Characterization of a Novel N-Acylhomoserine Lactonase RmmL from Ruegeria mobilis YJ3
Abstract
1. Introduction
2. Results
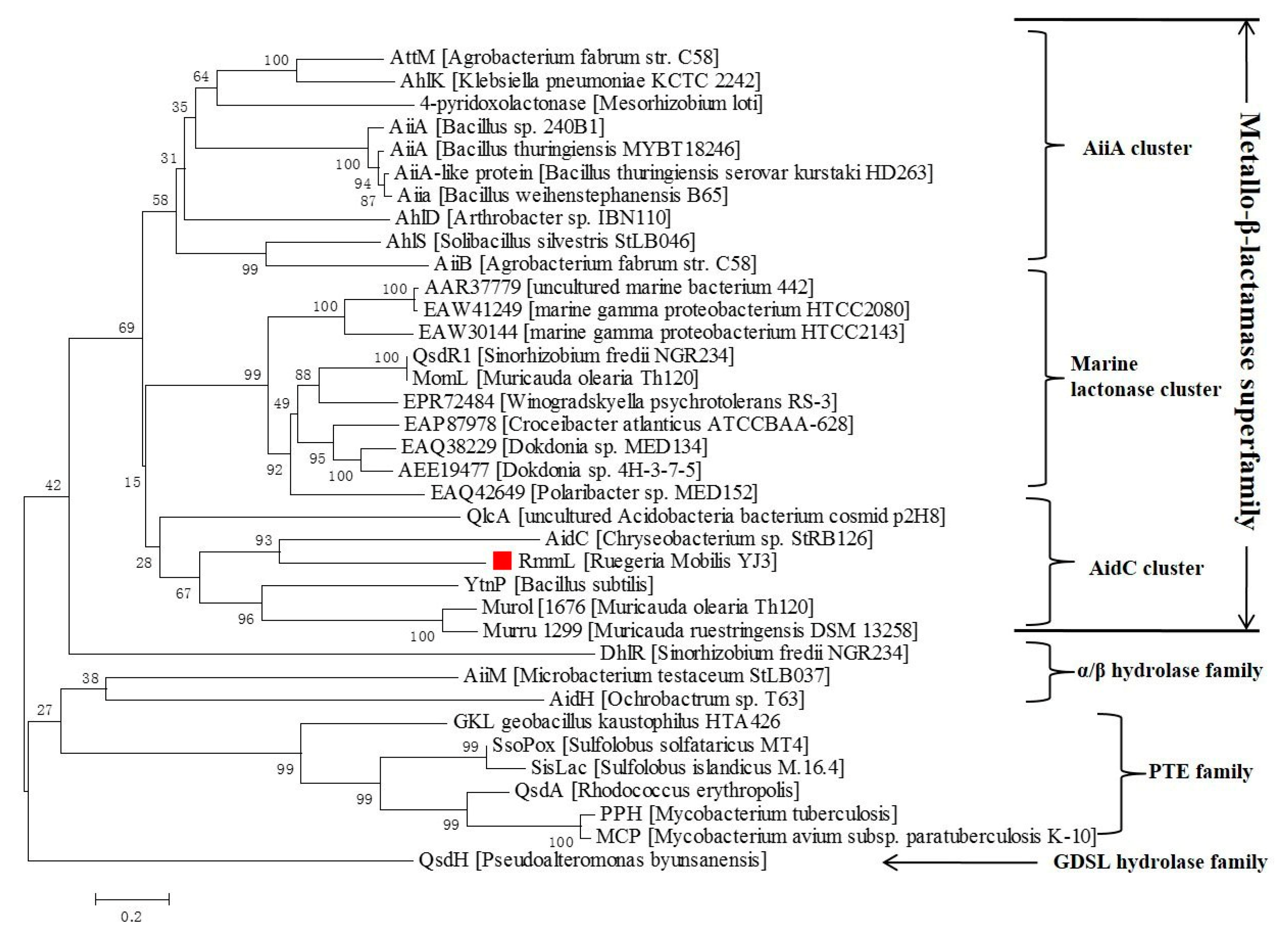
2.1. Identification of the Extracelluar QQ Enzyme in YJ3
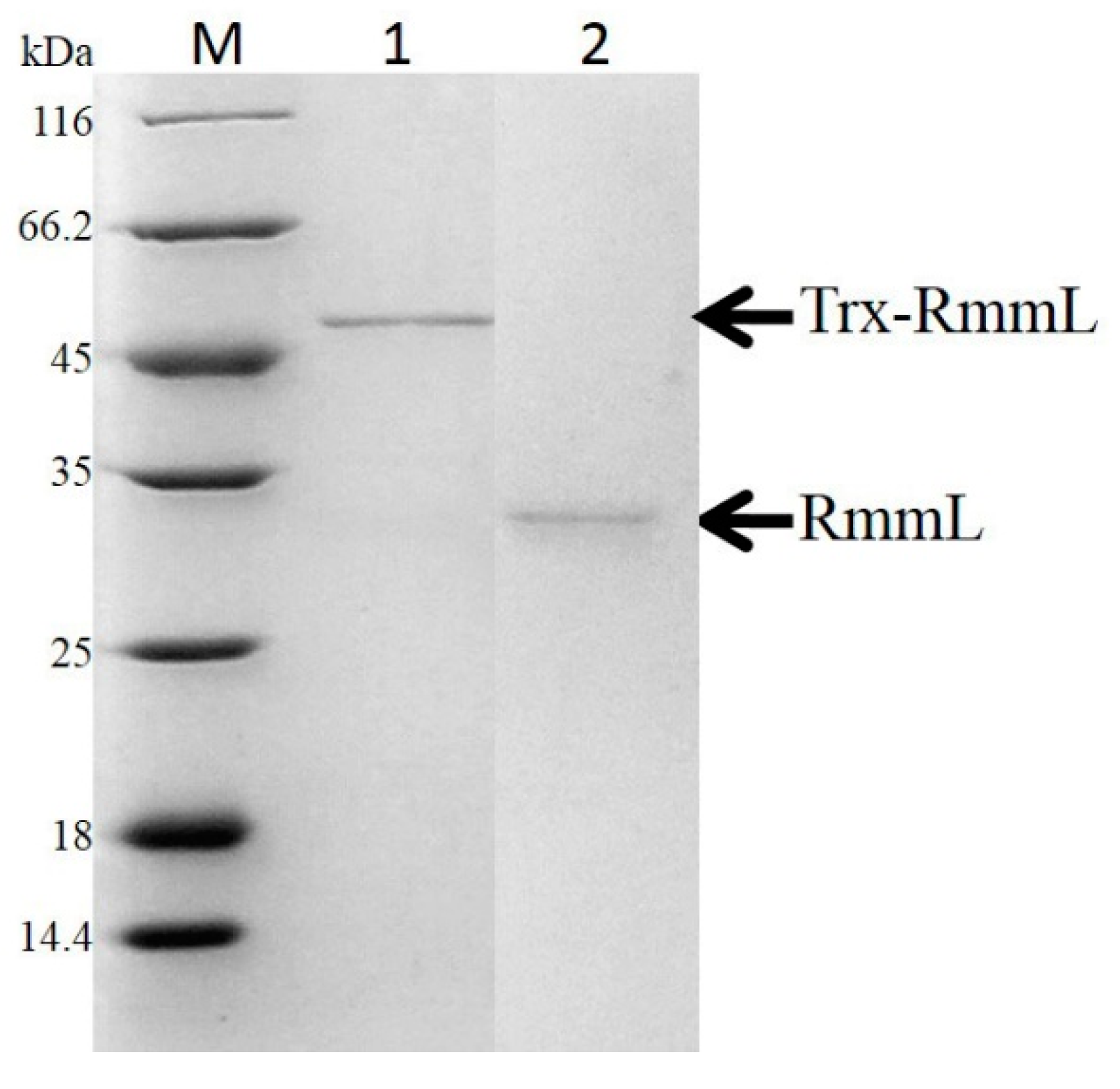
2.2. Expression, Purification and Activity Test of RmmL in YJ3
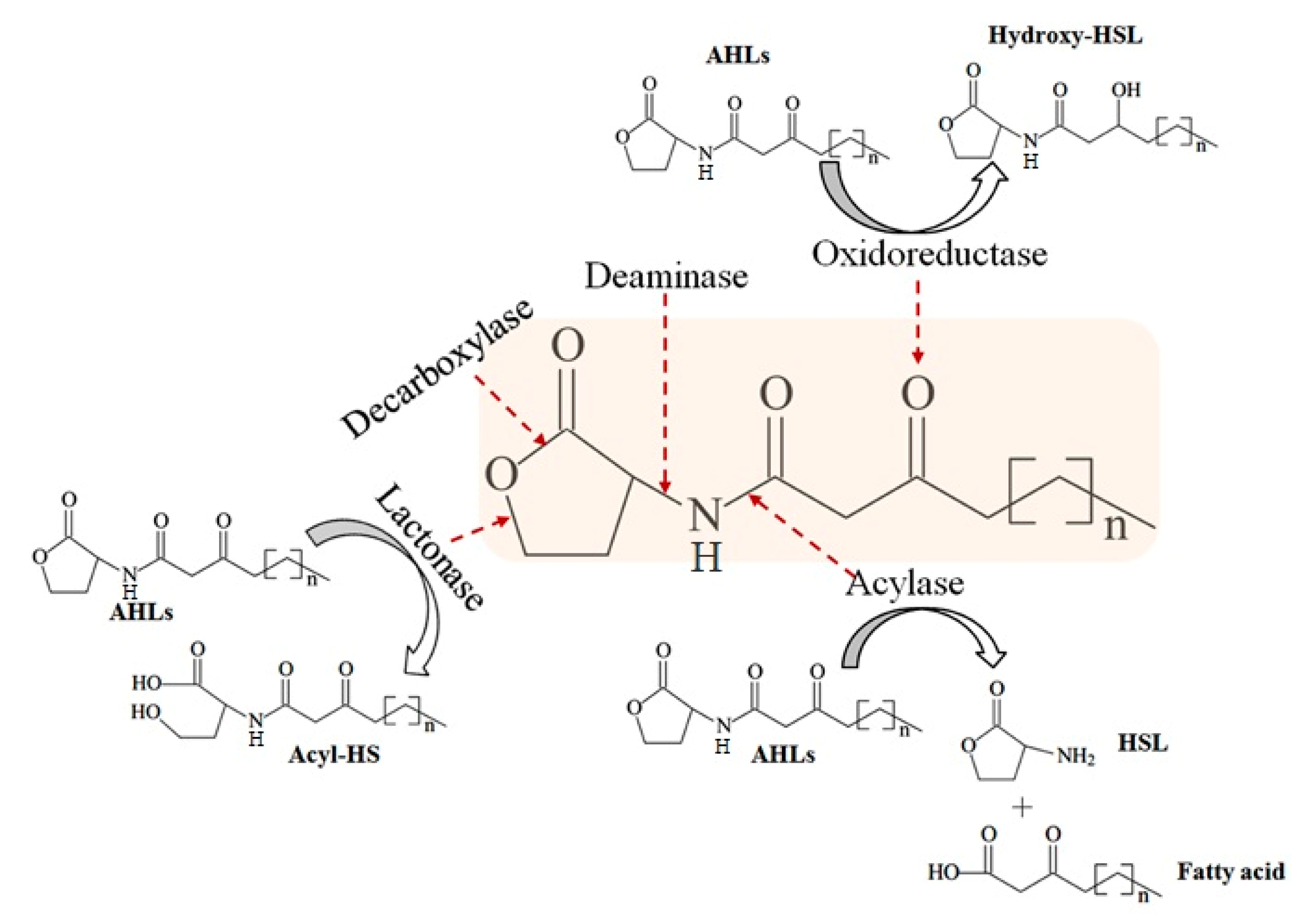
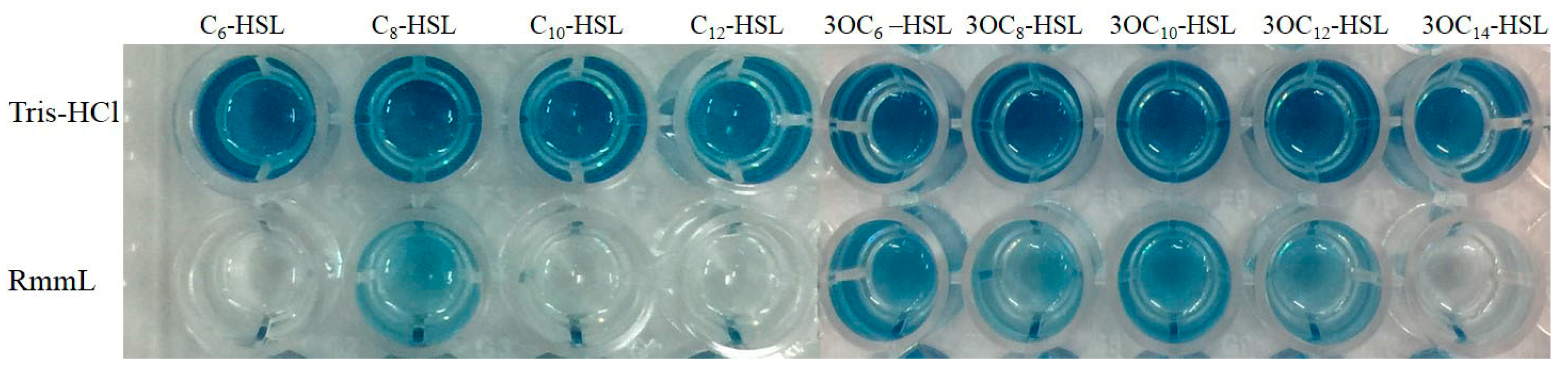
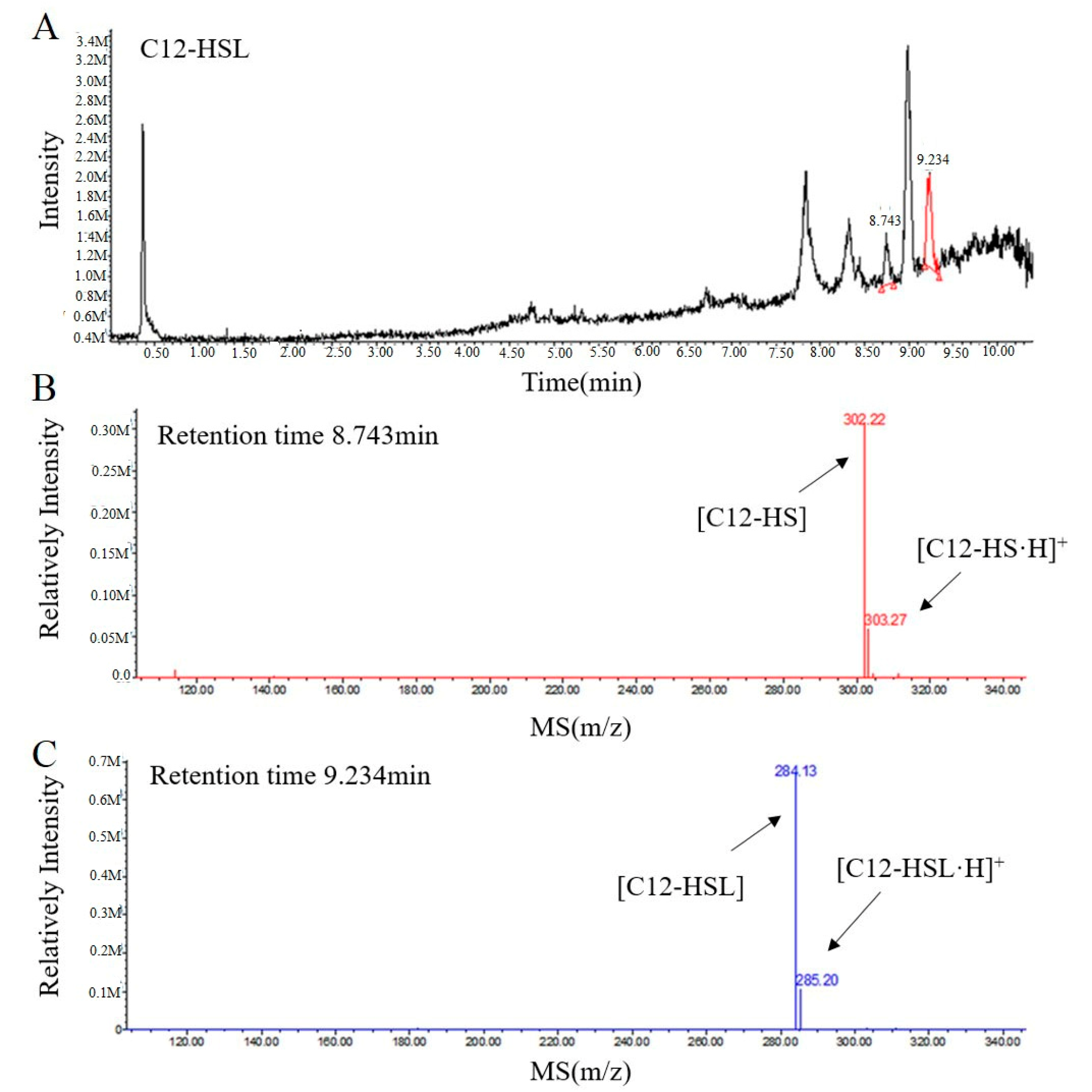
2.3. RmmL is an AHL Lactonase
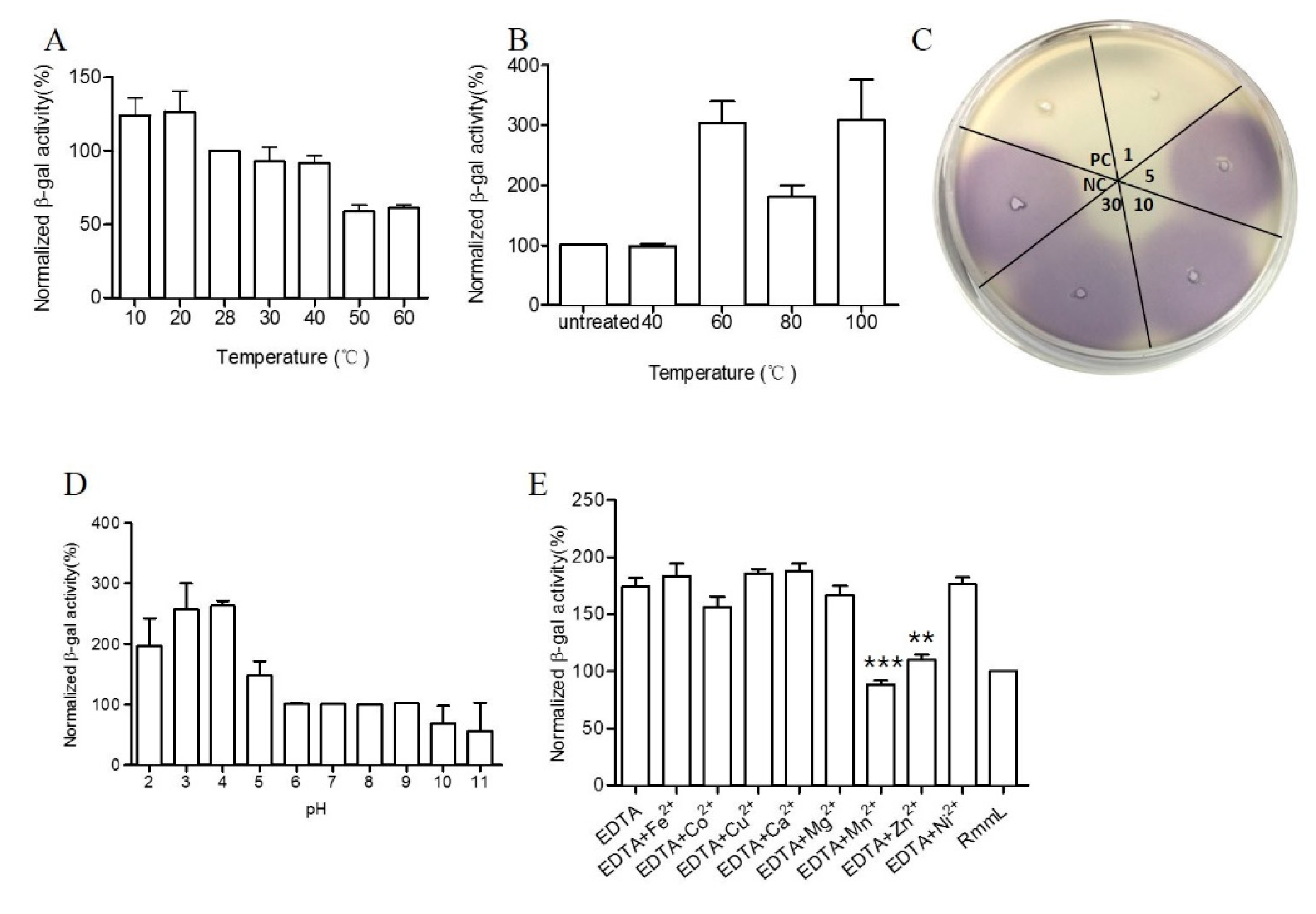
2.4. Biochemical Characterization of RmmL
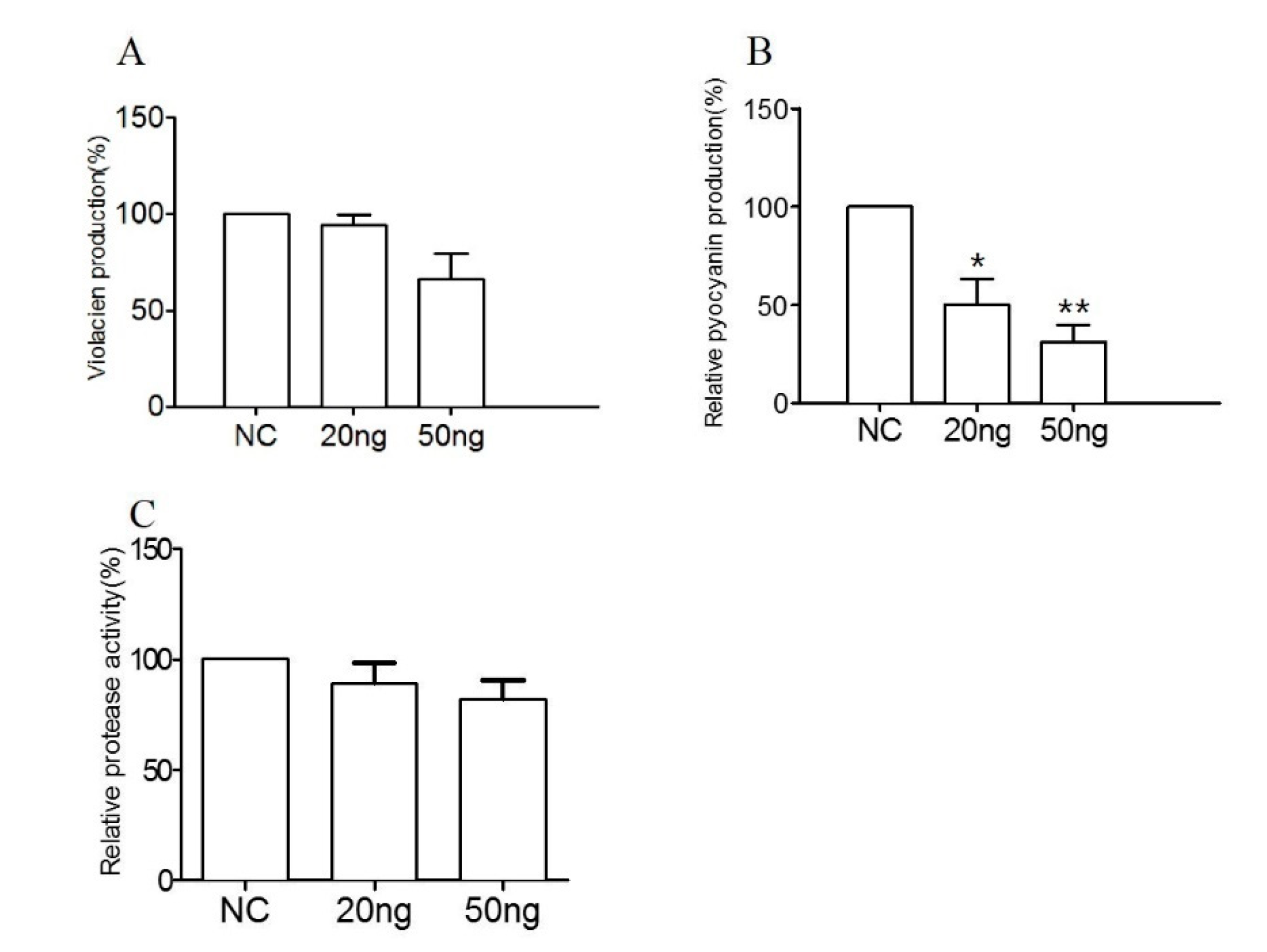
2.5. The Effect of RmmL on In Vitro Production of Virulence Factors
3. Discussion
4. Materials and Methods
4.1. Bacterial Strains, Media, Growth Conditions, and Chemicals
4.2. Cloning, Expression, and Purification of QQ Enzyme of YJ3
4.3. Liquid Chromatography-Mass Spectrometry (LC-MS) Analysis of AHL Degradation Products by RmmL
4.4. Detection of AHL-Degrading Activity of RmmL against Different Signaling Molecules
4.5. Physical and Chemical Parameters That Affect RmmL Activity
4.6. The Effect of RmmL on Violacien and Pyocyanin Production
4.7. The Effect of RmmL on the Extracellular Proteolytic Activity of V. anguillarum VIB72
