Development and Validation of a Simple and Reliable HPLC-UV Method for Determining Gemcitabine Levels: Application in Pharmacokinetic Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Instrumental Configuration and Chromatographic Conditions
2.3. Preparation of Standard Solutions
2.4. Sample Preparation
2.5. Method Validation
2.6. Pharmacokinetic Study
2.7. Data Acquisition and Statistical Analysis
3. Results
3.1. Method Validation
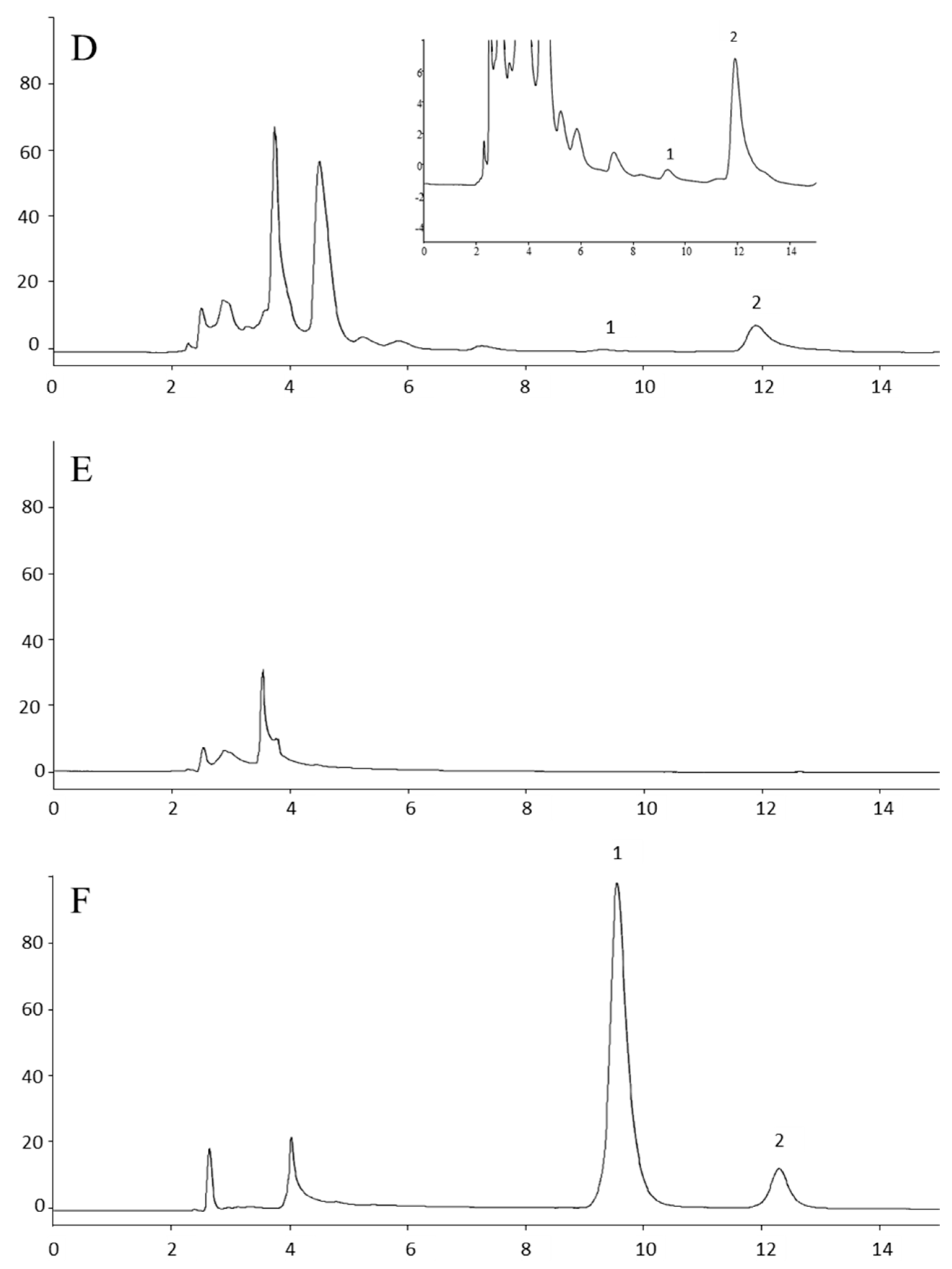
3.1.1. Selectivity
3.1.2. Calibration Curve
3.1.3. Recovery
3.1.4. Precision and Accuracy
3.1.5. Determination of the Lower Limit of Quantitation (LLOQ) and the Limit of Detection (LOD)
3.1.6. Stability
3.1.7. Carry-Over
3.2. Pharmacokinetics
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mini, E.; Nobili, S.; Caciagli, B.; Landini, I.; Mazzei, T. Cellular pharmacology of gemcitabine. Ann. Oncol. 2006, 17 (Suppl. S5), v7–v12. [Google Scholar] [CrossRef]
- Burris, H.A.; Moore, M.J.; Andersen, J.; Green, M.R.; Rothenberg, M.L.; Modiano, M.R.; Cripps, M.C.; Portenoy, R.K.; Storniolo, A.M.; Tarassoff, P.; et al. Improvements in survival and clinical benefit with gemcitabine as first-line therapy for patients with advanced pancreas cancer: A randomized trial. J. Clin. Oncol. 1997, 15, 2403–2413. [Google Scholar] [CrossRef]
- Li, Y.; Li, P.; Li, Y.; Zhang, R.; Yu, P.; Ma, Z.; Kainov, D.E.; de Man, R.A.; Peppelenbosch, M.P.; Pan, Q. Drug screening identified gemcitabine inhibiting hepatitis E virus by inducing interferon-like response via activation of STAT1 phosphorylation. Antivir. Res. 2020, 184, 104967. [Google Scholar] [CrossRef]
- Kirstein, M.N.; Hassan, I.; Guire, D.E.; Weller, D.R.; Dagit, J.W.; Fisher, J.E.; Remmel, R.P. High-performance liquid chromatographic method for the determination of gemcitabine and 2′,2′-difluorodeoxyuridine in plasma and tissue culture media. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2006, 835, 136–142. [Google Scholar] [CrossRef] [PubMed]
- Committee for Medicinal Products for Human Use; European Medicines Agency (EMA). EMEA/CHMP/EWP/192217/2009 Rev. 1 Corr. 2**. 2011, Chapters 4.1.3 and 4.1.9. in Guideline on Bioanalytical Method Validation. Available online: https://www.ema.europa.eu/en/documents/scientific-guideline/guideline-bioanalytical-method-validation_en.pdf (accessed on 23 October 2023).
- Snyder, L.R.; Kirkland, J.J.; Glajch, L.J. Limits of Detection and Quantitation. In Practical HPLC Method Development, 2nd ed.; John Wiley & Sons INC.: New York, NY, USA, 1997; pp. 670–710. [Google Scholar]
