Musculoskeletal Pain, Physical Activity, Muscle Mass, and Mortality in Older Adults: Results from the Korean Longitudinal Study on Health and Aging (KLoSHA)
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Subjects
2.2. Demographics and Anthropometric Measures
2.3. Knee Pain, Low Back Pain, and Regular Exercise
2.4. Statistical Analyses
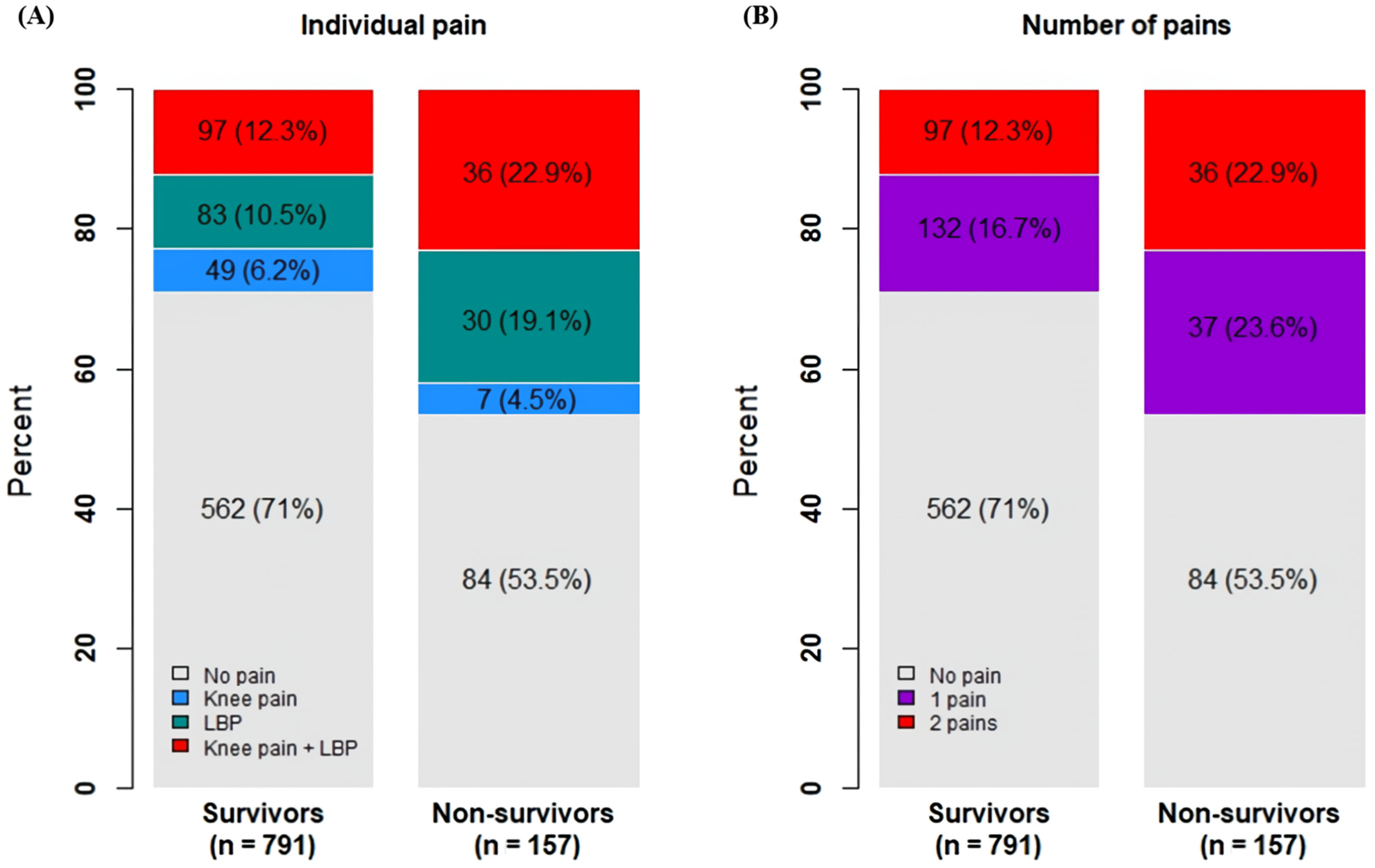
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Woolf, A.D.; Pfleger, B. Burden of major musculoskeletal conditions. Bull. World Health Organ. 2003, 81, 646–656. [Google Scholar] [PubMed]
- Cimmino, M.A.; Ferrone, C.; Cutolo, M. Epidemiology of chronic musculoskeletal pain. Best Pract. Res. Clin. Rheumatol. 2011, 25, 173–183. [Google Scholar] [CrossRef] [PubMed]
- Cieza, A.; Causey, K.; Kamenov, K.; Hanson, S.W.; Chatterji, S.; Vos, T. Global estimates of the need for rehabilitation based on the Global Burden of Disease study 2019: A systematic analysis for the Global Burden of Disease Study 2019. Lancet 2021, 396, 2006–2017. [Google Scholar] [CrossRef]
- Global, regional, and national burden of low back pain, 1990-2020, its attributable risk factors, and projections to 2050: A systematic analysis of the Global Burden of Disease Study 2021. Lancet Rheumatol. 2023, 5, e316–e329. [CrossRef] [PubMed]
- Fejer, R.; Ruhe, A. What is the prevalence of musculoskeletal problems in the elderly population in developed countries? A systematic critical literature review. Chiropr. Man. Ther. 2012, 20, 31. [Google Scholar] [CrossRef] [PubMed]
- Blyth, F.M.; Briggs, A.M.; Schneider, C.H.; Hoy, D.G.; March, L.M. The global burden of musculoskeletal pain—Where to from here? Am. J. Public Health 2019, 109, 35–40. [Google Scholar] [CrossRef]
- Briggs, A.M.; Cross, M.J.; Hoy, D.G.; Sànchez-Riera, L.; Blyth, F.M.; Woolf, A.D.; March, L. Musculoskeletal health conditions represent a global threat to healthy aging: A report for the 2015 World Health Organization world report on ageing and health. Gerontologist 2016, 56 (Suppl. S2), S243–S255. [Google Scholar] [CrossRef]
- GBD 2019 Diseases and Injuries Collaborators. Global burden of 369 diseases and injuries in 204 countries and territories, 1990–2019: A systematic analysis for the Global Burden of Disease Study 2019. Lancet 2020, 396, 1204–1222. [Google Scholar] [CrossRef]
- Sokka, T.; Abelson, B.; Pincus, T. Mortality in rheumatoid arthritis: 2008 update. Clin. Exp. Rheumatol. 2008, 26 (Suppl. S51), S35–S61. [Google Scholar]
