Antioxidant Properties of Polysaccharide from the Brown Seaweed Sargassum graminifolium (Turn.), and Its Effects on Calcium Oxalate Crystallization
Abstract
:Abbreviations
| SGP | polysaccharide from Sargassum graminifolium |
| IR | infrared spectrum |
| DPPH | 1,1-diphenyl-2-picrylhydrazyl free radical |
| Ox | oxalate |
| CaOx | calcium oxalate |
| OD | optical density |
| SN | maximum increase of optical density with time |
| SA | rate of aggregation derived from the maximum decrease in optical density |
| tmax | maximum time |
| COM | Calcium oxalate monohydrate |
| COD | Calcium oxalate dehydrate |
| COT | Calcium oxalate trihydrate |
1. Introduction
2. Results and Discussion
2.1. Properties of SGP
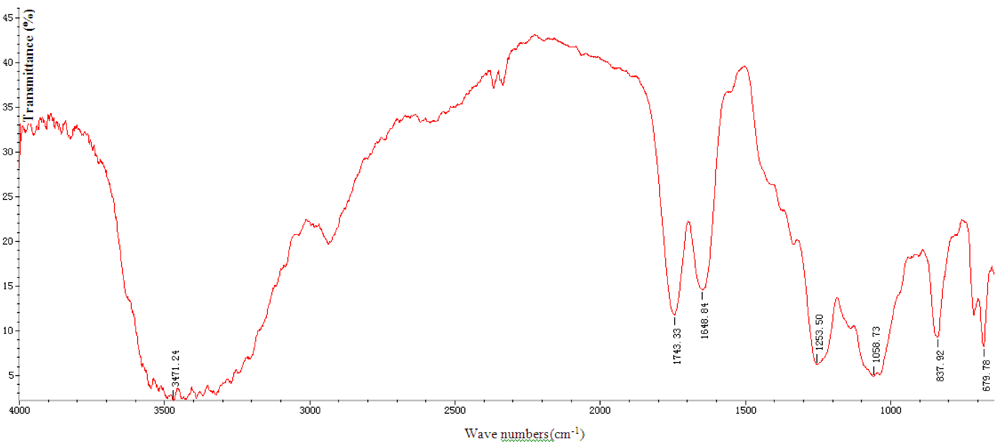
2.2. Effect on Calcium Oxalate Crystallization
| 4 mmol/L calcium/ 0.5 mmol/L oxalate | 4 mmol/L calcium/ 0.5 mmol/L oxalate + 0.25 mmol/L trisodium citrate | 4 mmol/L calcium/ 0.5 mmol/L oxalate + 0.01 g/100 mL SGP | P | |
