Potential Usefulness of a Wakame/Carob Functional Snack for the Treatment of Several Aspects of Metabolic Syndrome: From In Vitro to In Vivo Studies
Abstract
:1. Introduction
2. Results
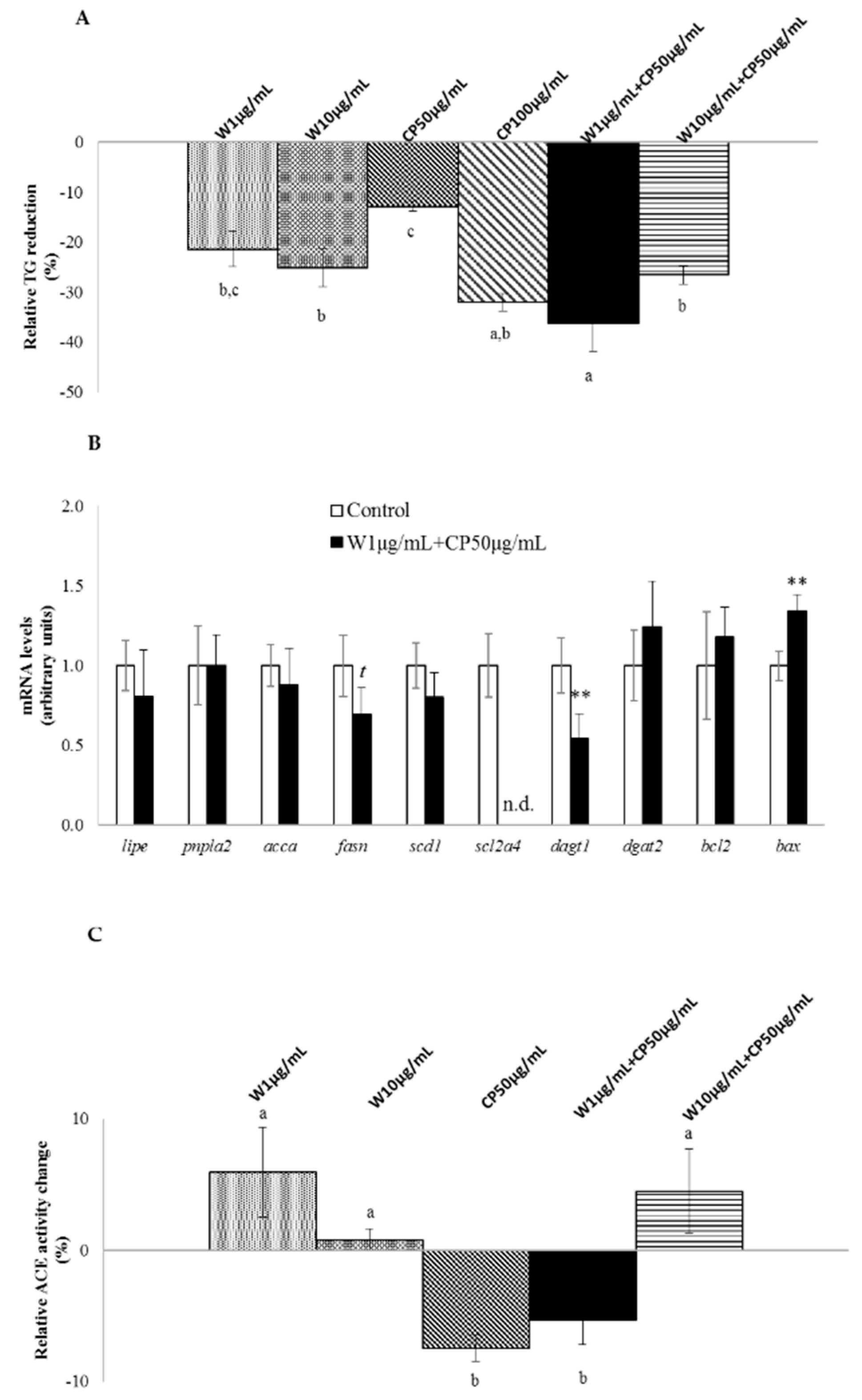
2.1. In Vitro Fat-Lowering Effect Evaluation and Anti-Hypertensive Activity of W, CP, and Their Combined Extracts
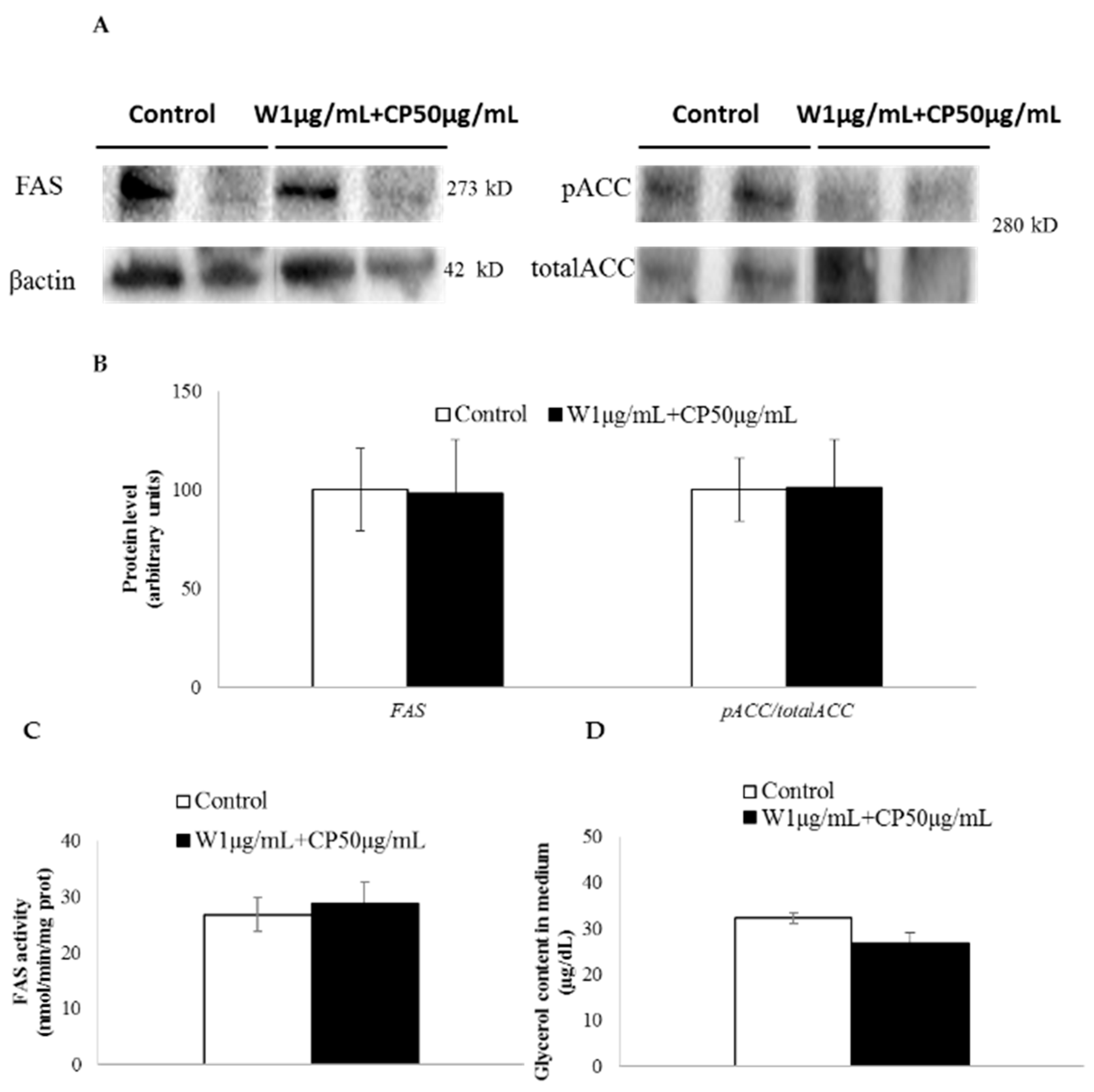
2.2. Gene Expression, Post-Transcriptional Lipolysis, and de Novo Lipogenesis Evaluation in Mature Adipocytes Treated with W and CP Combined Extracts
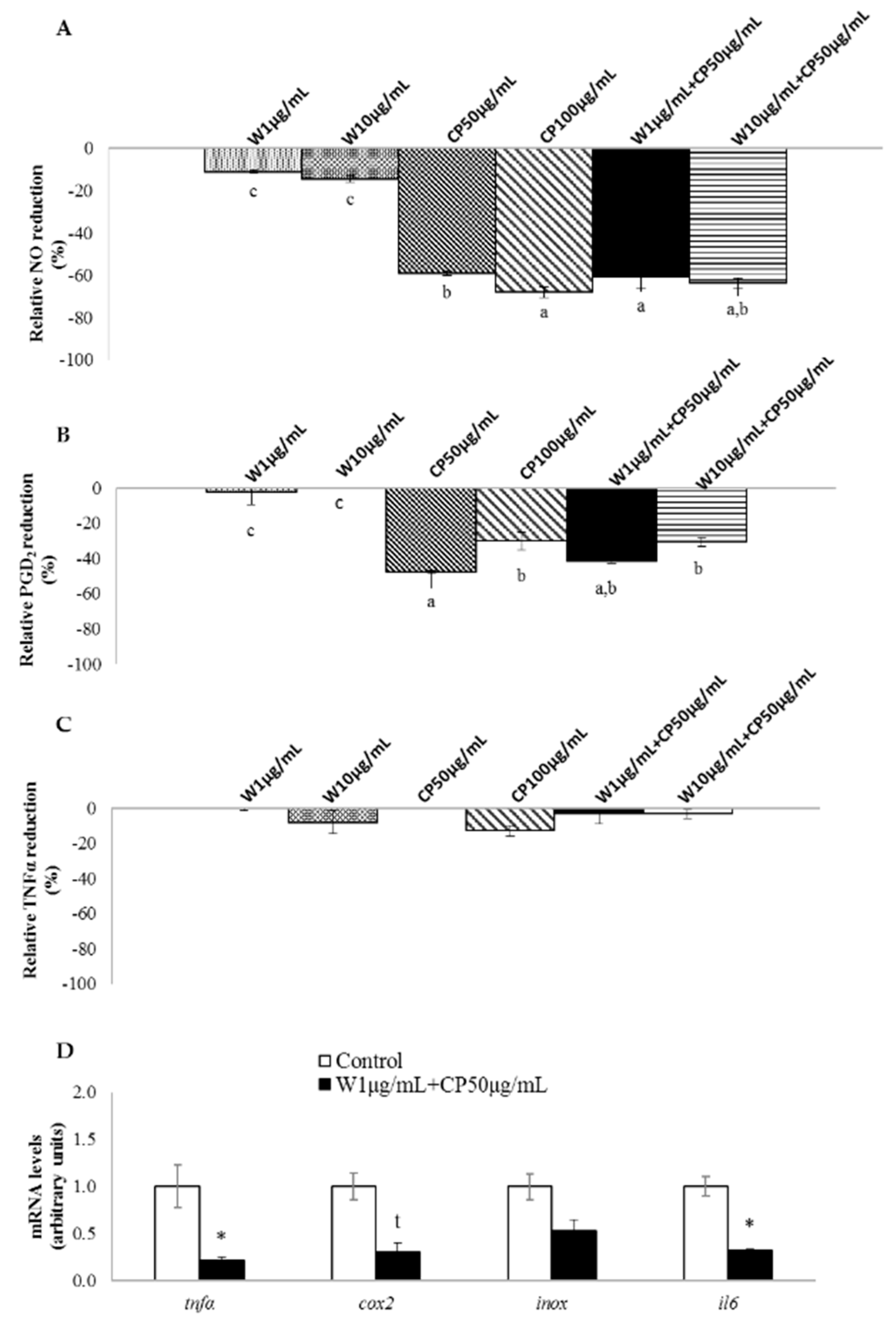
2.3. In Vitro Anti-Inflammatory of W, CP, and Their Combined Extracts
2.4. Gene Expression of RAW 264.7 Cells Treated with W and CP Combination
2.5. Composition of Functional Snacks
2.6. Food/Energy/Liquid Intake, Total Body Weights, White Adipose Tissue Weights, Interscapular Brown Adipose Tissue Weights, and Gastrocnemius Muscle Weights in Rats Treated with Functional Snacks
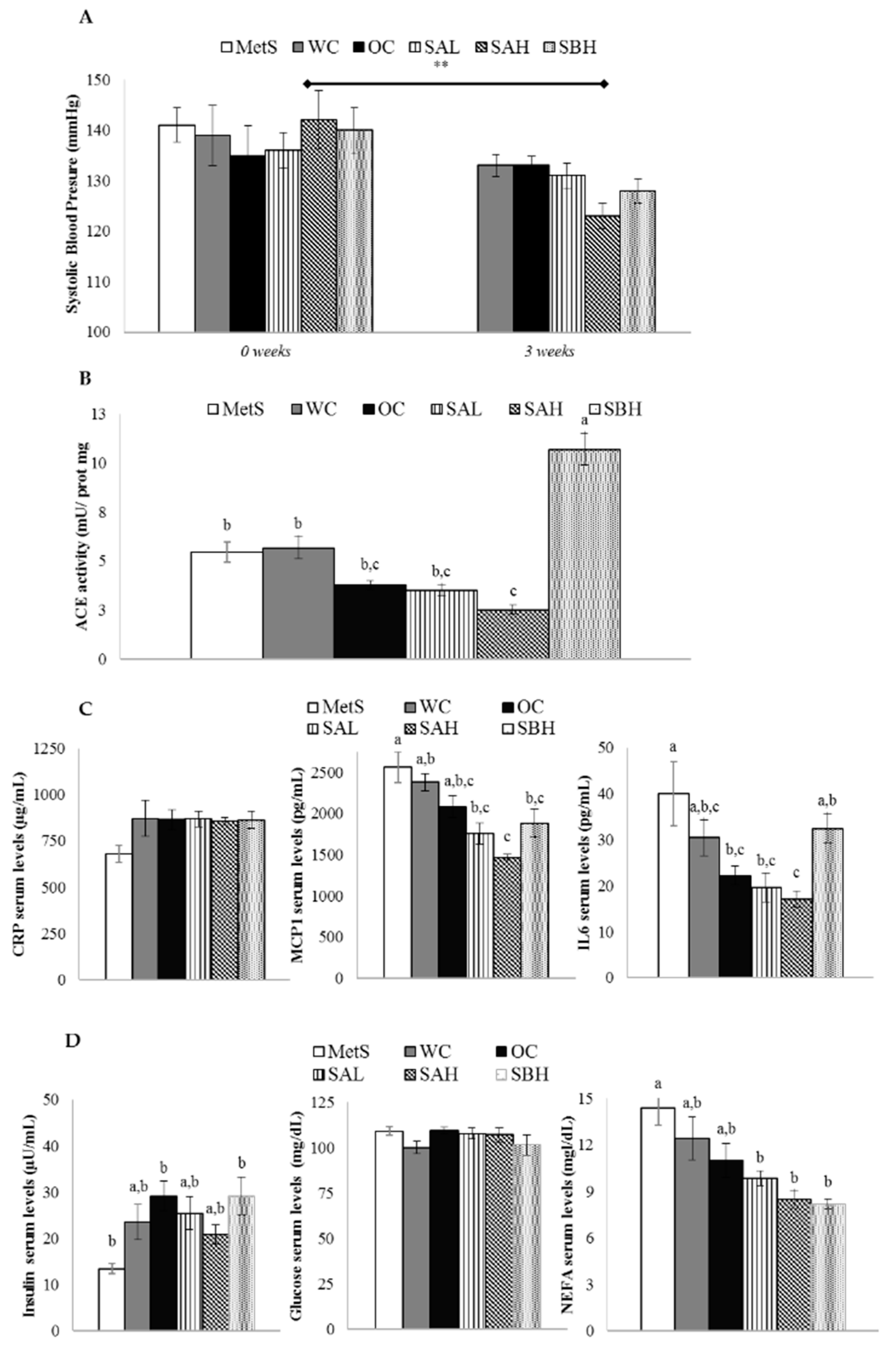
2.7. Blood Pressure and Renal ACE Activity in Rats Treated with Functional Snacks
2.8. Anti-Inflammatory Activity in Rats Treated with Functional Snacks
2.9. Functional Snacks Effect on Glycemic Control in MetS Rats
3. Discussion
4. Materials and Methods
4.1. Ethical Approval
4.2. Reagents
4.3. Raw Material
4.4. Carob Pod and Wakame Extract Preparation
4.5. In Vitro Assays: Anti-Hypertensive, Triacylglycerol-Lowering, and Anti-Inflammatory Activities
4.6. RNA Extraction and Reverse Transcription Polymerase Chain Reaction (RT-PCR)
4.7. Western Blot Analysis
4.8. Fatty Acid Synthase Activity
4.9. Snack Preparation
4.10. Animals and Treatment Protocols
4.11. Anti-Hypertensive, Anti-Inflammatory, and Glycemic Control Improvement Properties Evaluation of Functional Snack in MetS Rats
4.12. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Saklayen, M.G. The global epidemic of the metabolic syndrome. Curr. Hypertens. Rep. 2018, 20, 12. [Google Scholar] [CrossRef]
- National Cholesterol Education Program (NCEP) Expert Panel on Detection, E.a., and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). Third report of the national cholesterol education program (NCEP) expert panel on detection, evaluation, and treatment of high blood cholesterol in adults (adult treatment panel III) final report. Circulation 2002, 106, 3143–3421. [Google Scholar] [CrossRef]