- Rasch, E.K.; Hirsch, R.; Paulose-Ram, R.; Hochberg, M.C. Prevalence of rheumatoid arthritis in persons 60 years of age and older in the United States: Effect of different methods of case classification. Arthritis Rheum. Off. J. Am. Coll. Rheumatol. 2003, 48, 917–926. [Google Scholar] [CrossRef]
- Leyland, K.M.; Gates, L.S.; Sanchez-Santos, M.T.; Nevitt, M.C.; Felson, D.; Jones, G.; Jordan, J.M.; Judge, A.; Prieto-Alhambra, D.; Yoshimura, N.; et al. Knee osteoarthritis and time-to all-cause mortality in six community-based cohorts: An international meta-analysis of individual participant-level data. Aging Clin. Exp. Res. 2021, 33, 529–545. [Google Scholar] [CrossRef]
- Roseen, E.J.; Rajendran, I.; Stein, P.; Fredman, L.; Fink, H.A.; LaValley, M.P.; Saper, R.B. Association of back pain with mortality: A systematic review and meta-analysis of cohort studies. J. Gen. Intern. Med. 2021, 36, 3148–3158. [Google Scholar] [CrossRef] [PubMed]
- Stubbs, B.; Binnekade, T.T.; Soundy, A.; Schofield, P.; Huijnen, I.P.; Eggermont, L.H. Are older adults with chronic musculoskeletal pain less active than older adults without pain? A systematic review and meta-analysis. Pain Med. 2013, 14, 1316–1331. [Google Scholar] [CrossRef] [PubMed]
- Baumgartner, R.N.; Waters, D.L.; Gallagher, D.; Morley, J.E.; Garry, P.J. Predictors of skeletal muscle mass in elderly men and women. Mech. Ageing Dev. 1999, 107, 123–136. [Google Scholar] [CrossRef] [PubMed]
- Chuang, S.-Y.; Chang, H.-Y.; Lee, M.-S.; Chen, R.C.-Y.; Pan, W.-H. Skeletal muscle mass and risk of death in an elderly population. Nutr. Metab. Cardiovasc. Dis. 2014, 24, 784–791. [Google Scholar] [CrossRef] [PubMed]
- Pan, F.; Tian, J.; Scott, D.; Cicuttini, F.; Jones, G. Muscle function, quality, and relative mass are associated with knee pain trajectory over 10.7 years. Pain 2022, 163, 518–525. [Google Scholar] [CrossRef] [PubMed]
- Law, L.F.; Sluka, K.A. How does physical activity modulate pain? Pain 2017, 158, 369. [Google Scholar] [CrossRef]
- Xue, Q.L. The frailty syndrome: Definition and natural history. Clin. Geriatr. Med. 2011, 27, 1–15. [Google Scholar] [CrossRef]
- Vermeiren, S.; Vella-Azzopardi, R.; Beckwée, D.; Habbig, A.K.; Scafoglieri, A.; Jansen, B.; Bautmans, I. Frailty and the Prediction of Negative Health Outcomes: A Meta-Analysis. J. Am. Med. Dir. Assoc. 2016, 17, 1163.e1–1163.e17. [Google Scholar] [CrossRef]
- Nessighaoui, H.; Lilamand, M.; Patel, K.V.; Vellas, B.; Laroche, M.L.; Dantoine, T.; Cesari, M. Frailty and Pain: Two Related Conditions. J. Frailty Aging 2015, 4, 144–148. [Google Scholar] [CrossRef]
