Two Novel Aspochalasins from the Gut Fungus Aspergillus sp. Z4
Abstract
1. Introduction
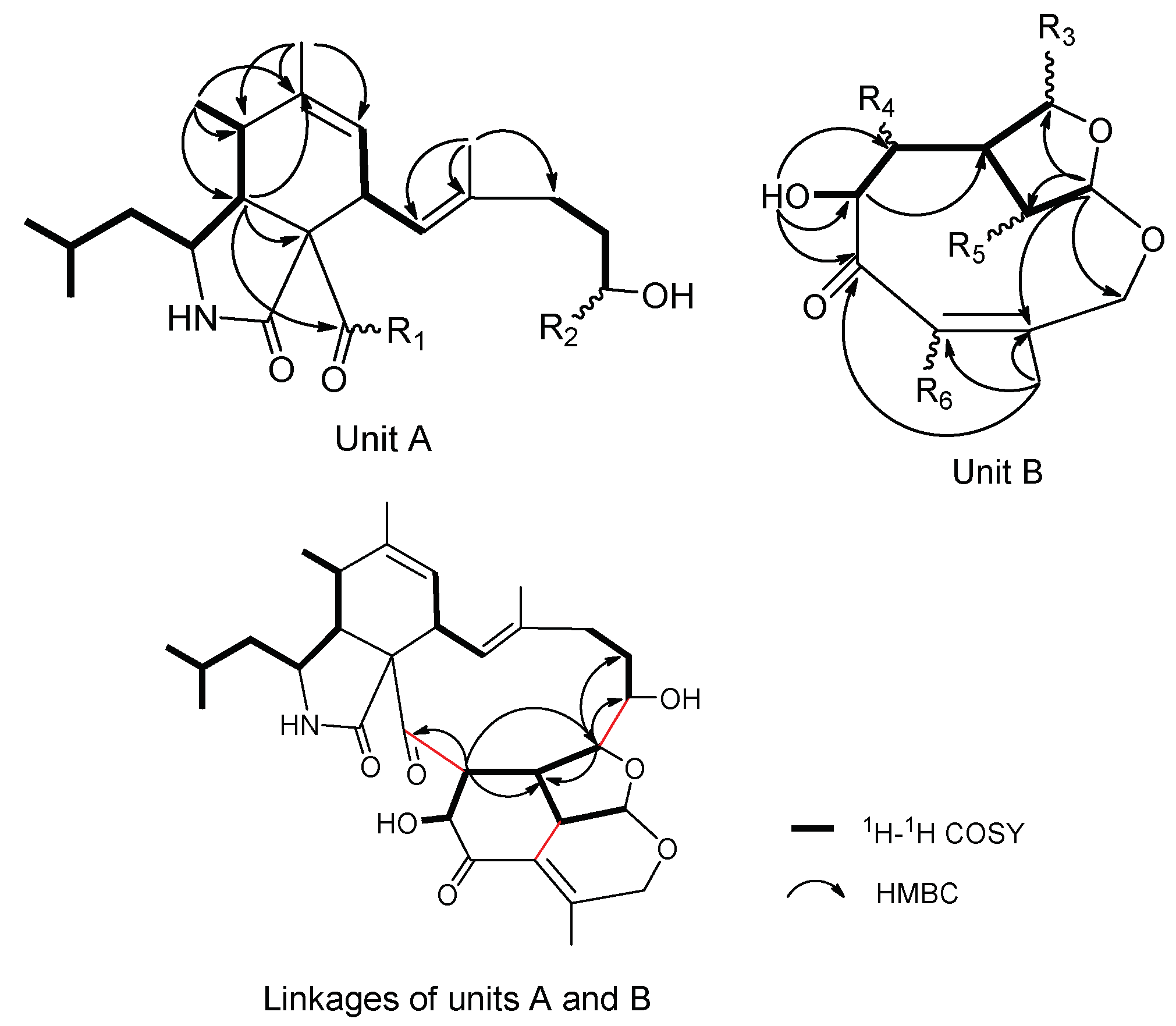
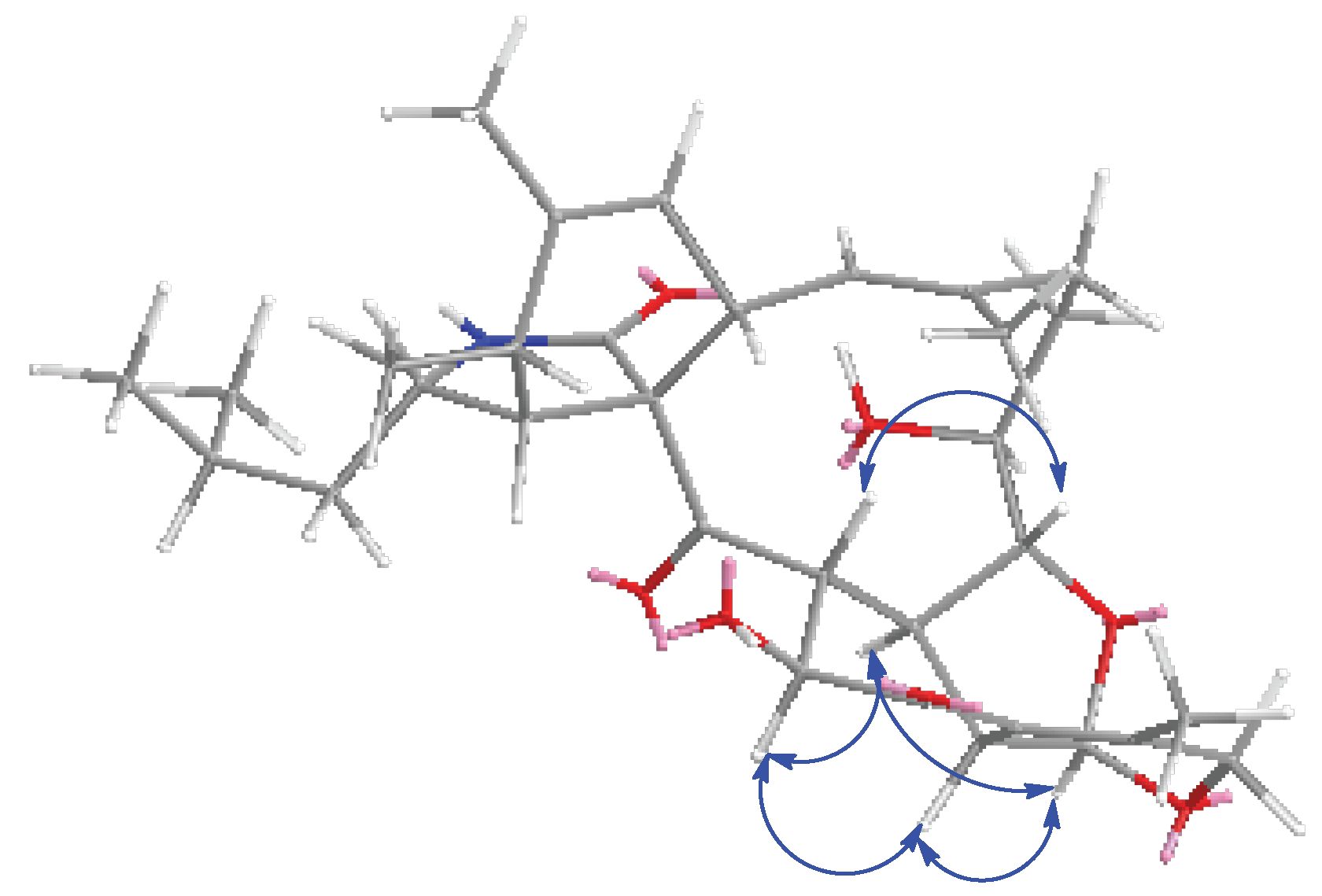
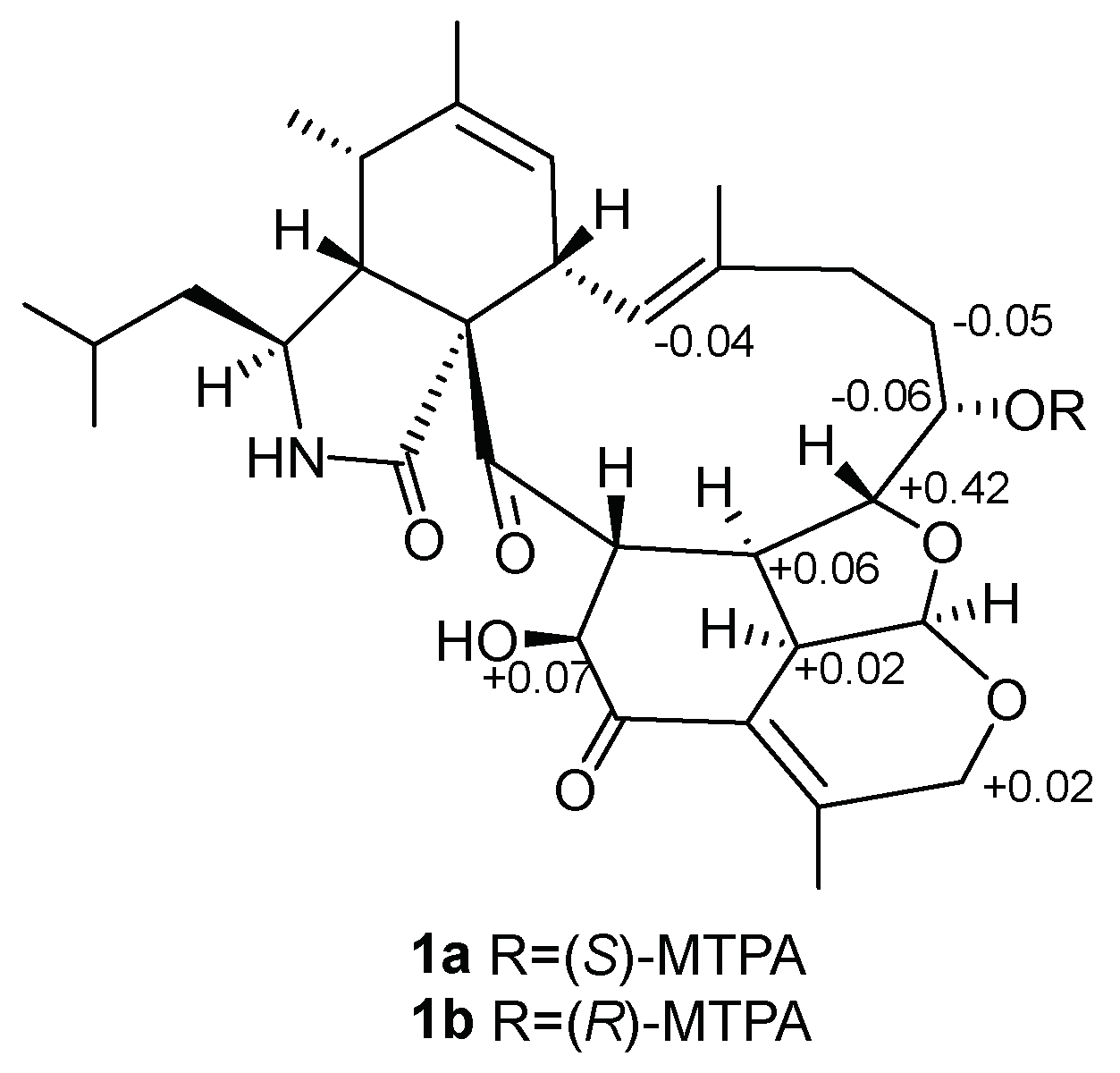
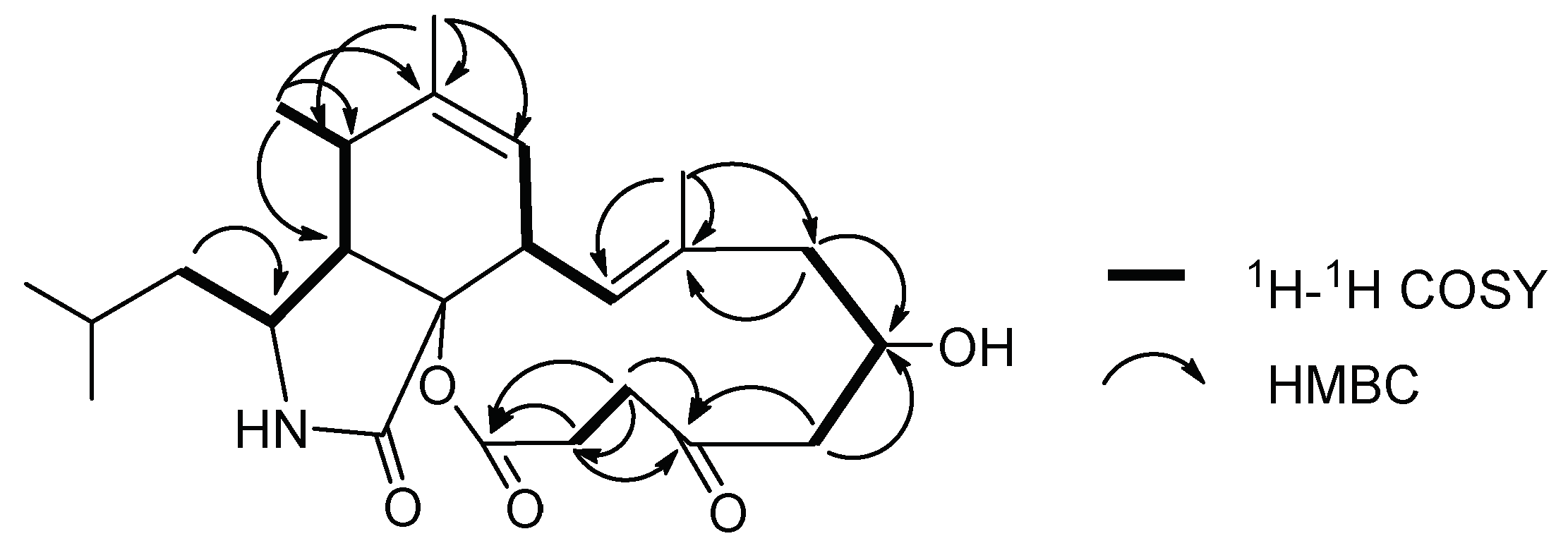
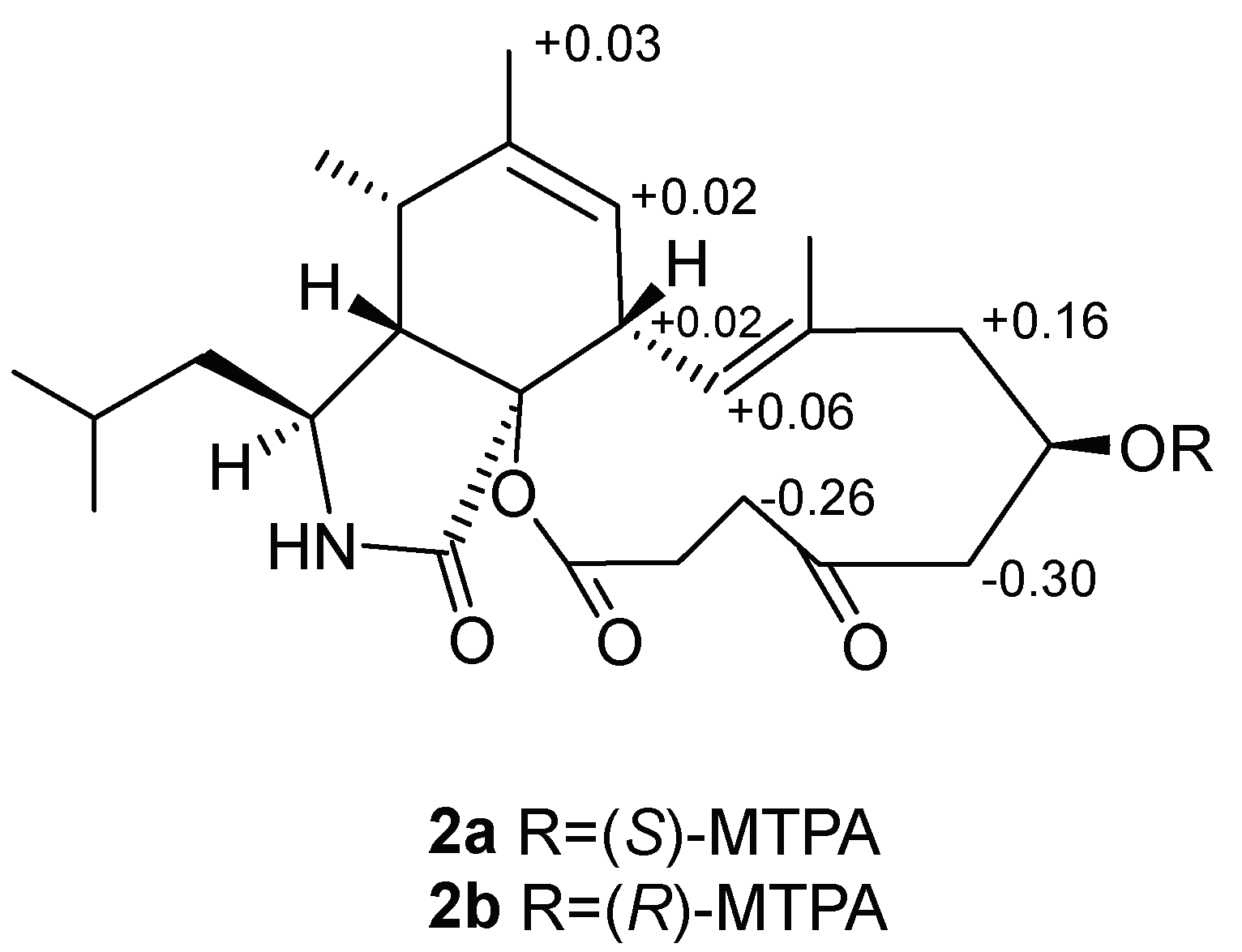
2. Results and Discussion
3. Materials and Methods
3.1. General Experimental Procedures
3.2. Fungal Material
