Cytotoxic and Antibacterial Compounds from the Coral-Derived Fungus Aspergillus tritici SP2-8-1
Abstract
:1. Introduction
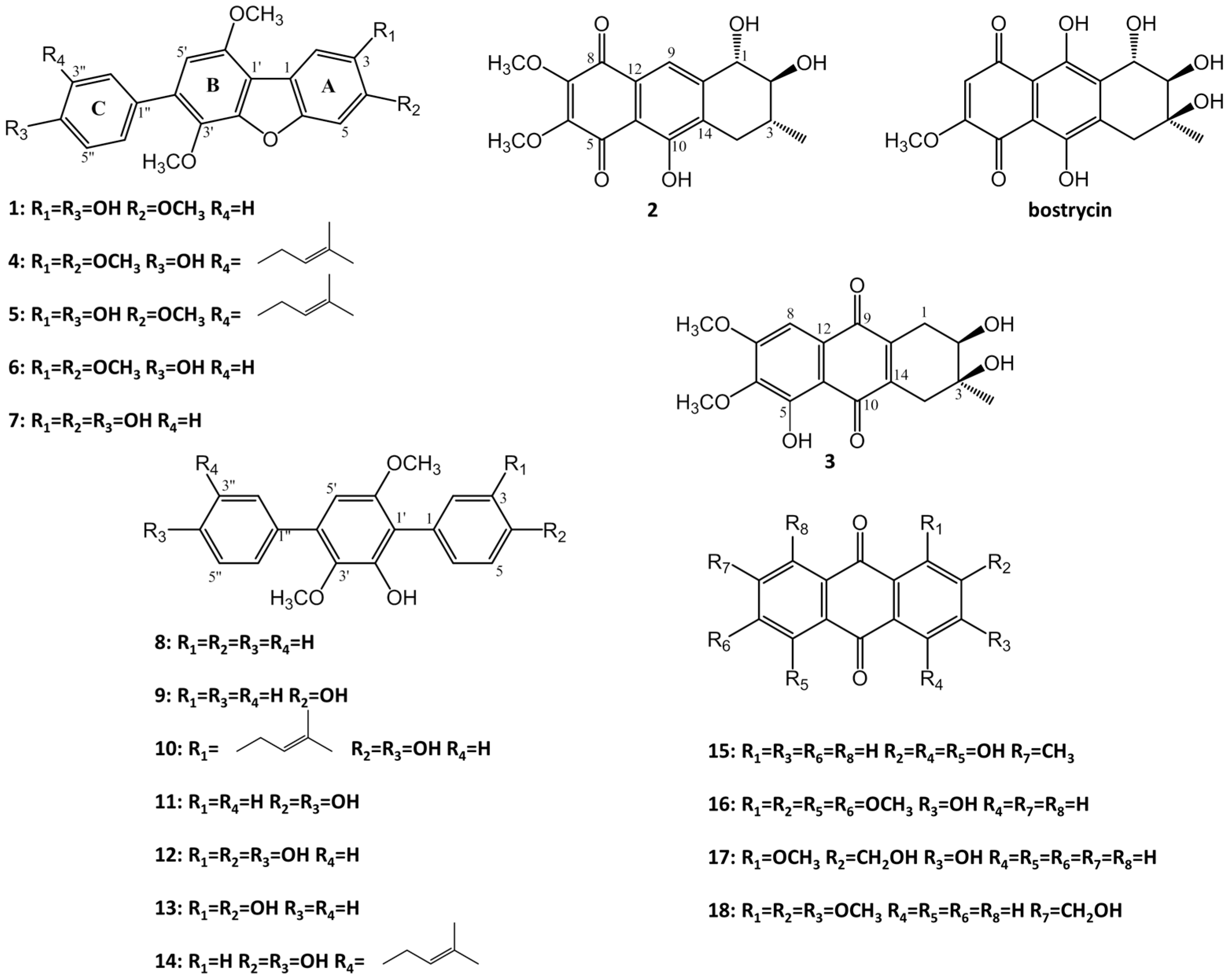
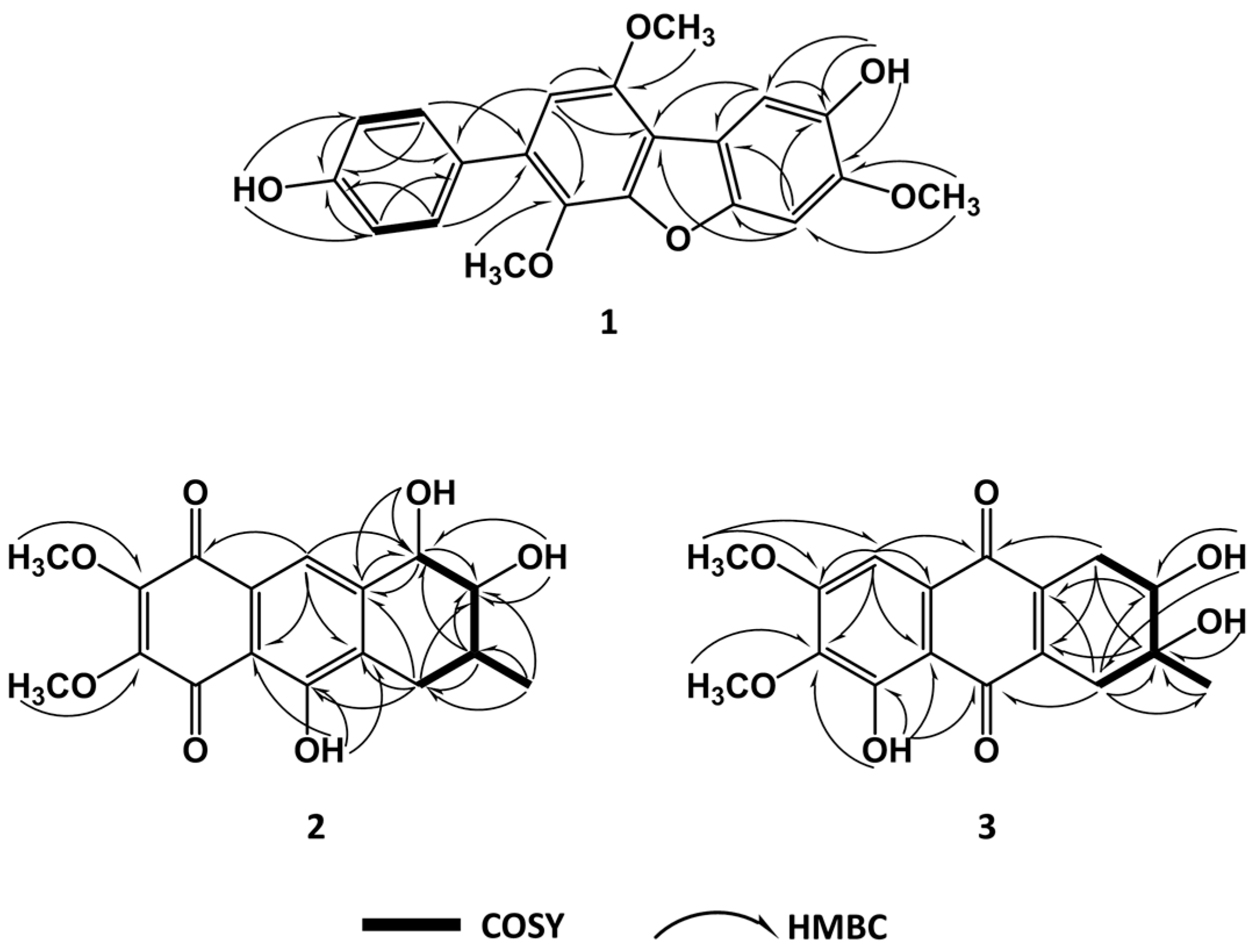
2. Results and Discussion
3. Materials and Methods
3.1. General Experimental Procedures
3.2. Fungal Material
3.3. Fermentation, Extraction, and Isolation
3.4. Antibacterial Assay
3.5. Cytotoxicity Assay
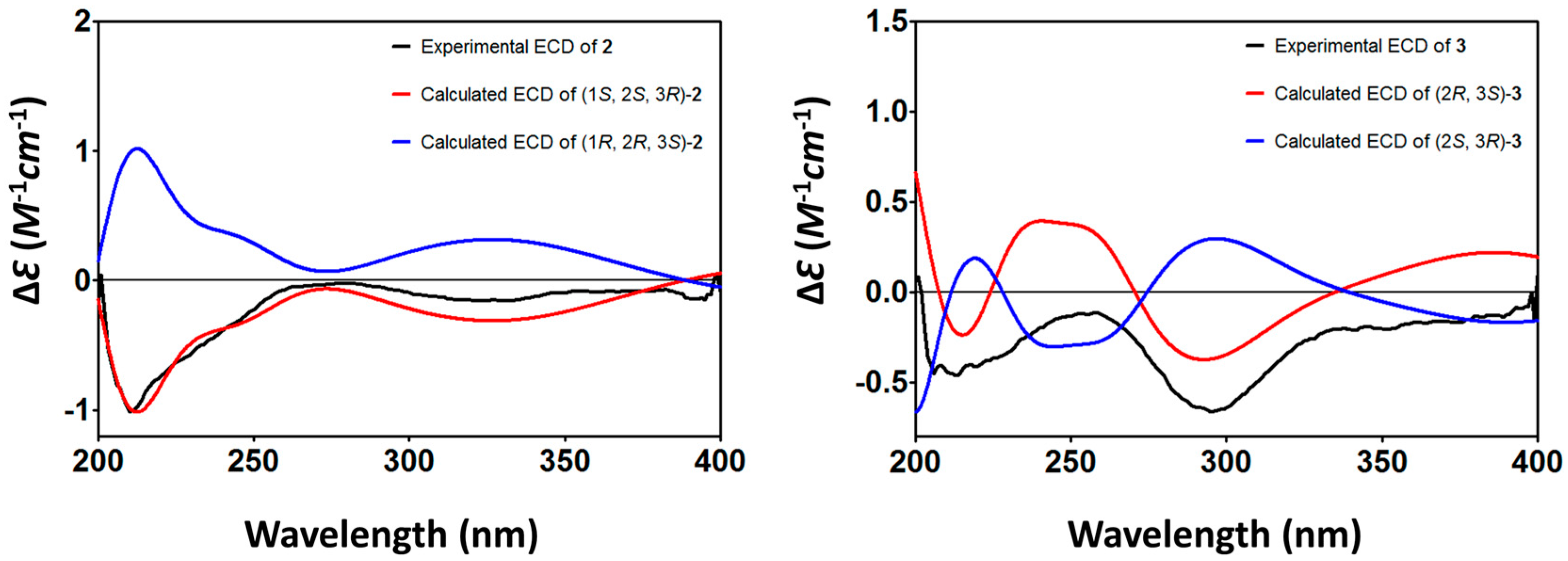
3.6. ECD Calculation
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Fouillaud, M.; Venkatachalam, M.; Girard-Valenciennes, E.; Caro, Y.; Dufosse, L. Anthraquinones and derivatives from marine-derived fungi: Structural diversity and selected biological activities. Mar. Drugs 2016, 14, 64. [Google Scholar] [CrossRef] [PubMed]
- Schueffler, A.; Anke, T. Fungal natural products in research and development. Nat. Prod. Rep. 2014, 31, 1425–1448. [Google Scholar] [CrossRef] [PubMed]
- Cai, S.; Sun, S.; Zhou, H.; Kong, X.; Zhu, T.; Li, D.; Gu, Q. Prenylated polyhydroxy-p-terphenyls from Aspergillus taichungensis ZHN-7-07. J. Nat. Prod. 2011, 74, 1106–1110. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.S.; Zhao, B.B.; Lu, C.H.; Huang, J.J.; Shen, Y.M. Two new p-terphenyl derivatives from the marine fungal strain Aspergillus sp. AF119. Nat. Prod. Commun. 2012, 7, 1057–1062. Available online: https://www.ncbi.nlm.nih.gov/pubmed/22978228 (accessed on 1 August 2012). [PubMed]