4.8. Nucleotide Sequence Accession Number
Author Contributions
Funding
Conflicts of Interest
References
- Hastings, J.W. Bioluminescence. Annu. Rev. Biochem. 1968, 37, 597–630. [Google Scholar] [CrossRef]
- Tang, K.; Zhang, X.H. Quorum quenching agents: Resources for antivirulence therapy. Mar. Drugs 2014, 6, 3245–3282. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.H.; Xu, J.L.; Li, X.Z.; Zhang, L.H. AiiA, an enzyme that inactivates the acylhomoserine lactone quorum-sensing signal and attenuates the virulence of Erwinia carotovora. Proc. Natl. Acad. Sci. USA 2000, 7, 3526–3531. [Google Scholar] [CrossRef]
- Park, S.Y.; Lee, S.J.; Oh, T.K.; Oh, J.W.; Koo, B.T.; Yum, D.Y.; Lee, J.K. AhlD, an N-acylhomoserine lactonase in Arthrobacter sp., and predicted homologues in other bacteria. Microbiology 2003, 6, 1541–1550. [Google Scholar] [CrossRef] [PubMed]
- Tang, K.; Su, Y.; Brackman, G.; Cui, F.; Zhang, Y.; Shi, X.; Coenye, T.; Zhang, X.H. MomL, a novel marine-derived N-acyl homoserine lactonase from Muricauda olearia. Appl. Environ. Microbiol. 2015, 2, 774–782. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.Z.; Morohoshi, T.; Ikenoya, M.; Someya, N.; Ikeda, T. AiiM, a novel class of N-acylhomoserine lactonase from the leaf-associated bacterium Microbacterium testaceum. Appl. Environ. Microbiol. 