Marine Cyanobacteria Compounds with Anticancer Properties: A Review on the Implication of Apoptosis
Abstract
:1. Introduction
| Compound | Source | Class of compound | Cytoxicity assay | Human cell line tested | Reference |
|---|---|---|---|---|---|
| Ankaraholide A | Geitlerinema | Glycosilated swinholide | MTT | NCI-H460 lung tumor | [14] |
| SRB | MDA-MB-435 breast carcinoma | [14] | |||
| Apratoxin A | Lyngbya majuscula | Cyclic depsipeptide | SRB | KB oral epidermoid cancer and LoVo colon cancer | [40,41] |
| MTT | U2OS osteosarcoma, HT29 colon adenocarcinoma and HeLa cervical carcinoma | [42] | |||
| Apratoxins B-C | Lyngbya sp. | Cyclic depsipeptides | MTT | KB oral epidermoid cancer and LoVo colon cancer | [40] |
| Apratoxin D | Lyngbya majuscula and Lyngbya sordida | Cyclic depsipeptide | MTT | H-460 lung cancer | [26] |
| Apratoxin E | Lyngbya bouilloni | Cyclic depsipeptide | MTT | U2OS osteosarcoma, HT29 colon adenocarcinoma and HeLa epithelial carcinoma | [42] |
| Apratoxins F and G | Lyngbya bouilloni | Cyclic depsipeptides | MTT | H-460 lung cancer | [43] |
| Hemocytometer counting | HCT-116 colorectal cancer cells | [43] | |||
| Aurilide B | Lyngbya majuscula | Cyclic depsipeptide | MTT | H-460 lung tumor | [24] |
| Aurilide C | Lyngbya majuscula | Cyclic depsipeptide | MTT | NCI-H460 lung tumor | [24] |
| Belamide A | Symploca sp. | Linear tetrapeptide | Non-specified | HCT-116 colon cancer | [8] |
| Bisebromoamide | Lyngbya sp. | Peptide | SRB | HeLa S3 epithelial carcinoma | [44] |
| Biselyngbyaside | Lyngbya sp. | Glicomacrolide | SRB | HeLa S3 epithelial carcinoma, SNB-78 central nervous system cancer and NCI H522 lung cancer | [15] |
| Calothrixin A | Calothrix | Pentacyclic indolophenanthridine | 3H-thymidine incorporation | HeLa epithelial carcinoma | [45] |
| MTT | Leukemia CEM | [46] | |||
| Calothrixin B | Calothrix | Pentacyclic indolophenanthridine | MTT | HeLa epithelial carcinoma | [47] |
| Leukemia CEM | [46] | ||||
| Caylobolide A | Lyngbya majuscula | Macrolactone | Non-specified | HCT-116 colon tumor | [48] |
| Caylobolide B | Phormidium spp. | Macrolactone | MTT | HT29 colorectal adenocarcinoma and HeLa cervical carcinoma | [16] |
| Coibamide A | Leptolyngbya sp. | Cyclic depsipeptide | MTT | Lung cancer NCI-H460, breast cancer MDA-MB-231, melanoma LOX IMVI, leukemia HL-60 and astrocytoma SNB75 | [49] |
| Cryptophycin 1 | Nostoc spp. | Cyclic depsipeptide | Cell morphology examination | MDA-MB-435 mammary adenocarcinoma and SKOV3 ovarian carcinoma | [50] |
| AlamarBlue dye reduction | Leukemia U937, CCRF-CEM and HL-60, colon carcinoma HT-29, GC3 and Caco-2, mammary carcinoma MCF-7 and MDA-MB-231 and cervical carcinoma HeLa | [51] | |||
| Dolastatin 10 | Symploca sp. | Linear Pentapeptide | MTT | Lung A549 carcinoma | [52] |
| Human lung cancer cells: NCI-H69, -H82, -H446 and -H510 | [53] | ||||
| Human DU-145 prostate cancer cell line | [54] | ||||
| [3H] Thymidine | Several lymphoma cell lines | [55] | |||
| Trypan blue dye | Reh lymphoblastic leukemia | [56] | |||
| Dolastatin 12 | Leptolyngbya sp. | Cyclic depsipeptide | MTT | A549 lung carcinoma | [52] |
| Dragonamide | Lyngbya majuscula | Lipopeptide | Non-specified | A-549 lung epithelial adenocarcinoma, HT-29 colon adenocarcinoma and MEL-28 melanoma | [57] |
| Ethyl Tumonoate A | Oscillatoria margaritifera | Peptide | MTT | H-460 lung cancer | [58] |
| Hoiamide A | Assemblage of Lyngbya majuscule and Phormidium gracile | Cyclic depsipeptide | Non-specified | H-460 lung cancer | [59] |
| Hoiamide B | Cyanobacterial sample | Cyclic depsipeptide | Non-specified | H-460 lung cancer | [59] |
| Homodolastatin 16 | Lyngbya majuscula | Cyclic depsipeptide | MTT | WHCO1 and WHCO6 esophageal cancer and ME180 cervical cancer | [60] |
| Isomalyngamide A and A-1 | Lyngbya majuscula | Fatty acid amides | MTT | Breast cancer MCF-7 and MDA-MB-231 | [13] |
| Jamaicamides A-C | Lyngbya majuscula | Polyketide-Peptides | MTT | H-460 lung cancer | [61] |
| Kalkitoxin | Lyngbya majuscula | Lipopeptide | Trypan blue dye | HCT-116 colon | [62] |
| Lagunamide C | Lyngbya majuscula | Cyclic depsipeptide | MTT | Lung adenocarcinoma A549, cancer prostate PC3, ileocecal colorectal cancer HCT8 and ovary cancer SK-OV | [63] |
| Largazole | Symploca sp. | Cyclic depsipeptide | MTT | MDA-MB-23I breast cancer and U2OS osteosarcoma | [64] |
| A549 lung cancer and HCT-116 colorectal carcinoma | [65] | ||||
| Lyngbyabellin A | Lyngbya majuscula | Cyclic depsipeptide | Non-specified | KB nasopharyngeal carcinoma and LoVo colon adenocarcinoma | [66] |
| Lyngbyaloside | Lyngbya sp. | Glicomacrolide | Non-specified | KB nasopharyngeal carcinoma and LoVo colon adenocarcinoma | [67] |
| Majusculamide C | Lyngbya majuscula | Cyclic depsipeptide | Non-specified | Ovarian carcinoma OVCAR-3, kidney cancer A498, lung cancer NCI-H460, colorectal cancer KM20L2 and glioblastoma SF-295 | [68] |
| Malevamide D | Symploca hydnoides | Peptide ester | Non-specified | Lung cancer A-549, colon cancer HT-29 and melanoma MEL-28. | [69] |
| Malyngamide 2 | Lyngbya sordida | Fatty acid amine | MTT | H-460 lung cancer | [23] |
| Malyngamide C, J and K | Lyngbya majuscula | Fatty acid amines | MTT | H-460 lung cancer | [70] |
| Malyngolide dimmer | Lyngbya majuscula | Cyclodepside | MTT | H-460 lung cancer | [71] |
| Nostocyclopeptide A1 and A2 | Nostoc sp. | Cyclic heptapeptides | Non-specified | KB oral epidermoid cancer and LoVo colon cancer | [72] |
| Obyanamide | Lyngbya confervoides | Cyclic depsipeptide | Non-specified | KB oral epidermoid cancer and LoVo colon cancer | [73] |
| Palauamide | Lyngbya sp. | Cyclic depsipeptide | Non-specified | Cervical carcinoma HeLa, lung adenocarcinoma A549 and gastrocarcinoma BGC | [74] |
| KB oral epidermoid cancer | [75] | ||||
| Palmyramide A | Lyngbya majuscula | Cyclic depsipeptide | MTT | H-460 lung cancer | [76] |
| Pitipeptolides A-B | Lyngbya majuscula | Cyclic depsipeptides | Non-specified | LoVo colon cancer | [77] |
| MTT | HT29 colon adenocarcinoma and MCF-7 breast cancer | [17] | |||
| Pitipeptolide C | Lyngbya majuscula | Cyclic depsipeptide | MTT | HT29 colon adenocarcinoma and MCF-7 breast cancer | [17] |
| Pitiprolamide | Lyngbya majuscula | Cyclic depsipeptide | MTT | HCT116 colorectal carcinoma and MCF7 breast adenocarcinoma | [78] |
