Sea Cucumber Glycosides: Chemical Structures, Producing Species and Important Biological Properties
Abstract
:1. Introduction
2. Taxonomy, Distribution and Nutritive Value of Sea Cucumbers
3. Extraction, Purification and Characterization
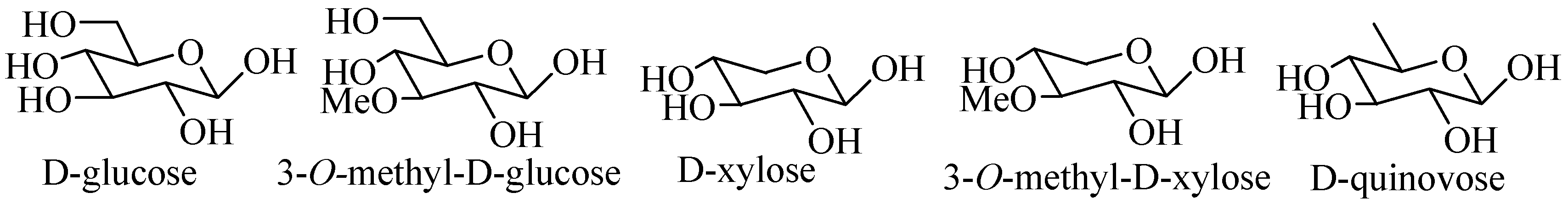
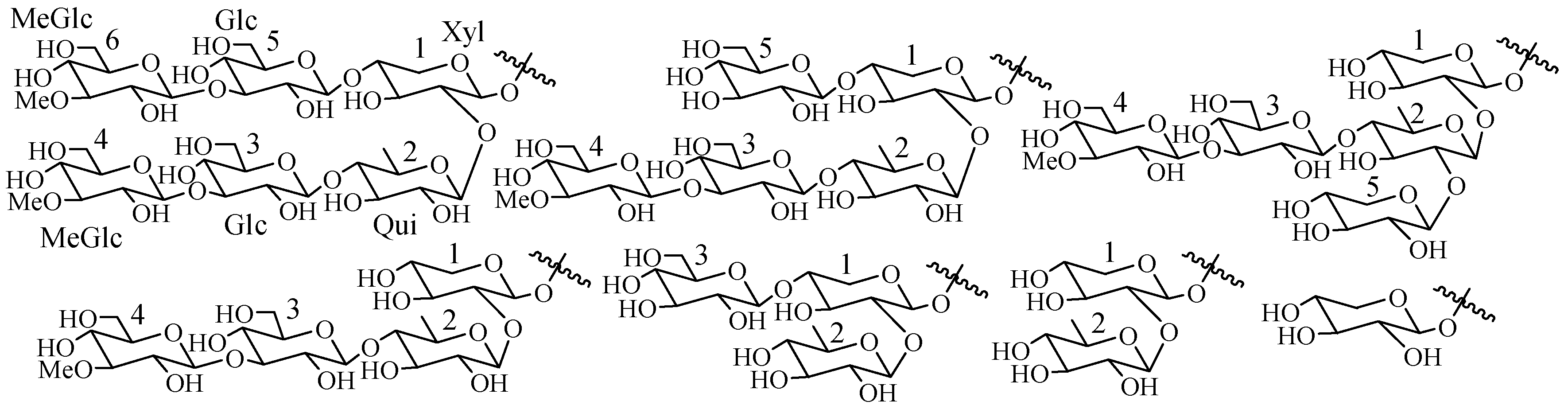
4. Structural Features of Triterpene Glycosides Isolated from Sea Cucumbers
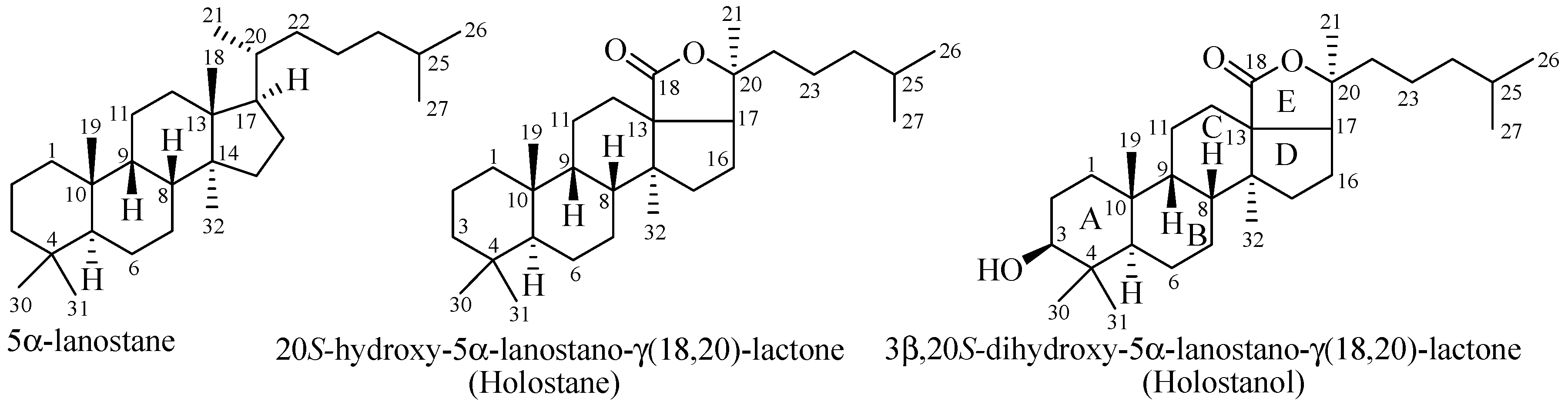
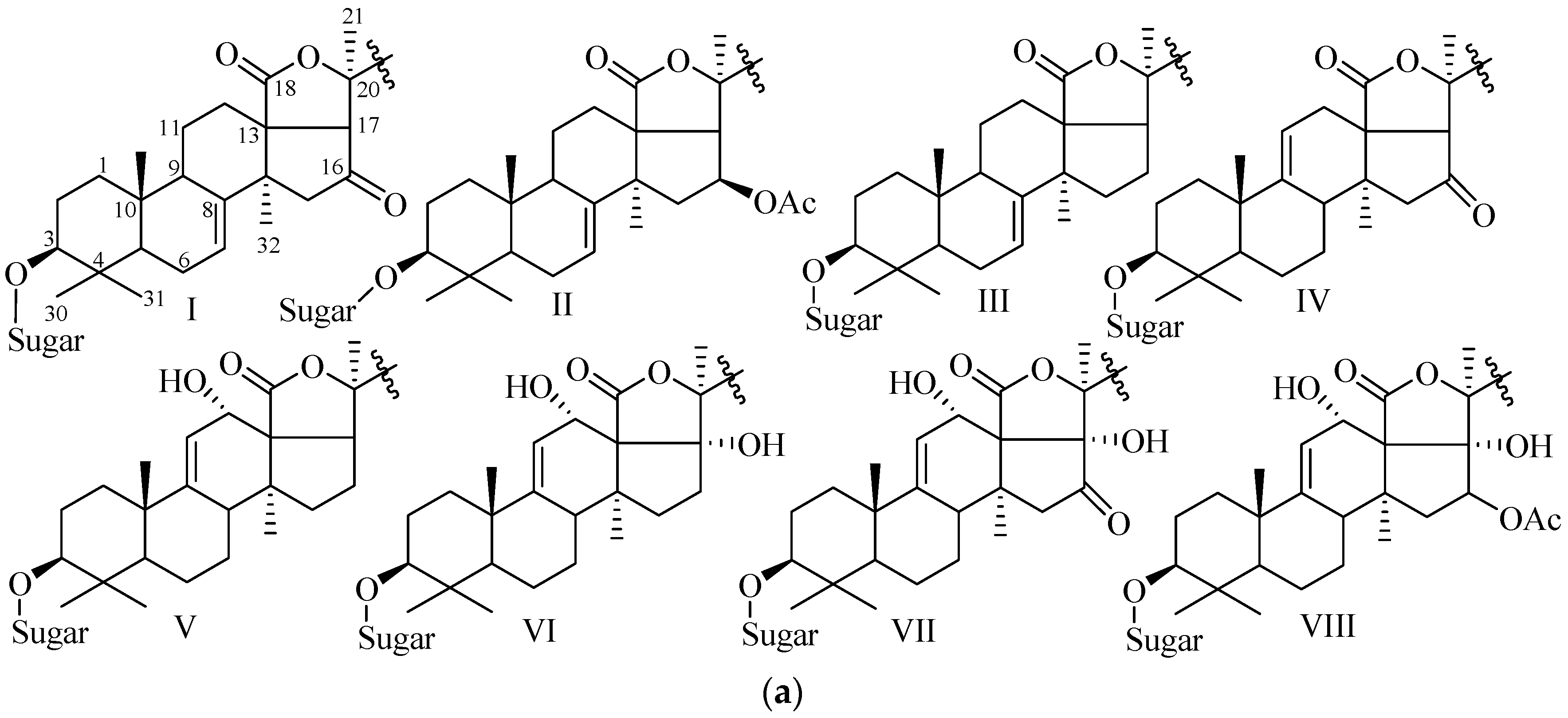
4.1. Holostane Type Triterpene Glycosides
4.1.1. 3β-Hydroxyholost-7(8)-ene Skeleton Containing Holostane Glycosides
Holostane Glycosides with 3β-Hydroxyholost-7(8)-ene Skeleton and Six Sugar Units
Holostane Glycosides with 3β-Hydroxyholost-7(8)-ene Skeleton and Five Sugar Units
Holostane Glycosides with 3β-Hydroxyholost-7(8)-ene Skeleton and Four Sugar Units
Holostane Glycosides with 3β-Hydroxyholost-7(8)-ene Skeleton and 1–3 Sugar Units
4.1.2. 3β-Hydroxyholost-9(11)-ene Skeleton Containing Holostane Glycosides
Holostane Glycosides with 3β-Hydroxyholost-9(11)-ene Skeleton and Six Sugar Units
Holostane Glycosides with 3β-Hydroxyholost-9(11)-ene Skeleton and Five Sugar Units
Holostane Glycosides with 3β-Hydroxyholost-9(11)-ene Skeleton and Four Sugar Units
Holostane Glycosides with 3β-Hydroxyholost-9(11)-ene Skeleton and 1–3 Sugar Units
4.1.3. Holostane Glycosides with 3β-Hydroxyholost-8(9)-ene Skeleton
4.2. Nonholostane Glycosides
5. The Important Biological Properties of Sea Cucumber Glycosides
6. Mechanisms of Action
7. Structure–Activity Relationships (SARs)
8. Conclusions
Acknowledgments
Conflicts of Interest
References
- Gomes, A.R.; Freitas, A.C.; Rocha-Santos, T.A.P.; Duarte, A.C. Bioactive compounds derived from echinoderms. RSC Adv. 2014, 4, 29365–29382. [Google Scholar] [CrossRef]
- Brusca, R.C.; Brusca, G.J. Invertebrates, 2nd ed.; Sinauer: Sunderland, MA, USA, 2003; pp. 808–826. [Google Scholar]
- Leal, M.C.; Madeira, C.; Brandão, C.A.; Puga, J.; Calado, R. Bioprospecting of marine invertebrates for new natural products-A chemical and zoogeographical perspective. Molecules 2012, 17, 9842–9854. [Google Scholar] [CrossRef] [PubMed]
- Shixiu, L.; Nina, A.; Yongjun, M.; Shujuan, S.; Wenjing, F.; Song, H. Bioactive compounds of sea cucumbers and their therapeutic effects. Chin. J. Oceanol. Limnol. 2016, 34, 549–558. [Google Scholar] [CrossRef]
- Janakiram, N.B.; Mohammed, A.; Rao, C.V. Sea cucumbers metabolites as potent anti-cancer agents. Mar. Drugs 2015, 13, 2909–2923. [Google Scholar] [CrossRef] [PubMed]
- Caulier, G.; Dyck, S.; Gerbaux, P.; Eeckhaut, I.; Flammang, P. Review of saponin diversity in sea cucumbers belonging to the family Holothuriidae. SPC Beche-de-mer Inf. Bull. 2011, 31, 48–54. [Google Scholar]
- Hyman, L.H. The Invertebrates. In Echinodermata; McGraw Hill: New York, NY, USA, 1955; Volume 4. [Google Scholar]
- Bruckner, A.W.; Johnson, K.; Field, J. Conservation strategies for sea cucumbers: Can a CITES Appendix II listing promote sustainable international trade? SPC Beche-de-mer Inf. Bull. 2003, 18, 24–33. [Google Scholar]
- Lawrence, J. A Functional Biology of Echinoderms = Functional Biology Series 6; Calow, P., Ed.; Croom Helm Ltd.: London, UK, 1987; p. 340. ISBN 0-7099-1642-6. [Google Scholar]
- Higgins, M. Sea cucumbers in a deep pickle. Environmental News Network, 30 August 2000. [Google Scholar]
- Purcell, S.W.; Samyn, Y.; Conand, C. Commercially important sea cucumbers of the world. In FAO Species Catalogue for Fishery Purposes; FAO: Rome, Italy, 2012; Volume 6, p. 150. [Google Scholar]
- Weici, T. Chinese medicinal materials from the sea. Abstr. Chin. Med. 1987, 4, 571–600. [Google Scholar]
- Yaacob, H.B.; Kim, K.H.; Shahimi, M.; Aziz, N.S.; Sahil, S.M. Malaysian sea cucumber (Gamat): A prospect in health food and therapeutic. In Proceedings of the Asian Food Technology Seminar, Kuala Lumpur, Malaysia, 6–7 October 1997; p. 6. [Google Scholar]
- Bordbar, S.; Anwar, F.; Saari, N. High-value components and bioactives from sea cucumbers for functional foods—A review. Mar. Drugs 2011, 9, 1761–1805. [Google Scholar] [CrossRef] [PubMed]
- Wen, J.; Hu, C.; Fan, S. Chemical composition and nutritional quality of sea cucumbers. J. Sci. Food Agric. 2010, 90, 2469–2474. [Google Scholar] [CrossRef] [PubMed]
- Silchenko, A.S.; Avilov, S.A.; Antonov, A.A.; Kalinin, V.I.; Kalinovsky, A.I.; Smirnov, A.V.; Riguera, R.; Jimenez, C. Triterpene Glycosides from the deep-water North-Pacific sea cucumber Synallactes nozawai Mitsukuri. J. Nat. Prod. 2002, 65, 1802–1808. [Google Scholar] [CrossRef] [PubMed]
- Yun, S.H.; Park, E.S.; Shin, S.W.; Na, Y.W.; Han, J.Y.; Jeong, J.S.; Shastina, V.V.; Stonik, V.A.; Park, J.I.; Kwak, J.Y. Stichoposide C induces apoptosis through the generation of ceramide in leukemia and colorectal cancer cells and shows in vivo antitumor activity. Clin. Cancer Res. 2012, 18, 5934–5948. [Google Scholar] [CrossRef] [PubMed]
- Stonik, V.A.; Maltsev, I.I.; Kalinovsky, A.I.; Conde, K.; Elyakov, G.B. Glycosides of marine-invertebrates. XI. Two novel triterpene glycosides from holothurians of Stichopodidae family. Chem. Nat. Prod. 1982, 2, 194–199. [Google Scholar]
- Mal’tsev, I.I.; Stonik, V.A.; Kalinovskii, A.I. Stichoposide E-A new triterpene glycoside from holothurians of the family Stichopodidae. Chem. Nat. Prod. 1983, 19, 292–295. [Google Scholar] [CrossRef]
- Kitagawa, I.; Kobuyashi, M.; Inamoto, T.; Yusuzawa, T.; Kyogoku, Y.; Kido, M. The structures of six antifungal oligoglycosides, stichlorosides A1, A2, B1, B2, C1 and C2 from the sea cucumber Stichopus chloronotus (Brandt). Chem. Pharm. Bull. 1981, 29, 2387–2391. [Google Scholar] [CrossRef]
- Wang, X.-H.; Zou, Z.-R.; Yi, Y.-H.; Han, H.; Li, L.; Pan, M.-X. Variegatusides: New non-sulphated triterpene glycosides from the sea cucumber Stichopus variegates. Mar. Drugs 2014, 12, 2004–2018. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Zhang, H.; Yuan, W.; Gong, W.; Tang, H.; Liu, B.; Krohn, K.; Li, L.; Yi, Y.; Zhang, W. Antifungal nortriterpene and triterpene glycosides from the sea cucumber Apostichopus japonicas Selenka. Food Chem. 2012, 1, 295–300. [Google Scholar] [CrossRef] [PubMed]
- Drozdova, O.A.; Avilov, S.A.; Kalinovskii, A.I.; Stonik, V.A.; Mil’grom, Y.M.; Rashkes, Y.V. New glycosides from the holothurian Cucumaria japonica. Chem. Nat. Compd. 1993, 29, 200–205. [Google Scholar] [CrossRef]
- Avilov, S.A.; Stonik, V.A.; Kalinovskii, A.I. Structures of four new triterpene glycosides from the holothurian Cucumaria japonica. Chem. Nat. Compd. 1990, 26, 670–675. [Google Scholar] [CrossRef]
- Avilov, S.; Tishchenko, L.Y.; Stonik, V.A. Structure of cucumarioside A2-2-A triterpene glycoside from the holothurian Cucumaria japonica. Chem. Nat. Compd. 1984, 20, 759–760. [Google Scholar] [CrossRef]
- Avilov, S.A.; Antonov, A.S.; Silchenko, A.S.; Kalinin, V.I.; Kalinovsky, A.I.; Dmitrenok, P.S.; Stonik, V.A.; Riguera, R.; Jimenez, C. Triterpene Glycosides from the Far Eastern sea cucumber Cucumaria conicospermium. J. Nat. Prod. 2003, 66, 910–916. [Google Scholar] [CrossRef] [PubMed]
- Drozdova, O.A.; Avilov, S.A.; Kalinin, V.I.; Kalinovsky, A.I.; Stonik, V.A.; Riguera, R.; Jiméne, C. Cytotoxic triterpene glycosides from Far-Eastern sea cucumbers belonging to the genus Cucumaria. Liebigs Ann. Chem. 1997, 11, 2351–2356. [Google Scholar] [CrossRef]
- Drozdova, O.A.; Avilov, S.A.; Kalinovskii, A.I.; Stonik, V.A.; Mil’grom, Y.M.; Rashkes, Y.V. Trisulfated glycosides from the holothurians Cucumaria japonica. Chem. Nat. Compd. 1993, 29, 309–313. [Google Scholar] [CrossRef]
- Silchenko, A.S.; Kalinovsky, A.I.; Avilov, S.A.; Andryjaschenko, P.V.; Dmitrenok, P.S.; Yurchenko, E.A.; Kalinin, V.I. Structure of cucumariosides H5, H6, H7 and H8, triterpene glycosides from the sea cucumber Eupentacta fraudatrix and unprecedented aglycone with 16,22-epoxy-group. Nat. Prod. Commun. 2011, 6, 1075–1082. [Google Scholar] [PubMed]