- Lanz, C.; Fruh, M.; Thormann, W.; Cerny, T.; Lauterburg, B.H. Rapid determination of gemcitabine in plasma and serum using reversed-phase HPLC. J. Sep. Sci. 2007, 12, 1811–1820. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Huo, M.; Zhou, J.; Xie, S. PKSolver: An add-in program for pharmacokinetic and pharmacodynamic data analysis in Microsoft Excel. Comput. Methods Programs Biomed. 2010, 99, 306–314. [Google Scholar] [CrossRef]
- Sereti, E.; Karagianellou, T.; Kotsoni, I.; Magouliotis, D.; Kamposioras, K.; Ulukaya, E.; Sakellaridis, N.; Zacharoulis, D.; Dimas, K. Patient Derived Xenografts (PDX) for personalized treatment of pancreatic cancer: Emerging allies in the war on a devastating cancer? J. Proteom. 2018, 188, 107–118. [Google Scholar] [CrossRef] [PubMed]
- Grunewald, R.; Kantarjian, H.; Keating, M.J.; Abbruzzese, J.; Tarassoff, P.; Plunkett, W. Pharmacologically directed design of the dose rate and schedule of 2′,2′-difluorodeoxycytidine (gemcitabine) administration in leukemia. Cancer Res. 1990, 50, 6823–6826. [Google Scholar]
- Grunewald, R.; Abbruzzese, J.L.; Tarassoff, P.; Plunkett, W. Saturation of 2′,2′-difluorodeoxycytidine 5′-triphosphate accumulation by mononuclear cells during a phase I trial of gemcitabine. Cancer Chemother. Pharmacol. 1991, 27, 258–262. [Google Scholar] [CrossRef]
- Grunewald, R.; Kantarjian, H.; Du, M.; Faucher, K.; Tarassoff, P.; Plunkett, W. Gemcitabine in leukemia: A phase I clinical, plasma, and cellular pharmacology study. J. Clin. Oncol. 1992, 10, 406–413. [Google Scholar] [CrossRef]
- Bengala, C.; Guarneri, V.; Giovannetti, E.; Lencioni, M.; Fontana, E.; Mey, V.; Fontana, A.; Boggi, U.; Del Chiaro, M.; Danesi, R.; et al. Prolonged fixed dose rate infusion of gemcitabine with autologous haemopoietic support in advanced pancreatic adenocarcinoma. Br. J. Cancer 2005, 93, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Abbruzzese, J.L.; Grunewald, R.; Weeks, E.A.; Gravel, D.; Adams, T.; Nowak, B.; Mineishi, S.; Tarassoff, P.; Satterlee, W.; Raber, M.N.; et al. A phase I clinical, plasma, and cellular pharmacology study of gemcitabine. J. Clin. Oncol. 1991, 9, 491–498. [Google Scholar] [CrossRef] [PubMed]
- Venook, P.; Egorin, M.J.; Rosner, G.L.; Hollis, D.; Mani, S.; Hawkins, M.; Byrd, J.; Hohl, R.; Budman, D.; Meropol, N.J.; et al. Phase I and pharmacokinetic trial of gemcitabine in patients with hepatic or renal dysfunction: Cancer and Leukemia Group B 9565. J. Clin. Oncol. 2000, 18, 2780–2787. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; He, S.; Wang, P.; Zhou, Y. The efficacy and safety of gemcitabine-based combination therapy vs. gemcitabine alone for the treatment of advanced pancreatic cancer: A systematic review and meta-analysis. J. Gastrointest. Oncol. 2022, 13, 1967–1980. [Google Scholar] [CrossRef] [PubMed]