|---|---|---|---|---|
| tmax (min) | 8.67 ± 0.94 | 13.44 ± 0.57 | 14.02 ± 0.82 | P < 0.05 a |
| P < 0.05 b | ||||
| SN (×10−3/min) | 5.30 ± 1.23 | 2.20 ± 0.36 | 1.63 ± 0.28 | P < 0.05 a |
| P < 0.05 b | ||||
| SA (×10−3/min) | 1.87 ± 0.62 | 0.53 ± 0.19 | 0.43 ± 0.05 | P < 0.05 a |
| P < 0.05 b |
2.3. Effect on Crystal Morphology
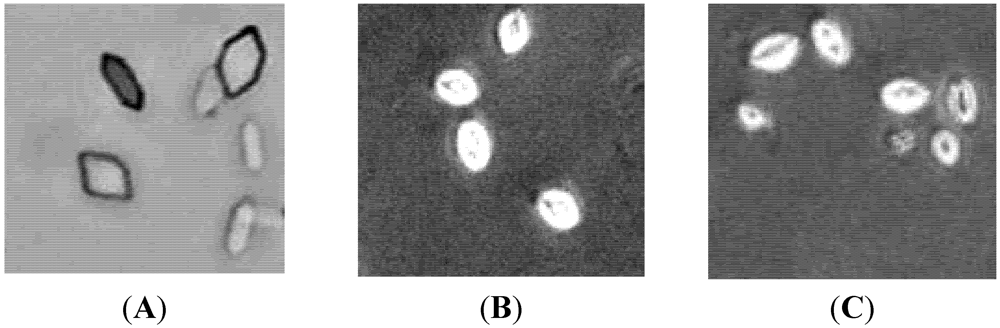
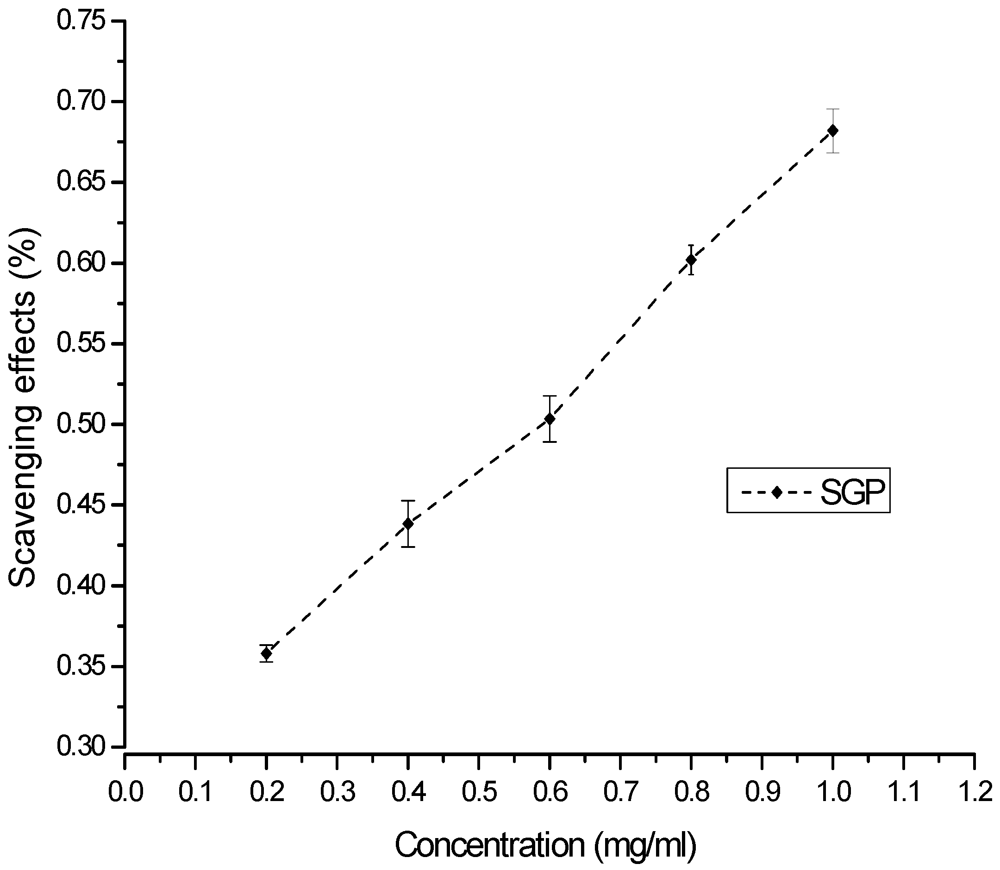
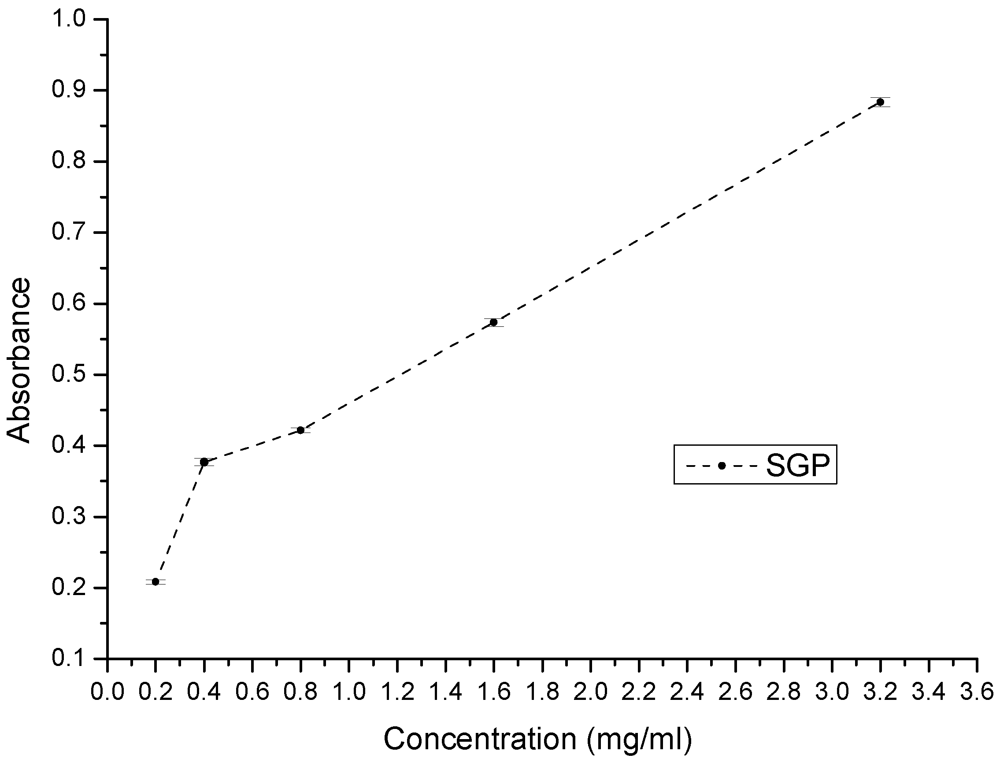
2.4. Antioxidant Effects of SGP

3. Experimental Section
3.1. Chemicals and Reagents
3.2. Extraction of Polysaccharides from S. graminifolium and Its Properties
3.3. Calcium Oxalate Crystallization Assay
3.4. Image Analysis of Crystal Morphology
3.5. Antioxidant Assays
3.5.1. DPPH Assay
3.5.2. Superoxide Radical Scavenging Assay
3.5.3. Reducing Powers of SGP
3.6. Statistical Analysis
4. Conclusions
Acknowledgments
- Samples Availability: Available from the authors.