3.3. Fermentation, Extraction, and Isolation of Strain Z4
- Tricochalasin A (1): colorless solid; −2.39 (c 0.67, MeOH); UV (MeOH) λmax (log ε) 204 (3.4), 245 (1.6) nm; IR(film) νmax 3391, 2931, 1753, 1681, 1452, 1380, 1189, 1095 cm−1; 1H and 13C data see Table 1; HR-ESIMS m/z 554.3107 [M + H]+, calcd. 554.3073 for C32H44NO7.
- Aspochalasin A2 (2): colorless solid; 8.08 (c 0.98, CHCl3); UV (MeOH) λmax (log ε) 202 (3.6) nm; IR(KBr) νmax 3454, 3273, 2924, 2852, 1709, 1435, 1384, 1324, 1263, 1142 cm−1; 1H and 13C data see Table 1; HR-ESIMS m/z 418.2589 [M + H]+, calcd. 418.2549 for C24H36NO5.
- Aspochalasins D (3): colorless solid; UV (MeOH) λmax (log ε) 204 (3.1) nm.
- Aspergillulactone (4): colorless solid; UV (MeOH) λmax (log ε) 204 (3.6), 245 (1.3) nm.
- Aspochalasins T (5): colorless solid; UV (MeOH) λmax (log ε) 205 (3.7) nm.
3.4. Preparation of MTPA Esters
- 1a:1H NMR (500 MHz, pyridine-d5): δ 6.68 (1H, H-13), δ 1.37 (1H, H-16a), δ 7.86 (1H, H-17), δ 4.51 (1H, H-18), δ 2.77 (1H, H-19), δ 4.95 (1H, H-27a), δ 4.03 (1H, H-30), δ 4.32 (1H, H-32).
- 1b:1H NMR (500 MHz, pyridine-d5): δ 6.72 (1H, H-13), δ 1.42 (1H, H-16a), δ 7.92 (1H, H-17), δ 4.09 (1H, H-18), δ 2.71 (1H, H-19), δ 4.93 (1H, H-27a), δ 4.01 (1H, H-30), δ 4.25 (1H, H-32).
- 2a:1H NMR (500 MHz, pyridine-d5): δ 5.42 (1H, H-7), δ 3.26 (1H, H-8), δ 1.79 (3H, H-12), δ 6.61 (1H, H-13), δ 2.35 (1H, H-15a), δ 2.25 (1H, H-17a), δ 2.14 (1H, H-19a).
- 2b:1H NMR (500 MHz, pyridine-d5): δ 5.40 (1H, H-7), δ 3.24 (1H, H-8), δ 1.76 (3H, H-12), δ 6.55 (1H, H-13), δ 2.19 (1H, H-15a), δ 2.55 (1H, H-17a), δ 2.40 (1H, H-19a).
