Carotenoids, Phenolic Compounds and Tocopherols Contribute to the Antioxidative Properties of Some Microalgae Species Grown on Industrial Wastewater
Abstract
:1. Introduction
2. Results and Discussion
2.1. Extraction of Phenolics and Carotenoids

2.2. Total Phenolics, Flavonoids, and Phenolic Composition
| Species | Total Phenolics (mg/g) * | Total Tocopherols (µg/g) | Total Carotenoids (mg/g) | Total Flavonoids (mg/g) ** |
|---|---|---|---|---|
| De.S | 7.72 ± 0.08 a | 361.9 ± 23 a | 6.70 ± 0.01 a | 4.03 ± 1.10 a |
| Du.S | 4.52 ± 0.05 d | 125.2 ± 23.5 b | 4.83 ± 0.01 d | 3.61 ± 1.09 a |
| N.L | 5.78 ± 0.12 c | 21.18 ± 0.05 d,e | 2.56 ± 0.02 g,f | 2.6 ± 0.30 a,b |
| P.T | 3.16 ± 0.04 f | 13.12 ± 0.01 e | 4.60 ± 0.03 e | 0.84 ± 0.12 a |
| N.S | 6.45 ± 0.25 b | 44.08 ± 3.11 c | 5.296 ± 0.01 b | 3.18 ± 0.59 a |
| C.S1 | 5.86 ± 0.06 c | 34.13 ± 0.37 c,d | 4.978 ± 0.06 c | 2.49 ± 0.7 a,b |
| C.S2 | 5.76 ± 0.12 c | 33.74 ± 0.27 c,d,e | 2.92 ± 0.15 f | 2.41 ± 0.9 a,b |
| Phenolic Compounds (µg/g) | De.S | Du.S | N.L | P.T | N.S | C.S1 | C.S2 |
|---|---|---|---|---|---|---|---|
| Gallic acid | 4.32 ± 0.01 | nd | 2.30 ± 0.02 | nd | 2.75 ± 0.03 | nd | nd |
| 2,5 dihydroxy benzoic acid | nd | nd | nd | nd | nd | nd | nd |
| 3,4 dihydroxy benzoic acid | nd | nd | nd | 1.64 ± 0.02 | 2.90 ± 0.07 | nd | nd |
| Caffeic acid | 1.11 ± 0.01 | 1.34 ± 0.04 | 1.37 ± 0.04 | nd | nd | 3.81 ± 0.03 | 3.12 ± 0.20 |
| Ferulic acid | 1.41 ± 0.04 | 4.07 ± 0.03 | 2.45 ± 0.04 | nd | 2.90 ± 0.05 | 2.81 ± 0.03 | 2.80 ± 0.20 |
| p-Coumaric acid | 1.91 ± 0.01 | 0.67 ± 0.02 | nd | 1.56 ± 0.12 | 0.29 ± 0.09 | 1.97 ± 0.05 | 1.16 ± 0.09 |
| Salycilic acid | nd | nd | 0.55 ± 0.07 | 1.91 ± 0.20 | 1.32 ± 0.01 | nd | nd |
| Cinnamic acid | 0.64 ± 0.01 | nd | 0.92 ± 0.01 | nd | nd | 0.47 ± 0.02 | 0.13 ± 0.04 |
| Total | 9.40 ± 0.09 b | 6.09 ± 0.04 f | 7.60 ± 0.05 d | 5.10 ± 0.12 g | 10.07 ± 0.03 a | 9.06 ± 0.09 c | 7.26 ± 0.08 e |
2.3. Total Tocopherols
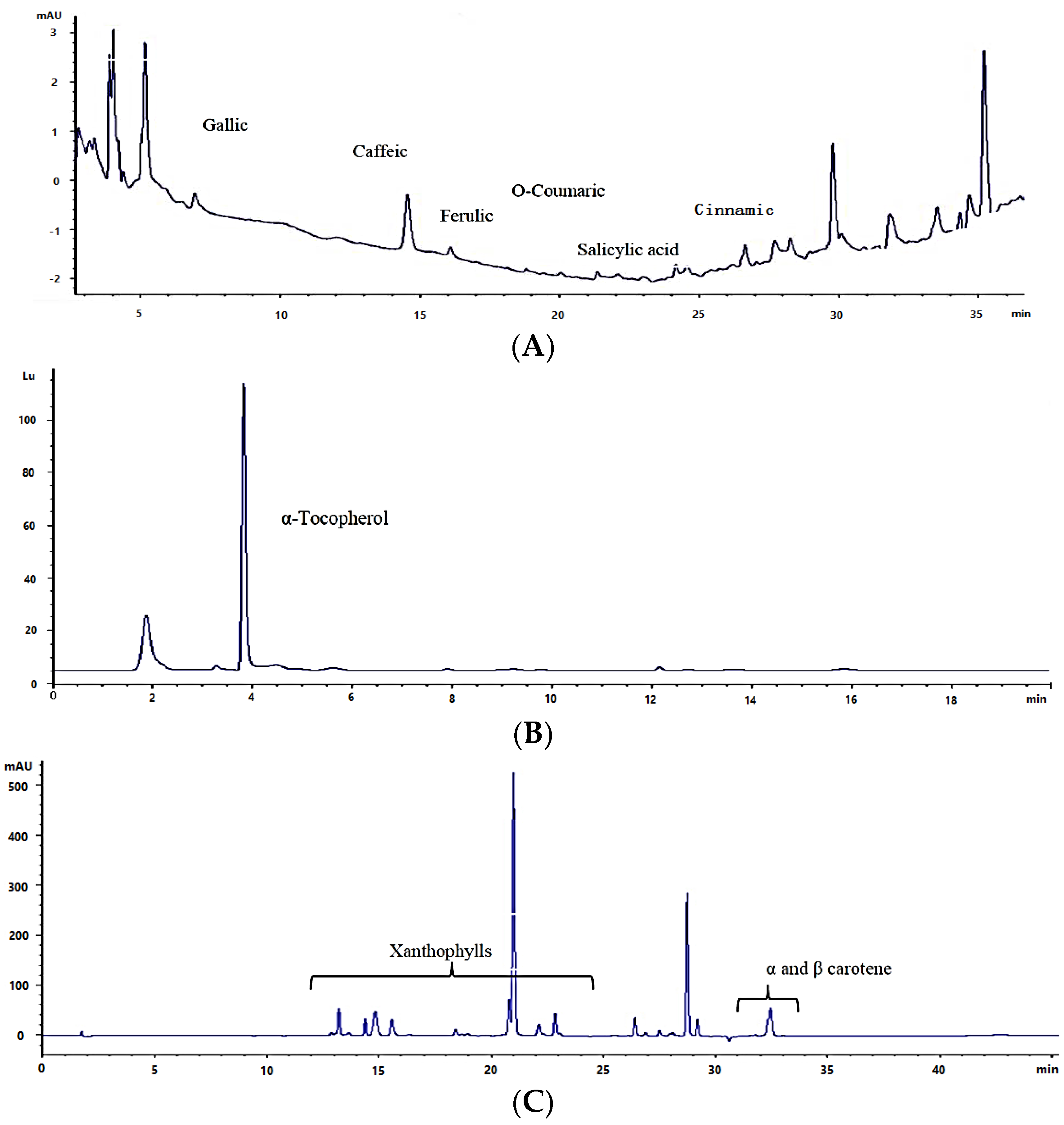
2.4. Total Carotenoids and Carotenoid Composition
| Pigments (µg/g) | De.S | Du.S | N.L | P.T | N.S | C.S1 | C.S2 |
|---|---|---|---|---|---|---|---|
| Chlorophyll c3 | nd | nd | nd | nd | nd | nd | nd |
| Unknown | 212.2 ± 5 | 351.0 ± 7.5 | nd | nd | nd | 269.7 ± 4.6 | 80.78 ± 1.3 |
| Chlorophyllide | nd | nd | nd | nd | nd | nd | nd |
| Peridin | nd | nd | nd | nd | nd | 16.70 ± 0.1 | 4.69 ± 0.1 |
| Vaucheriaxanthin | nd | nd | 164.8 ± 2.6 | nd | 85.16 ± 0.6 | nd | nd |
| 19-But-Fucoxanthin | nd | nd | nd | 50.75 ± 2.2 | nd | nd | nd |
| Fucoxanthin | nd | nd | 183.2 ± 9.8 | 264.5 ± 29 | 13.05 ± 0.1 | 104.8 ± 5.5 | 22.2 ± 0.2 |