- Mascherona, I.; Maggioli, C.; Biggiogero, M.; Mora, O.; Marelli, L. A Severe Case of Drug-Induced Liver Injury after Gemcitabine Administration: A Highly Probable Causality Grading as Assessed by the Updated RUCAM Diagnostic Scoring System. Case Rep. Hepatol. 2020, 2020, 8812983. [Google Scholar] [CrossRef] [PubMed]
- Jha, V.K.; Akal, R.S.; Mahapatra, D.; Sharma, A.; Singh, B.P.; Arora, R. Nephrotic Syndrome and Posterior Reversible Encephalopathy Syndrome as Clinical Presentations of Gemcitabine-Induced Thrombotic Micro-Angiopathy. Indian J. Nephrol. 2024, 34, 74–78. [Google Scholar] [CrossRef]
- Mercier, C.; Raynal, C.; Dahan, L.; Ortiz, A.; Evrard, A.; Dupuis, C.; Blesius, A.; Duluc Manceschini, F.; Giacometti, S.; Salas, S.; et al. Toxic death case in a patient undergoing gemcitabine-based chemotherapy in relation with cytidine deaminase downregulation. Pharmacogenet. Genom. 2007, 17, 841–844. [Google Scholar] [CrossRef]
- Hryciuk, B.; Szymanowski, B.; Romanowska, A.; Salt, E.; Wasąg, B.; Grala, B.; Jassem, J.; Duchnowska, R. Severe acute toxicity following gemcitabine administration: A report of four cases with cytidine deaminase polymorphisms evaluation. Oncol. Lett. 2018, 15, 1912–1916. [Google Scholar] [CrossRef] [PubMed]
- Freeman, K.B.; Anliker, S.; Hamilton, M.; Osborne, D.; Dhahir, P.H.; Nelson, R.; Allerheiligen, S.R. Validated assays for the determination of gemcitabine in human plasma and urine using high-performance liquid chromatography with ultraviolet detection. J. Chromatogr. B Biomed. Appl. 1995, 665, 171–181. [Google Scholar] [CrossRef]
- Keith, B.; Xu, Y.; Grem, J.L. Measurement of the anti-cancer agent gemcitabine in human plasma by high-performance liquid chromatography. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2003, 785, 65–72. [Google Scholar] [CrossRef]
- Yılmaz, B.; Kadıoğlu, Y.; Aksoy, Y. Simultaneous determination of gemcitabine and its metabolite in human plasma by high-performance liquid chromatography. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2003, 791, 103–109. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.Z.; Goh, B.C.; Lee, H.S.; Noordhuis, P.; Godefridus, J.P. An Expedient Assay for Determination of Gemcitabine and Its Metabolite in Human Plasma Using Isocratic Ion-Pair Reversed-Phase High-Performance Liquid Chromatography. Ther. Drug Monit. 2003, 25, 552–557. [Google Scholar] [CrossRef] [PubMed]