3.5. Cytotoxicity Bioassays
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Scherlach, K.; Boettger, D.; Remme, N.; Hertweck, C. The chemistry and biology of cytochalasans. Nat. Prod. Rep. 2010, 27, 869–886. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Ge, H.; Xie, D.; Chen, R.; Zou, J.H.; Tao, X.; Dai, J. Periconiasins A–C, new cytotoxic cytochalasans with an unprecedented 9/6/5 tricyclic ring system from endophytic fungus periconia sp. Org. Lett. 2013, 15, 1674–1677. [Google Scholar] [CrossRef] [PubMed]
- Knudsen, P.B.; Hanna, B.; Ohl, S.; Sellner, L.; Zenz, T.; Dohner, H.; Stilgenbauer, S.; Larsen, T.O.; Lichter, P.; Seiffert, M. Chaetoglobosin A preferentially induces apoptosis in chronic lymphocytic leukemia cells by targeting the cytoskeleton. Leukemia 2014, 28, 1289–1298. [Google Scholar] [CrossRef] [PubMed]
- Zhou, G.X.; Wijeratne, E.M.K.; Bigelow, D.; Pierson, L.S.; Vanetten, H.D.; Guantilaka, A.A.L. Aspochalasins I, J, and K: Three new cytotoxic cytochalasans of Aspergillus flavipes from the rhizosphere of Ericameria laricifolia of the sonoran desert. J. Nat. Prod. 2004, 67, 328–332. [Google Scholar] [CrossRef] [PubMed]
- Rochfort, S.; Ford, J.; Ovenden, S.; Wan, S.S.; George, S.; Wildman, H.; Tait, R.M.; Meurer-Grimes, B.; Cox, S.; Coates, J.; et al. A novel aspochalasin with HIV-1 integrase inhibitory activity from Aspergillus flavipes. J. Antibiot. 2005, 58, 279–283. [Google Scholar] [CrossRef] [PubMed]
- Hua, C.Y.; Yang, Y.H.; Sun, L.; Dou, H.; Tan, R.X.; Hou, Y.Y. Chaetoglobosin F, a small molecule compound, possesses immunomodulatory properties on bone marrow-derived dendritic cells via TLR9 signaling pathway. Immunobiology 2013, 218, 292–302. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Zhang, W.P.; Zhang, P.; Ruan, W.B.; Zhu, X.D. Nematicidal Activity of Chaetoglobosin A Poduced by Chaetomium globosum NK102 against Meloidogyne incognita. J. Agric. Food Chem. 2013, 61, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Qiao, K.J.; Chooi, Y.H.; Tang, Y. Identification and engineering of the cytochalasin gene cluster from Aspergillus clavatus NRRL 1. Metab. Eng. 2011, 13, 723–732. [Google Scholar] [CrossRef] [PubMed]
- Schumann, J.; Hertweck, C. Molecular basis of cytochalasan biosynthesis in fungi: Gene cluster analysis and evidence for the involvement of a PKS-NRPS hybrid synthase by RNA silencing. J. Am. Chem. Soc. 2007, 129, 9564–9565. [Google Scholar] [CrossRef] [PubMed]
- Oh, H.; Swenson, D.C.; Gloer, J.B.; Wicklow, D.T.; Dowd, P.F. Chaetochalasin A: A new bioactive metabolite from Chaetomium brasiliense. Tetrahedron. Lett. 1998, 39, 7633–7636. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, T.; Pei, Y.H.; Hua, H.M.; Feng, B.M. Aspergillin PZ, a novel isoindole-alkaloid from Aspergillus awamori. J. Antibiot. 2002, 55, 693–695. [Google Scholar] [CrossRef] [PubMed]
- Lin, Z.J.; Zhu, T.J.; Wei, H.J.; Zhang, G.J.; Wang, H.; Gu, Q.Q. Spicochalasin A and New Aspochalasins from the Marine-Derived Fungus Spicaria elegans. Eur. J. Org. Chem. 2009, 18, 3045–3051. [Google Scholar] [CrossRef]