2010, 8, 2524–2530. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.Z.; Morohoshi, T.; Someya, N.; Ikeda, T. AidC, a novel N-acylhomoserine lactonase from the potato root-associated cytophaga-flavobacteria-bacteroides (CFB) group bacterium Chryseobacterium sp. strain StRB126. Appl. Environ. Microbiol. 2012, 22, 7985–7992. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.H.; Xu, J.L.; Hu, J.; Wang, L.H.; Ong, S.L.; Leadbetter, J.R.; Zhang, L.H. Acyl-homoserine lactone acylase from Ralstonia strain XJ12B represents a novel and potent class of quorum-quenching enzymes. Mol. Microbiol. 2003, 3, 849–860. [Google Scholar] [CrossRef]
- Romero, M.; Diggle, S.P.; Heeb, S.; Camara, M.; Otero, A. Quorum quenching activity in Anabaena sp. PCC 7120: Identification of AiiC, a novel AHL-acylase. FEMS Microbiol. Lett. 2008, 1, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Park, S.Y.; Kang, H.O.; Jang, H.S.; Lee, J.K.; Koo, B.T.; Yum, D.Y. Identification of extracellular N-acylhomoserine lactone acylase from a Streptomyces sp. and its application to quorum quenching. Appl. Environ. Microbiol. 2005, 5, 2632–2641. [Google Scholar] [CrossRef] [PubMed]