- Lin, N.M.; Zeng, S.; Ma, S.L.; Fan, Y.; Zhong, H.J.; Fang, L. Determination of gemcitabine and its metabolite in human plasma using high-pressure liquid chromatography coupled with a diode array detector. Acta Pharmacol. Sin. 2004, 25, 1584–1589. [Google Scholar] [PubMed]
- Vidal, H.; Goncalinho, H.; Monteiro, J.; Neves, J.; Sarmento, B.; Diniz, C.; Fresco, P. Development and validation of a rapid and simple reversed-phase HPLC method for the determination of gemcitabine in human plasma. Int. J. Pharm. Pharm. Sci. 2014, 6, 59–65. [Google Scholar]
- Wang, M.; Hiraki, R.; Nakamura, N.; Hayama, T.; Yasuda, T.; Miyata, Y.; Sakai, H.; Nakashima, M.N.; Nakashima, M.; Ohyama, K. Determination of Gemcitabine in Plasma of Bladder Cancer Patients by Hydrophilic Interaction Chromatography with Ultraviolet Detection. J. Chromatogr. Sci. 2020, 58, 587–590. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Zhen, L.; Peng, Y.; Wang, J.; Fei, F.; Aa, L.; Jiang, W.; Pei, X.; Lu, L.; Liu, J.; et al. Simultaneous determination of gemcitabine prodrug, gemcitabine and its major metabolite 2′,2′-difluorodeoxyuridine in rat plasma by UFLC-MS/MS. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2018, 1084, 4–13. [Google Scholar] [CrossRef] [PubMed]
- Kozo, D.; Ross, M.; Jarrah, J.; Berrett, M.; Harney, R.; Courney, J.; Baburina, I.; Holleran, J.; Beumer, J.; Peters, G.; et al. A rapid homogeneous immunoassay to quantify gemcitabine in plasma for therapeutic drug monitoring (TDM). Ther. Drug Monit. 2017, 39, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Palakeeti, Β.; Amara Babu, N.L.A.; Chinta, J.P. Development of a magnetic solid phase extraction method for gemcitabine from rat plasma by using magnetic graphene oxide calix[6]arene composite and its application to pharmacokinetics. Sep. Sci. Technol. 2023, 58, 1596–1606. [Google Scholar] [CrossRef]
- Meyer, V.R. Theoretical Principles. In Practical High-Performance Liquid Chromatography; John Wiley & Sons Ltd.: West Sussex, UK, 1994; pp. 17–55. [Google Scholar]
- Turner, P.V.; Brabb, T.; Pekow, C.; Vasbinder, M.A. Administration of substances to laboratory animals: Routes of administration and factors to consider. J. Am. Assoc. Lab. Anim. Sci. 2011, 50, 600–613. [Google Scholar]
- McDonald, Τ.A.; Monica, L.; Zepeda, Μ.L.; Tomlinson, M.J.; Bee, W.H.; Ivens, I.A. Subcutaneous administration of biotherapeutics: Current experience in animal models. Curr. Opin. Mol. Ther. 2010, 12, 461–470. [Google Scholar]