| Neoxanthin | 158.3 ± 2.5 | 103.2 ± 12 | 423.4 ± 28 | nd | 53.45 ± 3.2 | 48.29 ± 1.2 | 20.0 ± 0.1 |
| Prasinoxanthin | nd | nd | nd | nd | nd | 22.64 ± 0.20 | 41.13 ± 0.2 |
| Violaxanthin | 54.60 ± 2.3 | 83.01 ± 11.7 | 1228 ± 61 | nd | 1679 ± 83 | nd | nd |
| 19-hex-fuco | nd | nd | nd | nd | nd | nd | nd |
| Dinoxanthin | nd | nd | nd | nd | nd | nd | nd |
| Antheraxanthin | nd | 344.4 ± 4.6 | nd | nd | nd | nd | nd |
| Astaxanthin | nd | nd | nd | nd | nd | 48.42 ± 0.5 | nd |
| Diadinoxanthin | 256.7 ± 3 | 43.44 ± 1.0 | nd | 2166 ± 68 | 140.5 ± 2.4 | nd | nd |
| Alloxanthin | 17.64 ± 0.1 | 55.73 ± 0.5 | nd | nd | 130.5 ± 2.9 | 94.06 ± 0.8 | 24.33 ± 0.1 |
| Diatoxanthin | 26.75 ± 1.0 | nd | 136.3 ± 1.0 | 1558 ± 88 | nd | nd | nd |
| Lutein | 5111 ± 61 | 3014± 76 | nd | nd | nd | 3220 ± 54 | 2069 ± 34 |
| Zeaxanthin | 284.5 ± 2.5 | 195.8 ± 6.7 | 136.8 ± 1.0 | nd | 584.9 ± 3.6 | 151.1 ± 1.7 | 15.52 ± 1.0 |
| Dihydro lutein | 145.0 ± 2.9 | 175.6 ± 2.5 | nd | 216.2 ± 2.0 | nd | 248.7 ± 9 | 111.1 ± 17 |
| Unknown | nd | nd | nd | nd | 165.2 ± 3.5 | nd | nd |
| Chlorophyll b | 862.6 ± 8.6 | 1454 ± 23 | nd | nd | nd | 725 ± 35 | 389.3 ± 19 |
| Chlorophyll a | 2993 ± 14 | 3424 ± 87 | 1065 ± 22 | 2714 ± 23 | 2001 ± 54 | 615.3 ± 4.1 | 1455 ± 3.9 |
| β-carotene | 647.3 ± 13 | 743.5 ± 44.3 | 284.5 ± 2 | 348.7 ± 4.4 | 2223 ± 88 | 1039 ± 17 | 614.4 ± 8.3 |
| Canthaxanthin | nd | nd | 3.40 ± 0.05 | nd | 136.5 ± 12.1 | nd | nd |
| α-carotene | nd | 76.01 ± 0.1 | nd | nd | 84.16 ± 3.2 | nd | nd |
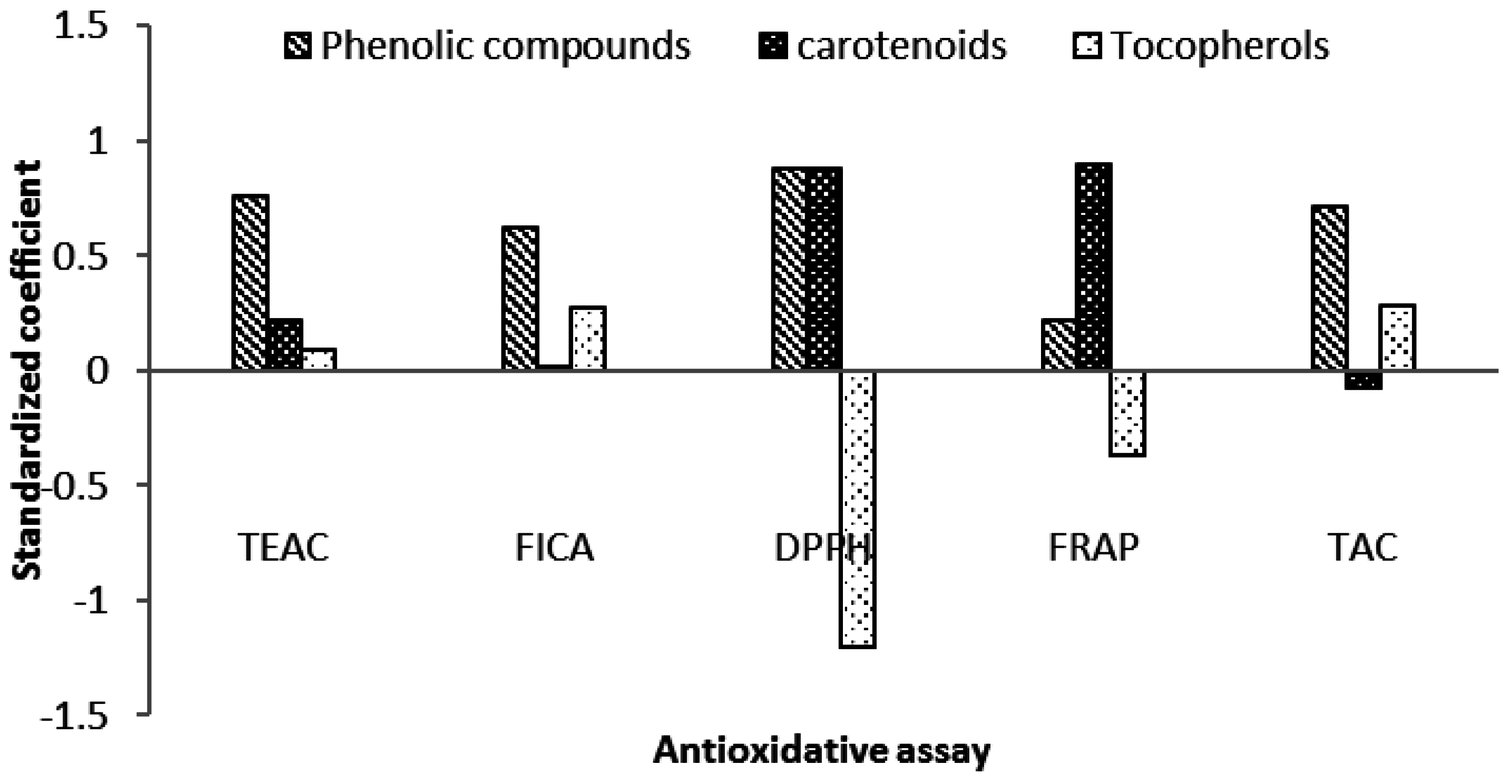
2.5. Antioxidative Properties
2.5.1. ABTS-Radical Scavenging Capacity (TEAC)
2.5.2. Ferrous Ion-Chelating Ability (FICA)
| Concentration * TEAC | De.S a | Du.S c,d | N.L c,d | P.T e | N.S a,b | C.S1 d | C.S2 b,c |
| 1.0 a | 24.26 ± 0.60 | 14.38 ± 0.40 | 16.60 ± 0.20 | 6.79 ± 0.02 | 20.34 ± 0.15 | 18.75 ± 0.16 | 13.48 ± 0.38 |
| 0.50 b | 10.05 ± 0.30 | 6.58 ± 0.03 | 5.48 ± 0.23 | 3.35 ± 0.10 | 8.68 ± 0.10 | 6.32 ± 0.11 | 7.30 ± 0.20 |
| 0.25 b | 7.05 ± 0.10 | 5.01 ± 0.05 | 5.16 ± 0.01 | 2.73 ± 0.20 | 6.66 ± 0.14 | 5.24 ± 0.14 | 6.80 ± 0.010 |
| Concentration FICA | De. S a | Du.S c | N.L d | P.T e | N.S b | C.S1 c,d | C.S2 b |
| 1.0 a | 20.10 ± 0.51 | 13.99 ± 0.18 | 11.61 ± 0.97 | 9.67 ± 0.60 | 16.90 ± 0.14 | 12.15 ± 0.042 | 17.91 ± 0.21 |
| 0.5 b | 16.87 ± 0.60 | 11.77 ± 0.31 | 9.45 ± 0.49 | 7.55 ± 0.21 | 14.25 ± 0.35 | 10.57 ± 0.60 | 14.92 ± 0.17 |
| 0.25 c | 8.57 ± 0.17 | 5.42 ± 0.17 | 3.95 ± 0.21 | 3.35 ± 0.21 | 7.17 ± 0.10 | 5.40 ± 0.14 | 7.53 ± 0.19 |
| Concentration DPPH | De. S c | Du.S d | N.L a,b | P.T d | N.S b | C.S1 a | C.S2 c,d |