- Silchenko, A.S.; Kalinovsky, A.I.; Avilov, S.A.; Andryjaschenko, P.V.; Dmitrenok, P.S.; Yurchenko, E.A.; Kalinin, V.I. Structures and cytotoxic properties of cucumariosides H2, H3 and H4 from the sea cucumber Eupentacta fraudatrix. Nat. Prod. Res. 2012, 26, 1765–1774. [Google Scholar] [CrossRef] [PubMed]
- Silchenko, A.S.; Kalinovsky, A.I.; Avilov, S.A.; Andryjaschenko, P.V.; Dmitrenok, P.S.; Martyyas, E.A.; Kalinin, V.I. Triterpene glycosides from the sea cucumber Eupentacta fraudatrix. Structure and biological action of cucumariosides I1, I3, I4, three new minor disulfated pentaosides. Nat. Prod. Commun. 2013, 8, 1053–1058. [Google Scholar] [PubMed]
- Silchenko, A.S.; Kalinovsky, A.I.; Avilov, S.A.; Andryjaschenko, P.V.; Dmitrenok, P.S.; Menchinskaya, E.S.; Aminin, D.L.; Kalinin, V.I. Structure of cucumarioside I2 from the sea cucumber Eupentacta fraudatrix (Djakonov et Baranova) and cytotoxic and immunostimulatory activities of this saponin and relative compounds. Nat. Prod. Res. 2013, 27, 1776–1783. [Google Scholar] [CrossRef] [PubMed]
- Girard, M.; Bélanger, J.; ApSimon, J.W.; Garneau, F.X.; Harvey, C.; Brisson, J.R. Frondoside A, a novel triterpene glycoside from the holothurians Cucumaria frondosa. Can. J. Chem. 1990, 68, 11–18. [Google Scholar] [CrossRef]
- Findlay, J.A.; Yayli, N.; Radics, L. Novel sulfated oligosaccharides from the sea cucumber Cucumaria frondosa. J. Nat. Prod. 1992, 55, 93–101. [Google Scholar] [CrossRef] [PubMed]
- Silchenko, A.S.; Avilov, S.A.; Antonov, A.S.; Kalinovsky, A.I.; Dmitrenok, P.S.; Kalinin, V.I.; Stonik, V.A.; Woodward, C.; Collin, P.D. Glycosides from the sea cucumber Cucumaria frondosa. III. Structure of frondosides A2-1, A2-2, A2-3, and A2-6, four new minor monosulfated triterpene glycosides. Can. J. Chem. 2005, 83, 21–27. [Google Scholar] [CrossRef]
- Silchenko, A.S.; Avilov, S.A.; Antonov, A.S.; Kalinovsky, A.I.; Dmitrenok, P.S.; Kalinin, V.I.; Stonik, V.A.; Woodward, C.; Collin, P.D. Glycosides from the sea cucumber Cucumaria frondosa. IV. Structure of frondosides A2-4, A2-7, and A2-8, three new minor monosulfated triterpene glycosides. Can. J. Chem. 2005, 83, 2120–2126. [Google Scholar] [CrossRef]
- Avilov, S.A.; Antonov, A.S.; Drozdova, O.A.; Kalinin, V.I.; Kalinovsky, A.I.; Stonik, V.A.; Riguera, R.; Lenis, L.A.; Jiménez, C. Triterpene glycosides from the Far-Eastern sea cucumber Pentamera calcigera. 1. Monosulfated glycosides and cytotoxicity of their unsulfated derivatives. J. Nat. Prod. 2000, 63, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Avilov, S.A.; Antonov, A.S.; Drozdova, O.A.; Kalinin, V.I.; Kalinovsky, A.I.; Riguera, R.; Lenis, L.A.; Jiménez, C. Triterpene Glycosides from the far eastern sea cucumber Pentamera calcigera II: Disulfated glycosides. J. Nat. Prod. 2000, 63, 1349–1355. [Google Scholar] [CrossRef] [PubMed]
- Yong-Juan, Z.; Yang-Hua, Y. Antitumor activities in vitro of the triterpene glycoside colochiroside A from sea cucumber Colochirus anceps. J. Mod. Oncol. 2011, 2, 205–207. [Google Scholar]
- Afiyatullov, S.S.; Kalinovskii, A.I.; Stonik, V.A. Structures of cucumariosides C1 and C2-Two new triterpene glycosides from the holothurians Eupentacta fraudatrix. Chem. Nat. Compd. 1987, 23, 691–696. [Google Scholar] [CrossRef]
- Avilov, S.A.; Silchenko, A.S.; Antonov, A.S.; Kalinin, V.I.; Kalinovsky, A.I.; Smirnov, A.V.; Dmitrenok, P.S.; Evtushenko, E.V.; Fedorov, S.N.; Savina, A.S.; et al. Synaptosides A and A1, triterpene glycosides from the sea cucumber Synapta maculata containing 3-O-methylglucuronic acid and their cytotoxic activity against tumor cells. J. Nat. Prod. 2008, 71, 525–531. [Google Scholar] [CrossRef] [PubMed]
- Silchenko, A.S.; Avilov, S.A.; Kalinin, V.I.; Stonik, V.A.; Kalinovskiĭ, A.I.; Dmitrenok, P.S.; Stepanov, V.G. Monosulfated triterpene glycosides from Cucumaria okhotensis Levin et Stepanov, a new species of sea cucumbers from sea of Okhotsk. Russ. J. Bioorg. Chem. 2007, 33, 73–82. [Google Scholar] [CrossRef]
- Silchenko, A.S.; Avilov, S.A.; Kalinovsky, A.I.; Dmitrenok, P.S.; Kalinin, V.I.; Morre, J.; Deinzer, M.L.; Woodward, C.; Collin, P.D. Glycosides from the North Atlantic sea cucumber Cucumaria frondosa V-Structures of five new minor trisulfated triterpene oligoglycosides, frondosides A7-1, A7-2, A7-3, A7-4, and isofrondoside C. Can. J. Chem. 2007, 85, 626–636. [Google Scholar] [CrossRef]
- Maier, M.S.; Roccatagliata, A.J.; Kuriss, A.; Chludil, H.; Seldes, A.M.; Pujol, C.A.; Damonte, E.B. Two new cytotoxic and virucidal trisulfated triterpene glycosides from the Antarctic sea cucumber Staurocucumis liouvillei. J. Nat. Prod. 2001, 64, 732–736. [Google Scholar] [CrossRef] [PubMed]
- Antonov, A.S.; Avilov, S.A.; Kalinovsky, A.I.; Anastyuk, S.D.; Dmitrenok, P.S.; Evtushenko, E.V.; Kalinin, V.I.; Smirnov, A.V.; Taboada, S.; Ballesteros, M.; et al. Triterpene glycosides from Antarctic sea cucumbers. 1. Structure of liouvillosides A1, A2, A3, B1, and B2 from the sea cucumber Staurocucumis liouvillei: New procedure for separation of highly polar glycoside fractions and taxonomic revision. J. Nat. Prod. 2008, 71, 1677–1685. [Google Scholar] [CrossRef] [PubMed]
- Antonov, A.S.; Avilov, S.A.; Kalinovsky, A.I.; Dmitrenok, P.S.; Kalinin, V.I.; Taboada, S.; Ballesteros, M.; Avila, C. Triterpene glycosides from Antarctic sea cucumbers III. Structures of liouvillosides A4 and A5, two minor disulphated tetraosides containing 3-O-methylquinovose as terminal monosaccharide units from the sea cucumber Staurocucumis liouvillei (Vaney). Nat. Prod. Res. 2011, 25, 1324–1333. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.Y.; Yi, Y.H.; Tang, H.F.; Li, L.; Sun, P.; Wu, J. Two new bioactive triterpene glycosides from the sea cucumber Pseudocolochirus violaceus. J. Asian Nat. Prod. Res. 2006, 8, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.Y.; Yi, Y.H.; Tang, H.F. Cytotoxic sulfated triterpene glycosides from the sea cucumber Pseudocolochirus violaceus. Chem. Biodivers. 2006, 3, 807–817. [Google Scholar] [CrossRef] [PubMed]
- Zou, Z.-R.; Yi, Y.-H.; Wu, H.-M.; Wu, J.-H.; Liaw, C.C.; Lee, K.H. Intercedensides A–C, three new cytotoxic triterpene glycosides from the sea cucumber Mensamaria intercedens Lampert. J. Nat. Prod. 2003, 66, 1055–1060. [Google Scholar] [CrossRef] [PubMed]
- Zou, Z.; Yi, Y.; Wu, H.; Yao, X.; Du, L.; Jiuhong, W.; Liaw, C.C.; Lee, K.H. Intercedensides D–I, cytotoxic triterpene glycosides from the sea cucumber Mensamaria intercedens Lampert. J. Nat. Prod. 2005, 68, 540–546. [Google Scholar] [CrossRef] [PubMed]
- Murray, A.P.; Muniaı́n, C.; Seldes, A.M.; Maier, M.S. Patagonicoside A: A novel antifungal disulfated triterpene glycoside from the sea cucumber Psolus patagonicus. Tetrahedron 2001, 57, 9563–9568. [Google Scholar] [CrossRef]
- Careaga, V.P.; Muniain, C.; Maier, M.S. Patagonicosides B and C, two antifungal sulfated triterpene glycosides from the sea cucumber Psolus patagonicus. Chem. Biodivers. 2011, 8, 467–475. [Google Scholar] [CrossRef] [PubMed]
- Yi, Y.H.; Xu, Q.Z.; Li, L.; Zhang, S.L.; Wu, H.M.; Ding, J.; Tong, Y.G.; Tan, W.F.; Li, M.H.; Tian, F.; et al. Philinopsides A and B, two new sulfated triterpene glycosides from the sea cucumber Pentacta quadrangularis. Helv. Chim. Acta 2006, 9, 54–63. [Google Scholar] [CrossRef]
- Zhang, S.L.; Li, L.; Yi, Y.H.; Sun, P. Philinopsides E and F, two new sulfated triterpene glycosides from the sea cucumber Pentacta quadrangularis. Nat. Prod. Res. 2006, 20, 399–407. [Google Scholar] [CrossRef] [PubMed]
- Moraes, G.; Northcote, P.T.; Silchenko, A.S.; Antonov, A.S.; Kalinovsky, A.I.; Dmitrenok, P.S.; Avilov, S.A.; Kalinin, V.I.; Stonik, V.A. Mollisosides A, B1, and B2: Minor triterpene glycosides from the New Zealand and South Australian sea cucumber Australostichopus mollis. J. Nat. Prod. 2005, 68, 842–847. [Google Scholar] [CrossRef] [PubMed]