- Huh, Y.; Yang, E.J.; Lee, S.A.; Lim, J.Y.; Kim, K.W.; Paik, N.J. Association between executive function and physical performance in older Korean adults: Findings from the Korean Longitudinal Study on Health and Aging (KLoSHA). Arch. Gerontol. Geriatr. 2011, 52, e156–e161. [Google Scholar] [CrossRef]
- Lohman, T.G.; Roche, A.F.; Martorell, R. Anthropometric Standardization Reference Manual; Human Kinetics Books: Champaign, IL, USA, 1988. [Google Scholar]
- Lukaski, H.C.; Johnson, P.E.; Bolonchuk, W.W.; Lykken, G.I. Assessment of fat-free mass using bioelectrical impedance measurements of the human body. Am. J. Clin. Nutr. 1985, 41, 810–817. [Google Scholar] [CrossRef]
- Mijnarends, D.M.; Meijers, J.M.; Halfens, R.J.; ter Borg, S.; Luiking, Y.C.; Verlaan, S.; Schoberer, D.; Cruz Jentoft, A.J.; van Loon, L.J.; Schols, J.M. Validity and reliability of tools to measure muscle mass, strength, and physical performance in community-dwelling older people: A systematic review. J. Am. Med. Dir. Assoc. 2013, 14, 170–178. [Google Scholar] [CrossRef] [PubMed]
- Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults. Executive Summary of The Third Report of The National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). JAMA 2001, 285, 2486–2497. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.Y.; Park, H.S.; Kim, D.J.; Han, J.H.; Kim, S.M.; Cho, G.J.; Kim, D.Y.; Kwon, H.S.; Kim, S.R.; Lee, C.B.; et al. Appropriate waist circumference cutoff points for central obesity in Korean adults. Diabetes Res. Clin. Pract. 2007, 75, 72–80. [Google Scholar] [CrossRef] [PubMed]
- Bellamy, N.; Buchanan, W.W.; Goldsmith, C.H.; Campbell, J.; Stitt, L.W. Validation study of WOMAC: A health status instrument for measuring clinically important patient relevant outcomes to antirheumatic drug therapy in patients with osteoarthritis of the hip or knee. J. Rheumatol. 1988, 15, 1833–1840. [Google Scholar] [PubMed]
- Fairbank, J.C.; Couper, J.; Davies, J.B.; O’Brien, J.P. The Oswestry low back pain disability questionnaire. Physiotherapy 1980, 66, 271–273. [Google Scholar]
- Craig, C.L.; Marshall, A.L.; Sjöström, M.; Bauman, A.E.; Booth, M.L.; Ainsworth, B.E.; Pratt, M.; Ekelund, U.; Yngve, A.; Sallis, J.F.; et al. International physical activity questionnaire: 12-country reliability and validity. Med. Sci. Sports Exerc. 2003, 35, 1381–1395. [Google Scholar] [CrossRef] [PubMed]
- Dahlhamer, J.; Lucas, J.; Zelaya, C.; Nahin, R.; Mackey, S.; DeBar, L.; Kerns, R.; Von Korff, M.; Porter, L.; Helmick, C. Prevalence of Chronic Pain and High-Impact Chronic Pain Among Adults—United States, 2016. MMWR Morb. Mortal. Wkly. Rep. 2018, 67, 1001–1006. [Google Scholar] [CrossRef]