| 1.0 a | 29.11 ± 0.01 | 26.95 ± 0.10 | 35.17 ± 0.08 | 28.35 ± 0.07 | 30.32 ± 0.02 | 34.09 ± 0.08 | 28.06 ± 0.04 |
| 0.5 b | 18.24 ± 0.14 | 15.55 ± 0.21 | 19.44 ± 0.14 | 15.58 ± 0.01 | 21.26 ± 0.08 | 26.05 ± 0.07 | 15.38 ± 0.26 |
| 0.25 c | 10.29 ± 0.24 | 12.66 ± 0.09 | 14.27 ± 0.09 | 8.41 ± 0.12 | 12.55 ± 0.07 | 11.74 ± 0.05 | 9.34 ± 0.15 |
| Concentration FRAP | De.S a | Du.S b,c | N.L e | P.T d | N.S a | C.S1 c,d | C.S2 b |
| 1.0 a | 0.45 ± 0.01 | 0.31 ± 0.02 | 0.30 ± 0.01 | 0.27 ± 0.01 | 0.39 ± 0.021 | 0.32 ± 0.01 | 0.35 ± 0.01 |
| 0.5 b | 0.32 ± 0.01 | 0.27 ± 0.01 | 0.16 ± 0.01 | 0.26 ± 0.01 | 0.35 ± 0.02 | 0.25 ± 0.01 | 0.27 ± 0.01 |
| 0.25 c | 0.25 ± 0.01 | 0.25 ± 0.01 | 0.14 ± 0.01 | 0.17 ± 0.01 | 0.30 ± 0.01 | 0.18 ± 0.01 | 0.22 ± 0.01 |
| Time ** TAC | De.S a | Du.S c | N.L b | P.T d | N.S a | C.S1 c | C.S2 c |
| 60 a | 5.22 ± 0.14 | 3.24 ± 0.13 | 2.65 ± 0.13 | 1.83 ± 0.01 | 6.30 ± 0.012 | 2.96 ± 0.08 | 3.47 ± 0.04 |
| 90 b | 8.56 ± 0.21 | 5.17 ± 0.05 | 6.57 ± 0.41 | 2.55 ± 0.21 | 7.29 ± 0.13 | 4.32 ± 0.09 | 4.65 ± 0.16 |
| 120 b | 8.95 ± 0.01 | 5.27 ± 0.01 | 7.00 ± 0.02 | 2.99 ± 0.06 | 7.30 ± 0.14 | 4.31 ± 0.26 | 5.36 ± 0.06 |
2.5.3. DPPH Radical Scavenging Activity Assay (DPPH)
2.5.4. Ferrous Reduction Power (FRAP)
2.5.5. Total Antioxidant Capacity Assay (TAC)
2.6. Contribution of Carotenoids in Antioxidative Activity of Microalgae Biomass
2.7. Effects of Source of Nitrogen on Productivity of Biomass and Bioactive Compounds
3. Experimental Section
3.1. Chemicals and Reagents
3.2. Microalgae Biomass
| Item | Unit | Amount |
|---|---|---|
| pH | - | 8.1 |
| Suspended solids | mg/L | 20 |
| Total N | mg/L | 190 |
| Ammonia + ammonium-N | mg/L | 150 |
| Nitrite + nitrate | mg/L | <0.1 |
| Total P | mg/L | 11 |
| Sulphate | mg/L | 3.6 |
| Total cyanide | µg/L | 2.5 |
| Total Alkalinity | mmol/L | 62.5 |
| EDTA | mg/L | <0.5 |
| Sodium(Na) | mg/L | 1500 |
| Cadmium (Cd) | µg/L | <0.05 |
| Copper (Cu) | µg/L | 3.4 |
| Iron (Fe) | mg/l | 0.23 |
| Cobolt (Co) | µg/L | <0.5 |
3.3. Sample Preparations
3.3.1. Antioxidative Properties, Tocopherols, and Phenolic Compounds (HPLC)
3.3.2. Pigments
3.4. Analytical Methods
3.4.1. Total Phenolic Content
3.4.2. Phenolic Compounds (HPLC)
3.4.3. Total Carotenoids and Pigment Composition
3.4.4. Total Flavonoids
3.4.5. Total Tocopherols
3.4.6. Total Antioxidant Capacity Assay (TAC)
3.4.7. DPPH Radical Scavenging Activity Assay (DPPH)
3.4.8. Ferrous Ion-Chelating Ability (FICA)
3.4.9. Ferrous Ion Reduction Power (FRAP)
3.4.10. ABTS-Radical Scavenging (TEAC)
3.4.11. Statistical Analyses