References
- Hasui, M.; Matsuda, M.; Okutani, K.; Shigeta, S. In vitro antiviral activities of sulfated polysaccharides from a marine microalga (Cochlodinium polykrikoides) against human immunodeficiency virus and other enveloped viruses. Int. J. Biol. Macromol. 1995, 17, 293–297. [Google Scholar]
- Karnjanapratum, S.; You, S. Molecular characteristics of sulfated polysaccharides from Monostroma nitidum and their in vitro anticancer and immunomodulatory activities. Int. J. Biol. Macromol. 2010, 48, 311–318. [Google Scholar]
- Rajeswari, A.; Varalakshmi, P. Low molecular weight heparin protection against oxalate-induced oxidative renal insult. Clin. Chim. Acta 2006, 370, 108–114. [Google Scholar]
- Wijesekara, I.; Pangestuti, R.; Kim, S.K. Biological activities and potential health benefits of sulfated polysaccharides derived from marine algae. Carbohydr. Polym. 2011, 84, 14–21. [Google Scholar]
- Zhang, Z.; Zhang, Q.; Wang, J.; Zhang, H.; Niu, X.; Li, P. Preparation of the different derivatives of the low-molecular-weight porphyran from Porphyra haitanensis and their antioxidant activities in vitro. Int. J. Biol. Macromol. 2009, 45, 22–26. [Google Scholar]
- Zhang, Q.; Li, N.; Liu, X.; Zhao, Z.; Li, Z.; Xu, Z. The structure of a sulfated galactan from Porphyra haitanensis and its in vivo antioxidant activity. Carbohydr. Res. 2004, 339, 105–111. [Google Scholar]
- Coothan, K.V.; Anthony, J.; Sreenivasan, P.; Preetha, P.V.; Rajaguru, S. Renal peroxidative changes mediated by oxalate: The protective role of fucoidan. Life Sci. 2006, 79, 1789–1795. [Google Scholar]
- Das, I.; Gupta, S.K.; Ansari, S.A.; Pandey, V.N.; Rastogi, R.P. In vitro inhibition and dissolution of calcium oxalate by edible plant Trianthema monogyna and pulse Macrotyloma uniflorum extracts. J. Cryst. Growth 2005, 273, 546–554. [Google Scholar]
- Basavaraj, D.R.; Biyani, C.S.; Browning, A.J.; Cartledge, J.J. The role of urinary kidney stone inhibitors and promoters in the pathogenesis of calcium containing renal stones. EAU-EBU Update Ser. 2007, 5, 126–136. [Google Scholar] [CrossRef]
- Bihl, G.; Meyers, A. Recurrent renal stone disease—advances in pathogenesis and clinical management. Lancet 2001, 358, 651–656. [Google Scholar]
- Atmani, F.; Khan, S.R. Effects of an extract from Herniaria hirsuta on calcium oxalate crystallization in vitro. BJU Int. 2000, 85, 621–625. [Google Scholar]
- Moe, O.W. Kidney stones: Pathophysiology and medical management. Lancet 2006, 367, 333–344. [Google Scholar]
- Borghi, L.; Meschi, T.; Guerra, A.; Bergamaschi, E.; Mutti, A.; Novarini, A. Effects of urinary macromolecules on the nucleation of calcium oxalate in idiopathic stone formers and healthy controls. Clin. Chim. Acta 1995, 239, 1–11. [Google Scholar]
- Edyvane, K.A.; Hibberd, C.M.; Harnett, R.M.; Marshall, V.R.; Ryall, R.L. Macromolecules inhibit calcium oxalate crystal growth and aggregation in whole human urine. Clin. Chim. Acta 1987, 167, 329–338. [Google Scholar]
- Liu, J.; Wang, T.; Chen, J.; Wang, S.; Ye, Z. Decreased inhibitory activity of prothrombin to calcium oxalate crystallization by specific chemical modification of its gamma-carboxyglutamic acid residues. Urology 2006, 67, 201–203. [Google Scholar]
- Hackett, R.L.; Shevock, P.N.; Khan, S.R. Madin-Darby ca nine kidney cells are injured by exposure to oxalate and to calcium oxalate crystals. Urol. Res. 1994, 22, 197–203. [Google Scholar]