| Pseudodysidenin | Lyngbya majuscula | Lipopeptide | Non-specified | A-549 lung adenocarcinoma, HT-29 colon adenocarcinoma and MEL-28 melanoma | [57] |
| Somocystinamide A | Lyngbya majuscula | Lipopeptide | XTT | Jurkat and CEM leukemia, A549 lung carcinoma, Molt4 T leukemia, M21 melanoma and U266 myeloma | [79] |
| Symplocamide | Symploca sp. | Cyclic peptide | Non-specified | H-460 lung cancer | [27] |
| Symplostatin 1 | Symploca hydnoides | Linear Pentapeptide | SRB | MDA-MB-435 breast carcinoma and NCI/ADR ovarian carcinoma | [25] |
| Epidermoid carcinoma cell line | [80] | ||||
| Tasiamide | Symploca sp. | Cyclic peptide | Non-specified | KB oral epidermoid cancer and LoVo colon cancer | [81] |
| Tasiamide B | Symploca sp. | Peptide | Non-specified | KB oral epidermoid cancer | [82] |
| Tasipeptins A-B | Symploca sp. | Cyclic depsipeptides | Non-specified | KB oral epidermoid cancer | [83] |
| Ulongapeptin | Lyngbya sp. | Cyclic depsipeptide | Non-specified | KB oral epidermoid cancer | [84] |
| Veraguamides A-G | Symploca cf. hydnoides | Cyclic depsipeptides | MTT | H-460 lung cancer | [85] |
| Wewakazole | Lyngbya sordida | Cyclic dodecapeptide | MTT | H-460 lung cancer | [23] |
| Wewakpeptins | Lyngbya semiplena | Depsipeptides | MTT | H-460 lung cancer | [11] |
| Compound | Source | Class of compound | Model tested | Cell effect | Reference |
|---|---|---|---|---|---|
| Alotamide | Lyngbya bouillonii | Cyclic depsipeptide | Murine cerebrocortical neurons | Calcium influx promotion | [12] |
| Ankaraholide A | Geitlerinema | Glycosilated swinholide | Rat aorta A-10 cells | Loss of filamentous (F)-actin | [14] |
| Antillatoxin | Lyngbya majuscula | Lipopeptide | Primary rat cerebellar granule cells | Voltage-gated sodium channel activation | [86] |
| CHL 1610 Chinese hamster lung cells | [87] | ||||
| Antillatoxin B | Lyngbya majuscula | Lipopeptide | neuro-2a mouse neuroblastoma cells | Sodium channel activation | [10] |
| Apratoxin A | Lyngbya majuscula | Cyclic depsipeptide | Human HeLa cervical carcinoma cells | Cell cycle inhibition | [88] |
| Human U2OS osteosarcoma cells | Secretory pathway inhibition | [89] | |||
| Aurilide B | Lyngbya majuscula | Cyclic depsipeptide | Rat aorta A-10 cells | Microfilament disruption | [24] |
| Belamide A | Symploca sp. | Linear tetrapeptide | Rat aorta A-10 cells | Microtubule disruption | [8] |
| Bisebromoamide | Lyngbya sp. | Peptide | Human HeLa epithelial carcinoma cells | Actin filaments stabilization | [90] |
| Normal rat kidney cells extracellular signal regulated protein kinase | Protein kinase inhibition | [44] | |||
| Bouillomides A-B | Lyngbya bouillonii | Depsipeptides | Elastase and chymotrypsin | Serine proteases inhibition | [91] |
| Calothrixin A | Calothrix | Pentacyclic indolophenanthridine | Human leukemia CEM cells | Cell cycle inhibition | [46] |
| Calothrixin B | Calothrix | Pentacyclic indolophenanthridine | Human HeLa epithelial carcinoma cells | Cell cycle inhibition | [45] |
| Human HeLa epithelial carcinoma cells | Oxidative stress induction | [45] | |||
| Coibamide A | Leptolyngbya sp. | Cyclic depsipeptide | Human NCI-H460 lung cancer cell line | Cell cycle inhibition | [49] |
| Cryptophycin 1 | Nostoc spp. | Cyclic depsipeptide | Human MDA-MB-435 mammary adenocarcinoma and SKOV3 ovarian carcinoma cells | Cell cycle inhibition | [50] |
| Human MDA-MB-435 mammary adenocarcinoma | Caspase-3 protein activation | [50] | |||
| Curacin A | Lyngbya majuscula | Lipopeptide | Tubulin | Tubulin polymerization inhibition | [92] |
| Human A549 lung carcinoma cells | Bad protein levels increase | [52] | |||
| Human A549 lung carcinoma cells | Caspase-3 protein activation | [52] | |||
| Bovine β-tubulin | Tubulin polymerization inhibition | [93] | |||
| Dolastatin 10 | Symploca sp. | Linear Pentapeptide | Human Reh lymphoblastic leukemia cells | Bcl-2 protein reduction | [56] |
| Human lung cancer cells: NCI-H69 and -H510 | Bcl-2 protein phosphorylation | [53] | |||
| Human A549 lung carcinoma cells | Bad protein levels increase | [52] | |||
| Human A549 lung carcinoma cells | Caspase-3 protein activation | [52] | |||
| Dolastatin 12 | Leptolyngbya sp. | Cyclic depsipeptide | Rat aorta A-10 cells | Microfilament disruptor | [94] |
| Grassystatin A-B | Lyngbya confervoides | Linear depsipeptides | Cathepsins D and E | Proteases inhibition | [95] |
| Hectochlorin | Lyngbya majuscula | Lipopeptide | Human CA46 Burkitt lymphoma cells | Cell cycle inhibition | [18] |
| Hermitamides A-B | Lyngbya majuscula | Lipopeptide | Human HEK embryonic kidney cells | Voltage-gated sodium channel inhibition | [96] |
| Hoiamide A | Assemblage of Lyngbya majuscule and Phormidium gracile | Cyclic depsipeptide | Primary cultures of neocortical neurons from embryonic mice | Sodium channel activation | [59,97] |
| Hoiamide B | Cyanobacterial sample | Cyclic depsipeptide | Primary cultures of neocortical neurons from embryonic mice | Sodium influx stimulation | [59] |
| Kalkitoxin | Lyngbya majuscula | Lipopeptide | Primary rat cerebellar granule neuron cultures | Calcium influx inhibition | [98] |
| Kempopeptin A | Lyngbya sp. | Cyclic depsipeptide | Bovine pancreatic α-chymotrypsin, porcine pancreatic elastase | Serine Protease Inhibition | [99] |
| Kempopeptin B | Lyngbya sp. | Cyclic depsipeptide | Trypsin | Serine Protease Inhibition | [99] |
| Largamides A-C | Lyngbya confervoides | Cyclic depsipeptides | Porcine pancreatic elastase | Serine protease inhibition | [100] |
| Largamides D-G | Oscillatoria sp. | Cyclic depsipeptides | α-chymotrypsin | Serine protease inhibition | [101] |
| Lyngbyabellin A | Lyngbya majuscula | Cyclic depsipeptide | Human CA46 Burkitt lymphoma cells | Cell cycle inhibition | [18] |
| Rat aorta A-10 cells | Microfilament disruption | [66] | |||
| Lyngbyabellin B | Lyngbya majuscula | Cyclic depsipeptide | Human CA46 Burkitt lymphoma cells | Cell cycle inhibition | [18] |
| Lyngbyastatin 1 | Lyngbya majuscula | Cyclic depsipeptide | Rat aorta A-10 cells | Microfilament disruption | [94] |
| Lyngbyastatin 4 | Lyngbya confervoides | Cyclic depsipeptide | Bovine pancreatic α-chymotrypsin and porcine pancreatic elastase | Serine protease inhibition | [102] |