- Nadal Jimenez, P.; Koch, G.; Papaioannou, E.; Wahjudi, M.; Krzeslak, J.; Coenye, T.; Cool, R.H.; Quax, W.J. Role of PvdQ in Pseudomonas aeruginosa virulence under iron-limiting conditions. Microbiology 2010, 1, 49–59. [Google Scholar] [CrossRef] [PubMed]
- Morohoshi, T.; Nakazawa, S.; Ebata, A.; Kato, N.; Ikeda, T. Identification and characterization of N-acylhomoserine lactone-acylase from the fish intestinal Shewanella sp. strain MIB015. Biosci. Biotechnol. Biochem. 2008, 7, 1887–1893. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Li, C. Exploiting quorum sensing interfering strategies in Gram-negative bacteria for the enhancement of environmental applications. Front. Microbiol. 2016, 6, 1535. [Google Scholar] [CrossRef] [PubMed]
- Sonnenschein, E.C.; Nielsen, K.F.; D’Alvise, P.; Porsby, C.H.; Melchiorsen, J.; Heilmann, J.; Kalatzis, P.G.; Lopez-Perez, M.; Bunk, B.; Sproer, C.; et al. Global occurrence and heterogeneity of the Roseobacter-clade species Ruegeria mobilis. ISME J. 2017, 2, 569–583. [Google Scholar] [CrossRef] [PubMed]
- Bruhn, J.B.; Gram, L.; Belas, R. Production of antibacterial compounds and biofilm formation by Roseobacter species are influenced by culture conditions. Appl. Environ. Microbiol. 2007, 2, 442–450. [Google Scholar] [CrossRef] [PubMed]