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Stengel, D.B.; Connan, S.; Popper, Z.A. Algal chemo diversity and bioactivity: Sources of natural variability and implications for commercial application. J. Biotechnol. Adv. 2011, 29, 483–501. [Google Scholar] [CrossRef] [PubMed]
- Rong, T. Review, Chemistry and Biochemistry of Dietary Polyphenols. Nutrients 2010, 2, 1231–1246. [Google Scholar]
- Li, H.B.; Cheng, K.W.; Wong, C.C. Evaluation of antioxidant capacity and total phenolic content of different fractions of selected microalgae. Food Chem. 2007, 102, 771–776. [Google Scholar] [CrossRef]
- Rao, A.R.; Sarada, R.; Baskaran, V.; Ravishankar, G.A. Antioxidant activity of Botryococcus braunii extract elucidated in vitro models. J. Agric. Food Chem. 2006, 54, 4593–4599. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Ho, J.A.; Shieh, M.; Lu, I. Antioxidant and antiproliferative activities of Spirulina and Chlorella water extracts. J. Agric. Food Chem. 2005, 53, 4207–4212. [Google Scholar] [CrossRef] [PubMed]
- Manivannan, K.; Anantharaman, P.; Balasubramanian, T. Evaluation of antioxidant properties of marine microalga Chlorella marina (Butcher, 1952). Asian Pac. J. Trop. Biomed. 2012, 2, S342–S346. [Google Scholar] [CrossRef]
- Anantharaman, P.; Hemalatha, A.; Girija, K.; Parthiban, C.; Saranya, C. Antioxidant properties and total phenolic content of a marine diatom, Navicula clavata and green microalgae, Chlorella marina and Dunaliella salina. Adv. Appl. Sci. Res. 2013, 4, 151–157. [Google Scholar]
- Herrero, M.J.L.; Martin-Alvarez, P.J.; Cifuentes, A.; Ibanez, E. Optimization of the extraction of antioxidants from Dunaliella salina microalga by pressurized liquids. J. Agric. Food Chem. 2006, 54, 5597–5603. [Google Scholar] [CrossRef] [PubMed]
- Choochote, W.; Suklampoo, L.; Ochaikul, D. Evaluation of antioxidant capacities of green microalgae. J. Appl. Phycol. 2014, 26, 43–48. [Google Scholar] [CrossRef]
- Guzman, S.; Gato, A.; Galleja, J.M. Anti-inflammatory, analgesic and free radical scavenging activities of the marine microalgae Chlorella stigmatophora and Phaeodactylum tricorntum. Phytother. Res. 2001, 15, 224–230. [Google Scholar] [CrossRef] [PubMed]
- Jaime, L.; Mendiola, J.A.; Herrero, M.; Soler-Rivas, C.; Santoyo, S.; Senorans, F.J. Separation and characterization of antioxidants from Spirulina platensis microalga combining pressurized liquid extraction, TLC and HPLC-DAD. J. Sep. Sci. 2005, 28, 2111–2119. [Google Scholar] [CrossRef] [PubMed]