| Lyngbyastatin 5-7 | Lyngbya spp. | Cyclic depsipeptides | Porcine pancreatic elastase | Serine protease inhibition | [103] |
| Lyngbyastatin 8-10 | Lyngbya semiplena | Cyclic depsipeptides | Porcine pancreatic elastase | Serine protease inhibition | [104] |
| Malevamide E | Symploca laete-viridis | Depsipeptide | Human HEK embryonic kidney cells | Calcium influx inhibition | [105] |
| Molassamide | Dichothrix utahensis | Depsipeptide | Bovine pancreatic α-chymotrypsin and porcine pancreatic elastase | Serine protease inhibition | [106] |
| Palmyramide A | Lyngbya majuscula | Cyclic depsipeptide | Mouse neuroblastoma neuro-2a cells | Sodium channel inhibition | [76] |
| Palmyrolide | Assemblage of Leptolyngbya cf. and Oscillatoria spp. | Macrolide | Mouse neuroblastoma neuro-2a cells | Sodium influx inhibition | [107] |
| Murine cerebrocortical neurons | Inhibition of calcium oscillations | [107] | |||
| Pitipeptolides A and B | Lyngbya majuscula | Cyclic depsipeptides | Elastase | Serine protease stimulation | [77] |
| Pompanopeptin A | Lyngbya confervoides | Cyclic peptide | Porcine pancreatic trypsin | Serine protease inhibition | [108] |
| Symplocamide | Symploca sp. | Cyclic peptide | Chymotrypsin | Serine protease inhibition | [27] |
| Symplostatin 1 | Symploca hydnoides | Linear Pentapeptide | Rat aorta A-10 and human HeLa cervical carcinoma cells | Cell cycle inhibition | [25] |
| Rat aorta A-10 cells | Microtubule depolymerization | [109] | |||
| Human MDA-MB-435 breast carcinoma cells | Bcl-2 phosphorylation | [25] | |||
| Human MDA-MB-435 breast carcinoma cells | Caspase-3 protein activity stimulation | [25] | |||
| Symplostatin 3 | Symploca sp. | Rat aorta A-10 cells | Microtubule depolymerization | [110] | |
| Tiglicamides A-C | Lyngbya confervoides | Cyclic depsipeptides | Porcine pancreatic elastase | Serine protease inhibition | [111] |
2. Cell Cycle Arrest
3. Mitochondrial Dysfunctions and Oxidative Damage
4. Alterations in Caspase Cascade
5. Non-Caspases Proteases Involvement
| Compound | Source | Class of compound | Serine protease inhibition | Reference | ||
|---|---|---|---|---|---|---|
| Elastase | Chymotripsin | Thrypsin | ||||
| Bouillomide A | Lyngbya bouillonii | Depsipeptide | IC50 = 1.9 μM | IC50 = 0.17 μM | No inhibition at 100 μM | [91] |
| Bouillomide B | Lyngbya bouillonii | Depsipeptide | IC50 = 1.0 μM | IC50 = 9.3 μM | No inhibition at 100 μM | [91] |
| Kempopeptin A | Lyngbya sp. | Cyclic depsipeptide | IC50 = 0.32 μM | IC50 = 2.6 μM | IC50 > 67 μM | [99] |
| Kempopeptin B | Lyngbya sp. | Cyclic depsipeptide | IC50 > 67 μM | IC50 > 67 μM | IC50 = 8.4 μM | [99] |
| Largamide A | Lyngbya confervoides | Cyclic depsipeptide | IC50 = 1.41 μM | No inhibition at 50 μM | No inhibition at 50 μM | [100] |
| Largamide B | Lyngbya confervoides | Cyclic depsipeptide | IC50 = 0.53 μM | No inhibition at 50 μM | No inhibition at 50 μM | [100] |
| Largamide C | Lyngbya confervoides | Cyclic depsipeptide | IC50 = 1.15 μM | No inhibition at 50 μM | No inhibition at 50 μM | [100] |
| Largamide D | Oscillatoria sp. | Cyclic depsipeptide | Not described | IC50 = 10.0 μM | No inhibition | [101] |
| Largamide E | Oscillatoria sp. | Cyclic depsipeptide | Not described | IC50 = 10.0 μM | No inhibition | [101] |
| Largamide F | Oscillatoria sp. | Cyclic depsipeptide | Not described | IC50 = 4.0 μM | No inhibition | [101] |
| Largamide G | Oscillatoria sp. | Cyclic depsipeptide | Not described | IC50 = 25.0 μM | No inhibition | [101] |
| Lyngbyastatin 4 | Lyngbya confervoides | Cyclic depsipeptide | IC50 = 0.03 μM | IC50 = 0.30 μM | No inhibition at 30 μM | [102] |
| Lyngbyastatin 5 | Lyngbya spp. | Cyclic depsipeptide | IC50 = 3.2 μM | IC50 = 2.8 μM | No inhibition at 30 μM | [103] |
| Lyngbyastatin 6 | Lyngbya spp. | Cyclic depsipeptide | IC50 = 2.0 μM | IC50 = 2.5 μM | No inhibition at 30 μM | [103] |
| Lyngbyastatin 7 | Lyngbya spp. | Cyclic depsipeptide | IC50 = 3.3 μMIC50 = 0.47 μM | IC50 = 2.5 μM | No inhibition at 30 μM | [103,104] |
| Lyngbyastatin 8 | Lyngbya semiplena | Cyclic depsipeptide | IC50 = 0.12 μM | Not described | Not described | [104] |
| Lyngbyastatin 9 | Lyngbya semiplena | Cyclic depsipeptide | IC50 = 0.21 μM | Not described | Not described | [104] |
| Lyngbyastatin 10 | Lyngbya semiplena | Cyclic depsipeptide | IC50 = 0.12 μM | Not described | Not described | [104] |
| Molassamide | Dichothrix utahensis | Depsipeptide | IC50 = 0.032 μM | IC50 = 0.234 μM | No inhibition at 10 μM | [106] |
| Pompanopeptin A | Lyngbya confervoides | Cyclic peptide | Not described | Not described | IC50 = 2.4 μM | [108] |
| Symplocamide A | Symploca sp. | Cyclic peptide | Not described | IC50 = 0.38 μM | IC50 = 80.2 μM | [27] |
| Somamide B | Lyngbya majuscula and Schizothrix assemblage | Depsipeptide | IC50 = 9.5 μM | IC50 = 4.2 μM | No inhibition at 30 μM | [103] |
| Tiglicamide A | Lyngbya confervoides | Cyclic depsipeptide | IC50 = 2.14 μM | Not described | Not described | [111] |
| Tiglicamide B | Lyngbya confervoides | Cyclic depsipeptide | IC50 = 6.99 μM | Not described | Not described | [111] |
| Tiglicamide C | Lyngbya confervoides | Cyclic depsipeptide | IC50 = 7.28 μM | Not described | Not described | [111] |

6. Alterations in the Bcl-2 Protein Family
7. Alterations in Membrane Sodium Channel Dynamics
8. Conclusions
Acknowledgements
References
- Tsygankov, A.A. Nitrogen-Fixing cyanobacteria: Producents of hydrogen. Prikl. Biokhim. Mikrobiol. 2007, 43, 279–288. [Google Scholar]
- Carmichael, W.W. Cyanobacteria secondary metabolites—the cyanotoxins. J. Appl. Bacteriol. 1992, 72, 445–459. [Google Scholar]
- Codd, G.A.; Morrison, L.F.; Metcalf, J.S. Cyanobacterial toxins: Risk management for health protection. Toxicol. Appl. Pharmacol. 2005, 203, 264–272. [Google Scholar]
- Blaha, L.; Babica, P.; Marsalek, B. Toxins produced in cyanobacterial water blooms—toxicity and risks. Interdiscip. Toxicol. 2009, 2, 36–41. [Google Scholar]
- Singh, R.K.; Tiwari, S.P.; Rai, A.K.; Mohapatra, T.M. Cyanobacteria: An emerging source for drug discovery. J. Antibiot. (Tokyo) 2011, 64, 401–412. [Google Scholar] [CrossRef]
- Nunnery, J.K.; Mevers, E.; Gerwick, W.H. Biologically active secondary metabolites from marine cyanobacteria. Curr. Opin. Biotechnol. 2010, 21, 787–793. [Google Scholar]