- Xu, J.; Nakazawa, T.; Ukai, K.; Kobayashi, H.; Mangindaan, R.E.P.; Wewengkang, D.S.; Rotinsulu, H.; Namikoshi, M. Tetrahydrobostrycin and 1-Deoxytetrahydrobostrycin, Two New Hexahydroanthrone Derivatives, from a Marine-derived Fungus Aspergillus sp. J. Antibiot. 2008, 61, 415–419. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Zhong, L.; Long, Y.; Li, J.; Wu, J.; Liu, L.; Chen, S.; Lin, Y.; Li, M.; Zhu, X.; et al. Studies on the synthesis of derivatives of marine-derived bostrycin and their structure-activity relationship against tumor cells. Mar. Drugs 2012, 10, 932–952. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Ding, X.; Feng, S.X.; Guan, Q.; Zhang, X.P.; Du, C.; Di, Y.T.; Chen, T. Seven new tetrahydroanthraquinones from the root of Prismatomeris connata and their cytotoxicity against lung tumor cell growth. Molecules 2015, 20, 22565–22577. [Google Scholar] [CrossRef] [PubMed]
- Ondeyka, J.; Buevich, A.V.; Williamson, R.T.; Zink, D.L.; Polishook, J.D.; Occi, J.; Vicente, F.; Basilio, A.; Bills, G.F.; Donald, R.G.; et al. Isolation, structure elucidation, and biological activity of altersolanol P using Staphylococcus aureus fitness test based genome-wide screening. J. Nat. Prod. 2014, 77, 497–502. [Google Scholar] [CrossRef] [PubMed]
- Debbab, A.; Aly, A.H.; Edrada-Ebel, R.; Wray, V.; Muller, W.E.; Totzke, F.; Zirrgiebel, U.; Schachtele, C.; Kubbutat, M.H.; Lin, W.H.; et al. Bioactive metabolites from the endophytic fungus Stemphylium globuliferum isolated from Mentha pulegium. J. Nat. Prod. 2009, 72, 626–631. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.K.; Yan, T.; Guo, Y.; Song, Y.C.; Jiao, R.H.; Tan, R.X.; Ge, H.M. P-Terphenyl and diterpenoid metabolites from endophytic Aspergillus sp. YXf3. J. Nat. Prod. 2012, 75, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Feng, X.; Xiao, Z.; Liu, L.; Li, H.; Ma, L.; Lu, Y.; Ju, J.; She, Z.; Lin, Y. Azaphilones and p-terphenyls from the mangrove endophytic fungus Penicillium chermesinum (ZH4-E2) isolated from the South China Sea. J. Nat. Prod. 2011, 74, 997–1002. [Google Scholar] [CrossRef] [PubMed]
- Marchelli, R.; Vining, L.C. Terphenyllin, A novel p-terphenyl metabolite from Aspergillus candidus. J. Antibiot. (Tokyo) 1975, 28, 328–331. [Google Scholar] [CrossRef] [PubMed]
- Kurobane, I.; Vining, L.C.; McInnes, A.G.; Smith, D.G. 3-Hydroxyterphenyllin, a new metabolite of Aspergillus candidus Structure elucidation by H and C nuclear magnetic resonance spectroscopy. J. Antibiot. (Tokyo) 1979, 32, 559–564. [Google Scholar] [CrossRef] [PubMed]
- Kamigauchi, T.; Sakazaki, R.; Nagashima, K.; Kawamura, Y.; Yasuda, Y.; Matsushima, K.; Tani, H.; Takahashi, Y.; Ishii, K.; Suzuki, R.; et al. Terprenins, Novel immunosuppressants produced by Aspergillus candidus. J. Antibiot. (Tokyo) 1998, 51, 445–450. [Google Scholar] [CrossRef]
- Zhang, W.; Wei, W.; Shi, J.; Chen, C.; Zhao, G.; Jiao, R.; Tan, R. Natural phenolic metabolites from endophytic Aspergillus sp. IFB-YXS with antimicrobial activity. BioOrg. Med. Chem. Lett. 2015, 25, 2698–2701. [Google Scholar] [CrossRef] [PubMed]
- Feng, S.-X.; Hao, J.; Chen, T.; Qiu, S.X. A New Anthraquinone and two new tetrahydroanthraquinones from the Roots of Prismatomeris connata. Helv. Chim. Acta 2011, 94, 1843–1849. [Google Scholar] [CrossRef]
- Lai, X.; Cao, L.; Tan, H.; Fang, S.; Huang, Y.; Zhou, S. Fungal communities from methane hydrate-bearing deep-sea marine sediments in South China Sea. ISME J. 2007, 1, 756–762. [Google Scholar] [CrossRef] [PubMed]