| Calibrator Concentration (μΜ) | Mean Measured Concentration (μΜ) | CV (%) | Bias (%) |
|---|---|---|---|
| 1 | 1.06 | 0.31 | 6.43 |
| 10 | 9.63 | 1.97 | −3.66 |
| 50 | 48.23 | 0.13 | −3.54 |
| 100 | 100.60 | 1.72 | 0.60 |
| 200 | 208.34 | 1.52 | 4.17 |
| 400 | 401.96 | 2.91 | 0.49 |
| INTRA-DAY | MEAN | SD | CV (%) | BIAS (%) |
|---|---|---|---|---|
| QC-3 μΜ | 3.50 | 0.17 | 4.89 | 16.55 |
| QC-25 μΜ | 27.81 | 2.05 | 5.98 | 11.25 |
| QC-125 μM | 126.84 | 3.70 | 2.92 | 1.47 |
| QC-375 μΜ | 347.81 | 26.16 | 7.52 | −7.25 |
| INTER-DAY | MEAN | SD | CV (%) | BIAS (%) |
| QC-3 μΜ | 3.44 | 0.33 | 9.56 | 14.70 |
| QC-25 μΜ | 26.05 | 1.61 | 6.16 | 4.21 |
| QC-125 μM | 126.01 | 4.51 | 3.58 | 0.80 |
| QC-375 μΜ | 366.87 | 14.69 | 4.00 | −2.17 |
| Parameter (Unit) | IP | SC | p * |
|---|---|---|---|
| T1/2 (min) | 59.34 (45.89–134.52) | 64.49 (55.13–69.43) | 1.00 |
| Tmax (min) | 5.00 (5.00–5.00) | 5.00 (5.00–5.00) | 1.00 |
| Cmax (μmol/L) | 300.73 (279.17–305.66) | 264.88 (234.37–339.90) | 0.31 |
| AUC0–t (μmol/L*min) | 8981.35 (7735.79–9354.28) | 9351.95 (7811.82–9851.25) | 0.31 |
| Cl (mg)/(μmol/L)/min | 0.0108 (0.0103–0.0125) | 0.0103 (0.0098–0.0120) | 0.15 |
| Reference | Column | Elution Mode | Mobile Phase | Sample Matrix | Sample Pre-Treatment | Internal Standard | Sample Volume | Analysis Time | Analytical Range (μΜ) | LLOQ | LLOD |
|---|---|---|---|---|---|---|---|---|---|---|---|
| [4] | C18 | Isocratic | 10% v/v acetonitrile/90% v/v sodium phosphate | Human plasma | protein precipitation (perchloric acid) | 2′-Deoxycytidine | 250 μL | 10 min | 2–100 | 2 μM | 0.02 μΜ |
| [7] | C18 | Gradient | 0.3–3% v/v acetonitrile /phosphate buffer | Human plasma & Rat serum | protein precipitation (acetonitrile) | 2′-Deoxyuridine | 200 μL | 7 min | 0.076–75.99 | 0.076 μΜ | 0.038 μΜ |
| [21] | NH2 (Amino) | Isocratic | 630 mL cyclohexane, 150 mL 1,2-dichloroethane, 220 mL methanol, 1 mL purified water, 0.5 mL glacial acetic acid, 1 mL triethylamine. | Human plasma | protein precipitation (isopropanol—ethyl acetate) | 2′-Deoxycytidine | 200 μL | 15 min | 0.19–9.1 | 0.19 μΜ | - |
| [22] | C18 | Gradient | Solvent A: 98% v/v sodium acetate/2% v/v methanol—Solvent B: 90% v/v sodioum acetate/10% v/v methanol | Human plasma | protein precipitation (acetic acid) | 2′-Fluorodeoxycytidine (FdC) | 500 μL | 17.5 min | 0.5–150 | - | - |
| [23] | NH2 (Amino) | Isocratic | 30% v/v methanol/50% v/v cyclohexane/20% v/v 1,2-dichloroethane | Human plasma | protein precipitation (isopropanol) | - | 200 μL | 10 min | 0.76–189.97 | 0.57 μΜ | 0.38 μΜ |