- Jones, A.C.; Monroe, E.A.; Eisman, E.B.; Gerwick, L.; Sherman, D.H.; Gerwick, W.H. The unique mechanistic transformations involved in the biosynthesis of modular natural products from marine cyanobacteria. Nat. Prod. Rep. 2010, 27, 1048–1065. [Google Scholar]
- Simmons, T.L.; McPhail, K.L.; Ortega-Barria, E.; Mooberry, S.L.; Gerwick, W.H. Belamide A, a new antimitotic tetrapeptide from a Panamanian marine cyanobacterium. Tetrahedron Lett. 2006, 47, 3387–3390. [Google Scholar]
- Sisay, M.T.; Hautmann, S.; Mehner, C.; Konig, G.M.; Bajorath, J.; Gutschow, M. Inhibition of human leukocyte elastase by brunsvicamides A–C: Cyanobacterial cyclic peptides. ChemMedChem 2009, 4, 1425–1429. [Google Scholar] [CrossRef]
- Nogle, L.M.; Okino, T.; Gerwick, W.H. Antillatoxin B, a neurotoxic lipopeptide from the marine cyanobacterium Lyngbya majuscula. J. Nat. Prod. 2001, 64, 983–985. [Google Scholar] [CrossRef]
- Han, B.; Goeger, D.; Maier, C.S.; Gerwick, W.H. The wewakpeptins, cyclic depsipeptides from a Papua New Guinea collection of the marine cyanobacterium Lyngbya semiplena. J. Org. Chem. 2005, 70, 3133–3139. [Google Scholar] [CrossRef]
- Soria-Mercado, I.E.; Pereira, A.; Cao, Z.; Murray, T.F.; Gerwick, W.H. Alotamide A, a novel neuropharmacological agent from the marine cyanobacterium Lyngbya bouillonii. Org. Lett. 2009, 11, 4704–4707. [Google Scholar] [CrossRef]
- Chang, T.T.; More, S.V.; Lu, I.H.; Hsu, J.C.; Chen, T.J.; Jen, Y.C.; Lu, C.K.; Li, W.S. Isomalyngamide A, A-1 and their analogs suppress cancer cell migration in vitro. Eur. J. Med. Chem. 2011, 46, 3810–3819. [Google Scholar] [CrossRef]
- Andrianasolo, E.H.; Gross, H.; Goeger, D.; Musafija-Girt, M.; McPhail, K.; Leal, R.M.; Mooberry, S.L.; Gerwick, W.H. Isolation of swinholide A and related glycosylated derivatives from two field collections of marine cyanobacteria. Org. Lett. 2005, 7, 1375–1378. [Google Scholar]
- Teruya, T.; Sasaki, H.; Kitamura, K.; Nakayama, T.; Suenaga, K. Biselyngbyaside, a macrolide glycoside from the marine cyanobacterium Lyngbya sp. Org. Lett. 2009, 11, 2421–2424. [Google Scholar] [CrossRef]
- Salvador, L.A.; Paul, V.J.; Luesch, H. Caylobolide B, a macrolactone from symplostatin 1-producing marine cyanobacteria Phormidium spp. from Florida. J. Nat. Prod. 2010, 73, 1606–1609. [Google Scholar] [CrossRef]
- Montaser, R.; Paul, V.J.; Luesch, H. Pitipeptolides C–F, antimycobacterial cyclodepsipeptides from the marine cyanobacterium Lyngbya majuscula from Guam. Phytochemistry 2011, 72, 2068–2074. [Google Scholar] [CrossRef]
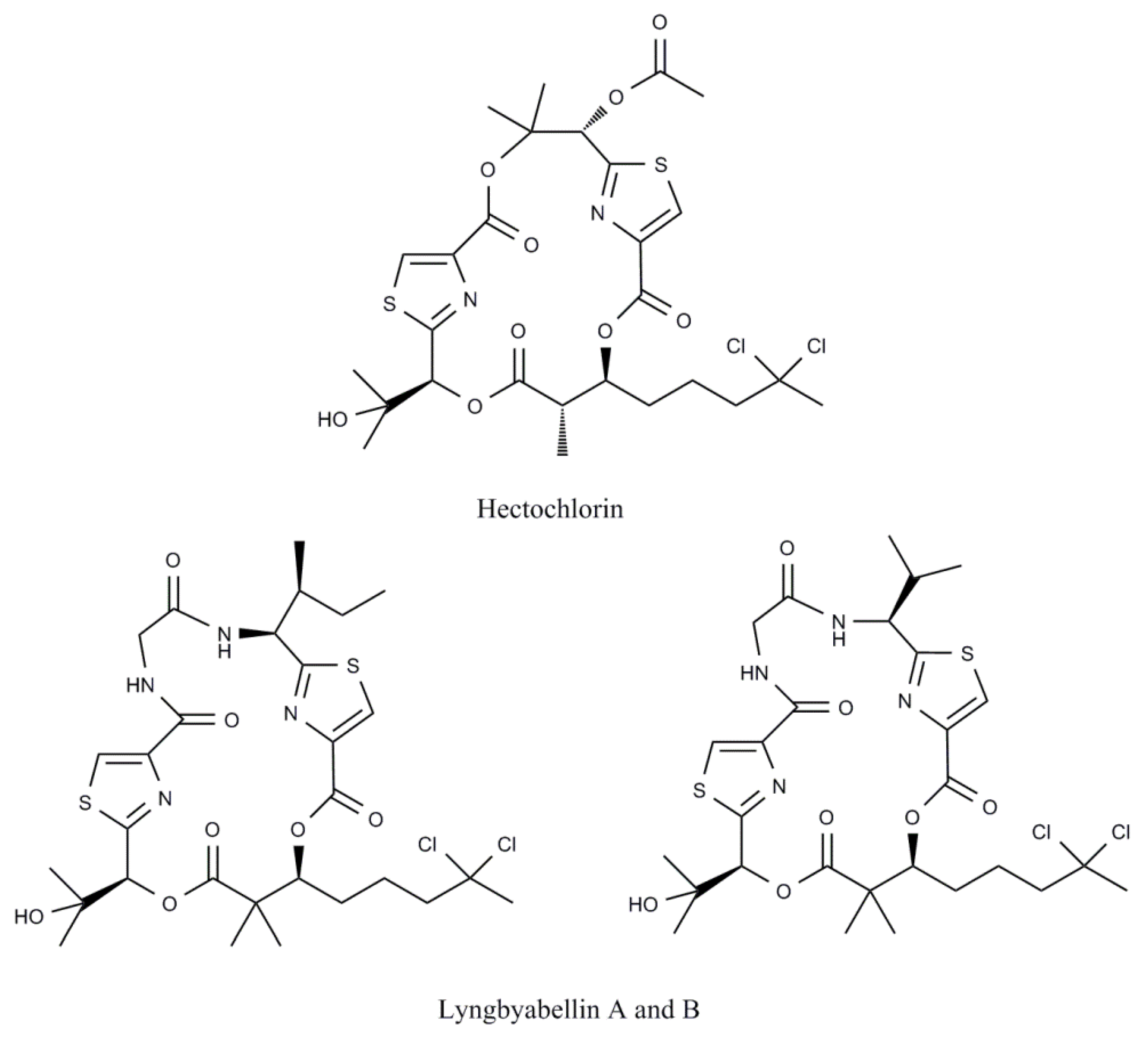
- Marquez, B.L.; Watts, K.S.; Yokochi, A.; Roberts, M.A.; Verdier-Pinard, P.; Jimenez, J.I.; Hamel, E.; Scheuer, P.J.; Gerwick, W.H. Structure and absolute stereochemistry of hectochlorin, a potent stimulator of actin assembly. J. Nat. Prod. 2002, 65, 866–871. [Google Scholar] [CrossRef]
- Milligan, K.E.; Marquez, B.L.; Williamson, R.T.; Gerwick, W.H. Lyngbyabellin B, a toxic and antifungal secondary metabolite from the marine cyanobacterium Lyngbya majuscula. J. Nat. Prod. 2000, 63, 1440–1443. [Google Scholar] [CrossRef]
- Tripathi, A.; Puddick, J.; Prinsep, M.R.; Rottmann, M.; Tan, L.T. Lagunamides A and B: Cytotoxic and antimalarial cyclodepsipeptides from the marine cyanobacterium Lyngbya majuscula. J. Nat. Prod. 2010, 73, 1810–1814. [Google Scholar] [CrossRef]
- Capper, A.; Cruz-Rivera, E.; Paul, V.; Tibbetts, I. Chemical deterrence of a marine cyanobacterium against sympatric and non-sympatric consumers. Hydrobiologia 2006, 553, 319–326. [Google Scholar]
- Tan, L.T.; Goh, B.P. Chemical ecology of marine cyanobacterial secondary metabolites: A mini-review. J. Coast. Dev. 2009, 13, 1–9. [Google Scholar]
- Malloy, K.L.; Villa, F.A.; Engene, N.; Matainaho, T.; Gerwick, L.; Gerwick, W.H. Malyngamide 2, an oxidized lipopeptide with nitric oxide inhibiting activity from a Papua New Guinea marine cyanobacterium. J. Nat. Prod. 2011, 74, 95–98. [Google Scholar]
- Han, B.; Gross, H.; Goeger, D.E.; Mooberry, S.L.; Gerwick, W.H. Aurilides B and C, cancer cell toxins from a Papua New Guinea collection of the marine cyanobacterium Lyngbya majuscula. J. Nat. Prod. 2006, 69, 572–575. [Google Scholar] [CrossRef]
- Mooberry, S.L.; Leal, R.M.; Tinley, T.L.; Luesch, H.; Moore, R.E.; Corbett, T.H. The molecular pharmacology of symplostatin 1: A new antimitotic dolastatin 10 analog. Int. J. Cancer 2003, 104, 512–521. [Google Scholar]
- Gutierrez, M.; Suyama, T.L.; Engene, N.; Wingerd, J.S.; Matainaho, T.; Gerwick, W.H. Apratoxin D, a potent cytotoxic cyclodepsipeptide from papua new guinea collections of the marine cyanobacteria Lyngbya majuscula and Lyngbya sordida. J. Nat. Prod. 2008, 71, 1099–1103. [Google Scholar] [CrossRef]
- Linington, R.G.; Edwards, D.J.; Shuman, C.F.; McPhail, K.L.; Matainaho, T.; Gerwick, W.H. Symplocamide A, a potent cytotoxin and chymotrypsin inhibitor from the marine cyanobacterium Symploca sp. J. Nat. Prod. 2008, 71, 22–27. [Google Scholar] [CrossRef]
- Ulukaya, E.; Acilan, C.; Yilmaz, Y. Apoptosis: Why and how does it occur in biology? Cell Biochem. Funct. 2011, 29, 468–480. [Google Scholar] [CrossRef]
- Han, S.I.; Kim, Y.S.; Kim, T.H. Role of apoptotic and necrotic cell death under physiologic conditions. BMB Rep. 2008, 41, 1–10. [Google Scholar]
- Kim, M.K.; Suh, D.H.; Seoung, J.; Kim, H.S.; Chung, H.H.; Song, Y.S. Autophagy as a target for anticancer therapy and its modulation by phytochemicals. J. Food Drug Anal. 2012, 20, 241–245. [Google Scholar]
- Zhang, J.Y. Apoptosis-Based anticancer drugs. Nat. Rev. Drug Discov. 2002, 1, 101–102. [Google Scholar]
- Fischer, U.; Schulze-Osthoff, K. Apoptosis-Based therapies and drug targets. Cell Death Differ. 2005, 12, 942–961. [Google Scholar]
- Chen, D.; Daniel, K.G.; Chen, M.S.; Kuhn, D.J.; Landis-Piwowar, K.R.; Dou, Q.P. Dietary flavonoids as proteasome inhibitors and apoptosis inducers in human leukemia cells. Biochem. Pharmacol. 2005, 69, 1421–1432. [Google Scholar]
- Mao, Y.B.; Song, G.; Cai, Q.F.; Liu, M.; Luo, H.H.; Shi, M.X.; Ouyang, G.; Bao, S.D. Hydrogen peroxide-induced apoptosis in human gastric carcinoma MGC803 cells. Cell Biol. Int. 2006, 30, 332–337. [Google Scholar]
- Lytvyn, D.I.; Yemets, A.I.; Blume, Y.B. UV-B overexposure induces programmed cell death in a BY-2 tobacco cell line. Environ. Exp. Bot. 2010, 68, 51–57. [Google Scholar]