- Blyth, F.M.; Noguchi, N. Chronic musculoskeletal pain and its impact on older people. Best Pract. Res. Clin. Rheumatol. 2017, 31, 160–168. [Google Scholar] [CrossRef] [PubMed]
- Eggermont, L.H.; Bean, J.F.; Guralnik, J.M.; Leveille, S.G. Comparing pain severity versus pain location in the MOBILIZE Boston study: Chronic pain and lower extremity function. J. Gerontol. A Biol. Sci. Med. Sci. 2009, 64, 763–770. [Google Scholar] [CrossRef] [PubMed]
- Croft, P.; Jordan, K.; Jinks, C. “Pain elsewhere” and the impact of knee pain in older people. Arthritis Rheum. 2005, 52, 2350–2354. [Google Scholar] [CrossRef] [PubMed]
- Rundell, S.D.; Patel, K.V.; Krook, M.A.; Heagerty, P.J.; Suri, P.; Friedly, J.L.; Turner, J.A.; Deyo, R.A.; Bauer, Z.; Nerenz, D.R.; et al. Multi-site Pain Is Associated with Long-term Patient-Reported Outcomes in Older Adults with Persistent Back Pain. Pain Med. 2019, 20, 1898–1906. [Google Scholar] [CrossRef] [PubMed]
- de Vitta, A.; Machado Maciel, N.; Bento, T.P.F.; Genebra, C.; Simeão, S. Multisite musculoskeletal pain in the general population: A cross-sectional survey. Sao Paulo Med. J. 2022, 140, 24–32. [Google Scholar] [CrossRef]
- Chen, L.; Ferreira, M.L.; Nassar, N.; Preen, D.B.; Hopper, J.L.; Li, S.; Bui, M.; Beckenkamp, P.R.; Shi, B.; Arden, N.K.; et al. Association of chronic musculoskeletal pain with mortality among UK adults: A population-based cohort study with mediation analysis. EClinicalMedicine 2021, 42, 101202. [Google Scholar] [CrossRef]
- Patterson, R.; McNamara, E.; Tainio, M.; de Sá, T.H.; Smith, A.D.; Sharp, S.J.; Edwards, P.; Woodcock, J.; Brage, S.; Wijndaele, K. Sedentary behaviour and risk of all-cause, cardiovascular and cancer mortality, and incident type 2 diabetes: A systematic review and dose response meta-analysis. Eur. J. Epidemiol. 2018, 33, 811–829. [Google Scholar] [CrossRef]
- Martin, R.R.; Hadjistavropoulos, T.; McCreary, D.R. Fear of pain and fear of falling among younger and older adults with musculoskeletal pain conditions. Pain Res. Manag. 2005, 10, 211–219. [Google Scholar] [CrossRef]
- Karttunen, N.; Lihavainen, K.; Sipilä, S.; Rantanen, T.; Sulkava, R.; Hartikainen, S. Musculoskeletal pain and use of analgesics in relation to mobility limitation among community-dwelling persons aged 75 years and older. Eur. J. Pain 2012, 16, 140–149. [Google Scholar] [CrossRef]
- Panhale, V.P.; Gurav, R.S.; Nahar, S.K. Association of Physical Performance and Fear-Avoidance Beliefs in Adults with Chronic Low Back Pain. Ann. Med. Health Sci. Res. 2016, 6, 375–379. [Google Scholar]