- Kalinin, V.I.; Avilov, S.A.; Kalinina, E.Y.; Korolkova, O.G.; Kalinovsky, A.I.; Stonik, V.A.; Riguera, R.; Jiménez, C. Structure of eximisoside A, a novel triterpene glycoside from the far-eastern sea cucumber Psolus eximius. J. Nat. Prod. 1997, 60, 817–819. [Google Scholar] [CrossRef] [PubMed]
- Kalinin, V.I.; Stonik, V.A.; Kalinovskii, A.I.; Isakov, V.V. Structure of pseudostichoposide A-The main triterpene glycoside from the holothurian Pseudostichopus trachus. Chem. Nat. Compd. 1989, 25, 577–582. [Google Scholar] [CrossRef]
- Popov, R.S.; Avilov, S.A.; Silchenko, A.S.; Kalinovsky, A.I.; Dmitrenok, P.S.; Grebnev, B.B.; Ivanchina, N.V.; Kalinin, V.I. Cucumariosides F1 and F2, two new triterpene glycosides from the sea cucumber Eupentacta fraudatrix and their LC-ESI MS/MS identification in the starfish Patiria pectinifera, a predator of the sea cucumber. Biochem. Syst. Ecol. 2014, 57, 191–197. [Google Scholar] [CrossRef]
- Careaga, V.P.; Bueno, C.; Muniain, C.; Alché, L.; Maier, M.S. Pseudocnoside A, a new cytotoxic and antiproliferative triterpene glycoside from the sea cucumber Pseudocnus dubiosus leoninus. Nat. Prod. Res. 2014, 28, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Silchenko, A.S.; Kalinovsky, A.I.; Avilov, S.A.; Andryjaschenko, P.V.; Dmitrenok, P.S.; Martyyas, E.A.; Kalinin, V.I.; Jayasandhya, P.; Rajan, G.C.; Padmakumar, K.P. Structures and biological activities of typicosides A1, A2, B1, C1 and C2, triterpene glycosides from the sea cucumber Actinocucumis typica. Nat. Prod. Commun. 2013, 8, 301–310. [Google Scholar] [PubMed]
- Aminin, D.L.; Silchenko, A.S.; Avilov, S.A.; Stepanov, V.G.; Kalinin, V.I. Immunomodulatory action of monosulfated triterpene glycosides from the sea cucumber Cucumaria okhotensis: Stimulation of activity of mouse peritoneal macrophages. Nat. Prod. Commun. 2010, 5, 1877–1880. [Google Scholar] [PubMed]
- Silchenko, A.S.; Avilov, S.A.; Kalinin, V.I.; Kalinovsky, A.I.; Dmitrenok, P.S.; Fedorov, S.N.; Stepanov, V.G.; Dong, Z.; Stonik, V.A. Constituents of the sea cucumber Cucumaria okhotensis. Structures of okhotosides B1–B3 and cytotoxic activities of some glycosides from this species. J. Nat. Prod. 2008, 71, 351–356. [Google Scholar] [CrossRef] [PubMed]
- Silchenkoa, A.S.; Kalinovskya, A.I.; Avilova, S.A.; Andryjaschenko, P.V.; Dmitrenok, P.S.; Yurchenko, E.A.; Kalinin, V.I.; Dolmatov, I.Y. Colochirosides A1, A2, A3, and D, four novel sulfated triterpene glycosides from the sea cucumber Colochirus robustus (Cucumariidae, Dendrochirotida). Nat. Prod. Commun. 2016, 11, 381–386. [Google Scholar]
- Silchenkoa, A.S.; Kalinovskya, A.I.; Avilova, S.A.; Andryjaschenko, P.V.; Dmitrenok, P.S.; Yurchenko, E.A.; Kalinin, V.I.; Dolmatov, I.Y. Colochirosides B1, B2, B3 and C, novel sulfated triterpene glycosides from the sea cucumber Colochirus robustus (Cucumariidae, Dendrochirotida). Nat. Prod. Commun. 2015, 10, 687–694. [Google Scholar]
- Silchenko, A.S.; Kalinovsky, A.I.; Avilov, S.A.; Andryjaschenko, P.V.; Dmitrenok, P.S.; Kalinin, V.I.; Yurchenko, E.A.; Dautov, S.S. Structures of violaceusosides C, D, E and G, sulfated triterpene glycosides from the sea cucumber Pseudocolochirus violaceus (Cucumariidae, Dendrochirotida). Nat. Prod. Commun. 2014, 9, 391–399. [Google Scholar] [PubMed]
- Silchenko, A.S.; Kalinovsky, A.I.; Avilov, S.A.; Andryjaschenko, P.V.; Dmitrenok, P.S.; Martyyas, E.A.; Kalinin, V.I. Triterpene glycosides from the sea cucumber Eupentacta fraudatrix. Structure and biological action of cucumariosides A1, A3, A4, A5, A6, A12 and A15, seven new minor non-sulfated tetraosides and unprecedented 25-keto, 27-norholostane aglycone. Nat. Prod. Commun. 2012, 7, 517–525. [Google Scholar] [PubMed]
- Silchenko, A.S.; Kalinovsky, A.I.; Avilov, S.A.; Andryjaschenko, P.V.; Dmitrenok, P.S.; Martyyas, E.A.; Kalinin, V.I. Triterpene glycosides from the sea cucumber Eupentacta fraudatrix. Structure and cytotoxic action of cucumariosides A2, A7, A9, A10, A11, A13 and A14, seven new minor non-sulfated tetraosides and an aglycone with an uncommon 18-hydroxy group. Nat. Prod. Commun. 2012, 7, 845–852. [Google Scholar] [PubMed]
- Afiyatullov, S.S.; Tishchenko, L.Y.; Stonik, V.A.; Kalinovskii, A.I.; Elyakov, G.B. Structure of cucumarioside G1-A new triterpene glycoside from the holothurians Cucumaria fraudatrix. Chem. Nat. Compd. 1985, 21, 228–232. [Google Scholar] [CrossRef]
- Kalinin, V.I.; Avilov, S.A.; Kalinovskii, A.I.; Stonik, V.A. Cucumarioside G3-A minor triterpene glycoside from the holothurian Eupentacta fraudatrix. Chem. Nat. Compd. 1992, 28, 635–636. [Google Scholar] [CrossRef]
- Kalinin, V.I.; Avilov, S.A.; Kalinovskii, A.I.; Stonik, V.A.; Mil’grom, Y.M.; Rashkes, Y.V. Cucumarioside G4-A new triterpenglycoside from the holothurian Eupentacta fraudatrix. Chem. Nat. Compd. 1992, 28, 600–603. [Google Scholar] [CrossRef]
- Han, H.; Xu, Q.Z.; Yi, Y.H.; Gong, W.; Jiao, B.H. Two new cytotoxic disulfated holostane glycosides from the sea cucumber Pentacta quadrangularis. Chem. Biodivers. 2010, 7, 158–167. [Google Scholar] [CrossRef] [PubMed]
- Silchenko, A.S.; Avilov, S.A.; Kalinin, V.I.; Kalinovsky, A.I.; Stonik, V.A.; Smirnov, A.V. Pseudostichoposide B-New triterpene glycoside with unprecedent type of sulfatation from the deep-water North-Pacific Sea cucumber Pseudostichopus trachus. Nat. Prod. Res. 2004, 18, 565–570. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.-H.; Li, L.; Yi, Y.-H.; Sun, P.; Yan, B.; Pan, M.-X.; Han, H.; Wang, X.-D. Two new triterpene glycosides from sea cucumber Stichopus variegatus. Chin. J. Nat. Med. 2006, 4, 177–180. [Google Scholar]
- Stonik, V.A.; Mal’tsev, I.I.; Elyakov, G.B. The structure of thelenotosides A and B from the holothurians Thelenota ananas. Chem. Nat. Compd. 1982, 18, 590–593. [Google Scholar] [CrossRef]
- Miyamoto, T.; Togawa, K.; Higuchi, R.; Komori, T.; Sasaki, T. Constituents of holothuroidea, II. Six newly identified biologically active triterpenoid glycoside sulfates from the sea cucumber Cucumaria echinata. Liebigs Ann. Chem. 1990, 5, 453–460. [Google Scholar] [CrossRef]
- Rodriguez, J.; Riguera, R. Lefevreiosides: Four new triterpene glycosides from the sea cucumber Cucumaria lefevrei. J. Chem. Res. 1989, 11, 342–343. [Google Scholar]
- Han, H.; Xu, Q.Z.; Tang, H.F.; Yi, Y.H.; Gong, W. Cytotoxic holostane-type triterpene glycosides from the sea cucumber Pentacta quadrangularis. Planta Med. 2010, 76, 1900–1904. [Google Scholar] [CrossRef] [PubMed]
- Silchenko, A.S.; Kalinovsky, A.I.; Avilov, S.A.; Andryjaschenko, P.V.; Dmitrenok, P.S.; Martyyas, E.A.; Kalinin, V.I. Triterpene glycosides from the sea cucumber Eupentacta fraudatrix. Structure and biological activity of cucumariosides B1 and B2, two new minor non-sulfated unprecedented triosides. Nat. Prod. Commun. 2012, 7, 1157–1162. [Google Scholar] [PubMed]
- Sharypov, V.F.; Chumak, A.D.; Stonik, V.A.; Elyakov, G.B. Glycosides of marine invertebrates. X. The structure of stichoposides A and B from the holothurians Stichopus cloronotus. Chem. Nat. Compd. 1981, 17, 139–142. [Google Scholar] [CrossRef]
- Cuong, N.X.; Vien, L.T.; Hoang, L.; Hanh, T.T.H.; Thao, D.T.; Thanh, N.V.; Nam, N.H.; Thung, D.C.; Kiem, P.V.; Minh, C.V. Cytotoxic triterpene diglycosides from the sea cucumber Stichopus horrens. Bioorg. Med. Chem. Lett. 2017, 27, 2939–2942. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Yi, Y.H.; Tang, H.F.; Wu, H.M.; Zhou, Z.R. Hillasides A and B, two new cytotoxic triterpene glycosides from the sea cucumber Holothuria hilla Lesson. J. Asian Nat. Prod. Res. 2007, 9, 609–615. [Google Scholar] [CrossRef] [PubMed]