- Elmore, S. Apoptosis: A review of programmed cell death. Toxicol. Pathol. 2007, 35, 495–516. [Google Scholar]
- Martins, R.F.; Ramos, M.F.; Herfindal, L.; Sousa, J.A.; Skaerven, K.; Vasconcelos, V.M. Antimicrobial and cytotoxic assessment of marine cyanobacteria—Synechocystis and Synechococcus. Mar. Drugs 2008, 6, 1–11. [Google Scholar]
- Yonezawa, T.; Mase, N.; Sasaki, H.; Teruya, T.; Hasegawa, S.; Cha, B.Y.; Yagasaki, K.; Suenaga, K.; Nagai, K.; Woo, J.T. Biselyngbyaside, isolated from marine cyanobacteria, inhibits osteoclastogenesis and induces apoptosis in mature osteoclasts. J. Cell Biochem. 2012, 113, 440–448. [Google Scholar] [CrossRef]
- Oftedal, L.; Selheim, F.; Wahlsten, M.; Sivonen, K.; Doskeland, S.O.; Herfindal, L. Marine benthic cyanobacteria contain apoptosis-inducing activity synergizing with daunorubicin to kill leukemia cells, but not cardiomyocytes. Mar. Drugs 2010, 8, 2659–2672. [Google Scholar]
- Luesch, H.; Yoshida, W.Y.; Moore, R.E.; Paul, V.J. New apratoxins of marine cyanobacterial origin from Guam and Palau. Bioorg. Med. Chem. 2002, 10, 1973–1978. [Google Scholar]
- Luesch, H.; Yoshida, W.Y.; Moore, R.E.; Paul, V.J.; Corbett, T.H. Total structure determination of apratoxin A, a potent novel cytotoxin from the marine cyanobacterium Lyngbya majuscula. J. Am. Chem. Soc. 2001, 123, 5418–5423. [Google Scholar]
- Matthew, S.; Schupp, P.J.; Luesch, H. Apratoxin E, a cytotoxic peptolide from a guamanian collection of the marine cyanobacterium Lyngbya bouillonii. J. Nat. Prod. 2008, 71, 1113–1116. [Google Scholar] [CrossRef]
- Tidgewell, K.; Engene, N.; Byrum, T.; Media, J.; Doi, T.; Valeriote, F.A.; Gerwick, W.H. Evolved diversification of a modular natural product pathway: Apratoxins F and G, two cytotoxic cyclic depsipeptides from a Palmyra collection of Lyngbya bouillonii. ChemBioChem 2010, 11, 1458–1466. [Google Scholar] [CrossRef]
- Teruya, T.; Sasaki, H.; Fukazawa, H.; Suenaga, K. Bisebromoamide, a potent cytotoxic peptide from the marine cyanobacterium Lyngbya sp.: isolation, stereostructure, and biological activity. Org. Lett. 2009, 11, 5062–5065. [Google Scholar]
- Chen, X.X.; Smith, G.D.; Waring, P. Human cancer cell (Jurkat) killing by the cyanobacterial metabolite calothrixin A. J. Appl. Phycol. 2003, 15, 269–277. [Google Scholar]
- Khan, Q.A.; Lu, J.; Hecht, S.M. Calothrixins, a new class of human DNA topoisomerase I poisons. J. Nat. Prod. 2009, 72, 438–442. [Google Scholar]
- Bernardo, P.H.; Chai, C.L.; Le Guen, M.; Smith, G.D.; Waring, P. Structure-Activity delineation of quinones related to the biologically active Calothrixin B. Bioorg. Med. Chem. Lett. 2007, 17, 82–85. [Google Scholar]
- MacMillan, J.B.; Molinski, T.F. Caylobolide A, a unique 36-membered macrolactone from a Bahamian Lyngbya majuscula. Org. Lett. 2002, 4, 1535–1538. [Google Scholar] [CrossRef]
- Medina, R.A.; Goeger, D.E.; Hills, P.; Mooberry, S.L.; Huang, N.; Romero, L.I.; Ortega-Barria, E.; Gerwick, W.H.; McPhail, K.L. Coibamide A, a potent antiproliferative cyclic depsipeptide from the Panamanian marine cyanobacterium Leptolyngbya sp. J. Am. Chem. Soc. 2008, 130, 6324–6325. [Google Scholar]
- Mooberry, S.L.; Busquets, L.; Tien, G. Induction of apoptosis by cryptophycin 1, a new antimicrotubule agent. Int. J. Cancer 1997, 73, 440–448. [Google Scholar] [CrossRef]
- Wagner, M.M.; Paul, D.C.; Shih, C.; Jordan, M.A.; Wilson, L.; Williams, D.C. In vitro pharmacology of cryptophycin 52 (LY355703) in human tumor cell lines. Cancer Chemother. Pharmacol. 1999, 43, 115–125. [Google Scholar] [CrossRef]
- Catassi, A.; Cesario, A.; Arzani, D.; Menichini, P.; Alama, A.; Bruzzo, C.; Imperatori, A.; Rotolo, N.; Granone, P.; Russo, P. Characterization of apoptosis induced by marine natural products in non small cell lung cancer A549 cells. Cell. Mol. Life Sci. 2006, 63, 2377–2386. [Google Scholar] [CrossRef]
- Kalemkerian, G.P.; Ou, X.L.; Adil, M.R.; Rosati, R.; Khoulani, M.M.; Madan, S.K.; Pettit, G.R. Activity of dolastatin 10 against small-cell lung cancer in vitro and in vivo: Induction of apoptosis and bcl-2 modification. Cancer Chemother. Pharm. 1999, 43, 507–515. [Google Scholar] [CrossRef]
- Turner, T.; Jackson, W.H.; Pettit, G.R.; Wells, A.; Kraft, A.S. Treatment of human prostate cancer cells with dolastatin 10, a peptide isolated from a marine shell-less mollusc. Prostate 1998, 34, 175–181. [Google Scholar] [CrossRef]
- Beckwith, M.; Urba, W.J.; Longo, D.L. Growth inhibition of human lymphoma cell lines by the marine products, dolastatins 10 and 15. J. Natl. Cancer Inst. 1993, 85, 483–488. [Google Scholar] [CrossRef]
- Wall, N.R.; Mohammad, R.M.; Al-Katib, A.M. Bax:Bcl-2 ratio modulation by bryostatin 1 and novel antitubulin agents is important for susceptibility to drug induced apoptosis in the human early pre-B acute lymphoblastic leukemia cell line, Reh. Leuk. Res. 1999, 23, 881–888. [Google Scholar] [CrossRef]
- Jimenez, J.I.; Scheuer, P.J. New lipopeptides from the Caribbean cyanobacterium Lyngbya majuscula. J. Nat. Prod. 2001, 64, 200–203. [Google Scholar] [CrossRef]
- Engene, N.; Choi, H.; Esquenazi, E.; Byrum, T.; Villa, F.A.; Cao, Z.; Murray, T.F.; Dorrestein, P.C.; Gerwick, L.; Gerwick, W.H. Phylogeny-Guided isolation of ethyl tumonoate A from the marine cyanobacterium cf. Oscillatoria margaritifera. J. Nat. Prod. 2011, 74, 1737–1743. [Google Scholar] [CrossRef]
- Choi, H.; Pereira, A.R.; Cao, Z.; Shuman, C.F.; Engene, N.; Byrum, T.; Matainaho, T.; Murray, T.F.; Mangoni, A.; Gerwick, W.H. The hoiamides, structurally intriguing neurotoxic lipopeptides from Papua New Guinea marine cyanobacteria. J. Nat. Prod. 2010, 73, 1411–1421. [Google Scholar] [CrossRef]
- Davies-Coleman, M.T.; Dzeha, T.M.; Gray, C.A.; Hess, S.; Pannell, L.K.; Hendricks, D.T.; Arendse, C.E. Isolation of homodolastatin 16, a new cyclic depsipeptide from a Kenyan collection of Lyngbya majuscula. J. Nat. Prod. 2003, 66, 712–715. [Google Scholar] [CrossRef]
- Edwards, D.J.; Marquez, B.L.; Nogle, L.M.; McPhail, K.; Goeger, D.E.; Roberts, M.A.; Gerwick, W.H. Structure and biosynthesis of the jamaicamides, new mixed polyketide-peptide neurotoxins from the marine cyanobacterium Lyngbya majuscula. Chem. Biol. 2004, 11, 817–833. [Google Scholar] [CrossRef]