- Koul, H.; Kenington, L.; Honeyman, T.; Jonassen, J.; Menon, M.; Scheid, C.R. Activation of the c-myc gene mediates the mitogenic effects of Ox in LLC-PK1 cells, a line of renal epithelial cells. Kidney Int. 1996, 50, 1525–1530. [Google Scholar]
- Bashir, S.; Gilani, A.H. Antiurolithic effect of Bergenia ligulata rhizome: An explanation of the underlying mechanisms. J. Ethnopharmacol. 2009, 122, 106–116. [Google Scholar]
- Saeed, R.K. Hyperoxaluria-induced oxidative stress and antioxidants for renal protection. Urol. Res. 2005, 33, 349–357. [Google Scholar]
- Vanachayangkul, P.; Byer, K.; Khan, S.; Butterweck, V. An aqueous extract of Ammi visnaga fruits and its constituents khellin and visnagin prevent cell damage caused by oxalate in renal epithelial cells. Phytomedicine 2010, 17, 653–658. [Google Scholar]
- Srinivasan, S.; Pragasam, V.; Jenita, X.; Kalaiselvi, P.; Muthu, V.; Varalakshmi, P. Oxidative stress in urogenital tuberculosis patients: a predisposing factor for renal stone formation—amelioration by vitamin E supplementation. Clin. Chim. Acta 2004, 350, 57–63. [Google Scholar]
- Rodrigo, R.; Rivera, G. Renal damage mediated by oxidative stress: A hypothesis of protective effects of red wine. Free Radic. Biol. Med. 2002, 33, 409–422. [Google Scholar]
- Siener, R.; Hesse, A. The effect of different diets on urine composition and the risk of calcium oxalate crystallisation in healthy subjects. Eur. Urol. 2002, 42, 289–296. [Google Scholar]
- Wang, J.; Zhang, Q.B.; Zhang, Z.S.; Zhang, H.; Niu, X.Z. Structural studies on a novel fucogalactan sulfate extracted from the brown seaweed Laminaria japonica. Int. J. Biol. Macromol. 2010, 47, 126–131. [Google Scholar]
- Samee, H.; Li, Z.X.; Lin, H.; Khalid, J.; Wang, B.P. In vivo study of antiallergenicity of ethanol extracts from Sargassum tenerrimum, Sargassum cervicorne and Sargassum graminifolium Turn. Eur. Food Res. Technol. 2009, 229, 435–441. [Google Scholar]
- Ouyang, J.M.; Zheng, H.; Deng, S.P. Simultaneous formation of calcium oxalate (mono-, di-, and trihydrate) induced by potassium tartrate in gelatinous system. J. Cryst. Growth 2006, 293, 118–123. [Google Scholar]
- Yu, J.; Tang, H.; Cheng, B. Influence of PSSS additive and temperature on morphology and phase structures of calcium oxalate. J. Colloid Interface Sci. 2005, 288, 407–411. [Google Scholar]
- Liang, M.; Bai, Y.; Huang, L.; Zheng, W.; Liu, J. Inhibition of the crystal growth and aggregation of calcium oxalate by elemental selenium nanoparticles. Colloids Surf. B Biointerfaces 2009, 74, 366–369. [Google Scholar]
- Liochev, S.I.; Fridovich, I. The effects of superoxide dismutase on H2O2 formation. Free Radic. Biol. Med. 2007, 42, 1465–1469. [Google Scholar]
- Hu, F.L.; Lu, R.L.; Huang, B.; Ming, L. Free radical scavenging activity of extracts prepared from fresh leaves of selected Chinese medicinal plants. Fitoterapia 2004, 75, 14–23. [Google Scholar]
- Bortolomeazzi, R.; Sebastianutto, N.; Toniolo, R.; Pizzariello, A. Comparative evaluation of the antioxidant capacity of smoke flavouring phenols by crocin bleaching inhibition, DPPH radical scavenging and oxidation potential. Food Chem. 2007, 100, 1481–1489. [Google Scholar]