- Zhu, H.C.; Chen, C.M.; Tong, Q.Y.; Li, X.N.; Yang, J.; Xue, Y.B.; Luo, Z.W.; Wang, J.P.; Yao, G.M.; Zhang, Y.H. Epicochalasines A and B: Two bioactive merocytochalasans bearing caged epicoccine dimer units from Aspergillus flavipes. Angew. Chem. Int. Ed. 2016, 55, 3486–3490. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zhao, Z.; Ding, W.; Ye, B.; Wang, P.; Xu, J. Aspochalazine A, a novel polycyclic aspochalasin from the fungus Aspergillus sp. Z4. Tetrahedron Lett. 2017, 58, 2405–2408. [Google Scholar] [CrossRef]
- Liu, Y.; Zhao, S.; Ding, W.; Wang, P.; Yang, X.; Xu, J. Methylthio-aspochalasins from a marine-derived fungus Aspergillus sp. Mar. Drugs 2014, 12, 5124–5131. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.Z.; Zhao, S.Z.; Yang, X.W. A new cyclopeptide metabolite of marine gut fungus from Ligia oceanica. Nat. Prod. Res. 2014, 28, 994–997. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.M.; Zhao, S.Z.; Liu, Y.; Ding, W.J.; Qiu, F.; Xu, J.Z. Asperginine, an unprecedented alkaloid from the marine-derived fungus Aspergillus sp. Nat. Prod. Commun. 2015, 10, 1363–1364. [Google Scholar] [PubMed]
- Wang, T.; Zhang, Y.; Pei, Y.h. Two novel trichothecenes from Myrothecium roridum. Med. Chem. Res. 2007, 16, 155–161. [Google Scholar] [CrossRef]
- Ding, G.; Wang, H.; Li, L.; Chen, A.J.; Chen, L.; Chen, H.; Zhang, H.; Liu, X.; Zou, Z. Trichoderones A and B: Two pentacyclic cytochalasans from the plant endophytic fungus Trichoderma gamsii. Eur. J. Org. Chem. 2012, 2012, 2516–2519. [Google Scholar] [CrossRef]
- Liu, R.; Gu, Q.; Zhu, W.; Cui, C.; Fan, G.; Fang, Y.; Zhu, T.; Liu, H. 10-Phenyl-[12]-cytochalasins Z7, Z8, and Z9 from the marine-derived fungus Spicaria elegans. J. Nat. Prod. 2006, 69, 871–875. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Tong, Q.; Zhu, H.; Tan, D.; Zhang, J.; Xue, Y.; Yao, G.; Luo, Z.; Wang, J.; Wang, Y.; et al. Nine new cytochalasan alkaloids from Chaetomium globosum TW1-1 (Ascomycota, Sordariales). Sci. Rep. 2016, 6, 18711. [Google Scholar] [CrossRef] [PubMed]
- Zheng, C.J.; Shao, C.L.; Wu, L.Y.; Chen, M.; Wang, K.L.; Zhao, D.L.; Sun, X.P.; Chen, G.Y.; Wang, C.Y. Bioactive phenylalanine derivatives and cytochalasins from the soft coral-derived fungus, Aspergillus elegans. Mar. Drugs 2013, 11, 2054–2068. [Google Scholar] [CrossRef] [PubMed]
- Tomikawa, T.; Shin-ya, K.; Kinoshita, T.; Miyajima, A.; Seto, H.; Hayakawa, Y. Selective cytotoxicity and stereochemistry of aspochalasin D. J. Antibiot. 2001, 54, 379–381. [Google Scholar] [CrossRef] [PubMed]
- Rukachaisirikul, V.; Rungsaiwattana, N.; Klaiklay, S.; Phongpaichit, S.; Borwornwiriyapan, K.; Sakayaroj, J. γ-Butyrolactone, cytochalasin, cyclic carbonate, eutypinic acid, and phenalenone derivatives from the soil fungus Aspergillus sp. PSU-RSPG185. J. Nat. Prod. 2014, 77, 2375–2382. [Google Scholar] [CrossRef] [PubMed]