- White, J.D.; Xu, Q.; Lee, C.S.; Valeriote, F.A. Total synthesis and biological evaluation of (+)-kalkitoxin, a cytotoxic metabolite of the cyanobacterium Lyngbya majuscula. Org. Biomol. Chem. 2004, 2, 2092–2102. [Google Scholar] [CrossRef]
- Tripathi, A.; Puddick, J.; Prinsep, M.R.; Rottmann, M.; Chan, K.P.; Chen, D.Y.; Tan, L.T. Lagunamide C, a cytotoxic cyclodepsipeptide from the marine cyanobacterium Lyngbya majuscula. Phytochemistry 2011, 72, 2369–2375. [Google Scholar] [CrossRef]
- Taori, K.; Paul, V.J.; Luesch, H. Structure and activity of largazole, a potent antiproliferative agent from the Floridian marine cyanobacterium Symploca sp. J. Am. Chem. Soc. 2008, 130, 1806–1807. [Google Scholar] [CrossRef]
- Zeng, X.; Yin, B.; Hu, Z.; Liao, C.; Liu, J.; Li, S.; Li, Z.; Nicklaus, M.C.; Zhou, G.; Jiang, S. Total synthesis and biological evaluation of largazole and derivatives with promising selectivity for cancers cells. Org. Lett. 2010, 12, 1368–1371. [Google Scholar] [CrossRef]
- Luesch, H.; Yoshida, W.Y.; Moore, R.E.; Paul, V.J.; Mooberry, S.L. Isolation, structure determination, and biological activity of Lyngbyabellin A from the marine cyanobacterium Lyngbya majuscula. J. Nat. Prod. 2000, 63, 611–615. [Google Scholar] [CrossRef]
- Luesch, H.; Yoshida, W.Y.; Harrigan, G.G.; Doom, J.P.; Moore, R.E.; Paul, V.J. Lyngbyaloside B, a new glycoside macrolide from a Palauan marine cyanobacterium, Lyngbya sp. J. Nat. Prod. 2002, 65, 1945–1948. [Google Scholar] [CrossRef]
- Pettit, G.R.; Hogan, F.; Xu, J.P.; Tan, R.; Nogawa, T.; Cichacz, Z.; Pettit, R.K.; Du, J.; Ye, Q.H.; Cragg, G.M.; et al. Antineoplastic agents. 536. New sources of naturally occurring cancer cell growth inhibitors from marine organisms, terrestrial plants, and microorganisms (1a,). J. Nat. Prod. 2008, 71, 438–444. [Google Scholar] [CrossRef]
- Horgen, F.D.; Kazmierski, E.B.; Westenburg, H.E.; Yoshida, W.Y.; Scheuer, P.J. Malevamide D: Isolation and structure determination of an isodolastatin H analogue from the marine cyanobacterium Symploca hydnoides. J. Nat. Prod. 2002, 65, 487–491. [Google Scholar] [CrossRef]
- Gross, H.; McPhail, K.L.; Goeger, D.E.; Valeriote, F.A.; Gerwick, W.H. Two cytotoxic stereoisomers of malyngamide C, 8-epi-malyngamide C and 8-O-acetyl-8-epi-malyngamide C, from the marine cyanobacterium Lyngbya majuscula. Phytochemistry 2010, 71, 1729–1735. [Google Scholar] [CrossRef]
- Gutierrez, M.; Tidgewell, K.; Capson, T.L.; Engene, N.; Almanza, A.; Schemies, J.; Jung, M.; Gerwick, W.H. Malyngolide dimer, a bioactive symmetric cyclodepside from the panamanian marine cyanobacterium Lyngbya majuscula. J. Nat. Prod. 2010, 73, 709–711. [Google Scholar] [CrossRef]
- Golakoti, T.; Yoshida, W.Y.; Chaganty, S.; Moore, R.E. Isolation and structure determination of nostocyclopeptides A1 and A2 from the terrestrial cyanobacterium Nostoc sp. ATCC53789. J. Nat. Prod. 2001, 64, 54–59. [Google Scholar] [CrossRef]
- Williams, P.G.; Yoshida, W.Y.; Moore, R.E.; Paul, V.J. Isolation and structure determination of obyanamide, a novel cytotoxic cyclic depsipeptide from the marine cyanobacterium Lyngbya confervoides. J. Nat. Prod. 2002, 65, 29–31. [Google Scholar] [CrossRef]
- Zou, B.; Long, K.; Ma, D.W. Total synthesis and cytotoxicity studies of a cyclic depsipeptide with proposed structure of palau’amide. Org. Lett. 2005, 7, 4237–4240. [Google Scholar]
- Williams, P.G.; Yoshida, W.Y.; Quon, M.K.; Moore, R.E.; Paul, V.J. The structure of Palau’amide, a potent cytotoxin from a species of the marine cyanobacterium Lyngbya. J. Nat. Prod. 2003, 66, 1545–1549. [Google Scholar] [CrossRef]
- Taniguchi, M.; Nunnery, J.K.; Engene, N.; Esquenazi, E.; Byrum, T.; Dorrestein, P.C.; Gerwick, W.H. Palmyramide A, a cyclic depsipeptide from a Palmyra Atoll collection of the marine cyanobacterium Lyngbya majuscula. J. Nat. Prod. 2010, 73, 393–398. [Google Scholar] [CrossRef]
- Luesch, H.; Pangilinan, R.; Yoshida, W.Y.; Moore, R.E.; Paul, V.J. Pitipeptolides A and B, new cyclodepsipeptides from the marine cyanobacterium Lyngbya majuscula. J. Nat. Prod. 2001, 64, 304–307. [Google Scholar] [CrossRef]
- Montaser, R.; Abboud, K.A.; Paul, V.J.; Luesch, H. Pitiprolamide, a proline-rich dolastatin 16 analogue from the marine cyanobacterium Lyngbya majuscula from Guam. J. Nat. Prod. 2011, 74, 109–112. [Google Scholar] [CrossRef]
- Wrasidlo, W.; Mielgo, A.; Torres, V.A.; Barbero, S.; Stoletov, K.; Suyama, T.L.; Klemke, R.L.; Gerwick, W.H.; Carson, D.A.; Stupack, D.G. The marine lipopeptide somocystinamide A triggers apoptosis via caspase 8. Proc. Natl. Acad. Sci. USA 2008, 105, 2313–2318. [Google Scholar]
- Harrigan, G.G.; Luesch, H.; Yoshida, W.Y.; Moore, R.E.; Nagle, D.G.; Paul, V.J.; Mooberry, S.L.; Corbett, T.H.; Valeriote, F.A. Symplostatin 1: A dolastatin 10 analogue from the marine cyanobacterium Symploca hydnoides. J. Nat. Prod. 1998, 61, 1075–1077. [Google Scholar] [CrossRef]
- Williams, P.G.; Yoshida, W.Y.; Moore, R.E.; Paul, V.J. Tasiamide, a cytotoxic peptide from the marine cyanobacterium Symploca sp. J. Nat. Prod. 2002, 65, 1336–1339. [Google Scholar] [CrossRef]
- Williams, P.G.; Yoshida, W.Y.; Moore, R.E.; Paul, V.J. The isolation and structure elucidation of Tasiamide B, a 4-amino-3-hydroxy-5-phenylpentanoic acid containing peptide from the marine Cyanobacterium Symploca sp. J. Nat. Prod. 2003, 66, 1006–1009. [Google Scholar] [CrossRef]
- Williams, P.G.; Yoshida, W.Y.; Moore, R.E.; Paul, V.J. Tasipeptins A and B: New cytotoxic depsipeptides from the marine cyanobacterium Symploca sp. J. Nat. Prod. 2003, 66, 620–624. [Google Scholar] [CrossRef]
- Williams, P.G.; Yoshida, W.Y.; Quon, M.K.; Moore, R.E.; Paul, V.J. Ulongapeptin, a cytotoxic cyclic depsipeptide from a Palauan marine cyanobacterium Lyngbya sp. J. Nat. Prod. 2003, 66, 651–654. [Google Scholar] [CrossRef]
- Mevers, E.; Liu, W.T.; Engene, N.; Mohimani, H.; Byrum, T.; Pevzner, P.A.; Dorrestein, P.C.; Spadafora, C.; Gerwick, W.H. Cytotoxic veraguamides, alkynyl bromide-containing cyclic depsipeptides from the marine cyanobacterium cf. Oscillatoria margaritifera. J. Nat. Prod. 2011, 74, 928–936. [Google Scholar] [CrossRef]
- Li, W.I.; Berman, F.W.; Okino, T.; Yokokawa, F.; Shioiri, T.; Gerwick, W.H.; Murray, T.F. Antillatoxin is a marine cyanobacterial toxin that potently activates voltage-gated sodium channels. Proc. Natl. Acad. Sci. USA 2001, 98, 7599–7604. [Google Scholar] [CrossRef]