- Leveille, S.G.; Guralnik, J.M.; Hochberg, M.; Hirsch, R.; Ferrucci, L.; Langlois, J.; Rantanen, T.; Ling, S. Low back pain and disability in older women: Independent association with difficulty but not inability to perform daily activities. J. Gerontol. A Biol. Sci. Med. Sci. 1999, 54, M487–M493. [Google Scholar] [CrossRef]
- Kitayuguchi, J.; Kamada, M.; Hamano, T.; Nabika, T.; Shiwaku, K.; Kamioka, H.; Okada, S.; Mutoh, Y. Association between knee pain and gait speed decline in rural Japanese community-dwelling older adults: 1-year prospective cohort study. Geriatr. Gerontol. Int. 2016, 16, 55–64. [Google Scholar] [CrossRef] [PubMed]
- Yanardag, M.; Şimşek, T.T.; Yanardag, F. Exploring the Relationship of Pain, Balance, Gait Function, and Quality of Life in Older Adults with Hip and Knee Pain. Pain Manag. Nurs. 2021, 22, 503–508. [Google Scholar] [CrossRef] [PubMed]
- Biswas, A.; Oh, P.I.; Faulkner, G.E.; Bajaj, R.R.; Silver, M.A.; Mitchell, M.S.; Alter, D.A. Sedentary time and its association with risk for disease incidence, mortality, and hospitalization in adults: A systematic review and meta-analysis. Ann. Intern. Med. 2015, 162, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Piercy, K.L.; Troiano, R.P.; Ballard, R.M.; Carlson, S.A.; Fulton, J.E.; Galuska, D.A.; George, S.M.; Olson, R.D. The Physical Activity Guidelines for Americans. JAMA 2018, 320, 2020–2028. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Veeranki, S.P.; Magnussen, C.G.; Xi, B. Recommended physical activity and all cause and cause specific mortality in US adults: Prospective cohort study. BMJ 2020, 370, m2031. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.; Chen, M. Gender-specific impact of cognitive impairment on all-cause mortality in older persons: A meta-analysis. Exp. Gerontol. 2022, 165, 111860. [Google Scholar] [CrossRef] [PubMed]
- Walker, E.R.; McGee, R.E.; Druss, B.G. Mortality in mental disorders and global disease burden implications: A systematic review and meta-analysis. JAMA Psychiatry 2015, 72, 334–341. [Google Scholar] [CrossRef]
- Soriano, J.B.; Kendrick, P.J.; Paulson, K.R.; Gupta, V.; Abrams, E.M.; Adedoyin, R.A.; Adhikari, T.B.; Advani, S.M.; Agrawal, A.; Ahmadian, E.; et al. Prevalence and attributable health burden of chronic respiratory diseases, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet Respir. Med. 2020, 8, 585–596. [Google Scholar] [CrossRef]
- Jones, N.R.; Roalfe, A.K.; Adoki, I.; Hobbs, F.D.R.; Taylor, C.J. Survival of patients with chronic heart failure in the community: A systematic review and meta-analysis. Eur. J. Heart Fail. 2019, 21, 1306–1325. [Google Scholar] [CrossRef]