- Chhillar, A.K.; Gahlaut, A. Evaluation of antibacterial potential of plant extracts using resazurin based microtiter dilution assay. Int. J. Pharm. Pharm. Sci. 2013, 5, 372–376. [Google Scholar]
- Wibowo, A.; Ahmat, N.; Hamzah, A.S.; Low, A.L.; Mohamad, S.A.; Khong, H.Y.; Sufian, A.S.; Manshoor, N.; Takayama, H.; Malaysianol, B. An oligostilbenoid derivative from Dryobalanops lanceolata. Fitoterapia 2012, 83, 1569–1575. [Google Scholar] [CrossRef] [PubMed]
- Coban, A.Y. Rapid determination of methicillin resistance among Staphylococcus aureus clinical isolates by colorimetric methods. J. Clin. Microbiol. 2012, 50, 2191–2193. [Google Scholar] [CrossRef] [PubMed]
- Han, S.B.; Shin, Y.J.; Hyon, J.Y.; Wee, W.R. Cytotoxicity of voriconazole on cultured human corneal endothelial cells. Antimicrob. Agents. Chemother. 2011, 55, 4519–4523. [Google Scholar] [CrossRef] [PubMed]
- Ishiyama, M.; Tominaga, H.; Shiga, M.; Sasamoto, K.; Ohkura, Y.; Ueno, K. A combined assay of cell viability and in vitro cytotoxicity with a highly water-soluble tetrazolium salt, neutral red and crystal violet. Biol. Pharm. Bull. 1996, 19, 1518–1520. [Google Scholar] [CrossRef] [PubMed]
- O’Boyle, N.M.; Vandermeersch, T.; Flynn, C.J.; Maguire, A.R.; Hutchison, G.R. Confab-Systematic generation of diverse low-energy conformers. J. Cheminform. 2011, 3, 8. [Google Scholar] [CrossRef] [PubMed]