- Hosseini, Z.; Whiting, S.J.; Vatanparast, H. Current evidence on the association of the metabolic syndrome and dietary patterns in a global perspective. Nutr. Res. Rev. 2016, 29, 152–162. [Google Scholar] [CrossRef] [PubMed]
- Monteiro, R.; Azevedo, I. Chronic inflammation in obesity and the metabolic syndrome. Mediat. Inflamm. 2010, 2010. [Google Scholar] [CrossRef] [PubMed]
- Arner, P. The adipocyte in insulin resistance: Key molecules and the impact of the thiazolidinediones. Trends Endocrinol. Metab. 2003, 14, 137–145. [Google Scholar] [CrossRef]
- Rutkowski, J.M.; Stern, J.H.; Scherer, P.E. The cell biology of fat expansion. J. Cell Biol. 2015, 208, 501. [Google Scholar] [CrossRef] [PubMed]
- Pucci, A.; Finer, N. New medications for treatment of obesity: Metabolic and cardiovascular effects. Can. J. Cardiol. 2015, 31, 142–152. [Google Scholar] [CrossRef] [PubMed]
- Amiot, M.J.; Riva, C.; Vinet, A. Effects of dietary polyphenols on metabolic syndrome features in humans: A systematic review. Obesity Reviews 2016, 17, 573–586. [Google Scholar] [CrossRef] [PubMed]
- Governa, P.; Baini, G.; Borgonetti, V.; Cettolin, G.; Giachetti, D.; Magnano, R.A.; Miraldi, E.; Biagi, M. Phytotherapy in the management of diabetes: A review. Molecules 2018, 23, 105. [Google Scholar] [CrossRef]
- Wan-Loy, C.; Siew-Moi, P. Marine algae as a potential source for anti-obesity agents. Mar. Drugs 2016, 14, 222. [Google Scholar] [CrossRef] [PubMed]
- Fung, A.; Hamid, N.; Lu, J. Fucoxanthin content and antioxidant properties of Undaria pinnatifida. Food Chem. 2013, 136, 1055–1062. [Google Scholar] [CrossRef]
- Billakanti, J.M.; Catchpole, O.J.; Fenton, T.A.; Mitchell, K.A.; MacKenzie, A.D. Enzyme-assisted extraction of fucoxanthin and lipids containing polyunsaturated fatty acids from Undaria pinnatifida using dimethyl ether and ethanol. Process Biochem. 2013, 48, 1999–2008. [Google Scholar] [CrossRef]