- Porsby, C.H.; Nielsen, K.F.; Gram, L. Phaeobacter and Ruegeria species of the Roseobacter clade colonize separate niches in a Danish Turbot (Scophthalmus maximus)-rearing farm and antagonize Vibrio anguillarum under different growth conditions. Appl. Environ. Microbiol. 2008, 23, 7356–7364. [Google Scholar] [CrossRef] [PubMed]
- Carlier, A.; Uroz, S.; Smadja, B.; Fray, R.; Latour, X.; Dessaux, Y.; Faure, D. The Ti plasmid of Agrobacterium tumefaciens harbors an attM-paralogous gene, aiiB, also encoding N-Acyl homoserine lactonase activity. Appl. Environ. Microbiol. 2003, 8, 4989–4993. [Google Scholar] [CrossRef]
- Zhang, H.B.; Wang, L.H.; Zhang, L.H. Genetic control of quorum-sensing signal turnover in Agrobacterium tumefaciens. Proc. Natl. Acad. Sci. USA 2002, 7, 4638–4643. [Google Scholar] [CrossRef] [PubMed]
- Morohoshi, T.; Tominaga, Y.; Someya, N.; Ikeda, T. Complete genome sequence and characterization of the N-acylhomoserine lactone-degrading gene of the potato leaf-associated Solibacillus silvestris. J. Biosci. Bioeng. 2012, 1, 20–25. [Google Scholar] [CrossRef] [PubMed]
- Petersen, T.N.; Brunak, S.; Heijne, G.; von Nielsen, H. SignalP 4.0: Discriminating signal peptides from transmembrane regions. Nat. Methods. 2011, 10, 785–786. [Google Scholar] [CrossRef] [PubMed]