- Bruhn, T.; Schaumloffel, A.; Hemberger, Y.; Bringmann, G. SpecDis: Quantifying the comparison of calculated and experimental electronic circular dichroism spectra. Chirality 2013, 25, 243–249. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Mukwaya, E.; Wong, M.S.; Zhang, Y. A systematic review on biological activities of prenylated flavonoids. Pharm. Biol. 2014, 52, 655–660. [Google Scholar] [CrossRef] [PubMed]

| Position | 1 | 2 | 3 | |||
|---|---|---|---|---|---|---|
| δH, mult. (J in Hz) | δC | δH, mult. (J in Hz) | δC | δH, mult. (J in Hz) | δC | |
| 1 | 114.9 | 4.26, d (7.03) | 73.8 | 2.54, dd (19.84, 4.88), Heq 2.69, dd (19.84, 11.63), Hax | 30.1 | |
| 2 | 7.39, s | 107.3 | 3.13, m | 76.3 | 3.59, m | 69.7 |
| 3 | 144.0 | 1.76, m | 33.5 | 69.1 | ||
| 4 | 148.4 | 2.19, dd (18.45, 11.46), Hax 2.92, dd (18.45, 5.40), Heq | 31.3 | 2.45, br d (13.30) | 33.0 | |
| 5 | 7.38, s | 96.6 | 187.2 | 155.1 | ||
| 6 | 149.7 | 148.8 | 140.8 | |||
| 7 | 146.9 | 157.9 | ||||
| 8 | 181.1 | 7.16, s | 104.0 | |||
| 9 | 7.65, s | 118.1 | 183.5 | |||
| 10 | 158.0 | 189.2 | ||||
| 11 | 111.0 | 110.8 | ||||
| 12 | 128.5 | 127.9 | ||||
| 13 | 148.6 | 142.8 | ||||
| 14 | 131.7 | 142.6 | ||||
| 1′ | 114.2 | |||||
| 2′ | 149.0 | |||||
| 3′ | 136.4 | |||||
| 4′ | 131.4 | |||||
| 5′ | 6.72, s | 106.0 | ||||
| 6′ | 150.0 | |||||
| 1″ | 129.0 | |||||
| 2″, 6″ | 7.42, d (8.53) | 130.8 | ||||
| 3″, 5″ | 6.85, d (8.53) | 115.5 | ||||
| 4″ | 157.2 | |||||
| CH3-3 | 1.08, d (6.53) | 18.5 | 1.21, s | 26.0 | ||
| OCH3-4 | 3.87, s | 56.4 | ||||
| OCH3-6 | 3.99, s | 61.4 | 3.79, s | 60.8 | ||
| OCH3-7 | 3.95, s | 61.7 | 3.92, s | 56.8 | ||
| OCH3-3′ | 3.77, s | 61.0 | ||||
| OCH3-6′ | 3.97, s | 56.3 | ||||
| OH-1 | 5.82, d (6.27) | |||||
| OH-2 | 5.06, d (5.02) | 4.73, br s | ||||