- Hijikata, Y.; Kamitani, T.; Sekiguchi, M.; Otani, K.; Konno, S.I.; Takegami, M.; Fukuhara, S.; Yamamoto, Y. Association of kyphotic posture with loss of independence and mortality in a community-based prospective cohort study: The Locomotive Syndrome and Health Outcomes in Aizu Cohort Study (LOHAS). BMJ Open 2022, 12, e052421. [Google Scholar] [CrossRef]
- Bhaskaran, K.; Dos-Santos-Silva, I.; Leon, D.A.; Douglas, I.J.; Smeeth, L. Association of BMI with overall and cause-specific mortality: A population-based cohort study of 3·6 million adults in the UK. Lancet Diabetes Endocrinol. 2018, 6, 944–953. [Google Scholar] [CrossRef]
- World Health Organization. The Asia-Pacific Perspective: Redefining Obesity and Its Treatment; World Health Organization: Geneva, Switzerland, 2000. [Google Scholar]
- Veronese, N.; Cereda, E.; Solmi, M.; Fowler, S.A.; Manzato, E.; Maggi, S.; Manu, P.; Abe, E.; Hayashi, K.; Allard, J.P.; et al. Inverse relationship between body mass index and mortality in older nursing home residents: A meta-analysis of 19,538 elderly subjects. Obes. Rev. 2015, 16, 1001–1015. [Google Scholar] [CrossRef] [PubMed]
- Ades, P.A.; Savage, P.D. The obesity paradox: Perception vs knowledge. Mayo Clin. Proc. 2010, 85, 112–114. [Google Scholar] [CrossRef] [PubMed]
- Tieland, M.; Trouwborst, I.; Clark, B.C. Skeletal muscle performance and ageing. J. Cachexia Sarcopenia Muscle 2018, 9, 3–19. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, C.P.; Hirsch, M.S.; Moeny, D.; Kaul, S.; Mohamoud, M.; Joffe, H.V. Testosterone and “Age-Related Hypogonadism”—FDA Concerns. N. Engl. J. Med. 2015, 373, 689–691. [Google Scholar] [CrossRef]
- Hermann, M.; Berger, P. Hormonal changes in aging men: A therapeutic indication? Exp. Gerontol. 2001, 36, 1075–1082. [Google Scholar] [CrossRef]
- Curcio, F.; Ferro, G.; Basile, C.; Liguori, I.; Parrella, P.; Pirozzi, F.; Della-Morte, D.; Gargiulo, G.; Testa, G.; Tocchetti, C.G.; et al. Biomarkers in sarcopenia: A multifactorial approach. Exp. Gerontol. 2016, 85, 1–8. [Google Scholar] [CrossRef]
- Mitchell, W.K.; Williams, J.; Atherton, P.; Larvin, M.; Lund, J.; Narici, M. Sarcopenia, dynapenia, and the impact of advancing age on human skeletal muscle size and strength; a quantitative review. Front. Physiol. 2012, 3, 260. [Google Scholar] [CrossRef]
- Wang, D.X.M.; Yao, J.; Zirek, Y.; Reijnierse, E.M.; Maier, A.B. Muscle mass, strength, and physical performance predicting activities of daily living: A meta-analysis. J. Cachexia Sarcopenia Muscle 2020, 11, 3–25. [Google Scholar] [CrossRef]
- Berger, M.J.; Doherty, T.J. Sarcopenia: Prevalence, mechanisms, and functional consequences. Interdiscip. Top. Gerontol. 2010, 37, 94–114. [Google Scholar]