- Miranda, M.S.; Cintra, R.G.; Barros, S.B.M.; Mancini-Filho, J. Antioxidant activity of the microalga Spirulina maxima. Braz. J. Med. Biol. Res. 1998, 31, 1075–1079. [Google Scholar] [CrossRef] [PubMed]
- Goh, S.; Yusoff, M.F.; Loh, S.P. A Comparison of the Antioxidant Properties and Total Phenolic Content in a Diatom, Chaetoceros sp. and a Green Microalga, Nannochloropsis sp. J. Agric. Sci. 2010, 2, 1–8. [Google Scholar] [CrossRef]
- Lee, S.; Lee, J.B.; Lee, K.W.; Jeon, Y.J. Antioxidant properties of tidal pool microalgae, Halochlorococcum porphyrae and Oltamannsiellopsis unicellularis from Jeju Island, Korea. Algae 2010, 25, 45–56. [Google Scholar] [CrossRef]
- Barredo, J.L. Microbial Carotenoids from Bacteria and Microalgae. In Methods and Protocols; Humana Press: New York, NY, USA, 2012; p. 5. [Google Scholar]
- Goiris, K.; Muylaert, K.; Fraeye, I.; Foubert, I.; de Brabanter, J.; de Cooman, L. Antioxidant potential of microalgae in relation to their phenolic and carotenoid content. J. Appl. Phycol. 2012, 24, 1477–1486. [Google Scholar] [CrossRef]
- Takaichi, S. Carotenoids in Algae: Distributions, Biosyntheses and Functions. Mar. Drugs 2011, 9, 1101–1118. [Google Scholar] [CrossRef] [PubMed]
- Jahnke, L.S. Massive carotenoid accumulation in Dunaliella bardawil induced by ultraviolet-A radiation. J. Photochem. Photobiol. 1999, 48, 68–74. [Google Scholar] [CrossRef]
- Natrah, F.M.; Yusoff, F.M.; Shariff, M.; Abbas, F.; Mariana, N.S. Screening of Malaysian indigenous microalgae for antioxidant properties and nutritional value. J. Appl. Phycol. 2007, 19, 711–718. [Google Scholar] [CrossRef]
- Klejdus, B.; Kopeckýb, J.; Benešová, L.; Vaceka, J. Solid-phase/supercritical-fluid extraction for liquid chromatography of phenolic compounds in freshwater microalgae and selected cyanobacterial species. J. Chromatogr. 2009, 1216, 763–771. [Google Scholar] [CrossRef] [PubMed]
- Chaudhuri, D.; Baban, N.; Deb, S.; Panja, S.; Sarkar, R.; Rout, J.; Mandal, N. Assessment of the phytochemical constituents and antioxidant activity of a bloom forming microalgae Euglena tuba. Biol. Res. 2014, 47, 24–27. [Google Scholar] [CrossRef] [PubMed]
- Goiris, K.; Muylaert, K.; Voorspoels, S.; de Paepe, D.J.E.; Baart, G.; de Cooman, L. Detection of flavonoids in microalgae from different evolutionary lineages. J. Phycol. 2014, 50, 483–492. [Google Scholar] [CrossRef]
- Liu, J.; Sun, Z.; Gerken, H. Potential Applications of Microalgae in Wastewater Treatments. In Recent Advances in Microalgal Biotechnology; OMICS Group e-Books: Hyderabad, India, 2014. [Google Scholar]
- Zepka, L.Q.; Rodrigues, D.B.; Flores, E.M.M.; Barin, J.S. Production of carotenoids from microalgae cultivated using agro industrial wastes. Food Res. Int. 2014, 65, 144–148. [Google Scholar]