| OH-3 | 9.06, brs | 5.03, br s | ||||
| OH-5 | 12.09, s | |||||
| OH-10 | 12.18, s | |||||
| OH-4″ | 9.55, brs | |||||
| Compound | MIC (μg/mL) | IC50 (μM) | ||||||
|---|---|---|---|---|---|---|---|---|
| MRSA 1 | MRSA 2 | VV | VR | VC | HeLa | A549 | Hep G2 | |
| 1 | 31.33 ± 0.61 | 30.97 ± 0.78 | 31.47 ± 1.22 | NA | 15.10 ± 0.44 | 30.23 ± 1.32 | 24.53 ± 1.10 | 27.50 ± 1.57 |
| 2 | 7.53 ± 0.31 | 7.63 ± 0.21 | 15.61 ± 0.48 | 31.17 ± 0.35 | 15.53 ± 0.60 | 2.67 ± 0.60 | 3.13 ± 0.68 | 3.87 ± 0.74 |
| 3 | 15.27 ± 0.35 | 15.63 ± 0.45 | 15.47 ± 0.51 | 31.33 ± 0.23 | 15.77 ± 0.29 | 10.57 ± 0.93 | 4.67 ± 0.60 | 8.57 ± 0.83 |
| 4 | 15.67 ± 0.50 | 7.57 ± 0.73 | 15.58 ± 0.33 | 15.57 ± 0.30 | NA | 16.77 ± 0.45 | 21.07 ± 0.76 | 27.17 ± 0.29 |
| 5 | 3.80 ± 0.13 | 3.80 ± 0.22 | 7.77 ± 0.10 | 7.75 ± 0.18 | 15.57 ± 0.30 | 10.20 ± 0.50 | 13.07 ± 0.72 | 35.10 ± 1.00 |
| 6 | NA | NA | NA | NA | NA | NA | NA | NA |
| 7 | 31.47 ± 0.24 | 31.23 ± 0.10 | 31.42 ± 0.23 | 31.33 ± 0.19 | NA | 25.07 ±0.81 | 19.07 ± 0.64 | 32.10 ± 2.00 |
| 8 | NA | NA | NA | NA | NA | NA | NA | NA |
| 9 | 31.30 ± 0.26 | 31.45 ± 0.22 | 31.37 ± 0.14 | 31.53 ± 0.31 | 31.47 ± 0.25 | NA | NA | NA |
| 10 | 15.53 ± 0.31 | 15.47 ± 0.23 | 31.43 ± 0.32 | 31.37 ± 0.21 | NA | 3.23 ± 0.40 | 3.87 ± 0.15 | 2.10 ± 0.20 |
| 11 | 31.47 ± 0.24 | 31.27 ± 0.16 | 31.37 ± 0.25 | NA | NA | 18.87 ± 1.27 | 12.33 ± 0.68 | 21.2 ± 0.35 |
| 12 | 31.30 ± 0.17 | 31.33 ± 0.12 | 31.43 ± 0.21 | NA | NA | 23.37 ± 0.84 | 36.07 ± 1.67 | 32.10 ± 2.65 |
| 13 | NA | NA | NA | NA | NA | NA | NA | 45.20 ± 1.00 |
| 14 | 31.33 ± 0.23 | 31.28 ± 0.10 | 31.25 ± 0.13 | NA | 31.43 ± 0.20 | 38.30 ± 1.50 | NA | 40.10 ± 0.90 |