- Dong, P.; Xue, C.-H.; Yu, L.-F.; Xu, J.; Chen, S.-G. Determination of triterpene glycosides in sea cucumber (Stichopus japonicus) and its related products by high-performance liquid chromatography. J. Agric. Food Chem. 2008, 56, 4937–4942. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Gong, W.; Sun, G.; Tang, H.; Liu, B.; Li, L.; Yi, Y.; Zhang, W. New holostan-type triterpene glycosides from the sea cucumber Apostichopus japonicus. Nat. Prod. Commun. 2012, 7, 1431–1434. [Google Scholar] [PubMed]
- Iñiguez-Martinez, A.M.; Guerra-Rivas, G.; Rios, T.; Quijano, L. Triterpenoid oligoglycosides from the sea cucumber Stichopus parvimensis. J. Nat. Prod. 2005, 68, 1669–1673. [Google Scholar] [CrossRef] [PubMed]
- Ohta, T.; Hikino, H. Structures of four new triterpenoidal oligosides, bivittosides A, B, C and D from the sea cucumber Bohadschia bivittata Mitsukuri. Chem. Pharm. Bull. 1981, 29, 282–285. [Google Scholar]
- Yuan, W.H.; Yi, Y.H.; Tang, H.F.; Liu, B.S.; Wang, Z.L.; Sun, G.Q.; Zhang, W.; Li, L.; Sun, P. Antifungal triterpene glycosides from the sea cucumber Bohadschia marmorata. Planta Med. 2009, 75, 168–173. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.S.; Yi, Y.H.; Li, L.; Sun, P.; Yuan, W.H.; Sun, G.Q.; Han, H.; Xue, M. Argusides B and C, two new cytotoxic triterpene glycosides from the sea cucumber Bohadschia argus. Chem. Biodivers. 2008, 5, 1288–1297. [Google Scholar] [CrossRef] [PubMed]
- Bahrami, Y.; Franco, C.M.M. Structure elucidation of new acetylated saponins, lessoniosides A, B, C, D, and E, and non-acetylated saponins, lessoniosides F and G, from the viscera of the sea cucumber Holothuria lessoni. Mar. Drugs 2015, 13, 597–617. [Google Scholar] [CrossRef] [PubMed]
- Dyck, S.V.; Gerbaux, P.; Flammang, P. Qualitative and quantitative saponin contents in five sea cucumbers from the Indian Ocean. Mar. Drugs 2010, 8, 173–189. [Google Scholar] [CrossRef] [PubMed]
- Silchenko, A.S.; Kalinovsky, A.I.; Avilov, S.A.; Andryjaschenko, P.V.; Dmitrenok, P.S.; Yurchenko, E.A.; Dolmatov, I.Y.; Kalinin, V.I.; Stonik, V.A. Structure and biological action of cladolosides B1, B2, C, C1, C2 and D, six new triterpene glycosides from the sea cucumber Cladolabes schmeltzii. Nat. Prod. Commun. 2013, 8, 1527–1534. [Google Scholar] [PubMed]
- Silchenko, A.S.; Kalinovsky, A.I.; Avilov, S.A.; Andryjaschenko, P.V.; Dmitrenok, P.S.; Yurchenko, E.A.; Dolmatov, I.Y.; Kalinin, V.I. Structures and biological activities of cladolosides C3, E1, E2, F1, F2, G, H1 and H2, eight triterpene glycosides from the sea cucumber Cladolabes schmeltzii with one known and four new carbohydrate chains. Carbohydr. Res. 2015, 23, 22–31. [Google Scholar] [CrossRef] [PubMed]
- Silchenko, A.S.; Kalinovsky, A.I.; Avilov, S.A.; Andryjaschenko, P.V.; Dmitrenok, P.S.; Chingizova, E.A.; Dolmatov, I.Y.; Kalinin, V.I. Cladolosides I1, I2, J1, K1, K2 and L1, monosulfated triterpene glycosides with new carbohydrate chains from the sea cucumber Cladolabes schmeltzii. Carbohydr. Res. 2017, 5, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, J.; Castro, R.; Riguera, R. Holothurinosides: New antitumour non sulphated triterpenoid glycosides from the sea cucumber Holothuria forskalii. Tetrahedron 1991, 47, 4753–4762. [Google Scholar] [CrossRef]
- Sun, G.-Q.; Li, L.; Yi, Y.-H.; Yuan, W.-H.; Liu, B.-S.; Weng, Y.-Y.; Zhang, S.-L.; Sun, P.; Wang, Z.-L. Two new cytotoxic nonsulfated pentasaccharide holostane (=20-hydroxylanostan-18-oic acid gamma-lactone) glycosides from the sea cucumber Holothuria grisea. Helv. Chim. Acta 2008, 9, 1453–1460. [Google Scholar] [CrossRef]
- Bahrami, Y.; Zhang, W.; Franco, C. Discovery of novel saponins from the viscera of the sea cucumber Holothuria lessoni. Mar. Drugs 2014, 12, 2633–2667. [Google Scholar] [CrossRef] [PubMed]
- Silchenko, A.S.; Kalinovsky, A.I.; Avilov, S.A.; Andryjashchenko, P.V.; Fedorov, S.N.; Dmitrenok, P.S.; Yurchenko, E.A.; Kalinin, V.I.; Rogacheva, A.V.; Gebruk, A.V. Kolgaosides A and B, two new triterpene glycosides from the Arctic deep water sea cucumber Kolga hyalina (Elasipodida: Elpidiidae). Nat. Prod. Commun. 2014, 9, 1259–1264. [Google Scholar] [PubMed]
- Yuan, W.H.; Yi, Y.H.; Tan, R.X.; Wang, Z.L.; Sun, G.Q.; Xue, M.; Zhang, H.W.; Tang, H.F. Antifungal triterpene glycosides from the sea cucumber Holothuria (Microthele) axiloga. Planta Med. 2009, 75, 647–653. [Google Scholar] [CrossRef] [PubMed]
- Antonov, A.S.; Avilov, S.A.; Kalinovsky, A.I.; Anastyuk, S.D.; Dmitrenok, P.S.; Kalinin, V.I.; Taboada, S.; Bosh, A.; Avila, C.; Stonik, V.A. Triterpene glycosides from Antarctic sea cucumbers. 2. Structure of achlioniceosides A1, A2, and A3 from the sea cucumber Achlionice violaecuspidata (=Rhipidothuria racowitzai). J. Nat. Prod. 2009, 72, 33–38. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, T.; Togawa, K.; Higuchi, R.; Komori, T.; Sasaki, T. Structures of four new triterpenoid oligoglycosides: DS-Penaustrosides A, B, C, and D from the sea cucumber Pentacta australis. J. Nat. Prod. 1992, 55, 940–946. [Google Scholar] [CrossRef] [PubMed]
- Cuong, N.X.; Vien, L.T.; Hanh, T.T.; Thao, N.P.; Thaodo, T.; Thanh, N.V.; Nam, N.H.; Thungdo, C.; Kiem, P.V.; Minh, C.V. Cytotoxic triterpene saponins from Cercodemas anceps. Bioorg. Med. Chem. Lett. 2015, 15, 3151–3156. [Google Scholar] [CrossRef] [PubMed]
- Chanley, J.D.; Ledeen, R.; Wax, J.; Nigrelli, R.F.; Sobotka, H. Holothurin I. The isolation, properties and sugar components of holothurin A. J. Am. Chem. Soc. 1959, 81, 5180–5183. [Google Scholar] [CrossRef]
- Oleinikova, G.K.; Kuznetsova, T.A.; Ivanova, N.S.; Kalinovskii, A.I.; Rovnykh, N.V.; Elyakov, G.B. Glycosides of marine invertebrates. XV. A new triterpene glycoside-Holothurin A1-from Caribbean holothurians of the family Holothuriidae. Chem. Nat. Compd. 1982, 18, 430–434. [Google Scholar] [CrossRef]
- Dang, N.H.; Thanh, N.V.; Kiem, P.V.; Huong, M.; Minh, C.V.; Kim, Y.H. Two new triterpene glycosides from the Vietnamese sea cucumber Holothuria scabra. Arch. Pharm. Res. 2007, 30, 1387–1391. [Google Scholar] [CrossRef] [PubMed]
- Han, H.; Yi, Y.; Xu, Q.; La, M.; Zhang, H. Two new cytotoxic triterpene glycosides from the sea cucumber Holothuria scabra. Planta Med. 2009, 75, 1608–1612. [Google Scholar] [CrossRef] [PubMed]
- Han, H.; Li, L.; Yi, Y.-H.; Wang, X.-H.; Pan, M.-X. Triterpene glycosides from sea cucumber Holothuria scabra with cytotoxic activity. Chin. Herb. Med. 2012, 4, 183–188. [Google Scholar] [CrossRef]
- Zhang, S.-Y.; Yi, Y.-H.; Tang, H.-F. Bioactive Triterpene glycosides from the sea cucumber Holothuria fuscocinerea. J. Nat. Prod. 2006, 69, 1492–1495. [Google Scholar] [CrossRef] [PubMed]
- Yuan, W.; Yi, Y.; Tang, H.; Xue, M.; Wang, Z.; Sun, G.; Zhang, W.; Liu, B.; Li, L.; Sun, P. Two new holostan-type triterpene glycosides from the sea cucumber Bohadschia marmorata. Chem. Pharm. Bull. 2008, 56, 1207–1211. [Google Scholar] [CrossRef] [PubMed]
- Kitagawa, I.; Inamoto, T.; Fuchida, M.; Okada, S.; Kobayashi, M.; Nishino, T.; Kyogoku, Y. Structures of echinosides A and B, two antifungal oligoglycosides from the sea cucumber Actinopyga echinites. Chem. Pharm. Bull. 1980, 28, 1651–1653. [Google Scholar] [CrossRef]
- Zhao, Q.; Liu, Z.-D.; Xue, Y.; Wang, J.F.; Li, H.; Tang, Q.J.; Wang, Y.M.; Dong, P.; Xue, C.H. Ds-echinoside A, a new triterpene glycoside derived from sea cucumber, exhibits antimetastatic activity via the inhibition of NF-κB-dependent MMP-9 and VEGF expressions. J. Zhejiang Univ. Sci. B 2011, 12, 534–544. [Google Scholar] [CrossRef] [PubMed]
- Bhatnagar, S.; Dudouet, B.; Ahond, A.; Poupat, C.; Thoison, O.; Clastres, A.; Laurent, D.; Potier, P. Invertébrés marins du lagon néocalédonien. IV: Saponines et sapogénines d’une holothurie, Actinopyga flammea. Bull. Soc. Chim. Fr. 1985, 1, 124–129. (In French) [Google Scholar]
- Kitagawa, I.; Kobayashi, M.; Son, B.W.; Siuzukia, S.; Kyogokub, Y. Marine Natural Products. XIX. Pervicosides A, B, and C, lanostane-type triterpene-oligoglycoside sulfates from the sea cucumber Holothuria pervicax. Chem. Pharm. Bull. 1989, 37, 1230–1234. [Google Scholar] [CrossRef]