- Sánchez-Machado, D.I.; López-Hernández, J.; Paseiro-Losada, P.; López-Cervantes, J. An HPLC method for the quantification of sterols in edible seaweeds. Biomed. Chromatogr. 2004, 18, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Suetsuna, K.; Nakano, T. Identification of an antihypertensive peptide from peptic digest of wakame (Undaria pinnatifida). J. Nutr. Biochem. 2000, 11, 450–454. [Google Scholar] [CrossRef]
- Grasa-López, A.; Miliar-García, Á.; Quevedo-Corona, L.; Paniagua-Castro, N.; Escalona-Cardoso, G.; Reyes-Maldonado, E.; Jaramillo-Flores, M.E. Undaria pinnatifida and fucoxanthin ameliorate lipogenesis and markers of both inflammation and cardiovascular dysfunction in an animal model of diet-induced obesity. Mar. Drugs 2016, 14, 148. [Google Scholar] [CrossRef]
- Kim, B.-M.; Park, J.-H.; Kim, D.-S.; Kim, Y.-M.; Jun, J.-Y.; Jeong, I.-H.; Chi, Y.-M. Effects of the polysaccharide from the sporophyll of brown alga Undaria Pinnatifida on serum lipid profile and fat tissue accumulation in rats fed a high-fat diet. J. Food Sci. 2016, 81, H1840–H1845. [Google Scholar] [CrossRef]
- Rico, D.; Diana, A.B.M.; Milton-Laskibar, I.; Fernández-Quintela, A.; Silván, J.M.; Rai, D.K.; Choudhary, A.; Peñas, E.; de Luis, D.A.; Martínez-Villaluenga, C. Characterization and in vitro evaluation of seaweed species as potential functional ingredients to ameliorate metabolic syndrome. J. Funct. Foods 2018, 46, 185–194. [Google Scholar] [CrossRef]
- Clark, L.J.; Taylor, G.C.; Zahradka, P. Rebelling against the (Insulin) resistance: A review of the proposed insulin-sensitizing actions of soybeans, chickpeas, and their bioactive compounds. Nutrients 2018, 10, 434. [Google Scholar] [CrossRef]
- Goulas, V.; Stylos, E.; Chatziathanasiadou, V.M.; Mavromoustakos, T.; Tzakos, G.A. Functional components of carob fruit: Linking the chemical and biological space. Int. J. Mol. Sci. 2016, 17, 1875. [Google Scholar] [CrossRef]
- Papagiannopoulos, M.; Wollseifen, H.R.; Mellenthin, A.; Haber, B.; Galensa, R. Identification and quantification of polyphenols in carob fruits (Ceratonia siliqua L.) and derived products by HPLC-UV-ESI/MSn. J. Agric. Food Chem. 2004, 52, 3784–3791. [Google Scholar] [CrossRef]