- Cao, Z.; Gerwick, W.H.; Murray, T.F. Antillatoxin is a sodium channel activator that displays unique efficacy in heterologously expressed rNav1.2, rNav1.4 and rNav1.5 alpha subunits. BMC Neurosci. 2010, 11, 154. [Google Scholar] [CrossRef]
- Ma, D.; Zou, B.; Cai, G.; Hu, X.; Liu, J.O. Total synthesis of the cyclodepsipeptide apratoxin A and its analogues and assessment of their biological activities. Chemistry 2006, 12, 7615–7626. [Google Scholar] [CrossRef]
- Liu, Y.; Law, B.K.; Luesch, H. Apratoxin a reversibly inhibits the secretory pathway by preventing cotranslational translocation. Mol. Pharmacol. 2009, 76, 91–104. [Google Scholar] [CrossRef]
- Sumiya, E.; Shimogawa, H.; Sasaki, H.; Tsutsumi, M.; Yoshita, K.; Ojika, M.; Suenaga, K.; Uesugi, M. Cell-Morphology profiling of a natural product library identifies bisebromoamide and miuraenamide A as actin filament stabilizers. ACS Chem. Biol. 2011, 6, 425–431. [Google Scholar] [CrossRef]
- Rubio, B.K.; Parrish, S.M.; Yoshida, W.; Schupp, P.J.; Schils, T.; Williams, P.G. Depsipeptides from a Guamanian Marine Cyanobacterium, Lyngbya bouillonii, with selective inhibition of serine proteases. Tetrahedron Lett. 2010, 51, 6718–6721. [Google Scholar] [CrossRef]
- Gerwick, W.H.; Proteau, P.J.; Nagle, D.G.; Hamel, E.; Blokhin, A.V.; Slate, D.L. Structure of Curacin A, a novel antimitotic, antiproliferative, and brine shrimp toxic natural product from the Marine Cyanobacterium Lyngbya majuscula. J. Org. Chem. 1994, 59, 1243–1245. [Google Scholar] [CrossRef]
- Mitra, A.; Sept, D. Localization of the antimitotic peptide and depsipeptide binding site on beta-tubulin. Biochemistry 2004, 43, 13955–13962. [Google Scholar] [CrossRef]
- Harrigan, G.G.; Yoshida, W.Y.; Moore, R.E.; Nagle, D.G.; Park, P.U.; Biggs, J.; Paul, V.J.; Mooberry, S.L.; Corbett, T.H.; Valeriote, F.A. Isolation, structure determination, and biological activity of dolastatin 12 and lyngbyastatin 1 from Lyngbya majuscula/Schizothrix calcicola cyanobacterial assemblages. J. Nat. Prod. 1998, 61, 1221–1225. [Google Scholar] [CrossRef]
- Kwan, J.C.; Eksioglu, E.A.; Liu, C.; Paul, V.J.; Luesch, H. Grassystatins A–C from marine cyanobacteria, potent cathepsin E inhibitors that reduce antigen presentation. J. Med. Chem. 2009, 52, 5732–5747. [Google Scholar]
- De Oliveira, E.O.; Graf, K.M.; Patel, M.K.; Baheti, A.; Kong, H.S.; MacArthur, L.H.; Dakshanamurthy, S.; Wang, K.; Brown, M.L.; Paige, M. Synthesis and evaluation of hermitamides A and B as human voltage-gated sodium channel blockers. Bioorg. Med. Chem. 2011, 19, 4322–4329. [Google Scholar]
- Pereira, A.; Cao, Z.; Murray, T.F.; Gerwick, W.H. Hoiamide a, a sodium channel activator of unusual architecture from a consortium of two papua new Guinea cyanobacteria. Chem. Biol. 2009, 16, 893–906. [Google Scholar]
- LePage, K.T.; Goeger, D.; Yokokawa, F.; Asano, T.; Shioiri, T.; Gerwick, W.H.; Murray, T.F. The neurotoxic lipopeptide kalkitoxin interacts with voltage-sensitive sodium channels in cerebellar granule neurons. Toxicol. Lett. 2005, 158, 133–139. [Google Scholar]
- Taori, K.; Paul, V.J.; Luesch, H. Kempopeptins A and B, serine protease inhibitors with different selectivity profiles from a marine cyanobacterium, Lyngbya sp. J. Nat. Prod. 2008, 71, 1625–1629. [Google Scholar] [CrossRef]
- Matthew, S.; Paul, V.J.; Luesch, H. Largamides A–C, tiglic acid-containing cyclodepsipeptides with elastase-inhibitory activity from the marine cyanobacterium Lyngbya confervoides. Planta Med. 2009, 75, 528–533. [Google Scholar] [CrossRef]
- Plaza, A.; Bewley, C.A. Largamides A–H, unusual cyclic peptides from the marine cyanobacterium Oscillatoria sp. J. Org. Chem. 2006, 71, 6898–6907. [Google Scholar] [CrossRef]
- Matthew, S.; Ross, C.; Rocca, J.R.; Paul, V.J.; Luesch, H. Lyngbyastatin 4, a dolastatin 13 analogue with elastase and chymotrypsin inhibitory activity from the marine cyanobacterium Lyngbya confervoides. J. Nat. Prod. 2007, 70, 124–127. [Google Scholar] [CrossRef]
- Taori, K.; Matthew, S.; Rocca, J.R.; Paul, V.J.; Luesch, H. Lyngbyastatins 5–7, potent elastase inhibitors from Floridian marine cyanobacteria, Lyngbya sp. J. Nat. Prod. 2007, 70, 1593–1600. [Google Scholar] [CrossRef]
- Kwan, J.C.; Taori, K.; Paul, V.J.; Luesch, H. Lyngbyastatins 8–10, elastase inhibitors with cyclic depsipeptide scaffolds isolated from the marine cyanobacterium Lyngbya semiplena. Mar. Drugs 2009, 7, 528–538. [Google Scholar] [CrossRef]
- Adams, B.; Porzgen, P.; Pittman, E.; Yoshida, W.Y.; Westenburg, H.E.; Horgen, F.D. Isolation and structure determination of malevamide E, a dolastatin 14 analogue, from the marine cyanobacterium Symploca laete-viridis. J. Nat. Prod. 2008, 71, 750–754. [Google Scholar] [CrossRef]
- Gunasekera, S.P.; Miller, M.W.; Kwan, J.C.; Luesch, H.; Paul, V.J. Molassamide, a depsipeptide serine protease inhibitor from the marine cyanobacterium Dichothrix utahensis. J. Nat. Prod. 2010, 73, 459–462. [Google Scholar] [CrossRef]
- Pereira, A.R.; Cao, Z.; Engene, N.; Soria-Mercado, I.E.; Murray, T.F.; Gerwick, W.H. Palmyrolide A, an unusually stabilized neuroactive macrolide from Palmyra Atoll cyanobacteria. Org. Lett. 2010, 12, 4490–4493. [Google Scholar]
- Matthew, S.; Ross, C.; Paul, V.J.; Luesch, H. Pompanopeptins A and B, new cyclic peptides from the marine cyanobacterium Lyngbya confervoides. Tetrahedron 2008, 64, 4081–4089. [Google Scholar] [CrossRef]
- Luesch, H.; Moore, R.E.; Paul, V.J.; Mooberry, S.L.; Corbett, T.H. Isolation of dolastatin 10 from the marine cyanobacterium Symploca species VP642 and total stereochemistry and biological evaluation of its analogue symplostatin 1. J. Nat. Prod. 2001, 64, 907–910. [Google Scholar] [CrossRef]
- Luesch, H.; Yoshida, W.Y.; Moore, R.E.; Paul, V.J.; Mooberry, S.L.; Corbett, T.H. Symplostatin 3, a new dolastatin 10 analogue from the marine cyanobacterium Symploca sp. VP452. J. Nat. Prod. 2002, 65, 16–20. [Google Scholar] [CrossRef]
- Matthew, S.; Paul, V.J.; Luesch, H. Tiglicamides A–C, cyclodepsipeptides from the marine cyanobacterium Lyngbya confervoides. Phytochemistry 2009, 70, 2058–2063. [Google Scholar] [CrossRef]