| [24] | C18 | Isocratic (ion pair) | 10 mM sodium 1-heptanesulfonate in ammonioum dihydrogen phosphate buffer solution (20 mM, pH 3.1): methanol (83:17% v/v) | Human plasma | protein precipitation (trichloroacetic acid) | - | 100 μL | 24 min | 0.3–75.99 | 0.3 μΜ | 0.19 μΜ |
| [25] | C18 | Isocratic | Acetate ammonium buffer solution (pH 5.5)—acetonitrile (97.5:2.5% v/v) | Human plasma | protein precipitation (methanol-acetonitrile 1:9 v/v) | Floxuridine | 900 μL | 18 min | 0.76–37.98 | 0.76 μΜ | 0.38 μΜ |
| [26] | C18 | Gradient | Solvent A: Sodium acetate buffer pH = 5 Solvent B: acetonitrile. %A:B%: 98.5:1.5 (v/v) | Human plasma | protein precipitation (methanol) | Cytarabine crystalline (4-amino-1-β-D-arabinofuranosyl-2(1H)-pyrimidinone | 200 μL | 13 min | 00.98–37.99 | 0.95 μΜ | 0.84 μΜ |
| [27] | HILIC-Amide | Isocratic | 90% v/v acetonitrile/10% v/v ammonioum acetate | Human plasma | liquid—liquid extraction | Cytarabine | 190 μL | 8.5 min | 1.9–189.97 | - | - |
| Proposed method | C18 | Isocratic | 3% v/v methanol/97% v/v phosphate buffer solution | Mouse serum | protein precipitation (perchloric acid) | 1,7-Dimethyluric Acid (1,7U) | 200 μL | 12.5 min | 1–400 | 1 μΜ | 0.17 μΜ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lafazanis, K.; Begas, E.; Papapostolou, I.; Iatrou, H.; Sakellaridis, N.; Vlassopoulos, D.; Dimas, K. Development and Validation of a Simple and Reliable HPLC-UV Method for Determining Gemcitabine Levels: Application in Pharmacokinetic Analysis. Medicina 2024, 60, 864. https://doi.org/10.3390/medicina60060864
Lafazanis K, Begas E, Papapostolou I, Iatrou H, Sakellaridis N, Vlassopoulos D, Dimas K. Development and Validation of a Simple and Reliable HPLC-UV Method for Determining Gemcitabine Levels: Application in Pharmacokinetic Analysis. Medicina. 2024; 60(6):864. https://doi.org/10.3390/medicina60060864
Chicago/Turabian StyleLafazanis, Konstantinos, Elias Begas, Irida Papapostolou, Hermis Iatrou, Nikos Sakellaridis, Dimitrios Vlassopoulos, and Konstantinos Dimas. 2024. "Development and Validation of a Simple and Reliable HPLC-UV Method for Determining Gemcitabine Levels: Application in Pharmacokinetic Analysis" Medicina 60, no. 6: 864. https://doi.org/10.3390/medicina60060864
APA StyleLafazanis, K., Begas, E., Papapostolou, I., Iatrou, H., Sakellaridis, N., Vlassopoulos, D., & Dimas, K. (2024). Development and Validation of a Simple and Reliable HPLC-UV Method for Determining Gemcitabine Levels: Application in Pharmacokinetic Analysis. Medicina, 60(6), 864. https://doi.org/10.3390/medicina60060864