- Oh, J.H.; Kim, J.; Lee, Y. Anti-inflammatory and anti-diabetic effects of brown seaweeds in high-fat diet-induced obese mice. Nutr. Res. Pract. 2016, 10, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Braithwaite, M.C.; Tyagi, C.; Tomar, L.K.; Kumar, P.; Choonara, Y.E.; Pillay, V. Nutraceutical-based therapeutics and formulation strategies augmenting their efficiency to complement modern medicine: An overview. J. Funct. Foods 2014, 6, 82–99. [Google Scholar] [CrossRef]
- Dandona, P.; Aljada, A.; Chaudhuri, A.; Mohanty, P.; Garg, R. Metabolic syndrome: A comprehensive perspective based on interactions between obesity, diabetes, and inflammation. Circulation 2005, 111, 1448–1454. [Google Scholar] [CrossRef] [PubMed]
- Rico, D.; Alonso-De Linaje, A.; Herrero, A.; Asensio-Vegas, C.; Miranda, J.; Martínez-Villaluenga, C.; De Luis, D.A.; Martin-Diana, A.B. Carob by-products and seaweeds for the development of functional bread. J. Food Process. Preserv. 2018, 42. [Google Scholar] [CrossRef]
- Scalbert, A.; Williamson, G. Dietary intake and bioavailability of polyphenols. J. Nutr. 2000, 130, 2073S–2085S. [Google Scholar] [CrossRef] [PubMed]
- Langhans, W. Food components in health promotion and disease prevention. J. Agric. Food Chem. 2018, 66, 2287–2294. [Google Scholar] [CrossRef]
- Zhang, H.; Tang, Y.; Zhang, Y.; Zhang, S.; Qu, J.; Wang, X.; Kong, R.; Han, C.; Liu, Z. Fucoxanthin: A promising medicinal and nutritional ingredient. Evid. Based Complement. Alternat. Med. 2015, 2015, 723515. [Google Scholar] [CrossRef]
- Ydjedd, S.; Bouriche, S.; López-Nicolás, R.; Sánchez-Moya, T.; Frontela-Saseta, C.; Ros-Berruezo, G.; Rezgui, F.; Louaileche, H.; Kati, D.E. Effect of in vitro gastrointestinal digestion on encapsulated and nonencapsulated phenolic compounds of carob (Ceratonia siliqua L.) pulp extracts and their antioxidant capacity. J. Agric. Food Chem. 2017, 65, 827–835. [Google Scholar] [CrossRef]
- Bajerska, J.; Chmurzynska, A.; Mildner-Szkudlarz, S.; Drzymała-Czyż, S. Effect of rye bread enriched with tomato pomace on fat absorption and lipid metabolism in rats fed a high-fat diet. J. Sci. Food Agric. 2015, 95, 1918–1924. [Google Scholar] [CrossRef]
- Paturi, G.; Butts, C.A.; Bentley-Hewitt, K.L.; Ansell, J. Influence of green and gold kiwifruit on indices of large bowel function in healthy rats. J. Food Sci. 2014, 79, H1611–H1620. [Google Scholar] [CrossRef]