- Garge, S.S.; Nerurkar, A.S. Attenuation of Quorum Sensing Regulated Virulence of Pectobacterium carotovorum subsp. carotovorum through an AHL Lactonase Produced by Lysinibacillus sp. Gs50. PLoS ONE 2016, 12, e0167344. [Google Scholar] [CrossRef] [PubMed]
- Stauff, D.L.; Bassler, B.L. Quorum sensing in Chromobacterium violaceum: DNA recognition and gene regulation by the CviR receptor. J. Bacteriol. 2011, 193, 3871–3878. [Google Scholar] [CrossRef] [PubMed]
- Smith, R.S.; Iglewski, B.H. Pseudomonas aeruginosa quorum sensing as a potential antimicrobial target. J. Clin. Investig. 2003, 112, 1460–1465. [Google Scholar] [CrossRef] [PubMed]
- Milton, D.L. Quorum sensing in vibrios: Complexity for diversification. Int. J. Med. Microbiol. 2006, 296, 61–71. [Google Scholar] [CrossRef] [PubMed]
- Zan, J.; Liu, Y.; Fuqua, C.; Hill, R.T. Acyl-homoserine lactone quorum sensing in the Roseobacter clade. Int. J. Mol. Sci. 2014, 1, 654–669. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Beaber, J.W.; Moré, M.I.; Fuqua, C.; Eberhard, A.; Winans, S.C. Analogs of the autoinducer 3-oxooctanoyl-homoserine lactone strongly inhibit activity of the TraR protein of Agrobacterium tumefaciens. J. Bacteriol. 1998, 20, 5398–5405. [Google Scholar]