- Zhou, H.H.; Liao, Y.; Peng, Z.; Liu, F.; Wang, Q.; Yang, W. Association of muscle wasting with mortality risk among adults: A systematic review and meta-analysis of prospective studies. J. Cachexia Sarcopenia Muscle 2023, 14, 1596–1612. [Google Scholar] [CrossRef] [PubMed]
- Stowe, R.P.; Peek, M.K.; Cutchin, M.P.; Goodwin, J.S. Plasma cytokine levels in a population-based study: Relation to age and ethnicity. J. Gerontol. A Biol. Sci. Med. Sci. 2010, 65, 429–433. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.M.; An, J. Cytokines, inflammation, and pain. Int. Anesthesiol. Clin. 2007, 45, 27–37. [Google Scholar] [CrossRef] [PubMed]
| Total (n = 1000) | Survivor (n = 829) | Non-Survivor (n = 171) | p-Value | |
|---|---|---|---|---|
| Age (years) (n = 1000) | 76.3 ± 8.7 | 74.8 ± 8.1 | 83.7 ± 7.8 | <0.001 * |
| Sex (n = 1000) | 0.061 | |||
| Male | 441 (44.1) | 354 (42.7) | 87 (50.9) | |
| Female | 559 (55.9) | 475 (57.3) | 84 (49.1) | |
| BMI (m2/kg) (n = 871) | 24.0 ± 3.3 | 24.1 ± 3.3 | 22.9 ± 3.3 | <0.001 * |
| ASM (kg) (n = 877) | 12.8 ± 3.3 | 12.9 ± 3.3 | 12.3 ± 3.5 | 0.098 |
| Metabolic syndrome (n = 996) | 0.753 | |||
| No | 631 (63.4) | 521 (63.1) | 110 (64.7) | |
| Yes | 365 (36.6) | 305 (36.9) | 60 (36.3) | |
| Low back pain (n = 950) | <0.001 * | |||
| No | 703 (74.0) | 612 (77.3) | 91 (57.6) | |
| Yes | 247 (26.0) | 180 (22.7) | 67 (42.4) | |
| Knee pain (n = 949) | 0.016 * | |||
| No | 760 (80.1) | 645 (81.5) | 115 (72.8) | |
| Yes | 189 (19.9) | 146 (18.5) | 43 (27.2) | |
| Alcohol (n = 992) | 0.185 | |||
| No | 761 (76.7) | 625 (75.8) | 136 (81.0) | |
| Yes | 231 (23.3) | 199 (24.2) | 32 (19.0) | |
| Smoking (n = 1000) | 1.000 | |||
| No | 711 (71.1) | 589 (71.0) | 122 (71.3) | |
| Yes | 289 (28.9) | 240 (29.0) | 49 (28.7) | |
| Regular exercise (n = 989) | <0.001 * | |||
| No | 492 (49.7) | 377 (45.9) | 115 (68.5) | |
| Yes | 497 (50.3) | 444 (54.1) | 53 (31.5) | |
| Variables | |||
|---|---|---|---|
| Variables | Low Back Pain | Knee Pain | Regular Exercise |
| Age | 0.220 ** | 0.160 ** | 0.152 ** |
| Sex | 0.255 ** | 0.279 ** | 0.291 ** |
| BMI | −0.008 | 0.086 * | 0.035 |
| ASM | −0.245 ** | −0.236 ** | 0.311 ** |
| Metabolic syndrome | 0.091 ** | 0.148 ** | −0.069 ** |
| Low back pain | - | 0.506 ** | −0.245 ** |
| Knee pain | 0.506 ** | - | −0.264 ** |
| Alcohol | −0.127 ** | −0.176 ** | 0.129 ** |
| Smoking | −0.122 ** | −0.163 ** | 0.119 ** |
| Regular exercise | −0.245 ** | −0.264 ** | - |
| Variables | |||
| Variables | Individual Pain † | MSK Pain ‡ | Regular Exercise |