- Liu, B.-S.; Yi, Y.-H.; Li, L.; Zhang, S.L.; Han, H.; Weng, Y.Y.; Pan, M. Arguside A: A new cytotoxic triterpene glycoside from the sea cucumber Bohadschia argus. Chem. Biodivers. 2007, 4, 2845–2851. [Google Scholar] [CrossRef] [PubMed]
- Chludil, H.D.; Muniain, C.C.; Seldes, A.M.; Maier, M.S. Cytotoxic and antifungal triterpene glycosides from the patagonian sea cucumber Hemoiedema spectabilis. J. Nat. Prod. 2002, 65, 860–865. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.-S.; Yi, Y.-H.; Li, L.; Sun, P.; Han, H.; Sun, G.Q.; Wang, X.H.; Wang, Z.L. Argusides D and E, two new cytotoxic triterpene glycosides from the sea cucumber Bohadschia argus. Chem. Biodivers. 2008, 5, 1425–1433. [Google Scholar] [CrossRef] [PubMed]
- Kalinin, V.I.; Kalinovsky, A.I.; Stonik, V.A. Structure of psolusoside A-the major triterpene glycoside from holothurian Psolus fabricii. Chem. Nat. Compd. 1985, 21, 197–202. [Google Scholar] [CrossRef]
- Bahrami, Y.; Zhang, W.; Chataway, T.; Franco, C. Structural elucidation of novel saponins in the sea cucumber Holothuria lessoni. Mar. Drugs 2014, 12, 4439–4473. [Google Scholar] [CrossRef] [PubMed]
- Silchenko, A.S.; Kalinovsky, A.I.; Avilov, S.A.; Andryjaschenko, P.V.; Dmitrenok, P.S.; Yurchenko, E.A.; Dolmatov, I.Y.; Savchenko, A.M.; Kalinin, V.I. Triterpene glycosides from the sea cucumber Cladolabes schmeltzii. II. Structure and biological action of cladolosides A1–A6. Nat. Prod. Commun. 2014, 9, 1421–1428. [Google Scholar] [PubMed]
- Yibmantasiri, P.; Leahy, D.C.; Busby, B.P.; Angermayr, S.A.; Sorgo, A.G.; Boeger, K.; Heathcott, R.; Barber, J.M.; Moraes, G.; Matthews, J.H.; et al. Molecular basis for fungicidal action of neothyonidioside, a triterpene glycoside from the sea cucumber, Australostichopus mollis. Mol. Biosyst. 2012, 8, 902–912. [Google Scholar] [CrossRef] [PubMed]
- Kitagawa, I.; Nishino, T.; Matsuno, T.; Akutsu, H.; Kyogoku, Y. Structure of holothurin B-a pharmacologically active triterpene-oligoglycoside from the sea cucumber holothuria leucospilota (Brandt). Tetrahedron Lett. 1978, 19, 985–988. [Google Scholar] [CrossRef]
- Kuznetsova, T.A.; Kalinovskaya, N.I.; Kalinovskii, A.I.; Oleinikova, G.K.; Rovnykh, N.V.; Elyakov, G.V. Glycosides of marine invertebrates. XIV. Structure of holothurin B1 from the holothurians Holothuria floridana. Chem. Nat. Compd. 1982, 18, 449–451. [Google Scholar] [CrossRef]
- Silchenko, A.S.; Stonik, V.A.; Avilov, S.A.; Kalinin, V.I.; Kalinovsky, A.I.; Zaharenko, A.M.; Smirnov, A.V.; Mollo, E.; Cimino, G. Holothurins B2, B3, and B4, new triterpene glycosides from mediterranean sea cucumbers of the genus Holothuria. J. Nat. Prod. 2005, 68, 564–567. [Google Scholar] [CrossRef] [PubMed]
- Hua, H.; Yi, Y.-H.; Li, L.; Liu, B.-S.; Pan, M.-X.; Yan, B.; Wang, X.-H. Triterpene glycosides from sea cucumber Holothuria leucospilota. Chin. J. Nat. Med. 2009, 7, 346–350. [Google Scholar] [CrossRef]
- Kobayashi, M.; Hori, M.; Kan, K.; Yasuzawa, T.; Matsui, M.; Shigeki, S.; Kitagawa, I. Marine Natural Products. XXVII. Distribution of lanostane-type triterpene oligoglycosides in ten kinds of okinawan sea cucumbers. Chem. Pharm. Bull. 1991, 39, 2282–2287. [Google Scholar] [CrossRef]
- Wu, J.; Yi, Y.H.; Tang, H.F.; Zou, Z.R.; Wu, H.M. Structure and cytotoxicity of a new lanostane-type triterpene glycoside from the sea cucumber Holothuria hilla. Chem. Biodivers. 2006, 3, 1249–1254. [Google Scholar] [CrossRef] [PubMed]
- Avilov, S.A.; Kalinin, V.I.; Makarieva, T.N.; Stonik, V.A.; Kalinovsky, A.I.; Rashkes, Y.W.; Milgrom, Y.M. Structure of cucumarioside G2, a novel nonholostane glycoside from the sea cucumber Eupentacta fraudatrix. J. Nat. Prod. 1994, 57, 1166–1171. [Google Scholar] [CrossRef] [PubMed]
- Avilov, S.A.; Kalinovsky, A.I.; Kalinin, V.I.; Stonik, V.A.; Riguera, R.; Jiménez, C. Koreoside A, a new nonholostane triterpene glycoside from the sea cucumber Cucumaria koraiensis. J. Nat. Prod. 1997, 60, 808–810. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.-S.; Zhang, H.-W.; Zhang, W.; Yi, Y.H.; Li, L.; Tang, H.; Wang, Z.-L.; Yuan, W.-H. Triterpene Glycosides-Antifungal Compounds of Sea Cucumber Holotoxins D-I and Preparation Method Thereof. Chinese Patent CN101671385 B, 30 May 2012. [Google Scholar]
- Avilov, S.A.; Drozdova, O.A.; Kalinin, V.I.; Kalinovsky, A.I.; Stonik, V.A.; Gudimova, E.N.; Riguera, R.; Jimenez, C. Frondoside C, a new nonholostane triterpene glycoside from the sea cucumber Cucumaria frondosa: Structure and cytotoxicity of its desulfated derivative. Can. J. Chem. 1998, 76, 137–141. [Google Scholar] [CrossRef]
- Kalinin, V.I.; Kalinovskii, A.I.; Stonik, V.A.; Dmitrenok, P.S.; El’kin, Y.N. Structure of psolusoside B-a nonholostane triterpene glycoside of the holothurian genus Psolus. Chem. Nat. Compd. 1989, 25, 311–317. [Google Scholar] [CrossRef]
- Avilov, S.A.; Kalinovskii, A.I.; Stonik, V.A. Two new triterpene glycosides from the holothurian Duasmodactyla kurilensis. Chem. Nat. Compd. 1991, 27, 188–192. [Google Scholar] [CrossRef]
- Silchenkoa, A.S.; Kalinovsky, A.I.; Avilov, S.A.; Dmitrenok, P.S.; Kalinin, V.I.; Berdyshev, D.V.; Chingizova, E.A.; Andryjaschenko, P.V.; Minin, K.V.; Stonika, V. Fallaxosides B1 and D3, triterpene glycosides with novel skeleton types of aglycones from the sea cucumber Cucumaria fallax. Tetrahedron 2017, 73, 2335–2341. [Google Scholar] [CrossRef]
- Silchenko, A.S.; Kalinovskya, A.I.; Avilov, S.A.; Andryjaschenko, P.V.; Dmitrenok, P.S.; Kalinin, V.I.; Martyyas, E.A.; Minin, K.V. Fallaxosides C1, C2, D1 and D2, unusual oligosulfated triterpene glycosides from the sea cucumber Cucumaria fallax (Cucumariidae, Dendrochirotida, Holothurioidea) and taxonomic status of this animal. Nat. Prod. Commun. 2016, 11, 939–945. [Google Scholar]
- Silchenko, A.S.; Kalinovskya, A.I.; Avilov, S.A.; Andryjaschenko, P.V.; Dmitrenok, P.S.; Kalinin, V.I.; Chingizova, E.A.; Minin, K.V.; Stonik, V.A. Structures and biogenesis of fallaxosides D4, D5, D6 and D7, trisulfated non-holostane triterpene glycosides from the sea cucumber Cucumaria fallax. Molecules 2016, 21, 939. [Google Scholar] [CrossRef] [PubMed]
- Silchenko, A.S.; Kalinovsky, A.I.; Avilov, S.A.; Kalinin, V.I.; Andrijaschenko, P.V.; Dmitrenok, P.S.; Chingizova, E.A.; Ermakova, S.P.; Malyarenko, O.S.; Dautova, T.N. Nine new triterpene glycosides, magnumosides A1–A4, B1, B2, C1, C2 and C4, from the Vietnamese sea cucumber Neothyonidium (=Massinium) magnum: Structures and activities against tumor cells independently and in synergy with radioactive irradiation. Mar. Drugs 2017, 16, 256. [Google Scholar] [CrossRef] [PubMed]
- Silchenko, A.S.; Kalinovsky, A.I.; Avilov, S.A.; Andryjaschenko, P.V.; Dmitrenok, P.S.; Yurchenko, E.A.; Dolmatov, I.Y.; Dautov, S.S.; Stonik, V.A.; Kalinin, V.I. Colochiroside E, an unusual non-holostane triterpene sulfated trioside from the sea cucumber Colochirus robustus and evidence of the impossibility of a 7(8)-double bond migration in lanostane derivatives having an 18(16)-lactone. Nat. Prod. Commun. 2016, 11, 741–746. [Google Scholar] [PubMed]
- Kalinin, V.I.; Prkofieva, N.G.; Likhatskaya, G.N. Hemolytic activities of triterpene glycosides from the holothurian order Dendrochirotida: Some trends in the evolution of this group of toxins. Toxicon 1996, 34, 475–483. [Google Scholar] [CrossRef]
- Kumar, R.; Chaturvedi, A.K.; Shukla, P.K.; Lakshmi, V. Antifungal activity in triterpene glycosides from the sea cucumber Actinopyga lecanora. Bioorg. Med. Chem. Lett. 2007, 17, 4387–4391. [Google Scholar] [CrossRef] [PubMed]
- Hua, H.; Yang-hua, Y.; Li, L.; Liu, B.S.; La, M.P.; Zhang, H.W. Antifungal active triterpene glycosides from sea cucumber Holothuria scabra. Acta Pharm. Sin. 2009, 44, 620–624. [Google Scholar]