- Mele, L.; Bidault, G.; Mena, P.; Crozier, A.; Brighenti, F.; Vidal-Puig, A.; Del Rio, D. Dietary (Poly)phenols, brown adipose tissue activation, and energy expenditure: A narrative review. Adv. Nutr. 2017, 8, 694–704. [Google Scholar] [CrossRef] [PubMed]
- Gammone, M.A.; D’Orazio, N. Anti-obesity activity of the marine carotenoid fucoxanthin. Mar. Drugs 2015, 13, 2196–2214. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.T.; Chou, H.N.; Huang, C.J. Dietary fucoxanthin increases metabolic rate and upregulated mRNA expressions of the PGC-1alpha network, mitochondrial biogenesis and fusion genes in white adipose tissues of mice. Mar. Drugs 2014, 12, 964–982. [Google Scholar] [CrossRef] [PubMed]
- Bartelt, A.; Widenmaier, S.B.; Schlein, C.; Johann, K.; Goncalves, R.L.S.; Eguchi, K.; Fischer, A.W.; Parlakgül, G.; Snyder, N.A.; Nguyen, T.B.; et al. Brown adipose tissue thermogenic adaptation requires Nrf1-mediated proteasomal activity. Nat. Med. 2018, 24, 292–303. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, T.W.; Miranda, J.; Teixeira, L.; Aiastui, A.; Matheu, A.; Gambero, A.; Portillo, M.P.; Ribeiro, M.L. Yerba mate stimulates mitochondrial biogenesis and thermogenesis in high-fat-diet-induced obese mice. Mol. Nutr. Food Res. 2018, e1800142. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Li, J.; Li, Z.; Sang, Y.; Niu, Y.; Zhang, Q.; Ding, H.; Yin, S. Fucoidan from Undaria pinnatifida prevents vascular dysfunction through PI3K/Akt/eNOS-dependent mechanisms in the l-NAME-induced hypertensive rat model. Food Funct. 2016, 7, 2398–2408. [Google Scholar] [CrossRef] [PubMed]
- Park, H.J.; Lee, M.K.; Park, Y.B.; Shin, Y.C.; Choi, M.S. Beneficial effects of Undaria pinnatifida ethanol extract on diet-induced-insulin resistance in C57BL/6J mice. Food Chem. Toxicol. 2011, 49, 727–733. [Google Scholar] [CrossRef] [PubMed]
- Milton-Laskibar, I.; Gómez-Zorita, S.; Aguirre, L.; Fernández-Quintela, A.; González, M.; Portillo, M.P. Resveratrol-induced effects on body fat differ depending on feeding conditions. Molecules 2017, 22, 2091. [Google Scholar] [CrossRef] [PubMed]