- Tan, L.T. Filamentous tropical marine cyanobacteria: a rich source of natural products for anticancer drug discovery. J. Appl. Phycol. 2010, 22, 659–676. [Google Scholar] [CrossRef]
- Drew, L.; Fine, R.L.; Do, T.N.; Douglas, G.P.; Petrylak, D.P. The novel antimicrotubule agent cryptophycin 52 (LY355703) induces apoptosis via multiple pathways in human prostate cancer cells. Clin. Cancer Res. 2002, 8, 3922–3932. [Google Scholar]
- Sato, M.; Sagawa, M.; Nakazato, T.; Ikeda, Y.; Kizaki, M. A natural peptide, dolastatin 15, induces G2/M cell cycle arrest and apoptosis of human multiple myeloma cells. Int. J. Oncol. 2007, 30, 1453–1459. [Google Scholar]
- Nagarajan, M.; Maruthanayagam, V.; Sundararaman, M. A review of pharmacological and toxicological potentials of marine cyanobacterial metabolites. J. Appl. Toxicol. 2012, 32, 153–155. [Google Scholar]
- Han, B.N.; McPhail, K.L.; Gross, H.; Goeger, D.E.; Mooberry, S.L.; Gerwick, W.H. Isolation and structure of five lyngbyabellin derivatives from a Papua New Guinea collection of the marine cyanobacterium Lyngbya majuscula. Tetrahedron 2005, 61, 11723–11729. [Google Scholar]
- Paradies, G.; Petrosillo, G.; Paradies, V.; Ruggiero, F.M. Mitochondrial dysfunction in brain aging: role of oxidative stress and cardiolipin. Neurochem. Int. 2011, 58, 447–457. [Google Scholar]
- Mammucari, C.; Rizzuto, R. Signaling pathways in mitochondrial dysfunction and aging. Mech. Ageing Dev. 2010, 131, 536–543. [Google Scholar]
- Sato, S.; Murata, A.; Orihara, T.; Shirakawa, T.; Suenaga, K.; Kigoshi, H.; Uesugi, M. Marine natural product aurilide activates the OPA1-mediated apoptosis by binding to prohibitin. Chem. Biol. 2011, 18, 131–139. [Google Scholar]
- Valko, M.; Leibfritz, D.; Moncol, J.; Cronin, M.T.; Mazur, M.; Telser, J. Free radicals and antioxidants in normal physiological functions and human disease. Int. J. Biochem. Cell Biol. 2007, 39, 44–84. [Google Scholar]
- Blokhina, O.; Virolainen, E.; Fagerstedt, K.V. Antioxidants, oxidative damage and oxygen deprivation stress: A review. Ann. Bot. 2003, 91, 179–194. [Google Scholar]
- Kelman, D.; Ben-Amotz, A.; Berman-Frank, I. Carotenoids provide the major antioxidant defence in the globally significant N2-fixing marine cyanobacterium Trichodesmium. Environ. Microbiol. 2009, 11, 1897–1908. [Google Scholar] [CrossRef]
- Patel, A.; Mishra, S.; Ghosh, P.K. Antioxidant potential of C-phycocyanin isolated from cyanobacterial species Lyngbya, Phormidium and Spirulina spp. Indian J. Biochem. Biophys. 2006, 43, 25–31. [Google Scholar]
- Donepudi, M.; Grutter, M.G. Structure and zymogen activation of caspases. Biophys. Chem. 2002, 101, 145–153. [Google Scholar]
- Fuentes-Prior, P.; Salvesen, G.S. The protein structures that shape caspase activity, specificity, activation and inhibition. Biochem. J. 2004, 384, 201–232. [Google Scholar] [CrossRef]
- Wilkinson, J.C.; Cepero, E.; Boise, L.H.; Duckett, C.S. Upstream regulatory role for XIAP in receptor-mediated apoptosis. Mol. Cell. Biol. 2004, 24, 7003–7014. [Google Scholar]
- Okuno, S.; Shimizu, S.; Ito, T.; Nomura, M.; Hamada, E.; Tsujimoto, Y.; Matsuda, H. Bcl-2 prevents caspase-independent cell death. J. Biol. Chem. 1998, 273, 34272–34277. [Google Scholar]
- Schrader, K.; Huai, J.; Jockel, L.; Oberle, C.; Borner, C. Non-Caspase proteases: Triggers or amplifiers of apoptosis? Cell. Mol. Life Sci. 2010, 67, 1607–1618. [Google Scholar] [CrossRef]
- Turk, B. Targeting proteases: Successes, failures and future prospects. Nat. Rev. Drug Discov. 2006, 5, 785–799. [Google Scholar]
- Williams, M.S.; Henkart, P.A. Apoptotic cell death induced by intracellular proteolysis. J. Immunol. 1994, 153, 4247–4255. [Google Scholar]
- Sagulenko, V.; Muth, D.; Sagulenko, E.; Paffhausen, T.; Schwab, M.; Westermann, F. Cathepsin D protects human neuroblastoma cells from doxorubicin-induced cell death. Carcinogenesis 2008, 29, 1869–1877. [Google Scholar]
- Bidere, N.; Lorenzo, H.K.; Carmona, S.; Laforge, M.; Harper, F.; Dumont, C.; Senik, A. Cathepsin D triggers Bax activation, resulting in selective apoptosis-inducing factor (AIF) relocation in T lymphocytes entering the early commitment phase to apoptosis. J. Biol. Chem. 2003, 278, 31401–31411. [Google Scholar]
- Zaidi, N.; Kalbacher, H. Cathepsin E: A mini review. Biochem. Biophys. Res. Commun. 2008, 367, 517–522. [Google Scholar]
- Ali, M.A.; Rosati, R.; Pettit, G.R.; Kalemkerian, G.P. Dolastatin 15 induces apoptosis and BCL-2 phosphorylation in small cell lung cancer cell lines. Anticancer Res. 1998, 18, 1021–1026. [Google Scholar]
- Pedersen, S.F.; Hoffmann, E.K.; Mills, J.W. The cytoskeleton and cell volume regulation. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2001, 130, 385–399. [Google Scholar] [CrossRef]
- Maeno, E.; Ishizaki, Y.; Kanaseki, T.; Hazama, A.; Okada, Y. Normotonic cell shrinkage because of disordered volume regulation is an early prerequisite to apoptosis. Proc. Natl. Acad. Sci. USA 2000, 97, 9487–9492. [Google Scholar]
- Fernandez-Segura, E.; Canizares, F.J.; Cubero, M.A.; Warley, A.; Campos, A. Changes in elemental content during apoptotic cell death studied by electron probe X-ray microanalysis. Exp. Cell Res. 1999, 253, 454–462. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Costa, M.; Costa-Rodrigues, J.; Fernandes, M.H.; Barros, P.; Vasconcelos, V.; Martins, R. Marine Cyanobacteria Compounds with Anticancer Properties: A Review on the Implication of Apoptosis. Mar. Drugs 2012, 10, 2181-2207. https://doi.org/10.3390/md10102181
Costa M, Costa-Rodrigues J, Fernandes MH, Barros P, Vasconcelos V, Martins R. Marine Cyanobacteria Compounds with Anticancer Properties: A Review on the Implication of Apoptosis. Marine Drugs. 2012; 10(10):2181-2207. https://doi.org/10.3390/md10102181
Chicago/Turabian StyleCosta, Margarida, João Costa-Rodrigues, Maria Helena Fernandes, Piedade Barros, Vitor Vasconcelos, and Rosário Martins. 2012. "Marine Cyanobacteria Compounds with Anticancer Properties: A Review on the Implication of Apoptosis" Marine Drugs 10, no. 10: 2181-2207. https://doi.org/10.3390/md10102181
APA StyleCosta, M., Costa-Rodrigues, J., Fernandes, M. H., Barros, P., Vasconcelos, V., & Martins, R. (2012). Marine Cyanobacteria Compounds with Anticancer Properties: A Review on the Implication of Apoptosis. Marine Drugs, 10(10), 2181-2207. https://doi.org/10.3390/md10102181