- Aminin, D.L.; Pislyagin, E.A.; Menchinskaya, E.S.; Silchenko, A.S.; Avilov, S.A.; Kalinin, V.I. Immunomodulatory and anticancer activity of sea cucumber triterpene glycosides. In Studies in Natural Products Chemistry; Atta-ur-Rahman, Ed.; Elsevier Science: Amsterdam, The Netherlands, 2013; Volume 41, pp. 75–94. [Google Scholar]
- Aminin, D.L.; Agafonova, I.G.; Kalinin, V.I.; Silchenko, A.S.; Avilov, S.A.; Stonik, V.A.; Collin, P.D.; Woodward, C. Immunomodulatory properties of frondoside A, a major triterpene glycoside from the North Atlantic commercially harvested sea cucumber Cucumaria frondosa. J. Med. Food 2008, 11, 443–453. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Han, H.; Chen, X.; Yi, Y.; Sun, H. Cytotoxic and apoptosis-inducing activity of triterpene glycosides from Holothuria scabra and Cucumaria frondosa against HepG2 cells. Mar. Drugs 2014, 24, 4274–4290. [Google Scholar] [CrossRef] [PubMed]
- Popov, A.; Kalinovskaia, N.; Kuznetsova, T.; Agafonova, I.; Anisimov, M. Role of sterols in the membranotropic activity of triterpene glycosides. Antibiotiki 1983, 28, 656–659. [Google Scholar] [PubMed]
- Segal, R.; Schlosser, E. Role of glycosides in the membranlytic, antifungal action of saponins. Arch. Microbiol. 1975, 104, 147–150. [Google Scholar] [CrossRef] [PubMed]
- Aminin, D.L.; Menchinskaya, E.S.; Pislyagin, E.A.; Silchenko, A.S.; Avilov, S.A.; Kalinin, V.I. Sea cucumber triterpene glycosides as anticancer agents. In Studies in Natural Products Chemistry, 1st ed.; Atta-ur-Rahman, Ed.; Elsevier Science: Amsterdam, The Netherlands, 2016; Volume 49, pp. 55–105. ISBN 978-0-444-63601-0. [Google Scholar]
- Kalinin, V.I.; Aminin, D.L.; Avilov, S.A.; Silchenko, A.S.; Stonik, V.A. Triterpene glycosides from sea cucumbers (Holothurioidae, Echinodermata), biological activities and functions. In Studies in Natural Product Chemistry (Bioactive Natural Products); Atta-ur-Rahman, Ed.; Elsevier Science: Amsterdam, The Netherlands, 2008; Volume 35, pp. 135–196. [Google Scholar]

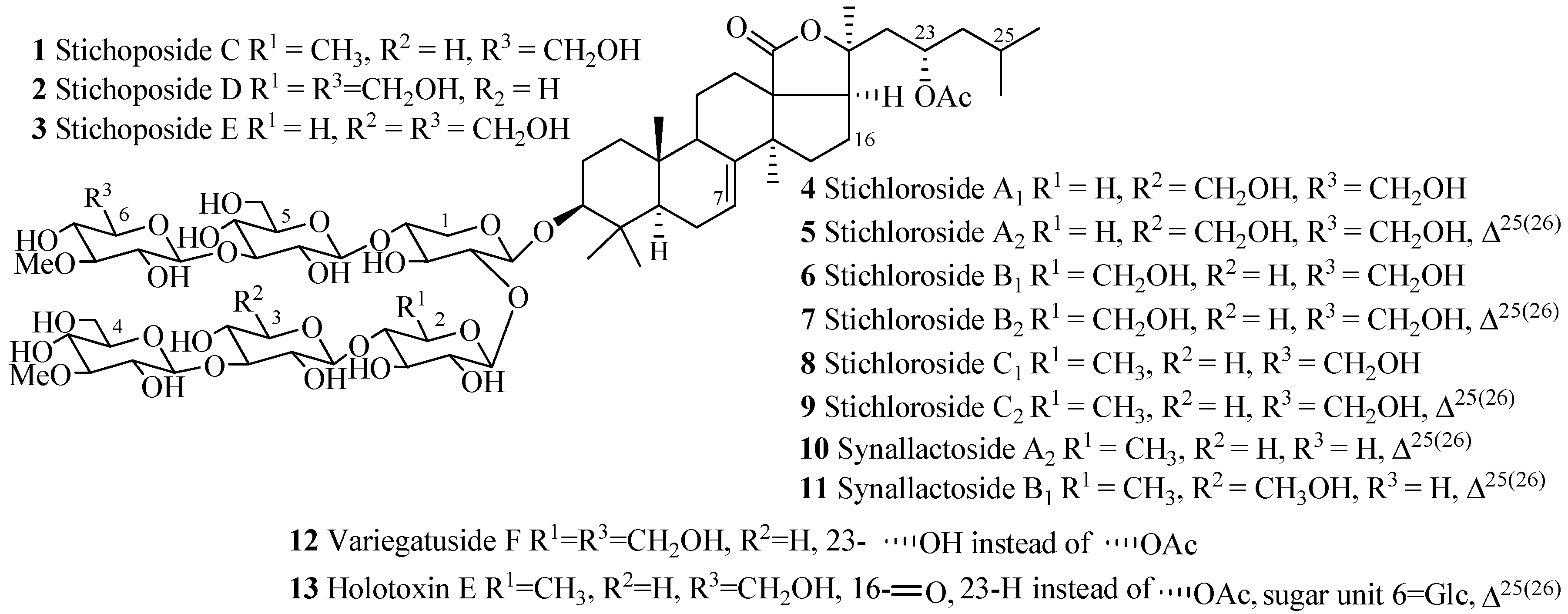

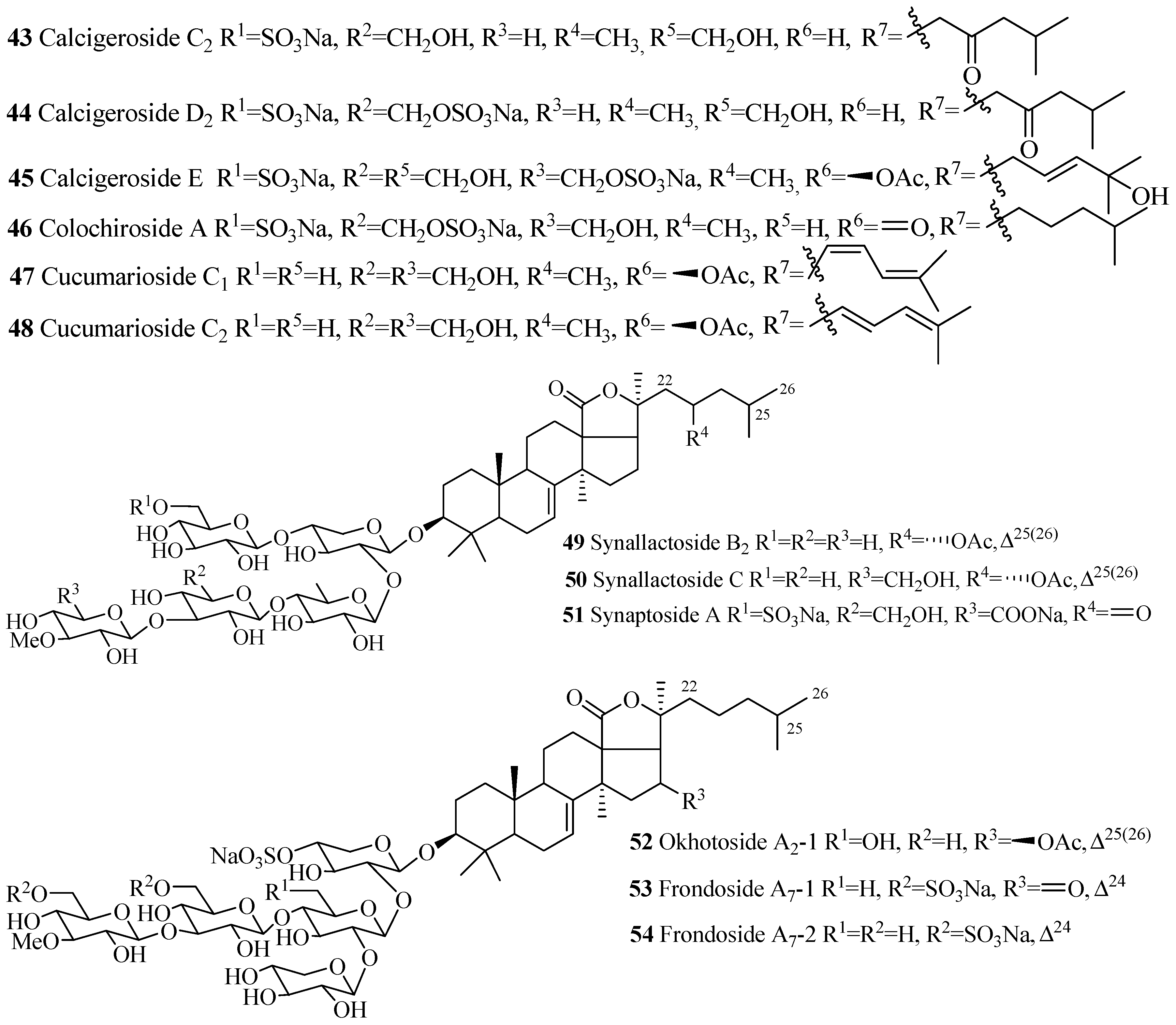
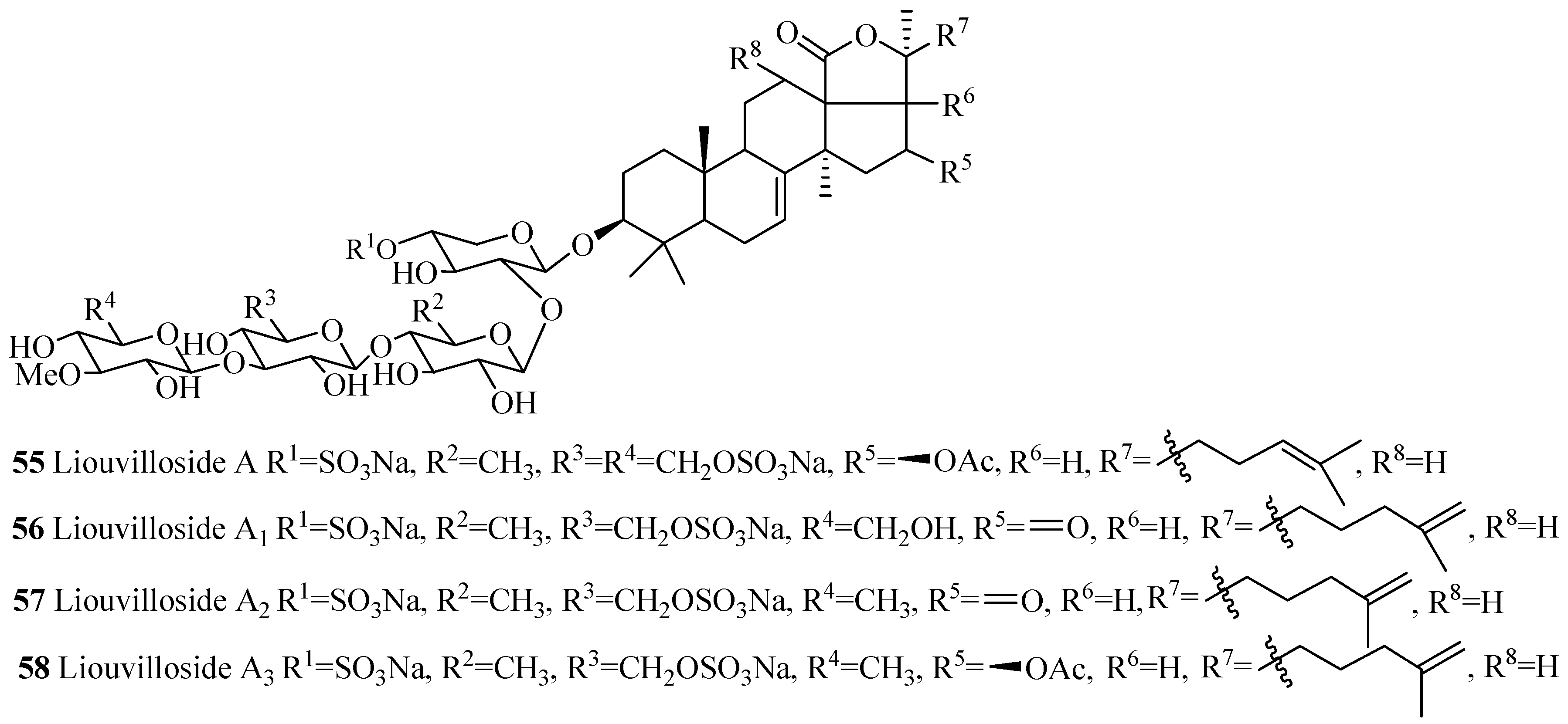

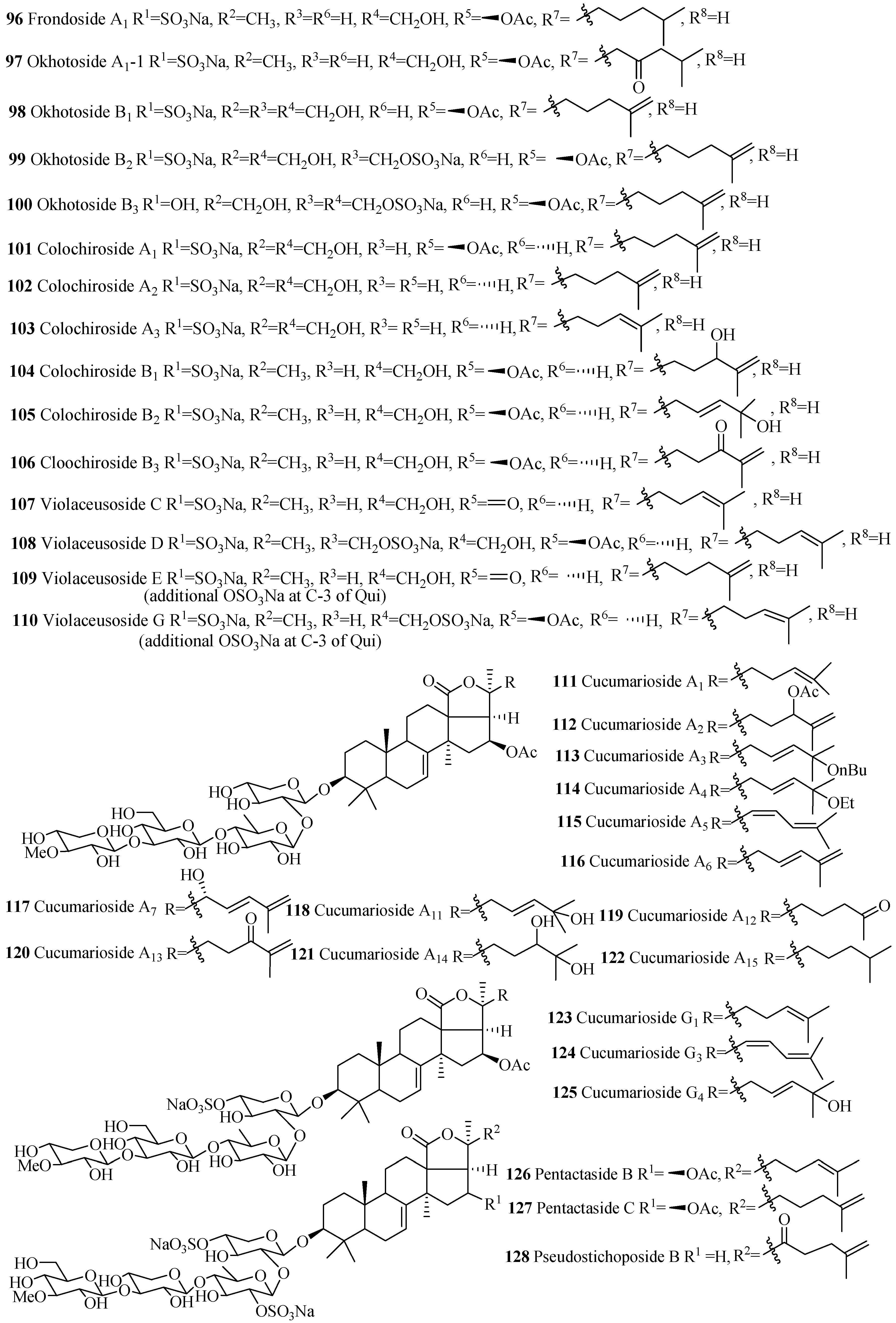

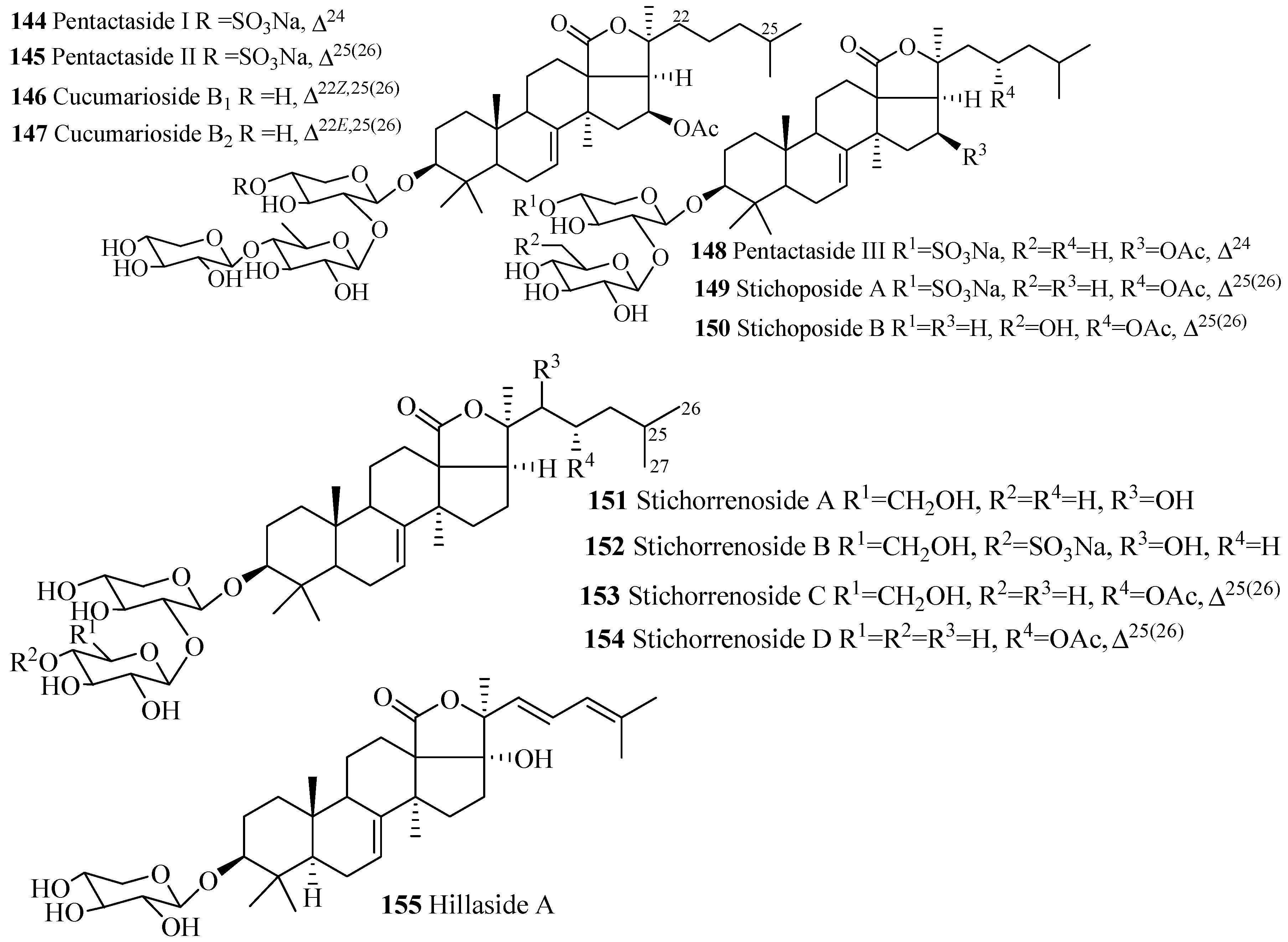
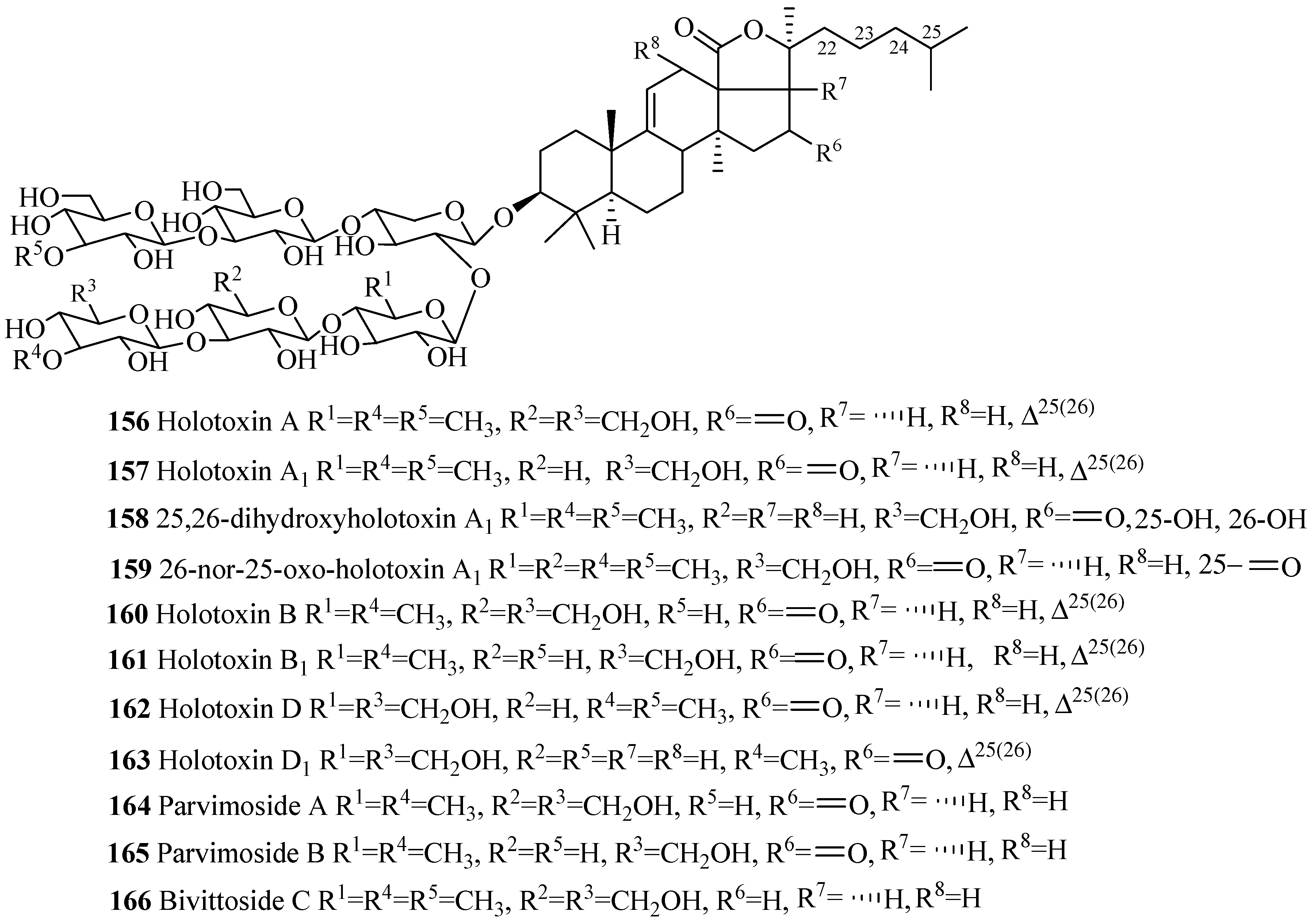
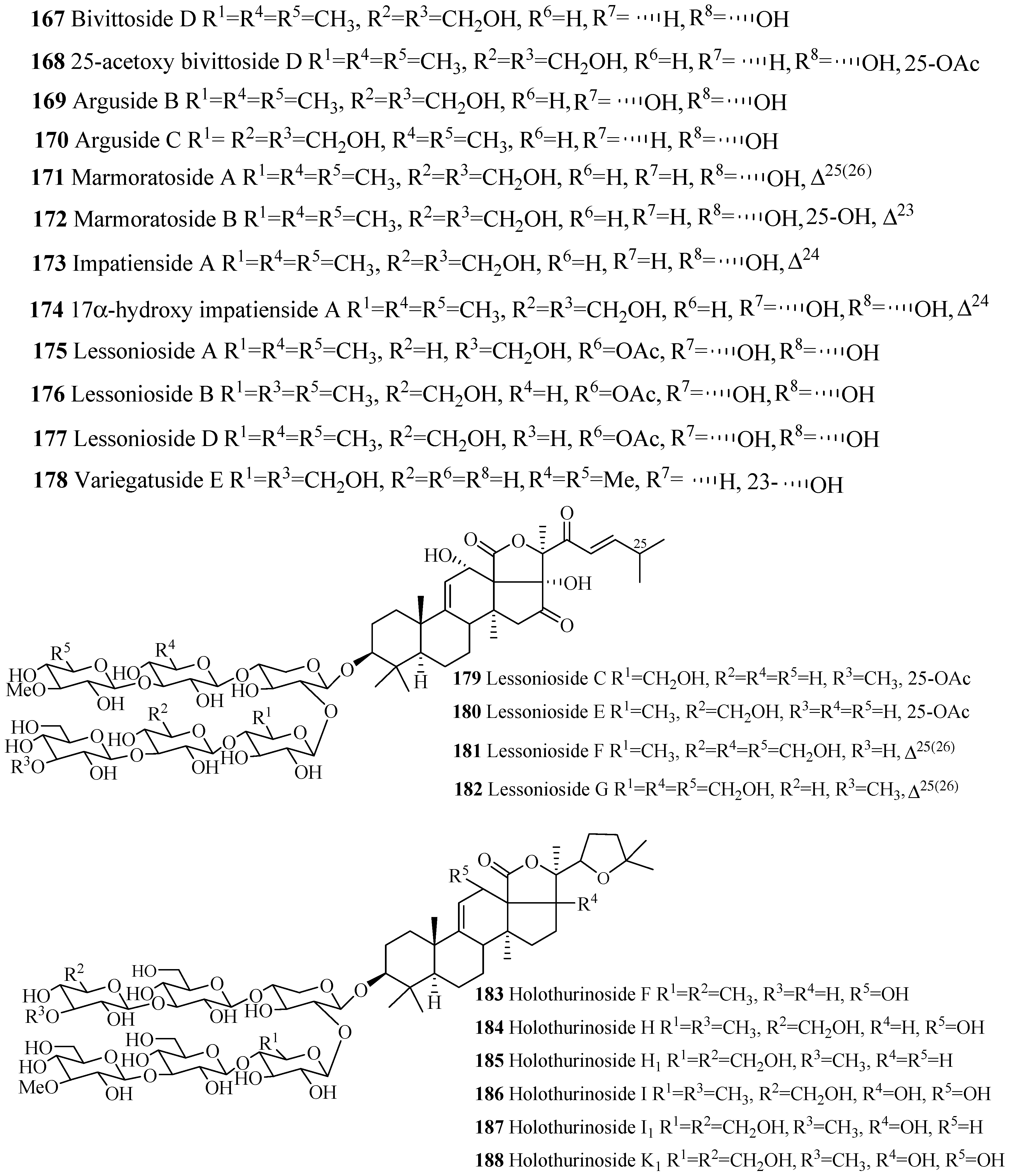
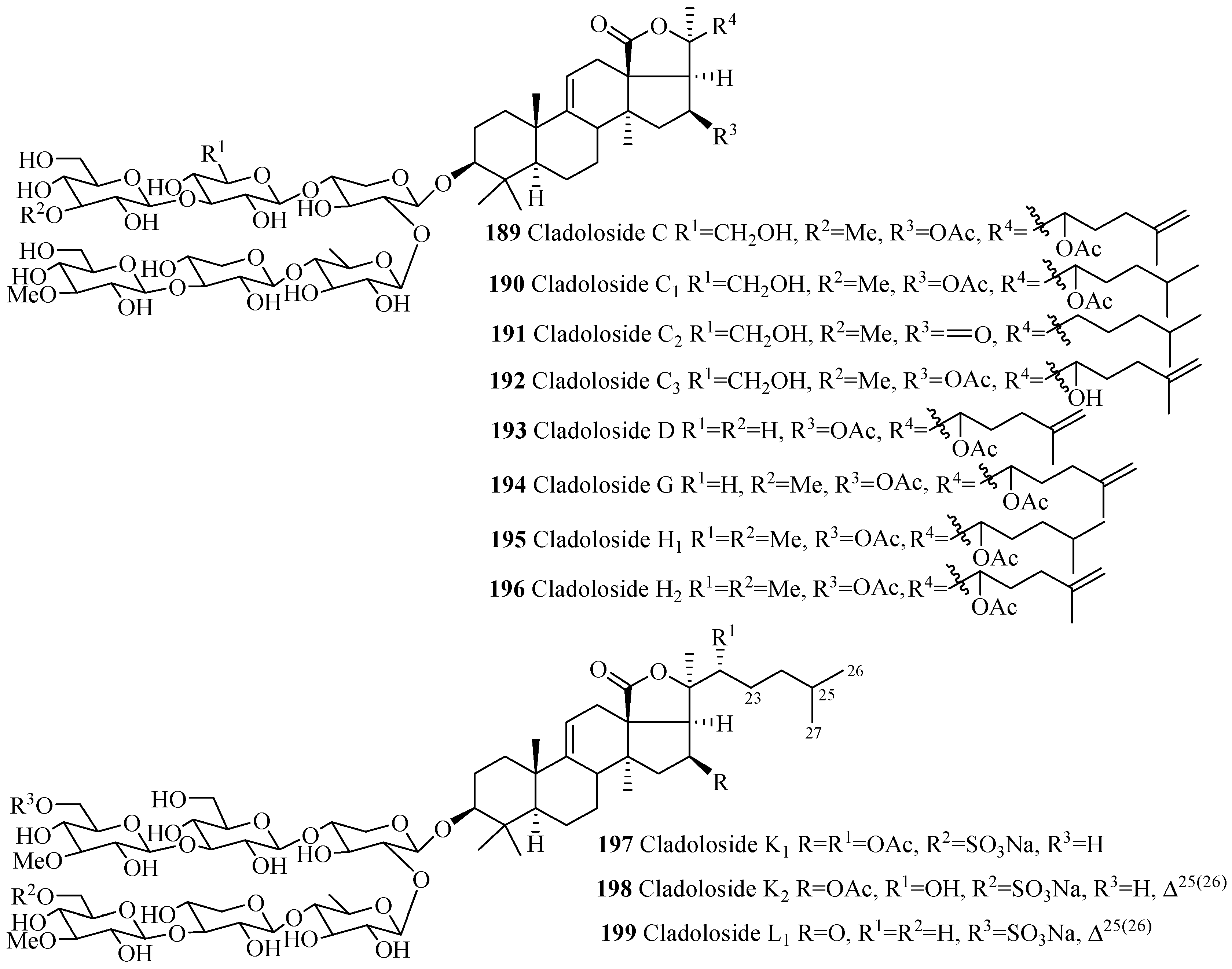
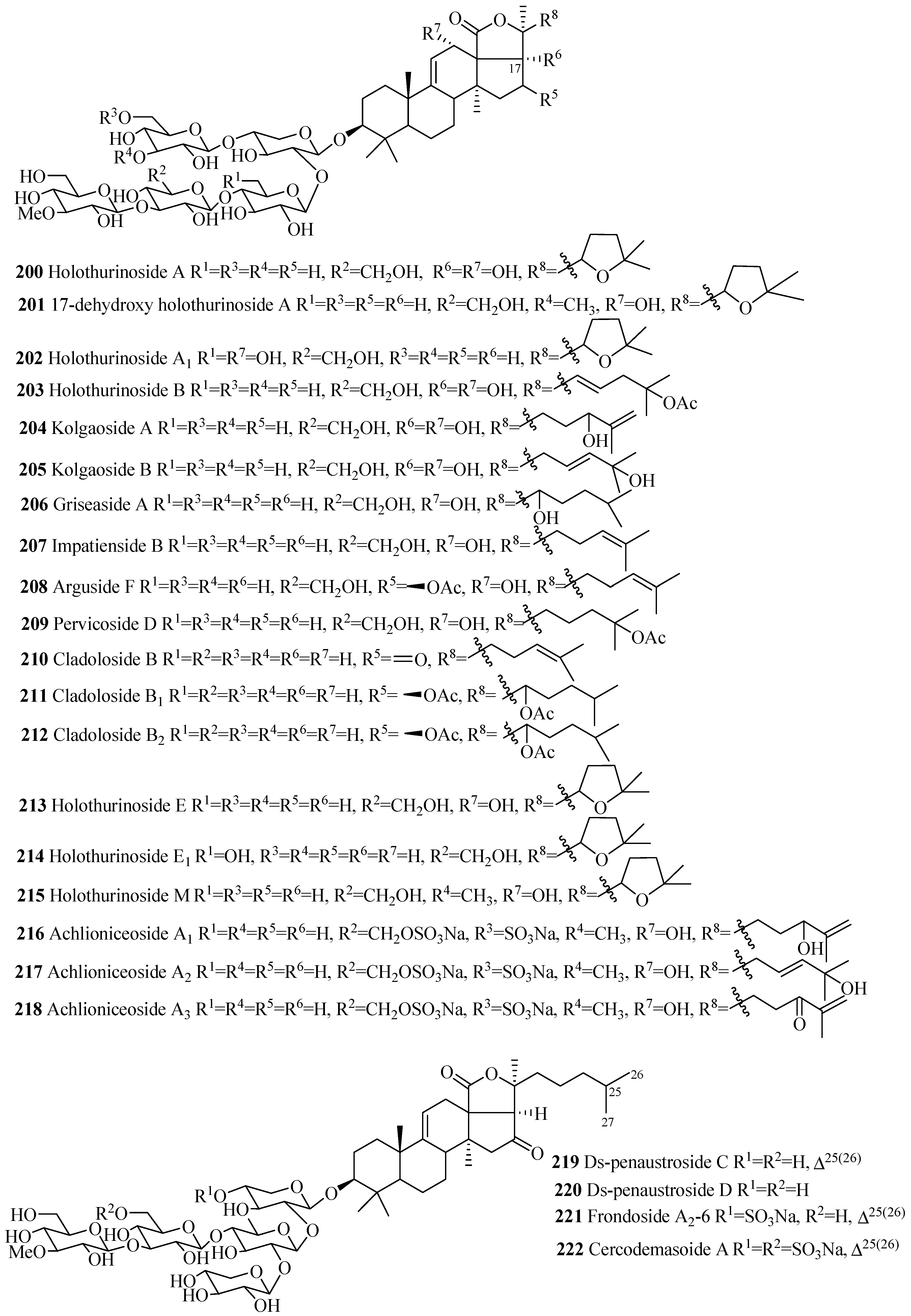

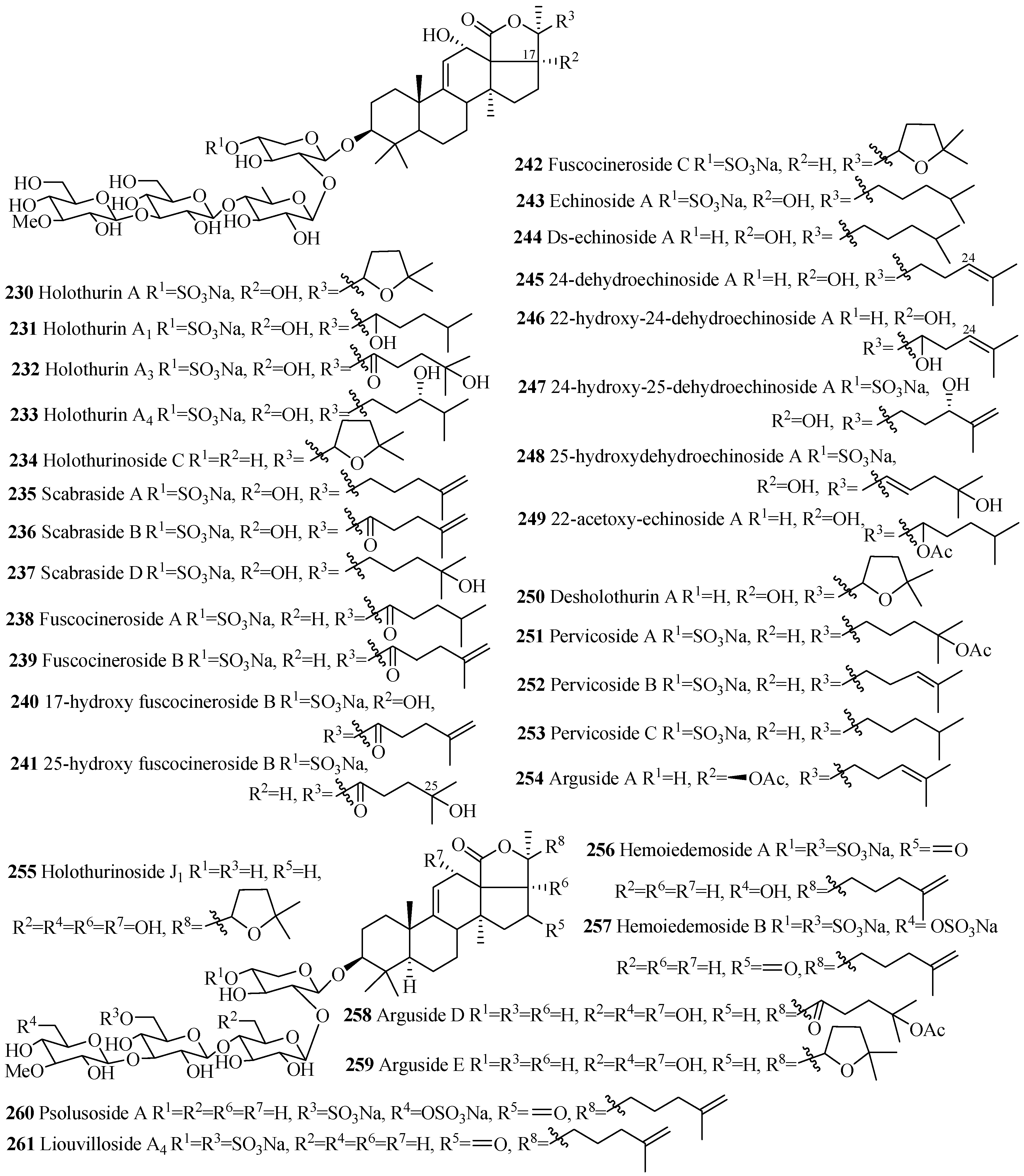
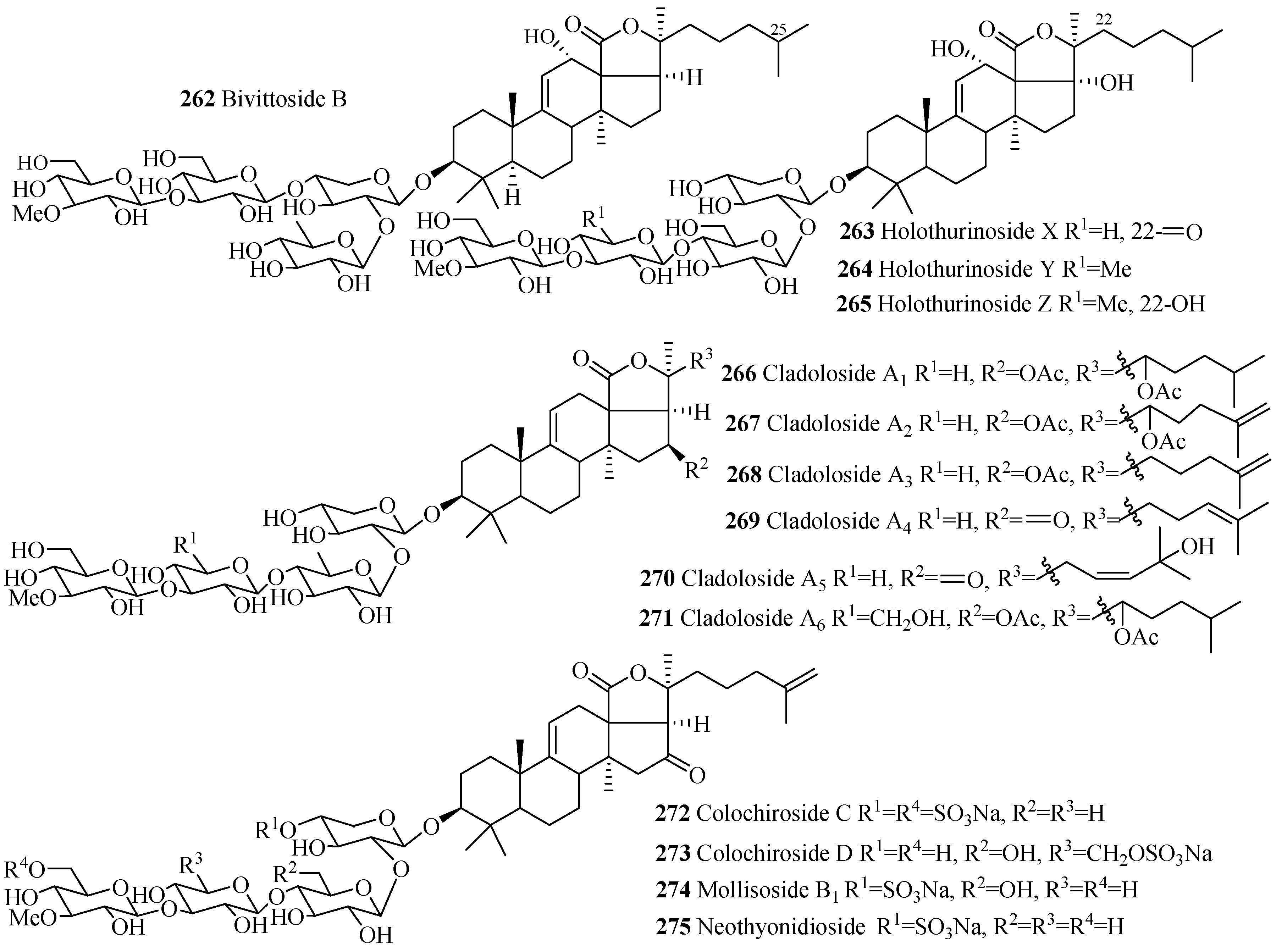
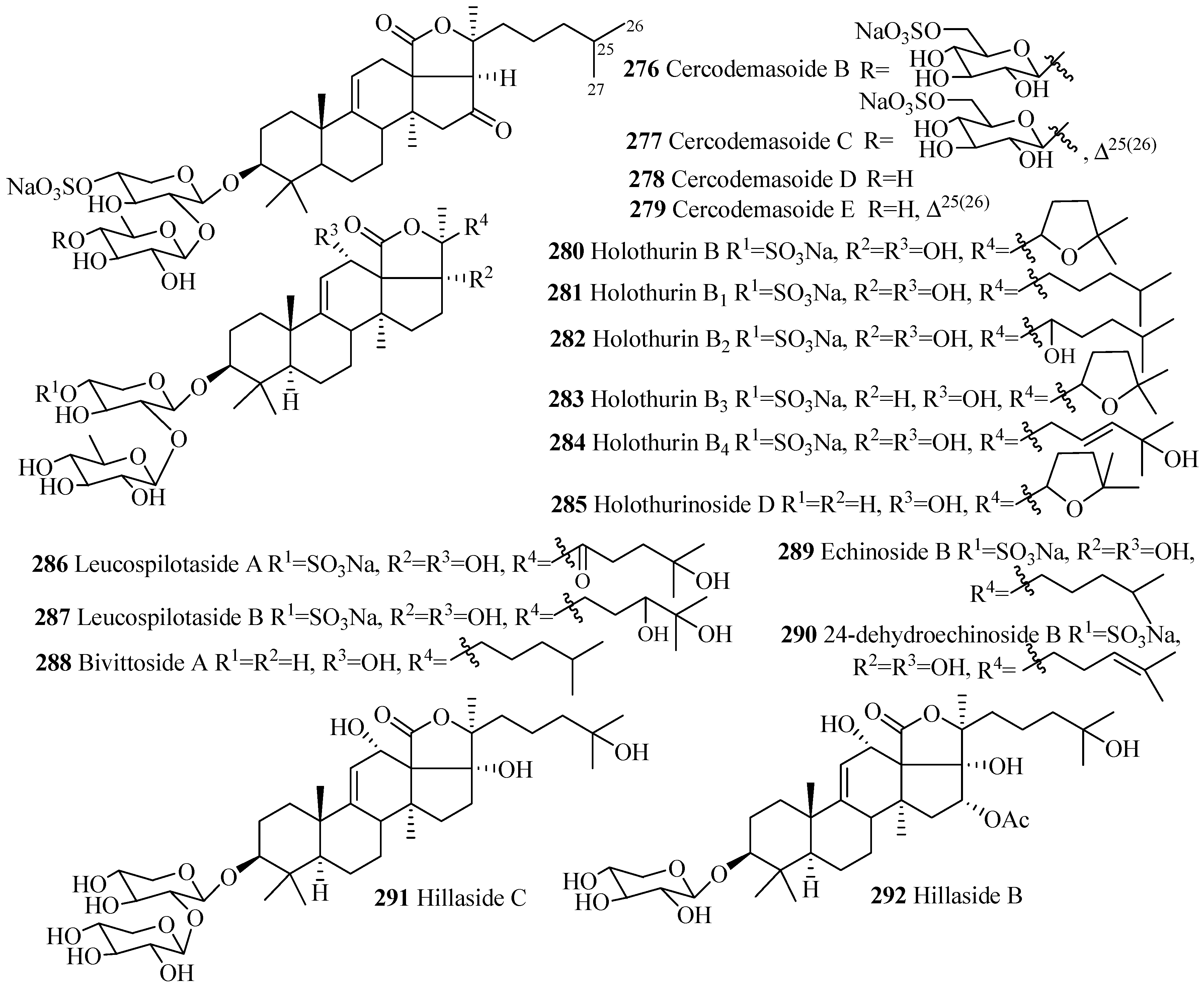
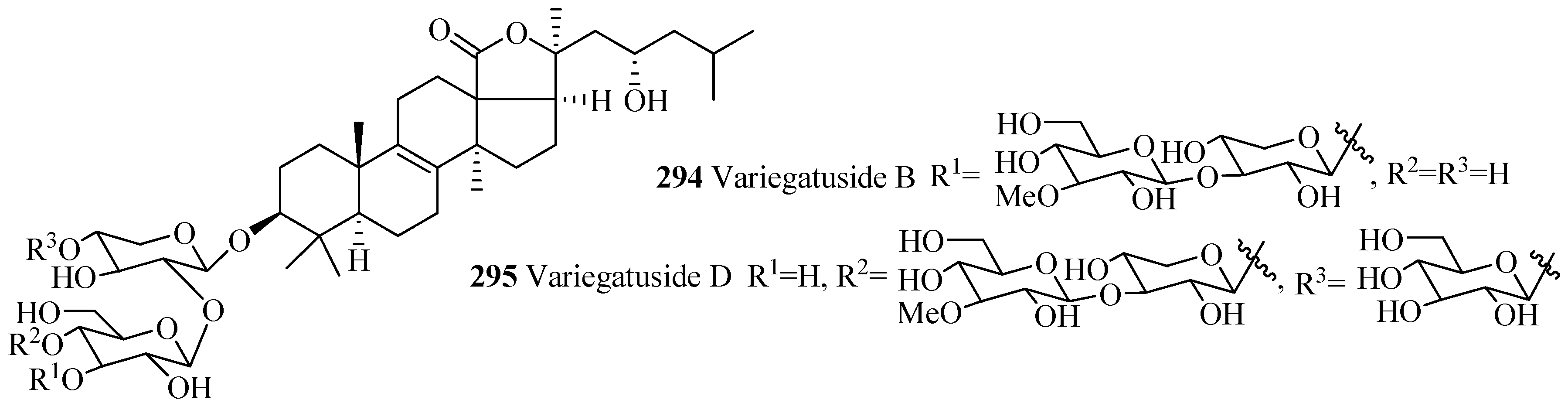
| Compound Name | Producing Species | Reference | Compound Name | Producing Species | Reference |
|---|---|---|---|---|---|
| Stichoposide C (1) | Thelenota anax | [17] | Stichoposide D (2) | Thelenota anax | [18] |
| Stichoposide E (3) | Stichopus chloronotus | [19] | Stichloroside A1 (4) | S. chloronotus | [20] |
| Stichloroside A2 (5) | S. chloronotus | [20] | Stichloroside B1 (6) | S. chloronotus | [20] |
| Stichloroside B2 (7) | S. chloronotus | [20] | Stichloroside C1 (8) | S. chloronotus | [20] |
| Stichloroside C2 (9) | S. chloronotus | [20] | Synallactoside A2 (10) | Synallactes nozawai | [16] |
| Synallactoside B1 (11) | S. nozawai | [16] | Variegatuside F (12) | S. variegates | [21] |
| Holotoxin E (13) | S. japonicus | [22] |
| Compound Name | Producing Species | Reference | Compound Name | Producing Species | Reference |
|---|---|---|---|---|---|
| Cucumarioside A0-1 (14) | Cucumaria japonica | [23] | Cucumarioside A0-2 (15) | C. japonica | [23] |
| Cucumarioside A0-3 (16) | C. japonica | [23] | Cucumarioside A1-2 (17) | C. japonica | [24] |
| Cucumarioside A2-2 (18) | C. japonica | [25] | Cucumarioside A2-3 (19) | C. japonica | [24] |
| Cucumarioside A2-4 (20) | C. japonica | [24] | Cucumarioside A2-5 (21) | C. conicospermium | [26] |
| Cucumarioside A4-2 (22) | C. japonica | [24] | Cucumarioside A6-2 (23) | C. japonica | [27] |
| Cucumarioside A7-1 (24) | C. japonica | [28] | Cucumarioside A7-2 (25) | C. japonica | [28] |
| Cucumarioside A7-3 (26) | C. japonica | [28] | Cucumarioside H (27) | E. fraudatrix | [29] |
| Cucumarioside H2 (28) | E. fraudatrix | [30] | Cucumarioside H4 (29) | E. fraudatrix | [30] |
| Cucumarioside H5 (30) | E. fraudatrix | [29] | Cucumarioside H6 (31) | E. fraudatrix | [29] |
| Cucumarioside H7 (32) | E. fraudatrix | [29] | Cucumarioside H8 (33) | E. fraudatrix | [29] |
| Cucumarioside I1 (34) | E. fraudatrix | [31] | Cucumarioside I2 (35) | E. fraudatrix | [32] |
| Cucumarioside I3 (36) | E. fraudatrix | [31] | Frondoside A (37) | C. frondosa | [33] |
| Frondoside B (38) | C. frondosa | [34] | Frondoside A2-1 (39) | C. frondosa | [35] |
| Frondoside A2-2 (40) | C. frondosa | [35] | Frondoside A2-3 (41) | C. frondosa | [35] |
| Frondoside A2-4 (42) | C. frondosa | [36] | Calcigeroside C2 (43) | P. calcigera | [37] |
| Calcigeroside D2 (44) | P. calcigera | [38] | Calcigeroside E (45) | P. calcigera | [38] |
| Colochiroside A (46) | C. anceps | [39] | Cucumarioside C1(47) | E. fraudatrix | [40] |
| Cucumarioside C2 (48) | E. fraudatrix | [40] | Synallactoside B2 (49) | S. nozawai | [16] |
| Synallactoside C (50) | S. nozawai | [16] | Synaptoside A (51) | Synapta maculata | [41] |
| Okhotoside A2-1 (52) | C. okhotensis | [42] | Frondoside A7-1 (53) | C. frondosa | [43] |
| Frondoside A7-2 (54) | C. frondosa | [43] |
| Compound Name | Producing Species | Reference | Compound Name | Producing Species | Reference |
|---|---|---|---|---|---|
| Liouvilloside A (55) | Staurocucumis liouvillei | [44] | Liouvilloside A1 (56) | S. liouviellei | [45] |
| Liouvilloside A2 (57) | S. liouvillei | [45] | Liouvilloside A3 (58) | S. liouvillei | [45] |
| Liouvilloside A5 (59) | S. liouvillei | [46] | Liouvilloside B (60) | S. liouvillei | [44] |
| Liouvilloside B1 (61) | S. liouvillei | [45] | Liouvilloside B2 (62) | S. liouvillei | [45] |
| Violaceuside A (63) | P. violeceus | [47] | Violaceuside B (64) | P. violeceus | [47] |
| Violaceuside I (65) | P. violeceus | [48] | Violaceuside II (66) | P. violeceus | [48] |
| Violaceuside III (67) | P. violeceus | [48] | Intercedenside A (68) | M. intercedens | [49] |
| Intercedenside B (67) | Mensamria intercedens | [49] | Intercedenside C (70) | M. intercedens | [49] |
| Intercedenside D (71) | M. intercedens | [50] | Intercedenside E (72) | M. intercedens | [50] |
| Intercedenside F (73) | M. intercedens | [50] | Intercedenside G (74) | M. intercedens | [50] |
| Intercedenside H (75) | M. intercedens | [50] | Intercedenside I (76) | M. intercedens | [50] |
| Patagonicoside A (77) | Psolus patagonicus | [51] | Patagonicoside B (78) | P. patagonicus | [52] |
| Patagonicoside C (79) | P. patagonicus | [52] | Philinopside A (80) | P. quadrangularis | [53] |
| Philinopside B (81) | Pentacta quadrangularis | [53] | Philinopside E (82) | P. quadrangularis | [54] |
| Philinopside F (83) | P. quadrangularis | [54] | Mollisoside A (84) | A. mollis | [55] |
| Mollisoside B2 (85) | Australostichopus mollis | [55] | Eximisoside A (86) | P. eximius | [56] |
| Pseudostichoposide A (87) | Pseudostichopus trachus | [57] | Cucumarioside F1 (88) | E. fraudatrix | [58] |
| Cucumarioside F2 (89) | E. fraudatrix | [58] | Pseudocnoside A (90) | P. leoninus | [59] |
| Typicoside A1 (91) | Actinocucumis typica | [60] | Typicoside A2 (92) | A. typica | [60] |
| Typicoside B1 (93) | A. typica | [60] | Typicoside C1 (94) | A. typica | [60] |
| Typicoside C2 (95) | A. typica | [60] | Frondoside A1 (96) | C. okhotensis | [61] |
| Okhotoside A1-1 (97) | Cucumaria okhotensis | [61] | Okhotoside B1 (98) | C. okhotensis | [62] |
| Okhotoside B2 (99) | C. okhotensis | [62] | Okhotoside B3 (100) | C. okhotensis | [62] |
| Colochiroside A1 (101) | Colochirus robustus | [63] | Colochiroside A2 (102) | C. robustus | [63] |
| Colochiroside A3 (103) | C. robustus | [63] | Colochiroside B1 (104) | C. robustus | [64] |
| Colochiroside B2 (105) | C. robustus | [64] | Colochiroside B3 (106) | C. robustus | [64] |