- Tempé, J.; Petit, A.; Holsters, M.; Montagu, M.; Schell, J. Thermosensitive step associated with transfer of the Ti plasmid during conjugation: Possible relation to transformation in crown gall. Proc. Natl. Acad. Sci. USA 1977, 7, 2848–2849. [Google Scholar] [CrossRef]
- McClean, K.H.; Winson, M.K.; Fish, L.; Taylor, A.; Chhabra, S.R.; Camara, M.; Daykin, M.; Lamb, J.H.; Swift, S.; Bycoft, B.W.; et al. Quorum sensing and Chromobacterium violaceum: Exploitation of violacein production and inhibition for the detection of N-acylhomoserine lactones. Microbiology 1997, 143, 3703–3711. [Google Scholar] [CrossRef] [PubMed]
- Someya, N.; Morohoshi, T.; Okano, N.; Otsu, E.; Usuki, K.; Sayama, M.; Sekiguchi, H.; Ikeda, T.; Ishida, S. Distribution of N-acylhomoserine lactone-producing fluorescent pseudomonads in the phyllosphere and rhizosphere of potato (Solanum tuberosum L.). Microbes Environ. 2009, 4, 305–314. [Google Scholar] [CrossRef]
- Tang, K.; Zhang, Y.; Yu, M.; Shi, X.; Coenye, T.; Bossier, P.; Zhang, X.H. Evaluation of a new high-throughput method for identifying quorum quenching bacteria. Sci. Rep. 2013, 3, 2935. [Google Scholar] [CrossRef] [PubMed]