- Fagali, N.; Catal, A. Antioxidant activity of conjugated linoleic acid isomers, linoleic acid and its methyl ester determined by photoemission and DPPH techniques. Biophys. Chem. 2008, 137, 56–62. [Google Scholar]
- Hassas-Roudsari, M.; Chang, P.R.; Pegg, R.B.; Tyler, R.T. Antioxidant capacity of bioactives extracted from canola meal by subcritical water, ethanolic and hot water extraction. Food Chem. 2009, 114, 717–726. [Google Scholar]
- Wang, H.; Gao, X.D.; Zhou, G.C.; Cai, L.; Yao, W.B. In vitro and in vivo antioxidant activity of aqueous extract from Choerospondias axillaris fruit. Food Chem. 2008, 106, 888–895. [Google Scholar]
- Zhuang, C.; Itoh, H.; Mizuno, T.; Ito, H. Antitumor active fucoidan from the brown seaweed, umitoranoo (Sargassum thunbergii). Biosci. Biotechnol. Biochem. 1995, 59, 563–567. [Google Scholar]
- Kulaksizoglu, S.; Sofikerim, M.; Cevik, C. In vitro effect of lemon and orange juices on calcium oxalate crystallization. Int. Urol. Nephrol. 2008, 40, 589–594. [Google Scholar]
- Ye, H.; Wang, K.; Zhou, C.; Liu, J.; Zeng, X. Purification, antitumor and antioxidant activities in vitro of polysaccharides from the brown seaweed Sargassum pallidum. Food Chem. 2008, 111, 428–432. [Google Scholar]
- Sun, T.; Yao, Q.; Zhou, D.X.; Mao, F. Antioxidant activity of N-carboxymethyl chitosan oligosaccharides. Bioorg. Med. Chem. Lett. 2008, 18, 5774–5776. [Google Scholar]
- Mushtaq, S.; Siddiqui, A.A.; Naqvi, Z.A.; Rattani, A.; Talati, J.; Palmberg, C.; Shafqat, J. Identification of myeloperoxidase, [alpha]-defensin and calgranulin in calcium oxalate renal stones. Clin. Chim. Acta 2007, 384, 41–47. [Google Scholar]
- Hess, B.; Meinhardt, U.; Zipperle, L.; Giovanoli, R.; Jaeger, P. Simultaneous measurements of calcium oxalate crystal nucleation and aggregation: impact of various modifiers. Urol. Res. 1995, 23, 231–238. [Google Scholar]
- Gohel, M.D.; Wong, S.P. Chinese herbal medicines and their efficacy in treating renal stones. Urol. Res. 2006, 34, 365–372. [Google Scholar]
- Kavanagh, J.P.; Jones, L.; Rao, P.N. Calcium oxalate crystallization kinetics studied by oxalate-induced turbidity in fresh human urine and artificial urine. Clin. Sci. (Lond.) 2000, 98, 151–158. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhang, C.-Y.; Wu, W.-H.; Wang, J.; Lan, M.-B. Antioxidant Properties of Polysaccharide from the Brown Seaweed Sargassum graminifolium (Turn.), and Its Effects on Calcium Oxalate Crystallization. Mar. Drugs 2012, 10, 119-130. https://doi.org/10.3390/md10010119
Zhang C-Y, Wu W-H, Wang J, Lan M-B. Antioxidant Properties of Polysaccharide from the Brown Seaweed Sargassum graminifolium (Turn.), and Its Effects on Calcium Oxalate Crystallization. Marine Drugs. 2012; 10(1):119-130. https://doi.org/10.3390/md10010119
Chicago/Turabian StyleZhang, Chao-Yan, Wen-Hui Wu, Jue Wang, and Min-Bo Lan. 2012. "Antioxidant Properties of Polysaccharide from the Brown Seaweed Sargassum graminifolium (Turn.), and Its Effects on Calcium Oxalate Crystallization" Marine Drugs 10, no. 1: 119-130. https://doi.org/10.3390/md10010119
APA StyleZhang, C.-Y., Wu, W.-H., Wang, J., & Lan, M.-B. (2012). Antioxidant Properties of Polysaccharide from the Brown Seaweed Sargassum graminifolium (Turn.), and Its Effects on Calcium Oxalate Crystallization. Marine Drugs, 10(1), 119-130. https://doi.org/10.3390/md10010119