- Shalaby, S.M.; Zakora, M.; Otte, J. Performance of two commonly used angiotensin-converting enzyme inhibition assays using FA-PGG and HHL as substrates. J. Dairy Res. 2006, 73, 178–186. [Google Scholar] [CrossRef]
- Eseberri, I.; Miranda, J.; Lasa, A.; Churruca, I.; Portillo, M.P. Doses of quercetin in the range of serum concentrations exert delipidating effects in 3T3-L1 preadipocytes by acting on different stages of adipogenesis, but not in mature adipocytes. Oxid. Med. Cell Longev. 2015, 2015, 480943. [Google Scholar] [CrossRef]
- González-Montoya, M.; Hernández-Ledesma, B.; Silván, J.M.; Mora-Escobedo, R.; Martínez-Villaluenga, C. Peptides derived from in vitro gastrointestinal digestion of germinated soybean proteins inhibit human colon cancer cells proliferation and inflammation. Food Chem. 2018, 242, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Yoon, J.H.; Won, H.J.; Ji, H.S.; Yuk, H.J.; Park, K.H.; Park, H.Y.; Jeong, T.S. Isotrifoliol inhibits pro-inflammatory mediators by suppression of TLR/NF-κB and TLR/MAPK signaling in LPS-induced RAW264.7 cells. Int. Immunopharmacol. 2017, 45, 110–119. [Google Scholar] [CrossRef]
- Ariemma, F.; D’Esposito, V.; Liguoro, D.; Oriente, F.; Cabaro, S.; Liotti, A.; Cimmino, I.; Longo, M.; Beguinot, F.; Formisano, P.; et al. Low-dose bisphenol—A impairs adipogenesis and generates dysfunctional 3T3-L1 Adipocytes. PLoS ONE 2016, 11, e0150762. [Google Scholar] [CrossRef] [PubMed]
- Zhong, W.; Zhao, Y.; Sun, X.; Song, Z.; McClain, C.J.; Zhou, Z. Dietary zinc deficiency exaggerates ethanol-induced liver injury in mice: Involvement of intrahepatic and extrahepatic factors. PLoS ONE 2013, 8, e76522. [Google Scholar] [CrossRef] [PubMed]
- Martin-Diana, A.B.; Izquierdo, N.; Albertos, I.; Sanchez, M.S.; Herrero, A.; Sanz, M.A.; Rico, D. Valorization of carob’s germ and seed peel as natural antioxidant ingredients in gluten-free crackers. J. Food Process. Preserv. 2016, 41, e12770. [Google Scholar] [CrossRef]
- Reeves, P.; Nielsen, F.; Fahey, G.J. AIN-93 purified diets for laboratory rodents: Final report of the American Institute of Nutrition ad hoc writing committee on the reformulation of the AIN-76A rodent diet. J. Nutr. 1993, 123, 1939–1951. [Google Scholar] [CrossRef] [PubMed]