- Francisco, J.B.; Grimi, N.; Vorobiev, E. New Approaches for the Use of Non-conventional Cell Disruption Technologies to Extract Potential Food Additives and Nutraceuticals from Microalgae. Food Eng. Rev. 2015, 7, 45–62. [Google Scholar]
- Lebovka, N.; Parniakov, O.; Barba, F.J.; Grimi, N.; Marchal, L.; Jubeau, S.; Vorobiev, E. Pulsed electric field assisted extraction of nutritionally valuable compounds from microalgae Nannochloropsis spp. using the binary mixture of organic solvents and water. Innov. Food Sci. Emerg. Technol. 2015, 27, 79–85. [Google Scholar]
- Grimi, N.; Parniakov, O.; Apicella, E.; Koubaa, M.; Barb, J.; Lebovka, N.; Pataro, G.; Ferrari, G.; Vorobiev, E. Ultrasound-assisted green solvent extraction of high-added value compounds from microalgae Nannochloropsis spp. Bioresour. Technol. 2015, 198, 262–267. [Google Scholar]
- Craft, N.E.; Soares, J.H. Relative Solubility, Stability, and Absorptivity of Lutein and β-Carotene in Organic Solvents. J. Aagric. Food Chem. 1992, 40, 431–434. [Google Scholar] [CrossRef]
- Sava, C.; Sibru, R. Analytical study of the determination of flavonoids in Black Sea algae. Ovidius Univ. Ann. Chem. 2010, 21, 29–34. [Google Scholar]
- Klein, B.C.; Lange, H.A.; Buchholz, R. Microalgae as natural sources for antioxidative compounds. J. Appl. Phycol. 2012, 24, 1133–1139. [Google Scholar] [CrossRef]
- Duan, X.; Zhang, W.; Li, X.; Wang, B. Evaluation of antioxidant property of extract and fractions obtained from a red alga, Polysiphonia urceolata. Food Chem. 2006, 95, 37–43. [Google Scholar] [CrossRef]
- Kuda, T.; Tsunekawa, M.; Hishi, T.; Araki, Y. Antioxidant properties of dried Kayamo-nori, a brown alga Scytosiphon lomentaria (Scytosiphonales, Phaeophyceae). Food Chem. 2005, 89, 617–622. [Google Scholar] [CrossRef]
- Hajimahmoodi, M.; Faramarzi, M.A.; Mohammadi, N.; Soltani, N.; Oveisi, M.R.; Nafissi-Varcheh, N. Evaluation of antioxidant properties and total phenolic contents of some strains of microalgae. J. Appl. Phycol. 2010, 22, 43–50. [Google Scholar] [CrossRef]
- Anantharaman, P.; Saranya, C.; Hemalatha, A.; Parthiban, C. Evaluation of Antioxidant Properties, Total Phenolic and Carotenoid Content of Chaetoceros calcitrans, Chlorella salina and Isochrysis galbana. Int. J. Curr. Microbiol. App. Sci. 2014, 3, 365–377. [Google Scholar]
- Jimenez-Escrig, A.; Jimenez-Jimenez, I.; Pulido, R.; Saura-Calixto, F. Antioxidant activity of fresh and processed edible seaweeds. J. Sci. Food Agric. 2001, 81, 530–534. [Google Scholar] [CrossRef]