- Lin, Z.J.; Zhu, T.J.; Chen, L.; Gu, Q.Q. Three new aspochalasin derivatives from the marine-derived fungus Spicaria elegans. Chin. Chem. Lett. 2010, 21, 824–826. [Google Scholar] [CrossRef]
- Wang, F.; Hua, H.M.; Pei, Y.H.; Chen, D.; Jing, Y.K. Triterpenoids from the resin of Styrax tonkinensis and their antiproliferative and differentiation effects in human leukemia HL-60 cells. J. Nat. Prod. 2006, 69, 807–810. [Google Scholar] [CrossRef] [PubMed]

| Pos. | 1 | 2 | ||
|---|---|---|---|---|
| δCa, Type | δH (J in Hz) b | δCa, Type | δH (J in Hz) b | |
| 1 | 176.4, C | 172.4, C | ||
| 2 | 6.10, brs | 5.96, brs | ||
| 3 | 52.9, CH | 2.97, m | 52.4, CH | 2.99, m |
| 4 | 53.9, CH | 2.81, t (4.4) | 52.3, CH | 2.56, dd (8.5,7.1) |
| 5 | 35.9, CH | 2.67, brs | 34.8, CH | 2.80, brs |
| 6 | 140.6, C | 140.9, C | ||
| 7 | 125.6, CH | 5.45, brs | 123.5, CH | 5.29, brs |
| 8 | 42.9, CH | 3.61, d (12.1) | 41.1, CH | 3.36, d (10.3) |
| 9 | 68.9, C | 86.5, C | ||
| 10 | 47.2, CH2 | 1.49, m; 1.25, m | 46.8, CH2 | 1.83, m; 1.23, m |
| 11 | 14.4, CH3 | 1.18, d (7.1) | 14.3, CH3 | 1.19, d (7.3) |
| 12 | 20.5, CH3 | 1.77, s | 20.1, CH3 | 1.77, s |
| 13 | 125.1, CH | 5.70, d (11.4) | 123.8, CH | 5.94, d (10.3) |
| 14 | 135.9, C | 138.2, C | ||
| 15 | 36.9, CH2 | 2.07, m | 33.1, CH2 | 2.27, m; 2.14, m |
| 16 | 29.3, CH | 1.50, m; 1.30, m | 75.1, CH | 4.42, brs |
| 17 | 68.6, CH | 4.35, d (6.3) | 32.3, CH2 | 2.04, m; 2.28, m |
| 18 | 88.8, CH | 3.86, d (11.3) | 208.9, C | |
| 19 | 44.0, CH | 2.25, m | 29.0, CH2 | 2.32, m; 3.02, m |
| 20 | 47.7, CH | 3.51, dd (4.9, 11.0) | 34.1, CH2 | 2.62, m; 2.44, m |
| 21 | 214.2, C | 171.9, C | ||
| 22 | 26.0, CH | 1.53, m | 25.6, CH | 1.59, m |
| 23 | 21.2, CH3 | 0.93, d (6.4) | 21.1, CH3 | 0.92, d (6.5) |
| 24 | 24.2, CH3 | 0.96, d (6.4) | 23.8, CH3 | 0.94, d (6.5) |
| 25 | 16.1, CH3 | 1.57, s | 17.7, CH3 | 1.56, s |
| 26 | 109.4, CH | 5.91, d (3.9) | ||
| 27 | 73.1, CH2 | 4.79, m | ||
| 28 | 158.1, C | |||
| 29 | 127.6, C | |||
| 30 | 51.4, CH | 3.59, m | ||
| 31 | 197.0, C | |||
| 32 | 77.3, CH | 4.42, dd (3.6, 11.1) | ||
| 33 | 14.0, CH3 | 1.85, s | ||
| Compounds | PC3 Cell Line (IC50 in μM) |
|---|---|
| 1 | >36 |
| 2 | >40 |
| 3 | 11.14 |
| 4 | >40 |
| 5 | >40 |
| ADR | 5.09 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, X.; Ding, W.; Wang, P.; Xu, J. Two Novel Aspochalasins from the Gut Fungus Aspergillus sp. Z4. Mar. Drugs 2018, 16, 343. https://doi.org/10.3390/md16100343
Li X, Ding W, Wang P, Xu J. Two Novel Aspochalasins from the Gut Fungus Aspergillus sp. Z4. Marine Drugs. 2018; 16(10):343. https://doi.org/10.3390/md16100343
Chicago/Turabian StyleLi, Xinyang, Wanjing Ding, Pinmei Wang, and Jinzhong Xu. 2018. "Two Novel Aspochalasins from the Gut Fungus Aspergillus sp. Z4" Marine Drugs 16, no. 10: 343. https://doi.org/10.3390/md16100343
APA StyleLi, X., Ding, W., Wang, P., & Xu, J. (2018). Two Novel Aspochalasins from the Gut Fungus Aspergillus sp. Z4. Marine Drugs, 16(10), 343. https://doi.org/10.3390/md16100343