| MetS n = 10 | WC n = 10 | OC n = 10 | SAL n = 10 | SAH n = 10 | SBH n = 10 | |
|---|---|---|---|---|---|---|
| Food intake (g/day) | 20.8 ± 0.5 b | 25.2 ± 1.4 a,b | 28.4 ± 1.3 a | 28.7 ± 1.2 a | 25.5 ± 1.0 a,b | 28.9 ± 1.5 a |
| Energy intake (kJ/day) | 391 ± 9 b | 418 ± 24 a,b | 471 ± 22 a | 473 ± 20 a,b | 409 ± 19 a,b | 463 ± 24 a,b |
| Water intake (mL/day) | 21.6 ± 0.9 b | 30.3 ± 0.2 a | 28.3 ± 1.1 a | 27.0 ± 1.1 a | 29.1 ± 0.7 a | 30.4 ± 1.9 a |
| Initial body weight (g) | 202 ± 2.5 | 202 ± 2.8 | 202 ± 2.5 | 203 ± 2.5 | 203 ± 2.6 | 204 ± 2.7 |
| Final body weight (g) | 411 ± 10.3 | 456 ± 13.8 | 449 ± 4.9 | 441 ± 10.5 | 441 ± 8.3 | 448 ± 11.8 |
| White adipose tissue weights | ||||||
| Epididymal (g) | 11.7 ± 1.6 | 11.2 ± 1.2 | 12.2 ± 1.0 | 11.3 ± 0.8 | 11.7 ± 1.2 | 10.6 ± 1.2 |
| Perirenal (g) | 15.5 ± 1.4 | 16.1 ± 1.8 | 14.9 ± 1.0 | 14.5 ± 1.2 | 16.3 ± 1.5 | 14.8 ± 0.9 |
| Mesenteric (g) | 4.1 ± 0.4 | 5.0 ± 0.6 | 4.8 ± 0.3 | 5.2 ± 0.6 | 4.9 ± 0.4 | 4.4 ± 0.3 |
| Subcutaneous (g) | 14.1 ± 1.3 | 14.8 ± 1.6 | 13.9 ± 1.0 | 13.8 ± 1.0 | 14.0 ± 0.8 | 12.7 ± 0.4 |
| ∑ Adipose tissue (g) | 45.4 ± 4.6 | 47.1 ± 4.9 | 45.8 ± 3.1 | 44.7 ± 3.5 | 46.9 ± 11.1 | 42.7 ± 2.9 |
| ∑ Adipose tissue/body weight (%) | 10.9 ± 0.8 | 10.3 ± 0.9 | 10.2 ± 0.6 | 10.1 ± 0.7 | 9.4 ± 1.3 | 9.6 ± 0.7 |
| IBAT weight (g) | 1.46 ± 0.04 | 1.56 ± 0.10 | 1.40 ± 0.14 | 1.52 ± 0.08 | 1.57 ± 0.06 | 1.56 ± 0.08 |
| ∑ Gastrocnemius muscle weight (g) | 4.58 ± 0.14 | 4.75 ± 0.16 | 4.18 ± 0.22 | 4.56 ± 0.13 | 4.90 ± 0.20 | 4.51 ± 0.26 |
| Primers | Sense Primer | Antisense Primer | Ref. | Annealing T(°C) |
|---|---|---|---|---|
| β-actin | 5′-ACG AGG CCC AGA GCA AGA G-3′ | 5′-GGT GTG GTG CCA GAT CTT CTC-3′ | 60.0 | |
| bax | 5′-GTG AGC GGC TGC TTG TCT-3′ | 5′-GTG GGG GTC CCG AAG TAG-3′ | 59.0 | |
| bcl2 | 5′-AGT ACC TGA ACC GGC ATC TG-3′ | 5′-GGG GCC ATA TAG TTC CAC AAA-3′ | 59.0 | |
| cox2 | 5′-GCT ACC TGG AGT ACA TGA AG-3′ | 5′-CTG TGA CTC CAG CTT ATC TG-3′ | [43] | 57.3 |
| il6 | 5′-GCT ACC TGG AGT ACA TGA AG-3′ | 5′-GCA TAA CGC ACT AGG TTT GCC-3′ | [44] | 60.0 |
| nos2 | 5′-CTT TGC CAC GGA CGA GAC-3′ | 5′-TCA TTG TAC TCT GAG GGC TGA C-3′ | 63.7 | |
| scd1 | 5′-TTC TTG CGA TAC ACT CTG GTG C-3′ | 5′-CGG GAT TGA ATG TTC TTG TCG T-3′ | [45] | 64.5 |