- Everette, J.D.; Bryant, Q.M.; Green, A.M.; Abbey, Y.A.; Wangila, G.W.; Walker, R.B. Thorough Study of Reactivity of Various Compound Classes toward the Folin Ciocalteu Reagent. J. Agric. Food Chem. 2010, 58, 8139–8144. [Google Scholar] [CrossRef] [PubMed]
- Durmaz, Y. Vitamin E (α-tocopherol) production by the marine microalgae Nannochloropsis oculata (Eustigmatophyceae) in nitrogen limitation. Aquaculture 2007, 272, 717–722. [Google Scholar] [CrossRef]
- Vismara, R.; Vestri, S.; Kusmic, C.; Brarsanti, L.; Gualtieri, P. Natural vitamin E enrichment of Artema salina fed freshwater and marine microalgae. J. Appl. Phycol. 2003, 15, 75–80. [Google Scholar] [CrossRef]
- Solovchenko, A.E.; Chivkunova, O.B.; Semenova, L.R. Stress-induced changes in pigment and fatty acid content in the microalga Desmodesmus sp. Isolated from a White Sea hydroid. Russ. J. Plant Physiol. 2013, 60, 313–321. [Google Scholar] [CrossRef]
- Mulders, K.J.M.; Lamers, P.P.; Martens, D.E.; Wijffels, R.H. Phototrophic pigment production with microalgae: Biological constraints and opportunitie. J. Phycol. 2014, 50, 229–242. [Google Scholar] [CrossRef]
- Cordero, B.F.; Obraztsova, I.; Couso, I.; Leon, R.; Vargas, M.A.; Rodriguez, H. Enhancement of lutein production in Chlorella sorokiniana (Chorophyta) by improvement of culture conditions and random mutagenesis. Mar. Drugs 2011, 9, 1607–1624. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Ho, S.; Chen, C.N. Phototrophic cultivation of a thermo-tolerant Desmodesmus sp. for lutein production: Effects of nitrate concentration, light intensity and fed-batch operation. Bioresour. Technol. 2013, 144, 435–440. [Google Scholar] [CrossRef] [PubMed]
- Prior, R.L.; Wu, X.; Schaich, K. Standardized methods for the determination of antioxidant capacity and phenolics in foods and dietary supplements. J. Agric. Food Chem. 2005, 53, 4290–4302. [Google Scholar] [CrossRef] [PubMed]
- Sindu, E.R.; Preethi, K.C.; Kuttan, R. Antioxidant activity of carotenoid lutein in vitro and in vivo. Indian J. Exp. Biol. 2010, 48, 843–848. [Google Scholar]
- Wang, L.; Min, M.; Li, Y.; Chen, P.; Chen, Y. Cultivation of green algae Chlorella sp. in different wastewaters from municipal wastewater treatment plant. Appl. Biochem. Biotechnol. 2010, 162, 1174–1186. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, E.; Galvan, A. Inorganic nitrogen assimilation in Chlamydomonas. J. Exp. Bot. 2007, 58, 2279–2287. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Sun, Z.; Sun, P.; Chen, T.; Zhang, J. Microalgal carotenoids: beneficial effects and potential in human health. Food Funct. 2014, 5, 413–415. [Google Scholar]