| Violaceusosides C (107) | P. violaceus | [65] | Violaceusosides D (108) | P. violaceus | [65] |
| Violaceusosides E (109) | P. violaceus | [65] | Violaceusosides G (110) | P. violaceus | [65] |
| Cucumarioside A1 (111) | E. fraudatrix | [66] | Cucumarioside A2 (112) | E. fraudatrix | [67] |
| Cucumarioside A3 (113) | E. fraudatrix | [66] | Cucumarioside A4 (114) | E. fraudatrix | [66] |
| Cucumarioside A5 (115) | E. fraudatrix | [66] | Cucumarioside A6 (116) | E. fraudatrix | [66] |
| Cucumarioside A7 (117) | E. fraudatrix | [67] | Cucumarioside A11 (118) | E. fraudatrix | [67] |
| Cucumarioside A12 (119) | E. fraudatrix | [66] | Cucumarioside A13 (120) | E. fraudatrix | [67] |
| Cucumarioside A14 (121) | E. fraudatrix | [67] | Cucumarioside A15 (122) | E. fraudatrix | [66] |
| Cucumarioside G1 (123) | C. fraudatrix | [68] | Cucumarioside G3 (124) | E. fraudatrix | [69] |
| Cucumarioside G4 (125) | E. fraudatrix | [70] | Pentactaside B (126) | P. quadrangularis | [71] |
| Pentactaside C (127) | P. quadrangularis | [71] | Pseudostichoposide B (128) | P. trachus | [72] |
| Variegatuside A (129) | S. variegates | [73] | Variegatuside C (130) | S. variegates | [21] |
| Synallactoside A1(131) | S. nozawai | [16] | Thelenotoside A (132) | Thelenota ananas | [74] |
| Thelenotoside B (133) | T. ananas | [74] | Cucumechinoside A (134) | C. echinata | [75] |
| Cucumechinoside B (135) | C. echinata | [75] | Cucumechinoside C (136) | C. echinata | [75] |
| Cucumechinoside D (137) | C. echinata | [75] | Cucumechinoside E (138) | C. echinata | [75] |
| Cucumechinoside F (139) | C. echinata | [75] | Lefevreoside A1 (140) | C. lefevrei | [76] |
| Lefevreoside A2 (141) | C. lefevrei | [76] | Lefevreoside C (142) | C. lefevrei | [76] |
| Lefevreoside D (143) | C. lefevrei | [76] |
| Compound Name | Producing Species | Reference | Compound Name | Producing Species | Reference |
|---|---|---|---|---|---|
| Pentactaside I (144) | Pentacta quadrangularis | [77] | Pentactaside II (145) | P. quadrangularis | [77] |
| Cucumarioside B1 (146) | E. fraudatrix | [78] | Cucumarioside B2 (147) | E. fraudatrix | [78] |
| Pentactaside III (148) | P. quadrangularis | [77] | Stichoposide A (149) | S. cloronotus | [79] |
| Stichoposide B (150) | Stichopus cloronotus | [79] | Stichorrenoside A (151) | Stichopus horrens | [80] |
| Stichorrenoside B (152) | S. horrens | [80] | Stichorrenoside C (153) | S. horrens | [80] |
| Stichorrenoside D (154) | S. horrens | [80] | Hillaside A (155) | H. hilla | [81] |
| Compound Name | Producing Species | Reference | Compound Name | Pro. Species | Reference |
|---|---|---|---|---|---|
| Holotoxin A (156) | Stichopus japonicus | [82] | Holotoxin A1 (157) | S. japonicus | [22] |
| 25,26-dihydroxyholotoxin A1 (158) | Apostichopus japonicus | [83] | Oxo-holotoxin A1 (159) | A. japonicus | [22] |
| Holotoxin B (160) | S. japonicus | [22] | Holotoxin B1 (161) | S. japonicus | [22] |
| Holotoxin D (162) | S. japonicus | [22] | Holotoxin D1 (163) | A. japonicus | [83] |
| Parvimoside A (164) | Stichopus parvimensis | [84] | Parvimoside B (165) | S. parvimensis | [84] |
| Bivittoside C (166) | Bohadschia bivittata | [85] | Bivittoside D (167) | B. bivittata | [85] |
| 25-acetoxybivittoside D (168) | Bohadschia marmorata | [86] | Arguside B (169) | B. argus | [87] |
| Arguside C (170) | B. argus | [87] | Marmoratoside A (171) | B. marmorata | [86] |
| Marmoratoside B (172) | B. marmorata | [86] | Impatienside A (173) | H. impatiens | [86] |
| 17α-hydroxyimpatienside A (174) | B. marmorata | [86] | Lessonioside A (175) | H. lessoni | [88] |
| Lessonioside B (176) | Holothuria lessoni | [88] | Lessonioside D (177) | H. lessoni | [88] |
| Variegatuside E (178) | S. variegates | [21] | Lessonioside C (179) | H. lessoni | [88] |
| Lessonioside E (180) | Holothuria lessoni | [88] | Lessonioside F (181) | H. lessoni | [88] |
| Lessonioside G (182) | H. lessoni | [88] | Holothurinoside F (183) | B. subrubra | [89] |
| Holothurinoside H (184) | B. subrubra | [89] | Holothurinoside H1 (185) | B. subrubra | [89] |
| Holothurinoside I (186) | B. subrubra | [89] | Holothurinoside I1 (187) | B. subrubra | [89] |
| Holothurinoside K1 (188) | B. subrubra | [89] | Cladoloside C (189) | C. schmeltzii | [90] |
| Cladoloside C1 (190) | Cladolabes schmeltzii | [90] | Cladoloside C2 (191) | C. schmeltzii | [90] |
| Cladoloside C3 (192) | C. schmeltzii | [91] | Cladoloside D (193) | C. schmeltzii | [90] |
| Cladoloside G (194) | C. schmeltzii | [91] | Cladoloside H1 (195) | C. schmeltzii | [91] |
| Cladoloside H2 (196) | C. schmeltzii | [91] | Cladoloside K1 (197) | C. schmeltzii | [92] |
| Cladoloside K2 (198) | C. schmeltzii | [92] | Cladoloside L1 (199) | C. schmeltzii | [92] |
| Compound Name | Producing Species | Reference | Compound Name | Producing Species | Reference |
|---|---|---|---|---|---|
| Holothurinoside A (200) | Holothuria forskalii | [93] | 17-dehydroxyholothurinoside A (201) | Holothuria grisea | [94] |
| Holothurinoside A1 (202) | H.lessoni | [95] | Holothurinoside B (203) | H. forskalii | [93] |
| Kolgaoside A (204) | Kolga hyalina | [96] | Kolgaoside B (205) | K. hyalina | [96] |
| Griseaside A (206) | H. grisea | [94] | Impatienside B (207) | H. axiloga | [97] |
| Arguside F (208) | Holothuria axiloga | [97] | Pervicoside D (209) | H. axiloga | [97] |
| Cladoloside B (210) | A. japonicus | [22] | Cladoloside B1 (211) | C. schmeltzii | [90] |
| Cladoloside B2 (212) | C. schmeltzii | [90] | Holothurinoside E (213) | H. lessoni | [95] |
| Holothurinoside E1 (214) | H. lessoni | [95] | Holothurinoside M (215) | H. lessoni | [95] |
| Achlioniceoside A1 (216) | A. violaecuspidata | [98] | Achlioniceoside A2 (217) | A. violaecuspidata | [98] |
| Achlioniceoside A3 (218) | A. violaecuspidata | [98] | Ds-penaustroside C (219) | P. australis | [99] |
| Ds-penaustroside D (220) | Pentacta australis | [99] | Frondoside A2-6 (221) | C. frondosa | [35] |
| Cladoloside E1 (222) | C. schmeltzii | [91] | Cladoloside E2 (223) | C. schmeltzii | [91] |
| Cladoloside F1 (224) | C. schmeltzii | [91] | Cladoloside F2 (225) | C. schmeltzii | [91] |
| Cercodemasoide A (226) | C. anceps | [100] | Cladoloside I1 (227) | C. schmeltzii | [92] |
| Cladoloside I2 (228) | C. schmeltzii | [92] | Cladoloside J1 (229) | C. schmeltzii | [92] |
| Compound Name | Producing Species | Reference | Compound Name | Producing Species | Reference |
|---|---|---|---|---|---|
| Holothurin A (230) | Actinopyga agassizi | [101] | Holothurin A1 (231) | H. grisea | [102] |
| Holothurin A3 (232) | Holothuria scabra | [103] | Holothurin A4 (233) | H. scabra | [103] |
| Holothurinoside C (234) | H. forskalii | [93] | Scabraside A (235) | H. scabra | [104] |
| Scabraside B (236) | [104] | 104] | Scabraside D (237) | H. scabra | [105] |
| Fuscocineroside A (238) | H. fuscocinerea | [106] | Fuscocineroside B (239) | H. fuscocinerea | [106] |
| 17-hydroxy fuscocineroside B (240) | B. marmorata | [107] | 25-hydroxy-fuscocineroside B (241) | B. marmorata | [107] |
| Fuscocineroside C (242) | H. fuscocinerea | [106] | Echinoside A (243) | A. echinites | [108] |
| Ds-echinoside A (244) | P. graeffei | [109] | 24-dehydroechinoside A (245) | H. scabra | [105] |
| 22-hydroxy-24-dehydroechinoside A (246) | Actinopyga flammea | [110] | 24-hydroxy-25-dehydroechinoside A (247) | A. flammea | [110] |
| 25-hydroxydehydroechinoside A (248) | A. flammea | [110] | 22-acetoxyechinoside A (249) | A. flammea | [110] |
| Desholothurin A (250) | P. graeffei | [93] | Pervicoside A (251) | H. pervicax | [111] |
| Pervicoside B (252) | H. pervicax | [111] | Pervicoside C (253) | H. pervicax | [111] |
| Arguside A (254) | Bohadschia argus | [112] | Holothurinoside J1 (255) | P. graeffei | [95] |
| Hemoiedemoside A (256) | H. spectabilis | [113] | Hemoiedemoside B (257) | H. spectabilis | [113] |
