamination |
| Baiting stations | Wood | 54–55 | Encrusting invertebrates can be problematic |
| In situ culturing | Any | 56–60 | Novel approach, has the potential to isolate truly unique species |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Overy, D.P.; Rämä, T.; Oosterhuis, R.; Walker, A.K.; Pang, K.-L. The Neglected Marine Fungi, Sensu stricto, and Their Isolation for Natural Products’ Discovery. Mar. Drugs 2019, 17, 42. https://doi.org/10.3390/md17010042
Overy DP, Rämä T, Oosterhuis R, Walker AK, Pang K-L. The Neglected Marine Fungi, Sensu stricto, and Their Isolation for Natural Products’ Discovery. Marine Drugs. 2019; 17(1):42. https://doi.org/10.3390/md17010042
Chicago/Turabian StyleOvery, David P., Teppo Rämä, Rylee Oosterhuis, Allison K. Walker, and Ka-Lai Pang. 2019. "The Neglected Marine Fungi, Sensu stricto, and Their Isolation for Natural Products’ Discovery" Marine Drugs 17, no. 1: 42. https://doi.org/10.3390/md17010042
APA StyleOvery, D. P., Rämä, T., Oosterhuis, R., Walker, A. K., & Pang, K.-L. (2019). The Neglected Marine Fungi, Sensu stricto, and Their Isolation for Natural Products’ Discovery. Marine Drugs, 17(1), 42. https://doi.org/10.3390/md17010042