| tnfa | 5′-TCA CCT GCT GCT ACT CAT TC-3′ | 5′-TAC AGA AGT GCT TGA GGT GG-3′ | [43] | 62.7 |
| Ingredients | WC (g/kg) | OC (g/kg) | SAL (g/kg) | SAH (g/kg) | SBH (g/kg) |
|---|---|---|---|---|---|
| Casein 1 | 200 | 200 | 200 | 200 | 200 |
| Sucrose 2 | 230 | 230 | 230 | 230 | 230 |
| Wheat starch 3 | 400 | 200 | 320 | - | - |
| Oat starch 3 | - | 200 | - | - | - |
| Snack A | - | - | 80 | 400 | - |
| Snack B | - | - | - | - | 400 |
| Sunflower oil 2 | 70 | 70 | 70 | 70 | 70 |
| Cellulose 3 | 46 | 46 | 46 | 46 | 46 |
| Mineral mix 4 | 35 | 35 | 35 | 35 | 35 |
| Vitamin mix 4 | 11 | 11 | 11 | 11 | 11 |
| l-methionine 1 | 4 | 4 | 4 | 4 | 4 |
| Choline chlorhydrate 1 | 4 | 4 | 4 | 4 | 4 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martínez-Villaluenga, C.; Peñas, E.; Rico, D.; Martin-Diana, A.B.; Portillo, M.P.; Macarulla, M.T.; De Luis, D.A.; Miranda, J. Potential Usefulness of a Wakame/Carob Functional Snack for the Treatment of Several Aspects of Metabolic Syndrome: From In Vitro to In Vivo Studies. Mar. Drugs 2018, 16, 512. https://doi.org/10.3390/md16120512
Martínez-Villaluenga C, Peñas E, Rico D, Martin-Diana AB, Portillo MP, Macarulla MT, De Luis DA, Miranda J. Potential Usefulness of a Wakame/Carob Functional Snack for the Treatment of Several Aspects of Metabolic Syndrome: From In Vitro to In Vivo Studies. Marine Drugs. 2018; 16(12):512. https://doi.org/10.3390/md16120512
Chicago/Turabian StyleMartínez-Villaluenga, Cristina, Elena Peñas, Daniel Rico, Ana Belén Martin-Diana, Maria P. Portillo, Maria Teresa Macarulla, Daniel Antonio De Luis, and Jonatan Miranda. 2018. "Potential Usefulness of a Wakame/Carob Functional Snack for the Treatment of Several Aspects of Metabolic Syndrome: From In Vitro to In Vivo Studies" Marine Drugs 16, no. 12: 512. https://doi.org/10.3390/md16120512
APA StyleMartínez-Villaluenga, C., Peñas, E., Rico, D., Martin-Diana, A. B., Portillo, M. P., Macarulla, M. T., De Luis, D. A., & Miranda, J. (2018). Potential Usefulness of a Wakame/Carob Functional Snack for the Treatment of Several Aspects of Metabolic Syndrome: From In Vitro to In Vivo Studies. Marine Drugs, 16(12), 512. https://doi.org/10.3390/md16120512