| Arguside D (258) | B. argus | [114] | Arguside E (259) | B. argus | [114] |
| Psolusoside A (260) | Psolus fabricii | [115] | Liouvilloside A4 (261) | S. liouvillei | [46] |
| Bivittoside B (262) | Bohadschia bivitta | [85] | Holothurinoside X (263) | H. lessoni | [116] |
| Holothurinoside Y (264) | H.lessoni | [116] | Holothurinoside Z (265) | H. lessoni | [116] |
| Cladoloside A1 (266) | Cladolabes chmeltzii | [117] | Cladoloside A2 (267) | C. chmeltzii | [117] |
| Cladoloside A3 (268) | C. chmeltzii | [117] | Cladoloside A4 (269) | C. chmeltzii | [117] |
| Cladoloside A5 (270) | C. chmeltzii | [117] | Cladoloside A6 (271) | C. chmeltzii | [117] |
| Colochiroside C (272) | C. chmeltzii | [64] | Colochiroside D (273) | C. robustus | [63] |
| Mollisoside B1 (274) | A. mollis | [55] | Neothyonidioside (275) | A. mollis | [118] |
| Compound Name | Producing Species | Reference | Compound Name | Producing Species | Reference |
|---|---|---|---|---|---|
| Cercodemasoide B (276) | Cercodemas anceps | [100] | Cercodemasoide C (277) | C. anceps | [100] |
| Cercodemasoide D (278) | C. anceps | [100] | Cercodemasoide E (279) | C. anceps | [100] |
| Holothurin B (280) | Holothuria lessoni | [119] | Holothurin B1 (281) | H. lessoni | [120] |
| Holothurin B2 (282) | H. polii | [121] | Holothurin B3 (283) | H. polii | [121] |
| Holothurin B4 (284) | H. polii | [121] | Holothurinoside D (285) | H. forskalii | [93] |
| Leucospilotaside A (286) | H. leucospilota | [122] | Leucospilotaside B (287) | H. leucospilota | [122] |
| Bivittoside A (288) | Bohadschia bivittata | [85] | Echinoside B (289) | A. echinites | [108] |
| 24-dehydroechinoside B (290) | Actinopyga mauritiana | [123] | Hillaside C (291) | Holothuria hilla | [124] |
| Hillaside B (292) | H. hilla | [81] |
| Compound Name | Producing Species | Reference | Compound Name | Producing Species | Reference |
|---|---|---|---|---|---|
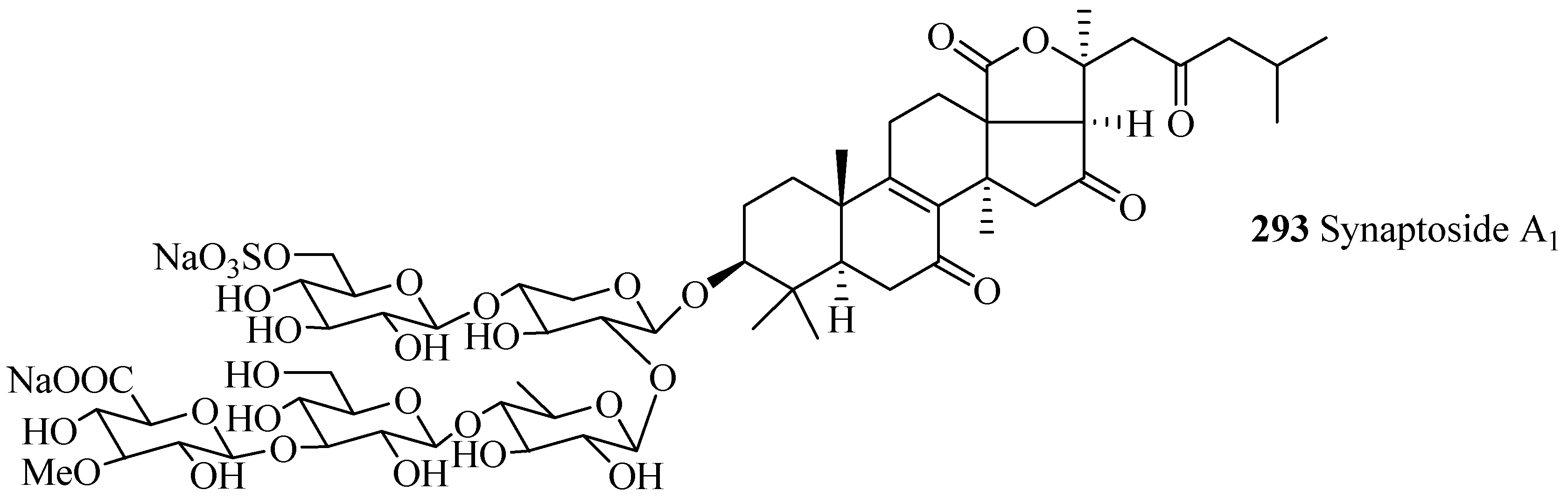
| Synaptoside A1 (293) | Synapta maculata | [41] | Variegatuside B (294) | Stichopus variegates | [73] |
| Variegatuside D (295) | Stichopus variegates | [21] |
| Compound Name | Producing Species | Reference | Compound Name | Producing Species | Reference |
|---|---|---|---|---|---|
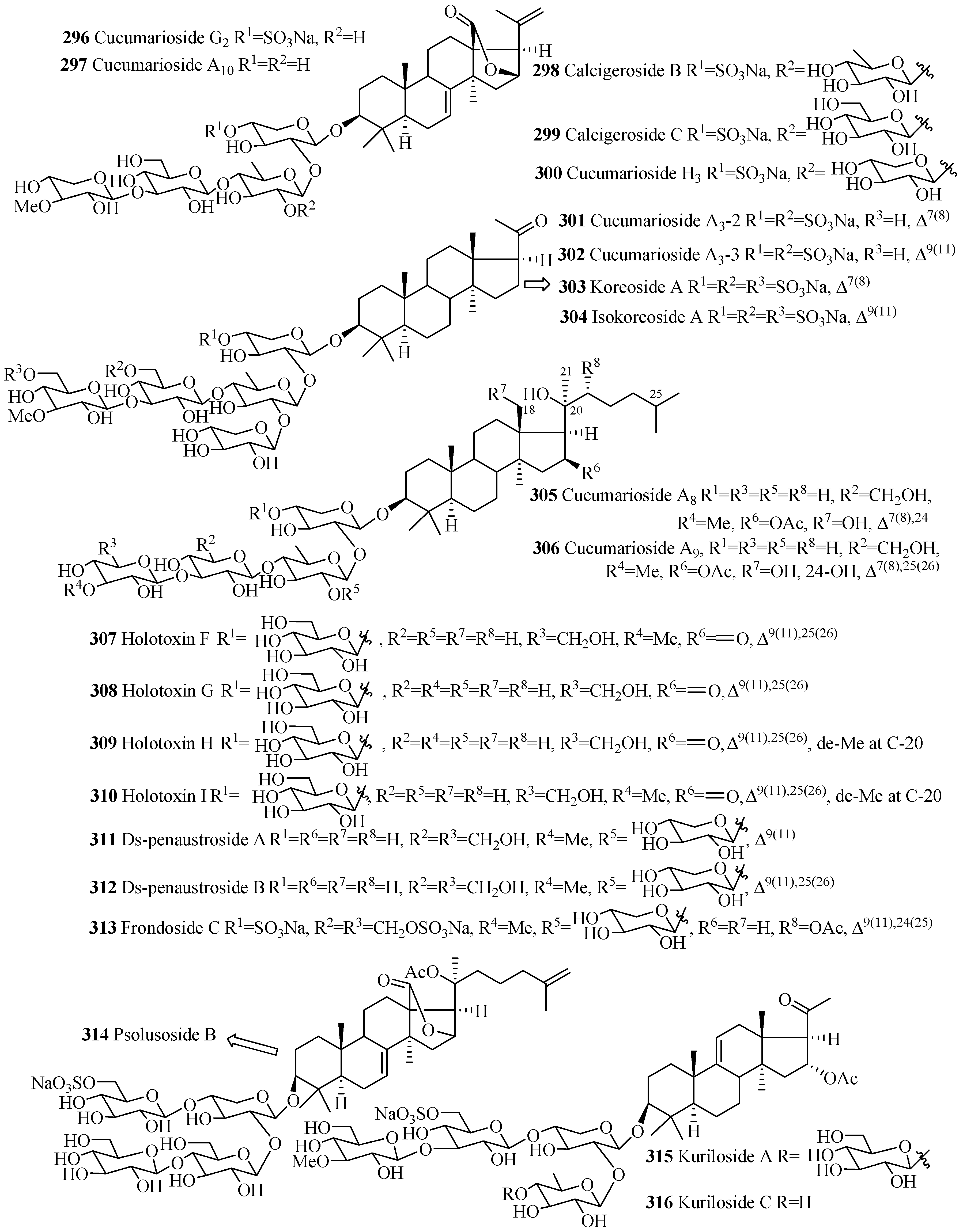
| Cucumarioside G2 (296) | E. fraudatrix | [125] | Cucumarioside A10 (297) | E. fraudatrix | [67] |
| Calcigeroside B (298) | P. calcigera | [37] | Calcigeroside C1 (299) | P. calcigera | [37] |
| Cucumarioside H3 (300) | E. fraudatrix | [30] | Cucumarioside A3-2 (301) | C. conicospermium | [26] |
| Cucumarioside A3-3 (302) | C. conicospermium | [26] | Koreoside A (303) | C. koraiensis | [126] |
| Isokoreoside A (304) | C. conicospermium | [26] | Cucumarioside A8 (305) | E. fraudatrix | [67] |
| Cucumarioside A9 (306) | E. fraudatrix | [67] | Holotoxin F (307) | A. japonicus | [22] |
| Holotoxin G (308) | A. japonicus | [22] | Holotoxin H (309) | S. japonicus | [127] |
| Holotoxin I (310) | S. japonicus | [127] | Ds-penaustroside A (311) | P. australis | [99] |
| Ds-penaustroside B (312) | P. australis | [99] | Frondoside C (313) | C. frondosa | [128] |
| Psolusoside B (314) | Psolus fabricii | [129] | Kuriloside A (315) | D. kurilensi | [130] |
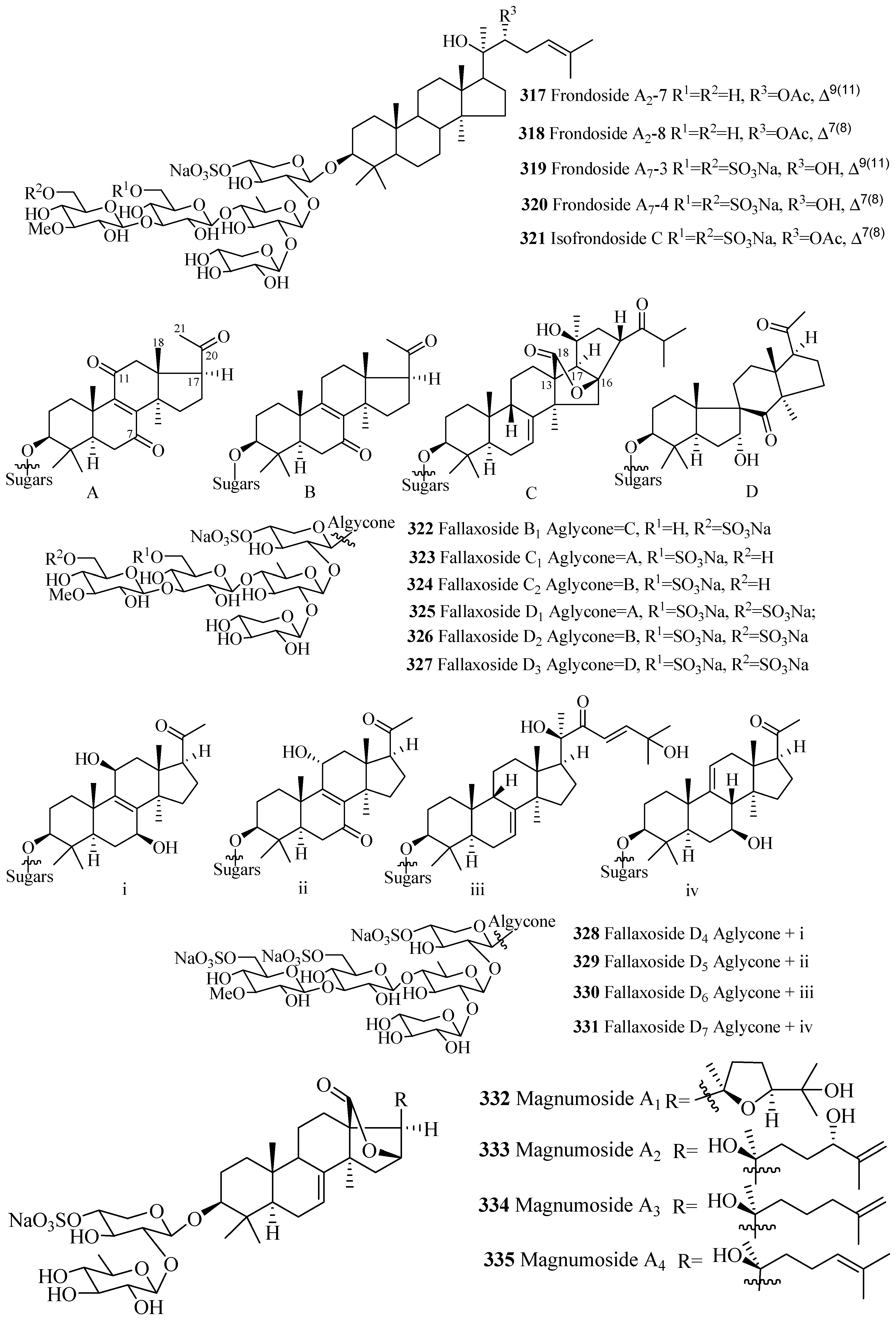
| Kuriloside C (316) | D. kurilensi | [130] | Frondoside A2-7 (317) | C. frondosa | [36] |
| Frondoside A2-8 (318) | C. frondosa | [36] | Frondoside A7-3 (319) | C. frondosa | [43] |
| Frondoside A7-4 (320) | C. frondosa | [43] | Isofrondoside C (321) | C. frondosa | [43] |
| Fallaxoside B1 (322) | C. fallax | [131] | Fallaxoside C1 (323) | C. fallax | [132] |
| Fallaxoside C2 (324) | C. fallax | [132] | Fallaxoside D1 (325) | C. fallax | [132] |
| Fallaxoside D2 (326) | C. fallax | [132] | Fallaxoside D3 (327) | C. fallax | [131] |
| Fallaxoside D4 (328) | C. fallax | [133] | Fallaxoside D5 (329) | C. fallax | [133] |
| Fallaxoside D6 (330) | C. fallax | [133] | Fallaxoside D7 (331) | C. fallax | [133] |
| Magnumoside A1 (332) | Massinium magnum | [134] | Magnumoside A2 (333) | M. magnum | [134] |
| Magnumoside A3 (334) | M. magnum | [134] | Magnumoside A4 (335) | M. magnum | [134] |
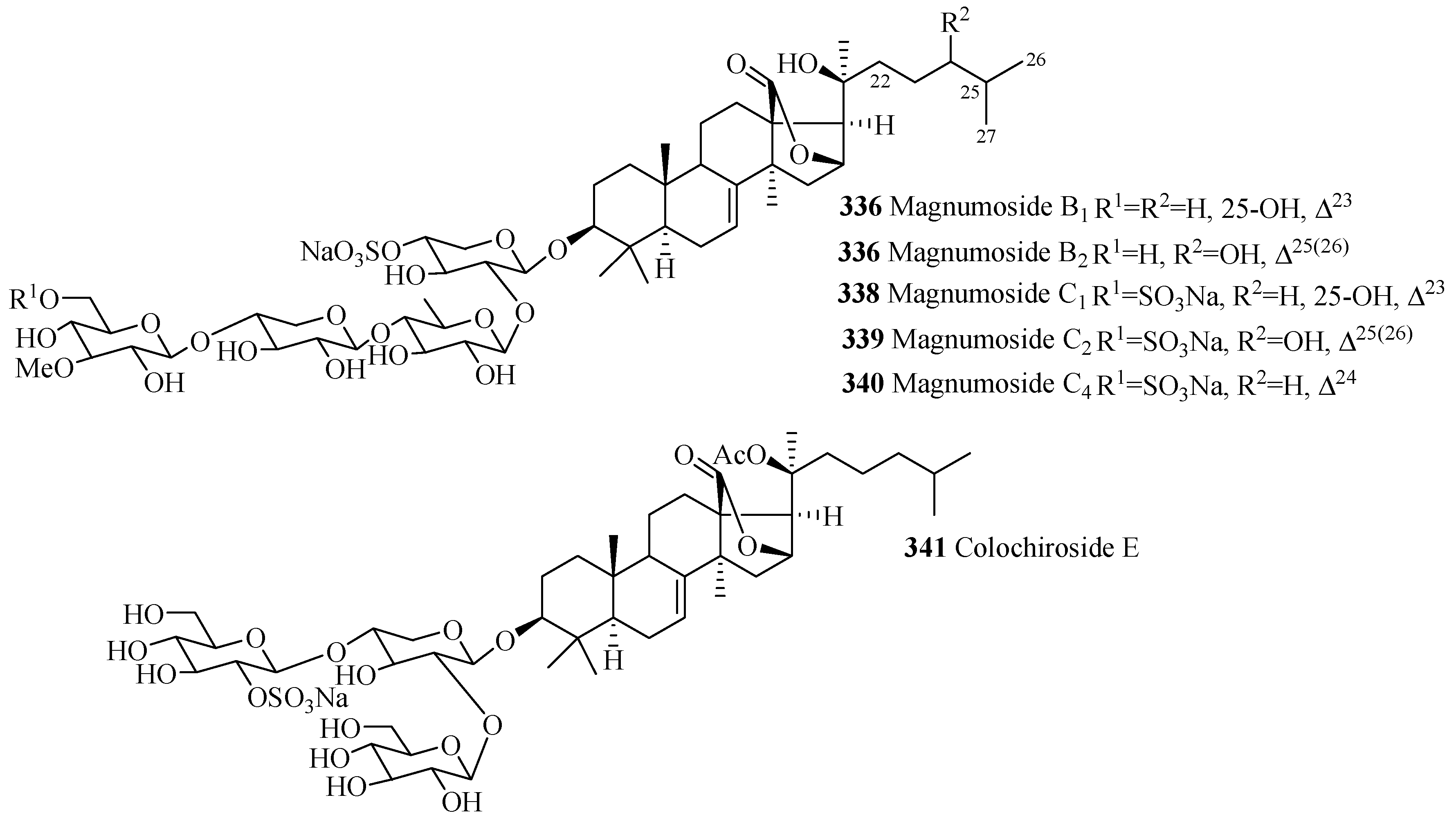
| Magnumoside B1 (336) | M. magnum | [134] | Magnumoside B2 (337) | M. magnum | [134] |
| Magnumoside C1 (338) | M. magnum | [134] | Magnumoside C2 (339) | M. magnum | [134] |
| Magnumoside C4 (340) | M. magnum | [134] | Colochiroside E (341) | C. robustus | [135] |
| Compound | Activity | Against/For | Activity Result | Reference |
|---|---|---|---|---|
| Hillaside C (285) | Cytotoxic | Human tumor cell lines | IC50: 0.15–3.20 µg/mL | [124] |
| Hemoiedemoside A (251) | Antifungal | C. cucumerinum | 20 µg/disc: 23 mm zone | [113] |
| Fuscocineroside C (237) | Cytotoxic | Human tumor cell lines | IC50: 0.88 and 0.58 µg/mL | [106] |
| Intercedenside A (66) | Cytotoxic | Human tumor cell lines | ED50: 0.96–4.0 µg/mL | [49] |
| Intercedenside B (67) | Cytotoxic | Human tumor cell lines | ED50: 0.61–2.0 µg/mL | [49] |
| Intercedenside C (68) | Cytotoxic | Human tumor cell lines | ED50: 0.96–4.0 µg/mL | [49] |
| Holothurinoside A (195) | Cytotoxic | Human tumor cell lines | IC50: 0.33–0.71 µg/mL | [93] |
| Holothurinoside C (229) | Cytotoxic | Human tumor cell lines | IC50: 0.16–0.93 µg/mL | [93] |
| Liouvilloside A (53) | Virucidal | Herpes simplex virus | <10 µg/mL | [44] |
| Leucospilotaside B (281) | Cytotoxic | Human tumor cell lines | IC50: 0.44–2.62 µg/mL | [122] |
| Arguside B (164) | Cytotoxic | Human tumor cell lines | IC50: 0.38–2.60 µg/mL | [87] |
| Arguside C (165) | Cytotoxic | Human tumor cell lines | IC50: 0.38–2.60 µg/mL | [87] |
| Philinopside A (78) | Cytotoxic | Human tumor cell lines | IC50: 1.70–3.50 µg/mL | [52] |
| Philinopside B (79) | Cytotoxic | Human tumor cell lines | IC50: 0.75–3.0 µg/mL | [53] |
| Cucumarioside A2-2 (18) | Hemolytic | Erythrocyte of mouse | ED50: 0.87 at 10−6 M | [136] |
| Holothurin B (274) | Antifungal | T. mentagrophytes | MIC-1.5 µg/mL | [137] |
| Holothurin A3 (227) | Cytotoxic | Human tumor cell lines | IC50 = 0.32–0.87 μg/mL | [103] |
| Holothurin A4 (228) | Cytotoxic | Human tumor cell lines | IC50 = 0.57–1.12 μg/mL | [103] |
| Scabraside A (230) | Antifungal | Eight pathogenic fungal strains | MIC80: 2–8 μg/mL | [138] |
| Echinoside A (238) | Antifungal | Eight pathogenic fungal strains | MIC80: 1–8 μg/mL | [108] |
| Cucumarioside A2-2 (18) | immunomodulatory | Increased lysosomal activity | 0.2–20 ng/mouse | [139] |
| Frondoside A (37) | immunomodulatory | Enhanced phagocytosis | 0.001 μg/mL | [140] |
| Philinopside E (80) | Cytotoxicity | Ten tumor cell lines | ED50: 0.75–3.50 μg/mL | [54] |
| Holotoxin A1 (152) | Antifungal | Five pathogenic fungi | MIC: 0.5–1.0 µg/mL | [22] |
| Cucumarioside A1 (106) | Hemolytic | Mouse erythrocytes | MEC100: 0.7 ± 0.1 µg/mL | [66] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mondol, M.A.M.; Shin, H.J.; Rahman, M.A.; Islam, M.T. Sea Cucumber Glycosides: Chemical Structures, Producing Species and Important Biological Properties. Mar. Drugs 2017, 15, 317. https://doi.org/10.3390/md15100317
Mondol MAM, Shin HJ, Rahman MA, Islam MT. Sea Cucumber Glycosides: Chemical Structures, Producing Species and Important Biological Properties. Marine Drugs. 2017; 15(10):317. https://doi.org/10.3390/md15100317
Chicago/Turabian StyleMondol, Muhammad Abdul Mojid, Hee Jae Shin, M. Aminur Rahman, and Mohamad Tofazzal Islam. 2017. "Sea Cucumber Glycosides: Chemical Structures, Producing Species and Important Biological Properties" Marine Drugs 15, no. 10: 317. https://doi.org/10.3390/md15100317
APA StyleMondol, M. A. M., Shin, H. J., Rahman, M. A., & Islam, M. T. (2017). Sea Cucumber Glycosides: Chemical Structures, Producing Species and Important Biological Properties. Marine Drugs, 15(10), 317. https://doi.org/10.3390/md15100317