| 15 | 15.65 ± 0.18 | 15.53 ± 0.12 | 15.73 ± 0.12 | 62.67 ± 0.15 | 31.35 ± 0.22 | 25.07 ± 0.81 | 22.17 ± 1.45 | 30.20 ± 0.87 |
| 16 | 31.32 ± 0.25 | 31.33 ± 0.23 | NA | NA | NA | NA | NA | NA |
| 17 | NA | NA | NA | 31.28 ± 0.14 | NA | NA | 45.63 ± 1.79 | NA |
| 18 | NA | NA | NA | NA | NA | NA | NA | 42.07 ± 1.07 |
| erythromycin | NT | NT | 1.92 ± 0.06 | 3.93 ± 0.03 | 7.68 ± 0.10 | NT | NT | NT |
| chloramphenicol | 7.67 ± 0.13 | 7.87 ± 0.08 | NT | NT | NT | NT | NT | NT |
| doxorubicin | NT | NT | NT | NT | NT | 0.50 ± 0.05 | 0.09 ± 0.01 | 1.06 ± 0.07 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, W.; Liao, Y.; Tang, C.; Huang, X.; Luo, Z.; Chen, J.; Cai, P. Cytotoxic and Antibacterial Compounds from the Coral-Derived Fungus Aspergillus tritici SP2-8-1. Mar. Drugs 2017, 15, 348. https://doi.org/10.3390/md15110348
Wang W, Liao Y, Tang C, Huang X, Luo Z, Chen J, Cai P. Cytotoxic and Antibacterial Compounds from the Coral-Derived Fungus Aspergillus tritici SP2-8-1. Marine Drugs. 2017; 15(11):348. https://doi.org/10.3390/md15110348
Chicago/Turabian StyleWang, Weiyi, Yanyan Liao, Chao Tang, Xiaomei Huang, Zhuhua Luo, Jianming Chen, and Peng Cai. 2017. "Cytotoxic and Antibacterial Compounds from the Coral-Derived Fungus Aspergillus tritici SP2-8-1" Marine Drugs 15, no. 11: 348. https://doi.org/10.3390/md15110348
APA StyleWang, W., Liao, Y., Tang, C., Huang, X., Luo, Z., Chen, J., & Cai, P. (2017). Cytotoxic and Antibacterial Compounds from the Coral-Derived Fungus Aspergillus tritici SP2-8-1. Marine Drugs, 15(11), 348. https://doi.org/10.3390/md15110348