- Blosser, R.S.; Gray, K.M. Extraction of violacein from Chromobacterium violaceum provides a new quantitative bioassay for N-acyl homoserine lactone autoinducers. J. Microbiol. Methods 2000, 1, 47–55. [Google Scholar] [CrossRef]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cai, X.; Yu, M.; Shan, H.; Tian, X.; Zheng, Y.; Xue, C.; Zhang, X.-H. Characterization of a Novel N-Acylhomoserine Lactonase RmmL from Ruegeria mobilis YJ3. Mar. Drugs 2018, 16, 370. https://doi.org/10.3390/md16100370
Cai X, Yu M, Shan H, Tian X, Zheng Y, Xue C, Zhang X-H. Characterization of a Novel N-Acylhomoserine Lactonase RmmL from Ruegeria mobilis YJ3. Marine Drugs. 2018; 16(10):370. https://doi.org/10.3390/md16100370
Chicago/Turabian StyleCai, Xiulei, Min Yu, Hu Shan, Xiaorong Tian, Yanfen Zheng, Chunxu Xue, and Xiao-Hua Zhang. 2018. "Characterization of a Novel N-Acylhomoserine Lactonase RmmL from Ruegeria mobilis YJ3" Marine Drugs 16, no. 10: 370. https://doi.org/10.3390/md16100370
APA StyleCai, X., Yu, M., Shan, H., Tian, X., Zheng, Y., Xue, C., & Zhang, X.-H. (2018). Characterization of a Novel N-Acylhomoserine Lactonase RmmL from Ruegeria mobilis YJ3. Marine Drugs, 16(10), 370. https://doi.org/10.3390/md16100370