| Mortality | 0.164 ** | 0.145 ** | −0.169 ** |
| Characteristics | Univariate Model | Multivariate Model 1 § | Multivariate Model 2 ⁋ | |||
|---|---|---|---|---|---|---|
| OR (95% CI) | p | OR (95% CI) | p | OR (95% CI) | p | |
| Age | 1.133 (1.108–1.159) | <0.001 | 1.095 (1.065–1.126) | <0.001 | 1.095 (1.065–1.126) | <0.001 |
| Sex (Female) * | 0.720 (0.517–1.001) | 0.051 | 0.138 (0.063–0.302) | <0.001 | 0.138 (0.063–0.302) | <0.001 |
| Metabolic syndrome | 0.932 (0.660–1.315) | 0.688 | ||||
| BMI | 0.897 (0.894–1.010) | 0.001 | 0.299 | 0.299 | ||
| ASM | 0.950 (0.383–0.751) | 0.098 | 0.822 (0.732–0.923) | 0.001 | 0.822 (0.732–0.923) | 0.001 |
| Low back pain | 2.503 (1.752–3.576) | <0.001 | 0.442 | |||
| Knee pain | 1.652 (1.114–2.499) | 0.012 | 0.956 | |||
| Alcohol | 0.739 (0.487–1.121) | 0.155 | 0.235 | 0.235 | ||
| Smoking | 0.986 (0.685–1.418) | 0.938 | ||||
| Regular exercise | 0.391 (0.275–0.557) | <0.001 | 0.465 (0.287–0.754) | 0.002 | 0.465 (0.287–0.754) | 0.002 |
| MSK pain (1) † | 1.875 (1.219–2.885) | 0.004 | 0.979 | |||
| MSK pain (2) ‡ | 2.483 (1.590–3.879) | <0.001 | 0.661 | |||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hwang, S.-W.; Kim, C.-W.; Jang, Y.-J.; Lee, C.-H.; Oh, M.-K.; Kim, K.-W.; Jang, H.-C.; Lim, J.-Y.; Chun, S.-W.; Lim, S.-K. Musculoskeletal Pain, Physical Activity, Muscle Mass, and Mortality in Older Adults: Results from the Korean Longitudinal Study on Health and Aging (KLoSHA). Medicina 2024, 60, 462. https://doi.org/10.3390/medicina60030462
Hwang S-W, Kim C-W, Jang Y-J, Lee C-H, Oh M-K, Kim K-W, Jang H-C, Lim J-Y, Chun S-W, Lim S-K. Musculoskeletal Pain, Physical Activity, Muscle Mass, and Mortality in Older Adults: Results from the Korean Longitudinal Study on Health and Aging (KLoSHA). Medicina. 2024; 60(3):462. https://doi.org/10.3390/medicina60030462
Chicago/Turabian StyleHwang, Sun-Woo, Chang-Woo Kim, Yun-Jeong Jang, Chang-Han Lee, Min-Kyun Oh, Ki-Woong Kim, Hak-Chul Jang, Jae-Young Lim, Se-Woong Chun, and Seung-Kyu Lim. 2024. "Musculoskeletal Pain, Physical Activity, Muscle Mass, and Mortality in Older Adults: Results from the Korean Longitudinal Study on Health and Aging (KLoSHA)" Medicina 60, no. 3: 462. https://doi.org/10.3390/medicina60030462
APA StyleHwang, S.-W., Kim, C.-W., Jang, Y.-J., Lee, C.-H., Oh, M.-K., Kim, K.-W., Jang, H.-C., Lim, J.-Y., Chun, S.-W., & Lim, S.-K. (2024). Musculoskeletal Pain, Physical Activity, Muscle Mass, and Mortality in Older Adults: Results from the Korean Longitudinal Study on Health and Aging (KLoSHA). Medicina, 60(3), 462. https://doi.org/10.3390/medicina60030462