- Miki, W. Biological functions and activities of animal carotenoids. Pure Appl. Chem. 1991, 63, 141–146. [Google Scholar] [CrossRef]
- Wagenen, J.V.; Mathias, L.P.; Angelidaki, I. Characterization of nutrient removal and microalgal biomass production on an industrial waste-stream by application of the deceleration-stat technique. Water Res. 2015, 75, 301–311. [Google Scholar] [CrossRef] [PubMed]
- Van huelkem, L.; Thomas, C.S. Computer-assisted high-performance liquid chromatography method development with applications to the isolation and analysis of phytoplankton pigments. J. Chromatogr. 2001, 910, 31–49. [Google Scholar]
- Firestone, D.; Society, A.O.C. Official Methods and Recommended Practices of the AOCS, 5th ed.; American Oil Chemists’ Society: Urbana, IL, USA, 2009. [Google Scholar]
- Pan, Y.; Zhu, J.; Wang, H.; Zhang, X.; Zhang, Y.; He, C.; Ji, X.; Li, H. Antioxidant activity of ethanolic extract of Cortex fraxini and use in peanut oil. Food Chem. 2007, 103, 913–918. [Google Scholar] [CrossRef]
- Benzie, I.F.; Strain, J.J. Ferric reducing antioxidant power assay: Direct measure of total antioxidant activity of biological fluids and modified version for simultaneous measurement of total antioxidant power and ascorbic acid concentration. Methods Enzymol. 1999, 299, 15–27. [Google Scholar] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Safafar, H.; Van Wagenen, J.; Møller, P.; Jacobsen, C. Carotenoids, Phenolic Compounds and Tocopherols Contribute to the Antioxidative Properties of Some Microalgae Species Grown on Industrial Wastewater. Mar. Drugs 2015, 13, 7339-7356. https://doi.org/10.3390/md13127069
Safafar H, Van Wagenen J, Møller P, Jacobsen C. Carotenoids, Phenolic Compounds and Tocopherols Contribute to the Antioxidative Properties of Some Microalgae Species Grown on Industrial Wastewater. Marine Drugs. 2015; 13(12):7339-7356. https://doi.org/10.3390/md13127069
Chicago/Turabian StyleSafafar, Hamed, Jonathan Van Wagenen, Per Møller, and Charlotte Jacobsen. 2015. "Carotenoids, Phenolic Compounds and Tocopherols Contribute to the Antioxidative Properties of Some Microalgae Species Grown on Industrial Wastewater" Marine Drugs 13, no. 12: 7339-7356. https://doi.org/10.3390/md13127069
APA StyleSafafar, H., Van Wagenen, J., Møller, P., & Jacobsen, C. (2015). Carotenoids, Phenolic Compounds and Tocopherols Contribute to the Antioxidative Properties of Some Microalgae Species Grown on Industrial Wastewater. Marine Drugs, 13(12), 7339-7356. https://doi.org/10.3390/md13127069








